Abstract
Background
Outcomes in chronic myeloid leukemia have improved with tyrosine kinase inhibitor treatment. However, little is known about outcomes of chronic myeloid leukemia in adolescent and young adult patients.
Design and Methods
We reviewed all 468 chronic myeloid leukemia patients treated at our institution with tyrosine kinase inhibitors as initial therapy: imatinib (n=281), nilotinib (n=98) or dasatinib (n=89).
Results
Median age was 47 years, median follow up 71 months and median treatment time with initial tyrosine kinase inhibitors 48 months. Adolescent and young adult was defined as aged 15–29 years. Sixty-one adolescent and young adult patients were identified. The only significant differences between adolescent and young adult and older patients were incidence of splenomegaly and distribution in Sokal risk groups. Only 3 adolescent and young adult patients have died. Rates of complete cytogenetic, major molecular and complete molecular response were significantly higher in older patients compared to adolescent and young adult patients, with a favorable trend in event-free survival for older patients. Transformation-free and overall survival were similar for the two groups.
Conclusions
The unfavorable trend in outcome for adolescent and young adult patients with chronic myeloid leukemia is unexpected. Additional research in this population is required to better define outcomes, understand the cause of this difference, and to help make better treatment recommendations.
Keywords: chronic myelogenous leukemia, tyrosine kinase inhibitor, adolescent, young adult, outcome
Introduction
Outcomes in chronic myeloid leukemia (CML) have dramatically improved since the introduction of imatinib as initial therapy in chronic phase.1 A complete cytogenetic response (CCyR) is achieved in over 80% of patients with a reported event-free survival (EFS) at eight years of 81%.2 More recently, two newer, second generation tyrosine kinase inhibitors (TKIs), dasatinib and nilotinib, have also been proven to be highly effective, not only for patients with resistance or intolerance to imatinib,3,4 but also as initial therapy for chronic phase CML.5–9
The median age of diagnosis of CML is reported to be approximately 67 years.10 However, all age groups can be affected by CML, and there is a small but significant group of patients diagnosed with CML at a much younger age, including some diagnosed at 15–29 years, a group frequently referred to as adolescents and young adults (AYA).11–13 AYA patients with malignancy are considered a group with unique features with different prognosis, outcomes, treatment strategies, and possibly different biology, to their younger or older counterparts. This has resulted in a growing awareness of this group as a unique patient population.14,15 Prior to the TKI era, stem cell transplantation (SCT) was the preferred option as initial treatment for many AYA patients with CML, with interferon alpha reserved for those with no available transplant option. This recommendation was based on the possibility of curing the disease with transplant and the relatively low response rate and high toxicity with interferon alpha. With the high efficacy and improved tolerability of TKIs, stem cell transplant has been mostly relegated to second-line or beyond treatment options in the general CML population.16
Among leukemia patients, because acute lymphoblastic leukemia (ALL) occurs more commonly in younger patients, much more attention has been devoted to the AYA patient population afflicted by this disease.17 Since most of the other leukemia subtypes are generally diseases involving older adults, little is known about the outcomes of AYA patients with other leukemia. There is currently very limited information on the outcome of AYA patients with CML, particularly those receiving initial therapy with TKI. We conducted this analysis in order to understand the characteristics and outcome of AYA patients with CML treated with TKI.
Design and Methods
From July 2000 to December 2010, 468 consecutive patients with CML in chronic phase (CP) were treated at our institution on a clinical trial of front-line therapy with TKI in sequential or parallel IRB-approved protocols with identical inclusion criteria. TKIs used included imatinib (n=281) either at standard (n=73) or high-dose (n=208), nilotinib (n=98), or dasatinib (n=89). Informed consent was obtained in accordance with the Declaration of Helsinki. The medical records of all patients were reviewed and AYA patients were identified. For this analysis, AYA was defined as those aged 15–29 years at the time treatment was initiated.
CP CML was defined as the presence in peripheral blood of all of the following: blasts less than 15%, basophils less than 20%, blasts plus promyelocytes less than 30%, and platelet count over 100×109/L.15 Patients were followed with weekly complete blood counts for the first 2–3 months, then every 4–6 weeks. A cytogenetic analysis was performed every three months for the first year, then every six months for 2–3 years, then every 2–3 years. Peripheral blood FISH was performed every three months for the first year, then every six months. Response criteria were as previously described.16 Briefly, a complete hematologic response (CHR) was defined as a white blood cell (WBC) count of below 10×109/L, a platelet count of below 450×109/L, absence of immature cells (blasts, promyelocytes, myelocytes) with less than 2% basophils in the peripheral blood, and disappearance of all signs and symptoms related to leukemia including palpable splenomegaly. Responses were further categorized by the best cytogenetic response as complete (0% Philadelphia-chromosome positive (Ph+) metaphases), partial (PCyR 1%–35% Ph+) or minor (36%–95% Ph+). A major cytogenetic response (MCyR) included complete plus partial cytogenetic responses (i.e. <35% Ph+). Major molecular response (MMR) was defined as a BCR-ABL/ABL ratio of 0.1% or under (international scale), and a complete molecular response (CMR) as undetectable transcripts with an assay with sensitivity of at least 4.5-log.16
Differences among variables were evaluated by the χ2 test and Mann-Whitney U test for categorical and continuous variables, respectively. Event-free survival (EFS) was measured from the start of treatment to the date of any of the following events while on therapy: loss of complete hematologic response, loss of complete or major cytogenetic response, discontinuation of therapy for toxicity or lack of efficacy, or progression to accelerated or blast phases, and death from any cause at any time. Survival was measured from the time treatment was started to the date of death from any cause at any time or date of last follow up. Transformation-free survival (TFS) was measured from the start of therapy to the date of transformation to accelerated or blast phases while on therapy, or death from any cause at any time, or to the date of last follow up. Survival probabilities were estimated by the Kaplan-Meier method, and compared by the log rank test. Multivariate analyses were conducted using the logistical regression model for CCyR, MMR, and CMR rates, and Cox’s proportional hazard model for survival. Proportional hazard assumption was verified by the Grambsch-Therneau test.18 Terms characterizing interactions between covariates were then added to the model and retained only with P<0.05.
Results
A total of 468 CML patients were included in one of the trials with TKI as initial therapy for CP CML. The median age of the entire cohort was 47 years (range 15–85 years). The majority of patients (322, 69%) had a low Sokal risk score, and 20 (4%) patients had additional chromosomal abnormalities besides the Philadelphia chromosome (i.e. clonal evolution) at the time of diagnosis. The median follow-up time for the entire cohort was 71 months (range 3–130 months), and the median treatment time with the first TKI was 48 months (range 0.1–130 months).
