Abstract
Objective
Type 1 diabetes (T1DM) is a pro-inflammatory state characterized by high C-reactive protein (CRP) levels. However, there is a paucity of data examining the role of CRP in promoting the pro-inflammatory state of diabetes. Thus, we examined the pro-inflammatory effects of human CRP using spontaneously diabetic bio-breeding (BB) rats.
Methods
Diabetic rats (n=9/group) were injected with Human serum albumin (huSA) or Human CRP (hCRP, 20 mg/kg body weight; i.p) for 3 consecutive days. Blood and peritoneal macrophages (MØ) were obtained following euthanasia. Peritoneal macrophages were used for measuring superoxide anion release, NF-κB DNA binding activity, proinflammatory mediator secretion.
Results
hCRP administration resulted in significantly increased superoxide anion production, along with increased release of cytokines/chemokines, and plasminogen activator inhibitor (PAI-1) and Tissue Factor (TF) activity in diabetic rats compared to huSA. hCRP-treated BB rat MØ showed significant induction of protein kinase C (PKC)-alpha, PKC-delta and p47 phox expression and NF-κB compared to huSA.
Conclusions
Thus, our data suggests that human CRP exacerbates in-vivo the pro-inflammatory, pro-oxidant and procoagulant states of diabetes predominantly via increased macrophage activity and this could have implications with respect to vascular complications and anti-inflammatory therapies.
Keywords: C-reactive protein, NF-κB, Macrophages, Tissue factor, PAI-1, PKC, p47Phox
1. Introduction
Type 1 diabetes (T1DM) confers an increased propensity to micro and macrovascular complications (Libby et al., 2005; Narayan et al., 2003; Centers for Disease Control and Prevention. National Diabetes Fact Sheet, 2005). Inflammation plays a crucial role in the pathogenesis of both microvascular and macrovascular complications of diabetes (Schram et al., 2005; Devaraj et al., 2007). C-reactive protein (CRP) is the prototypic marker of inflammation and several epidemiological studies have shown that levels in the highest quantile predict cardiovascular events (Bassuk et al., 2004; Libby & Ridker, 2004). More importantly, CRP levels have been shown to predict cardiovascular events and cardiovascular mortality in diabetic patients (Soinio et al., 2006; Schulze et al., 2004; Bruno et al., 2009).
CRP has been shown to induce pro-inflammatory effects in endothelial cells and macrophages both in-vitro and in-vivo (Devaraj et al., 2009b; Verma et al., 2006). CRP levels are increased in T1DM patients (Devaraj et al., 2007; Schalkwijk et al., 1999; Schram et al., 2005; Marcovecchio et al., 2008; Devaraj et al., 2006; Picardi et al., 2007), along with other biomarkers of inflammation, IL-1β, IL-6, TNF-α, and soluble intracellular adhesion molecule (sICAM-1) compared with healthy individuals. Previous studies have shown a significant association between increased intima-media thickness (IMT) and decreased flow mediated vasodilatation (FMD) with CRP in subjects with T1DM and recently CRP levels predicted progression of nephropathy in TIDM (Hayaishi-Okano et al., 2002; Targher et al., 2005; Colhoun et al., 2002; Hansen et al., 2010; Sundell et al., 2004; Romano et al., 2001; El Oudi et al., 2011).
The biobreeding diabetes-prone (BB-DP) rats spontaneously develop T1DM through a T-cell mediated autoimmune destruction of pancreatic β-cells (Mordes et al., 2004). The rat appears to be the preferred model to test the effects of human CRP (Griselli et al., 1999; Gill et al., 2004; Pepys et al., 2006; Devaraj et al., 2009b; Verma et al., 2006). Also, we showed that human CRP increases oxidized LDL (Ox-LDL) uptake, reactive oxygen species (ROS) generation, tissue factor release, and induces matrix metalloproteinase (MMP)-9 activity in macrophages and impairs vasoreactivity and the endothelial glycocalyx of non-diabetic Wistar rats (Hein et al, 2009; Devaraj et al, 2009a, 2009c; Singh et al, 2008). Furthermore, CRP has been shown in 2 different models to induce hypertension (Pravenec et al., 2011; Guan et al., 2009). CRP could promote vascular complications in diabetes via induction of oxidative stress and inflammation. However, it is unclear if CRP plays a role in exacerbating the inflammation and oxidative stress seen in diabetes or is merely a biomarker of inflammation. In this study, we tested the effect of human CRP in-vivo on biomediators of inflammation and oxidative stress in the spontaneously diabetic BB rat model to determine if like the non-diabetic state (Devaraj et al., 2009a; Singh et al., 2008), CRP induced increased inflammation and oxidative stress in a diabetic model since this has not been reported to date.
2. Materials and Methods
Biobreeding diabetes prone rats (males; weighing 125 –150gm) were obtained from BRM Inc (Boston, MA). BB rats were housed in a controlled sterile environment (pathogen-free conditions) at an ambient temperature of 21 ± 2 °C on a 12 h light: dark cycle. Food and water were provided ad libitum. Majority of the rats develop diabetes between 65-140 days of age (Bieg et al., 1998). Animals were weighed periodically and the diabetic condition was confirmed by measuring tail vein blood glucose levels using a commercial glucometer (Accuchek). Rats were considered diabetic when blood glucose levels exceeded 300mg/dL (non-fasting), on three consecutive readings. Animals were maintained in hyperglycemic condition (300-450 mg/dL) by subcutaneous insulin pellets (Linplant) (1-2U/day) to prevent decompensation and weight loss. The Institutional Animal Use and Care Committee at UC Davis approved all of the studies.
