Abstract
A novel hypothesis of obesity is suggested by consideration of diet-related inflammation and evolutionary medicine. The obese homeostatically guard their elevated weight. In rodent models of high-fat diet-induced obesity, leptin resistance is seen initially at vagal afferents, blunting the actions of satiety mediators, then centrally, with gastrointestinal bacterial-triggered SOCS3 signaling implicated. In humans, dietary fat and fructose elevate systemic lipopolysaccharide, while dietary glucose also strongly activates SOCS3 signaling. Crucially however, in humans, low-carbohydrate diets spontaneously decrease weight in a way that low-fat diets do not. Furthermore, nutrition transition patterns and the health of those still eating diverse ancestral diets with abundant food suggest that neither glycemic index, altered fat, nor carbohydrate intake can be intrinsic causes of obesity, and that human energy homeostasis functions well without Westernized foods containing flours, sugar, and refined fats. Due to being made up of cells, virtually all “ancestral foods” have markedly lower carbohydrate densities than flour- and sugar-containing foods, a property quite independent of glycemic index. Thus the “forgotten organ” of the gastrointestinal microbiota is a prime candidate to be influenced by evolutionarily unprecedented postprandial luminal carbohydrate concentrations. The present hypothesis suggests that in parallel with the bacterial effects of sugars on dental and periodontal health, acellular flours, sugars, and processed foods produce an inflammatory microbiota via the upper gastrointestinal tract, with fat able to effect a “double hit” by increasing systemic absorption of lipopolysaccharide. This model is consistent with a broad spectrum of reported dietary phenomena. A diet of grain-free whole foods with carbohydrate from cellular tubers, leaves, and fruits may produce a gastrointestinal microbiota consistent with our evolutionary condition, potentially explaining the exceptional macronutrient-independent metabolic health of non-Westernized populations, and the apparent efficacy of the modern “Paleolithic” diet on satiety and metabolism.
Keywords: carbohydrate density, metabolic syndrome, nutrition transition, Paleolithic diet
Introduction
Due to the complexity of a phenomenon like obesity, it is inevitable that most study assumes a “bottom-up” approach, working to elucidate detailed knowledge of the systems believed to be involved. During the assembly of such detailed knowledge into a working whole, a measure of conformity to prevailing views is natural, as researchers work in detail on their part of the puzzle and seek to integrate that work into the network of existing knowledge. Bottom-up approaches do not lend themselves to rapid paradigm shifts in understanding, as each novel contribution is an incremental advancement. This is especially true in research into the dietary causes of obesity and Western diseases, where implicating the wrong cause and advocating widespread dietary changes on the basis of that could lead to a significant worsening of the epidemiological situation.
With obesity and diabetes incidence and severity now at truly disconcerting rates, it is time to consider the possibility that implicating dietary fat as the primary cause and advocating that it be minimized has been a fundamental error, despite the large body of evidence supporting mechanisms by which high-fat diets might produce obesity.
The present paper attempts a “top-down” approach to obesity and provides a hypothesis that is not only consistent with available detailed mechanisms but also with some of the most confusing widely observed dietary phenomena. By comparing “ancestral diets,” which appear to promote universally effective homeostatic energy balance, with Western diets, a sharp delineation is found in the proportional mass of carbohydrate found in Western foods versus the locked-in low carbohydrate density of cellular plant foods. Since this characteristic does not correlate with glycemic index, the primary “organ” likely to be directly affected by such luminal concentration changes, and one already implicated in leptin resistance and obesity, is the gastrointestinal (GI) microbiota.
Background to the hypothesis
Energy homeostasis in the absence of Western foods
Obesity and non-insulin dependent diabetes are growing global phenomena, extending into the developing world following the spread of Westernized dietary patterns.1 The tendency toward overeating in the modern world is so common that it has led to the widely held belief that human energy homeostasis is maladapted to food excess and a sedentary lifestyle,2 and that constant voluntary caloric restriction is important to maintain healthy weight. Most epidemiological overviews of obesity examine Western populations, and it is often overlooked that even nominally healthy Westerners have leptin levels many times higher than those eating a non–Westernized diet, even in the presence of food abundance. For example, the Kitavan Islanders of Melanesia live as horticulturists with levels of activity comparable to manual workers who take moderate exercise; their dietary staples are root vegetables and fruit, with some consumption of meat and fish, but with little availability of Western foods. Hence as a population they consume virtually no grains or refined foods such as flour, sugar, or refined oils.3,4 Their carbohydrate intake is high, in the order of 60%–70% of energy intake, much of it root tubers or fruit with a moderately high glycemic index,5 while their saturated fat intake is also high (17% – largely from coconut). Despite food abundance and a clear overlap of macronutrients and glycemic index with Western diets, Kitavans are reported to possess leptin levels, fasting insulin, and blood glucose levels dramatically lower than those in Western populations deemed healthy,6,7 and appear to have a virtual absence of overweight, diabetes, and atherosclerotic disease.3,4,8,9 Environmental or genetic explanations for this metabolic health appear unlikely, since islanders who leave for the mainland and eat Western foods become overweight.7 People of this region may in fact be more susceptible to the effects of Western diet than Europeans, which may include increased susceptibility to effects subsequent to leptin resistance.10,11 Similar leanness and low leptin concentrations have also been recorded in the Ache hunter-gatherers of Paraguay, and in Shuar people of the Amazon who retain a traditional lifestyle.12–14 Those Shuar who have taken the initial steps toward agriculture have significant increases in leptin levels.15 Although leptin levels have not been recorded for other people eating traditional diets, it has been widely reported that unless grains or refined foods had arrived, all bore remarkably low incidences of “Western diseases,” including obesity (Table 1).3,4,6–9,16–29 Also notable is the apparent macronutrient-independence of the health associated with ancestral diets. The Kitavan case appears to argue against a primary causal role for carbohydrates or glycemic index in the genesis of obesity, at least when carbohydrates are in the form of root vegetables or fruit. Their diet is higher in carbohydrates than the estimated intake for our East African ancestors30 or modern hunter-gatherers,31,32 who tend to have carbohydrates provide around 35% of energy, ranging from 3% to 50% of energy intake, the proportion falling with northern latitude. Similarly, other analyses of modern hunter-gatherers33 have suggested that intake of unprocessed meat does not produce poor metabolic or cardiovascular health, in agreement with the reported historical diets and health of the Masai, Kavirondo, and Turkhana (Table 1).
Table 1.
Nutrition transition overview: obesity and “Western foods”
| People | Diet | Macronutrient summary | Acellular foods or grains | Overweight or obesity | Other health markers | Reference |
|---|---|---|---|---|---|---|
| Kitava 1990s | Starchy root vegetables, fruit, some fish and meat, coconuts. Food abundance | ~65% carbohydrate, high fibre, 17% saturated fat | − | 0% overweight | No stroke, diabetes or heart disease. No increase in weight or blood pressure in middle age | 3,4,6–9 |
| Machiguenga 1982 | Mostly root vegetables, fruit and nuts, very small amount of meat and fish. Food abundance | High carbohydrate, high fibre | − | None reported | Healthy teeth and gums, good general health, except for parasites and infections | 22 |
| Masai 1971 | Cow’s milk and blood, meat | 66% fat, similar cholesterol intake to US | − | Lean | Atherosclerosis rare at autopsy | 16 |
| Kavirondo Kenyans 1929 | Mostly meat | High protein, high fat | − | Lean | Aortic atheroma at autopsy rarer than in Westerners. No increase in blood pressure through middle age | 20 |
| !Kung 1972 | ~70% plant matter (mongongo nuts, fruits, roots, bulbs, leaves), some meat | Mongongo nuts: ~60% fat, 25% protein | − | Lean | No increase in blood pressure through middle age | 29 |
| Solomon Islanders 1974 | Root vegetables, fruit, fish, limited meat, tinned fish | Varied | −/+ | Lean (Western-influenced islands were heavier) | No hypertension or cardiovascular disease. Islands with Western influence showed increased blood pressure | 26 |
| Turkana 1980–83 | Mostly milk, blood, meat, but by 1983 also some traded sorghum, maize, millet, and sugar | High protein, high fat | + | Lean (<5th percentile of Western weight) | No increase in weight in middle age | 24,25 |
| Tarahumara 1978–79 | 90% from pinto beans and maize tortilla. Remainder from squash and other vegetables | High carbohydrate, high fibre, fat 12%, animal sources 6%, refined sugar 1% | ++ | 5% BMI > 26 | No hypertension, no rise in blood pressure with age | 17,19 |
| Mexican Pima 2006 | Beans, wheat-flour tortillas, corn tortillas, and potatoes | 62% carbohydrate, 25% fat, high fibre | +++ | Males: 7% obese Females: 20% obese |
7% diabetes incidence | 27 |
| Arizona Pima 1996–2006 | Refined Western diet, including fried breakfasts, processed meats, hamburgers, pork chops, beans, white bread, flour tortillas, fried or baked dough, cereals, canned foods, fruit juices | 49% carbohydrate, 15% protein, 34% fat, >10% saturated fat | ++++ | Males: 64% obese Females: 75% obese |
>30% diabetes | 27,28 |
Note: −, −/+, +,++, +++ and ++++: Approximate contribution of acellular foods or grains to diet, ranked from ‘absent’ (−), to ‘major component of diet’ (++++).
Those populations that transitioned to a Westernized diet invariably developed Western metabolic diseases,1,34 while sustaining high levels of physical activity appears to offer only a degree of protection against obesity,35,36 as would be expected if a perturbation of homeostatic set point were responsible. It is unlikely that environmental factors could effect an absence of overweight in non-Westernized populations by means of effectively enforcing caloric-restriction even in good times, and such an improbable constant food-shortage or increased physical exertion would not be sufficient explanation for the dramatically different leptin levels seen in hunter-gatherers and non-cereal horticulturists, which in the case of the Ache of Paraguay are significantly lower than Western distance runners despite the lower adiposity of the latter.13
Moreover, even small amounts of Western influence in diet are associated with significant adverse alterations in metabolic and physiological markers, often before any substantial lifestyle changes.15,26 The relationship between health and ancestral or Western diets is reviewed in detail by Lindeberg.37 The effects of Western diet and lifestyle do appear to be reversible, at least in part. The metabolic health of diabetic Australian Aborigines has been shown to be markedly improved by even a 7-week return to a hunter-gatherer lifestyle and diet.38 In addition, Westerners have also been shown to benefit from similar dietary changes.