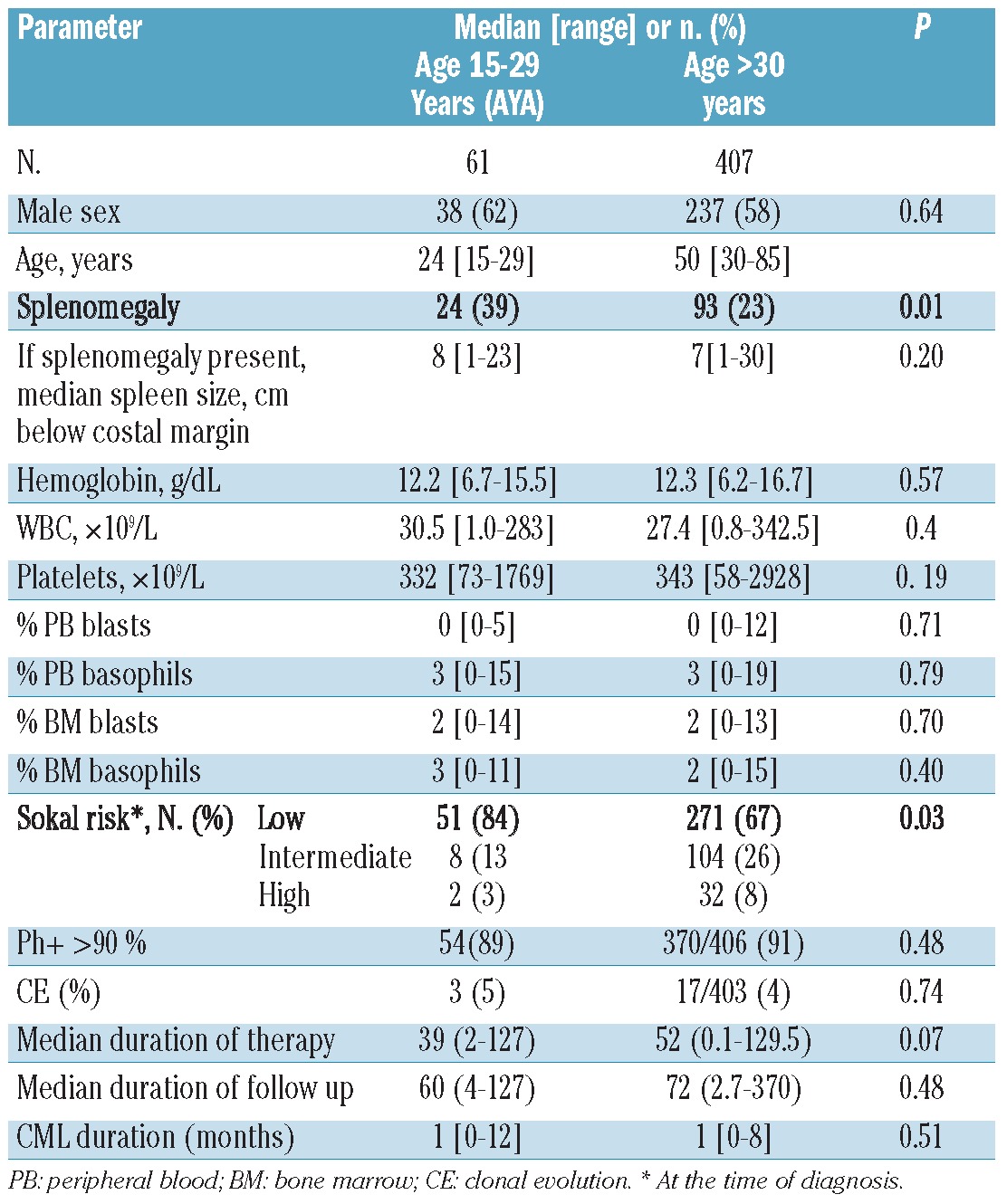
We identified 61 AYA patients who were included in these studies, representing 13% of the study population. Thirty-five AYA patients were treated with imatinib (11 with a starting dose of 400 mg daily and 24 with 800 mg daily), 13 with nilotinib and 13 with dasatinib. The median duration of therapy for the AYA group was 39 months (range 2–127 months). Compared to the older patient population, although AYA more frequently presented with splenomegaly (39% vs. 23%, P=0.01), significantly more AYA were in the low Sokal risk group (84 vs. 67, P=0.03). No other significant differences were identified between AYA and older patients (Table 1).
Table 1.
Patients’ characteristics according to age at the time of treatment initiation.
Response to therapy
Among the 468 patients treated, 11 (including 4 AYA) were not evaluable for response because of treatment discontinuation before reaching initial cytogenetic response evaluation at three months (2 for toxicity, 2 insurance issues, 2 non-adherence, 5 refused). Two of the patients who achieved CCyR did not have molecular tests available. Thus, overall response is based on 457 patients and molecular response is based on 455. The best cytogenetic response on therapy was CCyR for 420 patients (92%). The overall rate of CCyR was 87% for patients treated with imatinib 400 mg daily, 91% for those treated with 800 mg daily, 94% with nilotinib and 97% with dasatinib. MMR was achieved by 85%, including 39% who achieved a CMR. Rates of MMR were 77% for imatinib 400 mg, 86% for imatinib 800 mg, 81% for nilotinib and 91% for dasatinib.
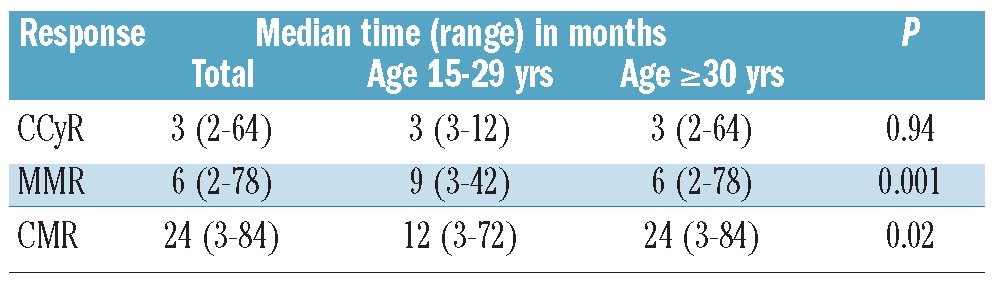
Response rates were generally lower for AYA patients (Table 2). The rate of CCyR was 84% for AYA compared to 93% for older patients (P=0.03). Corresponding rates for MMR were 75% and 86% (P=0.049), and for CMR 23% and 41% (P=0.01). The median time to CCyR was identical for the two age groups (3 months, range 2–64 months) but the median time to MMR was longer for AYA (9 months, range 3–42 months) than for older patients (6 months, range 2–78 months) (Table 4). CCyR rates among patients treated with second generation TKI were similar between the two groups (96% and 95%, respectively, P=0.85), albeit with a trend for lower rates of MMR in AYA (79% vs. 87%, P=0.34). We then compared the rate of response at times that have been identified as predictors of long-term outcome. The rate of CCyR at 12 months was 83% for AYA and 88% for older patients (P=0.36). The rates of MMR at 18 months were 65% and 78%, respectively (P=0.09). The rate of CCyR at 12 months for AYA was significantly higher for patients treated with dasatinib or nilotinib compared to imatinib, although no difference was seen in the rate of MMR at 12 or 18 months (Table 2).
