Summary
Background and objectives
The HLA system plays a central role in the distinction between self antigens and non-self antigens. This study aimed to investigate the association between HLA genes and the outcomes of patients with ANCA-associated vasculitis (AAV).
Design, setting, participants, & measurements
This study recruited 152 consecutive Chinese patients with AAV. The predictive value of the HLA alleles for renal outcome, response to treatment, and all-cause mortality were analyzed.
Results
The proportion of patients with treatment failure was significantly higher in DRB1*0405-positive patients than in DRB1*0405-negative patients (41.7% versus 12.9%; P=0.008; corrected P=0.02). After adjusting for the other potential predictors, DRB1*0405 was still an independent predictor for the poor response to treatment (hazard ratio [HR], 5.91; 95% confidence interval [95% CI], 1.23–28.52; P=0.03). Renal survival was significantly worse in patients with DRB1*0405 than those without DRB1*0405 (P<0.001; corrected P<0.001). After adjusting for the other potential predictors, DRB1*0405 was still an independent predictor for ESRD (HR, 5.50; 95% CI, 2.18–13.88; P<0.001). The probability of all-cause mortality in patients with DPB1*0402 was significantly higher than those without DPB1*0402 (P=0.02; corrected P=0.04). After adjusting for the other potential predictors, DPB1*0402 was still an independent predictor for all-cause mortality (HR, 2.52; 95% CI, 1.21–5.28; P=0.01).
Conclusions
In AAV patients, DRB1*0405 might be an independent risk factor for the poor response to treatment and the deterioration of renal function, whereas DPB1*0402 might be an independent risk factor for all-cause mortality.
Introduction
ANCA-associated vasculitis (AAV) comprises granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA), Churg-Strauss syndrome, and renal-limited vasculitis. The etiology of AAV remains unclear. As discussed in a review by Chen et al., environmental factors and certain drugs were assumed to contribute to the development of AAV (1). In addition to the factors above, the genetic background of AAV has long been of concern because some familial cases of AAV have been reported (2–7).
Among the genetic factors, HLA genes are considered a set of suspected genes in the development of AAV (8–14). A number of studies have shown an association of HLA genes, including both class I and class II, with the disease susceptibility of AAV (8–14). There are five loci of MHC class II genes in HLA, including HLA-DR, HLA-DP, HLA-DQ HLA-DO, and HLA-DM. Among the HLA class II genes, HLA-DR, HLA-DP, and HLA-DQ alleles were found responsible for both GPA and MPA (9,10,14). However, to the best of our knowledge, no association has been found between AAV and the HLA-DO and HLA-DM alleles. Previous studies mainly focused on the association between the frequency of HLA genes and disease susceptibility. Whether there is any association between HLA genes and outcomes of AAV patients is far from clear. This study aimed to investigate the association of some HLA class II genes with the outcomes of patients with AAV, including the response to immunosuppressive treatment, renal outcome, and mortality.
Materials and Methods
Patients and Controls
This study recruited 152 consecutive Chinese patients with AAV, diagnosed at Peking University First Hospital from 1996 to 2009. All of the patients met the criteria of the Chapel Hill Consensus Conference definition of AAV (15). Patients with secondary vasculitis or with other comorbid renal diseases were excluded. This study complied with the Declaration of Helsinki and was approved by the ethics committee of our hospital. Informed consent was obtained from each patient.
Detection of Serum ANCA
ANCA tests were performed by both indirect immunofluorescence assay and antigen-specific ELISA. Standard indirect immunofluorescence assays were performed according to the manufacturer’s instructions (Euroimmun, Lübeck, Germany). Ethanol-fixed human PMNs were used to detect ANCA and monkey liver sections were used to exclude antinuclear antibodies. Cytoplasmic ANCA and perinuclear ANCA were distinguished according to staining patterns by two experienced technicians. Two highly purified known ANCA antigens, proteinase 3 (PR3) and myeloperoxidase (MPO), purified as previously reported, were used as solid-phase ligands in ELISA (16).
DNA Samples
Peripheral blood samples (10 ml) from patients with AAV and normal controls were collected in the presence of EDTA. Genomic DNA was obtained from peripheral blood leukocytes with a salting-out procedure (17).
Sequence-Based Typing
Typing of HLA-DRB1, HLA-DPB1, HLA-DQB1, and HLA-DQA1 was performed by bidirectional sequencing of exon 2 using SeCore sequencing kits (Invitrogen, Brown Deer, WI), as previously described (14,18).
