Abstract
Wnt5a is a noncanonical member of the Wnt family of signaling molecules that has been linked to various physiological and pathological processes including cell differentiation, cell migration, cell growth, vascular remodeling, cancer and chronic inflammation. To understand the role of Wnt5a in these processes, it is necessary to determine the function and expression level of Wnt5a. In this study we developed a sensitive and specific sandwich enzyme-linked immunosorbent assay (ELISA) for detecting Wnt5a. We found that a rabbit anti-human Wnt5a is a suitable capture antibody for establishing a sandwich ELISA. We used two systems to detect Wnt5a: (1) goat anti-mouse Wnt5a and horseradish peroxidase (HRP) conjugated F(ab’)2 donkey anti-goat IgG as detection and enzyme-linked antibodies respectively, or (2) biotinylated goat anti-mouse Wnt5a and HRP-streptavidin as detection antibody and enzyme-linked avidin respectively. A sandwich ELISA using either of these systems failed to detect recombinant mouse (rm) - Wnt5a diluted in Hank’s balanced salt solution supplemented with Ca2+ and Mg2+ and 1% bovine serum albumin (HBBS+, 1% BSA). Addition of polyethylene glycol (PEG) to the HBBS+, 1% BSA buffer during the binding stage of rm-Wnt5a, afforded the detection of rm-Wnt5a. The use of PEG during both the binding of rm-Wnt5a and detection antibody stages of the assay yielded the maximum signal for rm-Wnt5a. The relationship between the ELISA signal and concentration of Wnt5a was linear with an R2 of 0.9934. In summary, we have developed a specific and sensitive sandwich ELISA that detects rm-Wnt5a.
Keywords: Wnt5a, Sandwich ELISA, Wnt, diagnostics, inflammation, cancer
1. Introduction
Wnts are lipid-modified, small, secreted signaling molecules that are known to be involved in development and disease (Logan and Nusse, 2004; Nusse, 2005; Clevers, 2006). The name Wnt is derived from the combination of names of two homologous genes; wingless (wg) and INT (Hausmann et al., 2007). Wnts regulate cellular interactions during embryogenesis and adult tissue homeostasis, however, disregulation of Wnt signaling has been implicated in cancer and pathological inflammation (Logan and Nusse, 2004; Nusse, 2005; Clevers, 2006). To date 19 mammalian Wnt genes have been identified and are conserved in a variety of species (Nusse, 2005; Clevers, 2006; van Amerongen et al., 2008). Wnts bind to frizzled (Fzd) receptors and stimulate canonical and noncanonical Wnt signaling pathways (Kuhl et al., 2000; Logan and Nusse, 2004; Nusse, 2005; Clevers, 2006). Canonical Wnt signaling is β-catenin dependent and involves transcriptional activation of several genes, some of which have been implicated in cell proliferation and differentiation (Logan and Nusse, 2004; Nusse, 2005; Clevers, 2006). Noncanonical Wnt signaling is β-catenin independent and activates the intracellular release of Ca2+, which in turn activates protein kinase C (PKC) and calcium/calmodulin kinase II (CaMKII) (Kuhl et al., 2000). Noncanonical Wnt signaling has been implicated in the regulation of cellular migration.
Wnt5a, a 38 kDa glycoprotein (Clark et al., 1993), belongs to the noncanonical Wnt family (Kuhl et al., 2000) and has been implicated in a variety of human cancers (Weeraratna et al., 2002; Dejmek et al., 2005; Kurayoshi et al., 2006; Da Forno et al., 2008; Liu et al., 2008; Pilarsky et al., 2008). However, the role of Wnt5a in cancer is controversial. For example, Wnt5a is down regulated in hepatocellular carcinoma (Liu et al., 2008) and Dukes B colon cancer (Dejmek et al., 2005) but upregulated in pancreatic, gastric, and melanoma cancers (Weeraratna et al., 2002; Kurayoshi et al., 2006; Da Forno et al., 2008; Pilarsky et al., 2008). These studies suggest that Wnt5a can mediate different signaling pathways in cancer, which depend on the tumor type. Recently it has been shown that Wnt5a is involved in the motility and invasion of melanoma (Weeraratna et al., 2002) and breast cancer (Pukrop et al., 2006) cell lines. The expression level of Wnt5a in cutaneous melanoma (Da Forno et al., 2008) and gastric cancer (Kurayoshi et al., 2006) has been shown to correlate with tumor grade.
Wnt5a has also been linked to inflammation [reviewed in (George, 2008)]. Several studies have shown that macrophages and dendritic cells upregulate Wnt5a in response to microbial stimuli (George, 2008). Moreover it has been demonstrated that Wnt5a can act both in an autocrine and paracrine fashion to initiate the production of various inflammatory cytokines which are involved in the progression of chronic inflammation (George, 2008). Overexpression of Wnt5a has been observed in various pathological conditions such as rheumatoid arthritis (Sen et al., 2000), psoriasis (Reischl et al., 2007), chronic hepatitis and cirrhosis (Liu et al., 2008), atherosclerosis (Christman et al., 2008) and sepsis (Pereira et al., 2008).