Table 2.
Response to therapy according to age group.
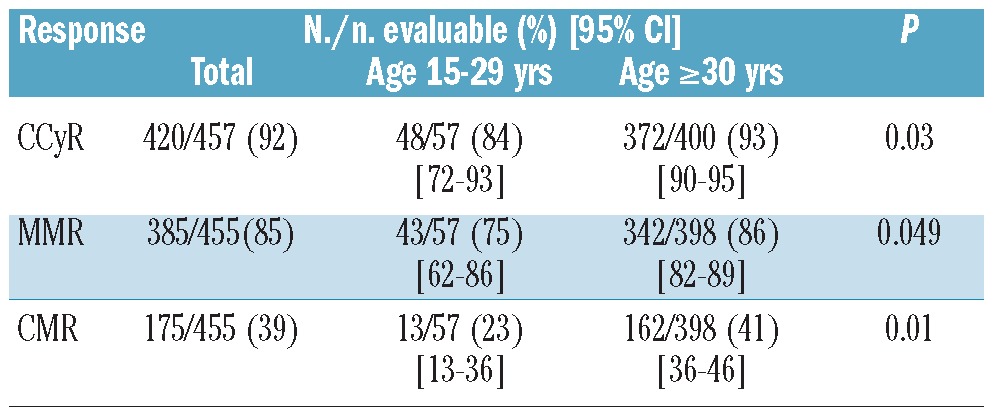
Table 4.
Median time to response according to age group.
After a median follow up of 48 months from the start of the first TKI, 333 (71%) patients continue receiving therapy with their initial TKI. Twenty-four of the 61 (39%) AYA patients discontinued their initial TKI: 11 due to resistance (4 transformed, 2 each to accelerated (AP) and blast phase (BP), and 7 still in CP), one due to insurance problems, 3 because of toxicity, 3 for non-adherence, 2 lost to follow up, 2 received an allogeneic SCT (one in CCyR after 9 months of therapy, one in PCyR after 6 months of therapy), one for other disease, and one died in a car accident.
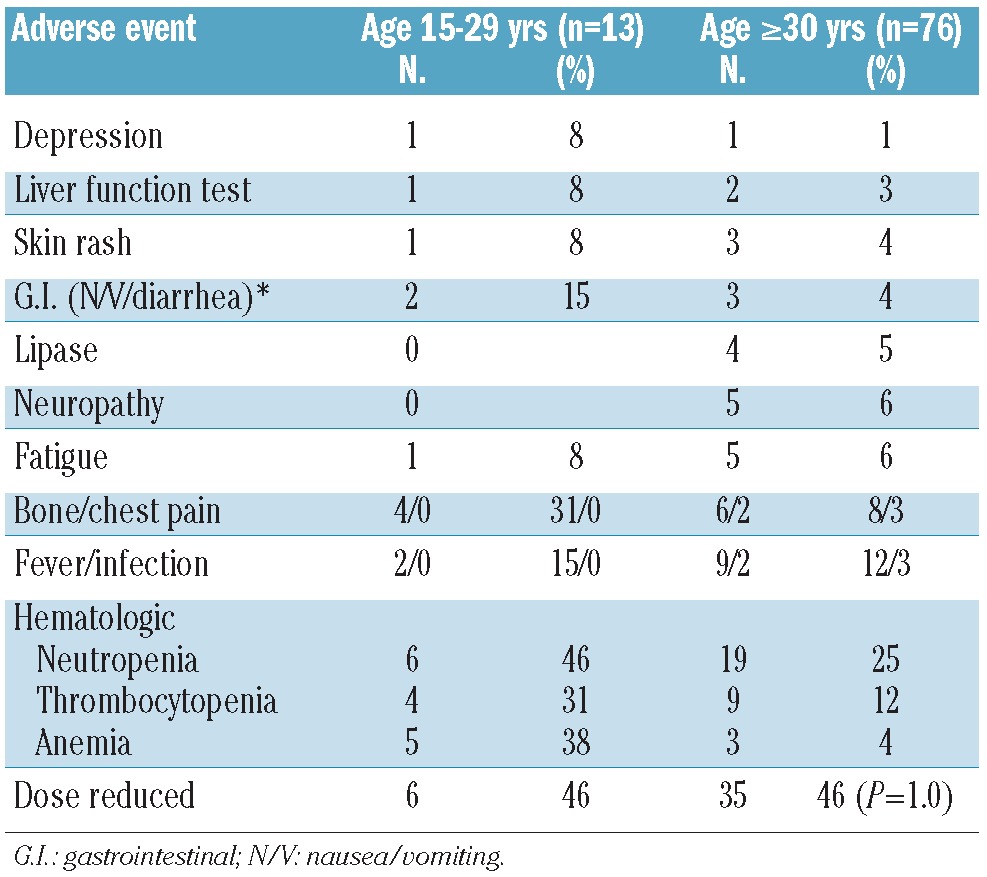
The most common grades 3 and 4 adverse events according to age group and to specific TKI are presented in Tables 5–7. Acknowledging the small numbers for some subsets, there was no evidence of a significant difference in frequency of these adverse events between AYA and their older counterparts. Interestingly, dose reductions were more frequently required for imatinib in the older patient population, particularly those treated with a higher starting dose. In contrast, there was no significant difference in dose reductions of second generation TKI between AYA (42%) and in the older (36%) patients (P=0.57).
Table 5.
Grade 3–4 toxicity and dose reductions according to age group. Nilotinib (n=98).
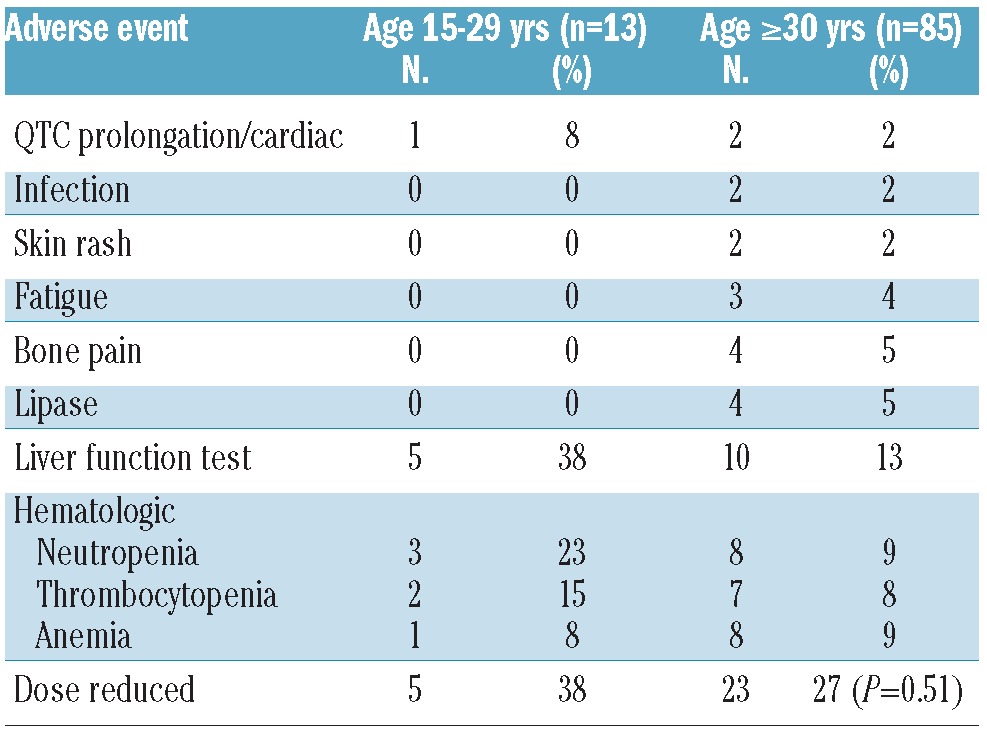
Table 7.
