Abstract
Araucana chickens are known for their rounded, tailless rumps and tufted ears. Inheritance studies have shown that the rumpless (Rp) and ear-tufted (Et) loci each act in an autosomal dominant fashion, segregate independently, and are associated with an increased rate of embryonic mortality. To find genomic regions associated with Rp and Et, we generated genome-wide SNP profiles for a diverse population of 60 Araucana chickens using the 60 K chicken SNP BeadChip. Genome-wide association studies using 40 rumpless and 11 tailed birds showed a strong association with rumpless on Gga 2 (P raw = 2.45×10−10, P genome = 0.00575), and analysis of genotypes revealed a 2.14 Mb haplotype shared by all rumpless birds. Within this haplotype, a 0.74 Mb critical interval containing two Iroquois homeobox genes, Irx1 and Irx2, was unique to rumpless Araucana chickens. Irx1 and Irx2 are central for developmental prepatterning, but neither gene is known to have a role in mechanisms leading to caudal development. A second genome-wide association analysis using 30 ear-tufted and 28 non-tufted birds revealed an association with tufted on Gga 15 (P raw = 6.61×10−7, P genome = 0.0981). We identified a 0.58 Mb haplotype common to tufted birds and harboring 7 genes. Because homozygosity for Et is nearly 100% lethal, we employed a heterozygosity mapping approach to prioritize candidate gene selection. A 60 kb region heterozygous in all Araucana chickens contains the complete coding sequence for TBX1 and partial sequence for GNB1L. TBX1 is an important transcriptional regulator of embryonic development and a key genetic determinant of human DiGeorge syndrome. Herein, we describe localization of Rp and Et and identification of positional candidate genes.
Introduction
There are hundreds of domestic chicken breeds worldwide [1]. Breeds were generally developed for meat and egg production, but morphological traits, plumage color, and other distinctive characteristics were also selected. The Araucana chicken, originally from Chile, is a multi-purpose breed initially established for its blue-shelled eggs [1], [2]. Araucana chickens are also known for two other distinguishing traits: a rounded, tailless rump and protruding ear-tufts. Although these traits segregate in the population, the United States Araucana breed standard requires show birds to possess both phenotypes.
The rumpless phenotype is characterized by the absence of all free caudal vertebrae and the uropygial gland [3]. Without underlying skeletal support, birds with caudal truncation lack a fleshy rump and tail feathers [3]. An intermediate rumpless phenotype, wherein some caudal vertebrae are present but irregularly fused together, is thought to result from a modifier gene introduced through crosses with non-Araucana tailed chickens [3], [4]. The rumpless phenotype arises from a defect in caudal patterning that is controlled by a dominant gene (Rp) [3]. Rumpless Araucana chickens may be heterozygous or homozygous for this locus. In test matings, all rumpless intermediates were determined to be heterozygous (Rp/rp +) [3]. Homozygosity is underrepresented among chicks from rumpless to rumpless matings, indicating that the Rp/Rp genotype has reduced viability [3], [5]. Birds having at least one copy of Rp have increased mortality in the embryo stage, with death occurring at 17 to 21 days of incubation [3]. Rumpless birds also have reduced fecundity as adults [3].
Ear-tufts are feather-covered, epidermal protrusions originating near the ear canal (Figure 1). The mass of tissue forming the protrusion, or peduncle, is believed to develop as a result of the incomplete fusion of the hyomandibular arches, and it can vary in position and length (from 2 mm to 2 cm) [6], [7]. Tufted chickens may also have structural rearrangement of the ears [6]. Abnormalities include irregularly shaped external ear openings and shortened or absent external auditory canals [6].
Figure 1. Araucana chicken.
(a) General appearance of a rumpless, tufted Araucana chicken. (b) For comparison, a tailed, non-tufted Araucana chicken.
Inheritance studies indicate that tufted is governed by a dominant locus, Et [6], [8]. Test matings show that all tufted birds are heterozygous (Et/et +) and that homozygosity for Et is lethal at about 17–19 days of incubation [6], [8]. Lethality among a portion of heterozygous birds is also reported, appearing to occur at 20–21 days of incubation [8]. Post-hatch mortality is significantly higher among tufted chickens [6], [8].