Grain-free whole-food diet: early results in Westerners
Whole grains are mooted to be healthier than refined grains,39 yet comparisons between grain consumption habits in industrialized societies indicate the effects of replacing refined grains with whole grains yield only modest improvements to health.40 These studies inevitably compare differing patterns of grain consumption against a background of almost universal flour, sugar, refined starches, and oils in the modern diet. Health patterns during nutrition transitions indicate that obesity, diabetes, and cardiovascular disease have characteristic incubation periods, but were virtually absent prior to the arrival of Western foods.18,21,37 The interindividual variability in “Western disease” incidence in a population eating Western foods is affected by genetic and other factors, but it is mostly these secondary factors that the epidemiology of industrialized populations would be studying if the root causes of metabolic disorders were now present throughout the population.
Removal of grains and all refined foods is one of the hallmarks of the “Paleolithic” diet, a modern way of eating that attempts to approximate the characteristics of ancestral diets. Although the literature of clinical studies of this dietary pattern in Westerners is currently small, it is also unanimous. Each published experimental comparison of a diet containing grains with one excluding grains has found significant favorable metabolic effects in the grain-restricted groups, with beneficial effects large enough to render the studies adequately powered despite their small test groups. The randomized clinical trials have shown significantly greater reductions in weight and waist circumference in an ad libitum Paleolithic-style diet compared with the consensus “Mediterranean” or “Diabetes” diets41,42 and significant improvements over the Mediterranean diet in blood glucose control, independently of the superior waist-circumference reduction.42 All three diets emphasize whole foods, but the restriction of grains in the Paleolithic diet is a principal difference, which correlated well with the reduced waist measurement42 and the 20%–30% increased satiety per calorie seen in the Paleolithic-diet groups. Importantly this increased satiety did not correlate with energy density, fiber, or macronutrient content,43 and significant spontaneous decreases in energy intake have been reported in all the ad libitum studies to date.41,42,44 However, the benefits of this dietary pattern do not appear dependent on this altered satiety or weight change; consumed in a calorie-matched manner to prevent weight loss, a Paleolithic-style diet produced significantly greater improvements in blood pressure, glucose tolerance, insulin sensitivity, and lipid profiles in a small group of healthy volunteers, with each individual participant showing improvements, indicating that these metabolic improvements occur independently of reduced caloric intake.45 Both the spontaneous 20%–30% decrease in caloric intake and the other metabolic improvements produced by a Paleolithic style of eating would be consistent with an increase in leptin sensitivity. Leptin levels dropped by 31% after 12 weeks of ad libitum Paleolithic diet, and were found to correlate best with consumption of cereals excluding rice.43 It is not known whether continued adherence to the regimen would produce leptin levels comparable to those reported in hunter-gatherers and noncereal horticulturists.
On the balance of evidence from the dietary patterns of non–Western populations and the initial findings of the modern Paleolithic diet, grains and refined foods therefore stand implicated as potential dietary causes of the Western obesity problem. It has been hypothesized that insulin receptor, epidermal growth factor receptor, or interleukin-2 receptor activities of cereal-grain lectins may be responsible for producing leptin resistance;46 the present paper proposes an alternative or additional mechanism with broad applicability to almost all Western foods.
Food-related inflammation and obesity
It is increasingly recognized that obesity is a disorder characterized by systemic low-level inflammation,47 with multiple elements of the metabolic syndrome strongly correlating with circulating bacterial lipopolysaccharide (LPS) concentrations.48 LPS levels are also significantly higher in nonalcoholic fatty-liver patients than controls, in conjunction with elevated GI permeability,49,50 while in metabolic syndrome, elevated downstream expression and activation of TLR4 and TLR2 on circulating monocytes is seen.51 After successful Roux-en-Y gastric bypass surgery, the reductions in weight and insulin resistance are associated with reductions in circulating LPS and inflammation.52 The source of the LPS is thought to be the GI tract, with ingestion of high-fat and high-carbohydrate Western-style meals found to produce postprandial “metabolic endotoxemia”: an increase in circulating LPS levels and other inflammatory changes.53 LPS and other gastrointestinally derived pathogen-associated molecular patterns (PAMPs) have also been suggested to play a role in the etiology of many “Western diseases,” including obesity. The small intestine has been implicated as a primary source for their systemic absorption, with possible PAMP sources including swallowed oral bacteria, small intestinal commensals, or PAMPs harbored inside foodstuffs. The far larger populations of Gram-negative bacteria in the large intestine are suggested to be less likely to act as a source for systemic PAMPs, due to the relatively small surface area of the bowel compared to the small intestine, the presumed evolutionary adaptations of the bowel to high concentrations of bacteria, and the fact that colonic translocatory PAMPs would undergo clearance by the liver, rather than bypassing hepatic detoxification via the lymphatic system, as small intestinal PAMPs would (reviewed by Erridge).54 The precise GI location of the alterations in microbiota underlying metabolic endotoxemia and inflammation in diet-induced obesity is not fully understood. The present hypothesis makes suggestions regarding the dietary triggers of these changes, wherever they are ultimately found to be located.
Dietary fat also appears able to facilitate translocation of PAMPs from the gut lumen into the circulation by promoting a microbiota that reduces the expression of tight-junction proteins.55 In humans, test drinks of cream (70% fat) produce a postmeal spike in circulating LPS, while a glucose drink does not. However, suppressor of cytokine signalling-3 (SOCS3) protein signaling and nuclear factor kappa B (NFκB) activation in circulating mononuclear cells were markedly elevated by both cream and glucose drinks, and were still elevated at 5 hours.56 The finding that glucose consumption elevates NFκB and inflammatory pathways57 without producing changes in LPS or TLR4 levels is important evidence that a glucose-induced non-LPS/TLR4 inflammatory pathway exists. SOCS3 has been implicated in leptin and insulin resistance, and both it and NFκB are significantly elevated in the obese.58 In animal studies, when conventional mice are compared with germ-free mice over 12 weeks, a saturated fat-free diet of ordinary rodent chow interacts with the microbiota of conventional mice to produce elevated LPS, increased white adipose tissue mass, increased levels of leptin, blood glucose and insulin, and increased infiltration of white adipose tissue by macrophages.59 In the same study, germ-free mice were monocolonized with either a wild-type Escherichia coli or a mutant form with less immunogenic penta-acylated LPS. The less immunogenic bacterium produced lower LPS levels in the portal vein, and was associated with lower macrophage infiltration and inflammation of white adipose tissue, indicating a role for LPS in this effect. However, leptin and insulin levels were even higher in mice monocolonized with the mutant E. coli, suggesting non-LPS PAMPs play a considerable role in leptin and insulin resistance.
Differing profiles of inflammation and PAMP translocation appear to be produced by different monosaccharides. In mice whose chow diet was supplemented with 30% glucose solution, larger weight gain and visceral adiposity were seen, but with no significant changes in hepatic portal LPS concentration. Conversely, fructose produced smaller changes in adiposity but markedly worse markers of hepatic inflammation, and significantly elevated hepatic portal LPS.60 Fructose is thought to have direct toxic effects on the liver,61,62 and its ability to generate circulatory LPS may be an important contributor to non-alcoholic fatty liver disease.
Inflammation of the gut itself plays a potential role, with early GI inflammation preceding and predicting obesity in diet-induced obesity models.63 This inflammation is associated with increased gastrointestinal permeability and changes in microbiota.64 Leptin resistance of the vagal afferent neurons innervating the upper GI tract is seen in diet-induced obese mice, apparently via SOCS3 signaling to inhibit STAT3 activation by leptin, with LPS shown to be able to increase SOCS3 in isolated nodose neurons (the cell bodies of the vagal afferents) in vitro.65,66 The onset of increased food intake in diet-induced obese animals coincides with this onset of leptin resistance, and substantially reduces the role of leptin in maintaining vagal afferent sensitivity to satiety mediators including cholecystokinin and peptide YY, by reducing the functional expression of their receptors, and increasing the expression of appetite-increasing receptors such as melanin-concentrating hormone 1 receptor and cannabinoid receptors.66 In similar diet-induced obesity models, central leptin resistance occurs some time later,67 with SOCS3 signaling again implicated.68 Modulation of the gastrointestinal microbiota in diet-induced obese and Ob/Ob mice with antibiotics is able to reduce the levels of LPS, inflammatory markers, and oxidative stress, along with reduced glucose intolerance, visceral fat, and body-weight gain.55,69 TLR4 and CD14 knockout mice are also protected from these effects, interestingly with subtly different effects on adipose tissue macrophage infiltration, indicating differing roles for these receptors in mediating LPS effects.70–72
The capacity for dysregulation of insulin, blood glucose, and other metabolic markers as a result of intestinal microbiotal changes may be highly conserved, with altered microbiota strongly affecting metabolism and growth patterns in the fruit fly Drosophila.73 In addition, microbiotal alterations appear able to influence the severity of myocardial infarction in a rodent model in a leptin-dependent manner.74 In humans, the species of bacteria that are present in fecal microbial communities appear to be relatively varied between individuals, with greater similarities between closely related people. However, the bacterial genes present across human populations appear to be more conserved, hence we possess a “core microbiome” composed more of similar bacterial genes rather than similar bacterial species. The microbiota of obese individuals showed alterations in the expression of bacterial genes, many involved in metabolism of carbohydrates, lipids, and amino acids.75 There is evidence that less Westernized diets can produce microbial communities with marked differences from those seen in industrialized countries. Children from rural Burkina Faso have a considerably different microbiota to those from Europe, a difference thought to be primarily dietarily mediated. With a lifestyle comparable to early Neolithic subsistence farmers, these children carry a large representation of Bacteroidetes, with an unusual prevalence of species suggested to bear genes for hydrolysis of dietary fiber.76
It is also known that the GI microbiota is also able to make use of fiber to affect the efficiency of energy absorption from the gut, which has been the subject of much study (reviewed by Backhed).77 By fermenting fiber into absorbable short-chain fatty acids, colonic bacteria are able to provide energy from fiber to the host that would otherwise not be available, and affect signaling via GPR41 and GPR43 receptors. Despite the trend toward reduced fiber intake in Western diets, the relevance of fermentable fiber mechanisms to obesity is questionable. The addition of supplementary fiber to Western diets has been studied extensively, without any improvements of the order of magnitude required for fiber to represent a significant part of the causal difference between ancestral and modern diets.78 While fermentable fiber is able to effect certain changes in colonic gastrointestinal microbiota, these currently seem unlikely to represent a useful intervention for obesity or metabolic disease.