Since Wnt5a plays a key role in many human cancers and pathological inflammation, it could serve as an important diagnostic tool. For example, Wnt5a in serum or proteins extracted from tissue could be used as a biomarker for the detection, characterization and/or staging of cancer and pathological inflammation. Detection of Wnt5a would also advance in vitro studies with cultured cells that seek to understand the regulation of Wnt5a expression. Such diagnostic/prognostic assays would likely be high throughput multi-well plate assays such as an enzyme-linked immunosorbent assay (ELISA). Thus, as a first step along this path, we sought to develop an ELISA that detects Wnt5a protein.
2. Materials and Methods
2.1. Materials
Rabbit polyclonal anti-human Wnt5a was obtained from Santa Cruz Biotechnology (Santa Cruz, CA). Recombinant mouse Wnt5a (rm-Wnt5a) and both biotin conjugated and unconjugated goat polyclonal anti-mouse Wnt5a were purchased from R and D Systems (Minneapolis, MN). Peroxidase conjugated affinity purified F(ab’)2 donkey anti-rabbit IgG and donkey anti-goat IgG were obtained from Jackson ImmunoResearch Laboratories (West Grove, PA). Streptavidin-peroxidase and polyethylene glycol (PEG) polymers were purchased from Sigma (St. Louis, MO). All other reagents were as described previously (Dagia et al., 2004). Washing buffer was Hank’s balanced salt solution (HBSS+) with Ca2+ and Mg2+ supplemented with 1% bovine serum albumin (BSA). Binding buffer was prepared by adding 5% PEG to HBSS+.
2.2. Direct binding ELISA
A direct binding ELISA was used to determine the binding of the Wnt5a antibodies to the 96 well plates (Yolken, 1982). In brief, 50 μL solutions containing anti-human Wnt5a or anti-mouse Wnt5a were added to 96 well plates and incubated overnight at 4°C. The next day, the 96 well plates were washed twice and blocked with washing buffer. Subsequently, the wells were treated with a 50 μL solution containing enzyme-linked antibody (HRP conjugated F(ab’)2 donkey anti-rabbit IgG or donkey anti-goat IgG at 4 μg/ml) for 30 min at 4°C and then washed multiple times with washing buffer. After washing, the wells were treated with 200 μL of OPD buffer and incubated in the dark at 4°C. Subsequently the absorbance (OD 450 nm) of the enzyme-substrate reaction solution was determined at different time points (15, 30, 45, 60 and 120 min). Different time points were used to optimize the signal to noise ratio. The Wnt5a antibodies and enzyme-linked secondary antibodies were diluted in HBSS+, and washing buffer respectively. In every experiment, each condition was performed in triplicate wells.
2.3. Wnt5a sandwich ELISA
A sandwich ELISA was employed to develop an assay that detects rm-Wnt5a (Yolken, 1982). In brief, a 50 μL solution containing the capture antibody (anti-human or anti-mouse Wnt5a at 2.5μg/ml) was added to 96 wells plate and incubated overnight at 4°C. The plates were then washed and blocked with washing buffer. Subsequently, a 50 μL solution containing rm-Wnt5a was added to the wells and the plate incubated at 4°C for 30 min. After washing, a 50 μL solution containing detection antibody (anti-mouse Wnt5a, biotinylated anti-mouse Wnt5a or anti-human Wnt5a at 2.5 μg/ml) was added and incubated at 4°C for 30 min. Subsequent to washing, the wells were incubated with a 50 μL solution containing an enzyme-linked antibody or enzyme-linked avidin at 4°C for 30 min depending on the type of detection antibody used. Following multiple washes, the wells were treated with 200 μL of OPD buffer and incubated in the dark at 4°C. The optical absorbance (OD 450 nm) of each well was determined at different time points (15, 30, 45, 60 and 120 min). Different time points were used to optimize the signal to noise ratio. The capture antibodies were diluted in HBSS+. All other reagents were diluted in HBSS+ or HBSS+, 1% BSA. In certain cases buffers were supplemented with 5% PEG. In every experiment, each condition was performed in triplicate wells.
2.4. Statistical analysis
One-way ANOVA with Tukey-Kramer multiple comparisons test was applied to evaluate the statistical significance among different treatments. A Student’s t-test was used to evaluate the statistical significance between two treatments. In both tests p ≤ 0.05 was considered significant.
