Abstract
Pneumocystis species are opportunistic fungal organisms that cause severe pneumonia in immune-compromised hosts, with resultant high morbidity and mortality. Recent work indicates that IL-17 responses are important components of host defense against fungal pathogens. In the present study, we demonstrate that cell-surface β-glucan components of Pneumocystis (PCBG) stimulate human dendritic cells (DCs) to secrete IL-23 and IL-6. These cytokines are well established to stimulate a T helper–17 (Th17) phenotype. Accordingly, we further observe that PCBG-stimulated human DCs interact with lymphocytes to drive the secretion of IL-17 and IL-22, both Th17-produced cytokines. The activation of DCs was shown to involve the dectin-1 receptor with a downstream activation of the Syk kinase and subsequent translocation of both the canonical and noncanonical components of the NF-κB transcription factor family. Finally, we demonstrate that glycosphingolipid-rich microdomains of the plasma membrane participate in the activation of DCs by PCBG through the accumulation of lactosylceramide at the cell surface during stimulation with PCBG. These data strongly support the idea that the β-glucan surface components of Pneumocystis drive the activation of the IL-23/IL-17 axis during this infection, through a glycosphingolipid-initiated mechanism.
Keywords: Pneumocystis, β-glucan, dendritic cells, IL-23, IL-17
Clinical Relevance
Pneumocystis pneumonia remains a serious infection in patients with impaired immunity. This study improves our understanding of how dendritic cells of the immune system respond to Pneumocystis.
Pneumocystis infection remains an all too common cause of pneumonia in immune-compromised hosts. Before the early 1990s, this infection was largely related to cases of childhood leukemia and infants with severe malnourishment. However, with the onset of the HIV pandemic, Pneumocystis pneumonia (PcP) has emerged as one of most serious causes of pneumonia among this patient population (1–3). In more recent years, an increasing number of PcP cases have been attributed to immunosuppressed individuals with malignancy, or as a result of immunosuppressive agents administered for organ transplantation or autoimmune diseases (4–10).
Earlier studies established the central importance of T cells in the host defense against Pneumocystis infection, where CD4 cell counts of less than 200 cells/mm3 were shown to place individuals at increased risk for this infection (11). More recently, CD4-independent mechanisms were also demonstrated to be important in clearing this infection (12). This is also supported by the fact that patients receiving B cell–suppressive therapy, and animal models of B-cell deficiency, also reveal a higher risk for developing PcP (13, 14). Thus, inadequate immune response across multiple components of host defense can render an individual susceptible to this infection.
In addition, exaggerated innate inflammatory responses in patients with PcP appear to be associated with a higher risk of developing respiratory failure, as indicated by early work from our laboratory showing that the degree of respiratory failure in immunosuppressed patients with PcP correlated best with the degree of inflammation, and not with the organism burden itself (15).
The innate immune system and, in particular, dendritic cells (DCs) are important orchestrators of the fungal inflammatory response, because they serve as a link between the innate and adaptive immune responses. Recent evidence indicates that DCs can prime T cells not only into a T helper–1 (Th1) and a T helper–2 (Th2) phenotype, but also into a newly identified T helper–17 (Th17) phenotype largely responsible for IL-17 and IL-22 secretion (16). This is important, because Th17 cells seem to play a major role in fungal clearance and defense (17, 18). An impaired IL-17 response was shown to be associated with the impaired clearance of Candida, Aspergillus, and Pneumocystis infections (19–21). The Th17 polarizing cytokine milieu for DCs is largely composed of IL-6, transforming growth factor (TGF)–β, and IL-23. Whereas IL-6 and TGF-β are major contributors to the initiation of Th17 responses, IL-23 seems to be responsible for the maintenance of the response (22–24). Despite observations that IL-17 is necessary for the clearance of fungi and other intracellular pathogens, an exaggerated response can also be deleterious to the host, because increased IL-17 concentrations have been associated with chronic inflammation and autoimmune disease (25–27). These data further indicate that a balanced immune response is necessary to clear these infections in an optimal manner. Accordingly, a better understanding of the host immune activation in response to fungal pathogens, and PcP in particular, will be required to develop better means to prevent and treat this infection in immunosuppressed patients.
Our laboratory has been studying host responses to Pneumocystis β-glucans (PCBG), to better define specific mechanisms by which these cell-wall components activate innate and adaptive immune systems. PCBGs are glucose homopolymers containing β-1,3 backbones with variously branched β-1,6 side chains that are present in the cell walls of certain fungi and bacteria (28–30). The main functions of β-glucans were originally believed to be related primarily to fungal cell-wall structural integrity. However, we know now that these cell-wall components are additionally able to trigger inflammatory responses in various immune and nonimmune cell types (31–37). In particular, we showed that DCs stimulated with particulate, nonsoluble PCBG are important inducers of Th1 responses, and that these responses are mainly driven by IL-1β secretion (31). However, the mechanisms by which PCBG induce Th17 responses in human DCs have not been fully characterized. To address this, we specifically evaluated the role of the dectin-1 receptor in DC stimulation leading to NF-κB activation and subsequent T-cell polarization, and further studied the role of lipid rafts in the PCBG activation of DCs. Lipid rafts are plasma membrane microdomains, enriched in cholesterol and sphingolipids that facilitate the downstream signaling of receptors that contain immunoreceptor tyrosine-based activation motif (ITAM) such as the T-cell receptor (TCR) (38–40). Because dectin-1 is one of the major receptors for β-glucan and possesses a hemi-ITAM motif, we sought to investigate the role of sphingolipid-enriched lipid rafts in PCBG-stimulated DCs.
In this report, we demonstrate that PCBG-activated DCs, in addition to polarizing T cells into a Th1 phenotype, can also induce a Th17 response that is at least partly mediated by dectin-1. Furthermore, we show that glycosphingolipids are important in facilitating the downstream signaling of these responses through the phosphorylation of dectin-1, Syk, and mitogen-activated protein kinases. Our findings also demonstrate for the first time, to the best of our knowledge, the relevance of glycosphingolipids for the PCBG activation of the human IL-23/IL-17 axis in DCs.
Materials And Methods
Reagents and Antibodies
Cells were cultured with RPMI-1640 (Sigma-Aldrich, St. Louis, MO) supplemented with 100 U/ml penicillin/streptomycin, 2 mM l-glutamine (Sigma-Aldrich), and 10% heat-inactivated FBS (Invitrogen Life Technologies, Carlsbad, CA). Pneumocystis carinii organisms were derived originally from American Type Culture Collection (Manassas, VA) stocks, and were subsequently maintained in our immunosuppressed rat colony (41). Endotoxin-free buffers and reagents were scrupulously used in all experiments. Human IL-23 p19 antibody, anti-human dectin-1 antibodies, and an IgG control were obtained from R&D Systems, Inc. (Minneapolis, MN). Anti–dectin-1 AF1859 was used for Western blot (WB) and for blocking assays, and MAB1859 was used for immunoprecipitation. The anti-phosphotyrosine antibody was obtained from Millipore (Temecula, CA). The Syk inhibitor piceatannol was from Calbiochem, Inc. (San Diego, CA). The glucosylceramide synthase inhibitor PDMP (D-threo-1-phenyl-2-decanoylamino-3-morpholino-1-propanol · HCl) was purchased from Matreya, LLC (Pleasant Gap, PA), and EDOP4 was a generous gift of Dr. Richard Pagano (Mayo Clinic, Rochester, MN). The IκBα inhibitor, BAY-11-7085, was obtained from SuperArray Bioscience Corporation (Frederick, MD). IL-23, IL-6, TGF-β, IL-1β, IL-22, and IL-17 ELISA kits were from eBiosciences, Inc. (San Diego, CA). Other reagents were obtained from Sigma-Aldrich, unless specified otherwise.