Two weeks after persistent hyperglycemia, human CRP (hCRP) or serum albumin (huSA control) were administered by intraperitoneal injection (20 mg/kg body weight for 3 days, n = 9 rats in each group), as previously described using the protocol of the Pepys group as we reported previously (Griselli et al., 1999; Gill et al., 2004; Pepys et al., 2006; Devaraj et al., 2009a). The rats were sacrificed on the 4th day by overdose of pentobarbital and peritoneal macrophages were isolated as described previously (Devaraj et al., 2009a) and used for further analysis. Serum levels of human CRP levels were measured using ELISA that only recognized human CRP (Hein et al., 2009; Devaraj et al., 2009a). The protocol was approved by the Animal Committee of University of California at Davis. Principles of laboratory animal care (NIH publication no. 85-23, revised 1985; http://grants1.nih.gov/grants/olaw/references/phspol.htm) were followed as well as US specific laws were adhered to.
2.1 Human CRP isolation and purification
CRP was purified from human pleural or ascites fluid by the method of Du Clos, Zlock, and Marnell (Devaraj et al., 2009a; Du Clos et al., 1991). Endotoxin was removed from CRP preparations with a Detoxigel column (Pierce Biochemicals) and found to contain <0.125 EU/ml (<12.5 pg/ml) by the Limulus assay (Biowhittaker), as described previously (Devaraj et al., 2009c). CRP protein was concentrated using Amicon concentrators (Amicon, Beverly, MA), dialyzed against Tris-buffered saline with 2 mM calcium, sterile-filtered, and stored at 4°C. None of the buffers contained sodium azide. The purity of pentameric CRP was checked by native gel electrophoresis under non-reducing conditions, yielding a single band at ~120 kDa, as described previously (Devaraj et al., 2009c; Dasu et al., 2007) and was consistently used in all experiments. All the reagents used were tested for endotoxin and found to have <0.125 EU/mL. This CRP preparation has been shown to exert pro-inflammatory effects in Toll like receptor (TLR) 4 knock down cells as described previously (Dasu et al., 2007). Also, Human serum albumin, fatty acid free and endotoxin free, was prepared in the same buffer, Tris-HCl with 2mM calcium, sterile filtered and used as protein control as described previously (Devaraj 2009a).
2.2 ELISA & Multiplex Cytokine assays
Peritoneal Macrophages were isolated following lavage with 5.0 ml warm PBS and showed F4/80 positivity (85%) by flow cytometry. We measured cytokines/chemokines in serum and peritoneal macrophage supernatant including, interleukin-1 beta (IL-1β), interleukin-6 (IL-6), interleukin-8 (KC/IL-8), Monocyte chemoattractant protein-1 (MCP-1), and tumor necrosis factor-alpha (TNF-α) using a multiplex cytokine assay (Millipore, St. Charles, MO) as described previously (Devaraj et al., 2009a, 2009c; Singh et al., 2008). Plasminogen activator inhibitor-1 (PAI-1) levels were measured using enzyme linked immunosorbent assay (ELISA) (American Diagnostica) (Devaraj et al., 2003). Intra and inter-assay coefficients of variation (CV) were determined to be <11%.
2.3 Superoxide anion release
Superoxide anion release from peritoneal macrophages was assessed by reduction of acetylated ferricytochrome c and dihydroethidium (DHE) fluorescence as described previously (Devaraj et al., 2009a).
2.4 Tissue factor (TF) activity
Peritoneal macrophages were stored in octylpyranoside to measure tissue factor activity as described earlier (Devaraj et al., 2009a). Briefly, cell lysates prepared in RIPA buffer were placed in a 96-well plate along with 10 nM FVIIa, 175nM of factor X, and incubated for 15 min at 37 °C. To measure Xa generation, 25 μL of S-2765 was added to each well and absorbance measured for 2 min at 405 nm. TF activity was calculated using relipidated TF as standard in the assay and standardized to milligram protein content.
2.5 NF-κB activity
Human CRP has been shown to induce NF-κB activity in endothelial cells, monocytes, and in non-diabetic rat peritoneal macrophages (Devaraj et al., 2009a; Devaraj et al., 2004; Liuzzo et al., 2007). Thus, in the present study, we also measured p65 NF-κB binding activity in the nuclear fractions of the peritoneal macrophages obtained from hCRP and huSA treated rats using the TransAM activity kits (Active Motif, Carlsbad, CA) as reported previously (Dasu et al., 2010).The results are expressed per milligram of cell protein of nuclear fractions.
2.6 Western Blot assays
Total protein was isolated from peritoneal macrophages obtained from treated rats to examine the effects of hCRP on PKC isoforms and p47 phox expression, as described earlier (Devaraj et al., 2005, 2009a; Dasu et al., 2007; Venugopal et al., 2002). Twenty μg of total protein isolated from peritoneal macrophages was resolved using sodium dodecyl sulphate-polyacrylamide gell electrophoresis (SDS-PAGE), transferred, and probed with antibodies for Protein kinase C (PKC) isoforms and p47phox (SantaCruz, CA), and stripped membranes were further incubated with β-actin to control for loading differences.
2.7 Statistical analysis
All assays were performed at least three times in duplicate. The comparisons between group means were analyzed using analysis of variance (ANOVA) followed by paired t-tests for parametric data and Wilcoxon signed rank tests for non-parametric data. The experimental results are presented as the mean ± standard deviation (S.D). Paired t-tests were used to compute differences in the variables, and the level of significance was set at P< 0.05.
