Abstract
Aurora B (Aur-B) plays multiple roles in mitosis, of which the best known are to ensure bi-orientation of sister chromatids by destabilizing incorrectly attached kinetochore microtubules and to participate in cytokinesis. Studies in Xenopus egg extracts, however, have indicated that Aur-B and the chromosome passenger complex play an important role in stabilizing chromosome-associated spindle microtubules. Aur-B stabilizes spindle microtubules in the egg extracts by inhibiting the catastrophe kinesin MCAK. Whether or not Aur-B plays a similar role in intact oocytes remains unknown. Here we have employed a dominant-negative Aur-B mutant (Aur-B122R, in which the ATP-binding lysine122 is replaced with arginine) to investigate the function of Aur-B in spindle assembly in Xenopus oocytes undergoing meiosis. Overexpression of Aur-B122R results in short bipolar spindles or monopolar spindles, with higher concentrations of Aur-B122R producing mostly the latter. Simultaneous inhibition of MCAK translation in oocytes overexpressing Aur-B122R results in suppression of monopolar phenotype, suggesting that Aur-B regulates spindle bipolarity by inhibiting MCAK. Furthermore, recombinant MCAK-4A protein, which lacks all four Aur-B phosphoryaltion sites and is therefore insensitive to Aur-B inhibition but not wild-type MCAK, recapitulated the monopolar phenotype in the oocytes. These results suggest that in vertebrate oocytes that lack centrosomes, one major function of Aur-B is to stabilize chromosome-associated spindle microtubules to ensure spindle bipolarity.
Keywords: Aur-B, MCAK, Xenopus, meiosis, monopolar spindle
Introduction
A key feature of mitosis is the pre-existence of two dominant microtubule-organizing centers (MTOCs): centrosomes (animal cells) or spindle pole bodies (yeasts). Microtubules emanating from the two centers capture each pair of sister kinetochores in a bipolar manner. Kinetochore microtubules that have strayed over the nearer kinetochore to attach to the farther kinetochore (merotelic attachment) are destabilized by active Aur-B, localized at the inner centromeres and hence sandwiched between the two sister kinetochores.1 Aur-B is thought to destabilize merotelic microtubules via the activity of the microtubule-depolymerizing kinesin MCAK, also localized at centromeres/kinetochores.1-3
Paradoxically, phosphorylation of MCAK by Aur-B inhibits its microtubule-depolymerizing activity.4-6 Studies of spindle assembly in Xenopus egg extracts have indicated that Aur-B is required for assembly of a bipolar spindle.7,8 Aur-B appear to stabilize chromosome-associated microtubules via inhibition of MCAK, since simultaneous inhibition of MCAK restores microtubule stability and normal spindle assembly.9 Similarly, depletion of other components of the CPC, borealin and Incenp, causes destabilization of chromosome-associated microtubules, and simultaneous depletion of MCAK restores microtubule stability and spindle assembly.10 Spindles assembled in Xenopus egg extracts resemble meiotic spindles in intact Xenopus oocytes (as well as in mouse oocytes),11,12 characterized by barrel shape, with chromosomes appearing “imbedded” within the dense microtubule network and with no prominent kinetochore microtubules. Studying spindle microtubule dynamics in the presence of excess MCAK or Op18/Stathmin, another microtubule-destabilizing protein, have revealed that only 5% of microtubules are kinetochore microtubules,13,14 and the rest belong to two functionally distinct non-kinetochore microtubules, polar array microtubules (similar to aster microtubules in mitosis) and barrel array microtubules.14,15 Barrel array microtubules are unique structural components of meiotic spindles. The abundance of barrel array microtubules and their close association with chromosomes gives the unique impression of chromosome “imbedding.” Barrel array microtubules control spindle length and spindle bipolarity. When barrel array microtubules are selectively destabilized by excess Op18/Stathmin or MCAK, the spindle become shorter, and kinetochore microtubules and polar array microtubules become more prominent.13,14 However, Xenopus egg extracts represent post-meiotic cytoplasm and, further, are usually supplemented with sperm nuclei and the associated centrosomes. Therefore, it remains unknown if Aur-B similarly regulates microtubule stability and spindle bipolarity in acentrosomal animal oocytes undergoing meiosis.
To determine if Aur-B is involved in stabilizing spindle microtubules in acentrosomal animal oocytes, we have employed a dominant-negative mutant of Aur-B (Aur-B122R) in combination of imaging live Xenopus oocytes undergoing meiosis.16 We demonstrated that inhibition of Aur-B results in short bipolar spindles or monopolar spindles. We also provide evidence supporting the notion that Aur-B controls spindle bipolarity via inhibition of endogenous MCAK.
