Abstract
Background
Progesterone receptor modulators (PRMs) delivered by contraceptive vaginal rings provide an opportunity for development of an estrogen-free contraceptive that does not require daily oral intake of steroids. The objective of this proof-of-concept study was to determine whether continuous delivery of 600–800 mcg of ulipristal acetate (UPA) from a contraceptive vaginal ring could achieve 80% to 90% inhibition of ovulation.
Study Design
This was a prospective, controlled, open-labeled, multicenter international trial to examine the effectiveness and safety of this prototype vaginal ring. Thirty-nine healthy women, 21–40 years old and not at risk of pregnancy, were enrolled at three clinic sites. Volunteers participated in a control cycle, a 12-week treatment period and a post-treatment cycle. Pharmacodynamic effects on follicular function and inhibition of ovulation, effects on endometrium, bleeding patterns and serum UPA levels were evaluated.
Results
Mean UPA levels during treatment were nearly constant, approximately 5.1 ng/mL throughout the study. Ovulation was documented in 32% of 111 “4-week treatment cycles.” A correlation was observed between serum UPA and degree of inhibition of ovarian activity. There was no evidence of hyperplasia of endometrium, but PRM-associated endometrial changes were frequently observed (41%).
Conclusion
In this study, the minimum effective contraceptive dose was not established. Further studies are required testing higher doses of UPA to attain ovulation suppression in a higher percentage of subjects.
Keywords: Progesterone receptor modulators, Ulipristal acetate, Contraceptive vaginal ring, Ovulation inhibition, Follicular development, PRM-associated endometrial changes
1. Introduction
Contraceptive prevalence surveys throughout the world reveal high levels of unmet needs for family planning and of unintended pregnancy [1]. Development of a wider range of safe and effective contraceptive options with user-friendly, acceptable characteristics has the potential to reduce unmet need for contraception and may bring additional health benefits to women. The study presented below indicates one promising route to such a development.
Progesterone antagonists and progesterone receptor modulators (PRMs) represent a class of progesterone ligands that may exert contraceptive action by different mechanisms. PRMs have been shown to inhibit ovulation, oppose the proliferation of the endometrium and induce amenorrhea [2–9]. In a study where the progesterone antagonist mifepristone was used orally at a low daily dose, ovulation was suppressed in over 90% of the cycles, and amenorrhea was observed in 65% to 90% of the cycles as well [10].
Several studies have evaluated the contraceptive potential of the PRM ulipristal acetate (UPA), previously known as VA-2914 or CDB-2914 [11]. UPA is a derivative of 19-norprogesterone that binds specifically to the human progesterone receptors (PRs) [12]. It is a potent, orally active modulator of the PR, known to block progesterone action in target tissues [13]. UPA is devoid of glucocorticoid activity but exhibits a modest antiglucocorticoid activity, lower than that of mifepristone [14–16]. A single dose of 10– 100 mg of UPA administered with a follicle of 14–16 mm caused a dose-dependent delay in the time interval from treatment to follicular rupture [17]. When given as emergency contraception in a single dose of 30 mg in the preovulatory phase, the luteinizing hormone (LH) peak was postponed, and follicle rupture was delayed [8]. In a study in mice subjected to gonadotrophin-induced superovulation, the administration of UPA 1 h before human chorionic gonadotrophin administration resulted in a greater than 90% inhibition of oocyte release, strongly suggesting a direct effect of the compound upon dominant follicles [7].
Two recent studies confirmed the effectiveness of UPA (30 mg) as an emergency contraceptive pill [18,19]. Another study evaluating a daily oral administration of UPA has demonstrated suppression of ovulation and amenorrhea in a majority of women [20]. This new lead appears to have the potential of being an “estrogen-free” contraceptive with possibly fewer side effects than contraceptives delivering ethinyl estradiol. It would also induce amenorrhea, which appears to be both acceptable and beneficial to many women [21].
The Population Council in collaboration with National Institute of Child Health and Human Development (NICHD) is developing a 3-month contraceptive vaginal ring (CVR) containing UPA as a method of estrogen-free hormonal contraception. Contraceptive vaginal rings offer advantages for drug delivery over those associated with oral administration. After insertion of a ring into the vagina, steroids are rapidly absorbed by vaginal tissues and pass into the systemic circulation. Vaginal drug delivery maintains relatively constant blood levels throughout ring use [22,23] and shows an increased bioavailability of steroids as compared with other routes of administration [24]. Additional benefits may result because CVRs do not require daily attention, and may thus improve compliance. Moreover, rings do not require a trained provider for insertion and removal, and they are controlled by the users [25,26].
A Phase 1 study of a CVR releasing 400–500 mcg per day of UPA (CDB-2914) was previously completed in 12 women [27]. CVR delivery of UPA permitted rapid absorption into the blood stream. UPA serum levels reached a plateau of 2–3 ng/mL within 72 h of ring insertion and remained at that level for the 1-month duration of the study. However, ovulation was suppressed in only 3 out of 12 subjects, indicating an insufficient dose of the PRM.
Based on those results, a new ring delivering a higher dose of UPA was designed and tested in a 3-month proof-of concept study. The main objective was to determine whether the continuous delivery of 600–800 mcg of UPA could achieve 80% to 90% inhibition of ovulation and induce amenorrhea in a similarly high proportion of women. The effect of this form of administration of UPA upon the endometrium was also evaluated as a key objective.
2. Method and materials
This prospective, open-labeled, three-center trial was conducted at Instituto Chileno de Medicina Reproductiva in Santiago, Chile; at USC Los Angeles, CA; and PROFAMILIA in Santo Domingo, Dominican Republic, in accordance with the guidelines of the Declaration of Helsinki. Approval was granted by the Ethics Committee of each center and by the Institutional Review Board of the Population Council (NY).
