Abstract
OBJECTIVES:
FTY720 modulates CD4+T cells by the augmentation of regulatory T cell activity, secretion of suppressive cytokines and suppression of IL-17 secretion by Th17 cells. To further understand the process of graft rejection/acceptance, we evaluated skin allograft survival and associated events after FTY720 treatment.
METHODS:
F1 mice (C57BL/6xBALB/c) and C57BL/6 mice were used as donors for and recipients of skin transplantation, respectively. The recipients were transplanted and either not treated or treated with FTY720 by gavage for 21 days to evaluate the allograft survival. In another set of experiments, the immunological evaluation was performed five days post-transplantation. The spleens, axillary lymph nodes and skin allografts of the recipient mice were harvested for phenotyping (flow cytometry), gene expression (real-time PCR) and cytokine (Bio-Plex) analysis.
RESULTS:
The FTY720 treatment significantly increased skin allograft survival, reduced the number of cells in the lymph nodes and decreased the percentage of Tregs at this site in the C57BL/6 recipients. Moreover, the treatment reduced the number of graft-infiltrating cells and the percentage of CD4+ graft-infiltrating cells. The cytokine analysis (splenocytes) showed decreased levels of IL-10, IL-6 and IL-17 in the FTY720-treated mice. We also observed a decrease in the IL-10, IL-6 and IL-23 mRNA levels, as well as an increase in the IL-27 mRNA levels, in the splenocytes of the treated group. The FTY720-treated mice exhibited increased mRNA levels of IL-10, IL-27 and IL-23 in the skin graft.
CONCLUSIONS:
Our results demonstrated prolonged but not indefinite skin allograft survival by FTY720 treatment. This finding indicates that the drug did not prevent the imbalance between Tr1 and Th17 cells in the graft that led to rejection.
Keywords: Transplantation, Rejection, Tolerance, FTY720, Foxp3, Th17
INTRODUCTION
The rejection responses to allografts are initiated by a specific T-cell recognition of antigens expressed on the cells within the graft (1-3). T-cell activation after allograft transplantation requires both antigen presentation and co-stimulatory signals (4,5), and, once they occur, several other factors are involved, leading to effector or regulatory immune responses. Recently, the immune responses to allografts have been reported to be modulated by regulatory T cells, such as CD4+CD25+ T cells, which comprise 5-10% of the peripheral CD4+ T cells in adult rodents and humans (6). These cells play an important role in the maintenance of self-tolerance, prevention of autoimmune diseases (7), and regulation of allogeneic immune responses (8,9).
Regulatory natural T cells (nTregs) are generated in the thymus, defined by their high expression of the IL-2 receptor α chain (CD25) and thought to be controlled by the forkhead box P3 (Foxp3) transcription factor. The population of thymus-derived Treg cells is supplemented with a population of inducible Tregs (iTregs) generated from naïve T cells activated in the presence of TGF-β. iTregs are believed to be functionally similar to thymus-derived nTregs (10).
In contrast with Treg-generated transplantation tolerance, the Th17 cell population has been associated with allograft rejection (11). Th17s are described as T cells producing interleukin-17 that are dependent on signals from cytokines, such as IL-6 (12). Moreover, the Th17 response is propagated by the cytokines IL-23 and IL-21 (13,14).
A member of the IL-12 family of cytokines, namely IL-27, has been shown to cause the immune-specific suppression of IL-17 secretion by down-regulating retinoid orphan nuclear receptor C expression. This inhibition is associated with the induction of the intracellular signaling factor STAT1 and the suppression of cytokine signaling protein 1 (15).
Investigations of the Treg-Th17 axis aim to identify potential pharmacological targets to promote graft acceptance.
The new compound FTY720 binds to S1P (sphingosine-1 phosphate) receptors, causing lymphocytes to be sequestered in secondary lymphoid organs. In addition, FTY720 inhibits the egress of lymphocytes from the thymus and secondary lymphoid tissues into the periphery, thereby preventing the migration of lymphocytes to inflammatory sites (16,17). Some additional mechanisms of action have been proposed for FTY720, such as its anti-proliferative effect on T cells through the augmentation of regulatory T cell activity and secretion of suppressive cytokines (18-20). FTY720 has also been shown to cause the suppression of IL-17 secretion by Th17 cells in vitro (15).
Jin et al. (21) showed that, although FTY720 improved CD4CD25Foxp3 expression after skin transplantation, the monotherapy was not sufficient to prevent rejection. However, when MR1 (anti-CD154 mAb) was administered in combination with FTY720, skin allograft survival increased. Interestingly, the expression of CD4CD25Foxp3 was similar when FTY720 monotherapy was compared with FTY720+MR1. These findings suggest that mechanisms other than CD4CD25Foxp3 expression occur during allograft acceptance. To further understand the process of graft acceptance/rejection, we evaluated skin allograft survival and associated events following treatment with FTY720.
MATERIALS AND METHODS
Experimental groups
Mice were cared for in accordance with the Principles of Laboratory Animal Care (NIH publication 86-23, revised 1985) and the regulations of the Brazilian Committee on Animal Experimentation. The project was submitted to and approved by the Animal Ethic Committee of UNIFESP Federal University of São Paulo (protocol 0125/08).