3. Results
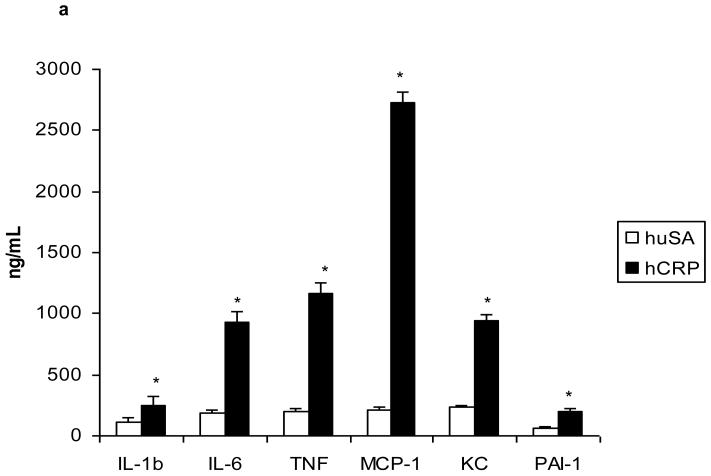
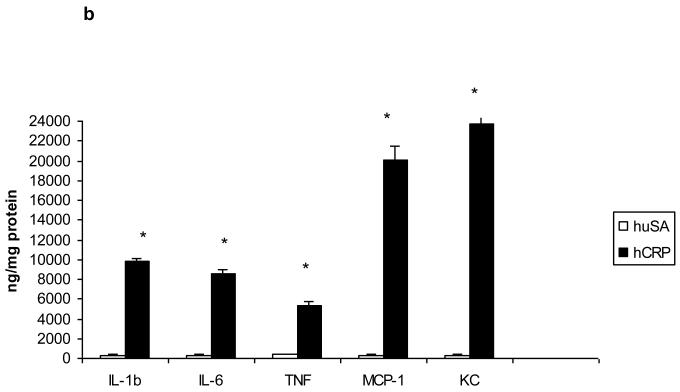
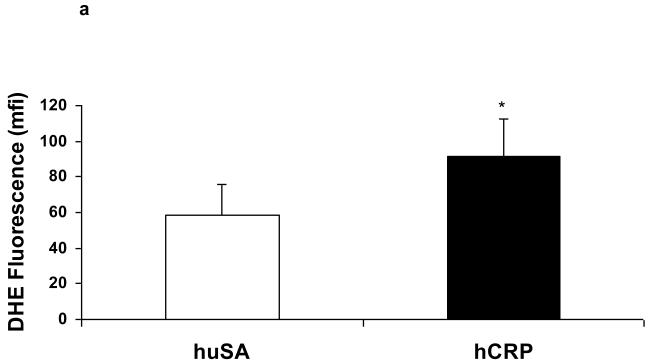
We tested the effects of hCRP administration in spontaneously diabetic BB rats. Average body weight of the BB-rat was 187± 41 g and average glucose levels was 399 ± 87 mg/dL. Administration of hCRP compared with huSA in diabetic rats led to significantly higher circulating levels of hCRP (23.7 ± 5.1 ug/mL vs. undetected, hCRP vs. huSA group, respectively; p<0.001). In the spontaneously diabetic BB rat, hCRP administration compared to hu-SA, resulted in significantly increase in biomarkers of inflammation as evidenced by increased circulating and macrophage biomediators including IL-1beta (IL-1b), TNF-alpha, MCP-1, IL-6, PAI-1 and IL-8 (KC) (Figures 1a and 1b). Also, compared to huSA, CRP stimulated an increase in macrophage superoxide anion release (Figure 2a). In addition, hCRP administration resulted in an increased TF activity (Figure 2b) and PAI-1 (Figure 2c) protein (n=9/group). Thus, using the BB rat, we show that hCRP accentuates the pro-inflammatory, pro-oxidant and prothrombotic state of diabetes also.
Fig 1a.
Circulating levels of pro-inflammatory cytokines and chemokines in sera following hCRP or huSA administration to diabetic BB rats (20mg/kg body weight for 3 days, n=9/group) were examined by multiplex assays as described in Methods. Values are expressed as ng/ml (mean ± SD). *P<0.005 vs. huSA. huSA=human serum albumin; hCRP=human C-reactive protein
Fig 1b.
Pro-inflammatory cytokines and chemokines release by peritoneal macrophages of hCRP or huSA administration to BB rats (n=9/group) were measured by Multiplex assay as described in Methods. Values are expressed as ng/mg cell protein (mean ± SD). *P<0.005 vs. huSA. huSA=human serum albumin; hCRP=human C-reactive protein
Fig 2a.
Superoxide anion release in hCRP or huSA (20 mg/kg b.w. for 3 days, n=9/group) administered by diabetic BB rat macrophages. Superoxide anion release was assessed by DHE fluorescence. Results are expressed as mean ± SD mean fluorescence intensity (MFI) *P< 0.005 vs. huSA. huSA=human serum albumin; hCRP=human C-reactive protein.
Fig 2b.
Tissue factor activity in hCRP or huSA (20 mg/kg b.w. for 3 days, n=9/group) administered by the BB rat macrophages. The rats were sacrificed on the 4th day and peritoneal macrophages were isolated and tissue factor was assessed as described in methods. Results are expressed as mean ± SD pg/mg cell protein. *P<0.005 vs. huSA. huSA=human serum albumin; hCRP=human C-reactive protein
Fig 2c.
Plasminogen activator inhibitor-1 (PAI-1) antigen in hCRP or huSA (20 mg/kg b.w. for 3 days, n=9/group) administered diabetic BB rat macrophages. The rats were sacrificed on the 4th day and peritoneal macrophages were isolated and PAI-1 levels were determined as described in methods. Results are expressed as mean ± SD pg/mg cell protein. *P<0.005 vs. huSA. huSA=human serum albumin; hCRP=human C-reactive protein.
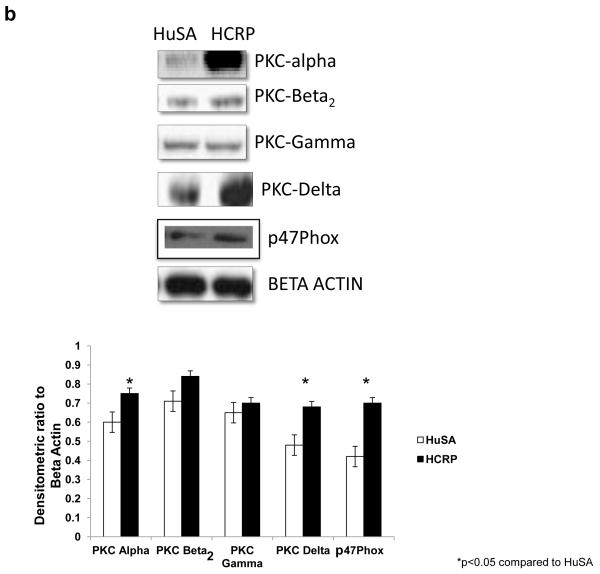
We then explored mechanisms for these effects in vivo. Previously, we have shown that hCRP induces NF-κB activity in-vivo in non-diabetic rats (Devaraj et al., 2009a; Singh et al., 2008). Thus, we tested the effect of hCRP administration compared to huSA on NF-κB activity in macrophages. As shown in Figure 3a, hCRP administration significantly increased NF-κB activity compared to huSA administration in BB rats. Furthermore, we have previously shown that PKC-alpha and beta2 and p47phox are involved in both superoxide production and cytokine release (Dasu et al., 2007; Devaraj et al., 2005; Venugopal et al., 2002). Thus, we examined PKC isoforms and p47phox expression in macrophages obtained from BB Rats that were administered hCRP or huSA. As shown in Figure 3b, in hCRP-administered BB rat macrophages, there is significant induction of PKC-alpha and PKC-delta and membrane p47phox compared to huSA.