Because tufts can occur unilaterally or bilaterally and may differ in size from one side to the other, Et is proposed to have variable expressivity [6]. In addition, a paucity of tufted progeny from mating studies in 1978 suggests reduced penetrance of the tufted locus [6]. In 1981, Somes and Pabilonia identified a tufted male that produced excessive tufted progeny when crossed with an et +/et + White Leghorn (86%), and they speculated that Et/Et birds may occasionally reach maturity [8]. The non-tufted chicks from the Et/Et male produced tufted progeny when crossed with an et +/et + White Leghorn, indicating that their predicted genotype does not match their phenotype, providing further evidence for variable penetrance.
The aim of our investigation was to localize the genetic bases for the rumpless and tufted phenotypes of the Araucana chicken. To this end, we generated genome-wide SNP profiles for 60 Araucana chickens using the 60 K chicken SNP BeadChip [9]. Using a genome-wide association approach, we elucidate the chromosomal regions harboring Rp and Et and identify strong candidate genes for each trait.
Results
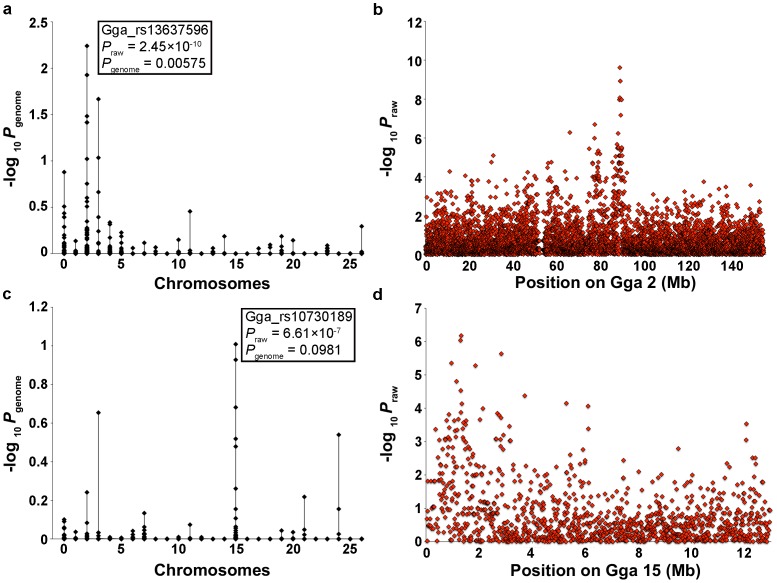
Case/control analyses were carried out using 40 rumpless and 11 tailed Araucana chickens (Figure 2a). Seven birds described as having partial tails by their breeders were excluded from the rumpless association analysis because of uncertainty concerning their phenotype. A total of 191 SNPs were associated with the rumpless phenotype (P raw ≤0.0001), 72 of which were located on Gga 2 (Figure 2b). The most significant result obtained was for SNP Gga_rs13637596, located on chromosome 2 at position 88.95 Mb (P raw = 2.45×10−10, P genome = 0.00575). The next two most significant results were for proximal SNPs located at 89.17 Mb (P raw = 1.20×10−9, P genome = 0.0119) and 89.19 Mb (P raw = 1.20×10−9, P genome = 0.0119).
Figure 2. Genome-wide association for Rp and Et.
After 100,000 permutations, the genome-wide adjusted P values (−log10 P genome) for each SNP are plotted by chromosome (left). The raw P values for the most strongly associated chromosomes are plotted against chromosomal position (right). (a,b) 40 rumpless versus 11 tailed Araucana chickens (c,d) 30 tufted versus 28 non-tufted Araucana chickens.
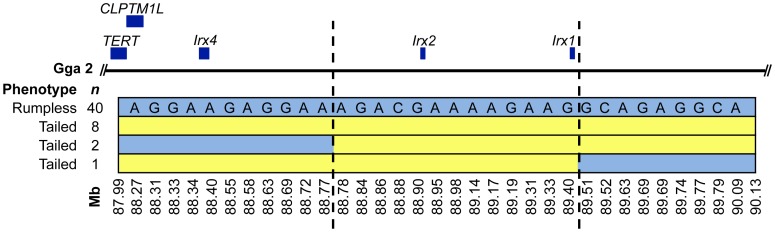
Analysis of genotypes in the Gga 2 region revealed a 2.14 Mb haplotype (87.99–90.13 Mb) predicted to contain five genes (Figure 3). All 40 rumpless birds had at least one copy of the haplotype: 18 were homozygous and 22 were heterozygous. Partial tailed birds were heterozygous. The haplotype was absent in its entirety from the 11 tailed birds. Three tailed birds were heterozygous for partial blocks of the haplotype and further delimit the critical interval to 0.74 Mb (88.77–89.51 Mb). This region contains two candidate genes: Irx1 and Irx2.