After providing informed consent, 39 healthy women, 13 at each center, were tested for normal ovulatory function and then initiated use of the ring. The subjects were 21 to 40 years old, with regular menstrual cycles of 25 to 35 days’ duration and a body mass index (BMI) <31. The women were not breastfeeding, had no contraindications to the use of oral contraceptives and were protected from pregnancy by sterilization of either partner, abstinence or consistent use of barrier methods.
The CVR studied is of a matrix design composed of micronized UPA mixed in a silicone rubber matrix. An external layer made of a silicone elastomer membrane served as a rate-limiting barrier, producing a near zero-order delivery rate of UPA. This flexible ring is 59 mm in diameter with ring body cross-section of 8.4 mm and an expected release rate of 600–800 mcg/day of UPA.
The study required a baseline control cycle to confirm normal ovulatory cycles, followed by 12 weeks of continuous ring use and a follow-up cycle to document return of ovulation. For analysis, each 4-week period of the 12 weeks of ring use was considered a “treatment cycle”; therefore, each participant contributed three treatment cycles for analysis.
2.1. Ovarian function
Ovarian function during ring use was assessed twice weekly by determination of estradiol (E2) and progesterone (P) serum concentrations and by vaginal ultrasounds. Whole blood was obtained during the morning and allowed to stand at room temperature for at least 1–2 h. The clotted whole blood was separated to obtain serum aliquot specimens of approximately 1 mL and frozen at −20°C. In Chile, estradiol and progesterone concentrations were measured by radioimmunoassay (DPC, Diagnostic Products Corporation, Los Angeles, CA, USA) with sensitivities of 21 pmol/L and 0.2 nmol/L, respectively, and interassay coefficients of variation (CVs) of 6.8%–9.3% for E2 and 5.9%–6.3% for P.
In the Dominican Republic, an electrochemiluminescence immunoassay (Roche Elecsys 2010, Mannheim, Germany) with sensitivities of 18 pmol/L and 0.1 nmol/L, respectively, and interassay CVs of 2.3%–6.2% for E2 and 3.7%–5.5% for P was used. At the Los Angeles site, estradiol was measured in serum by radioimmunoassay following extraction with ethyl acetate:hexane (3:2) [28] with an assay sensitivity of 10 pg/mL and an interassay CV of b10%. Serum progesterone levels for this site were measured by a competitive chemiluminescent immunoassay on the Immulite analyzer (Siemens Medical Solutions Diagnostics, Malvern, PA, USA) with a sensitivity of 0.2 ng/mL and an average interassay CV of 10.6%.
Transvaginal ultrasounds (TVUs) to evaluate follicular development were performed by a single individual at two sites at each semiweekly visit. In those centers, TVU was performed with a real-time scanner, Shimadzu SDU-400, using a 4–8-MHz vaginal transducer, or with a Medison Co. Ltd. SA 6000 ultrasound system, using a 7.5-MHz vaginal transducer. At the third site, semiweekly measurements of follicles were made by rotating staff members. Measurements proved frequently incompatible with preceding and succeeding follicular size. Accordingly, no follicular measurements from this site were used in this analysis.
Ovarian function was examined over five time periods: (1) a control cycle prior to ring initiation to establish eligibility; (2) weeks 1–4 of ring use (treatment cycle 1); (3) weeks 5–8 of ring use (treatment cycle 2); (4) weeks 9–12 of ring use (treatment cycle 3) and (5) a post-treatment cycle to establish return to normal menstruation. The analyses presented focus on the treatment period.
Two sets of data were used to categorize ovarian function. Treatment cycles from all three centers were classified according to the presence or absence of luteal activity, defined as P≥10 nmol/L in at least two samples.
A second ovarian function classification was employed based on both ultrasound and endocrine profiles observed in each “treatment cycle.” The following six classifications were used: (1) ovulation (follicular rupture immediately followed by an elevation of serum progesterone concentration that reached 10 nmol/L or more in at least two consecutive samples); (2) ovulatory dysfunction (a follicle ≥30 mm at rupture and preceded by progesterone levels ≥10 nmol/L); (3) luteinized unruptured follicle (LUF) (no follicular rupture but more than one P level ≥5 nmol/L); (4) persistent follicle (follicle ≥15 mm for ≥7 days without rupture or P increase); (5) no follicular development: no follicle >12 mm and (6) no follicular resolution (follicle development ≥10 mm with no resolution in the “treatment cycle”).
2.2. Serum UPA measurements
Serum levels of UPA were measured once a week during the 12 weeks of ring treatment. The serum samples were analyzed by radioimmunoassay developed at the Population Council according to the method of Larner et al. [12]. To correlate ovarian activity with UPA serum levels, mean levels of the weekly UPA samples taken during each “treatment cycle” were calculated.
2.3. Endometrial biopsies
An endometrial biopsy was taken by a Pipelle in the luteal phase of the control cycle prior to treatment (7–9 days following ovulation) and at the end of 12 weeks of treatment, when the ring was withdrawn. Endometrial biopsy specimens were fixed in formalin and processed in paraffin wax by standard methods in the local center laboratories. Paraffin-embedded tissue blocks were sent to the Edinburgh University Pathology Department in batches, and 3-μm sections were cut by standard microtomy for hematoxylin and eosin staining and for immunohistochemical assessment of proliferation markers. Preinsertion biopsies were assessed microscopically and categorized according to standard histological criteria, as described in Blaustein’s Pathology of the Female Genital Tract [29]. The end of treatment biopsies were assessed in the same way, but additional categories were used to take into account histological appearances that were evident in these biopsies. Appearances were categorized as inactive, proliferative or secretory, with a further subdivision of glandular architecture as normal or disordered. The term “disordered glandular architecture” was used to describe glands in which there was tortuosity of the normal gland structure. Recently, typical changes of the endometrium as observed here under the action of PRMs have been described as PRM-associated endometrial changes (PAECs) [30].