Results
Dynamic localization of Aur-A and Aur-B during frog oocyte maturation.
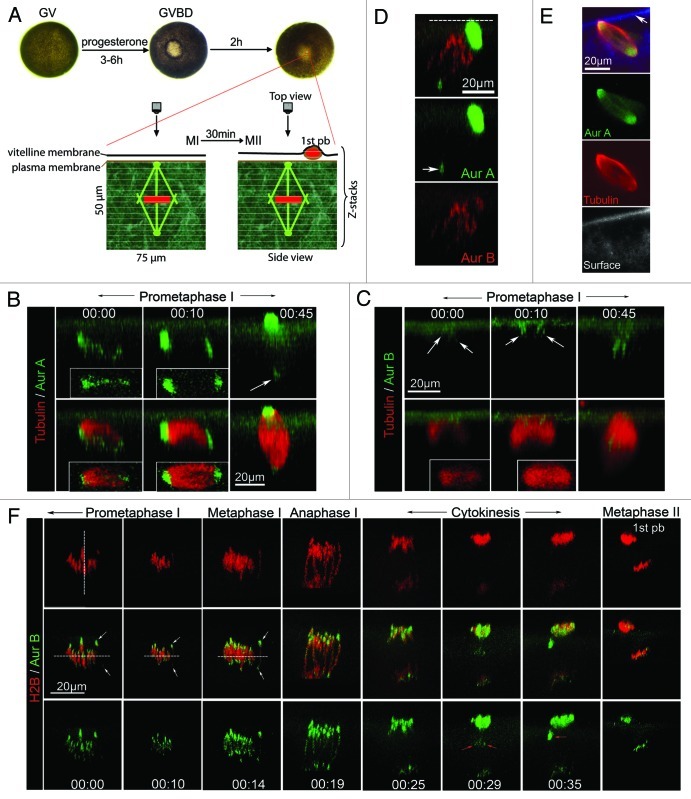
Xenopus oocytes are large and pigmented cells, measuring 1.4 mm in diameter. During oocyte maturation, the animal pole is de-pigmented at the time of GVBD, giving rise to a “maturation spot” (Fig. 1A). This provides a convenient landmark to position the oocyte for live cell imaging.17 We used 4D imaging18 to study the process of meiosis by collecting time series of confocal Z-stacks. After collection, stacks were rendered into 3D representations and, in the figures below, are presented either in the transverse direction (side view) or en face (top view).
Figure 1. Dynamic localization of Aur-A and Aur-B during polar body emission. (A) Live cell imaging during frog oocyte maturation: Prophase (aka germinal vesicle or GV) oocytes were injected with fluorescence probes and stimulated with progesterone to initiate oocyte maturation. Individual ocoytes were picked at GVBD and further incubated for various time (30–110 min) prior to the start of image acquisition by laser scanning confocal microscopy, focusing on a small square (typically 75 μm) centered around the “translucent spot” (visible ~1 h following GVBD, corresponding to spindle pole anchoring site). The Z stacks are volume-rendered into 3D representations. Most images in this paper are 3D images in the transverse direction (side view) with plasma membrane on top, unless otherwise indicated. (B) Aur-A signal transformed from multi-foci to bipolar: Side view of time series (00:00, indicating the start of image collection in hh:mm) of a representative oocyte injected with Alexa488-anti-Aur-A and rhodamin-tubulin-β. Images were acquired with a Z step of 3 μm. Insets show the respective top views. Arrow denotes cytoplasmic pole, which appeared much dimmer than the cortical pole due to its depth (30–40 μm) in the cytoplasm. (C) Aur-B associated with microtubules before spindle became bipolar: Time series (side view) of a representative oocyte injected with Alexa488-anti-Aur-B and rhodamin-tubulin-β. Images were acquired with a Z step of 2 μm. Insets show the respective top views. Arrows denote Aur-B signal. (D) Specificities of Aur-A and Aur-B probes: A representative oocyte injected with Alexa488-anti-Aur-A and Alexa594-anti-Aur-B, depicting the distinct localization of the two Aurora kinases. Only one time point (60 min after GVBD) is shown. Dashed line indicates plasma membrane. Arrow denotes cytoplasmic Aur-A (pole). (E) Spindle image of fixed and bisected oocyte: Uninjected oocytes were fixed 70 min after GVBD and bisected through the small translucent dot (spindle anchoring site) (Fig. 1A). The bisected oocytes were stained with anti-Aur-A and anti-α-tubulin. Single plane confocal images are presented, depicting relatively symmetric distribution of endogenous Aur-A and microtubles along the spindle axis. While Aur-A and tubulin signals are from the same focal plane, the bottom panel represents background fluorescence at a nearby focal plane (the bisection surface), depicting plasma membrane (arrow). (F) Aur-B was most prominently centromeric in metaphase I: Time series (side view) of a representative oocyte injected with RFP-H2B mRNA and Alexa488 anti-Aur-B. Images were acquired at Z step of 0.5 μm and XY pixel of 0.08 x 0.08 μm. Vertical dashed line denotes spindle axis. White arrows denote the two Aur-B domains (centromeres) of the same bivalent, which was used to place the proposed metaphase I plane (horizontal dashed lines). Red arrows denote central spindles (00:29) and midbody (00:35). The last image (metaphase II, 3h after GVBD) was from a different oocyte imaged in the same experiments.