Detailed Experimental Procedures
Detailed experimental procedures are outlined in the online supplement. Those procedures include the generation of a P. carinii β-glucan enriched cell wall isolate, the generation of human DCs and CD4 cells, cytokine detection, mixed lymphocyte reactions, cellular viability assays, protein extraction and immunoblotting, immunoprecipitation, the binding of NF-κB to DNA, the incorporation and localization of fluorescent lipids in DCs, and the detection of lipids by fluorescence microscopy.
Statistical Analyses
All data are presented as the means ± SEM of multiple experimental runs, unless otherwise stated. Data were assessed for differences using the Student t test or ANOVA, with relevant posttests applied as appropriate. Statistical differences were considered significant at P < 0.05. Three asterisks denote P < 0.001, two asterisks denote P = 0.001–0.01, one asterisk denotes P = 0.01–0.05, and “ns” denotes P > 0.05. Statistical analyses were performed using GraphPad Prism, version 5 (GraphPad Software, La Jolla, CA).
Results
PCBG Induce Activation of the Human IL-23/IL-17 Axis
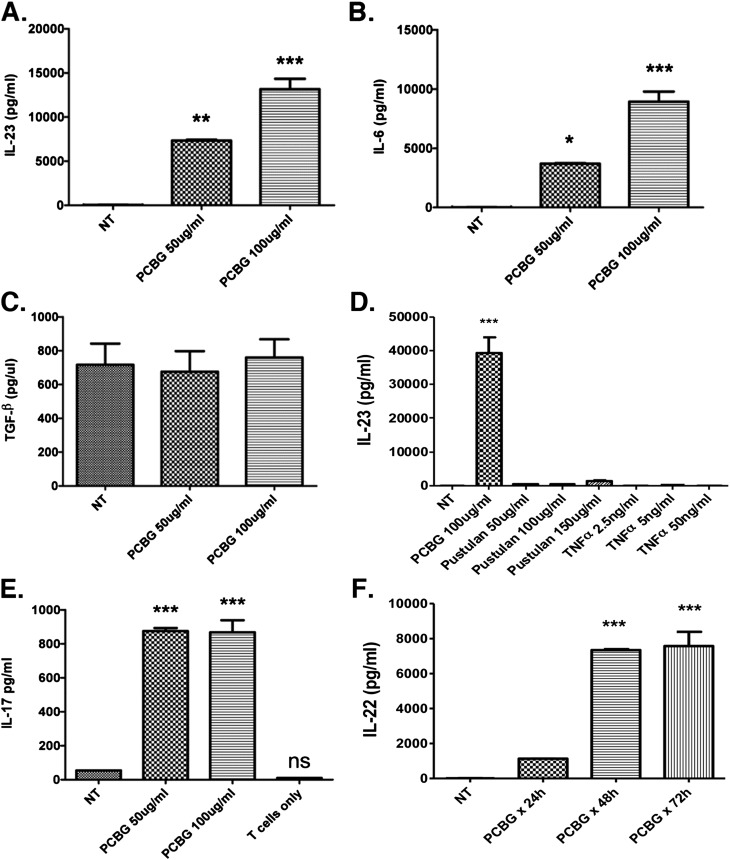
We previously demonstrated that human DCs stimulated with PCBG are potent inducers of a Th1 phenotype in the absence of IL-12 (31). Over the last several years, Th17 cells have been described as important components necessary for the clearance of fungal infections, including Pneumocystis pneumonia (19–21). We therefore investigated whether DCs produced IL-23, a key downstream inducer of Th17 responses, after stimulation with PCBG (Figure 1). Indeed, human monocyte–derived DCs stimulated with PCBG secreted IL-23 in a dose-dependent manner (Figure 1A). In addition, PCBG-stimulated DCs, even after washing off the free PCBG, were able to maintain IL-23 secretion for up to 48 hours (data not shown). Furthermore, IL-6 and TGF-β, two additional cytokines important in the initiation of Th17 responses, were also released from PCBG-stimulated DCs (Figures 1B and 1C). However, whereas IL-6 increased in a dose-dependent manner after PCBG stimulation, TGF-β (although present in DC culture supernatants) was not further induced after challenge with PCBG.
Figure 1.
Activation by β-glucan components of Pneumocystis (PCBG) of the IL-23/IL-17 axis. (A) IL-23, (B) IL-6, and (C) transforming growth factor (TGF)–β were measured by ELISA in the supernatant of PCBG-stimulated or unstimulated (NT) monocyte-derived dendritic cells (DCs). (D) As a control for specificity, IL-23 production by DCs was also measured after stimulation with the soluble β-glucan pustulan or with human TNF-α. For these experiments, PCBG served as positive control. (E and F) Next, particulate PCBG-activated and nonactivated DCs were cocultured in a mixed lymphocyte reaction with naive CD4 T cells for 72 hours, to measure subsequent PCBG-driven lymphocyte activation. The lymphocyte-derived cytokines, IL-17 (E) and IL-22 (F), were measured in the media supernatants of cell cultures, using ELISA. The data were analyzed using one-way ANOVA and posttest Bonferroni comparisons. Shown are data from a representative experimental run, typical of at least three separate experimental preparations, performed with different DC preparations. *P = 0.01–0.05. **P = 0.001–0.01. ***P < 0.001. ns, not significant (P > 0.05).
To investigate the specificity of PCBG stimulation, we further studied DCs stimulated with a soluble 1–6 β-glucan (Pustulan; Calbiochem), or with human TNF-α (Figure 1D). Notably, no significant IL-23 response was evident after either the soluble β-glucan or TNF-α stimulation. Thus, the stimulation of DCs with particulate PCBG appears to rather selectively stimulate the production of IL-23.
Next, we evaluated the production of IL-17 by CD4 cells cocultured with PCBG-activated DCs (Figures 1E and 1F). A mixed lymphocyte reaction of PCBG-activated DCs and naive CD4 cells released significantly more IL-17 than unstimulated DCs cocultured with CD4 or CD4 cells cultured alone. To assure that IL-17 was not being produced by DCs, IL-17 was measured in the supernatant of PCBG-stimulated DCs. As anticipated, no significant amounts of IL-17 were released in the absence of CD4 lymphocytes (data not shown). Because Th17 cells were shown to be potent secretors of IL-22, we investigated whether PCBG induced the IL-22 secretion of CD4 cells. As expected, PCBG-stimulated DCs were also potent inducers of IL-22 production from CD4 cells, in addition to IL-17 (Figures 1E and 1F). Taken together, these data suggest that PCBG induces a Th17 polarizing phenotype of DCs. When PCBG-stimulated DCs are cultured with naive CD4 cells, the lymphocytes are further stimulated to secrete IL-17 and IL-22 (Figures 1E and 1F).
To investigate the role of IL-23 further in the differentiation of Th17 cells, we used IL-23 antibodies to neutralize the IL-23 secretion of PCBG-stimulated DCs. Our results demonstrated that the preincubation of T cells with anti–IL-23 before coincubation with stimulated DCs, partly reduced IL-17 secretion by T cells (554 μg/ml of IL-17 in PCBG-stimulated cells without antibody, compared with 367 μg/ml in cells pretreated with neutralizing IL-23; P < 0.05). Furthermore, the physical contact of stimulated DCs with T cells was also required for IL-17 responses, because cells cultured using Transwell cell culture inserts (where T cells and DCs were not in direct contact) failed to elicit the secretion of IL-17 (data not shown).