Treatment
Treatment protocols were previously described (19–21). The induction therapy included corticosteroids in combination with cyclophosphamide. Oral prednisone was prescribed at an initial dosage of 1 mg/kg per day for 4–6 weeks, with reducing doses over time to 12.5–15 mg by 3 months. We administered 0.5–1.0 g/m2 of cyclophosphamide administered intravenously every month or orally daily (2 mg/kg per day). A 25% dose reduction of cyclophosphamide was made for those participants aged >65 years, those who developed leukocytopenia (<4000 cells/mm3), and those with renal insufficiency. Patients with ARF or pulmonary hemorrhage received pulses of intravenous methylprednisolone (7–15 mg/kg per day) before the above standard induction therapy. Patients with severe pulmonary hemorrhage received additional plasma exchanges. For maintenance therapy, daily oral azathioprine was given.
Definition of the Response to Immunosuppressive Treatment
The response to immunosuppressive treatment was defined as previously described (22). The vasculitic disease activity was measured by the Birmingham Vasculitis Activity Score (BVAS) (23). Remission was defined as the absence of disease activity attributable to active disease qualified by the need for ongoing stable maintenance immunosuppressive therapy (complete remission), or a 50% reduction of disease activity score and absence of new manifestations (partial remission). Those who failed to satisfy the above criteria for remission (complete or partial) were defined as having treatment failure.
Statistical Analyses
Chi-squared tests, one-way ANOVAs, and t tests were performed as appropriate. Kaplan–Meier survival analysis was used to assess patient and renal survival. Multivariate analyses of the treatment response were performed using the logistic regression model. Multivariate analyses of renal and patient survival were performed with the Cox regression model. Results were expressed as hazard ratios (HRs) with 95% confidence intervals (95% CIs). The following variables were assessed as potential predictors of treatment response, ESRD, and all-cause mortality: age, sex, ANCA specificities in ELISA, initial serum creatinine, HLA genes, plasma exchanges, pulses of intravenous methylprednisolone, and the way in which cyclophosphamide was used in the induction therapy (monthly intravenous dosage versus daily oral dosage). The Benjamini–Hochberg false discovery rate correction was applied to correct the P value (24,25).
Statistical power was estimated using Power and Sample Size Estimation Software (http://biostat.mc.vanderbilt.edu/PowerSampleSize).
Differences were considered significant if the P value was <0.05. Statistical analyses were performed using the SPSS statistical software package (version 11.0; SPSS Inc, Chicago, IL).
Results
Clinical Data
Among the 152 patients with AAV, 75 were male and 77 were female, with a median age of 61 years (range, 14–86) at diagnosis. According to the Chapel Hill Consensus Conference definition (15), 44 patients (28.9%) were classified as having GPA, 94 patients (61.8%) were classified as having MPA, and 14 patients (9.3%) were classified as having renal-limited vasculitis. Our results showed that 130 patients were MPO-ANCA positive, 19 were PR3-ANCA positive, and the remaining 3 were both MPO-ANCA and PR3-ANCA positive. Among the 107 patients with MPA, 105 (98.1%) were MPO-ANCA positive, and the other 2 (1.9%) were both MPO-ANCA and PR3-ANCA positive. Among the 44 patients with GPA, 25 (56.8%) were MPO-ANCA positive and 19 (43.2%) were PR3-ANCA positive. The BVAS level at diagnosis was 20 (interquartile range [IQR], 16–24].
HLA-DRB1, HLA-DPB1, HLA-DQB1, and HLA-DQA1 Alleles
A total of 56 HLA-DRB1, HLA-DPB1, HLA-DQB1, and HLA-DQA1 alleles were typed. The frequencies of these typed alleles are shown in Table 1.
Table 1.
