Abstract
The JIL-1 kinase is a multidomain protein that localizes specifically to euchromatin interband regions of polytene chromosomes and is the kinase responsible for histone H3S10 phosphorylation at interphase. Genetic interaction assays have suggested that the function of the epigenetic histone H3S10ph mark is to antagonize heterochromatization by participating in a dynamic balance between factors promoting repression and activation of gene expression as measured by position-effect variegation (PEV) assays. Interestingly, JIL-1 loss-of-function alleles can act either as an enhancer or indirectly as a suppressor of wm4 PEV depending on the precise levels of JIL-1 kinase activity. In this study, we have explored the relationship between PEV and the relative levels of the H3S10ph and H3K9me2 marks at the white gene in both wild-type and wm4 backgrounds by ChIP analysis. Our results indicate that H3K9me2 levels at the white gene directly correlate with its level of expression and that H3K9me2 levels in turn are regulated by H3S10 phosphorylation.
Keywords: ChIP, Drosophila, JIL-1 kinase, PEV, gene expression, heterochromatin
Introduction
In the absence of H3S10 phosphorylation by the JIL-1 kinase the major heterochromatin markers H3K9me2, HP1a and Su(var)3–7 spread to ectopic locations on the chromosome arms of Drosophila polytene chromosomes.1-3 These observations suggested a model for a dynamic balance between euchromatin and heterochromatin,1,3-5 where the level of gene expression is determined by antagonistic functions of the euchromatic H3S10ph mark and the heterochromatic H3K9me2 mark.3,5-8 Wang et al.5 tested this model by transgenically expressing various truncated versions of JIL-1, with or without kinase activity, and correlating their effect on PEV with the levels of the H3S10ph and H3K9me2 marks at a hsp70-white reporter gene as determined by ChIP assays in the pericentric insertion line 118E-10. At pericentric sites loss-of-function alleles of JIL-1 act as enhancers of PEV whereas gain-of-function alleles act as suppressors.7 As predicted by the model, the results of Wang et al.5 showed that the level of the H3K9me2 mark at the reporter was inversely proportional to the H3S10ph level. PEV was enhanced with increased levels of H3K9me2 in the absence of H3S10 phosphorylation and PEV was suppressed with increased levels of the H3S10ph mark and a concomitant decrease in the level of the H3K9me2 mark.5 However, it has been demonstrated that JIL-1 can act both as an enhancer as well as a suppressor of wm4 PEV depending on the precise levels of JIL-1,3,6 and that the genetic interactions between JIL-1 and the Su(var)3–9 and Su(var)2–5 alleles in regulating PEV of wm4 are more complex than in the case of 118E-10 where reduced levels of JIL-1 always act as an enhancer.7,8Therefore, in the present study we explored the relationship between PEV and the relative levels of the H3S10ph and H3K9me2 marks at the white gene in both wild-type and wm4 backgrounds by ChIP analysis.