Grade 3–4 toxicity and dose reductions according to age group. Imatinib (n=281).
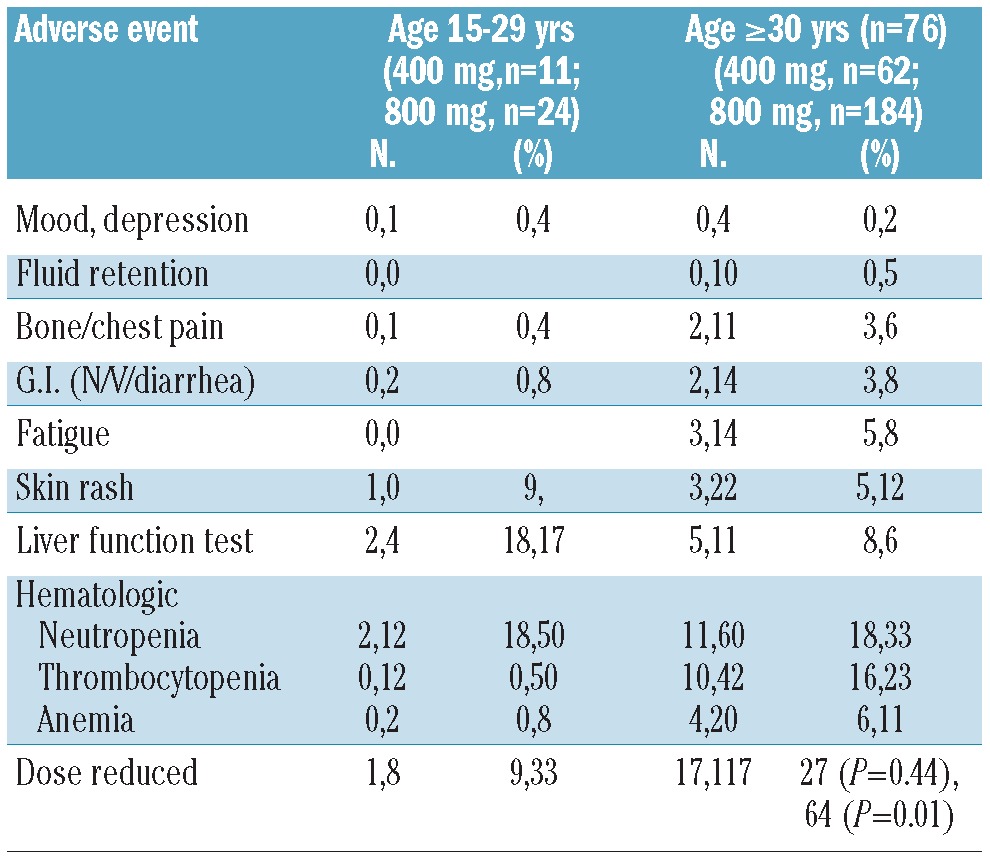
Prognostic significance of younger age
We then performed a multivariate analysis to investigate whether AYA had an independent prognostic value. Factors studied for the multivariate analysis included: hemoglobin, white blood cell count, platelet count, percentage of bone marrow blasts, percent age of bone marrow basophils, duration of CML diseases, percentage of peripheral blood blasts, percentage of peripheral blood basophils, percentage of Philadelphia chromosome metaphases, age group, treatment arm (imatinib standard or imatinib higher dose or 2nd generation TKI), splenomegaly, and Sokal risk score. AYA age group was significantly associated with lower probability of achieving MMR and CMR, with trends towards significance (P=0.06) for EFS and CCyR, after adjustment for covariates.
Long-term outcomes
The 5-year probability of EFS was 81% for the total patient population, with a rate of 93% alive and free from transformation at five years, and an overall survival of 93%. There was a trend for an inferior EFS for AYA compared to older adults, with 5-year rates of 71% and 82%, respectively (P=0.07), with no significant difference in survival free from transformation (87% for AYA vs. 94% for older adults, P=0.44) or overall survival (95 % vs. 93%, respectively, P=0.35) (Figure 1). The cause of death for the 3 AYA patients was one after transformation to BP, one from complications of allogeneic SCT, and one a car accident. Among the AYA group, a total of 7 patients have received allogeneic SCT after failing initial TKI therapy.
Figure 1.