Fig 3a.
NF-κB DNA binding activity in hCRP or huSA (20mg/kg b.w. for 3 days, n=9/group) administered diabetic BB rat macrophages. The rats were sacrificed on the 4th day and peritoneal macrophages were isolated and NF-κB DNA binding activity was assessed as described in methods. Results are expressed as mean ± SD ng/mg cell protein. *P<0.05 vs. huSA. huSA=human serum albumin; hCRP= human C-reactive protein; NF-κB=Nuclear factor-κB.
Fig 3b.
Representative Western blot depicting PKC isoforms, and p47phox protein expression in peritoneal macrophage cell lysates of hCRP or huSA (20mg/kg b.w. for 3 days, n=9/group) administered diabetic BB rats. Beta-actin was used as internal loading control.
4. Discussion
Inflammation appears to play a critical role in the pathogenesis of vascular complications, primarily cardiovascular disease (CVD). Monocyte/macrophages are recognized as key mediators of acute and chronic inflammatory responses (Moore & Tabas, 2011). Furthermore, high levels of CRP are associated with an increased risk for cardiovascular disease and increased cardiovascular events (CVE) in diabetes (Soinio et al., 2006; Schulze et al., 2004; Bruno et al., 2009). However, the role of CRP in contributing to the pro-oxidant and pro-inflammatory state of diabetes has not been studied. In the present study, we present novel data showing that hCRP exacerbates the pro-inflammatory and pro-oxidant and procoagulant states of T1DM using the BB rat model of T1DM suggesting that it is a participant in the increased risk for CVD it confers.
It is generally accepted that mice are not the optimum model to study CRP. The rat model is preferred to mice to document the pro-inflammatory and vascular effects of hCRP (Devaraj et al., 2009a, 2009b; Verma et al., 2008). hCRP has been shown to increase myocardial infarct size in a coronary artery ligation model and stroke volume following middle cerebral artery occlusion (Griselli et al., 1999; Gill et al., 2004). More recently 2 groups have independently shown that hCRP induces hypertension in rat models (Pravenec et al., 2011; Guan et al., 2009). With respect to mechanistic insights we have shown that hCRP delivery in non-diabetic rat models stimulated TF activity, NF-κB activity, ROS, MMP-9 activity, Ox-LDL uptake and cholesteryl ester accumulation, impairs vasoreactivity by uncoupling endothelial nitric oxide synthase (eNOS) perturbs the endothelial glycocalyx and promotes polarization of the macrophages to the pro-inflammatory M1 phenotype. Furthermore it appears that these effects are mediated by the FC-gamma receptors, CD32and CD64 (Hein et al., 2009; Devaraj et al., 2009a, 2009c; Singh et al., 2008; Devaraj & Jialal, 2011). Also, hCRP in rat models activate complement unlike rat CRP (Hirschfield et al., 2003). In fact, in a rat model a small molecular inhibitor to hCRP was shown to prevent the induction an increase in myocardial infarct size with hCRP (Pepys et al., 2006). Thus, several lines of evidence support and validate rat models to study in-vivo effects of hCRP. In the present study, we explored hCRPs effects in diabetes, using an established model of T1DM, the spontaneously diabetic BB rats. In this model we show pro-oxidant, pro-inflammatory and procoagulant effects with carefully purified CRP that exerts biological effects in TLR4-knock-down cells (Dasu et al., 2007; Devaraj & Jialal, 2011).
The levels of human CRP attained in diabetic rats in the present study are consistent with those attained in diabetic patients. In a recent report, Ridker’s group (Ridker & Cook, 2004) showed that CRP levels >20 μg/ml in diabetes patients predict future cardiovascular events. In the present study, CRP (a dose of 20mg/kg body weight) resulted in hCRP levels around 20 μg/ml and accentuated inflammation and oxidative stress in diabetic rats. Thus, the levels of CRP shown to induce inflammation in-vivo in this study can clearly be attained in patients especially diabetics with coronary artery disease (CAD). Therefore, it is likely that local CRP levels in diabetic patients exacerbate inflammation and oxidative stress via activation of macrophages.
There are studies demonstrating prooxidant effects of CRP in-vitro and in-vivo. Prasad’s group (Prasad, 2004) had shown that CRP dose-dependently induces superoxide anion release in neutrophils with no mechanistic details. Treatment of rat mesangial cells to CRP for 60 min led to a dose-dependent increase in superoxide anion release to control conditions (Trachtman et al., 2006). This increase in superoxide release was inhibited by co-incubation with diphenyliodinium (DPI) (10 μM). We reported that 25 μg/mL of human CRP induces superoxide anion release in Wistar rats and that this is attenuated by inhibition of PKC and apocyanin, an inhibitor of NADPH oxidase (Devaraj et al., 2009a). CRP treatment induces superoxide anion release from HAEC and uncouples eNOS; this is inhibited by siRNA to p22phox and p47phox (Singh et al., 2007). Also, Kobayashi et al. (2003) have shown that CRP colocalizes with p22phox, in lesions consistent with our data. In this report, we demonstrate that CRP induces p47phox expression and enhances superoxide anion release in diabetic rat macrophages. Thus we are extending these effects of CRP to the in-vivo diabetic state for the first time.