Figure 3. Localization of Rp.
Physical map showing the relative positions of mapped genes and informative SNP markers within the 2.14 Mb rumpless haplotype on Gga 2. Light blue shading denotes the rumpless haplotype (alleles are shown in the top row). Dashed lines flank the critical interval wherein no tailed birds share the rumpless haplotype.
Analyses for association with the tufted phenotype, using 30 cases and 28 controls, resulted in 31 significant SNPs, 11 of which map to Gga 15 (Figure 2c). The most significant results were for SNPs Gga_rs10730189 (P raw = 6.61×10−7, P genome = 0.0981) and Gga_rs15762547 (P raw = 9.19×10−7, P genome = 0.118), located at positions 1.33 Mb and 1.30 Mb on chromosome 15, respectively. Four other proximal SNPs also reached significance (Figure 2d).
Analysis of genotypes reveals that 29 of 30 tufted birds shared a haplotype extending from the telomere of Gga 15 to position 1.75 Mb. These birds were heterozygous for the complete haplotype. Two of 28 non-tufted birds were also heterozygous for the haplotype in its entirety. A single tufted bird shared only part of the 1.75 Mb haplotype, defining a 0.58 Mb (0.90–1.48 Mb) critical interval that is heterozygous in all 30 tufted birds and contains 7 genes. Because tufted is nearly always recessive lethal, blocks of homozygosity for the tufted haplotype were identified to reduce the number of candidate genes. Homozygosity blocks in three birds flank a 60 kb interval harboring two genes: TBX1 and GNB1L (Figure 4).
Figure 4. Localization of Et.
Physical map showing the relative positions of genes and informative SNP markers in the associated region of Gga 15. Alleles of the tufted haplotype and positions are shown. Pale green bars denote heterozygosity for the tufted haplotype. Black bars denote homozygosity for the tufted haplotype. Dashed lines mark a 60 kb interval wherein all tufted birds are heterozygous for the haplotype.
Discussion
In this study, we used genome-wide SNP profiles to localize genes causative for two breed-defining phenotypes of Araucana chickens, rumpless and ear-tufts. We took advantage of the fact that both traits segregate independently in the population by using a single data set to carry out an association analysis for each trait. Haplotype analyses based on inheritance patterns were used to identify positional candidate genes for both traits.
We identified a rumpless haplotype spanning 2.14 Mb and five genes on chromosome 2. The haplotype is present in the heterozygous or homozygous state in rumpless birds. All 7 birds with partial tails are heterozygous for the rumpless haplotype and likely represent the intermediate phenotype described by Dunn and Landauer [3]. Because rumpless is dominant and fully penetrant, we further delimited the critical interval by identifying regions of the haplotype shared by tailed birds. A 0.74 Mb region common to all rumpless birds, and absent from 11 tailed birds, harbors Rp.
These data reveal that Rp maps to a region of Gga 2 that is distinct from the predicted location of genes previously associated with caudal truncation [10]–[14]. The 0.74 Mb critical interval contains the Iroquois homeobox genes, Irx1 and Irx2. The Iroquois genes encode transcription factors that function in patterning and regionalization of tissues early in development [15]. Irx1 and Irx2 are prepattern and proneural genes first identified in Drosophila and Xenopus [16], [17]. Studies of gene function suggest that Irx genes have redundant yet distinct roles in development [18], [19]. Irx genes have been knocked out in mice and zebrafish with little effect on tail development [19]–[23]. However, the rumpless phenotype is dominant, suggesting that misexpression of Irx1 or Irx2 may underlie the trait, rather than loss of function.
We identified SNPs on Gga 15 that are strongly associated with the tufted phenotype and define a 0.58 Mb haplotype for which all tufted birds in our cohort are heterozygous. No birds are homozygous for the complete tufted haplotype. These data support conclusions from previous inheritance studies that suggest nearly 100% of tufted birds are heterozygous, and that Et/Et is lethal [6], [8].
