During the last two decades, multiple studies on DNA vaccines have revealed their efficacy in the induction of protective humoral and cellular immunity against a variety of viral and bacterial infections and certain tumors. Depending on the type of DNA vaccination, different CD4 T cell responses can be elicited. Injection of plasmid DNA primarily results in a T helper 1 (Th1) response, whereas Th2 responses are primarily induced when DNA vaccines are given via gene gun (1). An alternative form of DNA vaccination, in which the antigens are cloned in the CDR2 and CDR3 regions of Ig heavy-chain molecules, is termed somatic transgene immunization. Direct intrasplenic injection of the plasmid that contains promoters and enhancers that will drive expression of the transgene in B cells results in secretion of the transgene product in the serum and has been shown to induce a Th0 response. Interestingly, Gerloni and coworkers show in this issue of PNAS that this last form of DNA vaccination gives evidence for new modes of cross-talk between antigen-presenting cells (APC) and CD4 T helper cells (2).
APC are bone marrow-derived cells with the ability to induce primary immune responses. One of the most potent types of APC is the dendritic cell (DC). DC pick up antigens in the skin or other peripheral tissues and travel to the T cell zones of the lymphoid organs, where they stimulate naive CD4 and CD8 T cells (3). During this migration, the DC differentiate from an immature state, in which they can capture antigens and have low stimulatory capacity, to a mature state, in which they are no longer capable of taking up antigens but have an increased T cell stimulatory capacity. The high stimulatory capacity of activated DC is due to increased MHC expression, increased expression of costimulatory molecules such as CD80 and CD86, production of cytokines, and a change in chemokine receptor expression and chemokine production. The maturation of DC can be induced by a wide range of inflammatory stimuli and infectious agents, such as lipopolysaccharide (LPS) and tumor necrosis factor α (TNFα). In addition to these types of stimuli, CD4 T cells have been shown to play an important role in the activation of DC.
Helper T Cell Function
Classically, CD4 T cells have been shown to help B cells to isotype switch and to generate high affinity antibodies for the so-called T cell-dependent antigens. Because B cells can capture antigen via surface Ig and internalize and process it for presentation with MHC class II to CD4 T cells, a direct, antigen-specific T:B cell interaction can occur in this case. Interaction of CD40L on the T cell with CD40 on the B cell is essential for full B cell activation in this process (4). Similarly, CD8 T cell responses can be divided into CD4 T cell-independent and CD4 T cell-dependent responses. In this cellular cooperation, CD8 T cells cannot specifically capture antigen because they recognize only short peptide fragments of the antigen presented by MHC class I. Thus, the CD8 T cell recognizing antigen has no way to specifically attract the attention of CD4 T cells. The CD4 T cell-dependent type of CD8 T cell response revealed an important role for CD4 T cells in the activation of APC. The CD4 T cell help for CD8 T cell responses was shown to be mediated via an APC that presented both MHC class II- and MHC class I-restricted antigens to the CD4 and CD8 T cells, respectively. CD4 T cell help was blocked in the absence of CD40 signaling and could be reconstituted by agonistic CD40 antibodies (5–7). Furthermore, CD40 activation of DC was shown to lead to enhanced survival, increased expression of costimulatory molecules, and increased secretion of cytokines such as IL-12. These findings resulted in a model in which the CD4 T cell, recognizing antigen on the APC surface, activates the APC in such a way that it acquires the capacity to stimulate CD8 T cells.
Helping Each Other
The current work by Gerloni et al. (2) has suggested a new helper role for CD4 T cells in addition to the ones described above. Using somatic transgene immunization, they show that a strong CD4 T cell response toward a Plasmodium falciparum epitope can help the induction of an otherwise weak or absent CD4 T cell response toward an epitope derived from the MUC-1 protein that is expressed in malignant epithelial tumors. To observe this effect, the CD4 T cell responses toward the “strong” and the “weak” epitopes had to be induced by the same APC. This requirement is clearly reminiscent of the CD4 T cell help for CD8 T cell responses, in which CD4 T cell activation of the APC is crucial. Similarly, inhibition of CD40 signaling abrogated the CD4 T cell help for CD4 T cell stimulation. In contrast, whereas ligation of CD40 by a stimulatory antibody can mimic the CD4 T cell help for CD8 T cells, it did not completely reconstitute the CD4 T cell help for CD4 T cells. This difference suggests that the role of CD4 T cells in the activation of APC resulting in CD8 T cell priming is not identical to that resulting in the priming of other CD4 T cells and that different molecules may be involved in each function.