Dectin-1 and Syk Activation Participate in PCBG-Induced IL-23 Production in DCs
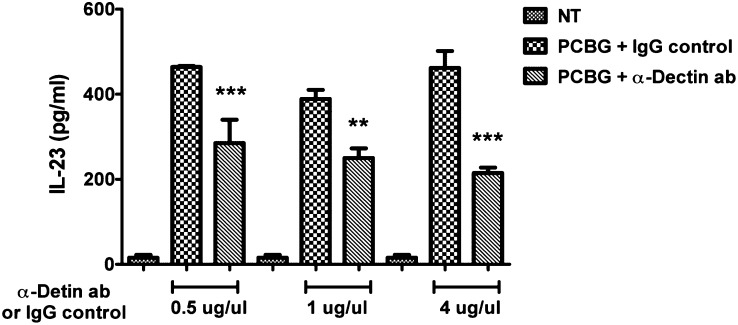
Dectin-1 is a major glucan receptor present on DCs (42, 43). Accordingly, we observed that the concentration of IL-23 was partly decreased by anti–dectin-1 antibodies compared with the IgG control (Figure 2). These results confirm a central role for dectin-1 in PCBG signaling in human DCs, polarizing responses toward the IL-23/IL-17 axis. However, the observed inhibition by anti–dectin-1 was only partial, indicating potential roles for other receptors in this process as well.
Figure 2.
Dectin-mediated IL-23 secretion by DCs. Human DCs were incubated for 1 hour with different concentrations of either anti–dectin-1 antibody (ab) or nonimmune IgG control, before stimulation with PCBG. IL-23 was measured using ELISA in the media supernatants of cells 18 hours after stimulation. The data were analyzed with one-way ANOVA and posttest Bonferroni comparisons. Shown are data from a representative experimental run, typical of at least three separate experimental preparations, conducted with different DC preparations. **P = 0.001–0.01. ***P < 0.001.
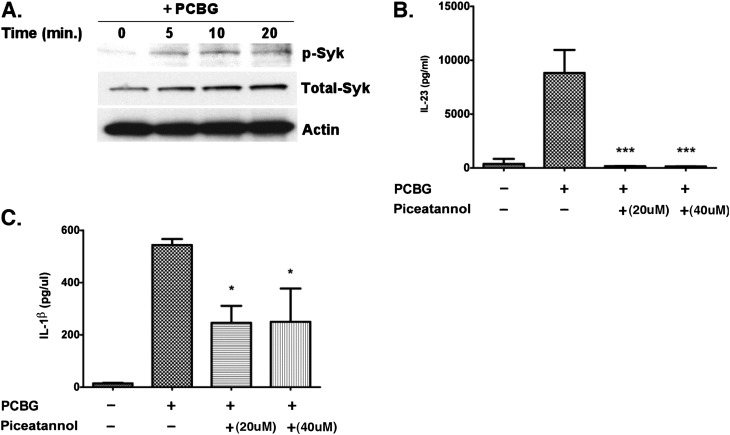
To elucidate further the downstream signaling pathways involved in PCBG–dectin-1 activation, we investigated the participation of the spleen kinase Syk after stimulation with PCBG. Syk is an upstream kinase implicated in NF-κB activation during certain dectin-1–based signaling responses (44–46). Of note, the phosphorylation of Syk was detected as early as 5 minutes after PCBG stimulation (Figure 3A). The Syk phosphorylation response was shown to decrease after 20 minutes (Figure 3A). In addition, Syk inhibition using piceatannol completely abrogated the secretion of IL-23 from PCBG-stimulated DCs, confirming the role of Syk in the PCBG activation of the IL-23/IL-17 axis (Figure 3B). We further compared the effect of the Syk inhibitor on IL-1β, and observed that although the production of IL-1β was deceased in the presence of piceatannol, the level of suppression was not as marked as that observed for IL-23 secretion, suggesting that IL-1β likely possesses additional Syk-independent regulatory mechanisms (Figure 3B). In all instances, viability assays using a colorimetric method based on the tetrazolium salt XTT confirmed that the suppressive effects were not the result of piceatannol cellular toxicity. Thus, the PCBG activation of IL-17 responses in DCs involves the Syk kinase, upstream of NF-κB.
Figure 3.
Syk-mediated activation of dendritic cell IL-23 pathway by PCBG. (A) Immunoblot analysis of phospho-Syk (p-Syk) and total Syk in unstimulated and PCBG-stimulated human DCs at indicated culture times. Total actin was used as loading control. (B and C) IL-23 and IL-1β concentrations from unstimulated or PCBG-stimulated DCs. The Syk inhibitor piceatannol was used before stimulation with PCBG for some of the conditions, as indicated. Data were analyzed with one-way ANOVA and posttest Bonferroni comparisons. Shown are data from a representative experimental run, typical of at least three separate experimental preparations, performed with different DC preparations. *P = 0.01–0.05. ***P < 0.001.
PCBG Activates Both the Canonical and Noncanonical NF-κB Pathways Leading to IL-23 Release
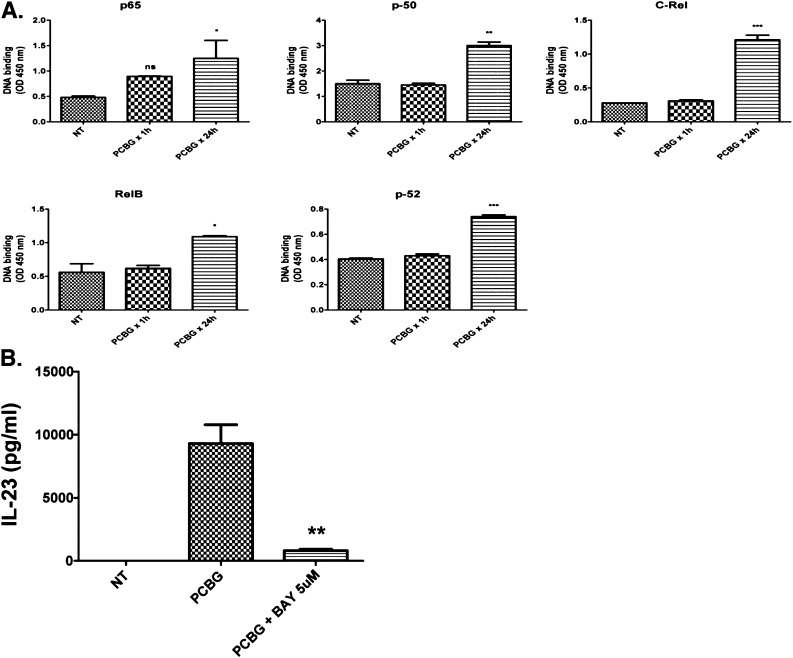
The activation of NF-κB can be achieved either by activation of the classic pathway, also known as the canonical pathway, or by activation of the noncanonical or alternative pathway (47). In activation of the classic pathway, IκBα is degraded via cellular proteosomes, and the p65 subcomponent is released and translocated into the nucleus. Within the nucleus, it forms heterodimeric complexes with other NF-κB family members that bind DNA and mediate subsequent gene transcription (48). In contrast, in the alternative or noncanonical pathway, p100 is cleaved into p52 that translocates into the nucleus. RelB, which is bound to p100, is also released by this cleavage, allowing it to be translocated into the nucleus (47, 49). The pattern of PCBG activation of NF-κB in DCs has not been completely defined. Using a DNA binding approach to detect NF-κB activation, we evaluated the PCBG induction of both NF-κB pathways (Figure 4). Interestingly, all major NF-κB subunits increased their DNA binding after the PCBG challenge of DCs, after 24 hours of steady-state stimulation. RelA, also known as p65, was the first unit to increase and was elevated after a 1-hour stimulation. However, after 24 hours of PCBG activation, all subunits had increased DNA binding in PCBG-stimulated DCs compared with unstimulated cells, indicating that both pathways were activated after PCBG stimulation (Figure 4A).
Figure 4.
PCBG activation of canonical and noncanonical NF-κB pathways in DCs leading to IL-23 secretion. (A) DNA binding of different NF-κB subunits (p65, p-50, C-Rel, RelB, and p-52) in the nuclear extracts of unstimulated and PCBG-stimulated DCs for 1 hour and 24 hours. RelB, the transcription factor protein encoded by the human RELB gene. C-Rel, the proto-oncogene protein encoded by the human REL gene. Data are expressed as optical density (OD) at 450 nm. Shown are data from a representative experimental run, typical of at least three separate experimental preparations, performed with different DC preparations. (B) The NF-κB inhibitor BAY-11-7085 was used before and throughout stimulation with PCBG, and IL-23 concentrations were measured in media supernatants by ELISA. *P = 0.01–0.05. **P = 0.001–0.01. ***P < 0.001.