Two non-tufted Araucana chickens are heterozygous for the tufted haplotype. These birds may signify reduced penetrance. Penetrance of the tufted allele is estimated to range from 86% to 96% [6], [8]. Based on the assigned phenotypes and the associated haplotype, we observed 94% penetrance in our cohort. Alternatively, these birds may have been incorrectly phenotyped by their breeders due to short peduncles or missing protruding feathers.
The 0.58 Mb haplotype harbors 7 protein-coding genes. Unlike rumpless, identification of the tufted haplotype in non-tufted birds could not be used to narrow the critical interval because of reduced penetrance. However, because homozygosity for Et is nearly always lethal, we were able to prioritize candidate gene selection using heterozygosity mapping. Tufted birds with blocks of homozygosity extending into the 0.58 Mb common haplotype were identified, and these regions were deemed less likely to harbor the Et locus. These data indicate that Et is located in a region containing partial coding sequence for GNB1L, which encodes a protein implicated in neuropsychiatric disorders [24], [25], and complete coding sequence for TBX1 [26], an important transcriptional regulator of embryonic development.
Haploinsufficiency for TBX1 is considered to be the key genetic determinant of human DiGeorge syndrome (DGS), which is caused by a heterozygous chromosomal deletion of 22q11.2 [27]. While the clinical phenotype is highly variable, DGS is characterized by craniofacial and cardiovascular abnormalities. Malformations in DGS are attributed to disturbed segmentation and patterning of the pharyngeal structures [28]. Auricular defects common in DGS include narrow or absent external ear canal and protruding ears [29]. Homozygosity for null mutations of TBX1 in mice and zebrafish causes a range of phenotypic effects similar to DGS, including abnormal ear development [30], [31]. Based on phenotypic similarities between the malformations causing ear tufts and DGS, TBX1 is a highly plausible candidate gene and the primary focus of ongoing work to identify the genetic basis for ear-tufts in Araucana chickens.
In conclusion, we used genome-wide association and haplotype analyses to localize Rp and Et to chicken chromosomes 2 and 15, respectively. In addition, we identified candidate genes that are immediate targets for future work.
Materials and Methods
Ethics Statement
This study was approved by the Clemson University IACUC protocol number 2011-041 and IBC protocol number 2010-041.
Study Cohort
Whole blood for DNA was collected from 6 different flocks of Araucana chickens from the United States. Phenotypic information and photographs, when available, were provided by owners. Birds with tufts of any size and on either side of the head were classified as tufted. Because both traits segregate in the Araucana population, birds were selected to ensure that the phenotypes were balanced. Our study cohort comprised 60 Araucana chickens: 21 rumpless/tufted birds, 20 rumpless/non-tufted birds, 7 tailed/non-tufted birds, 5 tailed/tufted birds, 5 partial/tufted birds, and 2 partial/non-tufted birds. Genomic DNA was isolated using the DNeasy blood and tissue kit (QIAGEN, Valencia, USA) and adjusted to a concentration of 50 ng/uL.
Genome-wide Association Mapping
SNP genotypes were generated using the Illumina 60 K chicken SNP BeadChip, which has 57,636 SNPs across chromosomes 1 through 28, Z, W, and two unmapped linkage groups [9]. BeadChips were processed by DNA Landmarks (Quebec, Canada), according to manufacturer’s protocols. Raw data files were analyzed using GenomeStudio’s Genotyping Module to generate SNP calls. The PLINK Input Report Plug-in v2.1.1 was used to format the data. For analysis, Gga 27, Gga 28, Gga Z, Gga W, and microchromosomes were all identified as chromosome zero. Case/control analyses using 56,685 SNPs were performed using PLINK [32]. Two birds with excessive missing data were excluded from all analyses. By convention, P raw values ≤0.0001 were considered significant. Permutation testing, using 100,000 iterations, was carried out using PLINK.
Acknowledgments
We are grateful to the Araucana Club of America and their members who provided samples, the Morgan Poultry Center at Clemson University for their assistance, and the Clemson University Genomics Institute for use of software and hardware.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: This study was supported by NIH/NIDCD (DC009236), with supplemental funding from the American Recovery and Reinvestment Act to SCC. Technical Contribution No. 6029 of the Clemson University Experiment Station. This material is based upon work supported by NIFA/USDA, under project number SC-1700374 to SCC. http://www.nidcd.nih.gov/Pages/default.aspx http://www.recovery.gov/Pages/default.aspx http://www.clemson.edu/public/experiment_station/faculty_staff/research_projects.html http://www.csrees.usda.gov/ The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Ekarius C. Storey’s Illustrated Guide to Poultry Breeds. North Adams, MA: Storey Publishing. 2007. pp. 23–24.