3. Results
3.1. Selection of capture antibody
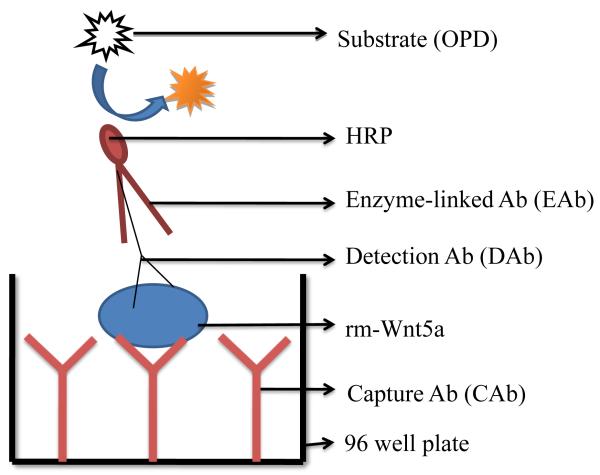
A typical sandwich ELISA has a capture antibody (CAb), a detection antibody (DAb), and an enzyme-linked antibody (EAb), which is schematically shown in Figure. 1. For this technique to be successful, the antigen for the capture and detection antibody must be distinct. We found two antibodies which met this criteria. One, available from Santa Cruz Biotechnology, binds to a 23-80 amino acid sequence at the amino terminus of human Wnt5a, and the other, available from R & D Systems, binds to a 255-335 amino acid sequence at the carboxyl terminus of mouse Wnt5a. Although one antibody was raised against human Wnt5a and the other against mouse Wnt5a, they both should bind to human and mouse Wnt5a based on homology. Human and mouse Wnt5a have 365 and 380 amino acids respectively, and a comparison of the amino acid sequences reveals nearly 98% of the amino acids are conserved (http://www.uniprot.org/uniprot/P41221 and http://www.uniprot.org/uniprot/P22725). Moreover, the amino acid sequence 23-80 in human Wnt5a, an antigen for the rabbit anti-human Wnt5a, matches the amino acid sequence 38-95 in mouse Wnt5a. The amino acid sequence 255-335 in mouse Wnt5a, an antigen for the goat anti-mouse Wnt5a, matches the amino acid sequence 240-320 in human Wnt5a. The amino acid sequence homology between human and mouse Wnt5a suggests that rabbit anti-human Wnt5a antibody will bind to mouse Wnt5a and goat anti-mouse Wnt5a antibody will bind to human Wnt5a. Therefore, we used these two antibodies to develop a sandwich ELISA for detecting Wnt5a.
Figure 1.
Schematic diagram of the sandwich ELISA. The sandwich ELISA used in this study involves (1) immobilization of capture antibody (CAb) to the 96 well plate, (2) capture of rm-Wnt5a via the CAb, (3) detection of rm-Wnt5a by detection antibody (DAb), (4) recognition of DAb by an enzyme-linked antibody (EAb), (5) addition of OPD and (6) optical analysis of signal (colored product) generated from the enzyme-substrate reaction.
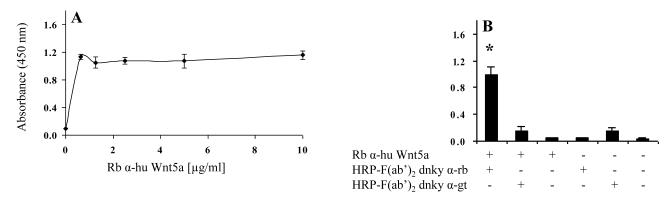
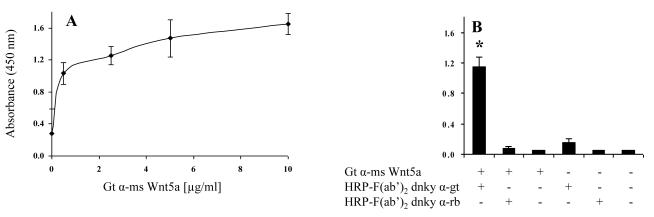
To develop an efficient sandwich ELISA, it is important to determine the level of capture antibody that can be bound to the wells of a multi-well plate. Therefore, we first determined the capture antibody concentration required to saturate the polystyrene surface of the 96 well plates. We coated the wells of 96 well plates with different concentrations of both rabbit anti-human Wnt5a and goat anti-mouse Wnt5a. From Figure 2A and 3A, it is clear that both anti-human Wnt5a and anti-mouse Wnt5a readily bind to 96 well plates at all concentrations tested. To evaluate the statistical significant difference among all treatment groups, one-way ANOVA with Tukey-Kramer multiple comparisons test was used. This analysis indicated that 0.625 μg/ml of anti-human Wnt5a (Fig. 2A) and 2.5 μg/ml of anti-mouse Wnt5a (Fig. 3A) is enough to saturate the wells. Therefore, we used 2.5 μg/ml as the concentration for the capture antibodies.
Figure 2.