Tissue factor (TF) is the key trigger of the coagulation cascade and thereby crucially involved in CVD (Steffel et al., 2006; Tilley & Mackman, 2006). NF-κB/p65 is a transcription factor that regulates TF expression and studies have established increased NF-κB activity and TF release following CRP treatment in cell culture and animal studies (Devaraj et al., 2009a); however, no data is available in diabetic conditions. Our data shows increased TF activity following CRP administration in diabetic BB rats. Hence, CRP may enhance TF expression especially in the inflammatory environment of diabetes and may add to cardiovascular risk factors and advanced atherosclerotic lesions associated tissue injury. Elevated levels of soluble TF are observed in patients with atherosclerosis as compared with controls (Cimmino et al., 2011). In addition, TF expression and activity is increased by cardiovascular risk factors such as diabetes and hypertension (Cimmino et al., 2011).
Many reports have shown that PAI-1 levels and activity is increased in diabetes (Grant, 2007; Kohler & Grant, 2000). Previously, CRP has been shown to induce procoagulant activity as observed by increased endothelial PAI-1 activity and decreased tPA (Devaraj et al., 2003; Singh et al., 2005). However, it is not clear if CRP enhances PAI-1 expression in diabetic models in-vivo. Our data suggests that CRP increases both plasma and macrophage PAI-1 thereby contributing to a procoagulant phenotype. Collectively the augmentation of both TF activity and PAI-1 in-vivo supports the notion that CRP exacerbates the well defined procoagulant state of diabetes.
Recent studies have indicated the involvement of PKC and NF-κB in the pro-inflammatory state of diabetes. There is no data showing the direct effects of human CRP on PKC expression in-vitro or in-vivo. In this report, we show for the first time that CRP induces PKC-alpha and delta expression in a diabetic model leading to downstream activation of NF-κB, and consequent inflammation. In future studies, we will explore the significance of the increased expression of these PKC isoforms with CRP treatment using anti-sense technology, since PKC isoforms have been implicated in diabetic complications and inflammation. Also, we have previously shown that CRP upregulates pro-inflammatory cytokines in-vivo (Hein et al., 2009; Devaraj et al., 2009a). In the present study under diabetic conditions, we demonstrate accentuated cytokine expression with CRP treatment in-vivo.
In this report we used diabetes prone BB rats, as they are a well characterized and established rodent T1DM model. In the present study, we show that CRP enhanced inflammation and oxidative stress in diabetic rats and report that this effect is possibly mediated via PKC-alpha, PKC-delta, p47phox, and NF-κB activation. However to establish causality we need to inhibit these pathways and determine the consequences. Our observation that CRP activates the NF-κB pathway in T1DM is appealing as its activation leads to increased IL-8, IL-6, 1L-1, Intercellular Adhesion Molecule-1 (ICAM-1), vascular cell adhesion molecule (VCAM), and plasminogen activator inhibitor-1. Thus, our findings suggest that CRP may have direct effects on many biological and pathological processes relevant to diabetic vasculopathies. Additionally, a number of biological mediators, such as cytokines and reactive oxygen species have been shown to be increased in diabetic cardiovascular diseases. In this regard, although we have not made direct comparisons of the effect of CRP in non-diabetic vs. diabetic conditions, in comparison with our previous reports (Devaraj et al., 2009a; Singh et al., 2008), we observe a trend to greater exacerbations of CRPs effects in the diabetic milieu compared to the non-diabetic state using an identical protocol, with regards to release of IL-1beta (3-fold in non-DM rats vs. 10-fold in BB rats), IL-6 (4-fold in non DM rats vs. 8-fold in BB rats), PAI-1 (1.2 fold in non-DM rats vs. 1.8 fold in BB rats); however, similar increases in superoxide anion release was observed in both diabetic and non-diabetic rats with human CRP administration. To determine if human CRP has a greater effect in diabetes, in future studies ,we need to undertake direct comparisons between both diabetic and non-diabetic animals in the same experiment. Also CRP administration to humans has been shown to promote the release of pro-inflammatory mediators thus further underscoring our posit that CRP promotes a pro-inflammatory phenotype (Bisoendial et al., 2009).
To conclude, in the present study, we make the novel observation that in a spontaneous model of T1DM, CRP exacerbates the increased oxidative stress, procoagulant diathesis, and enhanced inflammation, providing proof of concept to the human epidemiological studies in diabetic patients that CRP confers worse cardiovascular outcomes. Recently it has been shown that CRP transgenic mice have severe diabetic kidney injury than wild-type mice, associated with increased renal inflammation and fibrosis and activation of nuclear factor κB signaling pathways underscoring a mediator role for CRP in diabetic microvascular complications also (Liu et al., 2011).Thus, strategies aimed at decreasing CRP in diabetics may prove to be beneficial in forestalling vascular complications.
Acknowledgements
Grant support: National Institutes of Health ROI-HL074360. We thank Dr. R. Dasu and B. Syed for helping with the care of the animals and technical aspects of this study.
Footnotes
Funded by National Institutes of Health ROI-HL074360
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Author Contribution: I. Jialal: Designed experiments and helped with manuscript preparation and final submission; H. Kaur: Helped with data analyses and manuscript preparation; S. Devaraj: Performed rat experiments and manuscript preparation.