Dietary fat is an exacerbatory but not primary cause of human obesity
There is no question that fat has a role in obesity. The rodent diet-induced obesity models discussed above predominantly make use of high-fat chows to produce their effects, which for a proportion of dietary fat of 72% (lard and corn oil) produced around a 2.7-fold increase in circulating LPS, while 40% fat chows produced 1.4-fold changes in LPS.79 It has also been shown that diets of differing fatty acids are able to differentially produce endotoxemia and complex patterns of inflammatory processes.80 Saturated fatty acids themselves are also able to activate the LPS receptor TLR4; this appears to require concentrations in the high micromolar range.81–83 These data appear consistent with the dominant body of dietary belief since the late 1970s that overconsumption of dietary fats is one of the primary nutritional problems of the industrialized world. However, despite all the mechanistic evidence that has been amassed condemning dietary fat, two broader considerations nonetheless suggest fat is unlikely to be the most pertinent factor in human obesity and metabolic syndrome.
Firstly, if dietary fat were the primary determinant of leptin resistance in humans, one would expect a low-fat diet to produce meaningful spontaneous reductions in weight in the obese. Reduced dietary fat intake would substantially reduce vagal and central leptin resistance, correcting the appetite and energy-expenditure “set point,” increasing the effectiveness of satiety signals, reducing appetite, and correcting metabolic markers. However, instead it is low-carbohydrate diets that are reported to produce the greatest weight loss in ad libitum diets, while ad libitum low-fat diets perform less well and produce less improvement to metabolic markers.84–88 Low-fat diets work as effectively as low-carbohydrate diets only when caloric restriction is enforced in both regimens.89,90 A recent small study of a “prudent” reduced-fat diet produced a 38% reduction in endotoxin activity.91 However, despite having a similar sample size to some Paleolithic-diet studies, there were no significant differences in blood glucose or insulin levels between the “prudent” diet and an elevated-fat diet that increased circulating endotoxin activity by 71%, consistent with the modest metabolic improvements from reducing fat.86,88
The second consideration against dietary fat is the evidence of ancestral diets where some populations historically ate high levels of meat and/or fat, and some ate high levels of carbohydrates, but no overweight or poor metabolic health markers were reported (see Table 1). Similarly, modern hunter-gatherers do not have a gradient of metabolic health or overweight outcomes based on either fat or carbohydrate consumption,32,33 as would be expected to be manifest if simple increased proportions of one of these macronutrients were the primary causes of obesity.
These observations are consistent with some factor present in Westernized foods being responsible for obesity, with the macronutrient ratios of unprocessed foods having no obesogenic effects.
Background summary
Elevating dietary fat in a Western diet can cause obesity. However, the effects of low-carbohydrate diets and the failure of low-fat diets to match their ad libitum weight loss and metabolic effects indicate that carbohydrates somehow play a central role as initiators of leptin resistance in humans. However, the Kitavan Islanders, one of the metabolically healthiest populations yet studied, eat a high-glycemic-index, high-carbohydrate diet with staples of starchy tubers and fruits. A unified theory of obesity should (somehow) be consistent with all these observations.
The hypothesis
Carbohydrate density and diet-related inflammation
To find the cause of the modern obesity epidemic, the properties of modern foods that perturb the homeostatic energy set point should be sought. The universality of the negative effects of Western foods on indigenous populations indicates that these properties must be similarly widespread in modern foods.
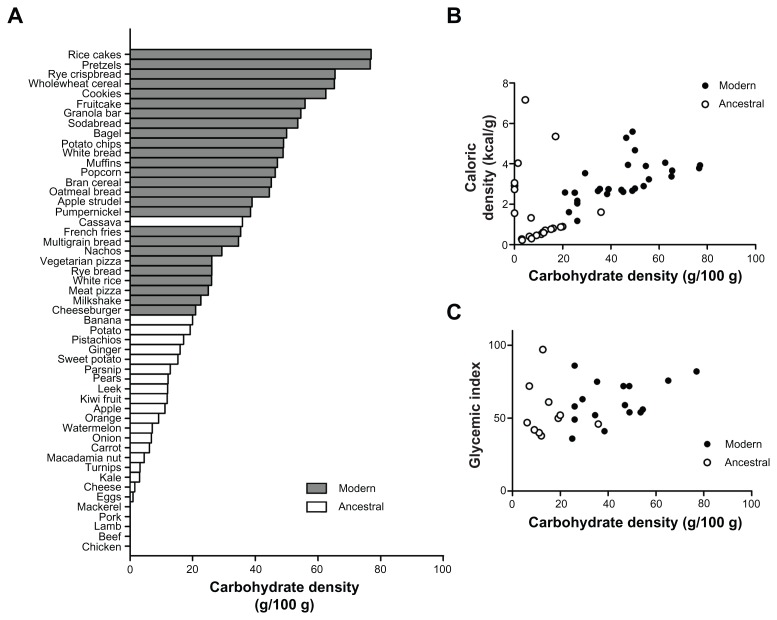
Among the most common elements of Westernized foods are the nonperishable products flour and sugar. A fundamental distinction between modern foods containing these and ancestral foods is carbohydrate density. Figure 1A shows the carbohydrate density of a broad selection of foods, with modern foods in grey (USDA data). Foods that would be permitted on a Paleolithic or “primal” diet – the “ancestral foods” – are those in the categories of root tubers, leafy vegetables, fruit, nuts, meats, eggs, and fish, and are shown in white. Tubers, fruits, or functional plant parts such as leaves and stems store their carbohydrates in organelles as part of fiber-walled living cells. These are thought to remain largely intact during cooking, which instead mostly breaks cell-to-cell adhesion.92,93 This cellular storage appears to mandate a maximum density of around 23% non-fibrous carbohydrate by mass, the bulk of the cellular weight being made up of water. The acellular carbohydrates of flour,94 sugar and processed plant-starch products are considerably more dense. Grains themselves are also highly dense, dry stores of starch designed for rapid macroscopic enzymic mobilization during germination.95 Whereas foods with living cells will have their low carbohydrate density “locked in” until their cell walls are breached by digestive processes, the chyme produced after consumption of acellular flour and sugar-based foods is thus suggested to have a higher carbohydrate concentration than almost anything the microbiota of the upper GI tract from mouth to small bowel would have encountered during our coevolution. This may stimulate differing bacterial species to prosper or be outcompeted, or increase some microbial metabolic pathways and waste products in preference to others. It is proposed that the effects of these enhanced carbohydrate concentrations will include a more inflammatory GI microbiota, initially causing leptin resistance, hence the greatly elevated leptin levels seen in Western populations when compared to those eating a wholly cellular diet.7,12–15
Figure 1.
The carbohydrate densities of ancestral foods are distinctly lower than those of the Westernized diet. (A) The carbohydrate density (excluding fiber) of a broad selection of foods, in descending order of carbohydrate density (data from USDA).112 Modern foods (gray bars) are those that have undergone refinement or desiccation, or are derived from grains. “Ancestral” foods (white bars) are unprocessed whole-foods from the categories of meats, eggs, fish, nuts, fruits, tubers, and leafy vegetables. (B) Carbohydrate density and caloric density of modern and ancestral foods. Some ancestral foods have caloric densities as high as modern foods, notably meats and nuts. (C) Carbohydrate density and glycemic index of modern and ancestral foods. Once again, there is no distinction between the two categories of food, and no correlation between the density of a carbohydrate and the nature of the blood glucose response it will elicit.
Figure 1B shows that the nutrient densities of ancestral foods have a good degree of overlap with those of modern food, with nuts, meats, and eggs having higher nutrient densities, but still retaining low carbohydrate density. Figure 1C shows a similar overlap of glycemic index between ancestral and modern foods for which data were available,5 with little correlation between carbohydrate density and glycemic index. Hence ancestral diets are strongly distinct from modern diets with regard to carbohydrate density, but not by either nutrient density or glycemic index.
Once an inflammatory microbiota is in place, consumption of refined dietary fats and oils may effect a double hit by increasing the absorption of inflammatory PAMPs including LPS into the circulation (Figure 2), or stabilizing and preserving the inflammatory microbiota itself. This would make a diet rich in both acellular carbohydrates and fat highly obesogenic, promoting a self-sustaining cycle of hyperphagia in an environment where such foods are abundant. Upon withdrawal of all acellular carbohydrates, the GI microbiota is proposed to return to a less inflammatory form consistent with our evolutionary experience, and one much more resistant to the exacerbatory effects of dietary fats, consistent with the interplay between fructose and fats seen in rats.96 From the sensitivity of ancestral peoples’ health markers to even small influences of Western foods,15,26 there may be a nonlinear relationship between the degree of diet-related inflammation and the amount of acellular carbohydrate in diet, with even small amounts of sugar or flour able to produce significant leptin resistance. This is also likely influenced by the concentration and nature of dietary fats present and heritable factors.