(A) Rabbit anti-human Wnt5a readily binds to 96 well plates. The binding of rabbit anti-human Wnt5a to 96 well plates at different concentrations was determined using a direct binding ELISA. HRP conjugated F(ab’)2 donkey anti-rabbit IgG was used to detect the rabbit anti-human Wnt5a. The absorbance was measured after 45 min of the enzyme-substrate reaction. (B) HRP conjugated F(ab’)2 donkey anti-goat IgG does not cross react with rabbit anti-human Wnt5a immobilized on the plate. OD was measured after 15 min of the enzyme-substrate reaction. All values are mean ± SD of triplicate wells. *p≤0.05, compared with the other bars. The results represent one of three experiments.
Figure 3.
(A) Goat anti-mouse Wnt5a readily binds to 96 well plates. The binding of goat anti-mouse Wnt5a to 96 well plates at different concentrations was determined using direct binding ELISA. HRP conjugated F(ab’)2 donkey anti-goat IgG was used to detect the goat anti-mouse Wnt5a. The absorbance was measured after 15 min of the enzyme-substrate reaction. (B) HRP conjugated F(ab’)2 donkey anti-rabbit IgG does not cross react with goat anti-mouse Wnt5a immobilized on the plate. OD was measured after 15 min of the enzyme-substrate reaction. All values are mean ± SD of triplicate wells. * p≤0.05, compared with the other bars. The results represent one of three experiments.
A complexity of sandwich ELISAs is the potential of cross reactivity between the various antibodies used (i.e. CAb, DAb, and EAb). This cross reactivity between antibodies could lead to false positive results. The possible cross reactions that could occur are between capture and detection antibodies, and/or between capture and enzyme-linked antibodies. We first determined whether or not the enzyme-linked antibodies would cross react with the capture antibodies. From Figures 2B and 3B, it appears that the enzyme-linked antibodies do not cross react with the capture antibodies. We next designed a set of experiments to determine if there is cross reactivity between detection and capture antibodies. To test this we used two different sandwich ELISA systems: (i) rabbit anti-human Wnt5a and goat anti-mouse Wnt5a as capture and detection antibodies respectively and HRP conjugated F(ab’)2 donkey anti-goat IgG as the enzyme-linked antibody, and (ii) goat anti-mouse Wnt5a and rabbit anti-human Wnt5a as capture and detection antibodies respectively and HRP conjugated F(ab’)2 donkey anti-rabbit IgG as the enzyme-linked antibody. The results in Figure 4A reveal that goat anti-mouse Wnt5a shows limited cross reactivity with rabbit anti-human Wnt5a adsorbed to the plate. In contrast, and somewhat surprisingly, the results in Figure 4B show that rabbit anti-human Wnt5a used as the detection antibody does cross react with goat anti-mouse Wnt5a used as the capture antibody. Apparently, this pair of antibodies has complementary sites that yield cross-reactivity. These sites are, for the most part, masked/not present when the rabbit anti-human Wnt5a is adsorbed to the plate. Therefore, we selected rabbit anti-human Wnt5a as the capture antibody for further development of the Wnt5a sandwich ELISA.
Figure 4.
Sandwich ELISA to determine whether the DAbs cross react with the CAbs. In each experiment anti-Wnt5a was adsorbed to the wells of the 96 wells plate. Subsequently the wells were treated as listed on the legends. In (A) rabbit anti-human Wnt5a served as the capture antibody and in (B) goat anti-mouse Wnt5a served as the capture antibody. (A) Goat anti-mouse Wnt5a shows limited cross reactivity with rabbit anti-human Wnt5a adsorbed to the plate; (B) Rabbit anti-human Wnt5a cross reacts with goat anti-mouse Wnt5a adsorbed to the plate. OD was measured after 15 min of enzyme-substrate reaction. All values are mean ± SD of triplicate wells. * p≤0.05, compared with the other bars. The results represent one of n≥2 experiments.