To assess further the participation of NF-κB in the secretion of IL-23, we measured cytokine production in the presence of the NF-κB inhibitor BAY-11-7085, and observed that IL-23 secretion was suppressed when NF-κB activation was inhibited (Figure 4B). These results indicate that PCBG-mediated IL-23 is NF-κB–mediated. In addition, both canonical and noncanonical NF-κB pathways are activated in DCs after PCBG stimulation.
Participation of Glycosphingolipids in IL-23 Secretion
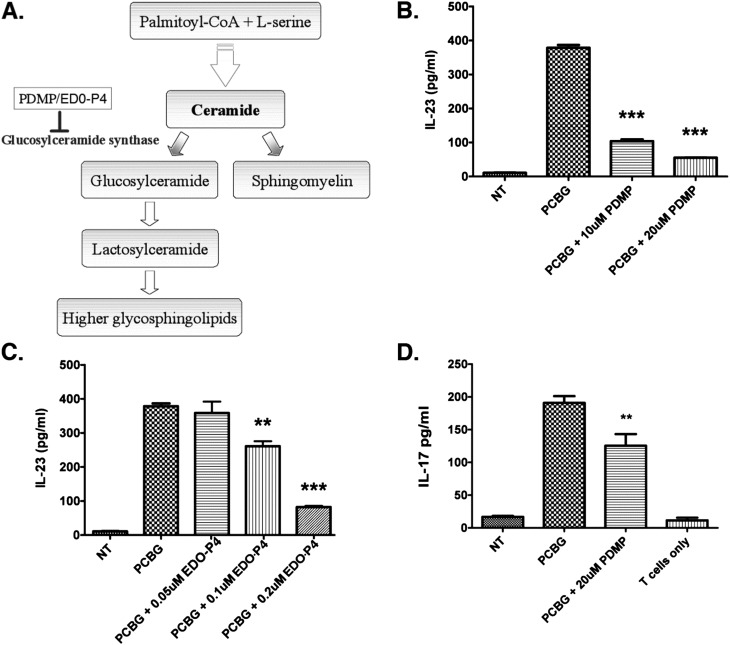
Lipid rafts are plasma membrane microdomains composed chiefly of cholesterol and sphingolipids (50). The relevance of these structures as facilitators of cell signaling and cell–cell interactions, among other functions, was only recently elucidated (51–53). Glycosphingolipids are important components of these domains, and may play a unique role in mediating these functions. We therefore investigated the activity of glycosphingolipids in PCBG signaling, and their potential contribution in the activation of the IL-23/IL-17 axis. To address this, we disrupted sphingolipid formation at the plasma membrane with PDMP, a pharmacological glycosphingolipid inhibitor that acts to inhibit glucosylceramide synthase, preventing the conversion of ceramide to glucosylceramide (Figure 5A). Although the formation of glycosphingolipids is affected by PDMP, the production of sphingomyelin is not thought to be impaired. To begin elucidating the role of glycosphingolipids in the IL-23/IL-17 axis, we first investigated whether PDMP abrogated the secretion of IL-23 from PCBG-stimulated DCs (Figure 5B). Interestingly, the presence of PDMP significantly reduced IL-23 secretion from PCBG-challenged DCs. This effect was also confirmed with EDOP4, an additional, specific sphingolipid synthesis inhibitor (Figure 5C). Moreover, we observed that CD4 cells cultured with PDMP-treated DCs also displayed substantially less IL-17 secretion, compared with nontreated DCs (Figure 5D). Together, these experiments suggest that glycosphingolipids participate in the activation of the IL-23/IL-17 axis in PCBG-stimulated DCs and CD4 lymphocytes.
Figure 5.
Glycosphingolipids are essential for optimal IL-23/IL-17 signaling. (A) Scheme of the sphingolipid biosynthesis pathway in mammalian cells from palmitoyl+coenzyme+A and L-serine. Glycosphingolipid synthesis inhibition by PDMP (D-threo-1-phenyl-2-decanoylamino-3-morpholino-1-propanol · HCl) and EDOP4 is outlined. (B) IL-23 secretion from PCBG-stimulated DCs in the presence of PDMP or (C) EDOP4. IL-23 was measured by ELISA 18 hours after stimulation in media supernatants of the cells. (D) Cells stimulated as already described were cocultured with CD4 T cells for 72 hours, and IL-17 was measured in the media supernatants of cultures. Shown are data from a representative experimental run, typical of at least three separate experimental preparations, performed with different DC preparations. **P = 0.001–0.01. ***P < 0.001.
PCBG Induce Lactosylceramide Cluster Formation at the Plasma Membrane
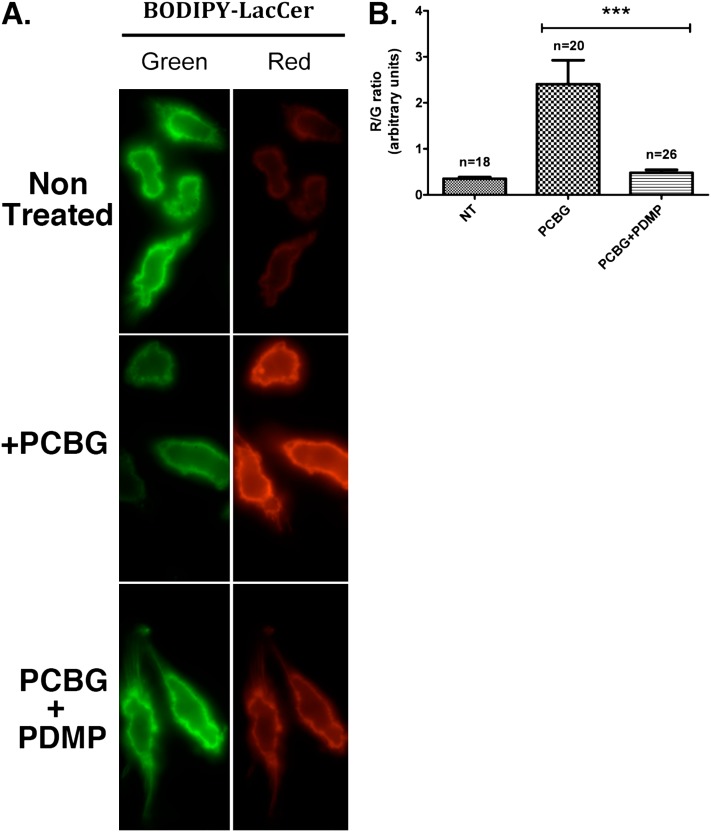
To investigate further the organization of glycosphingolipids in the plasma membrane after PCBG stimulation, we used the fluorescent sphingolipid analogue BODIPY–lactosylceramide [boron dipyrromethene (4,4-diXuoro-5,7-dimethyl-4-bora-3a,4a-diaza-s-indacene)] (BODIPY-LacCer). DCs were incubated with BODIPY-LacCer, and images were simultaneously acquired in both green and red fluorescence emission wavelengths. BODIPY-LacCer exhibits unique emission characteristics (54). Although lower concentrations of this glycosphingolipid are observed via green emission and represent monomers of the lactosylceramide, higher concentrations, as in clusters of lactosylceramide, are detected as red-emitting excimers (Figure 6). Untreated DCs exhibited a mostly green distribution of the BODIPY-LacCer tracer. In contrast, DCs stimulated with PCBG revealed a substantial shift to red emission, indicating clusters or higher concentrations of BODIPY-LacCer at the plasma membrane. Interestingly, DCs treated with the glycosphingolipid inhibitor PDMP revealed decreased clustering of BODIPY-LacCer, indicating that PDMP had abrogated the PCBG induction of sphingolipid clustering at the plasma membrane (Figure 6). To exclude the trivial possibility that the observed differences in green to red emission were attributable to differing incorporations of the BODIPY-LacCer tracer, membrane lipids were extracted under all experimental conditions and corrected for protein concentrations, and BODIPY-LacCer concentrations were measured by fluorometry, confirming equal incorporation of BODIPY-LacCer (data not shown). Thus, these data strongly indicate that PCBG induces glycosphingolipid mobilization to the plasma membrane, and that glycosphingolipid cluster formation is impaired by the glycosphingolipid inhibitor PDMP.