2.4. Proliferation markers
Immunohistochemical assessment of proliferation markers Ki-67 and phosphorylated histone protein 3 (PH3) was performed using mouse monoclonal antibody MIB-1 (Dako, Glostrup, Denmark) and Phospho-H3 (Upstate Cell Signalling Solutions, Charlottesville, VA, USA) 1:3200. Briefly, fresh 3-μm sections were dewaxed and submitted to antigen retrieval by immersion in Tris-EDTA (pH 8) and pressure cooked for 2.5 min at 1 m bar. Primary antibody was applied at 1 in 200 dilution (Ki-67) and 1 in 3200 (PH3). Negative control sections were performed in which the primary antibody was omitted. Tonsil or normal proliferative phase endometrium was used as positive control tissues. After incubation of primary antibody for 30 min, sections were washed, and the Dako EnVision-HRP visualization system was used (Dako), after which slides were counterstained with hematoxylin and mounted conventionally. For both markers, staining was assessed semiquantitatively for the three endometrial compartments (glandular epithelium, surface epithelium and stroma).
2.5. Safety
Clinical chemistries were evaluated prior to ring use and upon completion of treatment. Fasting blood samples were obtained during the screening visit, on the last day of ring use (treatment) and on the termination visit scheduled after the first spontaneous menses post ring use. Measurements included a complete clinical chemistry, complete blood count, amylase, prolactin, prothrombin time and cortisol. Urinalyses were also performed during these visits.
Diary cards were provided to subjects at the beginning of each cycle to record bleeding patterns and adverse events, if any.
2.6. Statistical methods
Each participant contributed three treatment cycles for analysis. Under the assumptions that the ring would prove 90% successful in inhibiting ovulation and 34 women would complete the study, 102 cycles would be analyzed. Under this null hypothesis, the probability of observing between 86 and 97 nonovulatory cycles (84% to 95.1%) would be 95.5%. Interpreting the null hypothesis to mean that the UPA ring would prove successful in preventing 90% of the women from ovulating during all three of their treatment cycles, one would expect to observe between 27 and 33 successes out of 34 subjects completing the study treatment. To account for possible early withdrawals and loss to followup, each clinic enrolled 13 women, a total of 39 subjects.
Data and analyses are presented only for the 37 women who completed the 12-week treatment period. Standard measures have been used for descriptive statistics. Statistical significance was set as p≤.05. Both parametric and nonparametric two-sided tests were employed as indicated in the text and tables. These include paired (t, Wilcoxon) and unpaired tests (t, Mann–Whitney), analysis of variance tests (F, Kruskal–Wallis), post hoc comparison and χ2 tests.
Additionally, we examined the sequence of UPA levels by week for each woman and computed Spearman (nonparametric) and Pearson correlation coefficients to measure the direction and magnitude of the trend for every subject.
3. Results
Thirty-seven women completed the study. Two volunteers discontinued early, citing personal reasons. The mean age and gravidity of participants who completed the study were 35.3±4.4 years (±SD) and 3.1±1.2, respectively. The mean body weight and BMI were 62.4±10.2 kg and 25.4±3.1 kg/m2, respectively. Participants from Chile were significantly older, while body weight and BMI were lowest in the Dominican Republic and highest in Los Angeles (Table 1).
Table 1.
Characteristics of subjects by site (n=37)
| Variable | Chile Mean±SD |
DR Mean±SD |
LA Mean±SD |
All sites Mean±SD |
p ANOVA |
|---|---|---|---|---|---|
| Age (years) | 38.1±1.9 | 33.8±4.0 | 33.9±5.6 | 35.3±4.4 | .0145 |
| Height (m) | 1.55±0.05 | 1.54±0.05 | 1.61±0.10 | 1.57±0.08 | .0419 |
| Weight (kg) | 61.3±6.3 | 57.6±9.5 | 69.5±11.5 | 62.4±10.2 | .0105 |
| BMI (kg/m2) | 25.4±2.4 | 24.2±3.3 | 26.7±3.5 | 25.4±3.1 | NS |
| Parity | 3.31±0.8 | 3.77±1.1 | 2.09±1.1 | 3.11±1.2 | .0008 |
One-way ANOVA: post hoc p values. Age: Chile vs. DR and LA, pb.05; parity: LA vs. Chile and DR, pb.02; weight: LA vs. DR p=.0118.
DR, Dominican Republic; LA, Los Angeles; ANOVA, analysis of variance; NS, not significant.
3.1. Ovarian activity
Table 2 summarizes the frequency of treatment periods with luteal activity for all three centers over the three successive “treatment cycles.” Luteal activity (as determined by P≥10 nmol/L in at least two samples) was observed in almost half of the 111 “treatment cycles.” There were significant differences between clinics, with the highest frequency of luteal activity reported in the Dominican Republic and the lowest in Chile. Seven women at the Santiago site and two in Los Angeles exhibited no rise in P>9.9 nmol/L in any treatment cycle. The other 28 women had elevated P levels ≥10 nmol/L at least once in the 12-week treatment period.
Table 2.