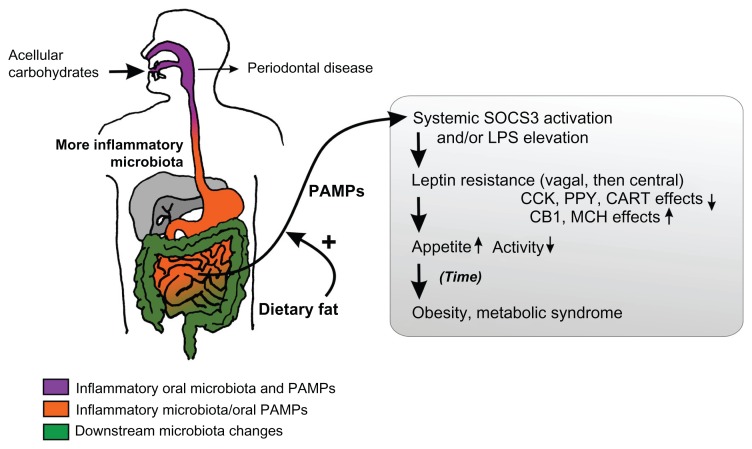
Figure 2.
Schematic of the hypothesis.
Notes: The acellular dense carbohydrates of modern foods are proposed to produce an inflammatory microbiota from the mouth onwards, initially producing periodontal disease. The small bowel is exposed to lipopolysaccharide (LPS) and other pathogen-associated molecular patterns (PAMPs) from the oral microbiota, and proinflammatory modulation of its own small populations of bacteria by concentrated acellular carbohydrates. With systemic absorption enhanced by dietary fat, the inflammatory bacterial compounds induce leptin resistance and hyperphagia. The contents of the gray box represent the existing understanding of the effects of diet-induced obesity on energy homeostasis.
Abbreviations: CCK, cholecystokinin; PPY, peptide YY; CART, cocaine and amphetamine related transcript; CB1, cannabinoid receptor type 1; MCH, melanin concentrating hormone.
In people of European descent, relatively high resistance to the diabesogenic effects of Western diet is seen.1,34 This is proposed to be due to thousands of years of exposure to the acellular carbohydrates from cereal agriculture, allowing limited adaptive mechanisms to evolve. People of non-European descent hence appear more susceptible to the effects of flour, sugar, and refinement, develop worse adipokine profiles from Western diets,10 and show higher incidences of obesity and diabetes. In Europeans, overt obesity or diabetes is minimal unless consumption of bread is compounded by replacement of remaining cellular carbohydrate sources (fruit and vegetables) with sugar, flour, and refined oil-bearing processed foods.
Archaeological evidence from Paleolithic African and European tools indicates that some groups of people did supplement their diet with ground plant starches from roots or gathered wild grass seeds.97–99 The pestle-and-mortar scale of this food preparation would likely have made the contribution of acellular carbohydrates to diet an order of magnitude smaller than that seen in the modern diet, where for many the overwhelming majority of plant foods consumed year-round are acellular processed products. Hence it is argued that any evolutionary pressures evoked by prior hand preparations of wild seeds was dwarfed by the advent of cereal agriculture, which in turn provided only a partial evolutionary preparation for the modern Western diet.
Dietary carbohydrate-density: a link between periodontal health and metabolic health?
When indigenous people adopted flours and sugars into their diets, along with the adverse metabolic effects they experienced a substantial downturn in their dental health.100,101 It is widely accepted that consumption of refined carbohydrates can cause tooth decay and gingival inflammation through bacterial means. While an inflammatory oral microbiota may alter oral inflammation, it increasingly appears that analogous microbial changes in the upper GI tract appear able to produce leptin resistance in vagal afferent endings, attenuating satiety signaling.65,66 The present hypothesis suggests that it is a diet of high-density carbohydrates that produces inflammatory microbiotal changes in both areas. Periodontal disease has long been known to associate with obesity,102 while elevated levels of a particular Gram-negative oral bacterium was found to predict 98.4% of obesity in one study.103 A 4-week experiment in “Stone Age” lifestyle, including removal of refined foods and consumption of reduced levels of whole cereal grains, produced significant reductions in markers of gingival inflammation despite an absence of modern oral hygiene practices during the study.104 Over the last decade, interest in the association between periodontal disease and systemic diseases has increased markedly, along with some reconsideration of the possibility of a role for dietary carbohydrate as a cause of both (reviewed by Hujoel).100 However, elsewhere this link was misrepresented, and some were marketing dental treatments as a means to improve cardiovascular health. This led to a recent American Heart Association scientific statement emphasizing the lack of evidence indicating a causal link between periodontal disease and heart disease, and suggesting independent causative factors were causing both.105 If the high carbohydrate density of modern foods produces an inflammatory microbiota in both the mouth and small bowel, it may be this that is the root cause of both periodontal and atherosclerotic disease, as well as obesity and other metabolic syndrome-linked “diseases of affluence.”
The hypothesis and existing dietary patterns
The consensus “prudent” or Mediterranean dietary patterns could be expected to offer slight improvements over an unrestrained Western diet, due to the inclusion of more cellular plant foods at the expense of some acellular carbohydrate and fat, and an emphasis on some less refined and thus very slightly less carbohydrate-dense breads (see Figure 1). However, enough breads and baked goods with acellular dense carbohydrate are retained to keep an inflammatory microbiota and prevent substantial correction to the endemic Western leptin resistance. This is consistent with the continued requirement for conscious caloric restriction when eating these consensus diets if a steady weight is to be maintained.
Low-carbohydrate diets will reduce acellular carbohydrate consumption as a by-product of markedly reducing all carbohydrates. This is suggested to result in a larger reduction in the inflammatory nature of the GI microbiota, weight loss without conscious caloric restriction, and improved metabolic syndrome markers.86,88,106 However, low-carbohydrate diets retain some carbohydrates (usually less than 20–50 g per day), often with no regard to their density. This means what little carbohydrate is eaten may still lay the basis for an inflammatory microbiota, which is proposed to receive a second hit via additional energy for bacteria and increased LPS translocation from the elevated fat content that often accompanies a low-carbohydrate diet. This is suggested to explain the “stall” process, where low-carbohydrate dieters can lose large amounts of weight, but then cease weight loss while still overweight or begin to slowly regain weight.84,85
Severe caloric restriction for 8 weeks with a diet of refined-liquid meal-replacement formula supplemented with some vegetables (510 kcal Optifast with vegetables to a total of 600 kcal per day) was reported to normalize beta-cell function and insulin sensitivity in a small group of type II diabetics (<4 years from diagnosis).107 This represents an intake of acellular carbohydrates of around 237 kcal per day (the liquid diet was 46.4% carbohydrate), which might represent a sufficient reduction to avoid adverse metabolic microbial effects. It is of note that a Paleolithic-style diet also normalized type II diabetic glucose tolerance in a randomized trial with a similar small group, but crucially these patients were eating ad libitum.42 Both 237 kcal per day of acellular carbohydrate and an ad libitum Paleolithic style of eating may produce similarly sparse concentrations of carbohydrate in the upper GI tract, but only the Paleolithic diet is practical or sustainable in the long term. Time of intervention may be of the essence due to beta-cell damage; in a later study, longer-established type II diabetics (mean = 8 years postdiagnosis) still showed greater improvements with Paleolithic eating than with a consensus diet, but they did not obtain fully normalized blood glucose regulation.41
Hence a grain-free whole-food diet would be predicted to restore the GI microbiota to the less inflammatory state that humans coevolved with. This is consistent with the larger falls in blood glucose response and leptin/insulin-resistance, and the 20%–30% spontaneous reduction in caloric intake seen in the Paleolithic diet when eating to satiety,41–43 as well as the population-wide maintenance of healthy weight in Kitava6,7 and in other grain-free whole food-eating populations.16,20,22,29,108,109 Upon initial adoption of dietary change, overweight Westerners with higher insulin resistance might initially benefit from a low-carbohydrate form of Paleolithic-style eating. However, over time (and assuming no permanent diabetes-related damage) the resulting restoration of insulin sensitivity should mean a diet with a higher level of unrefined whole fruit and starchy root vegetables should be compatible with maintaining optimal metabolic health and stable healthy weight. This is now being reported anecdotally by Paleolithic-diet eaters, and is consistent with the condition of the Kitavan Islanders. The proposed importance of the cellularity and low carbohydrate density of fruit and vegetables in maintaining an evolutionarily appropriate microbiota might explain the failure of supplementary fiber, vitamins, or antioxidants to replicate the health effects of a diet of fruit and vegetables when taken with a Western diet.110
Suggestions for the future
The available evidence suggests that whole-food grain-free diets are substantially metabolically healthier than modern diets. How generalizable these findings are to differing populations and to what extent Western diseases such as obesity, diabetes, and atherosclerosis can be reversed by adoption of this dietary pattern should now be assessed in large-scale clinical trials. Whether the means of action is as proposed in this paper should also be tested in humans, measuring levels of inflammation, SOCS3, NFκB and leptin with titrated intake of acellular carbohydrate and fat. Whether any effects of carbohydrate density are found to be dependent on cellularity will be a crucial determinant of whether processed foods can be made safer by reformulation. Potential interactions of the Paleolithic dietary pattern with common medications should be studied carefully.23 Whether the advice of some variants of the diet regarding exclusion of legumes or dairy products have demonstrable metabolic or anti-inflammatory roles should also be evaluated.
The complexity of the microbiome and its interplay with the host is hard to overstate. Detailed 16S RNA bacterial profiles from multiple levels of the GI tract, of both the luminal contents and mucosa, may be needed to identify any bacteria or bacterial genes that vary while carbohydrate density or cellularity are altered (holding macronutrient ratios as constant as possible). Putative microbiota-related signaling molecules and metabolic effectors such as fasting-induced adipocyte factor and adenosine monophosphate-activated protein kinase should also be monitored for correlations with carbohydrate density and fat. It may be possible to establish an index system for assessing the obesogenic potential of foods, based upon the rate of proliferation of relevant bacteria either in the gut or under simulated conditions in vitro.