Results and Discussion
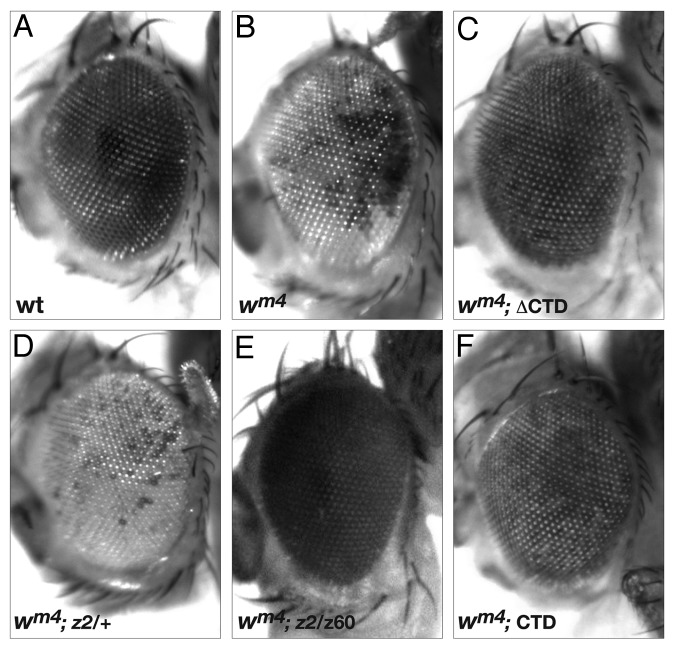
The In(1)wm4 X chromosome contains an inversion that juxtaposes the euchromatic white gene and centric heterochromatic sequences distal to the nucleolus organizer (Fig. 1).9,10 The resulting somatic variegation of wm4 expression occurs in clonal patches in the eye reflecting heterochromatic spreading from the inversion breakpoint that silences wm4 expression in the white patches and euchromatic packaging of the w gene in those patches that appear red (reviewed in ref. 11) (Fig. 2B). Studies of this effect suggest that the degree of spreading may depend on the amount of heterochromatic factors at the breakpoint (reviewed in refs. 12 and 13). Interestingly, strong hypomorphic combinations of JIL-1 alleles, in which heterochromatic factors spread to ectopic locations,1,2 act as suppressors not enhancers of PEV of the wm4 allele (Fig. 2E).6 Based on these findings, Lerach et al.6 proposed a model where the suppression of PEV of wm4 in strong JIL-1 hypomorphic backgrounds is due to a reduction in the level of heterochromatic factors at the pericentromeric heterochromatin near the inversion breakpoint site that reduces its potential for heterochromatic spreading and silencing (Fig. 1).
Figure 1. Diagrams of the distribution of heterochromatic factors relative to the white gene in wild-type and wm4 X chromosomes. (A) In wild-type high levels of heterochromatic factors are confined to pericentric heterochromatin (in black) and the distally located white gene is expressed at normal levels. (B) In wm4 an inversion juxtaposes the white gene to the pericentric heterochromatin where spreading of heterochromatic factors across the inversion breakpoint (in dark gray) represses white expression as reflected in PEV. (C) In a JIL-1 null wm4 background in the absence of H3S10 phosphorylation there is a redistribution of heterochromatic factors from the pericentric heterochomatin to ectopic locations on the chromosome arms (light gray). The resulting decrease in the level of heterochromatic factors at the pericentric heterochromatin (in dark gray) reduces its potential for heterochromatic spreading and gene silencing and under these conditions the white gene is expressed.
Figure 2. The effect on PEV of the wm4 allele by JIL-1 hypomorphic alleles or by expression of the CTD or the ΔCTD. Examples of the degree of PEV in the eyes of (A) wild-type flies, (B) wm4 flies, (C) wm4 flies expressing the ΔCTD, (D) wm4; JIL-1z2/+ flies, (E) wm4; JIL-1z2/ JIL-1z60 flies and (F) wm4 flies expressing the CTD. All pictures are from male flies. PEV of the wm4 allele in the strong hypomorphic JIL-1z2/ JIL-1z60 background is shown in (E) because JIL-1z2/ JIL-1z2 null mutant backgrounds have no adult escapers.