3.2. Sandwich ELISA performed using HBSS+, 1% BSA as the buffer does not appear to detect Wnt5a
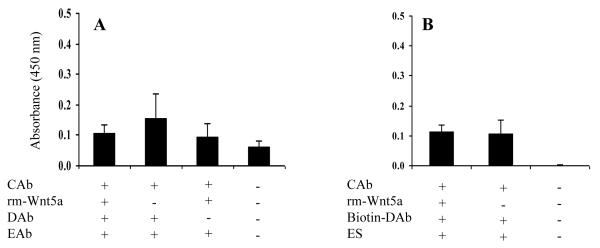
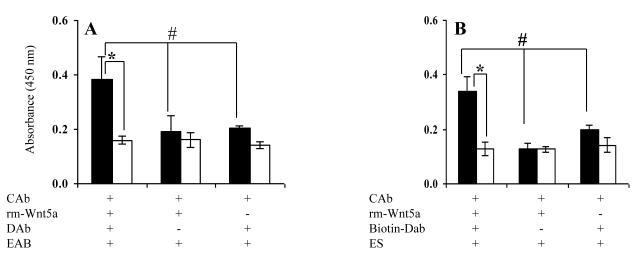
We next performed a sandwich ELISA in an attempt to detect recombinant mouse (rm) - Wnt5a using rabbit anti-human Wnt5a, goat anti-mouse Wnt5a and HRP conjugated F(ab’)2 donkey anti-goat IgG as capture, detection and enzyme-linked antibodies respectively. This assay failed to detect rm-Wnt5a as shown in Figure 5A. In an attempt to increase the sensitivity, we tried to detect rm-Wnt5a using biotinylated goat anti-mouse Wnt5a and HRP conjugated streptavidin as the detection system. Again this assay failed to detect rm-Wnt5a (Fig. 5B).
Figure 5.
Wnt5a sandwich ELISA does not detect rm-Wnt5a. Rabbit anti-human Wnt5a was used as the capture antibody (CAb). (A) Sandwich ELISA with goat anti-mouse Wnt5a (DAb) and HRP-F(ab’)2 donkey anti-goat IgG (EAb). (B) Sandwich ELISA with biotinylated goat anti-mouse Wnt5a (DAb) and HRP-streptavidin (ES). The concentration of rm-Wnt5a was 0.4 μg/ml in (A) and 1 μg/ml in (B). OD was measured after 15 min of enzyme-substrate reaction. All values are mean ± SD of triplicate wells.
3.3. Addition of PEG to the buffer affords detection of Wnt5a
Previous studies have shown that PEG enhances sensitivity of immunoassays (Lizana and Hellsing, 1974; Watanabe et al., 1999; Ochi et al., 2000). Therefore, we next determined if the addition of PEG to the buffer would result in detection of rm-Wnt5a in the sandwich ELISA. A binding buffer was prepared by dissolving 5% PEG powder in HBSS+. We diluted rm-Wnt5a and the detection antibody (i.e. goat anti-mouse Wnt5a or biotinylated goat anti-mouse Wnt5a) in the binding buffer. As shown in Figures 6A and 6B, the addition of PEG to rm-Wnt5a and the detection antibody afforded the detection of rm-Wnt5a.
Figure 6.
PEG affords detection of rm-Wnt5a in the sandwich ELISA. Rabbit anti-human Wnt5a served as the capture antibody (CAb) in both (A) and (B). Goat anti-mouse Wnt5a (DAb) and HRP conjugated F(ab’)2 donkey anti-goat IgG (EAb) detection system was used in (A), and biotinylated goat anti-mouse Wnt5a and HRP-streptavidin detection system was used in (B). Black bar indicates the presence of PEG during the rm-Wnt5a and detection antibody binding stages of the ELISA, white bar indicates no PEG. Concentration of rm-Wnt5a was 1 μg/ml. OD was measured after 2 hrs of enzyme-substrate reaction. * p≤0.05 via Student’s t-test # p≤0.05 via Tukey-Kramer. All values are mean ± SD of triplicates.
3.4. Adding PEG during rm-Wnt5a binding enhances detection, whereas adding PEG during both rm-Wnt5a and detection antibody binding maximizes detection
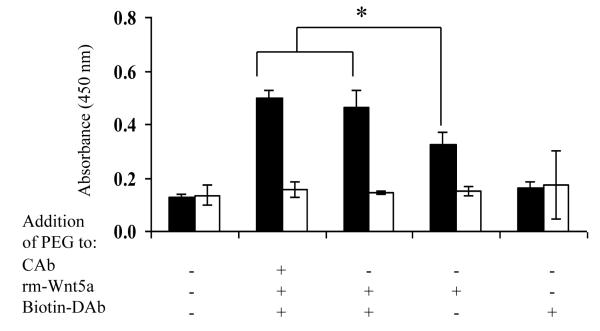
After determining that addition of PEG affords detection of Wnt5a, we next investigated which step in the ELISA protocol required PEG. Thus we tested the effect of PEG in five different ways: (1) no PEG used in the dilution of the capture antibody, rm-Wnt5a and detection antibody (essentially a negative control), (2) PEG used in the dilution of the capture antibody, rm-Wnt5a and the detection antibody, (3) PEG used in the dilution of rm-Wnt5a and the detection antibody, (4) PEG only used in the dilution of the rm-Wnt5a, (5) PEG only used in the dilution of the detection antibody. As shown in Figure 7, it is clear that (i) PEG affords detection of rm-Wnt5a, (ii) the use of PEG in the binding of rm-Wnt5a is required for detection, and (iii) the use of PEG in the binding of rm-Wnt5a and biotinylated goat anti-mouse Wnt5a maximizes the sensitivity.