Eight- to ten-week-old male (C57BL/6xBALB/c) F1 and C57BL/6 mice were used as donors for and recipients of skin transplantation, respectively. Skin from the donor tail (1.0 cm2) was placed on the C57BL/6 mice and fixed with 6-0 nylon sutures (Brasuture, Brazil) at each corner. The recipient mice were either transplanted only (Tx, n = 5) or transplanted and submitted to the daily administration of FTY720 (Novartis, Switzerland; 1 mg/kg/day diluted in distilled water) by gavage for 21 days, starting three days before the transplantation (Tx + FTY, n = 5). The grafts were observed daily to establish skin allograft survival (rejection = 90% skin necrosis). In another set of experiments, the immunological evaluation was performed five days post-transplantation.
Experimental design
1) To evaluate skin allograft survival, the following steps were completed: FTY720 treatment, skin transplantation, and graft follow-up.
2) An immunological evaluation of C57BL/6-transplanted mice either treated or not treated with FTY720 was performed.
Immunological evaluation
Five days post-transplantation, the spleens, axillary lymph nodes (ALNs) and skin allografts were harvested from the recipient mice. Single-cell suspensions from the spleen, ALNs and skin were counted in a Neubauer chamber. The spleen cells were used for in vitro stimulation and qPCR analysis, whereas the ALNs and graft-infiltrating cells were evaluated by flow cytometry.
Flow cytometry
Cell suspensions (1×106) from the ALNs and grafts were incubated for 30 min with rat anti-mouse monoclonal antibody CD4PerCP-Cy5.5 (eBioscience, San Diego, CA, USA). After this period, the cells were washed with PBS, fixed for 15 min with 1% paraformaldehyde, permeabilized for 6 min with 0.2% Triton X-100 and stained with rat anti-mouse Foxp3 FITC for 30 min (eBioscience, San Diego, CA, USA). The stained cells were acquired in a FACSCalibur Flow Cytometer (BD Biosciences, San Jose, CA, USA), and the data were analyzed using Cell Quest Pro software (BD Biosciences). At least 30,000 events were acquired for each analysis.
Quantitative analysis of gene expression by real-time PCR
The total RNA from the skin graft and spleen cells was extracted with Trizol (Invitrogen Life Technologies) according to the manufacturer's instructions and reverse-transcribed into complementary DNA with Superscript III (Invitrogen Life Technologies). For the real-time PCR of the IL-10, IL-6, FOXP3, IL-23 and IL-27 genes, 0.5 µL of primer, 1 µL of cDNA, 3.5 µL of RNAse/DNAse-free water and 5 µL of Taqman Fast Universal Master Mix (Applied Biosystems®) were used. The PCR was performed in an Applied Biosystems 7900HT Fast Real-Time PCR System®. The mRNA level was determined following normalization to β-actin. We performed qPCR in the spleen for IL-10, IL-6, FOXP3, IL-23 and IL-27. In the skin graft, we performed qPCR for IL-10, FOXP3, IL-23 and IL-27.
Bio-Plex
The spleen cells were harvested at day five post-skin transplantation and stimulated in vitro with anti-CD3. A Bio-Plex mouse cytokine assay kit (Bio-Rad Laboratories, Hercules, CA, USA) was used to test the samples for the presence of the specific cytokines IL-10, IL-6 and IL-17. The assay was performed in the Bio-Plex suspension array system, and the data were analyzed using Bio-Plex Manager software version 4.0. The standard curves ranged from 1.95 to 32 000 pg/ml.
Statistical analysis
The skin allograft survival curve was obtained using the Kaplan-Meier method. Other data were expressed as the means±SD. The statistical analyses were performed using Graph Pad Prism version 5.00 for Windows (Graph Pad Software, San Diego, CA, USA). The parametric Student's t test was employed, and p-values <0.05 were considered significant.
RESULTS
Treatment with FTY720 increases skin allograft survival
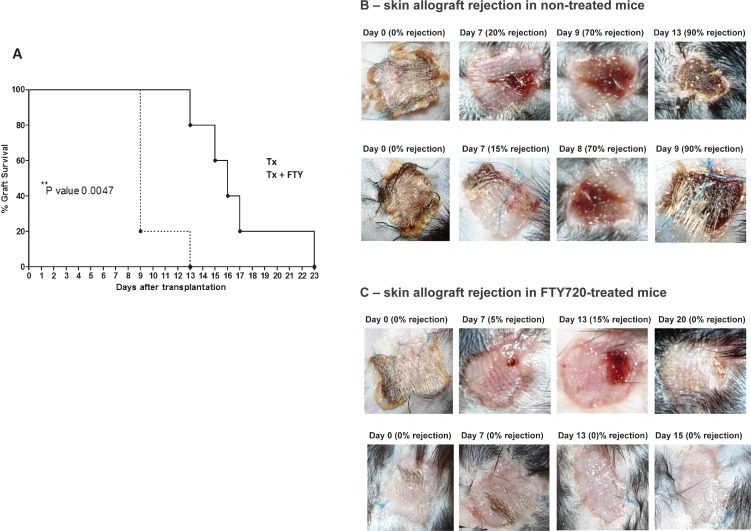
To evaluate the effect of FTY720 on the progression of skin allograft rejection, the recipient mice were either transplanted only (Tx, n = 5) or transplanted and submitted to the daily administration of FTY720 (Tx + FTY, n = 5) by gavage for 21 days, starting three days before the transplantation. We observed a significant (p-value = 0.0047) increase in the survival of allografts from F1 mice (BALB/c x C57BL/6) when the recipient C57BL/6 mice were treated with FTY720 (Figure 1A). The macroscopic examination shows that the first rejection (90% of skin necrosis) occurred on day 13 for the FTY720-treated mice, whereas the first rejection occurred as early as 9 days after transplantation in the non-treated mice (Figure 1B).
Figure 1.