Frequencies of all HLA-DRB1 and HLA-DPB1 alleles
| HLA Allele | Prevalence, n (%) | HLA Allele | Prevalence, n (%) | HLA Allele | Prevalence, n (%) |
|---|---|---|---|---|---|
| DRB1*1201 | 4 (2.6) | DPB1*0202 | 1 (0.7) | DQB1*0302 | 8 (5.3) |
| DRB1*0405 | 12 (7.9) | DPB1*0201 | 83 (54.6) | DQB1*0303 | 30 (19.7) |
| DRB1*1501 | 57 (37.5) | DPB1*0301 | 15 (9.9) | DQB1*0301 | 30 (19.7) |
| DRB1*0701 | 29 (19.1) | DPB1*0302 | 3 (2) | DQB1*0401 | 12 (7.9) |
| DRB1*1405 | 10 (6.6) | DPB1*0401 | 32 (21.1) | DQB1*0501 | 34 (22.4) |
| DRB1*0401 | 6 (3.9) | DPB1*0402 | 58 (38.2) | DQB1*0602 | 63 (41.4) |
| DRB1*0406 | 6 (3.9) | DPB1*0501 | 2 (1.3) | DQB1*0502 | 25 (16.4) |
| DRB1*0301 | 10 (6.6) | DPB1*0601 | 4 (2.6) | DQB1*0503 | 24 (15.8) |
| DRB1*0901 | 30 (19.7) | DPB1*0801 | 3 (2) | DQB1*0601 | 2 (1.3) |
| DRB1*1101 | 27 (17.8) | DPB1*0901 | 6 (3.9) | DQB1*0201 | 72 (47.4) |
| DRB1*0101 | 4 (2.6) | DPB1*1601 | 3 (2) | DQA1*0101 | 13 (8.6) |
| DRB1*0803 | 15 (9.9) | DPB1*1701 | 6 (3.9) | DQA1*0102 | 89 (58.6) |
| DRB1*1202 | 21 (13.8) | DPB1*1801 | 4 (2.6) | DQA1*0103 | 6 (3.9) |
| DRB1*0102 | 1 (0.7) | DPB1*2101 | 1 (0.7) | DQA1*0201 | 27 (17.8) |
| DRB1*1001 | 8 (5.3) | DPB1*2402 | 2 (1.3) | DQA1*0301 | 76 (50.0) |
| DRB1*1302 | 4 (2.6) | DPB1*3301 | 18 (11. 8) | DQA1*0302 | 1 (0.7) |
| DRB1*1403 | 1 (0.7) | DPB1*1301 | 6 (3.9) | DQA1*0401 | 9 (5.9) |
| DRB1*1301 | 4 (2.6) | DPB1*2001 | 6 (3.9) | DQA1*0501 | 49 (32.2) |
| DRB1*1502 | 2 (1.3) | DQA1*0601 | 6 (3.9) |
Association between HLA Genes and the Response to Immunosuppressive Treatment
At the sixth month after initiation of the induction therapy, 58 of 152 patients (38.2%) achieved complete remission, 80 of 152 patients (52.6%) achieved partial remission, and 14 of 152 (9.2%) patients had treatment failure. Of the 12 patients who were DRB1*0405 positive, 5 (41.7%) had treatment failure and 7 (58.3%) achieved remission. Of the 140 patients who were DRB1*0405 negative, 122 (87.1%) achieved remission and 18 (12.9%) had treatment failure. The proportion of patients with treatment failure was significantly higher in the DRB1*0405 positive group than in the DRB1* 0405 negative group (41.7% versus 12.9%; chi-squared test=7.14; P=0.008; corrected P=0.02). Power calculations indicated that this strategy would provide >70% power to detect HLA loci with a hazard risk >1.5.
Table 2 provides the clinical data of patients with and without DRB1*0405. The clinical characteristics were roughly comparable between these two groups.
Table 2.
Comparison of clinical data between DRB1*0405 positive and DRB1*0405 negative, DPB1*0402 positive, and DPB1*0402 negative patients
| DRB1*0405 Positive (n=12) | DRB1*0405 Negative (n=140) | P | DPB1*0402 Positive (n=58) | DPB1*0402 Negative (n=94) | P | |
|---|---|---|---|---|---|---|
| Male/female | 8/4 | 69/71 | 0.25 | 25/33 | 50/44 | 0.23 |
| Age at diagnosis (yr) | 58 (19–66) | 61 (52–68) | 0.43 | 59 (50–67) | 63 (52–69) | 0.76 |
| MPO-ANCA/PR3-ANCA | 1/11 | 22/118 | 0.56 | 47/11 | 83/11 | 0.22 |
| GPA/MPA/RLV | 0/10/2 | 44/84/12 | 0.06 | 24/32/2 | 20/62/12 | 0.01 |
| Muscle pain | 3 (25.0) | 42 (30.0) | 0.72 | 21 (46.7) | 24 (53.3) | 0.16 |
| Arthralgia | 5 (25.0) | 53 (37.9) | 0.38 | 25 (44.6) | 31 (55.4) | 0.21 |
| Skin rash | 1 (8.3) | 23 (16.4) | 0.46 | 14 (58.3) | 10 (41.7) | 0.03 |
| Pulmonary | 5 (41.7) | 76 (54.3) | 0.40 | 27 (33.3) | 54 (66.7) | 0.19 |
| Eye | 1 (8.3) | 33 (23.6) | 0.22 | 12 (35.3) | 22 (64.7) | 0.70 |
| Ear, nose, and throat | 3 (25.0) | 57 (40.7) | 0.29 | 27 (45.0) | 33 (55.0) | 0.16 |
| Gastrointestinal tract | 1 (8.3) | 18 (12.9) | 0.65 | 8 (42.1) | 11 (57.9) | 0.71 |
| Nervous system | 1 (8.3) | 19 (13.6) | 0.61 | 5 (25.0) | 15 (75.0) | 0.19 |
| Initial serum creatinine (μmol/L) | 560 (152–823) | 282 (123–596) | 0.004 | 259 (104–596) | 314 (135–620) | 0.15 |
| Urinary protein (g/24 h) | 2 (1.1–3) | 2.1 (0.98–3.1) | 0.91 | 2.33 (1.8–2.9) | 2.21 (1.3–3.3) | 0.44 |
| BVAS | 17 (15–20) | 20 (16–25) | 0.53 | 20 (15–23) | 20 (17–25) | 0.98 |
Data are given as the proportion, median (interquartile range), or n (%). MPO, myeloperoxidase; PR3, proteinase 3; GPA: granulomatosis with polyangiitis (previously nominated as Wegener’s granulomatosis); MPA, microscopic polyangiitis; RLV, renal-limited vasculitis; BVAS, Birmingham Vasculitis Activity Score.