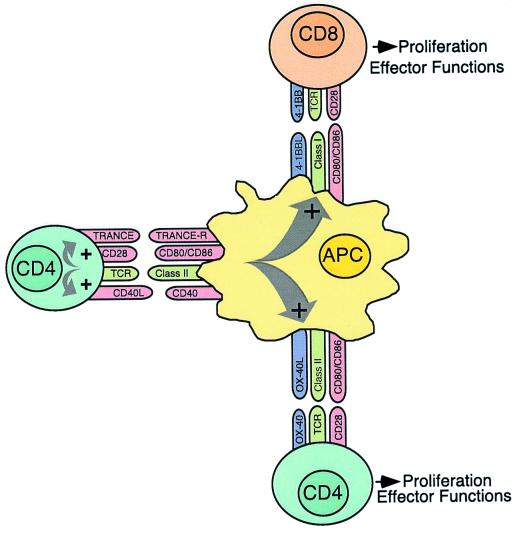
Two different molecules, 4-1BB and OX40, have already been shown to function as costimulators for CD8 and CD4 T cell responses, respectively. A schematic diagram of some of the receptor/ligand interactions between CD4 and CD8 T cells and the APC is presented in Fig. 1. Both ligands, 4-1BBL and OX40L, are induced on the APC by CD40 signaling (8). 4-1BBL-deficient mice have reduced CD8 T cell responses to lymphocytic choriomeningitis virus (LCMV) and diminished primary and memory CD8 T cell responses to peptides (9–11). OX40 is expressed by activated CD4 T cells, and both OX40- and OX40L-deficient mice have been shown to have impaired CD4 T cell priming (12–15). Gerloni et al. tested an agonistic OX40 antibody for its effect on the weak CD4 T cell response. Ligation of OX40 alone or in combination with ligation of CD40 led to measurable CD4 T cell responses toward the weak epitope, but the enhancing effect was considerably less compared with the CD4 T cell help from the strong CD4 T cell response. This finding implies that both activation of the APC by CD40 ligation and costimulation of the CD4 T cell by OX40 ligation could not reconstitute CD4 T cell help and suggests the involvement of other activation pathways on DC and CD4 T cells.
Figure 1.
Interactions of CD4 and CD8 T cells with DC serving as the APC. Recognition of a specific antigen/MHC class II complex on the APC leads to activation of the CD4 T cell with up-regulation of CD40L and TRANCE. Subsequently, CD40-CD40L and TRANCE–TRANCE-R interactions result in enhanced survival, production of cytokines, and increased expression of costimulatory molecules, such as CD80, CD86, OX40L, and 4-1BBL, by the APC. These changes allow the APC to activate other CD8 and CD4 T cells.
One potential receptor/ligand pair involved in the CD4 T cell help is (TRANCE) receptor (TRANCE-R) and TRANCE. Similar to CD40L and CD40, TRANCE and TRANCE-R were found to be expressed on activated CD4 T cells and DC, respectively. Signaling of TRANCE-R has been shown to result in enhanced survival of the DC (16, 17). Blockade of TRANCE signaling in the absence of CD40 interaction greatly decreased CD4 T cell stimulation in LCMV infection. In the presence of CD40 interaction, CD4 T cell responses were not significantly reduced when TRANCE signaling was prohibited (18). This finding suggests redundancy in the CD40 and TRANCE pathways for activation of APC. Because Gerloni et al. did not investigate whether this pathway was involved in the CD4 T cell help for CD4 T cells, the function of this receptor/ligand pair in this model is still unknown.
In addition to the question of which molecules are involved in CD4 T cell help for CD4 T cells, there is also the question of which APC is affected by this type of CD4 T cell help. Although the transfected B cells could possibly act as APC in this model, DC have a more potent ability to activate naive CD4 T cells. Murine splenic DC can be divided into two subsets with different phenotype and functions. Whereas myeloid DC primarily activate Th2 responses, lymphoid DC mainly activate Th1 responses (19–21). Recently, specifically the latter subset of splenic DC was shown to be involved in presenting antigen to CD8 T cells (J.M.M.d.H., S. M. Lehar, and M.J.B., unpublished results). These findings strongly suggest that different types of DC have different functions, and it would be of interest to know whether a specific DC subtype is involved. If indeed DC and not B cells are involved in the priming of CD4 T cells in somatic transgene immunization, it is unclear how the DC acquired the antigen. In principle, this could be due to uptake of soluble Ig molecules secreted by the transfected B cells. However, uptake of soluble Ig would not explain the absence of CD4 T cell help when the two T cell antigens were present on separate Ig molecules, as both were secreted and measurable in the serum. Alternatively, phagocytosis of apoptotic or necrotic cell debris derived from the transfected B cells may be the source of antigen presentation by the DC.
In conclusion, a new role for CD4 T cells in the activation of otherwise absent CD4 T cell responses has been indicated. Clearly, it remains to be established whether T:T cooperation is a mechanism for CD4 T cell priming in general and whether CD4 T cells specific for the same epitope may cooperate with each other. Furthermore, it is of interest whether this form of CD4 T cell help is involved in the broadening of an immune response as seen in the phenomenon of epitope spreading, observed in certain autoimmune diseases.