FTY720 treatment prolongs mouse skin allograft survival. F1 (BALB/c x C57BL/6) mice were donors to C57BL/6 recipients, and skin allograft rejection was determined macroscopically when the graft reached a necrosis level of 90%. A - Kaplan-Meier curve of skin allograft survival showing that FTY720 treatment significantly increased the survival of F1 (BALB/c x C57BL/6) skin allografts in the C57BL/6 recipients. B - The macroscopic signs of rejection were observed as early as day 7 in transplanted, non-treated (Tx) mice. C - Rejection signs occurred later in the transplanted, FTY720-treated (Tx+FTY) mice.
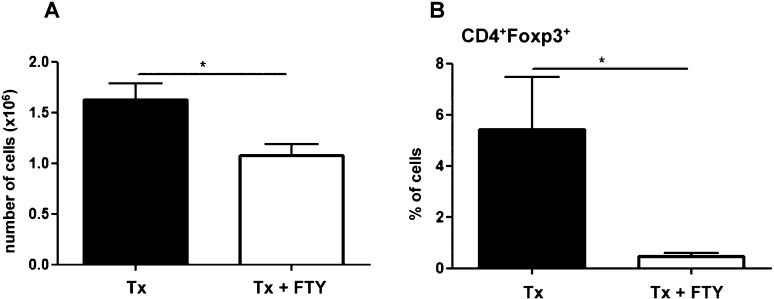
FTY720 reduced the number of cells in the lymph nodes
The total number of cells and percentage of regulatory T cells were analyzed in the lymph nodes from the Tx and Tx + FTY mice five days post-transplantation. Our results showed that the FTY720 treatment was able to significantly reduce both the number of cells and percentage of CD4+Foxp3+ T cells in the lymph nodes (Figure 2).
Figure 2.
Evaluation of the effect of transplantation and FTY720 treatment on draining lymph nodes (axillary) five days after F1 skin graft transplantation in the C57BL/6 mice. A - FTY720 treatment reduces the number of lymph node cells (p = 0.022). B - FTY720 treatment reduces the percentage of cells (p = 0.028) with the Treg phenotype (CD4+Foxp3+). The results are expressed as the means ± SD.
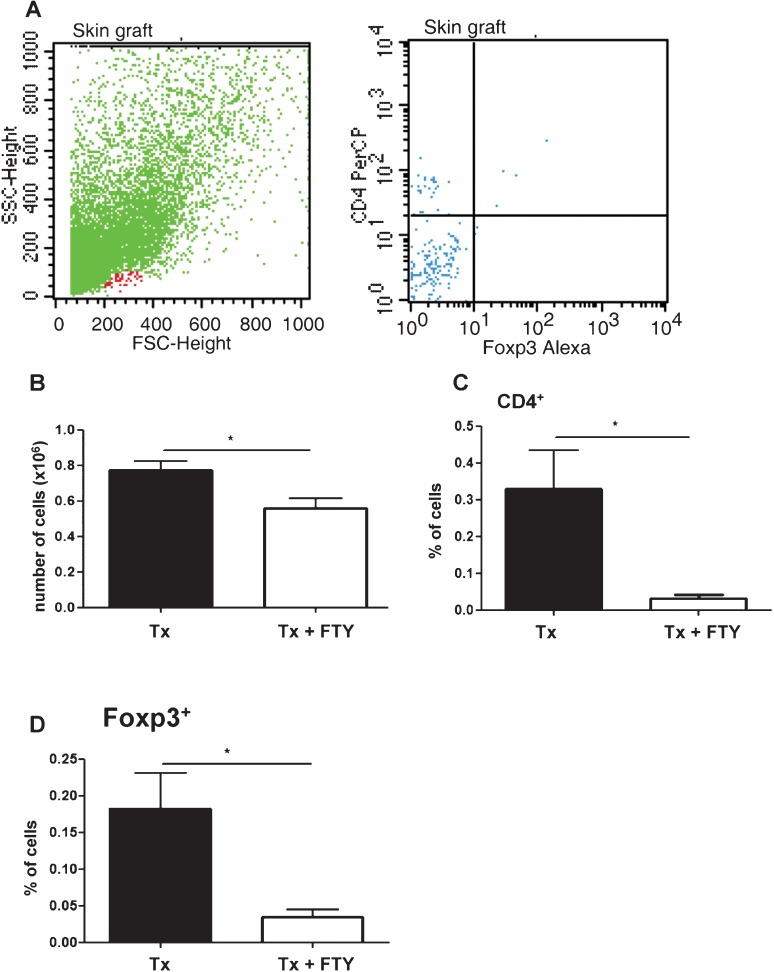
FTY720 reduced the number of graft-infiltrating cells
Five days after the skin transplantation, we analyzed the graft-infiltrating cells and observed that the FTY720 treatment reduced the number of graft-infiltrating cells and diminished the percentage of CD4+ T cells in the skin allograft. The FTY720 did not prevent the accumulation of Foxp3+ cells in the graft but did cause a decrease in the percentage of these cells (Figure 3).
Figure 3.
Evaluation of graft-infiltrating cells five days after F1 skin graft transplantation in the C57BL/6 mice. A - A representative flow cytometry dot plot showing the graft-infiltrating cells (FSC x SSC) and Treg cells (CD4+Foxp3+). B - FTY720 treatment reduces the number of graft-infiltrating cells in the F1 transplanted skin (p = 0.02). C - FTY720 treatment reduces the percentage of CD4+ (p = 0.014) graft-infiltrating cells in the F1 transplanted skin. D - FTY720 reduces but does not prevent the accumulation of Foxp3+ graft-infiltrating cells in the F1 transplanted skin (p = 0.011). The results are expressed as the means ± SD.