Figure 7.
The use of PEG in the binding stages of rm-Wnt5a and biotinylated goat anti-mouse Wnt5a is required to achieve maximum sensitivity. PEG was added in the different stages of sandwich ELISA as indicated and rm-Wnt5a was detected using the biotinylated goat anti-mouse Wnt5a (Biotin-DAb) and HRP streptavidin detection system. For each treatment group, the buffer in which the rm-Wnt5a was diluted was used as a negative control for rm-Wnt5a. The black bar indicates rm-Wnt5a in buffer whereas the white bar indicates negative control (no rm-Wnt5a in the buffer). OD was measured after 30 mins of the enzyme-substrate reaction. * p≤0.05, compared with other groups. All values are mean ± SD of triplicates.
3.5. Wnt5a calibration curve
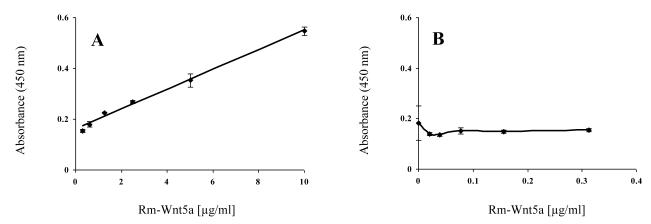
We next investigated the dose response curve in the sandwich ELISA. We used various concentrations of rm-Wnt5a and detected rm-Wnt5a using the above developed sandwich ELISA. The resulting OD for each concentration tested was then plotted against the rm-Wnt5a concentration. Least-squares regression was used to determine the relationship between measured absorbance and rm-Wnt5a concentration. Least-squares regression supports a simple linear dependence of OD on rm-Wnt5a concentration. The relationship is OD = 0.039 [rm-Wnt5a] + 0.1603 with an R2 (coefficient of determinant) = 0.9934 (Fig. 8A). The sandwich ELISA is not able to detect rm-Wnt5a if the concentration of rm-Wnt5a is less than 0.3125 μg/ml (Fig. 8B).
Figure 8.
Titration curve of the sandwich ELISA. The sandwich ELISA, using biotinylated goat anti-mouse Wnt5a and HRP-streptavidin detection system, was used to detect different concentrations of rm-Wnt5a (0 to 10 μg/ml). The absorbance was measured after 2 hrs of the enzyme-substrate reaction. The measured absorbance was plotted against rm-Wnt5a concentration from 0 .3125 to 10 μg/ml (A) and 0 to 0.3125
4. Discussion
In the present study we demonstrated that rm-Wnt5a, spiked into a HBSS+, 1% BSA buffer supplemented with PEG, can be detected and semi-quantified via a sandwich ELISA. The data presented in Figures 2A and 3A demonstrate that both anti-human and anti-mouse Wnt5a are readily absorbed to the wells of 96 well plates. It is reported for a variety of proteins that 50 to 500 ng/well is enough to saturate the wells (Crowther, 2000). In the present study we used 50 μL of a 2.5 μg/ml of capture antibody which corresponds to 125 ng/well. Thus our results are in line with previous observations (Crowther, 2000). Though both Wnt5a antibodies readily bind to the plates, and in theory either could be used as the capture antibody. Our study revealed that the rabbit anti-Wnt5a antibody recognized the goat anti-Wnt5a antibody adsorbed to the 96 well plate (Fig. 4B). Thus it appears to only be feasible to use the rabbit antibody and the goat antibody as the capture and detection antibodies respectively.
The results in Figure 6 demonstrate that the use of PEG during the binding stages of rm-Wnt5a and detection antibody allows the sandwich ELISA to detect rm-Wnt5a. This was true for both the detection systems used in the sandwich ELISA, i.e. goat anti-mouse Wnt5a and HRP-F(ab’)2 donkey anti-goat IgG (Fig. 6A) and biotinylated goat anti-mouse Wnt5a and HRP-streptavidin (Fig. 6B). This finding is consistent with several studies in which it has been reported that PEG increases sensitivity and reaction rates of immunoassays (Lizana and Hellsing, 1974; Watanabe et al., 1999; Ochi et al., 2000). Also, it has been suggested that PEG reorients membrane proteins and lipids and thereby promotes cell fusion (Strasser and Lefkowitz, 1985). Such studies suggest that PEG may modify the three dimensional structure of the Wnt5a and expose the binding sites available to the antibodies, causing an increase in sensitivity of the sandwich ELISA.
Our work is the first step towards developing a sandwich ELISA for the detection of Wnt5a in biological samples. Further development could lead to an efficient system for the detection of Wnt5a in blood samples, tissue samples, cell culture supernatants, and cultured cells. Such an assay could be used as a diagnostic or prognostic. For example, the expression levels of Wnt5a correlate with the tumor grade in cutaneous melanoma (Da Forno et al., 2008) and gastric cancers (Kurayoshi et al., 2006). In addition, our group has shown that Wnt5a is present in atherosclerotic plaques (Christman et al., 2008). Since Wnt5a is a soluble protein, it may be possible to detect vulnerable plaques or vascular disease via detection/quantification of Wnt5a.
