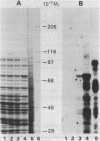
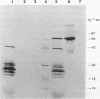
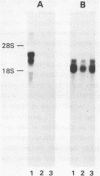
Abstract
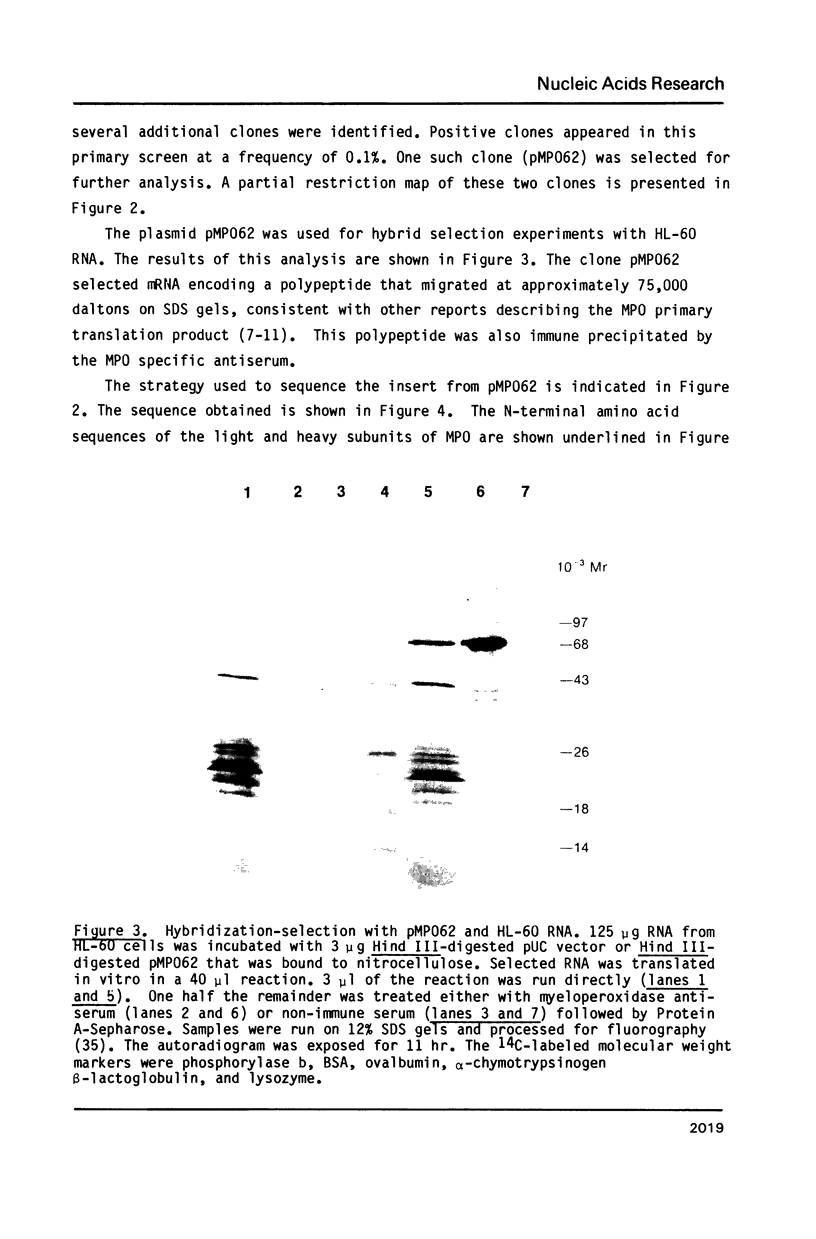
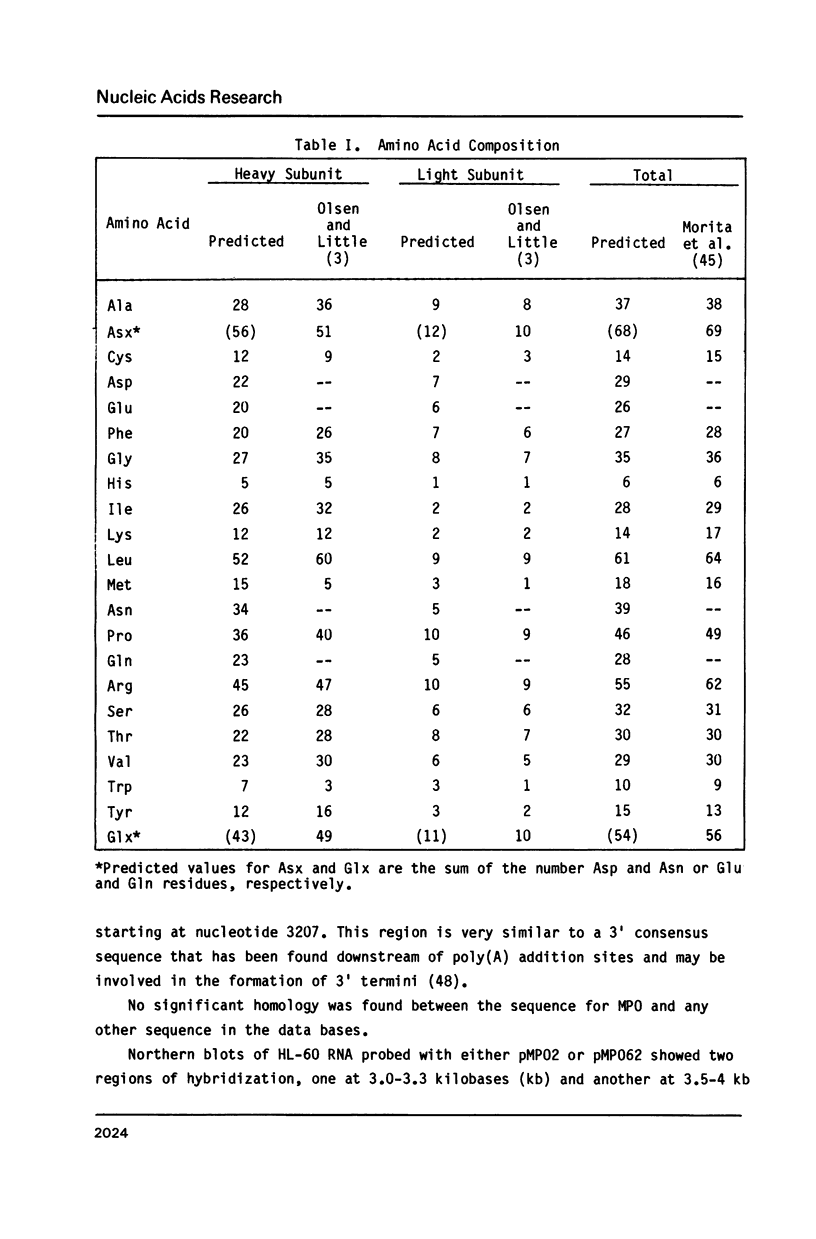
Myeloperoxidase is a component of the microbicidal network of polymorphonuclear leukocytes. The enzyme is a tetramer consisting of two heavy and two light subunits. A large proportion of humans demonstrate genetic deficiencies in the production of myeloperoxidase. As a first step in analyzing these deficiencies in more detail, we have isolated cDNA clones for myeloperoxidase from an expression library of the HL-60 human promyelocytic leukemia cell line. Two overlapping plasmids (pMP02 and pMP062) were identified as myeloperoxidase cDNA clones based on the detection with myeloperoxidase antiserum of 70 kDa protein expressed in pMP02-containing bacteria and a 75 kDa polypeptide produced by hybridization selection and translation using pMP062 and HL-60 RNA. Formal identification of the clones was made by matching the predicted amino acid sequences with the amino terminal sequences of the heavy and light subunits. Both subunits are encoded by one mRNA in the following order: pre-pro-sequences--light subunit--heavy subunit. The molecular weight of the predicted primary translation product is 83.7 kDa. Northern blots reveal two size classes of hybridizing RNAs (approximately 3.0-3.3 and 3.5-4.0 kilobases) whose expression is restricted to cells of the granulocytic lineage and parallels the changes in enzymatic activity observed during differentiation.