A simple start could be made by profiling the microbiota of Kitavan Islanders, and investigating whether comparable profiles can be produced by a Paleolithic diet in Westerners after a month or a year. Due to population constraints, we are currently dependent on grains to feed the world. Hence another question of importance is the relative obesogenic effects of different grains, and how much impact minimally processed rice, maize, millet, sorghum, or sprouting grains have on metabolism and microbiota.
Should carbohydrate density be found to correlate with increased appetite under experimental conditions, how rapidly a sugar- or flour-containing meal (against a background of otherwise constant grain-free whole foods) acts may be mechanistically and practically informative; how rapidly does a switch to flour or sugar produce leptin resistance in humans, is the localization pattern (vagal afferents, then central) the same as in rodents, and what is the duration of this effect? This will have implications for how strictly modern foods have to be avoided in order to maximize health. Whether acellular sugary drinks have similar effects, the nature of their concentration–inflammation relationships, and whether they undergo concentration or dilution in the gut lumen would likely also be of interest. The possibility that some effects of increased luminal carbohydrate density may be directly conveyed via enterochromaffin cell signaling to vagal afferents should be considered.
One complexity with the current hypothesis is that some effects that may be attributable to microbiota and carbohydrate density are hard to extricate from effects attributable to food reward. Changes in both have inevitably been present in many previous studies of food reward; for example, in the use of the highly refined and highly rewarding cafeteria diet in rodents.111 With such foods, both hypotheses predict central mechanisms to increase caloric intake, one through increased reward when a palatable food is encountered and eaten, the other through increased appetite and delayed satiety from microbiota-driven central mechanisms. One possible way to dissociate the contributions of the two might be to look at the effects of antibiotics on trials of cellular and acellular foods, while looking for correlations of microbiotal change with inflammatory and metabolic markers. A key difference to attempt to tease out may be the nature of intermeal hunger. People eating a grain-free whole-food diet anecdotally report a satiety that precludes intrusive thoughts about food, often until after a scheduled mealtime has passed. This is qualitatively different to the craving for almost anything to “fill the gap” so familiar to Westerners sensing a need for a snack between meals. An informative experiment might be one that tested whether refined diets increase subsequent intake of all foods, or only of highly palatable foods. Since all behaviors are expressed with input from the brain’s reward centers, food reward must naturally play a role in food intake, but surely so must homeostatic control of energy balance. At extremes of reward, it likely does affect meal size and short-term caloric intake. Otherwise, it certainly plays a major role in the choice between refined foods (with carefully tailored palatability) and the whole-food alternatives. That, of course, may be the most important choice of all.
Conclusion
The increased storage life and convenience of some of our oldest agricultural products may come with a hitherto unrecognized metabolic cost. The foods eaten by hunter-gatherers, non-cereal horticulturalists, and those following a modern Paleolithic or “primal” diet are sharply delineated from modern foods by their lower carbohydrate densities. Consumption of exclusively low-density carbohydrates is suggested to produce a less inflammatory GI microbiota, and may explain the apparent absence of overweight and metabolic disease in two of these groups, and the promising early data from the third. This hypothesis may also explain (1) why obesity incidence scales with refined food intake, but has such confusing correlatory patterns with macronutrients; (2) why calorie-controlled diets of Westernized foods require a perpetual fight with homeostatic correction mechanisms; (3) the link between periodontal disease and systemic atherosclerotic disease and obesity; (4) why the benefits of a diet of fruit and vegetables have not been replicated by supplements of the constituent antioxidants, vitamins, minerals, and fiber alongside a Western diet; (5) why low-carbohydrate diets produce ad libitum weight loss, but low-fat diets do not; and (6) the relative resistance of European people to obesity and diabetes from Westernized diets.
We should not settle for the meager improvements attainable from the consensus dietary advice when it is already clear that so much more might be achieved. Our sights should be set high, to see how close we can move levels of industrialized metabolic health toward those enjoyed by non-Westernized populations. While many will resist making dietary changes of such magnitude, official advice must nonetheless point in the correct direction, allowing individuals to make informed decisions.
The social and financial burden of the epidemic of obesity and metabolic syndrome threatens the long-term viability of our health-care systems and perniciously undermines the other benefits of modern civilization, including redirection of scarce financial resources. If this dietary pattern is confirmed to work, practical policy solutions must be sought. Some grain cultivars may be found to produce less inflammation, sprouting techniques may be found to be of benefit, or grain production may be replaced with root-vegetable cultivation where practical. Over time, the interplay between market forces, attainable agricultural yields, and the practicalities of food-distribution networks may allow shifting demand from a newly informed populace to reshape global agribusiness.
A dietary pattern with carbohydrates exclusively from cellular low-density sources may remove the root cause of a range of our most prevalent diseases. The potential savings in health-care costs should be borne in mind, and the hypothesis tested.
Acknowledgments
This work was not supported by funding. Thanks to Dr Francisco Bautista Cruz and Dr Andrew Samis for useful discussions over a long period, and to Dr Mike Beyak, Dr Japie Louw and Dr Philip Sherman for feedback on the evolving hypothesis.
Glossary
- Ad libitum (with regard to diet)
Eating whenever desired, and as much as desired.
- Ancestral diet
A diet of unprocessed animal and plant foods, with no grains, flour, sugar, or refined fats, as eaten by remnant modern hunter-gatherers, non-cereal horticulturists, and most humans before the adoption of cereal agriculture around 10,000 years ago.
- Energy homeostasis
The process whereby energy intake (eating) and expenditure (metabolic rate and physical activity) are monitored and controlled by the brain to maintain an appropriate balance in a similar feedback-driven manner to how body temperature or fluid balance are regulated (see leptin).
- Gastrointestinal microbiota
The community of bacteria that lives inside the gut.
- Glycemic index
A measure of how much a food elevates circulating blood glucose levels, typically expressed relative to the blood glucose-elevating properties of a meal of bread or pure glucose.
- Leptin
A hormone produced by the body’s fat stores that allows the hypothalamus of the brain to monitor the size of those stores, and regulate them by varying appetite and energy expenditure. In obesity, leptin often produces a smaller response at its receptors than it does in the lean, a phenomenon known as leptin resistance. This is believed to trigger hunger and reduced energy expenditure in an attempt to gain additional fat storage tissue, to increase leptin production levels, and overcome the leptin resistance. Thus, leptin resistance is analogous to a malfunctioning “fat thermostat.”
- Lipopolysaccharide (LPS)
A molecule that is part of the outer membrane of the Gram-negative category of bacteria. It acts as an endotoxin, at high concentrations producing powerful immune reactions and fever via the CD14/TLR4/MD2 complex of receptors. At lower concentrations, it plays a role in diet-induced inflammation.
- Paleolithic
The “old Stone Age,” extending from the first use of stone tools by human ancestors over 2 million years ago until the adoption of farming 10,000 years ago.
- Paleolithic/primal diet
Modern dietary styles that attempt to recreate key properties of ancestral diets. Their common elements are the exclusion of grain products, refined sugars, and processed foods, while encouraging whole-plant and animal foods.
- PAMPs
Pathogen-associated molecular patterns. Molecules that are commonly part of or shed by microbes, and are used by the innate immune system to recognize and mount a response to bacteria.
- Periodontal disease
A disorder of the gums, ligaments, or bone supporting the teeth.
- Vagal afferents
The fibers of the vagus nerve that carry information from the internal organs to the brain. From the upper gut, these fibers convey information about when food has been eaten, physical stretching of the gut by food, and the chemical nature of that food as it is digested. They are thought to play an important role in the signaling of satiety.
Footnotes
Disclosure
The author reports no conflicts of interest in this work.