Figure 6.
PCBG induces changes in the distribution of BODIPY–lactosylceramide [boron dipyrromethene (4,4-diXuoro-5,7-dimethyl-4-bora-3a,4a-diaza-s-indacene)] (BODIPY-LacCer) at the plasma membrane. (A) Distribution of BODIPY-LacCer at the plasma membrane after stimulation of DCs with PCBG. A group of DCs was preincubated with 20 μM of PDMP, as already indicated. Images were acquired simultaneously at both green and red wavelengths (see Materials and Methods). The dual acquisition of images permits the identification of microdomains enriched in lactosylceramide (seen as red), as distinguished from regions with lower concentrations of lactosylceramide (seen as green). (B) Red/green (R/G) fluorescent ratios were measured. The number of cells examined is shown as “n.” Representative images from at least three independent experiments are displayed. ***P < 0.001.
Glycosphingolipid Inhibition Affects IL-23 Signaling
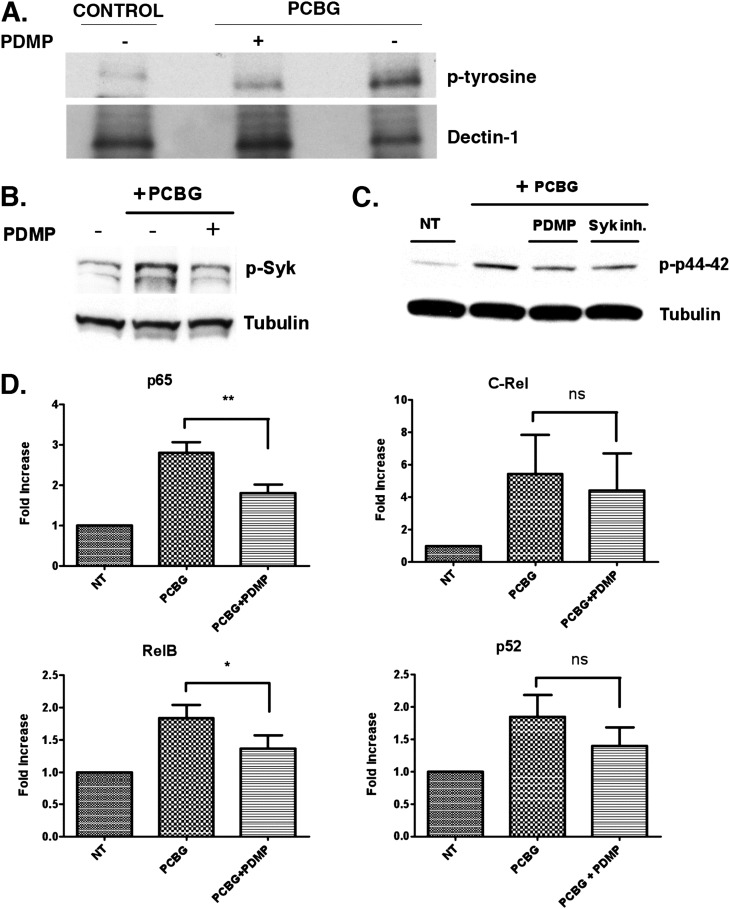
To investigate further the role of glycosphingolipid cluster formation and mobilization to the plasma membrane after PCBG stimulation, we evaluated whether PDMP, an agent that impairs glycosphingolipid synthesis and glycosphingolipid cluster formation, exerted any effect on the downstream signaling events that lead to IL-23 secretion. Because we know that dectin-1 mediates IL-23 secretion, and previous data from our laboratory showed that β-glucans induce the phosphorylation of dectin-1 (55), we investigated whether PDMP may act by affecting dectin-1 phosphorylation, to result in impaired IL-23 secretion (Figure 7). As predicted, PDMP treatment altered the phosphorylation of the dectin-1 receptor in PCBG-stimulated cells, indicating that lactosylceramide-enriched microdomains at the plasma membrane are important for dectin-1–mediated activation (Figure 7A). We observed that DC stimulation with PCBG in the presence of PDMP stimulation did induce some apparent increase in total dectin-1 protein expression, the mechanism of which is unclear. Nonetheless, the phosphorylation of dectin-1 was suppressed by the presence of PDMP.
Figure 7.
The glycosphingolipid inhibitor PDMP alters dectin-1/Syk signaling, and alters NF-κB binding. (A) Dectin-1 was immunoprecipitated from PCBG-stimulated DCs that were treated with or without PDMP (20 μM). Unstimulated cells were used as a control samples. Phosphotyrosine antibody was used to detect dectin-1 phosphorylation in the immunoprecipitates. Total dectin-1 expression was used as a control. (B and C) Immunoblot analyses of p-Syk and phospho-extracellular regulated kinase (p-ERK; p44–42) are shown after pretreatment of DCs with PDMP or piceatannol. All inhibitors were added 1 hour before and throughout subsequent stimulation. DCs were stimulated for 15 minutes with PCBG. Tubulin was used as loading control. The data shown are representative of at least three independent experiments. (D) DNA binding of the various NF-κB subunits in the nuclear extracts of unstimulated and PCBG-stimulated cells for 24 hours. Again, the PDMP was added 1 hour before and throughout the subsequent stimulation, as indicated in the experimental conditions. The data are expressed as the fold-increase, and represent the integrated results from three independent experiments. *P = 0.01–0.05. **P = 0.001–0.01. ns, not significant (P > 0.05).
We also know that the activation of Syk and NF-κB are required for IL-23 secretion. Therefore, we studied Syk phosphorylation in cells pretreated with PDMP before stimulation with PCBG, compared with control samples in the absence of PDMP. As anticipated, the phosphorylation of Syk was substantially impaired in the presence of PDMP (Figure 7B). Furthermore, the phosphorylation of p44/42 (Thr202/Tyr204) of extracellular regulated kinase (ERK), an integral signaling kinase in this pathway, was also impaired by PDMP (Figure 7C). Finally, we observed that the inhibition of Syk by piceatannol also significantly decreased ERK phosphorylation, suggesting that ERK activation occurs downstream of Syk activation after the PCBG stimulation of DCs (Figure 7C).
Because we demonstrated that IL-23 is partly dependent on NF-κB, we finally evaluated whether glycosphingolipid inhibition also affects NF-κB binding to cognate DNA sequences. Interestingly, we noticed that NF-κB binding was differentially impaired, depending of the NF-κB subunit being studied, with the p65 subunit as the main NF-κB family member most affected, whereas RelB showed only a mild reduction in DNA binding. Neither p52 nor C-Rel was significantly altered in the presence of PDMP (Figure 7D). These results suggest that the classic pathway of NF-κB, which is mainly controlled by p65, is primarily altered by glycosphingolipid disruption in our system.
Discussion
Our investigations demonstrate that cell-surface β-glucan components of Pneumocystis stimulate human DCs to secrete IL-23 and IL-6. These cytokines are well known to stimulate an IL-17 phenotype (56, 57). We further demonstrated that PCBG-stimulated human DCs interact with lymphocytes to drive IL-17 and IL-22 secretion. The activation of DCs was further shown to involve the dectin-1 receptor with Syk activation and the subsequent stimulation of both the canonical and noncanonical components of NF-κB. Finally, we learned that glycosphingolipid-rich microdomains participate in the PCBG activation of DCs. These data strongly support the idea that the β-glucan surface components of Pneumocystis drive the activation of the IL-23/IL-17 axis during this infection.