To further investigate the role of HLA-DRB1*0405 in predicting the treatment response, variables including age, sex, ANCA specificities in ELISA, initial serum creatinine, HLA genes, plasma exchanges, pulses of intravenous methylprednisolone, and the way in which cyclophosphamide was used in the induction therapy, in addition to DRB1*0405, were assessed as potential predictors of the treatment response in the multivariate analysis (Table 3). After adjusting for the other potential predictors described above, DRB1*0405 was still an independent predictor for the poor treatment response (HR, 5.91; 95% CI, 1.23–28.52; P=0.03).
Table 3.
Predictors of the treatment response in the multivariate analyses
| Predictor | P | Hazard Ratio | 95% Confidence interval |
|---|---|---|---|
| Age | 0.46 | 1.47 | 0.53–4.05 |
| Sex | 0.74 | 1.19 | 0.42–3.32 |
| ANCA specificity in ELISA | 0.22 | 5.03 | 0.38–66.31 |
| Initial serum creatinine (μmol/L) | 0.004 | ||
| first tertile (≤170) | 0.006 | 10.05 | 1.94–52.16 |
| second tertile (171–447) | 0.11 | 2.57 | 0.81–8.10 |
| third tertile (>447) | Reference group | ||
| Oral cyclophosphamide/pulse cyclophosphamide | 0.60 | 1.72 | 0.23–12.87 |
| Plasma exchange | 0.30 | 5.00 | 0.24–105.46 |
| Pulses of intravenous methylprednisolone | 0.12 | 0.30 | 0.07–1.34 |
| HLA-DRB1*0405 | 0.03 | 5.91 | 1.23–28.52 |
Association between HLA Genes and ESRD
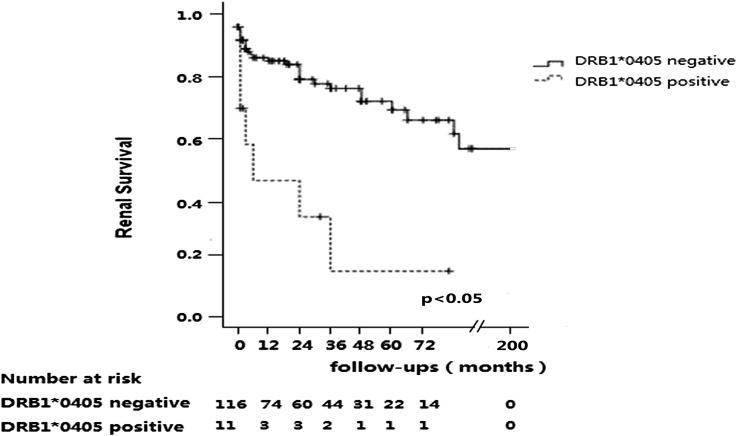
The median follow-up time was 17 months (IQR, 1–47]. During follow-up, 32 patients progressed to ESRD. As shown in Figure 1, the renal survival was significantly worse in patients with DRB1*0405 than those without DRB1*0405 (P<0.001; corrected P<0.001). The cumulative proportion of renal survival at 1 year was significantly lower in patients with DRB1*0405 than those without DRB1*0405 (52.4% versus 89.8%; P<0.01). This was also the case for the cumulative proportions of renal survival at 2 years (52.4% versus 86.3%; P<0.01) and 3 years (37.4% versus 80.7%; P<0.01). There was no significant association between other HLA alleles and ESRD.