References
- 1.Zimmet PZ, McCarty DJ, de Courten MP. The global epidemiology of non-insulin-dependent diabetes mellitus and the metabolic syndrome. J Diabetes Complications. 1997;11(2):60–68. doi: 10.1016/s1056-8727(96)00090-6. [DOI] [PubMed] [Google Scholar]
- 2.Chakravarthy MV, Booth FW. Eating, exercise, and “thrifty” genotypes: connecting the dots toward an evolutionary understanding of modern chronic diseases. J Appl Physiol. 2004;96(1):3–10. doi: 10.1152/japplphysiol.00757.2003. [DOI] [PubMed] [Google Scholar]
- 3.Lindeberg S, Lundh B. Apparent absence of stroke and ischaemic heart disease in a traditional Melanesian island: a clinical study in Kitava. J Intern Med. 1993;233(3):269–275. doi: 10.1111/j.1365-2796.1993.tb00986.x. [DOI] [PubMed] [Google Scholar]
- 4.Lindeberg S, Nilsson-Ehle P, Terent A, Vessby B, Schersten B. Cardiovascular risk factors in a Melanesian population apparently free from stroke and ischaemic heart disease: the Kitava study. J Intern Med. 1994;236(3):331–340. doi: 10.1111/j.1365-2796.1994.tb00804.x. [DOI] [PubMed] [Google Scholar]
- 5.Foster-Powell K, Holt SHA, Brand-Miller JC. International table of glycemic index and glycemic load values. Am J Clin Nutr. 2002;76(1):5–56. doi: 10.1093/ajcn/76.1.5. [DOI] [PubMed] [Google Scholar]
- 6.Lindeberg S, Eliasson M, Lindahl B, Ahren B. Low serum insulin in traditional Pacific Islanders – the Kitava Study. Metabolism. 1999;48(10):1216–1219. doi: 10.1016/s0026-0495(99)90258-5. [DOI] [PubMed] [Google Scholar]
- 7.Lindeberg S, Soderberg S, Ahren B, Olsson T. Large differences in serum leptin levels between nonwesternized and westernized populations: the Kitava study. J Intern Med. 2001;249(6):553–558. doi: 10.1046/j.1365-2796.2001.00845.x. [DOI] [PubMed] [Google Scholar]
- 8.Lindeberg S, Berntorp E, Carlsson R, Eliasson M, Marckmann P. Haemostatic variables in Pacific Islanders apparently free from stroke and ischaemic heart disease – the Kitava Study. Thromb Haemost. 1997;77(1):94–98. [PubMed] [Google Scholar]
- 9.Lindeberg S, Berntorp E, Nilsson-Ehle P, Terent A, Vessby B. Age relations of cardiovascular risk factors in a traditional Melanesian society: the Kitava Study. Am J Clin Nutr. 1997;66(4):845–852. doi: 10.1093/ajcn/66.4.845. [DOI] [PubMed] [Google Scholar]
- 10.Mente A, Razak F, Blankenberg S, et al. Ethnic variation in adiponectin and leptin levels and their association with adiposity and insulin resistance. Diabetes Care. 2010;33(7):1629–1634. doi: 10.2337/dc09-1392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tanaka M, Umezaki M, Natsuhara K, et al. No difference in serum leptin concentrations between urban-dwelling Austronesians and Non-Austronesians in Papua New Guinea. Am J Hum Biol. 2005;17(6):696–703. doi: 10.1002/ajhb.20445. [DOI] [PubMed] [Google Scholar]
- 12.Bribiescas RG. Serum leptin levels and anthropometric correlates in Ache Amerindians of eastern Paraguay. Am J Phys Anthropol. 2001;115(4):297–303. doi: 10.1002/ajpa.1085. [DOI] [PubMed] [Google Scholar]
- 13.Bribiescas RG, Hickey MS. Population variation and differences in serum leptin independent of adiposity: a comparison of Ache Amerindian men of Paraguay and lean American male distance runners. Nutr Metab (Lond) 2006;3:34. doi: 10.1186/1743-7075-3-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vessby B, Ahren B, Warensjo E, Lindgarde F. Plasma lipid fatty acid composition, desaturase activities and insulin sensitivity in Amerindian women. Nutr Metab Cardiovasc Dis. 2012;22(3):176–181. doi: 10.1016/j.numecd.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 15.Lindgarde F, Widen I, Gebb M, Ahren B. Traditional versus agricultural lifestyle among Shuar women of the Ecuadorian Amazon: effects on leptin levels. Metabolism. 2004;53(10):1355–1358. doi: 10.1016/j.metabol.2004.04.012. [DOI] [PubMed] [Google Scholar]
- 16.Biss K, Ho KJ, Mikkelson B, Lewis L, Taylor CB. Some unique biologic characteristics of the Masai of East Africa. N Engl J Med. 1971;284(13):694–699. doi: 10.1056/NEJM197104012841304. [DOI] [PubMed] [Google Scholar]
- 17.Cerqueira MT, Fry MM, Connor WE. The food and nutrient intakes of the Tarahumara Indians of Mexico. Am J Clin Nutr. 1979;32(4):905–915. doi: 10.1093/ajcn/32.4.905. [DOI] [PubMed] [Google Scholar]
- 18.Cleave TL. Conditions Caused by the Taking of Refined Carbohydrates Such as Sugar and White Flour. Bristol: Wright; 1974. The Saccharine Disease. [Google Scholar]
- 19.Connor WE, Cerqueira MT, Connor RW, Wallace RB, Malinow MR, Casdorph HR. The plasma lipids, lipoproteins, and diet of the Tarahumara indians of Mexico. Am J Clin Nutr. 1978;31(7):1131–1142. doi: 10.1093/ajcn/31.7.1131. [DOI] [PubMed] [Google Scholar]
- 20.Donnison CP. Blood pressure in the African native. Its bearing upon the aetiology of hyperpiesia and arterio-sclerosis. Lancet. 1929;213:6–7. [Google Scholar]
- 21.Eaton SB, Konner M. Paleolithic nutrition. A consideration of its nature and current implications. N Engl J Med. 1985;312(5):283–289. doi: 10.1056/NEJM198501313120505. [DOI] [PubMed] [Google Scholar]
- 22.Johnson A, Behrens C. Nutritional criteria in Machiguenga food production decisions: a linear-programming analysis. Hum Ecol. 1982;10(2):167–189. [Google Scholar]
- 23.Lindeberg S. Paleolithic diets as a model for prevention and treatment of western disease. Am J Hum Biol. 2012;24(2):110–115. doi: 10.1002/ajhb.22218. [DOI] [PubMed] [Google Scholar]
- 24.Little MA, Galvin K, Mugambi M. Cross-sectional growth of nomadic Turkana pastoralists. Hum Biol. 1983;55(4):811–830. [PubMed] [Google Scholar]
- 25.Murray MJ, Murray AB, Murray CJ. An ecological interdependence of diet and disease? A study of infection in one tribe consuming two different diets. Am J Clin Nutr. 1980;33(3):697–701. doi: 10.1093/ajcn/33.3.697. [DOI] [PubMed] [Google Scholar]
- 26.Page LB, Damon A, Moellering RC., Jr Antecedents of cardiovascular disease in six Solomon Islands societies. Circulation. 1974;49(6):1132–1146. doi: 10.1161/01.cir.49.6.1132. [DOI] [PubMed] [Google Scholar]
- 27.Schulz LO, Bennett PH, Ravussin E, et al. Effects of traditional and western environments on prevalence of type 2 diabetes in Pima Indians in Mexico and the US. Diabetes Care. 2006;29(8):1866–1871. doi: 10.2337/dc06-0138. [DOI] [PubMed] [Google Scholar]
- 28.Smith CJ, Nelson RG, Hardy SA, Manahan EM, Bennett PH, Knowler WC. Survey of the diet of Pima Indians using quantitative food frequency assessment and 24-hour recall. Diabetic Renal Disease Study. J Am Diet Assoc. 1996;96(8):778–784. doi: 10.1016/s0002-8223(96)00216-7. [DOI] [PubMed] [Google Scholar]
- 29.Truswell AS, Kennelly BM, Hansen JD, Lee RB. Blood pressures of !Kung bushmen in Northern Botswana. Am Heart J. 1972;84(1):5–12. doi: 10.1016/0002-8703(72)90299-2. [DOI] [PubMed] [Google Scholar]
- 30.Kuipers RS, Luxwolda MF, Dijck-Brouwer DA, et al. Estimated macronutrient and fatty acid intakes from an East African Paleolithic diet. Br J Nutr. 2010;104(11):1666–1687. doi: 10.1017/S0007114510002679. [DOI] [PubMed] [Google Scholar]
- 31.Strohle A, Hahn A, Sebastian A. Latitude, local ecology, and hunter-gatherer dietary acid load: implications from evolutionary ecology. Am J Clin Nutr. 2010;92(4):940–945. doi: 10.3945/ajcn.2010.29815. [DOI] [PubMed] [Google Scholar]
- 32.Strohle A, Hahn A. Diets of modern hunter-gatherers vary substantially in their carbohydrate content depending on ecoenvironments: results from an ethnographic analysis. Nutr Res. 2011;31(6):429–435. doi: 10.1016/j.nutres.2011.05.003. [DOI] [PubMed] [Google Scholar]
- 33.Cordain L, Eaton SB, Miller JB, Mann N, Hill K. The paradoxical nature of hunter-gatherer diets: meat-based, yet non-atherogenic. Eur J Clin Nutr. 2002;56(Suppl 1):S42–S52. doi: 10.1038/sj.ejcn.1601353. [DOI] [PubMed] [Google Scholar]
- 34.Diamond J. The double puzzle of diabetes. Nature. 2003;423(6940):599–602. doi: 10.1038/423599a. [DOI] [PubMed] [Google Scholar]
- 35.Hopping BN, Erber E, Mead E, Roache C, Sharma S. High levels of physical activity and obesity co-exist amongst Inuit adults in Arctic Canada. J Hum Nutr Diet. 2010;23(Suppl 1):110–114. doi: 10.1111/j.1365-277X.2010.01096.x. [DOI] [PubMed] [Google Scholar]
- 36.Mekary RA, Feskanich D, Hu FB, Willett WC, Field AE. Physical activity in relation to long-term weight maintenance after intentional weight loss in premenopausal women. Obesity (Silver Spring) 2010;18(1):167–174. doi: 10.1038/oby.2009.170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lindeberg S. Food and Western Disease: Health and Nutrition from an Evolutionary Perspective. Oxford: Wiley-Blackwell; 2010. [Google Scholar]
- 38.O’Dea K. Marked improvement in carbohydrate and lipid metabolism in diabetic Australian aborigines after temporary reversion to traditional lifestyle. Diabetes. 1984;33(6):596–603. doi: 10.2337/diab.33.6.596. [DOI] [PubMed] [Google Scholar]
- 39.Harris KA, Kris-Etherton PM. Effects of whole grains on coronary heart disease risk. Curr Atheroscler Rep. 2010;12(6):368–376. doi: 10.1007/s11883-010-0136-1. [DOI] [PubMed] [Google Scholar]
- 40.Williams PG. Evaluation of the evidence between consumption of refined grains and health outcomes. Nutr Rev. 2012;70(2):80–99. doi: 10.1111/j.1753-4887.2011.00452.x. [DOI] [PubMed] [Google Scholar]