References
- Bassuk SS, Rifai N, Ridker PM. High-sensitivity C-reactive protein: clinical importance. Curr Probl Cardiol. 2004;29:439–493. [PubMed] [Google Scholar]
- Bieg S, Koike G, Jiang J, et al. Genetic isolation of iddm 1 on chromosome 4 in the biobreeding (BB) rat. Mamm Genome. 1998;9:324–326. doi: 10.1007/s003359900759. [DOI] [PubMed] [Google Scholar]
- Bisoendial RJ, Birjmohun RS, Akdim F, et al. C - reactive protein elicits white blood cell activation in humans. Am J. Med. 2009;122(6):582e1–582e9. doi: 10.1016/j.amjmed.2008.11.032. [DOI] [PubMed] [Google Scholar]
- Bruno G, Fornengo P, Novelli G, et al. C-reactive protein and 5-year suvival in Type 2 diabetes: The Casale Monferrato study. Diabetes. 2009;58:926–933. doi: 10.2337/db08-0900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention . General Information and National Estimates on Diabetes in the United States. US Dept of Health and Human Services; Atlanta, GA: 2005. National Diabetes Fact Sheet. 2005. [Google Scholar]
- Cimmino G, Golino P, Badimon JJ. Pathophysiological role of blood-borne tissue factor: should the old paradigm be revisited? Intern Emerg Med. 2011;6:29–34. doi: 10.1007/s11739-010-0423-4. [DOI] [PubMed] [Google Scholar]
- Colhoun HM, Schalkwijk C, Rubens MB, Stehouwer CD. C-reactive protein in type 1 diabetes and its relationship to coronary artery calcification. Diabetes Care. 2002;25:1813–1817. doi: 10.2337/diacare.25.10.1813. [DOI] [PubMed] [Google Scholar]
- Dasu MR, Devaraj S, Du Clos TW, Jialal I. The biological effects of CRP are not attributable to endotoxin contamination: evidence from TLR4 knockdown human aortic endothelial cells. J Lipid Res. 2007;48:509–512. doi: 10.1194/jlr.C600020-JLR200. [DOI] [PubMed] [Google Scholar]
- Dasu MR, Devaraj S, Jialal I. High glucose induces IL-1beta expression in human monocytes: mechanistic insights. Am J Physiol Endocrinol Metab. 2007;293:E337–446. doi: 10.1152/ajpendo.00718.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dasu MR, Thangappan RK, Bourgette A, DiPietro LA, Isseroff R, Jialal I. TLR2 expression and signaling-dependent inflammation impair wound healing in diabetic mice. Lab Invest. 2010;90:1628–1636. doi: 10.1038/labinvest.2010.158. [DOI] [PubMed] [Google Scholar]
- Devaraj S, Cheung AT, Jialal I, et al. Evidence of increased inflammation and microcirculatory abnormalities in patients with type 1 diabetes and their role in microvascular complications. Diabetes. 2007;56:2790–2796. doi: 10.2337/db07-0784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaraj S, Dasu MR, Singh U, Rao LV, Jialal I. C-reactive protein stimulates superoxide anion release and tissue factor activity in vivo. Atherosclerosis. 2009a;203:67–74. doi: 10.1016/j.atherosclerosis.2008.05.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaraj S, Glaser N, Griffen S, Wang-Polagruto J, Miguelino E, Jialal I. Increased monocytic activity and biomarkers of inflammation in patients with type 1 diabetes. Diabetes. 2006;55:774–779. doi: 10.2337/diabetes.55.03.06.db05-1417. [DOI] [PubMed] [Google Scholar]
- Devaraj S, Jialal I. C-reactive protein polarizes human macrophages to an M1 phenotype and inhibits transformation to the M2 phenotype. Arterioscler Thromb Vasc Biol. 2011;31:1397–1402. doi: 10.1161/ATVBAHA.111.225508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaraj S, Kumaresan PR, Jialal I. Effect of C-reactive protein on chemokine expression in human aortic endothelial cells. J Mol Cell Cardiol. 2004;36:405–410. doi: 10.1016/j.yjmcc.2003.12.005. [DOI] [PubMed] [Google Scholar]
- Devaraj S, Singh U, Jialal I. The evolving role of C-reactive protein in atherothrombosis. Clin Chem. 2009b;55:229–38. doi: 10.1373/clinchem.2008.108886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaraj S, Tobias P, Kasinath BS, Ramsamooj R, Afify A, Jialal I. Knockout of toll-like receptor-2 attenuates both the proinflammatory state of diabetes and incipient diabetic nephropathy. Arterioscler Thromb Vasc Biol. 2011;31:1796–1804. doi: 10.1161/ATVBAHA.111.228924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaraj S, Venugopal SK, Singh U, Jialal I. Hyperglycemia induces monocytic release of interleukin-6 via induction of protein kinase c-{alpha} and - {beta} Diabetes. 2005;54:85–91. doi: 10.2337/diabetes.54.1.85. [DOI] [PubMed] [Google Scholar]
- Devaraj S, Xu DY, Jialal I. C-reactive protein increases plasminogen activator inhibitor-1 expression and activity in human aortic endothelial cells: implications for the metabolic syndrome and atherothrombosis. Circulation. 2003;107:398–404. doi: 10.1161/01.cir.0000052617.91920.fd. [DOI] [PubMed] [Google Scholar]
- Devaraj S, Yun JM, Adamson G, Galvez J, Jialal I. C-reactive protein impairs the endothelial glycocalyx resulting in endothelial dysfunction. Cardiovasc Res. 2009c;84:479–484. doi: 10.1093/cvr/cvp249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du Clos TW, Zlock LT, Marnell L. Definition of a C-reactive protein binding determinant on histones. J Biol Chem. 1991;266:2167–2171. [PubMed] [Google Scholar]
- El Oudi M, Aouni Z, Ouertani H, Mazigh C, Nsiri B, Zidi B, Machghoul S. CRP and micro-albumin: new markers of cardiovascular risk in type 2 diabetes] Ann Cardiol Angeiol (Paris) 2011;60(4):207–210. doi: 10.1016/j.ancard.2010.12.026. [DOI] [PubMed] [Google Scholar]