The importance of an adequate Th1 response for the clearance of Pneumocystis infection has been established (58, 59). We previously showed that human DCs stimulated with PCBG are potent inducers of some features of the Th1 phenotype, in the absence of IL-12 (31). The lack of the p35 subunit, rather than the abundant p40 subunit, was determined to be the main reason for deficiency of total IL-12. Over the past several years, Th17 cells were described as important in the immune response, mediating the clearance of fungal infections, including Pneumocystis pneumonia (19–21). Beta-glucans from bacteria and other fungi were also shown to induce Th17 responses strongly (60). TGF-β and IL-6 are also widely held to be responsible for the initiation of the Th17 response, and IL-23 is thought to play a major role in the expansion and maintenance of this response. We also found IL-6 expression in DCs to be induced by PCBG. Interestingly, IL-23 and IL-12 share the p40 subunit. However, whereas IL-12 has a p35 subunit, IL-23 possesses a unique p19 subunit. In our system, we demonstrated that PCBG-activated DCs produced large amounts of p40 in the absence of IL-12 (31). The present work indicates that this excess p40 subunit is likely diverted to the production of IL-23 by DCs after β-glucan stimulation.
Dectin-1 is a major glucan receptor on DCs and macrophages, and PCBG-induced IL-1β production by DCs can be inhibited by antibodies to dectin-1 (31, 42, 43). Using anti–dectin-1 antibodies, we observed that IL-23 was also partly decreased compared with the IgG control sample. These results establish a central role for dectin-1 in PCBG signaling in human DCs, polarizing responses toward the IL-23/IL-17 axis. However, the inhibition was only partial, suggesting that other receptors such as Toll-like receptor-2, dectin-2, or dendritic cell–specific intercellular adhesion molecule-3–grabbing nonintegrin may also participate in this process (61, 62). We further showed that the spleen kinase Syk leads to NF-κB activation and IL-23 responses after the stimulation of DCs with PCBG. A variety of other organisms have also been suggested to signal through Syk-dependent and NF-κB–dependent pathways (44–46). We continue to investigate other receptors and signaling intermediates that may be involved in this activation process.
Because β-glucans induce cytokine responses and polarize cells toward the IL-23/IL-17 axis, these naturally occurring substances may be exploitable for therapeutic benefit. Although IL-17 responses appear to be protective for Pneumocystis and other fungal infections, it remains unknown whether priming hosts toward these innate and adaptive immune responses with these noninfective cell wall components may actually ameliorate Pneumocystis pneumonia or other fungal infections. The data strongly suggest that PCBG or similar fungal cell-wall derivatives may hold promise as vaccine adjuvants or immune modulators to help prevent (or perhaps treat) these serious infections in immune-compromised hosts.
Our experiments further implicate glycosphingolipid-rich rafts (microdomains) containing lactosylceramide in the initiation of β-glucan cellular responses. Our fluorescence tracer data indicate that lactosylceramide is concentrated in the plasma membrane of DCs after stimulation with PCBG. Furthermore, the PCBG-induced concentration of lactosylceramide and subsequent cellular stimulation to release IL-23 is suppressed by the glycosphingolipid synthesis inhibitor PDMP. Our previous studies showed that agents blocking or suppressing lactosylceramide participate in the β-glucan–induced activation of macrophages and epithelial cells (36, 37, 55). Preliminary experiments in our laboratory did not support the idea that lactosylceramide acts directly as a receptor for fungal β-glucans (data not shown). However, β-glucan stimulation may act to mobilize and concentrate lactosylceramide within microdomains at the plasma membrane, and this glycosphingolipid may then act as a coreceptor to initiate inflammatory signaling, beginning at the membrane itself. Indeed, our previous work confirmed that the mere surface ligation of β-glucan is necessary for cellular activation, and that the internalization of β-glucan–rich particles is not required for this process (55).
Agents such as PDMP and EDOP4 provide unique tools to study the emerging activities of glycosphingolipids in cellular signaling events (63–65). These agents suppress glucosylceramide synthase, and thus block the subsequent generation of glycosphingolipids (64). Their activity may also affect the intracellular trafficking of glycosphingolipids and related mediators (66). Furthermore, their marked suppressive activities may in some situations exert beneficial effects during exuberant host inflammation, including activity in suppressing TNF-α and macrophage inflammatory protein-2 (36, 37). Inflammatory responses by macrophages and epithelial cells also provide important functions in Pneumocystis host defense (67, 68). However, caution must be exercised, because these agents may also suppress important DC/lymphocyte-based IL-23/IL-17 responses, which are required to clear fungal infections. Hence, additional studies will be required in whole animal model systems to understand the net benefits (or detriments) to the host with Pneumocystis and other fungal infections.
In conclusion, this study provides an enhanced understanding of the mechanisms by which β-glucans, important surface components of Pneumocystis, trigger IL-23 activation in DCs, which subsequently drive the Th17 phenotype in T-lymphocytes. These activities underlie important roles of DCs in bridging innate and adaptive immune responses in the lung during this important infection, which continues to affect increasing numbers of immune-compromised patients.
Supplementary Material
Acknowledgments
This study is dedicated to the memory of our recently departed collaborator and friend, Richard E. Pagano, Ph.D., who guided us in our studies of the roles of glycosphingolipids in innate immune activation. The authors thank Zvezdana Vuk-Pavlovic and David Marks for many helpful discussions in the planning of this work.
Footnotes
This work was supported by National Institutes of Health grants R01-HL62150 and R01-HL55934 (A.H.L.) and by the Mayo Foundation (A.H.L. and E.M.C.).