Figure 1.
Renal survival of patients with and without DRB1*0405.
To further investigate the role of HLA-DRB1*0405 in predicting ESRD, variables including age, sex, ANCA specificities, initial serum creatinine, plasma exchanges, pulses of intravenous methylprednisolone, and the way in which cyclophosphamide was used in induction therapy, in addition to DRB1*0405, were assessed as potential predictors of ESRD in the multivariate analysis (Table 4). After adjusting for the other variables described above, DRB1*0405 was still an independent predictor for ESRD (HR, 5.50; 95% CI, 2.18–13.88; P<0.001).
Table 4.
Predictors of ESRD in the multivariate analyses
| Predictor | P | Hazard Ratio | 95% Confidence Interval |
|---|---|---|---|
| Age | 0.81 | 0.90 | 0.39–2.11 |
| Sex | 0.35 | 1.41 | 0.68–2.92 |
| ANCA specificity in ELISA | 0.85 | 0.89 | 0.27–2.95 |
| Initial serum creatinine (μmol/L) | 0.007 | ||
| first tertile (≤170) | 0.002 | 0.17 | 0.06–0.51 |
| second tertile (171–447) | 0.20 | 0.58 | 0.26–1.33 |
| third tertile (>447) | Reference group | ||
| Oral cyclophosphamide/pulse cyclophosphamide | 0.24 | 1.43 | 0.79–2.59 |
| Plasma exchange | 0.88 | 1.19 | 0.13–10.82 |
| Intravenous methylprednisolone pulse therapy | 0.99 | 1.01 | 0.47–2.16 |
| HLA-DRB1*0405 | <0.001 | 5.50 | 2.18–13.88 |
Association between HLA Genes and All-Cause Mortality
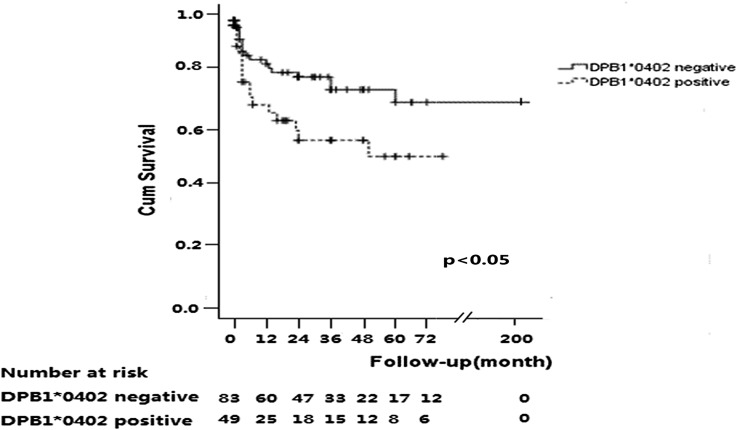
The median follow-up time was 24 months (IQR, 2–48). During follow-up, 34 of 152 (22.4%) patients died. As shown in Figure 2, the probability of all-cause mortality in patients with DPB1*0402 was significantly higher than those without DPB1*0402 (P=0.02; corrected P=0.04). The cumulative proportion of patient survival at 1 year was significantly lower in patients with DPB1*0402 than those without DPB1*0402 (79.6% versus 89.3%). This was also the case at 2 years (68.0% versus 85.0%) and 3 years (61.0% versus 83.4%). Causes of death of these patients are shown in Table 5. There was no significant association between other HLA genes and all-cause mortality.
Figure 2.
Survival of patients with and without DPB1*0402.
Table 5.
Causes of death in patients with and without DPB1*0402
| Cause of Death | DPB1*0402 Negative (n=17) | DPB1*0402 Positive (n=17) | P |
|---|---|---|---|
| Infection | 10 (58.8) | 8 (47.1) | 0.49 |
| Active vasculitis | 3 (17.6) | 2 (11.8) | 0.53 |
| ESRD complications | 2 (11.8) | 1 (5.9) | 0.47 |
| Cardiovascular events | 1 (5.9) | 3 (17.6) | 0.35 |
| Unknown | 1 (5.9) | 3 (17.6) | 0.35 |
Table 2 shows the clinical data of patients with and without DPB1*0402. The clinical characteristics were roughly comparable between these two groups.