- Gill R, Kemp JA, Sabin C, Pepys MB. Human C-reactive protein increases cerebral infarct size after middle cerebral artery occlusion in adult rats. J Cereb Blood Flow Metab. 2004;24:1214–1218. doi: 10.1097/01.WCB.0000136517.61642.99. [DOI] [PubMed] [Google Scholar]
- Grant PJ. Diabetes mellitus as a prothrombotic condition. J Intern Med. 2007;262:157–172. doi: 10.1111/j.1365-2796.2007.01824.x. [DOI] [PubMed] [Google Scholar]
- Griselli M, Herbert J, Hutchinson WL, et al. C-reactive protein and complement are important mediators of tissue damage in acute myocardial infarction. J Exp Med. 1999;190:1733–1740. doi: 10.1084/jem.190.12.1733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan H, Wang P, Hui R, Edin ML, Zeldin DC, Wang DW. Adeno-associated virus-mediated human C-reactive protein gene delivery causes endothelial dysfunction and hypertension in rats. Clin Chem. 2009;55:274–284. doi: 10.1373/clinchem.2008.115857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen TK, Forsblom C, Saraheimo M, et al. Association between mannose-binding lectin, high-sensitivity C-reactive protein and the progression of diabetic nephropathy in type 1 diabetes. Diabetologia. 2010;53:1517–1524. doi: 10.1007/s00125-010-1742-8. [DOI] [PubMed] [Google Scholar]
- Hayaishi-Okano R, Yamasaki Y, Katakami N, et al. Elevated C-reactive protein associates with early-stage carotid atherosclerosis in young subjects with type 1 diabetes. Diabetes Care. 2002;25:1432–1438. doi: 10.2337/diacare.25.8.1432. [DOI] [PubMed] [Google Scholar]
- Hein TW, Singh U, Vasquez-Vivar J, Devaraj S, Kuo L, Jialal I. Human C-reactive protein induces endothelial dysfunction and uncoupling of eNOS in vivo. Atherosclerosis. 2009;206:61–68. doi: 10.1016/j.atherosclerosis.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirschfield GM, Pepys MB. C-reactive protein and cardiovascular disease: new insights from an old molecule. QJM. 2003;96:793–807. doi: 10.1093/qjmed/hcg134. [DOI] [PubMed] [Google Scholar]
- Jialal I, Grundy SM. Preservation of the endogenous antioxidants in low density lipoprotein by ascorbate but not probucol during oxidative modification. J. Clin. Invest. 1991;87:597–601. doi: 10.1172/JCI115035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi S, Inoue N, Ohashi Y, et al. Interaction of oxidative stress and inflammatory response in coronary plaque instability: important role of C-reactive protein. Arterioscler Thromb Vasc Biol. 2003;23:1398–1404. doi: 10.1161/01.ATV.0000081637.36475.BC. [DOI] [PubMed] [Google Scholar]
- Kohler HP, Grant PJ. Plasminogen-activator inhibitor type 1 and coronary artery disease. N Engl J Med. 2000;342:1792–1801. doi: 10.1056/NEJM200006153422406. [DOI] [PubMed] [Google Scholar]
- Libby P, Nathan DM, Abraham K, et al. National Heart, Lung, and Blood Institute, National Institute of Diabetes and Digestive and Kidney Diseases: Working Group on Cardiovascular Complications of Type 1 Diabetes Mellitus. Circulation. 2005;11:3489–3493. doi: 10.1161/CIRCULATIONAHA.104.529651. [DOI] [PubMed] [Google Scholar]
- Libby P, Ridker PM. Inflammation and atherosclerosis: role of C-reactive protein in risk assessment. Am J Med. 2004;116(Suppl 6A):9S–16S. doi: 10.1016/j.amjmed.2004.02.006. [DOI] [PubMed] [Google Scholar]
- Liu F, Chen HY, Huang XR, Chung AC, Zhou L, Fu P, Szalai AJ, Lan HY. C-reactive protein promotes diabetic kidney disease in a mouse model of type 1 diabetes. Diabetologia. 2011;54(10):2713–23. doi: 10.1007/s00125-011-2237-y. [DOI] [PubMed] [Google Scholar]
- Liuzzo GM, Santamaria LM, Biasucci M, et al. Persistent activation of nuclear factor kappa-B signaling pathway in patients with unstable angina and elevated levels of C-reactive protein: evidence for a direct proinflammatory effect of azide and lipopolysaccharide-free C-reactive protein on human monocytes via nuclear factor kappa-B activation. J Am Coll Cardiol. 2007;49:185–194. doi: 10.1016/j.jacc.2006.07.071. [DOI] [PubMed] [Google Scholar]
- Marcovecchio ML, Giannini C, Widmer B, et al. C-reactive protein in relation to the development of microalbuminuria in type 1 diabetes: the Oxford Regional Prospective Study. Diabetes Care. 2008;31:974–976. doi: 10.2337/dc07-2101. [DOI] [PubMed] [Google Scholar]
- Moore KJ, Tabas I. Macrophages in the pathogenesis of atherosclerosis. Cell. 2011;145:341–355. doi: 10.1016/j.cell.2011.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mordes JP, Bortell R, Blankenhorn EP, Rossini AA, Greiner DL. Rat models of type 1 diabetes: genetics, environment, and autoimmunity. ILAR J. 2004;45:278–291. doi: 10.1093/ilar.45.3.278. [DOI] [PubMed] [Google Scholar]
- Narayan KM, Boyle JP, Thompson TJ, et al. Lifetime risk for diabetes mellitus in the United States. JAMA. 2003;290:1884–1890. doi: 10.1001/jama.290.14.1884. [DOI] [PubMed] [Google Scholar]
- Pepys MB, Hirschfield GM, Tennent GA, et al. Targeting C-reactive protein for the treatment of cardiovascular disease. Nature. 2006;440:1217–1221. doi: 10.1038/nature04672. [DOI] [PubMed] [Google Scholar]
- Picardi A, Valorani MG, Vespasiani GU, et al. Raised C-reactive protein levels in patients with recent onset type 1 diabetes. Diabetes Metab Res Rev. 2007;23:211–214. doi: 10.1002/dmrr.671. [DOI] [PubMed] [Google Scholar]