Full text
PDF












Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benjamin D., Magrath I. T., Maguire R., Janus C., Todd H. D., Parsons R. G. Immunoglobulin secretion by cell lines derived from African and American undifferentiated lymphomas of Burkitt's and non-Burkitt's type. J Immunol. 1982 Sep;129(3):1336–1342. [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Breitman T. R., Selonick S. E., Collins S. J. Induction of differentiation of the human promyelocytic leukemia cell line (HL-60) by retinoic acid. Proc Natl Acad Sci U S A. 1980 May;77(5):2936–2940. doi: 10.1073/pnas.77.5.2936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breton-Gorius J., Houssay D., Dreyfus B. Partial myeloperoxidase deficiency in a case of preleukaemia. I. Studies of fine structure and peroxidase synthesis of promyelocytes. Br J Haematol. 1975 Jul;30(3):273–278. doi: 10.1111/j.1365-2141.1975.tb00542.x. [DOI] [PubMed] [Google Scholar]
- Catovsky D., Galton D. A., Robinson J. Myeloperoxidase-deficient neutrophils in acute myeloid leukaemia. Scand J Haematol. 1972;9(2):142–148. doi: 10.1111/j.1600-0609.1972.tb00923.x. [DOI] [PubMed] [Google Scholar]
- Church G. M., Gilbert W. Genomic sequencing. Proc Natl Acad Sci U S A. 1984 Apr;81(7):1991–1995. doi: 10.1073/pnas.81.7.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cioe L., Curtis P. Detection and characterization of a mouse alpha-spectrin cDNA clone by its expression in Escherichia coli. Proc Natl Acad Sci U S A. 1985 Mar;82(5):1367–1371. doi: 10.1073/pnas.82.5.1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis A. T., Brunning R. D., Quie P. G. Polymorphonuclear leukocyte myeloperoxidase deficiency in a patient with myelomonocytic leukemia. N Engl J Med. 1971 Sep 30;285(14):789–790. doi: 10.1056/NEJM197109302851410. [DOI] [PubMed] [Google Scholar]
- El-Maallem H., Fletcher J. Impaired neutrophil function and myeloperoxidase deficiency in myeloid metaplasia. Br J Haematol. 1977 Nov;37(3):323–329. doi: 10.1111/j.1365-2141.1977.tb01002.x. [DOI] [PubMed] [Google Scholar]
- Erikson J., Griffin C. A., ar-Rushdi A., Valtieri M., Hoxie J., Finan J., Emanuel B. S., Rovera G., Nowell P. C., Croce C. M. Heterogeneity of chromosome 22 breakpoint in Philadelphia-positive (Ph+) acute lymphocytic leukemia. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1807–1811. doi: 10.1073/pnas.83.6.1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. "A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity". Addendum. Anal Biochem. 1984 Feb;137(1):266–267. doi: 10.1016/0003-2697(84)90381-6. [DOI] [PubMed] [Google Scholar]
- Gallagher R., Collins S., Trujillo J., McCredie K., Ahearn M., Tsai S., Metzgar R., Aulakh G., Ting R., Ruscetti F. Characterization of the continuous, differentiating myeloid cell line (HL-60) from a patient with acute promyelocytic leukemia. Blood. 1979 Sep;54(3):713–733. [PubMed] [Google Scholar]
- Gubler U., Hoffman B. J. A simple and very efficient method for generating cDNA libraries. Gene. 1983 Nov;25(2-3):263–269. doi: 10.1016/0378-1119(83)90230-5. [DOI] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Kalyanaraman V. S., Sarngadharan M. G., Robert-Guroff M., Miyoshi I., Golde D., Gallo R. C. A new subtype of human T-cell leukemia virus (HTLV-II) associated with a T-cell variant of hairy cell leukemia. Science. 1982 Nov 5;218(4572):571–573. doi: 10.1126/science.6981847. [DOI] [PubMed] [Google Scholar]
- Kitahara M., Eyre H. J., Simonian Y., Atkin C. L., Hasstedt S. J. Hereditary myeloperoxidase deficiency. Blood. 1981 May;57(5):888–893. [PubMed] [Google Scholar]