In order to measure how the relative H3S10ph and H3K9me2 levels at the white gene were affected in the experiments, we performed ChIP assays as in Legube et al.14 and in Wang et al.5 Chromatin was immunoprecipitated (ip) from larval salivary glands from wild-type, the inversion line wm4, the inversion line in either a heterozygous JIL-1 mutant background wm4; JIL1z2/+ or a homozygous JIL-1 mutant background wm4; JIL-1z2/JIL-1z2, or the inversion line with either a transgenic JIL-1 CTD or ΔCTD construct wm4; “JIL-1 transgene”/+; da-GAL4/+ using rabbit anti-H3S10ph antibody or purified rabbit IgG antibody (negative control) or mAbs to H3K9me2 or GST (negative control). Primers that amplify region -110 to +65 of the white gene were used to amplify the precipitated material. Experiments were done in triplicate and relative enrichment of white DNA from the H3S10ph and H3K9me2 ips were normalized to the corresponding control antibody ips performed in tandem for each experimental sample. Statistical comparisons using the various genotypes are provided in Tables 1 and 2. As illustrated in Figure 3 there is a relatively higher enrichment of H3S10ph (4.3 ± 0.8) than of H3K9me2 (2.0 ± 0.5) at the white gene in wild-type. However, compared with wild-type (Fig. 2A) the variegated eye phenotype in wm4 (Fig. 2B) is correlated with an almost 4-fold decrease in relative H3S10ph enrichment levels (1.4 ± 0.4) and a more than 3-fold increase in the relative H3K9me2 enrichment levels (6.7 ± 1.6). Interestingly, this distribution can be reversed by expression of a JIL-1 ΔCTD construct which lacks the COOH-terminal sequences required for proper chromatin localization leading to mislocalization of the protein.15 However, it does retain its kinase activity resulting in ectopic histone H3S10 phosphorylation15 and suppression of PEV (Fig. 2C).5 As shown in Figure 3 expression of JIL-1 ΔCTD in a wm4 background leads to a dramatic increase of almost 10-fold in the relative enrichment of H3S10ph (9.9 ± 0.9) at the white gene accompanied with a decrease in the relative enrichment of H3K9me2 (1.7 ± 0.1) to levels comparable to wild-type (Tables 1 and 2).
Table 1. Statistical comparison of H3K9me2 levels.
|
Genotype |
wm4 |
wm4; z2/+ |
wm4; z2/z2 |
wm4; CTD |
wm4; ΔCTD |
| wt |
p < 0.001 |
p < 0.001 |
p > 0.4 |
p > 0.5 |
p > 0.8 |
| wm4 |
- |
p > 0.9 |
p < 0.001 |
p < 0.001 |
p < 0.001 |
| wm4; z2/+ |
- |
- |
p < 0.001 |
p < 0.001 |
p < 0.001 |
| wm4; z2/z2 |
- |
- |
- |
p > 0.8 |
p > 0.5 |
| wm4; CTD | - | - | - | - | p > 0.7 |
For each genotype the average relative enrichment from three sets of measurements were compared using a two-tailed Student’s t-test.
Table 2. Statistical comparison of H3S10ph levels.
|
Genotype |
wm4 |
wm4; z2/+ |
wm4; z2/z2 |
wm4; CTD |
wm4; ΔCTD |
| wt |
p < 0.005 |
p < 0.002 |
p < 0.002 |
p < 0.001 |
p < 0.0001 |
| wm4 |
- |
p > 0.9 |
p > 0.7 |
p > 0.5 |
p < 0.0001 |
| wm4; z2/+ |
- |
- |
p > 0.8 |
p > 0.6 |
p < 0.0001 |
| wm4; z2/z2 |
- |
- |
- |
p > 0.7 |
p < 0.0001 |
| wm4; CTD | - | - | - | - | p < 0.0001 |
For each genotype the average relative enrichment from three sets of measurements were compared using a two-tailed Student’s t-test.