Number and percentage of cycles with luteal activity based on progesterone levels ≥10 nmol/L by clinic
| Luteal activity (hormonal data only) |
Chile % | DR % | LA % | Total % | UPA ng/mL |
|---|---|---|---|---|---|
| Yes | 28.2 | 66.7 | 54.5 | 49.5 | 4.8±1.0 |
| 11/39 | 26/39 | 18/33 | 55/111 | ||
| No | 71.8 | 33.3 | 45.5 | 50.5 | 5.5±1.6 |
| 28/39 | 13/39 | 15/33 | 56/111 | ||
| UPA levels | 5.6±1.7 | 5.1±1.0 | 4.6±1.0 | 5.1±1.4 |
Corresponding UPA levels are shown. Kruskal–Wallis ANOVA test of differences among clinics in luteal activity, p=.0026. UPA comparisons from factorial analysis by clinic and luteal activity: UPA by clinic, p=.0381; UPA by luteal activity, p=.0231.
Table 3 shows ovarian activity in six-category classification based upon both hormonal and ultrasound data at two centers during 78 “UPA treatment cycles.” Thirty-two percent of the 78 treatment periods were classified as ovulatory, while LUF occurred in 13%. Persistent follicles were observed in 31% of the periods, and there was no follicular development in 9% of the “cycles.” Significant differences in distribution of categories were observed by clinic. Follicular development occurred in all women during the study; the few cycles with no follicular development were temporary and not maintained for two consecutive treatment periods. Estradiol levels fluctuated within the normal range in all ultrasound classifications, except for lower estradiol levels (in the range of early follicular phase) observed in the no follicular development category (mean highest E2 level 178±71 pmol/L).
Table 3.
Ovarian activity classification based on both ultrasound and hormonal data in “4-week treatment cycles” in two clinics where ultrasound assessments were done by the same investigator
| Ovarian activity classification |
Clinic site |
Total |
UPA |
||
|---|---|---|---|---|---|
| Chile (n=39) |
DR (n=39) |
n (n=78) |
% | Mean ng/mL |
|
| Ovulation | 8 | 17 | 25 | 32 | 4.7 |
| Ovulatory dysfunction | 1 | 0 | 1 | 1 | 5.7 |
| LUF | 1 | 9 | 10 | 13 | 5.5 |
| Persistent follicle | 18 | 6 | 24 | 31 | 5.6 |
| No follicular resolution | 5 | 6 | 11 | 14 | 4.7 |
| No follicular development >10 mm |
6 | 1 | 7 | 9 | 7.6 |
Corresponding UPA levels are shown. Mann–Whitney test, p=.0046 (significant differences in distribution of categories by clinic); ANOVA, p=.0051 (significant differences in distribution of categories by clinic); UPA values by category, p<.0001; UPA post-hoc: No follicular development is significantly different from all other classes, p=.001.
3.2. UPA serum levels
Mean UPA levels over all cycles of treatment were nearly constant, approximately 5.1 ng/mL throughout the study (Fig. 1). Mean UPA serum levels were slightly higher in Chile, with levels ranging from 5.3 to 5.9 ng/mL per treatment cycle, whereas mean levels of 5.0 to 5.1 ng/mL were maintained at the Dominican Republic clinic. In Los Angeles, UPA concentrations decreased slightly from around 4.7 ng/mL in the first two treatment cycles to 4.4 ng/mL in the last treatment cycle (p<.02, paired tests).
Fig. 1.
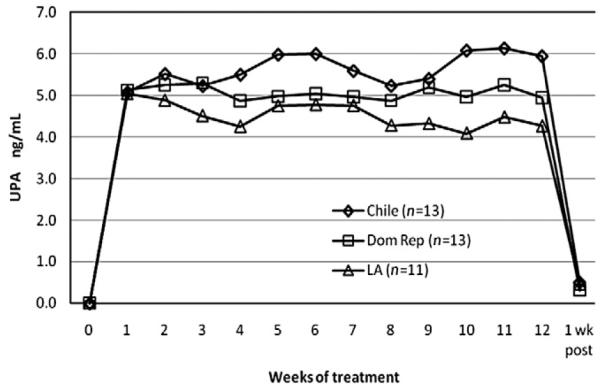
Mean UPA serum levels per site during 12 weeks of treatment.
As shown in Table 2, mean UPA levels differed significantly per clinic and were significantly lower in the cycles with luteal activity. A correlation was observed between serum UPA and the different classifications of ovarian activity based on both ultrasound and hormonal data (Table 3). As anticipated, the highest UPA levels were observed in cycles with the highest degree of inhibition of ovarian activity: no follicular development (7.6±1.3 ng/mL), while the lowest UPA levels were observed in ovulatory cycles (4.7±0.9 ng/mL). Over all categories, the differences in UPA levels by category proved significant (p<.0001).
3.3. Endometrial tissue evaluation
Histological assessment of pre- and post-treatment endometrial biopsy specimens taken from trial participants was performed by a gynecological pathologist (A.W.) with previous experience of effects of PRMs and antiprogestins on endometrium. A total of 36 pretreatment biopsies taken between days 19 and 29 of the control cycles were received. All except two specimens showed normal appearances of the secretory phase of the menstrual cycle; one was insufficient for histological assessment, and the remaining one showed appearance of chronic endometritis detected in a subject without any symptomatology and which disappeared without treatment in the biopsy taken at the end of treatment.
End of treatment biopsy specimens were obtained from 37 women after 12 weeks of continuous use of the UPA ring delivering 600–800 mcg/day. There was no evidence of hyperplasia of endometrium of either simple or complex types, and there was no evidence of cytological atypia. Cystic dilatation of glands was not commonly seen, but an inactive appearance was seen in many cases where tubular glands were widely interspersed with compact stroma. Fourteen women had end of treatment endometrial biopsies classified as “inactive,” 17 were proliferative, and 6 were secretory. However, benign glandular changes, described as PAECs, were frequently observed (41%) in a background of inactive or weakly proliferative endometrium. The percentage of endometrial biopsies that showed benign glandular changes was significantly greater among cycles with no luteal activity in the preceding 60 days (59%) as compared with those with luteal activity in the same period (25%) (Table 4).