The clinical data of patients with and without DPB1*0402 are presented in Table 6. To further investigate the role of HLA-DPB1*0402 in predicting all-cause mortality, variables including age, sex, ANCA specificities, initial serum creatinine, plasma exchanges, pulses of intravenous methylprednisolone, and the way in which cyclophosphamide was used in induction therapy, in addition to DPB1*0402, were assessed as potential predictors of all-cause mortality in the multivariate analysis (Table 6). After adjusting for the other variables described above, DPB1*0402 was still an independent predictor for all-cause mortality (HR, 2.52; 95% CI, 1.21–5.28; P=0.01).
Table 6.
Predictors of all-cause mortality in the multivariate analysis
| Predictor | P | Hazard Ratio | 95% Confidence Interval |
|---|---|---|---|
| Age | <0.001 | 3.76 | 1.82–7.78 |
| Sex | 0.33 | 0.69 | 0.33–1.45 |
| ANCA specificity in ELISA | 0.51 | 1.47 | 0.46–4.68 |
| Initial serum creatinine (μmol/L) | 0.001 | ||
| first tertile (≤170) | 0.0001 | 0.08 | 0.02–0.30 |
| second tertile (171–447) | 0.24 | 0.64 | 0.31–1.35 |
| third tertile (>447) | Reference group | ||
| Oral cyclophosphamide/pulse cyclophosphamide | 0.98 | 1.01 | 0.50–2.04 |
| Plasma exchange | 0.90 | 0.87 | 0.11–7.13 |
| Intravenous methylprednisolone pulse therapy | 0.02 | 0.32 | 0.13–0.81 |
| HLA-DPB1*0402 | 0.01 | 2.52 | 1.21–5.28 |
Discussion
This study investigated the association of some HLA class II genes, including HLA-DRB1, HLA-DPB1, HLA-DQA1, and HLA-DQB1, with the outcomes of patients with AAV. Of all of these HLA genes, DRB1*0405 might be an independent predictor for the deterioration of renal function. In our study, the treatment response was defined by BVAS, which reflects disease activity of multi-systems. Our study showed that DRB1*0405 was also associated with the treatment response. In the multivariate analysis, DRB1*0405 was still an independent predictor for the poor treatment response. This suggests that DRB1*0405 had a great effect not only on renal function progression but also on the systemic disease activity. The DRB1*0405 allele has been reported to be associated with various autoimmune disorders including rheumatoid arthritis and Vogt-Koyanagi-Harada disease (26–28). Gencik et al. showed that TNFα 2a/DRB1*04 individuals were found to be over-represented in a subgroup of patients with GPA in ESRD as a result of renal vasculitis. ANCA antigens are mainly located in azurophilic granules of polymorphonuclear granulocytes; after being primed by TNFα, there are more ANCA antigens expressed on the membrane of polymorphonuclear granulocytes (29,30). Over-representation of TNFα 2a/DRB1*04 might contribute to the severe courses of GPA (11). We speculated that the mechanism of HLA-DRB1*0405 contributing to the outcome of AAV is possibly through the over-representation of TNFα, resulting in poor prognosis of renal function.
HLA-DPB1*0402 has been reported to be associated with some other autoimmune diseases such as type 1 diabetes (31,32). The association with genes of the HLA class II region is explained by their role in the presentation of peptides derived from exogenous protein to CD4+ T helper cells. In the case of type 1 diabetes, this mechanism may result in an inappropriate T cell immune response against self-antigens on the pancreatic β cell. In this study, the probability of all-cause mortality in patients with DPB1*0402 was significantly higher than those without DPB1*0402, and DPB1*0402 was an independent predictor for all-cause mortality. To the best of our knowledge, this is the first report to show that a certain HLA allele is associated with mortality in AAV patients. The exact mechanism of such an association requires further study.
For the other HLA genes in this study, we conducted further investigations to calculate the power value in the Kaplan–Meier survival analysis. Some of the power values were <0.6 (Supplemental Material), which means there might be a possibility of false negative results for these alleles. Future studies with larger samples are needed. The limitation of our research was the relatively short duration of follow-up. Moreover, it is necessary to validate or replicate our association results using an independent cohort.
In conclusion, we found some associations between certain HLA genes and outcomes of AAV patients. DRB1*0405 might be an independent risk factor for the poor treatment response and the deterioration of renal function, whereas DPB1*0402 might be an independent risk factor for all-cause mortality.
Disclosures
None.
Supplementary Material
Acknowledgments
We are very grateful to Professor Xue-ying Li for assistance with medical statistics.
This study was supported Chinese 973 Project Grant 2012CB517702 and National Natural Science Fund Grants 30972733 and 81021004.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.13071211/-/DCSupplemental.