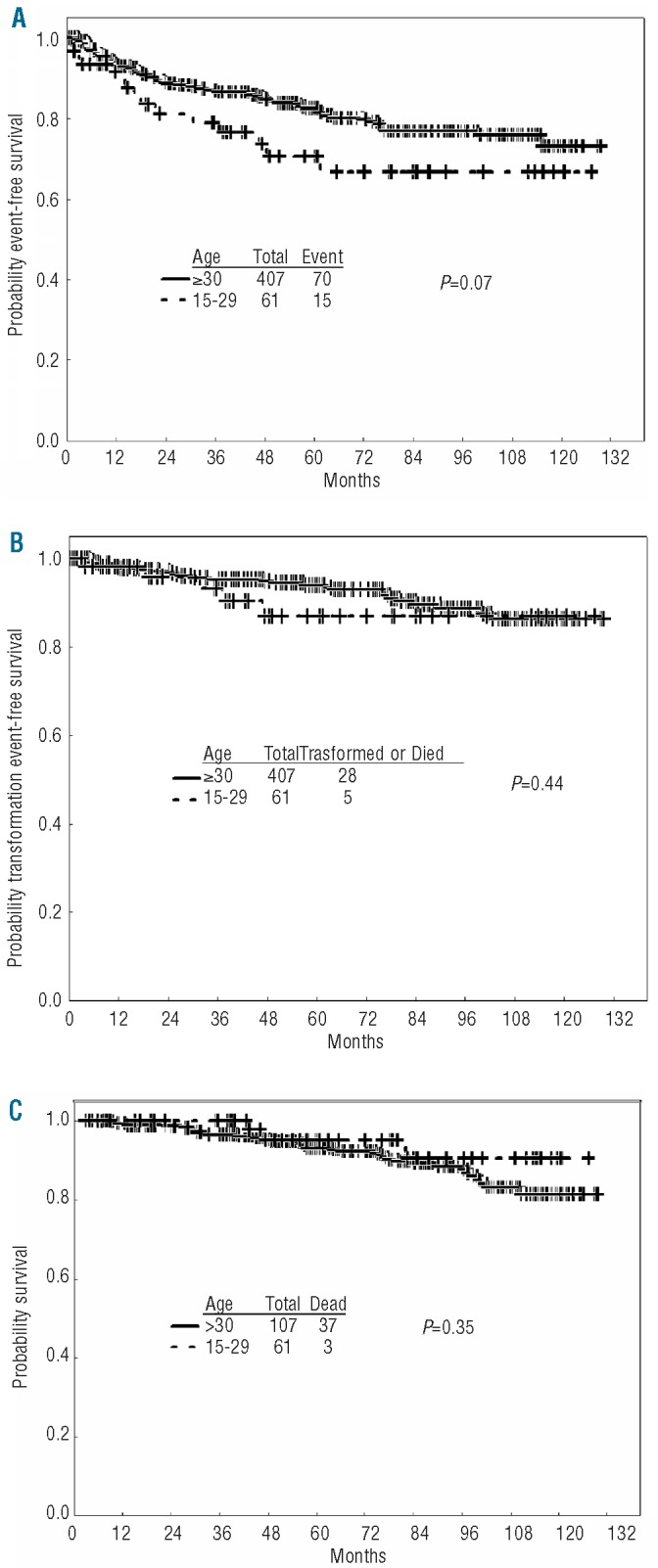
Long-term outcome of CML patients treated with TKI as initial therapy according to age group. (A) Event-free survival. (B) Transformation-free survival. (C) Overall survival.
Discussion
The outcome of patients with CML has improved significantly since the introduction of TKI as initial therapy for this disease. Front-line therapy with imatinib,2 nilotinib5,7,19 and dasatinib6,20 have yielded excellent results, with rates of CCyR of 80–90% and EFS of over 80% at five years. There has been considerable attention given to the prognostic significance of age in the outcome of patients treated with TKI. However, most of the attention has been devoted to the older patient population. This is probably because the median age of patients with CML is reported to be approximately 67 years,10,21 although the most recent studies report a median age of patients enrolled of between 45 to 55 years. Before the era of TKI, older patients were reported to have a poorer prognosis. This was due to the poor tolerance to interferon therapy in this age group22 and the limited availability of stem cell transplant options for older patients. With TKI, the impact of older age has been greatly minimized and nearly eliminated, with excellent results reported for older patients treated with imatinib.23,24 Still, it has been suggested that these patients may not tolerate TKI as well as younger patients,25 perhaps explaining why the older population is more frequently not offered therapy with TKI.26,27
Table 3.
Responses by TKI among AYA patients.
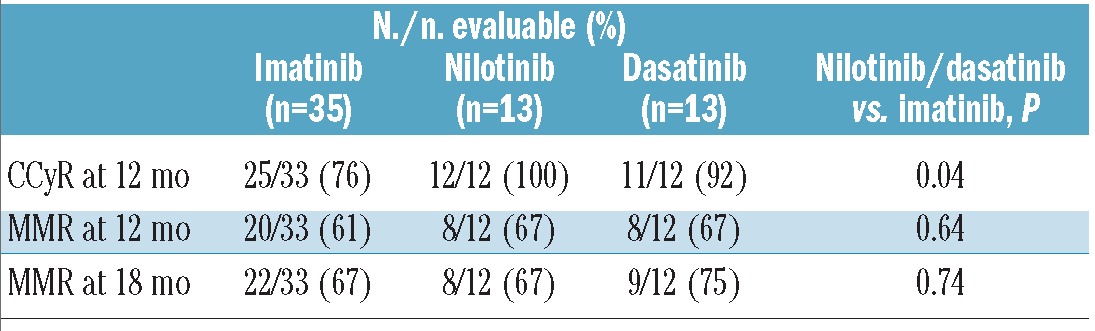
Table 6.
Grade 3–4 toxicity and dose reductions according to age group. Dasatinib (n=89).
The impact of age has been explored much less frequently in the younger population, and more specifically, in AYA (i.e. those in an age group variably defined but usually ranging from around 15 to around 29 years). An important reason for the paucity of reports in this patient population is the low frequency with which CML is found in this patient population. Still, this age group has been attracting growing interest in oncology as a distinct patient population with unique clinical characteristics, needs and outcome. The field of adolescent and young adult oncology has been emerging over the past decade in large part due to the fact that malignancy is recognized as the leading cause of disease-related death among young adults in the United States.11 Approximately 260,000 AYA are diagnosed with cancer every year.28–30 While it is encouraging that the overall survival for childhood cancer has greatly improved over the past four decades with 5-year survival rates now approaching 80%,31 the outcomes for young adults with many malignancies have not yet reflected this improvement in tumors as diverse as brain tumors, leukemias, some epithelial cancers, and sarcomas.11,29
There is a plethora of special issues significant to the AYA cancer population that has a profound impact on access, delivery and quality of care. One of these concerns the unique pharmacology, body and drug metabolism of the AYA population.32 One study in particular examined age and imatinib pharmacokinetics in children and young adults. In 41 patients taking imatinib, it was found that body weight was the only variable that was significantly related to imatinib clearance.33 While there was no statistically significant association with age, it is noteworthy that body weight can frequently be a rapidly changing parameter in many AYA patients.33
Adherence to therapy is a particularly important facet of care. Several studies have demonstrated that poor adherence and inadequate drug dosing is associated with increased relapse and decreased survival in younger age leukemia patients.24–26
In CML, recent studies have demonstrated that adherence to therapy might be the most important factor associated with outcome.37 Non-adherence has been reported in 27% to 60% of AYA cancer patients.36,38–41 Most studies have demonstrated that adherence rates are lower in adolescent cancer patients than with either younger or older cancer patients, even when taking into account treatment on similar protocols with the same disease entities.36,42–45 With an oral, self-administered therapy that is given for long (indefinite) periods of time, such as TKIs in CML, non-adherence becomes an even bigger issue. A multitude of other concerns should be considered for the AYA population including fertility,46,47 pharmacology and metabolism, development of late effects of treatment including secondary neoplasms, and access to health insurance and health care. These are all, indeed, considerations that have a huge impact on outcomes in this unique population. However, little information is available on these issues for the CML patients in general and AYA patients in particular.