- Prasad K. C-reactive protein increases oxygen radical generation by neutrophils. J Cardiovasc Pharmacol Ther. 2004;9:203–209. doi: 10.1177/107424840400900308. [DOI] [PubMed] [Google Scholar]
- Pravenec M, Kajiya T, Zídek V, et al. Effects of human C-reactive protein on pathogenesis of features of the metabolic syndrome. Hypertension. 2011;57:731–737. doi: 10.1161/HYPERTENSIONAHA.110.164350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridker PM, Cook N. Clinical usefulness of very high and very low levels of CRP across the full range of Framingham Risk Scores. Circulation. 2004;109:1955–1959. doi: 10.1161/01.CIR.0000125690.80303.A8. [DOI] [PubMed] [Google Scholar]
- Romano M, Pomilio M, Vigneri S. Endothelial perturbation in children and adolescents with type 1 diabetes: association with markers of the inflammatory reaction. Diabetes Care. 2001;24:1674–1678. doi: 10.2337/diacare.24.9.1674. [DOI] [PubMed] [Google Scholar]
- Sattar N. Inflammation and endothelial dysfunction: intimate companions in the pathogenesis of vascular disease? Clin Sci (Lond) 2004;106:443–445. doi: 10.1042/CS20040019. [DOI] [PubMed] [Google Scholar]
- Schalkwijk CG, Poland DC, van Dijk W, et al. Plasma concentration of C-reactive protein is increased in type I diabetic patients without clinical macroangiopathy and correlates with markers of endothelial dysfunction: evidence for chronic inflammation. Diabetologia. 1999;42:351–357. doi: 10.1007/s001250051162. [DOI] [PubMed] [Google Scholar]
- Schram MT, Chaturvedi N, Schalkwijk CG, Fuller JH, Stehouwer CD, EURODIAB Prospective Complications Study Group Markers of inflammation are cross-sectionally associated with microvascular complications and cardiovascular disease in type 1 diabetes--the EURODIAB Prospective Complications Study. Diabetologia. 2005;48:370–378. doi: 10.1007/s00125-004-1628-8. [DOI] [PubMed] [Google Scholar]
- Schram MT, Chaturvedi N, Schalkwijk CG, Fuller JH, Stehouwer CD, EURODIAB Prospective Complications Study Group Markers of inflammation are cross-sectionally associated with microvascular complications and cardiovascular disease in type 1 diabetes--the EURODIAB Prospective Complications Study. Diabetologia. 2005;48:370–378. doi: 10.1007/s00125-004-1628-8. [DOI] [PubMed] [Google Scholar]
- Schulze MB, Rimm EB, Li T, Rifai N, Stampfer MJ, Hu F. C-reactive protein and incident cardiovascular events in diabetes. Diabetes Care. 2004;27:889–894. doi: 10.2337/diacare.27.4.889. [DOI] [PubMed] [Google Scholar]
- Singh U, Dasu MR, Yancey PG, Afify A, Devaraj S, Jialal I. Human C-reactive protein promotes oxidized low density lipoprotein uptake and matrix metalloproteinase-9 release in Wistar rats. J Lipid Res. 2008;49:1015–1023. doi: 10.1194/jlr.M700535-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh U, Devaraj S, Jialal I. C-reactive protein decreases tissue plasminogen activator activity in human aortic endothelial cells: evidence that C-reactive protein is a procoagulant. Arterioscler Thromb Vasc Biol. 2005;25:2216–2221. doi: 10.1161/01.ATV.0000183718.62409.ea. [DOI] [PubMed] [Google Scholar]
- Singh U, Devaraj S, Vasquez-Vivar J, Jialal I. C-reactive protein decreases endothelial nitric oxide synthase activity via uncoupling. J Mol Cell Cardiol. 2007;43:780–791. doi: 10.1016/j.yjmcc.2007.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soinio M, Marnemi J, Laakso M, Lehto S, Ronnemaa T. High Sensitivity C-reactive protein and coronary heart disease mortality in patients with Type 2 diabetes. Diabetes Care. 2006;29:329–333. doi: 10.2337/diacare.29.02.06.dc05-1700. [DOI] [PubMed] [Google Scholar]
- Steffel J, Lüscher TF, Tanner FC. Tissue factor in cardiovascular diseases: molecular mechanisms and clinical implications. Circulation. 2006;113:722–731. doi: 10.1161/CIRCULATIONAHA.105.567297. [DOI] [PubMed] [Google Scholar]
- Sundell J, Rönnemaa T, Laine H, et al. High-sensitivity C-reactive protein and impaired coronary vasoreactivity in young men with uncomplicated type 1 diabetes. Diabetologia. 2004;47:1888–1894. doi: 10.1007/s00125-004-1543-z. [DOI] [PubMed] [Google Scholar]
- Targher G, Bertolini L, Zoppini G, Zenari L, Falezza G. Increased plasma markers of inflammation and endothelial dysfunction and their association with microvascular complications in Type 1 diabetic patients without clinically manifest macroangiopathy. Diabet Med. 2005;22:999–1004. doi: 10.1111/j.1464-5491.2005.01562.x. [DOI] [PubMed] [Google Scholar]
- Tilley R, Mackman N. Tissue factor in hemostasis and thrombosis. Semin Thromb Hemost. 2006;32:5–10. doi: 10.1055/s-2006-933335. [DOI] [PubMed] [Google Scholar]
- Trachtman H, Futterweit S, Arzberger C, et al. Nitric oxide and superoxide in rat mesangial cells: modulation by C-reactive protein. Pediatr Nephrol. 2006;21:619–626. doi: 10.1007/s00467-006-0066-x. [DOI] [PubMed] [Google Scholar]
- Venugopal SK, Devaraj S, Yang T, Jialal I. Alpha-tocopherol decreases superoxide anion release in human monocytes under hyperglycemic conditions via inhibition of protein kinase C-alpha. Diabetes. 2002;51:3049–3054. doi: 10.2337/diabetes.51.10.3049. [DOI] [PubMed] [Google Scholar]
- Verma S, Devaraj S, Jialal I. Is C-reactive protein an innocent bystander or proatherogenic culprit? C-reactive protein promotes atherothrombosis. Circulation. 2006;113:2135–50. [PubMed] [Google Scholar]