- 41.Jonsson T, Granfeldt Y, Ahren B, et al. Beneficial effects of a Paleolithic diet on cardiovascular risk factors in type 2 diabetes: a randomized cross-over pilot study. Cardiovasc Diabetol. 2009;8:35. doi: 10.1186/1475-2840-8-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lindeberg S, Jonsson T, Granfeldt Y, et al. A Palaeolithic diet improves glucose tolerance more than a Mediterranean-like diet in individuals with ischaemic heart disease. Diabetologia. 2007;50(9):1795–1807. doi: 10.1007/s00125-007-0716-y. [DOI] [PubMed] [Google Scholar]
- 43.Jonsson T, Granfeldt Y, Erlanson-Albertsson C, Ahren B, Lindeberg S. A paleolithic diet is more satiating per calorie than a mediterranean-like diet in individuals with ischemic heart disease. Nutr Metab (Lond) 2010;7:85. doi: 10.1186/1743-7075-7-85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Osterdahl M, Kocturk T, Koochek A, Wandell PE. Effects of a short-term intervention with a paleolithic diet in healthy volunteers. Eur J Clin Nutr. 2008;62(5):682–685. doi: 10.1038/sj.ejcn.1602790. [DOI] [PubMed] [Google Scholar]
- 45.Frassetto LA, Schloetter M, Mietus-Synder M, Morris RC, Jr, Sebastian A. Metabolic and physiologic improvements from consuming a paleolithic, hunter-gatherer type diet. Eur J Clin Nutr. 2009;63(8):947–955. doi: 10.1038/ejcn.2009.4. [DOI] [PubMed] [Google Scholar]
- 46.Jonsson T, Olsson S, Ahren B, Bog-Hansen TC, Dole A, Lindeberg S. Agrarian diet and diseases of affluence – do evolutionary novel dietary lectins cause leptin resistance? BMC Endocr Disord. 2005;5:10. doi: 10.1186/1472-6823-5-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lumeng CN, Saltiel AR. Inflammatory links between obesity and metabolic disease. J Clin Invest. 2011;121(6):2111–2117. doi: 10.1172/JCI57132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lassenius MI, Pietilainen KH, Kaartinen K, et al. Bacterial endotoxin activity in human serum is associated with dyslipidemia, insulin resistance, obesity, and chronic inflammation. Diabetes Care. 2011;34(8):1809–1815. doi: 10.2337/dc10-2197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Thuy S, Ladurner R, Volynets V, et al. Nonalcoholic fatty liver disease in humans is associated with increased plasma endotoxin and plasminogen activator inhibitor 1 concentrations and with fructose intake. J Nutr. 2008;138(8):1452–1455. doi: 10.1093/jn/138.8.1452. [DOI] [PubMed] [Google Scholar]
- 50.Volynets V, Kuper MA, Strahl S, et al. Nutrition, intestinal permeability, and blood ethanol levels are altered in patients with nonalcoholic fatty liver disease (NAFLD) Dig Dis Sci. doi: 10.1007/s10620-012-2112-9. Epub March 17, 2012. [DOI] [PubMed] [Google Scholar]
- 51.Jialal I, Huet BA, Kaur H, Chien A, Devaraj S. Increased toll-like receptor activity in patients with metabolic syndrome. Diabetes Care. 2012;35(4):900–904. doi: 10.2337/dc11-2375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Monte SV, Caruana JA, Ghanim H, et al. Reduction in endotoxemia, oxidative and inflammatory stress, and insulin resistance after Roux-en-Y gastric bypass surgery in patients with morbid obesity and type 2 diabetes mellitus. Surgery. 2012;151(4):587–593. doi: 10.1016/j.surg.2011.09.038. [DOI] [PubMed] [Google Scholar]
- 53.Ghanim H, Abuaysheh S, Sia CL, et al. Increase in plasma endotoxin concentrations and the expression of Toll-like receptors and suppressor of cytokine signaling-3 in mononuclear cells after a high-fat, high-carbohydrate meal: implications for insulin resistance. Diabetes Care. 2009;32(12):2281–2287. doi: 10.2337/dc09-0979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Erridge C. Diet, commensals and the intestine as sources of pathogen-associated molecular patterns in atherosclerosis, type 2 diabetes and non-alcoholic fatty liver disease. Atherosclerosis. 2011;216(1):1–6. doi: 10.1016/j.atherosclerosis.2011.02.043. [DOI] [PubMed] [Google Scholar]
- 55.Cani PD, Bibiloni R, Knauf C, et al. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes. 2008;57(6):1470–1481. doi: 10.2337/db07-1403. [DOI] [PubMed] [Google Scholar]
- 56.Deopurkar R, Ghanim H, Friedman J, et al. Differential effects of cream, glucose, and orange juice on inflammation, endotoxin, and the expression of Toll-like receptor-4 and suppressor of cytokine signaling-3. Diabetes Care. 2010;33(5):991–997. doi: 10.2337/dc09-1630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Aljada A, Friedman J, Ghanim H, et al. Glucose ingestion induces an increase in intranuclear nuclear factor kappaB, a fall in cellular inhibitor kappaB, and an increase in tumor necrosis factor alpha messenger RNA by mononuclear cells in healthy human subjects. Metabolism. 2006;55(9):1177–1185. doi: 10.1016/j.metabol.2006.04.016. [DOI] [PubMed] [Google Scholar]
- 58.Ghanim H, Aljada A, Daoud N, Deopurkar R, Chaudhuri A, Dandona P. Role of inflammatory mediators in the suppression of insulin receptor phosphorylation in circulating mononuclear cells of obese subjects. Diabetologia. 2007;50(2):278–285. doi: 10.1007/s00125-006-0508-9. [DOI] [PubMed] [Google Scholar]
- 59.Caesar R, Reigstad CS, Backhed HK, et al. Gut-derived lipopolysaccharide augments adipose macrophage accumulation but is not essential for impaired glucose or insulin tolerance in mice. Gut. 2012 Apr 25; doi: 10.1136/gutjnl-2011-301689. [Epub ahead of print.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bergheim I, Weber S, Vos M, et al. Antibiotics protect against fructose- induced hepatic lipid accumulation in mice: role of endotoxin. J Hepatol. 2008;48(6):983–992. doi: 10.1016/j.jhep.2008.01.035. [DOI] [PubMed] [Google Scholar]
- 61.Lim JS, Mietus-Snyder M, Valente A, Schwarz JM, Lustig RH. The role of fructose in the pathogenesis of NAFLD and the metabolic syndrome. Nat Rev Gastroenterol Hepatol. 2010;7(5):251–264. doi: 10.1038/nrgastro.2010.41. [DOI] [PubMed] [Google Scholar]
- 62.Lustig RH, Schmidt LA, Brindis CD. Public health: the toxic truth about sugar. Nature. 2012;482(7383):27–29. doi: 10.1038/482027a. [DOI] [PubMed] [Google Scholar]
- 63.Ding S, Chi MM, Scull BP, et al. High-fat diet: bacteria interactions promote intestinal inflammation which precedes and correlates with obesity and insulin resistance in mouse. PLoS One. 2010;5(8):e12191. doi: 10.1371/journal.pone.0012191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.de La Serre CB, Ellis CL, Lee J, Hartman AL, Rutledge JC, Raybould HE. Propensity to high-fat diet-induced obesity in rats is associated with changes in the gut microbiota and gut inflammation. Am J Physiol Gastrointest Liver Physiol. 2010;299(2):G440–G448. doi: 10.1152/ajpgi.00098.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.de Lartigue, Barbier de la SC, Espero E, Lee J, Raybould HE. Diet-induced obesity leads to the development of leptin resistance in vagal afferent neurons. Am J Physiol Endocrinol Metab. 2011;301(1):E187–E195. doi: 10.1152/ajpendo.00056.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.de Lartigue, Barbier de la SC, Espero E, Lee J, Raybould HE. Leptin resistance in vagal afferent neurons inhibits cholecystokinin signaling and satiation in diet induced obese rats. PLoS One. 2012;7(3):e32967. doi: 10.1371/journal.pone.0032967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wilsey J, Zolotukhin S, Prima V, Scarpace PJ. Central leptin gene therapy fails to overcome leptin resistance associated with diet-induced obesity. Am J Physiol Regul Integr Comp Physiol. 2003;285(5):R1011–R1020. doi: 10.1152/ajpregu.00193.2003. [DOI] [PubMed] [Google Scholar]
- 68.Reed AS, Unger EK, Olofsson LE, Piper ML, Myers MG, Jr, Xu AW. Functional role of suppressor of cytokine signaling 3 upregulation in hypothalamic leptin resistance and long-term energy homeostasis. Diabetes. 2010;59(4):894–906. doi: 10.2337/db09-1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Membrez M, Blancher F, Jaquet M, et al. Gut microbiota modulation with norfloxacin and ampicillin enhances glucose tolerance in mice. FASEB J. 2008;22(7):2416–2426. doi: 10.1096/fj.07-102723. [DOI] [PubMed] [Google Scholar]
- 70.Poggi M, Bastelica D, Gual P, et al. C3H/HeJ mice carrying a toll-like receptor 4 mutation are protected against the development of insulin resistance in white adipose tissue in response to a high-fat diet. Diabetologia. 2007;50(6):1267–1276. doi: 10.1007/s00125-007-0654-8. [DOI] [PubMed] [Google Scholar]
- 71.Spruss A, Kanuri G, Wagnerberger S, Haub S, Bischoff SC, Bergheim I. Toll-like receptor 4 is involved in the development of fructose-induced hepatic steatosis in mice. Hepatology. 2009;50(4):1094–1104. doi: 10.1002/hep.23122. [DOI] [PubMed] [Google Scholar]
- 72.Tsukumo DM, Carvalho-Filho MA, Carvalheira JB, et al. Loss-of-function mutation in Toll-like receptor 4 prevents diet-induced obesity and insulin resistance. Diabetes. 2007;56(8):1986–1998. doi: 10.2337/db06-1595. [DOI] [PubMed] [Google Scholar]
- 73.Shin SC, Kim SH, You H, et al. Drosophila microbiome modulates host developmental and metabolic homeostasis via insulin signaling. Science. 2011;334(6056):670–674. doi: 10.1126/science.1212782. [DOI] [PubMed] [Google Scholar]