Two points that also need to be considered regarding development to a diagnostic are the sensitivity of the assay and whether the assay will work in the presence of serum. To the latter point, we sought to detect rm-Wnt5a in buffers containing serum. In preliminary studies, we found that the sandwich ELISA was unable to detect rm-Wnt5a spiked into buffers containing 10% fetal bovine serum. This result could be due to a variety of factors including: (i) serum immunoglobulins masking the detection of rm-Wnt5a, (ii) serum proteases degrading rm-Wnt5a, (iii) the presence of extracellular Wnt inhibitors such as secreted frizzled-related protein (SFRP) and Wnt inhibitory factor (WIF) (Logan and Nusse, 2004) in the serum, (iv) displacement of capture antibodies by the serum proteins and/or, (v) serum proteins binding to the capture antibody and thus inhibiting the recognition of rm-Wnt5a. Our laboratory is currently working to determine which of these scenarios are operative in the sandwich ELISA conducted in the presence of serum.
In summary, we have developed a sandwich ELISA that detects Wnt5a in buffers. This is the first step towards the development of a high throughput Wnt5a based diagnostic/prognostic assay.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errorsmaybe discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Christman MA, 2nd, Goetz DJ, Dickerson E, McCall KD, Lewis CJ, Benencia F, Silver MJ, Kohn LD, Malgor R. Wnt5a is expressed in murine and human atherosclerotic lesions. Am J Physiol Heart Circ Physiol. 2008;294:H2864–70. doi: 10.1152/ajpheart.00982.2007. [DOI] [PubMed] [Google Scholar]
- Clark CC, Cohen I, Eichstetter I, Cannizzaro LA, McPherson JD, Wasmuth JJ, Iozzo RV. Molecular cloning of the human proto-oncogene Wnt-5A and mapping of the gene (WNT5A) to chromosome 3p14-p21. Genomics. 1993;18:249–60. doi: 10.1006/geno.1993.1463. [DOI] [PubMed] [Google Scholar]
- Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006;127:469–80. doi: 10.1016/j.cell.2006.10.018. [DOI] [PubMed] [Google Scholar]
- Crowther JR. The ELISA guidebook. Methods Mol Biol. 2000;149:III–IV. 1–413. doi: 10.1385/1592590497. [DOI] [PubMed] [Google Scholar]
- Da Forno PD, Pringle JH, Hutchinson P, Osborn J, Huang Q, Potter L, Hancox RA, Fletcher A, Saldanha GS. WNT5A expression increases during melanoma progression and correlates with outcome. Clin Cancer Res. 2008;14:5825–32. doi: 10.1158/1078-0432.CCR-07-5104. [DOI] [PubMed] [Google Scholar]
- Dagia NM, Harii N, Meli AE, Sun X, Lewis CJ, Kohn LD, Goetz DJ. Phenyl methimazole inhibits TNF-alpha-induced VCAM-1 expression in an IFN regulatory factor-1-dependent manner and reduces monocytic cell adhesion to endothelial cells. J Immunol. 2004;173:2041–9. doi: 10.4049/jimmunol.173.3.2041. [DOI] [PubMed] [Google Scholar]
- Dejmek J, Dejmek A, Safholm A, Sjolander A, Andersson T. Wnt-5a protein expression in primary dukes B colon cancers identifies a subgroup of patients with good prognosis. Cancer Res. 2005;65:9142–6. doi: 10.1158/0008-5472.CAN-05-1710. [DOI] [PubMed] [Google Scholar]
- George SJ. Wnt pathway: a new role in regulation of inflammation. Arterioscler Thromb Vasc Biol. 2008;28:400–2. doi: 10.1161/ATVBAHA.107.160952. [DOI] [PubMed] [Google Scholar]
- Hausmann G, Banziger C, Basler K. Helping Wingless take flight: how WNT proteins are secreted. Nat Rev Mol Cell Biol. 2007;8:331–6. doi: 10.1038/nrm2141. [DOI] [PubMed] [Google Scholar]
- Kuhl M, Sheldahl LC, Park M, Miller JR, Moon RT. The Wnt/Ca2+ pathway: a new vertebrate Wnt signaling pathway takes shape. Trends Genet. 2000;16:279–83. doi: 10.1016/s0168-9525(00)02028-x. [DOI] [PubMed] [Google Scholar]
- Kurayoshi M, Oue N, Yamamoto H, Kishida M, Inoue A, Asahara T, Yasui W, Kikuchi A. Expression of Wnt-5a is correlated with aggressiveness of gastric cancer by stimulating cell migration and invasion. Cancer Res. 2006;66:10439–48. doi: 10.1158/0008-5472.CAN-06-2359. [DOI] [PubMed] [Google Scholar]