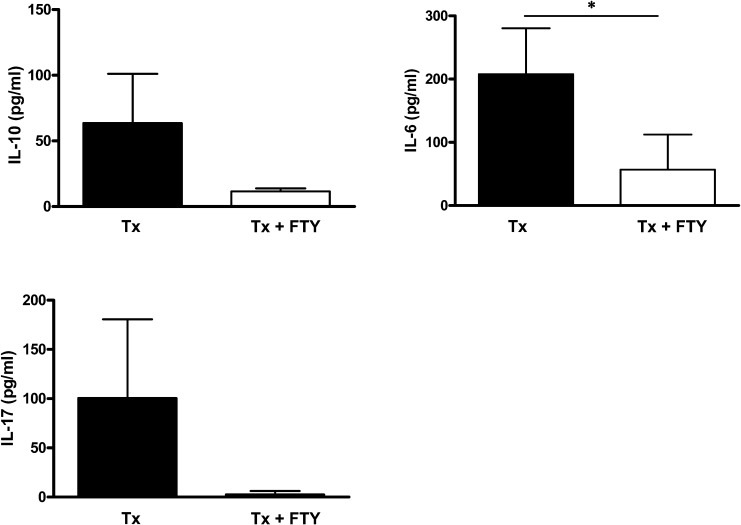
Decrease in the cytokine levels in mice treated with FTY720
We evaluated the cytokine levels in the spleen cells submitted to in vitro stimulation with anti-CD3. The analysis of the 72-h culture supernatants by Bio-Plex showed decreased levels of IL-10, IL-6 and IL-17 secretion in the FTY720-treated mice compared with the non-treated mice (Figure 4). The IL-6 secretion was significantly decreased in the Tx+FTY group (p<0.05).
Figure 4.
FTY720 decreases the cytokine production by splenocytes. Analysis of the IL-10, IL-6 and IL-17 secretion levels by the splenocytes. The spleen cells from the Tx non-treated and Tx + FTY720-treated mice were harvested at day 5 post-skin transplantation and stimulated in vitro with anti-CD3. The supernatants were collected 72 h later, and the IL-10, IL-6, and IL-17 levels were determined by multiplex ELISA. The results are expressed as the means ± SD (*p<0.05).
qPCR analysis
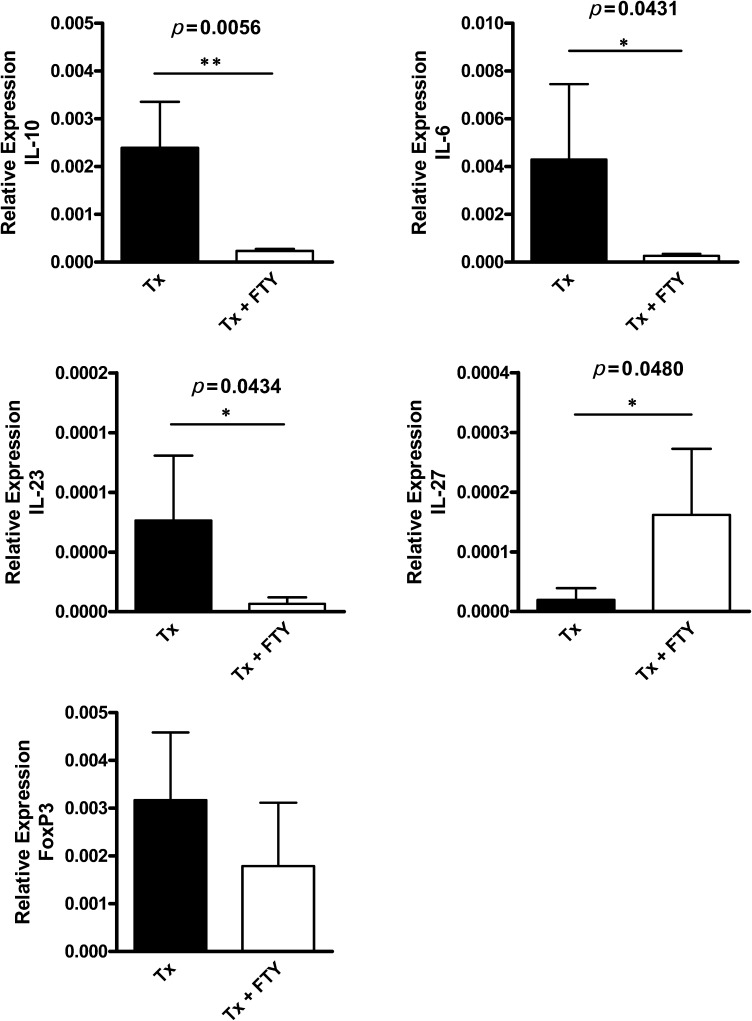
To confirm our results regarding cytokine levels, we evaluated the IL-10, IL-6, FOXP3, IL-23 and IL-27 gene expression in the spleen cells by real-time PCR. In agreement with our previous observation, there was a decrease in the IL-10 and IL-6 mRNA levels in the splenocytes of the FTY720-treated mice. In this group, we also observed a decrease in the IL-23 mRNA level but an increase in the IL-27 mRNA level. The FOXP3 expression was decreased, but this decrease did not reach statistical significance (Figure 5).
Figure 5.