- 74.Lam V, Su J, Koprowski S, et al. Intestinal microbiota determine severity of myocardial infarction in rats. FASEB J. 2012;26(4):1727–1735. doi: 10.1096/fj.11-197921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457(7228):480–484. doi: 10.1038/nature07540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.De Filippo C, Cavalieri D, Di Paola M, et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A. 2010;107(33):14691–14696. doi: 10.1073/pnas.1005963107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Backhed F. Programming of host metabolism by the gut microbiota. Ann Nutr Metab. 2011;58(Suppl 2):44–52. doi: 10.1159/000328042. [DOI] [PubMed] [Google Scholar]
- 78.Wanders AJ, van den Borne JJ, de Graaf C, et al. Effects of dietary fibre on subjective appetite, energy intake and body weight: a systematic review of randomized controlled trials. Obes Rev. 2011;12(9):724–739. doi: 10.1111/j.1467-789X.2011.00895.x. [DOI] [PubMed] [Google Scholar]
- 79.Cani PD, Amar J, Iglesias MA, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761–1772. doi: 10.2337/db06-1491. [DOI] [PubMed] [Google Scholar]
- 80.Laugerette F, Furet JP, Debard C, et al. Oil composition of high-fat diet affects metabolic inflammation differently in connection with endotoxin receptors in mice. Am J Physiol Endocrinol Metab. 2012;302(3):E374–E386. doi: 10.1152/ajpendo.00314.2011. [DOI] [PubMed] [Google Scholar]
- 81.Eguchi K, Manabe I, Oishi-Tanaka Y, et al. Saturated fatty acid and TLR signaling link β cell dysfunction and islet inflammation. Cell Metab. 2012;15(4):518–533. doi: 10.1016/j.cmet.2012.01.023. [DOI] [PubMed] [Google Scholar]
- 82.Lee JY, Sohn KH, Rhee SH, Hwang D. Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cyclooxygenase-2 mediated through Toll-like receptor 4. J Biol Chem. 2001;276(20):16683–16689. doi: 10.1074/jbc.M011695200. [DOI] [PubMed] [Google Scholar]
- 83.Suganami T, Tanimoto-Koyama K, Nishida J, et al. Role of the Toll-like receptor 4/NF-kappaB pathway in saturated fatty acid-induced inflammatory changes in the interaction between adipocytes and macrophages. Arterioscler Thromb Vasc Biol. 2007;27(1):84–91. doi: 10.1161/01.ATV.0000251608.09329.9a. [DOI] [PubMed] [Google Scholar]
- 84.Foster GD, Wyatt HR, Hill JO, et al. A randomized trial of a low-carbohydrate diet for obesity. N Engl J Med. 2003;348(21):2082–2090. doi: 10.1056/NEJMoa022207. [DOI] [PubMed] [Google Scholar]
- 85.Foster GD, Wyatt HR, Hill JO, et al. Weight and metabolic outcomes after 2 years on a low-carbohydrate versus low-fat diet: a randomized trial. Ann Intern Med. 2010;153(3):147–157. doi: 10.1059/0003-4819-153-3-201008030-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Gardner CD, Kiazand A, Alhassan S, et al. Comparison of the Atkins, Zone, Ornish, and LEARN diets for change in weight and related risk factors among overweight premenopausal women: the A TO Z Weight Loss Study: a randomized trial. JAMA. 2007;297(9):969–977. doi: 10.1001/jama.297.9.969. [DOI] [PubMed] [Google Scholar]
- 87.Hite AH, Berkowitz VG, Berkowitz K. Low-carbohydrate diet review: shifting the paradigm. Nutr Clin Pract. 2011;26(3):300–308. doi: 10.1177/0884533611405791. [DOI] [PubMed] [Google Scholar]
- 88.McAuley KA, Hopkins CM, Smith KJ, et al. Comparison of high-fat and high-protein diets with a high-carbohydrate diet in insulin-resistant obese women. Diabetologia. 2005;48(1):8–16. doi: 10.1007/s00125-004-1603-4. [DOI] [PubMed] [Google Scholar]
- 89.Brinkworth GD, Noakes M, Buckley JD, Keogh JB, Clifton PM. Long-term effects of a very-low-carbohydrate weight loss diet compared with an isocaloric low-fat diet after 12 mo. Am J Clin Nutr. 2009;90(1):23–32. doi: 10.3945/ajcn.2008.27326. [DOI] [PubMed] [Google Scholar]
- 90.de Souza RJ, Bray GA, Carey VJ, et al. Effects of 4 weight-loss diets differing in fat, protein, and carbohydrate on fat mass, lean mass, visceral adipose tissue, and hepatic fat: results from the POUNDS LOST trial. Am J Clin Nutr. 2012;95(3):614–625. doi: 10.3945/ajcn.111.026328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Pendyala S, Walker JM, Holt PR. A High-Fat Diet Is Associated With Endotoxemia That Originates From the Gut. Gastroenterology. 2012;142(5):1100–1101. doi: 10.1053/j.gastro.2012.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Ng A, Waldron KW. Effect of cooking and pre-cooking on cell-wall chemistry in relation to firmness of carrot tissues. J Sci Food Agric. 1997;73(4):513–502. [Google Scholar]
- 93.Parker CC, Parker ML, Smith AC, Waldron KW. Pectin distribution at the surface of potato parenchyma cells in relation to cell-cell adhesion. J Agric Food Chem. 2001;49(9):4364–4371. doi: 10.1021/jf0104228. [DOI] [PubMed] [Google Scholar]
- 94.Roman-Gutierrez AD, Guilbert S, Cuq B. Description of microstructural changes in wheat flour and flour components during hydration by using environmental scanning electron microscopy. Lebenson Wiss Technol. 2002;35:730–740. [Google Scholar]
- 95.Fincher GB. Molecular and cellular biology associatedwith endosperm mobilization in germinating cereal grains. Annu Rev Plant Physiol. 1989;40:305–346. [Google Scholar]
- 96.Shapiro A, Mu W, Roncal C, Cheng KY, Johnson RJ, Scarpace PJ. Fructose-induced leptin resistance exacerbates weight gain in response to subsequent high-fat feeding. Am J Physiol Regul Integr Comp Physiol. 2008;295(5):R1370–R1375. doi: 10.1152/ajpregu.00195.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Mercader J. Mozambican grass seed consumption during the Middle Stone Age. Science. 2009;326(5960):1680–1683. doi: 10.1126/science.1173966. [DOI] [PubMed] [Google Scholar]
- 98.Piperno DR, Weiss E, Holst I, Nadel D. Processing of wild cereal grains in the Upper Palaeolithic revealed by starch grain analysis. Nature. 2004;430(7000):670–673. doi: 10.1038/nature02734. [DOI] [PubMed] [Google Scholar]
- 99.Revedin A, Aranguren B, Becattini R, et al. Thirty thousand-year-old evidence of plant food processing. Proc Natl Acad Sci U S A. 2010;107(44):18815–18819. doi: 10.1073/pnas.1006993107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hujoel P. Dietary carbohydrates and dental-systemic diseases. J Dent Res. 2009;88(6):490–502. doi: 10.1177/0022034509337700. [DOI] [PubMed] [Google Scholar]
- 101.Klaus HD, Tam ME. Oral health and the postcontact adaptive transition: a contextual reconstruction of diet in Morrope, Peru. Am J Phys Anthropol. 2010;141(4):594–609. doi: 10.1002/ajpa.21179. [DOI] [PubMed] [Google Scholar]
- 102.Wood N, Johnson RB, Streckfus CF. Comparison of body composition and periodontal disease using nutritional assessment techniques: Third National Health and Nutrition Examination Survey (NHANES III) J Clin Periodontol. 2003;30(4):321–327. doi: 10.1034/j.1600-051x.2003.00353.x. [DOI] [PubMed] [Google Scholar]
- 103.Goodson JM, Groppo D, Halem S, Carpino E. Is obesity an oral bacterial disease? J Dent Res. 2009;88(6):519–523. doi: 10.1177/0022034509338353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Baumgartner S, Imfeld T, Schicht O, Rath C, Persson RE, Persson GR. The impact of the stone age diet on gingival conditions in the absence of oral hygiene. J Periodontol. 2009;80(5):759–768. doi: 10.1902/jop.2009.080376. [DOI] [PubMed] [Google Scholar]
- 105.Lockhart PB, Bolger AF, Papapanou PN, et al. Periodontal disease and atherosclerotic vascular disease: does the evidence support an independent association?: A scientific statement from the American Heart Association. Circulation. 2012;125(20):2520–2544. doi: 10.1161/CIR.0b013e31825719f3. [DOI] [PubMed] [Google Scholar]
- 106.Accurso A, Bernstein RK, Dahlqvist A, et al. Dietary carbohydrate restriction in type 2 diabetes mellitus and metabolic syndrome: time for a critical appraisal. Nutr Metab (Lond) 2008;5:9. doi: 10.1186/1743-7075-5-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lim EL, Hollingsworth KG, Aribisala BS, Chen MJ, Mathers JC, Taylor R. Reversal of type 2 diabetes: normalisation of beta cell function in association with decreased pancreas and liver triacylglycerol. Diabetologia. 2011;54(10):2506–2514. doi: 10.1007/s00125-011-2204-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ho KJ, Mikkelson B, Lewis LA, Feldman SA, Taylor CB. Alaskan Arctic Eskimo: responses to a customary high fat diet. Am J Clin Nutr. 1972;25(8):737–745. doi: 10.1093/ajcn/25.8.737. [DOI] [PubMed] [Google Scholar]
- 109.Schaefer O. Medical observations and problems in the Canadian Arctic. II. Can Med Assoc J. 1959;81:386–393. [PMC free article] [PubMed] [Google Scholar]
- 110.Bruckdorfer KR. Antioxidants and CVD. Proc Nutr Soc. 2008;67(2):214–222. doi: 10.1017/S0029665108007052. [DOI] [PubMed] [Google Scholar]
- 111.Sampey BP, Vanhoose AM, Winfield HM, et al. Cafeteria diet is a robust model of human metabolic syndrome with liver and adipose inflammation: comparison to high-fat diet. Obesity (Silver Spring) 2011;19(6):1109–1117. doi: 10.1038/oby.2011.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.www.ndb.nal.usda.gov. NDL/FNIC Food Composition Database. Agricultural Research Service National Agricultural Library; [Accessed July 2012]. [homepage on the internet] (modified Dec 7. 2011].Available from http://ndb.nal.usda.gov. [Google Scholar]