To study spindle assembly in live oocytes, we employed fluorescent tubulin and fluorescently tagged antibodies against Xenopus Aur-A19 and Xenopus Aur-B.16 Anti-Aur-A was used as a potential marker for microtubule organizing center (MTOC)/spindle poles,11 as others have shown that Aur-A is localized at the spindle poles of mouse oocytes.20 Thirty to forty (30-40) min after GVBD, at a time when Aur-B is fully activated21 (Fig. 2A), microtubules accumulate at the animal pole (Fig. 1B and C, 00:00), as previously reported.11 At this stage, Aur-A is distributed diffusely along the length of the prospective spindle (Fig. 1B, 00:00), while a slight enrichment of Aur-B is evident at the point where the microtubules contact the cortex (Fig. 1C, 00:00). Within ~10 min, Aur-A begins to accumulate at the two poles of the nascent spindle (Fig. 1B, 00:10) while the concentration of Aur-B near the middle of the region where the nascent spindle contacts the cortex becomes more pronounced (Fig. 1C, 00:10). The dynamic distribution of Aur-A is reminiscent of the transformation of multi-MTOCs to a bipolar spindle previously described in live mouse oocytes.22 Following spindle rotation,11 Aur-A is most prominent on the pole contacting the cortex but is nevertheless visible on the opposite pole ~30 μm deep in the cytoplasm (Fig. 1B, 00:45), while Aur-B is now concentrated at the middle of the spindle (Fig. 1C, 00:45), consistent with chromosomal localization. The distinct localization of Aur-A and Aur-B is confirmed by imaging experiments in which fluorescent anti-Aur-A and anti-Aur-B are injected in the same oocytes (Fig. 1D). The apparently asymmetrical distribution of Aur-A and microtubule signals in these live images are likely due to the limited tissue penetration capacity of the single photon confocal system and not any spindle asymmetry. This is best illustrated by the symmetrical distribution of endogenous Aur-A and microtubules in oocytes of similar maturation stages when imaging was performed in fixed and bisected oocytes (Fig. 1E).
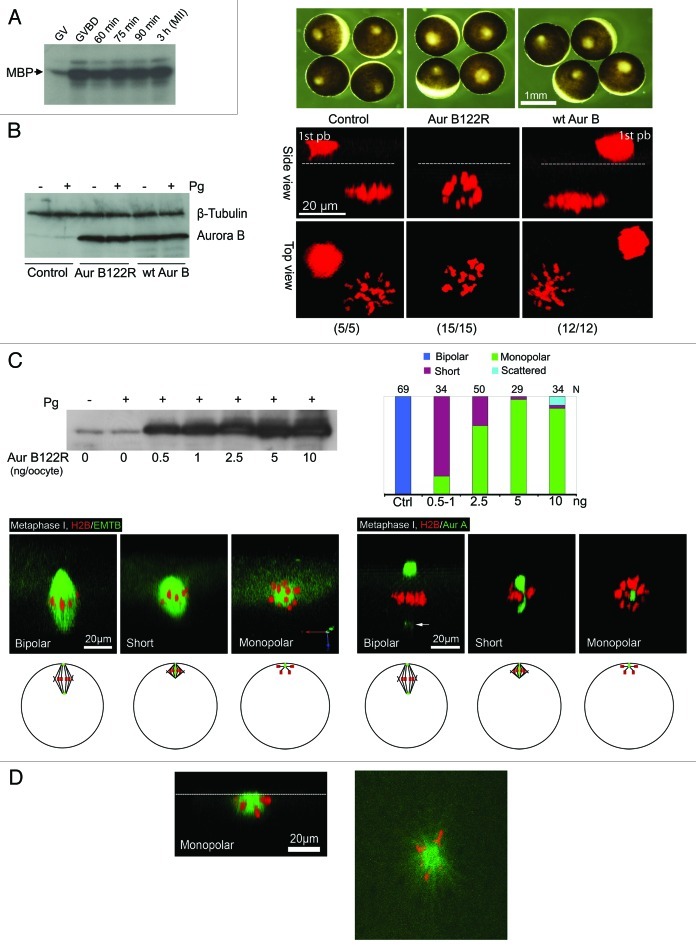
Figure 2. Aur-B122R interferes with spindle bipolarity in frog ocoytes. (A) Aur-B activation during first polar body emission: Oocytes were incubated with progesterone and monitored every 10 min for GVBD. GVBD oocytes were picked out and further incubated for the indicated period of time. Groups of 30 oocytes each were lysed and the extracts were subjected to immunoprecipitation with anti-Aur-B antibodies, followed by in vitro kinase assays using myelin basic protein (MBP) as substrate, as described in our previous publication.19 (B) Overexpression of Aur-B122R, but not WT Aur-B, inhibits polar body emission: Oocytes were injected with RFP-H2B mRNA (control), plus Aur-B122R mRNA (5 ng per oocyte) or WT Aur-B mRNA (5 ng per oocyte) and treated overnight with progesterone. Oocytes were imaged live and each of the representative oocytes is shown in both side view and top view (total numbers indicated). Dash lines denote plasma membrane. 