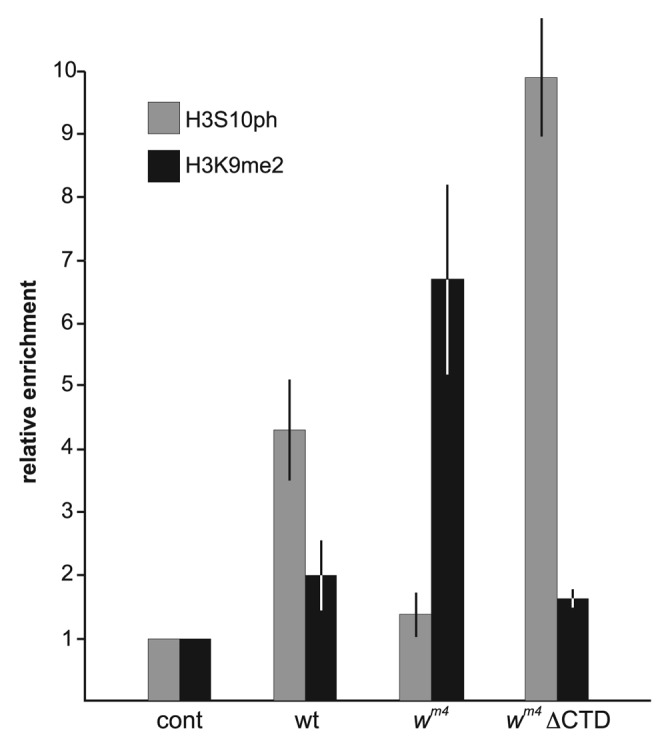
Figure 3. ChIP analysis of the white gene in wild-type and wm4 backgrounds. Histograms of the relative enrichment of chromatin immunoprecipitated by anti-H3S10ph or anti-H3K9me2 antibody from third instar larval salivary glands from wild-type, wm4 or wm4 expressing the ΔCTD. For each experimental condition the average relative enrichment normalized to the corresponding control immunoprecipitation from three independent experiments with SD is shown.
Because strong hypomorphic combinations of JIL-1 alleles act as suppressors of PEV (Fig. 2E), the prediction of the model of Lerach et al.6 (Fig. 1) is that the relative enrichment of H3K9me2 at the white gene in a wm4; JIL-1z2/JIL-1z2 background should be at or below wild-type levels. JIL-1z2 is a true null allele16 without any detectable H3S10 phosphorylation in interphase cells such as third instar salivary gland cells.15 As illustrated in Figure 4 we found almost no relative enrichment compared with the antibody control ips of both H3K9me2 (1.1 ± 0.1) and H3S10ph (1.1 ± 0.1) in agreement with the above hypothesis. Interestingly, it has been demonstrated that JIL-1z2 can act as an haplo-enhancer3,8 (Fig. 2D) and as shown in Figure 4 when only one copy of JIL-1z2 is present in a wm4 background the relative enrichment of H3K9me2 (6.7 ± 0.8) increases 6-fold compared with the homozygous JIL-1z2 condition whereas the relative enrichment of H3S10ph (1.3 ± 0.4) is indistinguishable from that in the wm4 and wm4; JIL-1z2/JIL-1z2 backgrounds (Tables 1 and 2). Furthermore, when a construct containing only the COOH-terminal domain (CTD) of JIL-1 is expressed in a wild-type background it has a dominant-negative effect and displaces endogenous JIL-1 leading to a striking decrease in histone H3S10ph levels15 and spreading of H3K9me2 to the chromosome arms as in JIL-1z2/JIL-1z2 null mutants.5 As illustrated in Figure 4, expression of the CTD in a wm4 background leads to a relative enrichment of the H3K9me2 (1.4 ± 0.3) and H3S10ph (0.9 ± 0.1) marks indistinguishable from that in the wm4; JIL-1z2/JIL-1z2 background (Tables 1 and 2). Furthermore, this low level of H3K9me2 mark at the white gene is correlated with suppression of PEV (Fig. 2F).5
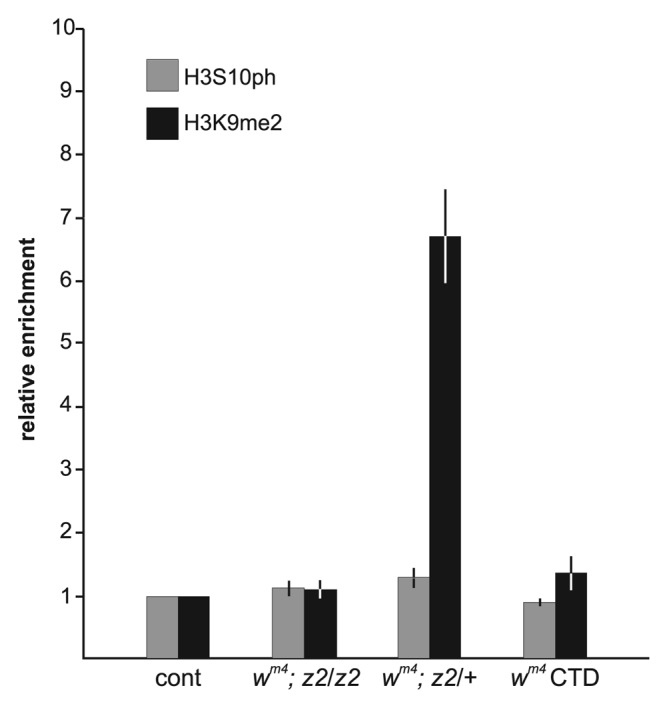
Figure 4. ChIP analysis of the white gene in wm4 , JIL-1 hypomorphic allelic backgrounds. Histograms of the relative enrichment of chromatin immunoprecipitated by anti-H3S10ph or antiH3K9me2 antibody from third instar larval salivary glands from wm4; JIL-1z2/ JIL-1z2 , wm4; JIL-1z2/+ or wm4 expressing the CTD. For each experimental condition the average relative enrichment normalized to the corresponding control immunoprecipitation from three independent experiments with SD is shown.