With regards to hematologic malignancy, and leukemia patients in particular, very few studies have specifically examined the AYA population.48 Several studies have highlighted the outcomes in ALL and, more recently, AML.49 However, to our knowledge, this is the first report to address CML in AYA. This group represents only 13% of all CML patients treated with TKI at our institution during the study period. While the median age of CML is reported to be approximately 66 years in the US, CML also occurs, albeit rarely, in childhood, making up less than 10% of all CML cases.50 Notable differences in presentation of the disease include higher median WBC in pediatric than adult populations, and the fact that splenomegaly is more commonly found in those patients with a higher WBC count.51,52 While there are now several scoring systems for the general/adult CML population, these have been developed in the more typical, older populations. None of them has been specifically developed or validated in the pediatric or AYA populations.50 In the past, stem cell transplant was thought to be the best upfront treatment for younger populations. But with the success of TKI in older patients, TKI therapy has now become standard front-line treatment for pediatric and AYA populations.50,53,54
Unexpectedly, we observed that the response rate for AYA patients is lower than that seen in older patients. In our analysis, younger patients tend to have a lower Sokal risk score, with 51 of 61 (84%) patients having low Sokal score. The EUTOS score55 was also calculated; this showed 92% of the AYA patients to have a low-risk EUTOS score. Notably, both scoring systems include spleen size, but only the Sokal score includes age as one of its parameters. Also, the median age in the cohort used to determine both scoring systems was older than the AYA group: median age EUTOS group 52 years, mean age range for original Sokal group 38–49 years.56 It would be of interest to investigate whether other biological or molecular determinants of the disease, such as the expression or activity of hOCT1 or the MDR phenotype, may vary in this patient population compared to older patients. Second generation TKIs appear to confer a better outcome compared to imatinib with a higher rate of CCyR. It is possible that other factors, such as adherence to therapy and availability of insurance, may play a role in the lower response rate. It is noteworthy that in all of the studies included in our analysis, TKI medication was provided as part of the study protocols in which these patients were included, at no cost to the participants. However, even routine clinic visits may represent a considerable financial and/or logistical burden to AYA patients with poor or no medical insurance coverage. Still, there was no significant difference in long-term outcome to that of the older patients. There was a trend for a shorter event-free survival that does not, however, reach statistical significance probably because of the small sample size. However, none of the patients transformed on therapy and therefore most patients could be salvaged with subsequent therapy, including SCT in 7 AYA patients. Outcome with SCT in younger patients with CML after imatinib failure has been reported to be excellent.57 Those not eligible for SCT may also have a favorable outcome with 2nd TKI such as dasatinib or nilotinib.
In conclusion, AYA patients with CML tend to have a lower response rate to initial TKI therapy. The reasons for this need to be further defined but may have both biological as well as non-biological features, for example, psychosocial elements including adherence to therapy and access to healthcare and medical insurance. AYA patients need to be followed closely to ensure adequate response to therapy and those who do not achieve optimal response should have the opportunity of being offered timely and appropriate alternative therapies. Future studies aimed at this unique patient population should address any differences in disease biology, as well as more specifically adherence and other factors that may affect outcome.
Acknowledgments
The authors would like to thank Sherry Pierce for her critical review of the manuscript.
Footnotes
Funding: this work was supported by grant funding provided through NIH grant P-01 CA049639.
Authorship and Disclosures
The information provided by the authors about contributions from persons listed as authors and in acknowledgments is available with the full text of this paper at www.haematologica.org.
Financial and other disclosures provided by the authors using the ICMJE (www.icmje.org) Uniform Format for Disclosure of Competing Interests are also available at www.haematologica.org.
References
- 1.O’Brien SG, Guilhot F, Larson RA, Gathmann I, Baccarani M, Cervantes F, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348(11):994–1004. doi: 10.1056/NEJMoa022457. [DOI] [PubMed] [Google Scholar]
- 2.Deininger M, O’Brien SG, Guilhot F, Goldman J, Hochhaus A, Hughes T, et al. International randomized study of interferon vs STI571 (IRIS) 8-Year follow up: sustained survival and low risk for progression or events in patients with newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP) treated with imatinib. Poster session presented at: Proceedings of the 51st American Society of Hematology; 2009 Dec 5–8; New Orleans, LA. [Google Scholar]
- 3.Shah NP, Kim DW, Kantarjian H, Rousselot P, Llacer PE, Enrico A, et al. Potent, transient inhibition of BCR-ABL with dasatinib 100 mg daily achieves rapid and durable cytogenetic responses and high transformation-free survival rates in chronic phase chronic myeloid leukemia patients with resistance, suboptimal response or intolerance to imatinib. Haematologica. 2010;95(2):232–40. doi: 10.3324/haematol.2009.011452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kantarjian HM, Giles FJ, Bhalla KN, Pinilla-Ibarz J, Larson RA, Gattermann N, et al. Nilotinib is effective in patients with chronic myeloid leukemia in chronic phase after imatinib resistance or intolerance: 24-month follow-up results. Blood. 2011;117(4):1141–5. doi: 10.1182/blood-2010-03-277152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rosti G, Palandri F, Castagnetti F, Breccia M, Levato L, Gugliotta G, et al. Nilotinib for the frontline treatment of Ph(+) chronic myeloid leukemia. Blood. 2009;114(24):4933–8. doi: 10.1182/blood-2009-07-232595. [DOI] [PubMed] [Google Scholar]