- Liu XH, Pan MH, Lu ZF, Wu B, Rao Q, Zhou ZY, Zhou XJ. Expression of Wnt-5a and its clinicopathological significance in hepatocellular carcinoma. Dig Liver Dis. 2008;40:560–7. doi: 10.1016/j.dld.2007.12.011. [DOI] [PubMed] [Google Scholar]
- Lizana J, Hellsing K. Polymer enhancement of automated immunological nephelometric analysis, as illustrated by determination of urinary albumin. Clin Chem. 1974;20:415–20. [PubMed] [Google Scholar]
- Logan CY, Nusse R. The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol. 2004;20:781–810. doi: 10.1146/annurev.cellbio.20.010403.113126. [DOI] [PubMed] [Google Scholar]
- Nusse R. Wnt signaling in disease and in development. Cell Res. 2005;15:28–32. doi: 10.1038/sj.cr.7290260. [DOI] [PubMed] [Google Scholar]
- Ochi Y, Kajita Y, Inui T, Yamashiro K, Takasu N, Sato Y, Nagata A. Sensitive thyroid-stimulating antibody assay in whole serum containing five percent polyethylene glycol using porcine thyroid cells. Thyroid. 2000;10:653–7. doi: 10.1089/10507250050137725. [DOI] [PubMed] [Google Scholar]
- Pereira C, Schaer DJ, Bachli EB, Kurrer MO, Schoedon G. Wnt5A/CaMKII signaling contributes to the inflammatory response of macrophages and is a target for the antiinflammatory action of activated protein C and interleukin-10. Arterioscler Thromb Vasc Biol. 2008;28:504–10. doi: 10.1161/ATVBAHA.107.157438. [DOI] [PubMed] [Google Scholar]
- Pilarsky C, Ammerpohl O, Sipos B, Dahl E, Hartmann A, Wellmann A, Braunschweig T, Lohr M, Jesnowski R, Friess H, Wente MN, Kristiansen G, Jahnke B, Denz A, Ruckert F, Schackert HK, Kloppel G, Kalthoff H, Saeger HD, Grutzmann R. Activation of Wnt signalling in stroma from pancreatic cancer identified by gene expression profiling. J Cell Mol Med. 2008;12:2823–35. doi: 10.1111/j.1582-4934.2008.00289.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pukrop T, Klemm F, Hagemann T, Gradl D, Schulz M, Siemes S, Trumper L, Binder C. Wnt 5a signaling is critical for macrophage-induced invasion of breast cancer cell lines. Proc Natl Acad Sci U S A. 2006;103:5454–9. doi: 10.1073/pnas.0509703103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reischl J, Schwenke S, Beekman JM, Mrowietz U, Sturzebecher S, Heubach JF. Increased expression of Wnt5a in psoriatic plaques. J Invest Dermatol. 2007;127:163–9. doi: 10.1038/sj.jid.5700488. [DOI] [PubMed] [Google Scholar]
- Sen M, Lauterbach K, El-Gabalawy H, Firestein GS, Corr M, Carson DA. Expression and function of wingless and frizzled homologs in rheumatoid arthritis. Proc Natl Acad Sci U S A. 2000;97:2791–6. doi: 10.1073/pnas.050574297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strasser RH, Lefkowitz RJ. Homologous desensitization of beta-adrenergic receptor coupled adenylate cyclase. Resensitization by polyethylene glycol treatment. J Biol Chem. 1985;260:4561–4. [PubMed] [Google Scholar]
- van Amerongen R, Mikels A, Nusse R. Alternative wnt signaling is initiated by distinct receptors. Sci Signal. 2008;1:re9. doi: 10.1126/scisignal.135re9. [DOI] [PubMed] [Google Scholar]
- Watanabe Y, Tada H, Hidaka Y, Takano T, Takeoka K, Fukata S, Kuma K, Amino N. Polyethylene glycol increases the detection of anti-thyrotropin receptor antibodies by a radioreceptor assay. Clin Chem. 1999;45:407–9. [PubMed] [Google Scholar]
- Weeraratna AT, Jiang Y, Hostetter G, Rosenblatt K, Duray P, Bittner M, Trent JM. Wnt5a signaling directly affects cell motility and invasion of metastatic melanoma. Cancer Cell. 2002;1:279–88. doi: 10.1016/s1535-6108(02)00045-4. [DOI] [PubMed] [Google Scholar]
- Yolken RH. Enzyme immunoassays for the detection of infectious antigens in body fluids: current limitations and future prospects. Rev Infect Dis. 1982;4:35–68. [Google Scholar]