Taken together the present experiments indicate that H3K9me2 levels at the white gene directly correlate with its level of expression and that H3K9me2 levels in turn are regulated by H3S10 phosphorylation providing strong support for the model depicted in Figure 1. In wild type, there are low levels of the epigenetic H3K9me2 mark at the white gene resulting in its normal expression. However, in the wm4 allele heterochromatic factors can spread across the inversion breakpoint leading to high levels of H3K9me2 at the white gene and silencing of gene expression. Interestingly, we show that this increase in H3K9me2 level can be counteracted by ectopic H3S10 phosphorylation at the white gene restoring gene expression. In contrast, in the absence of H3S10 phosphorylation as it occurs in strong JIL-1 hypomorphic mutant backgrounds there is a redistribution of heterochromatic factors to ectopic chromosome sites resulting in reduced levels of these factors at the pericentric heterochromatin. This leads to less heterochromatic spreading and low levels of H3K9me2 at the white gene in the wm4 inversion, thus allowing for white gene expression. This expression occurs without H3S10 phosphorylation providing additional evidence that the H3S10 mark is not required for RNA polymerase II-mediated transcription17 but rather regulates transcription indirectly by counteracting H3K9 dimethylation and gene silencing.
Materials and Methods
Drosophila melanogaster stocks
Fly stocks were maintained at 25°C according to standard protocols.18 The JIL-1z2 and the JIL-1z60 alleles are described in Wang et al.16 as well as in Zhang et al.19 The ΔCTD construct containing residue 1–926 and the CTD construct containing sequences from aa 927–1207 in the pYES vector are described in Wang et al.5 Expression of the transgenes were driven using a daGAL4 driver introduced by standard genetic crosses. The driver line and the In(1)wm4 allele was obtained from the Bloomington Stock Center. Balancer chromosomes and markers are described in Lindsley and Zimm.20 wm4 flies or larvae (wm4/Y or wm4) in combination with JIL-1 mutant alleles or expressing the CTD or ΔCTD constructs were generated by standard crossing. Eyes from representative individuals from these crosses were photographed using an Olympus Stereo Microscope and a Spot digital camera (Diagnostic Instruments).