Footnotes
See companion article on page 13269.
References
- 1.Liu M A, Ulmer J B. Mol Ther. 2000;1:497–500. doi: 10.1006/mthe.2000.0079. [DOI] [PubMed] [Google Scholar]
- 2.Gerloni M, Xiong S, Mukerjee S, Schoenberger S P, Croft M, Zanetti M. Proc Natl Acad Sci USA. 2000;97:13269–13274. doi: 10.1073/pnas.230429197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Banchereau J, Steinman R M. Nature (London) 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 4.van Kooten C, Banchereau J. J Leukocyte Biol. 2000;67:2–17. doi: 10.1002/jlb.67.1.2. [DOI] [PubMed] [Google Scholar]
- 5.Bennett S R, Carbone F R, Karamalis F, Flavell R A, Miller J F, Heath W R. Nature (London) 1998;393:478–480. doi: 10.1038/30996. [DOI] [PubMed] [Google Scholar]
- 6.Ridge J P, Di Rosa F, Matzinger P. Nature (London) 1998;393:474–478. doi: 10.1038/30989. [DOI] [PubMed] [Google Scholar]
- 7.Schoenberger S P, Toes R E, van der Voort E I, Offringa R, Melief C J. Nature (London) 1998;393:480–483. doi: 10.1038/31002. [DOI] [PubMed] [Google Scholar]
- 8.Lane P J, Brocker T. Curr Opin Immunol. 1999;11:308–313. doi: 10.1016/s0952-7915(99)80049-1. [DOI] [PubMed] [Google Scholar]
- 9.DeBenedette M A, Wen T, Bachmann M F, Ohashi P S, Barber B H, Stocking K L, Peschon J J, Watts T H. J Immunol. 1999;163:4833–4841. [PubMed] [Google Scholar]
- 10.Tan J T, Whitmire J K, Ahmed R, Pearson T C, Larsen C P. J Immunol. 1999;163:4859–4868. [PubMed] [Google Scholar]
- 11.Tan J T, Whitmire J K, Murali-Krishna K, Ahmed R, Altman J D, Mittler R S, Sette A, Pearson T C, Larsen C P. J Immunol. 2000;164:2320–2325. doi: 10.4049/jimmunol.164.5.2320. [DOI] [PubMed] [Google Scholar]
- 12.Chen A I, McAdam A J, Buhlmann J E, Scott S, Lupher M L, Jr, Greenfield E A, Baum P R, Fanslow W C, Calderhead D M, Freeman G J, Sharpe A H. Immunity. 1999;11:689–698. doi: 10.1016/s1074-7613(00)80143-0. [DOI] [PubMed] [Google Scholar]
- 13.Kopf M, Ruedl C, Schmitz N, Gallimore A, Lefrang K, Ecabert B, Odermatt B, Bachmann M F. Immunity. 1999;11:699–708. doi: 10.1016/s1074-7613(00)80144-2. [DOI] [PubMed] [Google Scholar]
- 14.Pippig S D, Pena-Rossi C, Long J, Godfrey W R, Fowell D J, Reiner S L, Birkeland M L, Locksley R M, Barclay A N, Killeen N. J Immunol. 1999;163:6520–6529. [PubMed] [Google Scholar]
- 15.Murata K, Ishii N, Takano H, Miura S, Ndhlovu L C, Nose M, Noda T, Sugamura K. J Exp Med. 2000;191:365–374. doi: 10.1084/jem.191.2.365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Josien R, Wong B R, Li H L, Steinman R M, Choi Y. J Immunol. 1999;162:2562–2568. [PubMed] [Google Scholar]
- 17.Josien R, Li H L, Ingulli E, Sarma S, Wong B R, Vologodskaia M, Steinman R M, Choi Y. J Exp Med. 2000;191:495–502. doi: 10.1084/jem.191.3.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bachmann M F, Wong B R, Josien R, Steinman R M, Oxenius A, Choi Y. J Exp Med. 1999;189:1025–1031. doi: 10.1084/jem.189.7.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Smith A L, de St Groth B F. J Exp Med. 1999;189:593–598. doi: 10.1084/jem.189.3.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pulendran B, Smith J L, Caspary G, Brasel K, Pettit D, Maraskovsky E, Maliszewski C R. Proc Natl Acad Sci USA. 1999;96:1036–1041. doi: 10.1073/pnas.96.3.1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Maldonado-Lopez R, De Smedt T, Michel P, Godfroid J, Pajak B, Heirman C, Thielemans K, Leo O, Urbain J, Moser M. J Exp Med. 1999;189:587–592. doi: 10.1084/jem.189.3.587. [DOI] [PMC free article] [PubMed] [Google Scholar]