Table 4.
Correlation of ovulatory status and presence of PAECs in the histological appearance of the endometrium after 12 weeks of treatment
| Ovulatory status | Normal glandular architecture n (%) |
PAEC benign glandular changes n (%) |
Total |
|---|---|---|---|
| Luteal activity (P>10 nmol/L) in preceding 60 days |
15 (75%) | 5 (25%) | 20 |
| No luteal activity in preceding 60 days |
7 (41%) | 10 (59%) | 17 |
| Total | 22 (59%) | 15 (41%) | 37 |
χ2 test, p=.0368; Fisher’s Exact Test one-sided, p=.0394; Fisher’s Exact Test two-sided, p=.0498.
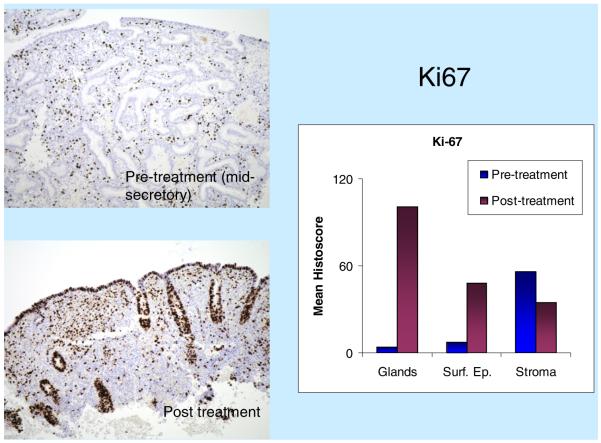
Immunohistochemical staining with proliferation marker Ki-67 showed a significant increase in the glandular epithelium post-treatment (Fig. 2). In pretreatment luteal phase biopsies, as expected, Ki-67 expression was absent from glandular epithelial cells but was expressed at relatively low levels in stromal cells. Post-treatment, there was a significant increase in Ki-67 staining in the glandular and surface epithelium, but no significant change in expression was seen in the stroma. The mitotic marker PH3 showed a significant increase in activity in glands and surface epithelium post-treatment, but not much change in the stroma.
Fig. 2.
Immunohistochemical assessment of the proliferation marker Ki-67 pre- and post-treatment of UPA. The summary chart shows mean Ki-67 histoscore, a semiquantitative score allocated to each endometrial compartment — glands, surface epithelium and stroma — according to the percentage of nuclei showing positive staining. Ki-67 gland histoscore=% of glands stained×nuclear score (NS) (NS: 0=no staining; 1=b10% of nuclei show staining in positively stained glands; 2=10%–50%; 3=N50%). Surface epithelium histoscore=% surface epithelial cells showing staining. Stroma histoscore=% stromal cells showing staining. Note that histoscore values allow comparison of Ki-67 labeling between pre- and post-treatment states, but not comparison between compartments.
3.4. Bleeding patterns
The mean number of bleeding onsets during the 12 weeks of treatment was 1.19, while the mean number of bleeding and spotting onsets was 1.41. These values are significantly below the minimum expected value of two onsets in the three 4-week periods (p<.05).
The mean days of bleeding and of bleeding and spotting in the 12 weeks of ring use for all 37 subjects were, respectively, 4.16 and 7.51. These overall values are about 33% to 50% of what one may expect over a 12-week period in normally menstruating women. The Santiago clinic had the lowest average number of days with a bleeding and/or spotting event.
Progesterone increase and drop were followed by a withdrawal bleeding with few exceptions (4/51).
3.5. Adverse events
A variety of minor conditions were reported throughout treatment; the most frequent were headache (60%), common cold symptoms (32%) and leukorrhea (19%). All of these events were considered mild or of moderate intensity. No events were considered probably related to the study drug. Leukorrhea was classified as possibly related to ring use or to high E2 levels in some subjects.
One serious adverse event (SAE), mild hyperthyroidism, was reported. However, levels of thyroid stimulating hormone, T3 and T4 were measured on a remaining aliquot of a baseline serum sample from the subject and were indicative of a preexisting subclinical hyperthyroidism at enrollment. Therefore, the SAE was not related to the study drug.
Blood pressure remained steady throughout the study, with no significant change in either systolic or diastolic blood pressure. Of more than 20 clinical chemistry and hematological parameters studied, two showed statistically significant changes in means by paired analysis, but none showed clinically relevant changes or marked increased over normal values (Table 5). Total cholesterol increased over baseline during treatment (171.6 vs. 178.9 mg/dL), while calcium levels decreased (9.1 vs. 8.9 mg/dL).
Table 5.