- 6.Cortes JE, Jones D, O’Brien S, Jabbour E, Ravandi F, Koller C, et al. Results of dasatinib therapy in patients with early chronic-phase chronic myeloid leukemia. J Clin Oncol. 2010;28(3):398–404. doi: 10.1200/JCO.2009.25.4920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cortes JE, Jones D, O’Brien S, Jabbour E, Konopleva M, Ferrajoli A, et al. Nilotinib as front-line treatment for patients with chronic myeloid leukemia in early chronic phase. J Clin Oncol. 2010;28(3):392–7. doi: 10.1200/JCO.2009.25.4896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Anderlini P, Luna M, Kantarjian HM, O’Brien S, Pierce S, Keating MJ, et al. Causes of initial remission induction failure in patients with acute myeloid leukemia and myelodysplastic syndromes. Leukemia. 1996;10(4):600–8. [PubMed] [Google Scholar]
- 9.Saglio G, Kim DW, Issaragrisil S, le Coutre P, Etienne G, Lobo C, et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med. 2010;362(24):2251–9. doi: 10.1056/NEJMoa0912614. [DOI] [PubMed] [Google Scholar]
- 10.Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60(5):277–300. doi: 10.3322/caac.20073. [DOI] [PubMed] [Google Scholar]
- 11.Thomas DM, Albritton KH, Ferrari A. Adolescent and young adult oncology: an emerging field. J Clin Oncol. 2010;28(32):4781–2. doi: 10.1200/JCO.2010.30.5128. [DOI] [PubMed] [Google Scholar]
- 12.Wein S, Pery S, Zer A. Role of palliative care in adolescent and young adult oncology. J Clin Oncol. 2010;28(32):4819–24. doi: 10.1200/JCO.2009.22.4543. [DOI] [PubMed] [Google Scholar]
- 13.Morgan S, Davies S, Palmer S, Plaster M. Sex, drugs, and rock ‘n’ roll: caring for adolescents and young adults with cancer. J Clin Oncol. 2010;28(32):4825–30. doi: 10.1200/JCO.2009.22.5474. [DOI] [PubMed] [Google Scholar]
- 14.Bleyer A, Barr R, Hayes-Lattin B, Thomas D, Ellis C, Anderson B. The distinctive biology of cancer in adolescents and young adults. Nat Rev Cancer. 2008;8(4):288–98. doi: 10.1038/nrc2349. [DOI] [PubMed] [Google Scholar]
- 15.Schmidt C. Lack of progress in teen and young adult cancers concerns researchers, prompts study. J Natl Cancer Inst. 2006;98(24):1760–3. doi: 10.1093/jnci/djj517. [DOI] [PubMed] [Google Scholar]
- 16.Baccarani M, Cortes J, Pane F, Niederwieser D, Saglio G, Apperley J, et al. Chronic myeloid leukemia: an update of concepts and management recommendations of European LeukemiaNet. J Clin Oncol. 2009;27(35):6041–51. doi: 10.1200/JCO.2009.25.0779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stock W. Adolescents and young adults with acute lymphoblastic leukemia. Hematology Am Soc Hematol Educ Program. 2010;2010:21–9. doi: 10.1182/asheducation-2010.1.21. [DOI] [PubMed] [Google Scholar]
- 18.Grambsch PM, Therneau TM. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika. 1994;81(3):515–26. [Google Scholar]
- 19.Kantarjian H, Shah NP, Hochhaus A, Cortes J, Shah S, Ayala M, et al. Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2010;362(24):2260–70. doi: 10.1056/NEJMoa1002315. [DOI] [PubMed] [Google Scholar]
- 20.SEER [Internet] 1975–2002. 2004. [updated 2011 Nov 10; cited 2012 Jan 3]. Available from: http://seer.cancer.gov/statfacts/html/cmyl.html.
- 21.Cortes J, Kantarjian H, O’Brien S, Robertson LE, Pierce S, Talpaz M. Result of interferon-alpha therapy in patients with chronic myelogenous leukemia 60 years of age and older. Am J Med. 1996;100(4):452–5. doi: 10.1016/s0002-9343(97)89522-8. [DOI] [PubMed] [Google Scholar]
- 22.Cortes J, Talpaz M, O’Brien S, Giles F, Beth Rios M, Shan J, et al. Effects of age on prognosis with imatinib mesylate therapy for patients with Philadelphia chromosome-positive chronic myelogenous leukemia. Cancer. 2003;98(6):1105–13. doi: 10.1002/cncr.11629. [DOI] [PubMed] [Google Scholar]
- 23.Gugliotta G, Castagnetti F, Palandri F, Breccia M, Intermesoli T, Capucci A, et al. Frontline imatinib treatment of chronic myeloid leukemia: no impact of age on outcome, a survey by the GIMEMA CML WP. Blood. 2011;117(21):5591–99. doi: 10.1182/blood-2010-12-324228. [DOI] [PubMed] [Google Scholar]
- 24.Latagliata R, Breccia M, Carmosino I, Cannella L, De Cuia R, Diverio D, et al. “Real-life” results of front-line treatment with Imatinib in older patients (>/=65 years) with newly diagnosed chronic myelogenous leukemia. Leuk Res. 2010;34(11):1472–5. doi: 10.1016/j.leukres.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 25.Cortes J, De Souza C, Ayala-Sanchez M, Bendit I, Best-Aguilera C, Enrico A, et al. Current patient management of chronic myeloid leukemia in Latin America: a study by the Latin American Leukemia Net (LAL-NET) Cancer. 2010;116(21):4991–5000. doi: 10.1002/cncr.25273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wiggins CL, Harlan LC, Nelson HE, Stevens JL, Willman CL, Libby EN, et al. Age disparity in the dissemination of imatinib for treating chronic myeloid leukemia. Am J Med. 2010;123(8):764.e1–9. doi: 10.1016/j.amjmed.2010.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tonorezos ES, Oeffinger KC. Research challenges in adolescent and young adult cancer survivor research. Cancer. 2011;117(10 Suppl):2295–300. doi: 10.1002/cncr.26058. [DOI] [PubMed] [Google Scholar]
- 28.Bleyer A, Morgan S, Barr R. Proceedings of a workshop: bridging the gap in care and addressing participation in clinical trials. Cancer. 2006;107(7 Suppl):1656–8. doi: 10.1002/cncr.22104. [DOI] [PubMed] [Google Scholar]
- 29.Oeffinger KC, Tonorezos ES. The cancer is over, now what?: Understanding risk, changing outcomes. Cancer. 2011;117(10 Suppl):2250–7. doi: 10.1002/cncr.26051. [DOI] [PubMed] [Google Scholar]
- 30.Robison LL, Armstrong GT, Boice JD, Chow EJ, Davies SM, Donaldson SS, et al. The Childhood Cancer Survivor Study: a National Cancer Institute-supported resource for outcome and intervention research. J Clin Oncol. 2009;27(14):2308–18. doi: 10.1200/JCO.2009.22.3339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Veal GJ, Hartford CM, Stewart CF. Clinical pharmacology in the adolescent oncology patient. J Clin Oncol. 2010;28(32):4790–9. doi: 10.1200/JCO.2010.28.3473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Menon-Andersen D, Mondick JT, Jayaraman B, Thompson PA, Blaney SM, Bernstein M, et al. Population pharmacokinetics of imatinib mesylate and its metabolite in children and young adults. Cancer Chemother Pharmacol. 2009;63(2):229–38. doi: 10.1007/s00280-008-0730-x. [DOI] [PubMed] [Google Scholar]