References
- 1.Chen M, Kallenberg CG: The environment, geoepidemiology and ANCA-associated vasculitides. Autoimmun Rev 9: A293–A298, 2010 [DOI] [PubMed] [Google Scholar]
- 2.Muniain MA, Moreno JC, Gonzalez Cámpora R: Wegener’s granulomatosis in two sisters. Ann Rheum Dis 45: 417–421, 1986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hay EM, Beaman M, Ralston AJ, Ackrill P, Bernstein RM, Holt PJ: Wegener’s granulomatosis occurring in siblings. Br J Rheumatol 30: 144–145, 1991 [DOI] [PubMed] [Google Scholar]
- 4.Franssen CF, ter Maaten JC, Hoorntje SJ: Brother and sister with myeloperoxidase associated autoimmune disease. Ann Rheum Dis 53: 213, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nowack R, Lehmann H, Flores-Suárez LF, Nanhou A, van der Woude FJ: Familial occurrence of systemic vasculitis and rapidly progressive glomerulonephritis. Am J Kidney Dis 34: 364–373, 1999 [DOI] [PubMed] [Google Scholar]
- 6.Hull CM, Couser WG, Knostman JD: A familial case of P-ANCA glomerulonephritis presenting in a father and daughter. Am J Kidney Dis 35: E23, 2000 [DOI] [PubMed] [Google Scholar]
- 7.Brener Z, Cohen L, Goldberg SJ, Kaufman AM: ANCA-associated vasculitis in Greek siblings with chronic exposure to silica. Am J Kidney Dis 38: E28, 2001 [DOI] [PubMed] [Google Scholar]
- 8.Heckmann M, Holle JU, Arning L, Knaup S, Hellmich B, Nothnagel M, Jagiello P, Gross WL, Epplen JT, Wieczorek S: The Wegener’s granulomatosis quantitative trait locus on chromosome 6p21.3 as characterised by tagSNP genotyping. Ann Rheum Dis 67: 972–979, 2008 [DOI] [PubMed] [Google Scholar]
- 9.Tsuchiya N, Kobayashi S, Kawasaki A, Kyogoku C, Arimura Y, Yoshida M, Tokunaga K, Hashimoto H: Genetic background of Japanese patients with antineutrophil cytoplasmic antibody-associated vasculitis: Association of HLA-DRB1*0901 with microscopic polyangiitis. J Rheumatol 30: 1534–1540, 2003 [PubMed] [Google Scholar]
- 10.Tsuchiya N, Kobayashi S, Hashimoto H, Ozaki S, Tokunaga K: Association of HLA-DRB1*0901-DQB1*0303 haplotype with microscopic polyangiitis in Japanese. Genes Immun 7: 81–84, 2006 [DOI] [PubMed] [Google Scholar]
- 11.Gencik M, Borgmann S, Zahn R, Albert E, Sitter T, Epplen JT, Fricke H: Immunogenetic risk factors for anti-neutrophil cytoplasmic antibody (ANCA)-associated systemic vasculitis. Clin Exp Immunol 117: 412–417, 1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen M, Kallenberg CG: New advances in the pathogenesis of ANCA-associated vasculitides. Clin Exp Rheumatol 27[Suppl 52]: S108–S114, 2009 [PubMed] [Google Scholar]
- 13.Kallenberg CG, Rarok A, Stegeman CA: Genetics of ANCA-associated vasculitides. Cleve Clin J Med 69[Suppl 2]: SII61–SII63, 2002 [DOI] [PubMed] [Google Scholar]
- 14.Luo H, Chen M, Yang R, Xu PC, Zhao MH: The association of HLA-DRB1 alleles with antineutrophil cytoplasmic antibody-associated systemic vasculitis in Chinese patients. Hum Immunol 72: 422–425, 2011 [DOI] [PubMed] [Google Scholar]
- 15.Jennette JC, Falk RJ, Andrassy K, Bacon PA, Churg J, Gross WL, Hagen EC, Hoffman GS, Hunder GG, Kallenberg CG, Mccluskey RT, Sinico RA, Rees AJ, Van Es LA, Waldherr R, Wiik A: Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheum 37: 187–192, 1994 [DOI] [PubMed] [Google Scholar]
- 16.Zhao MH, Lockwood CM: A comprehensive method to purify three major ANCA antigens: Proteinase 3, myeloperoxidase and bactericidal/permeability-increasing protein from human neutrophil granule acid extract. J Immunol Methods 197: 121–130, 1996 [DOI] [PubMed] [Google Scholar]