- Kitahara M., Kushner J. P. Acquired myeloperoxidase deficiency and recurrent infections in a patient with acute myelomonocytic leukemia. Cancer. 1979 Dec;44(6):2244–2248. doi: 10.1002/1097-0142(197912)44:6<2244::aid-cncr2820440636>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- Koeffler H. P., Ranyard J., Pertcheck M. Myeloperoxidase: its structure and expression during myeloid differentiation. Blood. 1985 Feb;65(2):484–491. [PubMed] [Google Scholar]
- Kornfeld R., Kornfeld S. Assembly of asparagine-linked oligosaccharides. Annu Rev Biochem. 1985;54:631–664. doi: 10.1146/annurev.bi.54.070185.003215. [DOI] [PubMed] [Google Scholar]
- Kozak M. Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell. 1986 Jan 31;44(2):283–292. doi: 10.1016/0092-8674(86)90762-2. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K., Favre M. Maturation of the head of bacteriophage T4. I. DNA packaging events. J Mol Biol. 1973 Nov 15;80(4):575–599. doi: 10.1016/0022-2836(73)90198-8. [DOI] [PubMed] [Google Scholar]
- Lehrach H., Diamond D., Wozney J. M., Boedtker H. RNA molecular weight determinations by gel electrophoresis under denaturing conditions, a critical reexamination. Biochemistry. 1977 Oct 18;16(21):4743–4751. doi: 10.1021/bi00640a033. [DOI] [PubMed] [Google Scholar]
- Lozzio C. B., Lozzio B. B. Human chronic myelogenous leukemia cell-line with positive Philadelphia chromosome. Blood. 1975 Mar;45(3):321–334. [PubMed] [Google Scholar]
- McLauchlan J., Gaffney D., Whitton J. L., Clements J. B. The consensus sequence YGTGTTYY located downstream from the AATAAA signal is required for efficient formation of mRNA 3' termini. Nucleic Acids Res. 1985 Feb 25;13(4):1347–1368. doi: 10.1093/nar/13.4.1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Michelson A. M., Markham A. F., Orkin S. H. Isolation and DNA sequence of a full-length cDNA clone for human X chromosome-encoded phosphoglycerate kinase. Proc Natl Acad Sci U S A. 1983 Jan;80(2):472–476. doi: 10.1073/pnas.80.2.472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan D. A., Ruscetti F. W., Gallo R. Selective in vitro growth of T lymphocytes from normal human bone marrows. Science. 1976 Sep 10;193(4257):1007–1008. doi: 10.1126/science.181845. [DOI] [PubMed] [Google Scholar]
- Morita Y., Iwamoto H., Aibara S., Kobayashi T., Hasegawa E. Crystallization and properties of myeloperoxidase from normal human leukocytes. J Biochem. 1986 Mar;99(3):761–770. doi: 10.1093/oxfordjournals.jbchem.a135535. [DOI] [PubMed] [Google Scholar]
- Nauseef W. M. Myeloperoxidase biosynthesis by a human promyelocytic leukemia cell line: insight into myeloperoxidase deficiency. Blood. 1986 Apr;67(4):865–872. [PubMed] [Google Scholar]
- Nauseef W. M., Root R. K., Malech H. L. Biochemical and immunologic analysis of hereditary myeloperoxidase deficiency. J Clin Invest. 1983 May;71(5):1297–1307. doi: 10.1172/JCI110880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen R. L., Little C. Studies on the subunits of human myeloperoxidase. Biochem J. 1984 Sep 15;222(3):701–709. doi: 10.1042/bj2220701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsson I., Olofsson T., Odeberg H. Myeloperoxidase-mediated iodination in granulocytes. Scand J Haematol. 1972;9(5):483–491. doi: 10.1111/j.1600-0609.1972.tb00974.x. [DOI] [PubMed] [Google Scholar]