Paired analysis of changes in clinical chemistry
| Measure | Units | Admission |
End of treatment |
p |
|---|---|---|---|---|
| Mean±SD | ||||
| Hematocrit | % | 39.1±3.2 | 39.3±2.8 | .6117 |
| Hemoglobin | g/dL | 13.1±1.2 | 13.1±1.2 | .8404 |
| Glucose | mg/dL | 80.2±7.7 | 78.7±7.6 | .1396 |
| Uric acid | mg/dL | 3.3±0.8 | 3.5±0.8 | .0655 |
| BUN | mg/dL | 10.7±3.0 | 11.1±3.4 | .3741 |
| Creatinine | mg/dL | 0.72±0.10 | 0.7±0.1 | .2889 |
| GGT | U/L | 13.8±8.7 | 13.6±9.1 | .7874 |
| ALT/SGPT | U/L | 15.5±6.8 | 15.9±7.6 | .7661 |
| AST/SGOT | U/L | 17.8±5.2 | 19.2±5.4 | .1905 |
| LDH | U/L | 247.1±162.6 | 236.0±140.6 | .0549 |
| Alk phos | U/L | 58.8±20.0 | 58.4±18.0 | .8291 |
| T bilirubin | mg/dL | 0.6±0.3 | 0.6±0.3 | .0914 |
| Total protein | g/dL | 7.2±0.4 | 7.2±0.5 | .5377 |
| Albumin | g/dL | 4.2±0.3 | 4.2±0.3 | .4518 |
| A/G ratio | 1.4±0.2 | 1.4±0.2 | .8140 | |
| Cholesterol | mg/dL | 171.6±31.0 | 178.9±30.4 | .0355 |
| Triglyceride | mg/dL | 86.0±36.1 | 92.5±40.9 | .1676 |
| Amylase | U/L | 87.3±45.4 | 89.3±46.0 | .1721 |
| Sodium | nmol/L | 140.4±1.9 | 140.2±1.6 | .3265 |
| Potassium | nmol/L | 4.1±0.4 | 4.2±0.3 | .1123 |
| Chloride | nmol/L | 103.4±6.5 | 103.7±2.0 | .4038 |
| Calcium | mg/dL | 9.1±0.5 | 8.9±0.4 | .0027 |
4. Discussion
The current study tested the effects on ovarian activity and menstrual bleeding of 600–800 mcg/day of UPA released from a newly formulated vaginal ring. Ovulation, as judged by ultrasound and hormonal data, was observed in one third of the treatment periods. Thus, the present ring formulation seems more effective for preventing ovulation than previously tested rings releasing lower dosages of UPA, but is not yet the fully effective dose. This is in agreement with UPA serum levels attained with this ring which were around 5 ng/mL, with a higher rate of ovulation suppression when levels reached or surpassed 7 ng/mL.
The observation of higher UPA levels among Chilean women cannot be explained by a difference in BMI, as BMI was similar in all three clinics. We cannot completely exclude the possibility of difference in compliance between centers. Although women were instructed to not remove the ring during the study, this might have occurred. However, the UPA levels per participant at each center were very constant during the study, and the presence of the ring in the vagina was assessed twice a week at each visit. If women were inserting the ring only for their visits, a much larger variability in UPA serum levels would have been observed. The possibility of a random difference cannot be excluded.
By comparing the rate of ovulation suppression attained with rings differing in their rate of release of UPA or by comparing the same ring tested in different women, a good dose–response relationship becomes quite apparent. A correlation was observed between UPA serum levels and the degree of ovulation inhibition. The highest UPA levels were observed in cycles with no follicular development, while the lowest levels were observed in ovulatory cycles. With oral doses, ovulation suppression in 80% of subjects has been observed with serum levels above 10 ng/mL [20]. Animal model studies have demonstrated a dose–response relationship between UPA and ovulation suppression [31]. A similar trend has been observed with oral doses administered to healthy female volunteers [17,20]. In any event, the potential for delivering UPA by a vaginal ring has been demonstrated, although its use for contraception will require higher delivery rates of the PRM in the ring design.
All periods characterized by persistent follicle and LUF had elevated E2 circulating levels typically associated with a growing dominant follicle destined to ovulate. This suggests normal gonadotropin stimulation during the follicular phase. The mechanism by which UPA prevented follicular rupture in 67% of cycles may be a direct effect of this PRM upon the ovary, as demonstrated in rodents deprived of the PR [7] or by blocking the onset of the LH surge. When given as emergency contraception in a single dose of 30 mg in the preovulatory phase, the LH peak was postponed, and follicle rupture was delayed [8]. Low daily doses of the antiprogestin mifepristone were shown to suppress the preovulatory surge of LH [10]. Chabbert-Buffet et al. [20] report little effect on gonadotropin with the PRM oral UPA at doses up to 10 mg, but they took twice a week blood sampling, which is not sufficient to detect the LH surge.
The endometrial changes were in line with those described with other PRMs [30]. In April 2006, an NICHD workshop convened a group of pathologists to define the histological appearances of PRM-treated endometrium and to develop new terminology [32]. Overall, a ‘class effect’ was noted, designated as PAECs [30]. There was no evidence of hyperplasia or cytological atypia in any of the endometrial biopsies. Expression of proliferation markers Ki-67 and PH3 was significantly increased in post-treatment endometrial glandular epithelium compared to baseline, but baseline biopsies were taken during the secretory phase, when proliferative activity is at very low levels.
Headache, common cold symptoms and leukorrhea were reported most frequently. No significant changes in systolic or diastolic blood pressure or biochemical and hematological parameters were reported. This is consistent with the findings reported in other studies [20,27].
In the current study, the minimum effective contraceptive dose was not established, and only the women with serum levels around 6–7 ng/mL experienced ovulation suppression. In those women, the changes of the endometrium reflected the effect of the PRM without the presence of P. Further studies are required testing higher doses of UPA to attain ovulation suppression in a higher percentage of subjects. Long-term follow-up studies of the endometrium are warranted.
Following the Woman’s Health Initiative study publication, it was suggested that the use of some progestins may increase the risk of breast cancer [33]. Later on, it was shown that various synthetic progestins combined with estrogen increased the risk of breast cancer, while the use of natural P did not [34]. Therefore, it has been suggested that the use of a PRM inhibiting the PR might also suppress proliferation of mammary gland cells and possibly decrease the risk for breast disease. Although this hypothesis remains to be demonstrated, the possibility of adding a major health benefit to a new contraceptive method would be a welcome addition [35].
Given the good tolerability of PRMs, and their capacity to inhibit ovulation without suppression of the endogenous E2 secretion, this category of compounds represents a novel and promising approach to contraception without synthetic estrogens.