- 17.Miller SA, Dykes DD, Polesky HF: A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16: 1215, 1988 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Luo H, Chen M, Cui Z, Yang R, Xu PC, Zhou XJ, Zhao MH: The association of HLA-DQB1, -DQA1 and -DPB1 alleles with anti-glomerular basement membrane (GBM) disease in Chinese patients. BMC Nephrol 12: 21, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chen M, Yu F, Zhang Y, Zhao MH: Clinical [corrected] and pathological characteristics of Chinese patients with antineutrophil cytoplasmic autoantibody associated systemic vasculitides: A study of 426 patients from a single centre. Postgrad Med J 81: 723–727, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chen M, Yu F, Zhang Y, Zhao MH: Antineutrophil cytoplasmic autoantibody-associated vasculitis in older patients. Medicine (Baltimore) 87: 203–209, 2008 [DOI] [PubMed] [Google Scholar]
- 21.Jennette JC: Rapidly progressive crescentic glomerulonephritis. Kidney Int 63: 1164–1177, 2003 [DOI] [PubMed] [Google Scholar]
- 22.Hellmich B, Flossmann O, Gross WL, Bacon P, Cohen-Tervaert JW, Guillevin L, Jayne D, Mahr A, Merkel PA, Raspe H, Scott DG, Witter J, Yazici H, Luqmani RA: EULAR recommendations for conducting clinical studies and/or clinical trials in systemic vasculitis: Focus on anti-neutrophil cytoplasm antibody-associated vasculitis. Ann Rheum Dis 66: 605–617, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Luqmani RA, Bacon PA, Moots RJ, Janssen BA, Pall A, Emery P, Savage C, Adu D: Birmingham Vasculitis Activity Score (BVAS) in systemic necrotizing vasculitis. QJM 87: 671–678, 1994 [PubMed] [Google Scholar]
- 24.Benjamini Y, Hochberg Y: Controlling the false discovery rate: A practical and powerful approach to multiple testing. J R Stat Soc Series B Stat Methodol 57: 289–300, 1995 [Google Scholar]
- 25.Benjamini Y, Yekutieli D: The control of the false discovery rate in multiple testing under dependency. Ann Stat 29: 1165–1188, 2001 [Google Scholar]
- 26.Kinikli G, Ateş A, Turgay M, Akay G, Kinikli S, Tokgöz G: HLA-DRB1 genes and disease severity in rheumatoid arthritis in Turkey. Scand J Rheumatol 32: 277–280, 2003 [DOI] [PubMed] [Google Scholar]
- 27.Gonzalez-Gay MA, Garcia-Porrua C, Hajeer AH: Influence of human leukocyte antigen-DRB1 on the susceptibility and severity of rheumatoid arthritis. Semin Arthritis Rheum 31: 355–360, 2002 [DOI] [PubMed] [Google Scholar]
- 28.Shindo Y, Inoko H, Yamamoto T, Ohno S: HLA-DRB1 typing of Vogt-Koyanagi-Harada’s disease by PCR-RFLP and the strong association with DRB1*0405 and DRB1*0410. Br J Ophthalmol 78: 223–226, 1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ralston DR, Marsh CB, Lowe MP, Wewers MD: Antineutrophil cytoplasmic antibodies induce monocyte IL-8 release. Role of surface proteinase-3, alpha1-antitrypsin, and Fcgamma receptors. J Clin Invest 100: 1416–1424, 1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Falk RJ, Terrell RS, Charles LA, Jennette JC: Anti-neutrophil cytoplasmic autoantibodies induce neutrophils to degranulate and produce oxygen radicals in vitro. Proc Natl Acad Sci USA 87: 4115–4119, 1990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Baschal EE, Aly TA, Babu SR, Fernando MS, Yu L, Miao D, Barriga KJ, Norris JM, Noble JA, Erlich HA, Rewers MJ, Eisenbarth GS: HLA-DPB1*0402 protects against type 1A diabetes autoimmunity in the highest risk DR3-DQB1*0201/DR4-DQB1*0302 DAISY population. Diabetes 56: 2405–2409, 2007 [DOI] [PubMed] [Google Scholar]
- 32.Varney MD, Valdes AM, Carlson JA, Noble JA, Tait BD, Bonella P, Lavant E, Fear AL, Louey A, Moonsamy P, Mychaleckyj JC, Erlich H, Type 1 Diabetes Genetics Consortium : HLA DPA1, DPB1 alleles and haplotypes contribute to the risk associated with type 1 diabetes: Analysis of the type 1 diabetes genetics consortium families. Diabetes 59: 2055–2062, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.