- 33.Peeters M, Koren G, Jakubovicz D, Zipursky A. Physician compliance and relapse rates of acute lymphoblastic leukemia in children. Clin Pharmacol Ther. 1988;43(3):228–32. doi: 10.1038/clpt.1988.25. [DOI] [PubMed] [Google Scholar]
- 34.Lilleyman JS, Lennard L. Non-compliance with oral chemotherapy in childhood leukaemia. BMJ. 1996;313(7067):1219–20. doi: 10.1136/bmj.313.7067.1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Butow P, Palmer S, Pai A, Goodenough B, Luckett T, King M. Review of adherence-related issues in adolescents and young adults with cancer. J Clin Oncol. 2010;28(32):4800–9. doi: 10.1200/JCO.2009.22.2802. [DOI] [PubMed] [Google Scholar]
- 36.Marin D, Bazeos A, Mahon FX, Eliasson L, Milojkovic D, Bua M, et al. Adherence is the critical factor for achieving molecular responses in patients with chronic myeloid leukemia who achieve complete cytogenetic responses on imatinib. J Clin Oncol. 2010;28(14):2381–8. doi: 10.1200/JCO.2009.26.3087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Festa RS, Tamaroff MH, Chasalow F, Lanzkowsky P. Therapeutic adherence to oral medication regimens by adolescents with cancer. I. Laboratory assessment. J Pediatr. 1992;120(5):807–11. doi: 10.1016/s0022-3476(05)80256-2. [DOI] [PubMed] [Google Scholar]
- 38.Lansky SB, Smith SD, Cairns NU, Cairns GF., Jr Psychological correlates of compliance. Am J Pediatr Hematol Oncol. 1983;5(1):87–92. [PubMed] [Google Scholar]
- 39.Tebbi CK, Cummings KM, Zevon MA, Smith L, Richards M, Mallon J. Compliance of pediatric and adolescent cancer patients. Cancer. 1986;58(5):1179–84. doi: 10.1002/1097-0142(19860901)58:5<1179::aid-cncr2820580534>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- 40.Phipps S, DeCuir-Whalley S. Adherence issues in pediatric bone marrow transplantation. J Pediatr Psychol. 1990;15(4):459–75. doi: 10.1093/jpepsy/15.4.459. [DOI] [PubMed] [Google Scholar]
- 41.Dolgin MJ, Katz ER, Doctors SR, Siegel SE. Caregivers’ perceptions of medical compliance in adolescents with cancer. J Adolesc Health Care. 1986;7(1):22–7. doi: 10.1016/s0197-0070(86)80090-0. [DOI] [PubMed] [Google Scholar]
- 42.Tebbi CK. Treatment compliance in childhood and adolescence. Cancer. 1993;71(10 Suppl):3441–9. doi: 10.1002/1097-0142(19930515)71:10+<3441::aid-cncr2820711751>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- 43.Jamison RN, Lewis S, Burish TG. Cooperation with treatment in adolescent cancer patients. J Adolesc Health Care. 1986;7(3):162–7. doi: 10.1016/s0197-0070(86)80032-8. [DOI] [PubMed] [Google Scholar]
- 44.Lancaster D, Lennard L, Lilleyman JS. Profile of non-compliance in lymphoblastic leukaemia. Arch Dis Child. 1997;76(4):365–6. doi: 10.1136/adc.76.4.365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ault P, Kantarjian H, O’Brien S, Faderl S, Beran M, Rios MB, et al. Pregnancy among patients with chronic myeloid leukemia treated with imatinib. J Clin Oncol. 2006;24(7):1204–8. doi: 10.1200/JCO.2005.04.6557. [DOI] [PubMed] [Google Scholar]
- 46.Pye SM, Cortes J, Ault P, Hatfield A, Kantarjian H, Pilot R, et al. The effects of imatinib on pregnancy outcome. Blood. 2008;111(12):5505–8. doi: 10.1182/blood-2007-10-114900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wood WA, Lee SJ. Malignant hematologic diseases in adolescents and young adults. Blood. 2011;117(22):5803–15. doi: 10.1182/blood-2010-12-283093. [DOI] [PubMed] [Google Scholar]
- 48.Pemmaraju N, Kantarjian H, Ravandi F, O’Brien S, Wierda W, Thomas D, et al. Acute myeloid leukemia (AML) in adolescents and young adults (AYA): The M. D. Anderson Cancer Center (MDACC) experience. Poster session presented at: Proceedings of the 45th American Society of Clinical Oncology (ASCO); 2009 May 29–June 2; Orlando, FL, USA. [Google Scholar]
- 49.Suttorp M, Millot F. Treatment of pediatric chronic myeloid leukemia in the year 2010: use of tyrosine kinase inhibitors and stem-cell transplantation. Hematology Am Soc Hematol Educ Program. 2010;2010:368–76. doi: 10.1182/asheducation-2010.1.368. [DOI] [PubMed] [Google Scholar]
- 50.Millot F, Traore P, Guilhot J, Nelken B, Leblanc T, Leverger G, et al. Clinical and biological features at diagnosis in 40 children with chronic myeloid leukemia. Pediatrics. 2005;116(1):140–3. doi: 10.1542/peds.2004-2473. [DOI] [PubMed] [Google Scholar]
- 51.Adler R, Viehmann S, Kuhlisch E, Martiniak Y, Rottgers S, Harbott J, et al. Correlation of BCR/ABL transcript variants with patients’ characteristics in childhood chronic myeloid leukaemia. Eur J Haematol. 2009;82(2):112–8. doi: 10.1111/j.1600-0609.2008.01170.x. [DOI] [PubMed] [Google Scholar]
- 52.Champagne MA, Capdeville R, Krailo M, Qu W, Peng B, Rosamilia M, et al. Imatinib mesylate (STI571) for treatment of children with Philadelphia chromosome-positive leukemia: results from a Children’s Oncology Group phase 1 study. Blood. 2004;104(9):2655–60. doi: 10.1182/blood-2003-09-3032. [DOI] [PubMed] [Google Scholar]
- 53.Pulsipher MA. Treatment of CML in pediatric patients: should imatinib mesylate (STI-571, Gleevec) or allogeneic hematopoietic cell transplant be front-line therapy? Pediatr Blood Cancer. 2004;43(5):523–33. doi: 10.1002/pbc.20062. [DOI] [PubMed] [Google Scholar]
- 54.Hasford J, Baccarani M, Hoffmann V, Guilhot J, Saussele S, Rosti G, et al. Predicting complete cytogenetic response and subsequent progression-free survival in 2060 patients with CML on imatinib treatment: the EUTOS score. Blood. 2011;118(3):686–92. doi: 10.1182/blood-2010-12-319038. [DOI] [PubMed] [Google Scholar]
- 55.Sokal JE, Cox EB, Baccarani M, Tura S, Gomez GA, Robertson JE, et al. Prognostic discrimination in “good-risk” chronic granulocytic leukemia. Blood. 1984;63(4):789–99. [PubMed] [Google Scholar]
- 56.Saussele S, Lauseker M, Gratwohl A, Beelen DW, Bunjes D, Schwerdtfeger R, et al. Allogeneic hematopoietic stem cell transplantation (allo SCT) for chronic myeloid leukemia in the imatinib era: evaluation of its impact within a subgroup of the randomized German CML Study IV. Blood. 2010;115(10):1880–5. doi: 10.1182/blood-2009-08-237115. [DOI] [PubMed] [Google Scholar]
- 57.Saussele S, Lauseker M, Gratwohl A, Beelen DW, Bunjes D, Schwerdtfeger R, et al. Allogeneic hematopoietic stem cell transplantation (allo SCT) for chronic myeloid leukemia in the imatinib era: evaluation of its impact within a subgroup of the randomized German CML Study IV. Blood. 2010;115(10):1880–5. doi: 10.1182/blood-2009-08-237115. [DOI] [PubMed] [Google Scholar]