Chromatin immunoprecipitation
For ChIP experiments 50 pairs of salivary glands per sample were dissected from third instar larvae and fixed for 15 min at room temperature in 1 mL of fixative (50 mM HEPES at pH 7.6, 100 mM NaCl, 0.1 mM EDTA at pH 8, 0.5 mM EGTA at pH 8, 2% formaldehyde). Preparation of chromatin for immunoprecipitation was performed as previously described in Legube et al.14 Rabbit anti-H3S10ph antibody (Active Motif), purified rabbit IgG antibody (Sigma), anti-H3K9me2 mAb (Abcam), or anti-GST mAb 8C721 was used for immunoprecipitation. For each sample the chromatin lysate was divided into equal amounts and immunoprecipitated with experimental and control antibody, respectively. DNA from the immunoprecipitated chromatin fragments (500 bp average) was purified by a Wizard SV DNA purification kit (Promega). The isolated DNA was used as template for quantitative real-time (qRT) PCR performed with the Stratagene Mx4000 real-time cycler. The PCR mixture contained Brilliant II SYBR Green QPCR Master Mix (Stratagene) as well as the corresponding primers: white-forward 5′-GTGCTGTGCCAAAACTCCTC-3′, white-reverse 5′-GATGCTCGGCAGATGGGTTGT-3′ which amplify region -110 to +65. Cycling parameters were 10 min at 95°C, followed by 40 cycles of 30 sec at 95°C, 30 sec at 55°C, and 30 sec at 72°C. Fluorescence intensities were plotted against the number of cycles using an algorithm provided by Stratagene. DNA levels were quantified using a calibration curve based on dilution of concentrated DNA. For each experimental condition the relative enrichment was normalized to the corresponding control immunoprecipitation from the same chromatin lysate.
Acknowledgments
We thank members of the laboratory for discussion, advice, and critical reading of the manuscript. We also wish to acknowledge Mr. Atrez Norwood for technical assistance. This work was supported by NIH Grant GM062916 (K.M.J./J.J.).
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Footnotes
Previously published online: www.landesbioscience.com/journals/fly/article/20029
References
- 1.Zhang W, Deng H, Bao X, Lerach S, Girton J, Johansen J, et al. The JIL-1 histone H3S10 kinase regulates dimethyl H3K9 modifications and heterochromatic spreading in Drosophila. Development. 2006;133:229–35. doi: 10.1242/dev.02199. [DOI] [PubMed] [Google Scholar]
- 2.Deng H, Bao X, Zhang W, Girton J, Johansen J, Johansen KM. Reduced levels of Su(var)3-9 but not Su(var)2-5 (HP1) counteract the effects on chromatin structure and viability in loss-of-function mutants of the JIL-1 histone H3S10 kinase. Genetics. 2007;177:79–87. doi: 10.1534/genetics.107.075143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Deng H, Cai W, Wang C, Lerach S, Delattre M, Girton J, et al. JIL-1 and Su(var)3-7 interact genetically and counteract each other’s effect on position-effect variegation in Drosophila. Genetics. 2010;185:1183–92. doi: 10.1534/genetics.110.117150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ebert A, Schotta G, Lein S, Kubicek S, Krauss V, Jenuwein T, et al. Su(var) genes regulate the balance between euchromatin and heterochromatin in Drosophila. Genes Dev. 2004;18:2973–83. doi: 10.1101/gad.323004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang C, Cai W, Li Y, Deng H, Bao X, Girton J, et al. The epigenetic H3S10 phosphorylation mark is required for counteracting heterochromatic spreading and gene silencing in Drosophila melanogaster. J Cell Sci. 2011;124:4309–17. doi: 10.1242/jcs.092585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lerach S, Zhang W, Bao X, Deng H, Girton J, Johansen J, et al. Loss-of-function alleles of the JIL-1 kinase are strong suppressors of position effect variegation of the wm4 allele in Drosophila. Genetics. 