This article has an online supplement, which is accessible from this issue's table of contents at www.atsjournals.org
Originally Published in Press as DOI: 10.1165/rcmb.2011-0159OC on February 16, 2012
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Baar HS. Interstitial plasmacellular pneumonia due to Pneumocystis carinii. J Clin Pathol 1955;8:19–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fischl MA, Dickinson GM, La Voie L. Safety and efficacy of sulfamethoxazole and trimethoprim chemoprophylaxis for Pneumocystis carinii pneumonia in aids. Jama 1988;259:1185–1189 [DOI] [PubMed] [Google Scholar]
- 3.Masur H, Ognibene FP, Yarchoan R, Shelhamer JH, Baird BF, Travis W, Suffredini AF, Deyton L, Kovacs JA, Falloon J, et al. CD4 counts as predictors of opportunistic pneumonias in human immunodeficiency virus (HIV) infection. Ann Intern Med 1989;111:223–231 [DOI] [PubMed] [Google Scholar]
- 4.Dungarwalla M, Marsh JC, Tooze JA, Lucas G, Ouwehand W, Pettengell R, Dearden CE, Gordon Smith EC, Elebute MO. Lack of clinical efficacy of rituximab in the treatment of autoimmune neutropenia and pure red cell aplasia: implications for their pathophysiology. Ann Hematol 2007;86:191–197 [DOI] [PubMed] [Google Scholar]
- 5.Harigai M, Koike R, Miyasaka N. Pneumocystis pneumonia associated with infliximab in Japan. N Engl J Med 2007;357:1874–1876 [DOI] [PubMed] [Google Scholar]
- 6.Kalyoncu U, Karadag O, Akdogan A, Kisacik B, Erman M, Erguven S, Ertenli AI. Pneumocystis carinii pneumonia in a rheumatoid arthritis patient treated with adalimumab. Scand J Infect Dis 2007;39:475–478 [DOI] [PubMed] [Google Scholar]
- 7.Kaur N, Mahl TC. Pneumocystis jiroveci (carinii) pneumonia after infliximab therapy: a review of 84 cases. Dig Dis Sci 2007;52:1481–1484 [DOI] [PubMed] [Google Scholar]
- 8.Kolstad A, Holte H, Fossa A, Lauritzsen GF, Gaustad P, Torfoss D. Pneumocystis jirovecii pneumonia in B-cell lymphoma patients treated with the rituximab–choep-14 regimen. Haematologica 2007;92:139–140 [DOI] [PubMed] [Google Scholar]
- 9.Lahiff C, Khiaron OB, Nolan N, Chadwick GA. Pneumocystis carinii pneumonia in a patient on etanercept for psoriatic arthritis. Ir J Med Sci 2007;176:309–311 [DOI] [PubMed] [Google Scholar]
- 10.Yale SH, Limper AH. Pneumocystis carinii pneumonia in patients without acquired immunodeficiency syndrome: associated illness and prior corticosteroid therapy. Mayo Clin Proc 1996;71:5–13 [DOI] [PubMed] [Google Scholar]
- 11.Thomas CF, Jr, Limper AH. Pneumocystis pneumonia. N Engl J Med 2004;350:2487–2498 [DOI] [PubMed] [Google Scholar]
- 12.Zheng M, Ramsay AJ, Robichaux MB, Norris KA, Kliment C, Crowe C, Rapaka RR, Steele C, McAllister F, Shellito JE, et al. CD4+ T cell–independent DNA vaccination against opportunistic infections. J Clin Invest 2005;115:3536–3544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Carmona EM, Limper AH. Update on the diagnosis and treatment of Pneumocystis pneumonia. Ther Adv Respir Dis 2011;5:41–59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.AbuAttieh M, Rebrovich M, Wettstein PJ, Vuk-Pavlovic Z, Limper AH, Platt JL, Cascalho M. Fitness of cell-mediated immunity independent of repertoire diversity. J Immunol 2007;178:2950–2960 [DOI] [PubMed] [Google Scholar]
- 15.Limper AH, Offord KP, Smith TF, Martin WJ., Jr Pneumocystis carinii pneumonia: differences in lung parasite number and inflammation in patients with and without AIDS. Am Rev Respir Dis 1989;140:1204–1209 [DOI] [PubMed] [Google Scholar]
- 16.Aggarwal S, Ghilardi N, Xie MH, de Sauvage FJ, Gurney AL. Interleukin-23 promotes a distinct CD4 T cell activation state characterized by the production of interleukin-17. J Biol Chem 2003;278:1910–1914 [DOI] [PubMed] [Google Scholar]
- 17.Milner JD, Brenchley JM, Laurence A, Freeman AF, Hill BJ, Elias KM, Kanno Y, Spalding C, Elloumi HZ, Paulson ML, et al. Impaired T(h)17 cell differentiation in subjects with autosomal dominant hyper-IgE syndrome. Nature 2008;452:773–776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ma CS, Chew GY, Simpson N, Priyadarshi A, Wong M, Grimbacher B, Fulcher DA, Tangye SG, Cook MC. Deficiency of Th17 cells in hyper IgE syndrome due to mutations in STAT3. J Exp Med 2008;205:1551–1557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Huang W, Na L, Fidel PL, Schwarzenberger P. Requirement of interleukin-17A for systemic anti–Candida albicans host defense in mice. J Infect Dis 2004;190:624–631 [DOI] [PubMed] [Google Scholar]
- 20.Zelante T, De Luca A, Bonifazi P, Montagnoli C, Bozza S, Moretti S, Belladonna ML, Vacca C, Conte C, Mosci P, et al. IL-23 and the Th17 pathway promote inflammation and impair antifungal immune resistance. Eur J Immunol 2007;37:2695–2706 [DOI] [PubMed] [Google Scholar]
- 21.Rudner XL, Happel KI, Young EA, Shellito JE. Interleukin-23 (IL-23)–IL-17 cytokine axis in murine Pneumocystis carinii infection. Infect Immun 2007;75:3055–3061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B. TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17–producing T cells. Immunity 2006;24:179–189 [DOI] [PubMed] [Google Scholar]
- 23.Acosta-Rodriguez EV, Napolitani G, Lanzavecchia A, Sallusto F. Interleukins 1beta and 6 but not transforming growth factor–beta are essential for the differentiation of interleukin 17–producing human T helper cells. Nat Immunol 2007;8:942–949 [DOI] [PubMed] [Google Scholar]
- 24.Zhou L, Ivanov II, Spolski R, Min R, Shenderov K, Egawa T, Levy DE, Leonard WJ, Littman DR. IL-6 programs T(h)-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat Immunol 2007;8:967–974 [DOI] [PubMed] [Google Scholar]
- 25.Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol 2006;177:566–573 [DOI] [PubMed] [Google Scholar]
- 26.Nakae S, Nambu A, Sudo K, Iwakura Y. Suppression of immune induction of collagen-induced arthritis in IL-17–deficient mice. J Immunol 2003;171:6173–6177 [DOI] [PubMed] [Google Scholar]
- 27.Rohn TA, Jennings GT, Hernandez M, Grest P, Beck M, Zou Y, Kopf M, Bachmann MF. Vaccination against IL-17 suppresses autoimmune arthritis and encephalomyelitis. Eur J Immunol 2006;36:2857–2867 [DOI] [PubMed] [Google Scholar]
- 28.Duffus JH, Levi C, Manners DJ. Yeast cell-wall glucans. Adv Microb Physiol 1982;23:151–181 [DOI] [PubMed] [Google Scholar]
- 29.Kottom TJ, Limper AH. Cell wall assembly by Pneumocystis carinii. evidence for a unique GSC-1 subunit mediating beta-1,3–glucan deposition. J Biol Chem 2000;275:40628–40634 [DOI] [PubMed] [Google Scholar]
- 30.Shematek EM, Braatz JA, Cabib E. Biosynthesis of the yeast cell wall: I. Preparation and properties of beta-(1 leads to 3)glucan synthetase. J Biol Chem 1980;255:888–894 [PubMed] [Google Scholar]
- 31.Carmona EM, Vassallo R, Vuk-Pavlovic Z, Standing JE, Kottom TJ, Limper AH. Pneumocystis cell wall beta-glucans induce dendritic cell costimulatory molecule expression and inflammatory activation through a Fas–Fas ligand mechanism. J Immunol 2006;177:459–467 [DOI] [PubMed] [Google Scholar]
- 32.Lebron F, Vassallo R, Puri V, Limper AH. Pneumocystis carinii cell wall beta-glucans initiate macrophage inflammatory responses through NF-kappaB activation. J Biol Chem 2003;278:25001–25008 [DOI] [PubMed] [Google Scholar]
- 33.Vassallo R, Standing J, Limper AH. Beta-glucan from Pneumocystis carinii stimulates TNF alpha release from alveolar macrophages. J Eukaryot Microbiol 1999;46:145S. [PubMed] [Google Scholar]