Acknowledgments
This study was supported by a grant from the National Institute of Child Health and Human Development of the National Institutes of Health (grant number U54 HD 29990). The authors would like to thank HRA Pharma, Paris, France, for supplying ulipristal acetate for the study.
References
- [1].Cleland J, Bernstein S, Ezeh A, Faundes A, Glasier A, Innis J. Family planning: the unfinished agenda. Lancet. 2006;18:1810–27. doi: 10.1016/S0140-6736(06)69480-4. [DOI] [PubMed] [Google Scholar]
- [2].Spitz IM, Croxatto HB, Robbins A. Antiprogestins: mechanism of action and contraceptive potential. Annu Rev Pharmacol Toxicol. 1996;36:47–81. doi: 10.1146/annurev.pa.36.040196.000403. [DOI] [PubMed] [Google Scholar]
- [3].Croxatto HB, Kovacs L, Massai R, et al. Effects of long-term low-dose mifepristone on reproductive function in women. Hum Reprod. 1998;13:793–8. doi: 10.1093/humrep/13.4.793. [DOI] [PubMed] [Google Scholar]
- [4].Chabbert-Buffet N, Meduri G, Bouchard P, Spitz IM. Selective progesterone receptor modulators and progesterone antagonists: mechanisms of action and clinical applications. Hum Reprod Update. 2005;11:293–307. doi: 10.1093/humupd/dmi002. [DOI] [PubMed] [Google Scholar]
- [5].Brenner RM, Slayden OD. Progesterone receptor antagonists and the endometrial antiproliferative effect. Semin Reprod Med. 2005;23:74–81. doi: 10.1055/s-2005-864035. [DOI] [PubMed] [Google Scholar]
- [6].Chwalisz K, Brenner RM, Fuhrmann UU, Hess-Stumpp H, Elger W. Antiproliferative effects of progesterone antagonists and progesterone receptor modulators on the endometrium. Steroids. 2000;65:741–51. doi: 10.1016/s0039-128x(00)00190-2. [DOI] [PubMed] [Google Scholar]
- [7].Palanisamy GS, Cheon YP, Kim J, et al. A novel pathway involving progesterone receptor, endothelin-2, and endothelin receptor B controls ovulation in mice. Mol Endocrinol. 2006;20:2784–95. doi: 10.1210/me.2006-0093. [DOI] [PubMed] [Google Scholar]
- [8].Brache V, Cochon L, Jesam C, et al. Immediate pre-ovulatory administration of 30 mg. ulipristal acetate significantly delays follicular rupture. Hum Reprod. 2010;25:2256–63. doi: 10.1093/humrep/deq157. [DOI] [PubMed] [Google Scholar]
- [9].Chwalisz K, Elger W, Stickler T, Mattia-Goldberg C, Larsen L. The effects of 1-month administration of asoprisnil (J867), a selective progesterone receptor modulator, in healthy premenopausal women. Hum Reprod. 2005;20:1090–9. doi: 10.1093/humrep/deh738. [DOI] [PubMed] [Google Scholar]
- [10].Brown A, Cheng L, Lin S, Baird D. Daily dose mifepristone has contraceptive potential suppressing ovulation and menstruation; a double-blind randomized control trial of 2 and 5 mg per day for 120 days. J Clin Endocrinol Metab. 2002;87:63–70. doi: 10.1210/jcem.87.1.8140. [DOI] [PubMed] [Google Scholar]
- [11].Blithe DL, Nieman LK, Blye RP, Stratton P, Passaro M. Development of the selective progesterone receptor modulator CDB-2914 for clinical indications. Steroids. 2003;68:1013–7. doi: 10.1016/s0039-128x(03)00118-1. [DOI] [PubMed] [Google Scholar]
- [12].Larner JM, Reel JR, Blye RP. Circulating concentrations of antiprogestins CDB-2914 and mifepristone in female rhesus monkey following various routes of administration. Hum Reprod. 2000;15:1100–6. doi: 10.1093/humrep/15.5.1100. [DOI] [PubMed] [Google Scholar]
- [13].Cook CE, Wani MC, Lee YW, Fail PA, Petrow V. Reversal of activity profile in analogs of the antiprogestin RU 486: effect of a 16α-substituent on progestational (agonist) activity. Life Sci. 1992;52:155–62. [PubMed] [Google Scholar]
- [14].Hild SA, Reel JR, Hoffman LH, Blye RP. CDB-2914: anti-progestational/anti-glucocorticoid profile and post coital antifertility activity in rats and rabbits. Hum Reprod. 2000;15:822–9. doi: 10.1093/humrep/15.4.822. [DOI] [PubMed] [Google Scholar]
- [15].Attardi BJ, Burgenson J, Hild SA, Reel JR, Blye RP. CDB-4124 and its putative monodemethylated metabolite, CDB-4453, are potent anti-progestins with reduced antiglucocorticoid activity: in vitro comparison to mifepristone and CDB-2914. Mol Cell Endocrinol. 2002;188:111–23. doi: 10.1016/s0303-7207(01)00743-2. [DOI] [PubMed] [Google Scholar]
- [16].Attardi BJ, Burgenson J, Hild SA, Reel JR. In vitro antiprogestational/ antiglucocorticoid activity and progestin and glucocorticoid receptor binding of the putative metabolites and synthetic derivatives of CDB-2914, CDB-4124, and mifepristone. J Steroid Biochem Mol Biol. 2004;88:277–88. doi: 10.1016/j.jsbmb.2003.12.004. [DOI] [PubMed] [Google Scholar]
- [17].Stratton P, Hartog B, Hajizadeh N, et al. A single mid-follicular dose of CDB2914, a new antiprogestin, inhibits folliculogenesis and endometrial differentiation in normally cycling women. Hum Reprod. 2000;15:1092–9. doi: 10.1093/humrep/15.5.1092. [DOI] [PubMed] [Google Scholar]