FTY720-treated mice exhibit a decrease in the relative gene expression (qPCR analysis) of IL-10, IL-6, and IL-23 in the spleen cells, whereas IL-27 is increased. The relative gene expression in the spleen cells was evaluated in the Tx non-treated and Tx + FTY720-treated mice. The results are expressed as the means ± SD.
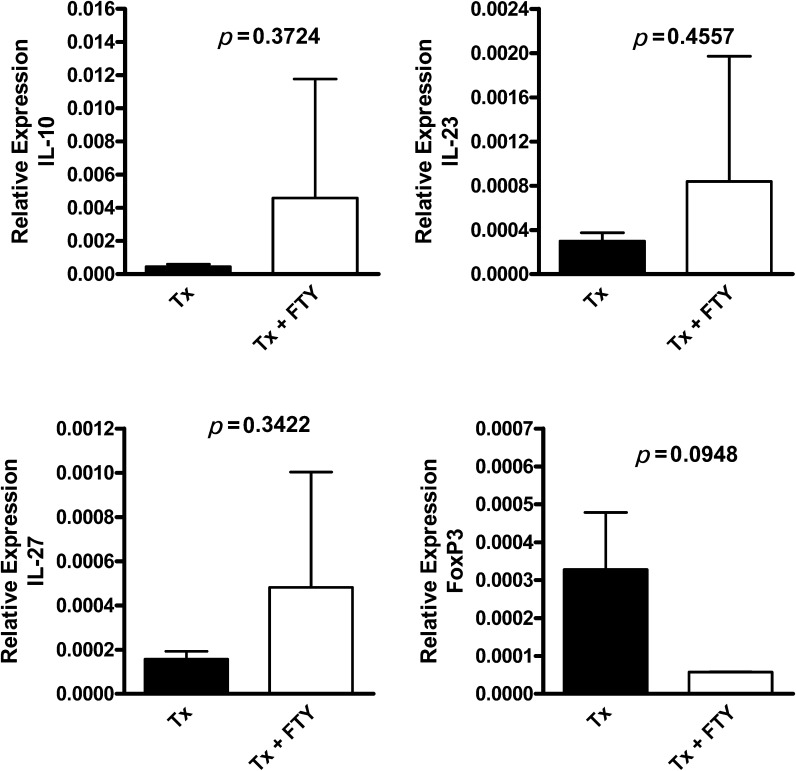
In the skin, we found increased mRNA levels of IL-10, IL-27, and IL-23 in mice treated with FTY720 (Figure 6), but this increase was not statistically significant. The FOXP3 expression was decreased in this group (p = 0.0948).
Figure 6.
FTY720-treated mice present an increase in the relative gene expression (qPCR analysis) of IL-10, IL-23, and IL-27 in the skin grafts. The relative gene expression (qPCR analysis) of FOXP3, IL-10, IL-23, and IL-27 in the F1 skin grafts was evaluated in the Tx non-treated and Tx + FTY720-treated mice. The results are expressed as the means ± SD.
DISCUSSION
In the present study, we investigated the events associated with FTY720 treatment in mice submitted to skin allograft transplantation. We confirmed the previous data showing increased skin allograft survival and delayed macroscopic signs of tissue necrosis in the mice treated with FTY720. In the same model, we showed previously that FTY720 increases skin allograft survival when administered as monotherapy, as well as when combined with cyclosporine, tacrolimus and sirolimus (22-25).
The prevention of T lymphocytes from migrating to inflammatory sites is an effect already described for FTY720. Accordingly, our present results demonstrate a reduction in the number of graft-infiltrating cells and in the percentage of CD4+ T and Foxp3+ cells.
In 2008, we demonstrated that the increased skin allograft survival in FTY720-treated mice was associated with diminished cell numbers in the spleen, blood and axillary lymph nodes (24). Our present results showed a reduction in the number of cells and percentage of CD4+Foxp3+ cells in the lymph nodes of mice treated with FTY720.
Recently, we demonstrated that FTY720 was effective in suppressing experimental autoimmune uveitis (EAU), and this finding was associated with a reduction in the eye-infiltrating cells with either Treg or Th17 phenotypes (26). In the present study, at the early time points (day +5) after transplantation, FOXP3 gene expression in the spleen and skin graft, as well as Foxp3 protein expression in the lymph nodes and graft-infiltrating cells, was decreased in mice treated with FTY720. These findings show that FTY720 does not inhibit but rather decreases the expression of Foxp3 systemically. The increase in skin allograft survival due to FTY720 without reaching indefinite tolerance could be justified by the following: the increased skin graft survival induced by FTY720 is mediated by other factors in addition to Foxp3 cells; and the decreased Foxp3 expression contributes to the rejection, even if over the long-term.