- Olsson I., Persson A. M., Strömberg K. Biosynthesis, transport and processing of myeloperoxidase in the human leukaemic promyelocytic cell line HL-60 and normal marrow cells. Biochem J. 1984 Nov 1;223(3):911–920. doi: 10.1042/bj2230911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parry M. F., Root R. K., Metcalf J. A., Delaney K. K., Kaplow L. S., Richar W. J. Myeloperoxidase deficiency: prevalence and clinical significance. Ann Intern Med. 1981 Sep;95(3):293–301. doi: 10.7326/0003-4819-95-3-293. [DOI] [PubMed] [Google Scholar]
- Proudfoot N. J., Brownlee G. G. 3' non-coding region sequences in eukaryotic messenger RNA. Nature. 1976 Sep 16;263(5574):211–214. doi: 10.1038/263211a0. [DOI] [PubMed] [Google Scholar]
- Ricciardi R. P., Miller J. S., Roberts B. E. Purification and mapping of specific mRNAs by hybridization-selection and cell-free translation. Proc Natl Acad Sci U S A. 1979 Oct;76(10):4927–4931. doi: 10.1073/pnas.76.10.4927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rovera G., Santoli D., Damsky C. Human promyelocytic leukemia cells in culture differentiate into macrophage-like cells when treated with a phorbol diester. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2779–2783. doi: 10.1073/pnas.76.6.2779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saxon A., Stevens R. H., Quan S. G., Golde D. W. Immunologic characterization of hairy cell leukemias in continuous culture. J Immunol. 1978 Mar;120(3):777–782. [PubMed] [Google Scholar]
- Selsted M. E., Harwig S. S., Ganz T., Schilling J. W., Lehrer R. I. Primary structures of three human neutrophil defensins. J Clin Invest. 1985 Oct;76(4):1436–1439. doi: 10.1172/JCI112121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss E. C., Kobori J. A., Siu G., Hood L. E. Specific-primer-directed DNA sequencing. Anal Biochem. 1986 Apr;154(1):353–360. doi: 10.1016/0003-2697(86)90536-1. [DOI] [PubMed] [Google Scholar]
- Taub F., Thompson E. B. An improved method for preparing large arrays of bacterial colonies containing plasmids for hybridization: in situ purification and stable binding of DNA on paper filters. Anal Biochem. 1982 Oct;126(1):222–230. doi: 10.1016/0003-2697(82)90133-6. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verdot J. J., Bayle J., Juhan I., Aillaud M. F., Guitard A. M., Muratore R. Hémogramme automatisé et déficit en myélopéroxydase. Analyse de trente-trois observations. Sem Hop. 1981 Mar 8;57(9-10):450–457. [PubMed] [Google Scholar]
- Vogelstein B., Gillespie D. Preparative and analytical purification of DNA from agarose. Proc Natl Acad Sci U S A. 1979 Feb;76(2):615–619. doi: 10.1073/pnas.76.2.615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woods D., Crampton J., Clarke B., Williamson R. The construction of a recombinant cDNA library representative of the poly(A)+ mRNA population from normal human lymphocytes. Nucleic Acids Res. 1980 Nov 25;8(22):5157–5168. doi: 10.1093/nar/8.22.5157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada M., Kurahashi K. Regulation of myeloperoxidase gene expression during differentiation of human myeloid leukemia HL-60 cells. J Biol Chem. 1984 Mar 10;259(5):3021–3025. [PubMed] [Google Scholar]
- Yamada M., Mori M., Sugimura T. Myeloperoxidases in cultured human promyelocytic leukemia cell line HL-60. Biochem Biophys Res Commun. 1981 Jan 15;98(1):219–226. doi: 10.1016/0006-291x(81)91891-x. [DOI] [PubMed] [Google Scholar]
- Yamada M., Mori M., Sugimura T. Purification and characterization of small molecular weight myeloperoxidase from human promyelocytic leukemia HL-60 cells. Biochemistry. 1981 Feb 17;20(4):766–771. doi: 10.1021/bi00507a018. [DOI] [PubMed] [Google Scholar]
- Yamada M. Myeloperoxidase precursors in human myeloid leukemia HL-60 cells. J Biol Chem. 1982 Jun 10;257(11):5980–5982. [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]