2006;173:2403–6. doi: 10.1534/genetics.106.059253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bao X, Deng H, Johansen J, Girton J, Johansen KM. Loss-of-function alleles of the JIL-1 histone H3S10 kinase enhance position-effect variegation at pericentric sites in Drosophila heterochromatin. Genetics. 2007;176:1355–8. doi: 10.1534/genetics.107.073676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang C, Girton J, Johansen J, Johansen KM. A balance between euchromatic (JIL-1) and heterochromatic [SU(var)2-5 and SU(var)3-9] factors regulates position-effect variegation in Drosophila. Genetics. 2011;188:745–8. doi: 10.1534/genetics.111.129353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Muller HJ. Types of visible variegations induced by X-rays in Drosophila. J Genet. 1930;22:299–335. doi: 10.1007/BF02984195. [DOI] [Google Scholar]
- 10.Pirrotta V, Hadfield C, Pretorius GHJ. Microdissection and cloning of the white locus and the 3B1-3C2 region of the Drosophila X chromosome. EMBO J. 1983;2:927–34. doi: 10.1002/j.1460-2075.1983.tb01523.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Grewal SI, Elgin SC. Heterochromatin: new possibilities for the inheritance of structure. Curr Opin Genet Dev. 2002;12:178–87. doi: 10.1016/S0959-437X(02)00284-8. [DOI] [PubMed] [Google Scholar]
- 12.Weiler KS, Wakimoto BT. Heterochromatin and gene expression in Drosophila. Annu Rev Genet. 1995;29:577–605. doi: 10.1146/annurev.ge.29.120195.003045. [DOI] [PubMed] [Google Scholar]
- 13.Girton JR, Johansen KM. Chromatin structure and the regulation of gene expression: the lessons of PEV in Drosophila. Adv Genet. 2008;61:1–43. doi: 10.1016/S0065-2660(07)00001-6. [DOI] [PubMed] [Google Scholar]
- 14.Legube G, McWeeney SK, Lercher MJ, Akhtar A. X-chromosome-wide profiling of MSL-1 distribution and dosage compensation in Drosophila. Genes Dev. 2006;20:871–83. doi: 10.1101/gad.377506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bao X, Cai W, Deng H, Zhang W, Krencik R, Girton J, et al. The COOH-terminal domain of the JIL-1 histone H3S10 kinase interacts with histone H3 and is required for correct targeting to chromatin. J Biol Chem. 2008;283:32741–50. doi: 10.1074/jbc.M806227200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang Y, Zhang W, Jin Y, Johansen J, Johansen KM. The JIL-1 tandem kinase mediates histone H3 phosphorylation and is required for maintenance of chromatin structure in Drosophila. Cell. 2001;105:433–43. doi: 10.1016/S0092-8674(01)00325-7. [DOI] [PubMed] [Google Scholar]
- 17.Cai W, Bao X, Deng H, Jin Y, Girton J, Johansen J, et al. RNA polymerase II-mediated transcription at active loci does not require histone H3S10 phosphorylation in Drosophila. Development. 2008;135:2917–25. doi: 10.1242/dev.024927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roberts DB. In Drosophila: A Practical Approach IRL Press, Oxford, UK, 1998. [Google Scholar]
- 19.Zhang W, Jin Y, Ji Y, Girton J, Johansen J, Johansen KM. Genetic and phenotypic analysis of alleles of the Drosophila chromosomal JIL-1 kinase reveals a functional requirement at multiple developmental stages. Genetics. 2003;165:1341–54. doi: 10.1093/genetics/165.3.1341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lindsley DL, Zimm GG. The genome of Drosophila melanogaster Academic Press, New York, NY, 1992. [Google Scholar]
- 21.Rath U, Wang D, Ding Y, Xu Y-Z, Qi H, Blacketer MJ, et al. Chromator, a novel and essential chromodomain protein interacts directly with the putative spindle matrix protein skeletor. J Cell Biochem. 2004;93:1033–47. doi: 10.1002/jcb.20243. [DOI] [PubMed] [Google Scholar]