In our model, the in vitro anti-CD3 stimulation of splenocytes from the mice submitted to skin transplantation and treated with FTY720 exhibited decreased secretion of IL-10 and IL-6, which was confirmed by qPCR analysis. In addition, the splenocytes from FTY720-treated mice secreted significantly less IL-17 after in vitro anti-CD3 stimulation, and the evaluation of the mRNA in the spleen cells showed a decreased level of IL-23. Chen et al. showed an enhanced expression of Th17-related cytokines at the early stage of cornea rejection either in the draining lymph nodes (LNs) or in the allograft. They also found that responder cells in the draining LNs of the transplanted recipients secreted high levels of IL-6, TGF-β and IL-21 compared with the controls and suggested that these factors could drive naïve T cells to differentiate into Th17 cells (27). Because Th17 cells have been implicated in renal allograft rejection (28) and IL-17 production has been shown to be dependent on IL-6 (29) and IL-23 levels (30), the decreased expression of these cytokines in our model suggests that the prolonged skin allograft survival obtained by FTY720 treatment was at least in part due to Th17 phenotype impairment in the periphery rather than Foxp3 enhancement. In agreement, Mehling et al. showed a decrease in the number of Th17 cells from the blood of patients with multiple sclerosis treated with FTY720 and of patients in disease remission (31). However, whether FTY720 increases the Treg cell population is still uncertain. Wolf et al. showed in vitro and in vivo that FTY720 inhibits the proliferation of Treg cells without affecting their viability, phenotype or in vitro immunosuppression (32). Zhou et al. showed that the addition of FTY720 to the mixed lymphocyte reaction (MLR) in mouse (BALB/c and C57BL/6) splenocytes increased the proportion of CD4+CD25+ T regulatory cells. Additionally, FTY720 added to the MLR caused the suppression of lymphocyte proliferation, decreased the IL-2 levels in the supernatant, and induced lymphocyte apoptosis (33).
Allograft acceptance/rejection is associated with multiple suppressor/effector mechanisms, and immunosuppressive drugs administered during transplantation have been shown to affect T cell populations.
In agreement, Hester et al. observed, in the follow-up of kidney recipients induced with alemtuzumab and treated post-transplantation with several drugs such as sirolimus or sirolimus + mycophenolate mofetil, that rejection and the long-term maintenance of immunosuppression influence the number of circulating Treg and Th17 cells (34).
Sewgobind et al. (35) showed that the relative mRNA expression of FOXP3 decreased in the blood of kidney-transplanted patients treated with rabbit anti-thymocyte globulin (rATG) or rATG + tacrolimus, whereas the levels of IL-10 and IL-27p28 increased compared with the levels in non-transplanted blood donors.
Battaglia et al. (36) showed that the administration of IL-10 in combination with the immunosuppressive drug rapamycin promoted tolerance in type 1 diabetic mice after pancreatic islet transplantation by the induction of Tr1 cells. In addition, IL-27 secretion from dendritic cells has been shown to enhance the development of Tr1 cells and, as observed by Yoshimoto et al. (37), inhibit not only Th2 commitment but also Th2 cytokine production from already polarized Th2 cells (down-regulation of GATA-3 and up-regulation of T-bet). Moreover, IL-27 was shown by Stumhofer et al. (38) to directly antagonize the development of the Th17-cell responses in experimental autoimmune models.
In our model, even though statistical significance could not be reached among groups, the prolonged skin allograft survival promoted by FTY720 treatment could be explained by the increased expression of IL-10 and IL-27 in the skin graft, suggesting the development of the Tr1 cells. However, we also found an increase in the IL-23 expression in the skin graft, confirming the presence of Th17 cells. In agreement with our data, Li et al. (39) found both Treg and Th17 cells in mouse liver transplants. They showed a time-dependent and significant increase in Th17-related cytokines (IL-17, IL-6 and IL-23), whereas the expression of Treg-related cytokines (IL-10, TGF-β1) was markedly low in the liver of transplanted mice prior to graft rejection.
Our results show that FTY720 treatment causes a different modulation of IL-10 and IL-23 in the spleen and skin graft; namely, the IL-10 and IL-23 expression decreased in the spleen but increased in the graft. In contrast, IL-27 and Foxp3 exhibited the same modulation in the spleen and skin, with an increase in IL-27 and decrease in Foxp3. These findings suggest that, at early time points (day +5) after transplantation, FTY720 controls the balance between Tr1 (IL-10 and IL-27) and Th17 (IL-23) in the graft, which prevents rejection for a definite period of time.
In conclusion, we demonstrated that skin allograft survival was prolonged but not indefinite in FTY720-treated mice, indicating that the drug did not prevent the long-term imbalance between the Treg and Th17 cells in the graft, which thus led to rejection.
ACKNOWLEDGMENTS
This study was supported by grants from Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and CAPES.
Footnotes
No potential conflict of interest was reported.