- 34.Vassallo R, Standing JE, Limper AH. Isolated Pneumocystis carinii cell wall glucan provokes lower respiratory tract inflammatory responses. J Immunol 2000;164:3755–3763 [DOI] [PubMed] [Google Scholar]
- 35.Carmona EM, Lamont JD, Xue A, Wylam M, Limper AH. Pneumocystis cell wall beta-glucan stimulates calcium-dependent signaling of IL-8 secretion by human airway epithelial cells. Respir Res 2010;11:95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hahn PY, Evans SE, Kottom TJ, Standing JE, Pagano RE, Limper AH. Pneumocystis carinii cell wall beta-glucan induces release of macrophage inflammatory protein–2 from alveolar epithelial cells via a lactosylceramide-mediated mechanism. J Biol Chem 2003;278:2043–2050 [DOI] [PubMed] [Google Scholar]
- 37.Evans SE, Hahn PY, McCann F, Kottom TJ, Pavlovic ZV, Limper AH. Pneumocystis cell wall beta-glucans stimulate alveolar epithelial cell chemokine generation through nuclear factor–kappaB–dependent mechanisms. Am J Respir Cell Mol Biol 2005;32:490–497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sheets ED, Holowka D, Baird B. Membrane organization in immunoglobulin e receptor signaling. Curr Opin Chem Biol 1999;3:95–99 [DOI] [PubMed] [Google Scholar]
- 39.Janes PW, Ley SC, Magee AI, Kabouridis PS. The role of lipid rafts in T cell antigen receptor (TCR) signalling. Semin Immunol 2000;12:23–34 [DOI] [PubMed] [Google Scholar]
- 40.Cheng PC, Dykstra ML, Mitchell RN, Pierce SK. A role for lipid rafts in B cell antigen receptor signaling and antigen targeting. J Exp Med 1999;190:1549–1560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Limper AH, Hoyte JS, Standing JE. The role of alveolar macrophages in Pneumocystis carinii degradation and clearance from the lung. J Clin Invest 1997;99:2110–2117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Brown GD, Taylor PR, Reid DM, Willment JA, Williams DL, Martinez-Pomares L, Wong SY, Gordon S. Dectin-1 is a major beta-glucan receptor on macrophages. J Exp Med 2002;196:407–412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Brown GD, Gordon S. Immune recognition: a new receptor for beta-glucans. Nature 2001;413:36–37 [DOI] [PubMed] [Google Scholar]
- 44.Leibund Gut-Landmann S, Gross O, Robinson MJ, Osorio F, Slack EC, Tsoni SV, Schweighoffer E, Tybulewicz V, Brown GD, Ruland J, et al. Syk- and CARD9-dependent coupling of innate immunity to the induction of T helper cells that produce interleukin 17. Nat Immunol 2007;8:630–638 [DOI] [PubMed] [Google Scholar]
- 45.Dennehy KM, Ferwerda G, Faro-Trindade I, Pyz E, Willment JA, Taylor PR, Kerrigan A, Tsoni SV, Gordon S, Meyer-Wentrup F, et al. Syk kinase is required for collaborative cytokine production induced through dectin-1 and Toll-like receptors. Eur J Immunol 2008;38:500–506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gringhuis SI, den Dunnen J, Litjens M, van der Vlist M, Wevers B, Bruijns SC, Geijtenbeek TB. Dectin-1 directs T helper cell differentiation by controlling noncanonical NF-kappaB activation through Raf-1 and Syk. Nat Immunol 2009;10:203–213 [DOI] [PubMed] [Google Scholar]
- 47.Vallabhapurapu S, Karin M. Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol 2009;27:693–733 [DOI] [PubMed] [Google Scholar]
- 48.Barnes PJ, Karin M. Nuclear factor–kappaB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 1997;336:1066–1071 [DOI] [PubMed] [Google Scholar]
- 49.Solan NJ, Miyoshi H, Carmona EM, Bren GD, Paya CV. RELB cellular regulation and transcriptional activity are regulated by p100. J Biol Chem 2002;277:1405–1418 [DOI] [PubMed] [Google Scholar]
- 50.Simons K, Ikonen E. Functional rafts in cell membranes. Nature 1997;387:569–572 [DOI] [PubMed] [Google Scholar]
- 51.Iwabuchi K, Nagaoka I. Lactosylceramide-enriched glycosphingolipid signaling domain mediates superoxide generation from human neutrophils. Blood 2002;100:1454–1464 [PubMed] [Google Scholar]
- 52.Brown DA. Lipid rafts, detergent-resistant membranes, and raft targeting signals. Physiology (Bethesda) 2006;21:430–439 [DOI] [PubMed] [Google Scholar]
- 53.Rajendran L, Simons K. Lipid rafts and membrane dynamics. J Cell Sci 2005;118:1099–1102 [DOI] [PubMed] [Google Scholar]
- 54.Singh RD, Holicky EL, Cheng ZJ, Kim SY, Wheatley CL, Marks DL, Bittman R, Pagano RE. Inhibition of caveolar uptake, SV40 infection, and beta1-integrin signaling by a nonnatural glycosphingolipid stereoisomer. J Cell Biol 2007;176:895–901 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.McCann F, Carmona E, Puri V, Pagano RE, Limper AH. Macrophage internalization of fungal beta-glucans is not necessary for initiation of related inflammatory responses. Infect Immun 2005;73:6340–6349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sutton CE, Lalor SJ, Sweeney CM, Brereton CF, Lavelle EC, Mills KH. Interleukin-1 and IL-23 induce innate IL-17 production from gammadelta T cells, amplifying Th17 responses and autoimmunity. Immunity 2009;31:331–341 [DOI] [PubMed] [Google Scholar]
- 57.Kolls JK, Kanaly ST, Ramsay AJ. Interleukin-17: an emerging role in lung inflammation. Am J Respir Cell Mol Biol 2003;28:9–11 [DOI] [PubMed] [Google Scholar]
- 58.Wright TW, Johnston CJ, Harmsen AG, Finkelstein JN. Analysis of cytokine mRNA profiles in the lungs of Pneumocystis carinii–infected mice. Am J Respir Cell Mol Biol 1997;17:491–500 [DOI] [PubMed] [Google Scholar]
- 59.Beck JM, Harmsen AG. Lymphocytes in host defense against Pneumocystis carinii. Semin Respir Infect 1998;13:330–338 [PubMed] [Google Scholar]
- 60.Leibundgut-Landmann S, Osorio F, Brown GD, Reis e Sousa C. Stimulation of dendritic cells via the dectin-1/Syk pathway allows priming of cytotoxic T-cell responses. Blood 2008;112:4971–4980 [DOI] [PubMed] [Google Scholar]
- 61.Gringhuis SI, den Dunnen J, Litjens M, van Het Hof B, van Kooyk Y, Geijtenbeek TB. C-type lectin DC-SIGN modulates Toll-like receptor signaling via Raf-1 kinase–dependent acetylation of transcription factor NF-kappaB. Immunity 2007;26:605–616 [DOI] [PubMed] [Google Scholar]
- 62.Underhill DM. Collaboration between the innate immune receptors dectin-1, TLRS, and NODS. Immunol Rev 2007;219:75–87 [DOI] [PubMed] [Google Scholar]
- 63.Cheng ZJ, Singh RD, Wang TK, Holicky EL, Wheatley CL, Bernlohr DA, Marks DL, Pagano RE. Stimulation of GLUT4 (glucose transporter isoform 4) storage vesicle formation by sphingolipid depletion. Biochem J 2010;427:143–150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rosenwald AG, Machamer CE, Pagano RE. Effects of a sphingolipid synthesis inhibitor on membrane transport through the secretory pathway. Biochemistry 1992;31:3581–3590 [DOI] [PubMed] [Google Scholar]
- 65.Chen CS, Rosenwald AG, Pagano RE. Ceramide as a modulator of endocytosis. J Biol Chem 1995;270:13291–13297 [DOI] [PubMed] [Google Scholar]
- 66.Rosenwald AG, Pagano RE. Effects of the glucosphingolipid synthesis inhibitor, PDMP, on lysosomes in cultured cells. J Lipid Res 1994;35:1232–1240 [PubMed] [Google Scholar]
- 67.Wang J, Gigliotti F, Bhagwat SP, Maggirwar SB, Wright TW. Pneumocystis stimulates MCP-1 production by alveolar epithelial cells through a JNK-dependent mechanism. Am J Physiol Lung Cell Mol Physiol 2007;292:L1495–1505 [DOI] [PubMed] [Google Scholar]
- 68.Lasbury ME, Durant PJ, Ray CA, Tschang D, Schwendener R, Lee CH. Suppression of alveolar macrophage apoptosis prolongs survival of rats and mice with Pneumocystis pneumonia. J Immunol 2006;176:6443–6453 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.