- 2.Browman DL. Advances in Andean Archaeology. Berlin: Mouton Publishers. 1978. pp. 189–196.
- 3.Dunn LC, Landauer W. The genetics of the rumpless fowl with evidence of a case of changing dominance. J Genet. 1934;29:217–243. [Google Scholar]
- 4.Dunn LC, Landauer W. Further data on genetic modification of rumplessness in the fowl. J Genet. 1936;33:401–405. [Google Scholar]
- 5.Zwilling E. The development of dominant rumplessness in chick embryos. Genetics. 1942;27:641–656. doi: 10.1093/genetics/27.6.641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Somes RG., Jr Ear-Tufts: a skin structure mutation of the Araucana fowl. J Hered. 1978;69:91–96. [Google Scholar]
- 7.Pabilonia MS, Somes RG., Jr The Embryonic Development of Ear-Tufts and Associated Structural Head and Neck Abnormalities of the Araucana Fowl. Poult Sci. 1983;62:1539–1542. doi: 10.3382/ps.0621539. [DOI] [PubMed] [Google Scholar]
- 8.Somes RG, Jr, Pabilonia MS. Ear tuftedness: a lethal condition in the Araucana fowl. J Hered. 1981;72:121–124. doi: 10.1093/oxfordjournals.jhered.a109439. [DOI] [PubMed] [Google Scholar]
- 9.Groenen MAM, Megens HJ, Zare Y, Warren WC, Hillier LW, et al. The development and characterization of a 60 K SNP chip for chicken. BMC Genomics. 2011;12:274. doi: 10.1186/1471-2164-12-274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Herrmann BG, Labeit S, Poustka A, King TR, Lehrach H. Cloning of the T gene required in mesoderm formation in the mouse. Nature. 1990;343:617–622. doi: 10.1038/343617a0. [DOI] [PubMed] [Google Scholar]
- 11.Greco TL, Takada S, Newhouse MM, McMahon JA, McMahon AP, et al. Analysis of the vestigial tail mutation demonstrates that Wnt-3a gene dosage regulates mouse axial development. Genes Dev. 1996;10:313–324. doi: 10.1101/gad.10.3.313. [DOI] [PubMed] [Google Scholar]
- 12.Ross AJ, Ruiz-Perez V, Wang Y, Hagan DM, Scherer S, et al. A Homeobox gene, HLXB9, is the major locus for dominantly inherited sacral agenesis. Nat Genet. 1998;20:358–361. doi: 10.1038/3828. [DOI] [PubMed] [Google Scholar]
- 13.Abu-Abed S, Dollé P, Metzger D, Beckett B, Chambon P, et al. The retinoic acid-metabolizing enzyme, CYP26A1, is essential for normal hindbrain patterning, vertebral identity, and development of posterior structures. Genes Dev. 2001;15:226–240. doi: 10.1101/gad.855001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.van den Akker E, Forlani S, Chawengsaksophak K, de Graaff W, Beck F, et al. Cdx1 and Cdx2 have overlapping functions in anteroposterior patterning and posterior axis elongation. Development. 2002;129:2181–2193. doi: 10.1242/dev.129.9.2181. [DOI] [PubMed] [Google Scholar]
- 15.Cavodeassi F, Modolell J, Gómez-Skarmeta JL. The Iroquois family of genes: from body building to neural patterning. Development. 2001;128:2847–2855. doi: 10.1242/dev.128.15.2847. [DOI] [PubMed] [Google Scholar]
- 16.Gómez-Skarmeta JL, Diez del Corral R, de la Calle-Mustienes E, Ferré-Marcó D, Modolell J. Araucan and caupolican, two members of the novel iroquois complex, encode homeoproteins that control proneural and vein-forming genes. Cell. 1996;85:95–105. doi: 10.1016/s0092-8674(00)81085-5. [DOI] [PubMed] [Google Scholar]
- 17.Gómez-Skarmeta JL, Modolell J. araucan and caupolican provide a link between compartment subdivisions and patterning of sensory organs and veins in the Drosophila wing. Genes Dev. 1996;10:2935–2945. doi: 10.1101/gad.10.22.2935. [DOI] [PubMed] [Google Scholar]