REFERENCES
- 1.Rosenberg AS, Singer A. Evidence that the effector mechanism of skin allograft rejection is antigen-specific. Proc Natl Acad Sci USA. 1988;85(20):7739–42. doi: 10.1073/pnas.85.20.7739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lee RS, Grusby MJ, Glimcher LH, WinnHJ, Auchincloss H., Jr Indirect recognition by helper cells can induce donor-specific cytotoxic T lymphocytes in vivo. J Exp Med. 1994;179(3):865–72. doi: 10.1084/jem.179.3.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chen W, Murphy B, Waaga AM, Willett TA, Russell ME, Khoury SJ, et al. Mechanisms of indirect allorecognition in graft rejection: class II MHC allopeptide-specific T cells clones transfer delayed-type hypersensitivity responses in vivo. Transplantation. 1996;62(6):705–10. doi: 10.1097/00007890-199609270-00001. [DOI] [PubMed] [Google Scholar]
- 4.Sayegh MH, Turka LA. The role of T-cell co stimulatory activation pathways in transplant rejection. N Engl J Med. 1998;338(25):1813–21. doi: 10.1056/NEJM199806183382506. [DOI] [PubMed] [Google Scholar]
- 5.Chattopadhyay K, Bhatia S, Fiser A, Almo SC, Nathenson SG. Structural basis of inducible costimulator ligand costimulatory function: determination of the cell surface oligomeric state and functional mapping of the receptor binding site of the protein. 2006;177(6):3920–9. doi: 10.4049/jimmunol.177.6.3920. J Immunol. [DOI] [PubMed] [Google Scholar]
- 6.Itoh M, Takahashi T, Sakaguchi N, Kuniyasu Y, Shimizu J, Otsuka F, et al. Thymus and autoimmunity: production of CD25+CD4+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologic self-tolerance. J Immunol. 1999;162(9):5317–26. [PubMed] [Google Scholar]
- 7.Jee Y, Piao WH, Liu R, Bai XF, Rhodes S, Rodebaugh R, et al. CD4(+)CD25(+) regulatory T cells contribute to the therapeutic effects of glatiramer acetate in experimental autoimmune encephalomyelitis. Clin Immunol. 2007;125(1):34–42. doi: 10.1016/j.clim.2007.05.020. [DOI] [PubMed] [Google Scholar]
- 8.Walsh PT, Taylor DK, Turka LA. Tregs and transplantation tolerance. J Clin Invest. 2004;114(10):1398–403. doi: 10.1172/JCI23238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Karim M, Feng G, Wood KJ, Bushell AR. CD25+CD4+ regulatory T cells generated by exposure to a model protein antigen prevent allograft rejection: antigen-specific reactivation in vivo is critical for bystander regulation. Blood. 2005;105(12):4871–7. doi: 10.1182/blood-2004-10-3888. [DOI] [PubMed] [Google Scholar]
- 10.Mitchell P, Afzali B, Lombardi G, Lechler RI. The T helper 17-regulatory T cell axis in transplant rejection and tolerance. Curr Opin Organ Transplant. 2009;14(4):326–31. doi: 10.1097/MOT.0b013e32832ce88e. [DOI] [PubMed] [Google Scholar]
- 11.Afzali B, Lombardi G, Lechler RI, Lord GM. The role of T helper 17 (Th17) and regulatory T cells (Treg) in human organ transplantation and autoimmune disease. Clin Exp Immunol. 2007;148(1):32–46. doi: 10.1111/j.1365-2249.2007.03356.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Weaver CT, Hatton RD, Mangan PR, et al. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol. 2007;25:821–52. doi: 10.1146/annurev.immunol.25.022106.141557. [DOI] [PubMed] [Google Scholar]
- 13.de Jong E, Suddason T, Lord GM. Translational mini-reviews series on Th17 cells: Development of mouse and human T helper 17 cells. Clin Exp Immunol. 2010;159(2):148–58. doi: 10.1111/j.1365-2249.2009.04041.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ouyang W, Kolls JK, Zheng Y. The biological functions of T helper 17 cells effector cytokines in inflammation. Immunity. 2008;28(4):454–67. doi: 10.1016/j.immuni.2008.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liao JJ, Huang MC, Goetzl EJ. Cutting Edge: Alternative Signaling of Th17 Cell Development by Sphingosine 1-Phosphate. J Immunol. 2007;178(9):5425–28. doi: 10.4049/jimmunol.178.9.5425. [DOI] [PubMed] [Google Scholar]
- 16.Isoyama N, Takai K, Tsuchida M, Matsumura M, Naito K. Evidence that FTY720 induces rat thymocyte apoptosis. Transpl Immunol. 2006;15(4):265–71. doi: 10.1016/j.trim.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 17.Xie JH, Nomura N, Koprak SL, Quackenbush EJ, Forrest MJ, Rosen H. Sphingosine-1-phosphate receptor agonist impairs the efficiency of the local immune response by altering trafficking of naive and antigen-activated CD4+ T cells. J Immunol. 2003;170(7):3662–70. doi: 10.4049/jimmunol.170.7.3662. [DOI] [PubMed] [Google Scholar]
- 18.Sawicka E, Dubois G, Jarai G, Edwards M, Thomas M, Nicholls A, et al. The sphingosine1-phosphate receptor agonist FTY720 differentially affects the sequestration of CD4+CD25+ T-regulatory cells and enhances their functional activity. J Immunol. 2005;175(12):7973–80. doi: 10.4049/jimmunol.175.12.7973. [DOI] [PubMed] [Google Scholar]
- 19.Fujiwara I, Nakajima H, Matsuda T, Mizuta N, Yamagishi H, Oka T. Prolongation of concordant xenograft survival by a newly developed drug, FTY720. Transplant Proc. 1999;31(7):2831–33. doi: 10.1016/s0041-1345(99)00582-5. [DOI] [PubMed] [Google Scholar]
- 20.Wang W, Graeler MH, Goetzl EJ. Type 4 sphingosine 1-phosphate G protein coupled receptor (S1P4) transduces S1P effects on T cell proliferation and cytokine secretion without signaling migration. FASEB J. 2005;19(12):1731–33. doi: 10.1096/fj.05-3730fje. [DOI] [PubMed] [Google Scholar]