- [18].Glasier AF, Cameron ST, Fine PM, et al. Ulipristal acetate versus levonorgestrel for emergency contraception: a randomized non-inferiority trial and meta-analysis. Lancet. 2010;375:555–62. doi: 10.1016/S0140-6736(10)60101-8. [DOI] [PubMed] [Google Scholar]
- [19].Fine P, Mathé H, Ginde S, Cullins V, Morfesis J, Gainer E. Ulipristal acetate taken 48-120 hours after intercourse for emergency contraception. Obstet Gynecol. 2010;115:257–63. doi: 10.1097/AOG.0b013e3181c8e2aa. [DOI] [PubMed] [Google Scholar]
- [20].Chabbert-Buffet N, Pintiaux-Kairis A, Bouchard P. Effects of the progesterone receptor modulator VA2914 in a continuous low dose on the hypothalamic–pituitary–ovarian axis and endometrium in normal women: a prospective, randomized, placebo-controlled trial. J Clin Endocrinol Metab. 2007;92:3582–9. doi: 10.1210/jc.2006-2816. [DOI] [PubMed] [Google Scholar]
- [21].Glasier AF, Smith KB, van der Spuy ZM, et al. Amenorrhea associated with contraception an international study on acceptability. Contraception. 2003;67:1–8. doi: 10.1016/s0010-7824(02)00474-2. [DOI] [PubMed] [Google Scholar]
- [22].Alvarez-Sanchez F, Brache V, Jackanicz T, Faundes A. Evaluation of four different contraceptive vaginal rings: steroid serum levels, luteal activity, bleeding control, and lipid profiles. Contraception. 1992;46:387–98. doi: 10.1016/0010-7824(92)90101-x. [DOI] [PubMed] [Google Scholar]
- [23].Fraser IS, Weisberg E, Brache V, et al. Serum Nestorone and ethinyl estradiol levels, and ovulation inhibition in women using three different dosage combinations of a Nestorone progestogen-ethinyl estradiol contraceptive vaginal ring on a bleeding-signaled regimen. Contraception. 2005;72:40–5. doi: 10.1016/j.contraception.2004.12.015. [DOI] [PubMed] [Google Scholar]
- [24].Timmer CJ, Mulders TM. Pharmacokinetics of etonogestrel and ethinylestradiol released from a combined contraceptive vaginal ring. Clin Pharmacokinet. 2000;39:233–42. doi: 10.2165/00003088-200039030-00005. [DOI] [PubMed] [Google Scholar]
- [25].Brache V, Faundes A. Contraceptive vaginal rings: a review. Contraception. 2010;82:418–27. doi: 10.1016/j.contraception.2010.04.012. [DOI] [PubMed] [Google Scholar]
- [26].Kerns J, Darney P. Vaginal ring contraception. Contraception. 2011;83:107–15. doi: 10.1016/j.contraception.2010.07.008. [DOI] [PubMed] [Google Scholar]
- [27].Croxatto HB, Brache V, Sitruk-Ware R, Kumar N, Sivin I. A study to evaluate the effect of a contraceptive vaginal ring delivering a daily dose of 400-500 μg of CDB-2914 on pharmacokinetics and pharmacodynamics in normal cycling women. Population Council; New York, New York: 2003. Summary study report. Protocol 312. [data on file] [Google Scholar]
- [28].Stanczyk FZ, Cho MM, Endres DB, Morrison JL, Patel S, Paulson RJ. Limitations of direct estradiol and testosterone immunoassay kits. Steroids. 2003;68:1173–8. doi: 10.1016/j.steroids.2003.08.012. [DOI] [PubMed] [Google Scholar]
- [29].Kurman R, editor. Blaustein’s pathology of the female genital tract. 5th ed Springer-Verlag; New York: 2002. pp. 383–420. [Google Scholar]
- [30].Mutter GL, Bergeron C, Deligdisch L, et al. The spectrum of endometrial pathology induced by progesterone receptor modulators. Mod Pathol. 2008;21:591–8. doi: 10.1038/modpathol.2008.19. [DOI] [PubMed] [Google Scholar]
- [31].Reel JR, Hild-Petito S, Blye RP. Antiovulatory and postcoital antifertility activity of the antiprogestin CDB-2914 when administered as single, multiple, or continuous doses to rats. Contraception. 1998;58:129–36. doi: 10.1016/s0010-7824(98)00067-5. [DOI] [PubMed] [Google Scholar]
- [32].Horne FM, Blithe DL. Progesterone receptor modulators and the endometrium: changes and consequences. Hum Reprod Update. 2007;13:567–80. doi: 10.1093/humupd/dmm023. [DOI] [PubMed] [Google Scholar]
- [33].Writing Group for the Women’s Health Initiative Investigators Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288:321–33. doi: 10.1001/jama.288.3.321. [DOI] [PubMed] [Google Scholar]
- [34].Fournier A, Berrino F, Riboli E, Avenel V, Clavel-Chapelon F. Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. Int J Cancer. 2005;114:448–54. doi: 10.1002/ijc.20710. [DOI] [PubMed] [Google Scholar]
- [35].Lakha F, Ho PC, Van der Spuy ZM, et al. A novel estrogen-free oral contraceptive pill for women: multicentre, double-blind, randomized controlled trial of mifepristone and progestogen-only pill (levonorgestrel) Hum Reprod. 2007;22:2428–36. doi: 10.1093/humrep/dem177. [DOI] [PubMed] [Google Scholar]