- 18.Costantini DL, Arruda EP, Agarwal P, Kim KH, Zhu Y, et al. The homeodomain transcription factor Irx5 establishes the mouse cardiac ventricular repolarization gradient. Cell. 2005;123:347–358. doi: 10.1016/j.cell.2005.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lebel M, Agarwal P, Cheng CW, Kabir MG, Chan TY. The Iroquois homeobox gene Irx2 is not essential for normal development of the heart and midbrain-hindbrain boundary in mice. Mol Cell Biol. 2003;23:8216–8225. doi: 10.1128/MCB.23.22.8216-8225.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Itoh M, Kudoh T, Dedekian M, Kim CH, Chitnis AB. A role for iro1 and iro7 in the establishment of an anteroposterior compartment of the ectoderm adjacent to the midbrain-hindbrain boundary. Development. 2002;129:2317–2327. doi: 10.1242/dev.129.10.2317. [DOI] [PubMed] [Google Scholar]
- 21.Peters T, Ausmeier K, Dildrop R, Rüther U. The mouse Fused toes (Ft) mutation is the result of a 1.6-Mb deletion including the entire Iroquois B gene cluster. Mamm Genome. 2002;13:186–188. doi: 10.1007/s00335-001-2142-7. [DOI] [PubMed] [Google Scholar]
- 22.Cheng CW, Yan CHM, Hui CC, Strähle U, Cheng SH. The Homeobox gene irx1a is required for the propagation of the neurogenic waves in the zebrafish retina. Mech Develop. 2006;123:252–263. doi: 10.1016/j.mod.2005.12.001. [DOI] [PubMed] [Google Scholar]
- 23.Kimura W, Machii M, Xue X, Sultana N, Hikosaka K, et al. Irx1 mutant mice show reduced tendon differentiation and no patterning defects in musculoskeletal system development. Genesis. 2011;49:2–9. doi: 10.1002/dvg.20688. [DOI] [PubMed] [Google Scholar]
- 24.Williams NM, Glaser B, Norton N, Williams H, Pierce T, et al. Strong evidence that GNB1L is associated with schizophrenia. Hum Mol Genet. 2008;17:555–566. doi: 10.1093/hmg/ddm330. [DOI] [PubMed] [Google Scholar]
- 25.Li Y, Zhao Q, Wang T, Liu J, Li J, et al. Association study between GNB1L and three major mental disorders in Chinese Han populations. Psychiat Res. 2011;187:457–459. doi: 10.1016/j.psychres.2010.04.019. [DOI] [PubMed] [Google Scholar]
- 26.Völker M, Backström N, Skinner BM, Langley EJ, Bunzey SK, et al. Copy number variation, chromosome rearrangement, and their association with recombination during avian evolution. Gen Res. 2010;20:503–511. doi: 10.1101/gr.103663.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yagi H, Furutani Y, Hamada H, Sasaki T, Asakawa S, et al. Role of TBX1 in human del22q11.2 syndrome. Lancet. 2003;362:1366–1373. doi: 10.1016/s0140-6736(03)14632-6. [DOI] [PubMed] [Google Scholar]
- 28.Wurdak H, Ittner LM, Sommer L. DiGeorge syndrome and pharyngeal apparatus development. BioEssays. 2006;28:1078–1086. doi: 10.1002/bies.20484. [DOI] [PubMed] [Google Scholar]
- 29.Butts SC. The facial phenotype of the velo-cardio-facial syndrome. Int J Pediatr Otorhinolaryngol. 2009;73:343–350. doi: 10.1016/j.ijporl.2008.10.011. [DOI] [PubMed] [Google Scholar]
- 30.Jerome LA, Papaioannou VE. DiGeorge syndrome phenotype in mice mutant for the T-box gene, Tbx1. Nat Genet. 2001;27:286–291. doi: 10.1038/85845. [DOI] [PubMed] [Google Scholar]
- 31.Piotrowski T, Ahn DG, Schilling TF, Nair S, Ruvinsky I, et al. The zebrafish van gogh mutation disrupts tbx1, which is involved in the DiGeorge deletion syndrome in humans. Development. 2003;130:5043–5052. doi: 10.1242/dev.00704. [DOI] [PubMed] [Google Scholar]
- 32.Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559–575. doi: 10.1086/519795. [DOI] [PMC free article] [PubMed] [Google Scholar]