- 21.Jin J, Yang SH, Park J, Lee C, Lee J, Kim SM, Kim YS. Fragile maintenance of allograft tolerance induced by lymphocyte sequestration and co-stimulation blockade. Transpl Immunol. 2009;21(4):221–7. doi: 10.1016/j.trim.2009.06.001. [DOI] [PubMed] [Google Scholar]
- 22.Silva FR, Silva LB, Cury PM, Burdmann EA, Bueno V. FTY720 in combination with cyclosporine - an analysis of skin allograft survival and renal function. Int Immunopharmacol. 2006;6(13-14):1911–8. doi: 10.1016/j.intimp.2006.07.014. [DOI] [PubMed] [Google Scholar]
- 23.Gallo AP, Silva LB, Franco M, Burdmann EA, Bueno V. Tacrolimus in combination with FTY720 – an analysis of renal and blood parameters. Int Immunopharmacol. 2006;6(13-14):1919–24. [PubMed] [Google Scholar]
- 24.Lopes CT, Gallo AP, Palma PV, Cury PM, Bueno V. Skin allograft survival and analysis of renal parameters after FTY720 + Tacrolimus treatment in mice. Transplant Proc. 2008;40(3):856–60. doi: 10.1016/j.transproceed.2008.02.051. [DOI] [PubMed] [Google Scholar]
- 25.Lopes CT, Rosin EC, Cordeiro JA, Bueno V. Cell phenotype evaluation at various sites after skin transplantation and FTY720 plus Sirolimus therapy. Transplant Proc. 2010;42(2):573–7. doi: 10.1016/j.transproceed.2010.01.027. [DOI] [PubMed] [Google Scholar]
- 26.Commodaro AG, Peron JP, Lopes CT, Arslanian C, Belfort R, Jr, Rizzo LV, Bueno V. Evaluation of experimental autoimmune uveitis in mice treated with FTY720. Invest Ophtalmol Vis Sci. 2010;51(5):2568–74. doi: 10.1167/iovs.09-4769. [DOI] [PubMed] [Google Scholar]
- 27.Chen H, Wang W, Xie H, Xu X, Wu J, Jiang Z, Zhang M, Zhou L, Zheng S. A pathogenic role of IL- 17 at the early stage of corneal allograft rejection. Transpl Immunol. 2009;21(3):155–61. doi: 10.1016/j.trim.2009.03.006. [DOI] [PubMed] [Google Scholar]
- 28.Loong CC, Hsieh HG, Lui WY, Chen A, Lin CY. Evidence for the early involvement of interleukin 17 in human and experimental renal allograft rejection. J Pathol. 2002;197(3):322–32. doi: 10.1002/path.1117. [DOI] [PubMed] [Google Scholar]
- 29.Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B. TGF beta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity. 2006;24(2):179–89. doi: 10.1016/j.immuni.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 30.Aggarwal S, Ghilardi N, Xie MH, de Sauvage FJ, Gurney AL. Interleukin-23 promotes a distinct CD4 T cell activation state characterized by the production of interleukin-17. J Biol Chem. 2003;278(3):1910–4. doi: 10.1074/jbc.M207577200. [DOI] [PubMed] [Google Scholar]
- 31.Mehling M, Lindberg R, Raul F, Kuhle J, Hess C, Kappos L, et al. Th17 central memory T cells are reduced by FTY720 in patients with multiple sclerosis. Neurology. 2010;75(5):403–10. doi: 10.1212/WNL.0b013e3181ebdd64. [DOI] [PubMed] [Google Scholar]
- 32.Wolf AM, Eller K, Zeiser R, Dürr C, Gerlach UV, Sixt M, Markut L, Gastl G, Rosenkranz AR, Wolf D. The sphingosine 1-phosphate receptor agonist FTY720 potently inhibits regulatory T cell proliferation <italic>in vitro and in vivo. J Immunol. 2009;183(6):3751–60. doi: 10.4049/jimmunol.0901011. [DOI] [PubMed] [Google Scholar]
- 33.Zhou PJ, Wang H, Shi GH, Wang XH, Shen ZJ, Xu D. Immunomodulatory drug FTY720 induces regulatory CD4(+)CD25(+) T cells <italic>in vitro. Clin Exp Immunol. 2009;157(1):40–7. doi: 10.1111/j.1365-2249.2009.03942.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hester J, Mills N, Shankar S, Carvalho-Gaspar M, Friend P, Wood KJ. Th17 cells in Alemtuzumab-treated patients: the effect of long-term maintenance immunosuppressive therapy. Transplantation. 2011;91(7):744–50. doi: 10.1097/TP.0b013e31820c85dc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sewgobind VDKD, van der Laan LJW, Kho MML, Kraaijeveld R, Korevaar SS, Mol W, Weimar W, Baam CC. The calcineurin inhibitor tacrolimus allows the induction of functional CD4+CD25+ regulatory T cells by rabbit anti-thymocyte globulins. Clin Exp Immunol. 2010;161(2):364–77. doi: 10.1111/j.1365-2249.2010.04183.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Battaglia M, Stabilini A, Draghici E, Gregori S, Mocchetti C, Bonifacio E. Rapamycin and interleukin-10 treatment induces T regulatory type 1 cells that mediate antigen-specific transplantation tolerance. Diabetes. 2006;55(1):40–9. [PubMed] [Google Scholar]
- 37.Yoshimoto T, Yoshimoto T, Yasuda K. IL-27 suppresses Th2 cell development and Th2 cytokines production from polarized Th2 cells: a novel therapeutic way for Th2-mediated allergic inflammation. J Immunol. 2007;179(7):4415–23. doi: 10.4049/jimmunol.179.7.4415. [DOI] [PubMed] [Google Scholar]
- 38.Stumhofer JS, Laurence A, Wilson EH, Huang E, Tato CM, Johnson LM, et al. Interleukin 27 negatively regulates the development of interleukin 17-producing T helper cells during chronic inflammation of the central nervous system. Nat Immunol. 2006;7(9):937–45. doi: 10.1038/ni1376. [DOI] [PubMed] [Google Scholar]
- 39.Li J, Lai X, Liao W, He Y, Liu Y, Gong J. The dynamic changes of Th17/Treg cytokines in rat liver transplant rejection and tolerance. Int Immunopharmacol. 2011;11(8):962–7. doi: 10.1016/j.intimp.2011.02.010. [DOI] [PubMed] [Google Scholar]