1st pb = first polar body (trapped between the vitelline membrane and plasma membrane, See Fig. 1A). Also shown are bright field images of representative oocytes at time of confocal imaging, as well as a representative immunoblot depicting the levels of overexpression of WT Aur-B and Aur-B122R, compared with endogenous Aur-B (control). β-tubulin blotting served as internal control. (C) Phenotypes of Aur-B122R overexpression: Control oocytes and oocytes injected with the indicated amount of Aur-B122R mRNA, plus the indicated fluorescence probes. Individual oocytes were examined live between 90 and 110 min after GVBD and classified as bipolar (spindle), short (spindle), monopolar (spindle) and scattered (chromosomes). The graph summarizes 20 experiments, with typical images of the indicated phenotypes (“scattered” not shown). The left three images (H2B/EMTB) are 3-D views slightly tilted upward to show that all spindles (bipolar, short and monopolar) were attached to the cortex. Arrow points to Aur-A signal at the cytoplasmic pole. Also shown is a representative anti-Aur-B immunoblot of extracts of control oocytes or oocytes injected with the indicated amounts of Aur-B122R (+ or – progesterone stimulation). Five oocytes were lysed in each group but only 1/5 of the extracts (equivalent to one oocyte) were analyzed. The schematics depict the various phenotypes in intact oocytes (side view), with animal pole at top. Green: Aur-A (spindle pole); red: chromosomes; black lines: microtubules. (D) Surface view of a monopolar Aur-B122R oocyte: The left panel is a 3-D rendered image (side view) with a dash line depicting the oocyte surface. The right panel is a single plane confocal image of the oocyte surface, depicting chromosomes attaching to a central core of microtubules (presumably kinetochore microtubules) and radial polar microtubules growing beyond the chromosomes.
To precisely localize endogenous Aur-B in relation to chromosomes in live oocytes, the distribution of Aur-B was compared with the distribution of a fluorescent histone, RFP-H2B.23 In prometaphase and metaphase I (Fig. 1F, 00:00 to 00:14), endogenous Aur-B can be seen on the whole chromosomes, but is most prominently localized to the two ends of each bivalent. Furthermore, each chromosome bivalent was aligned axial to the spindle axis (vertical dashed line, Fig. 1F, 00:00) with homologous centromeres pointing to the opposite poles. There was progressively more alignment of the two centromere foci of each bivalent to the metaphase plane (horizontal dashed line) as the oocyte approached anaphase. At anaphase, the two sets of chromosomes rapidly and synchronously moved apart toward the opposite poles. Aur-B appeared at central spindles (Fig. 1F, 00:29) and midbody (Fig. 1F, 00:35) as well as remaining associated with the segregating chromosomes, as previously shown.16 At metaphase II, Aur-B remained prominently on chromosomes, particularly at the sister centromeres.
Inhibition of Aur-B caused monopolar spindles.
To directly address potential roles of Aur-B in this system, a dominant-negative Aur-B construct was used. This construct (K122R), in which the catalytically essential lysine (K122) is replaced with arginine, has previously been employed as a potent and specific inhibitor of Aur-B function in mammalian cells.24 A similar dominant-negative mutant for Aur-C has been employed to inhibit Aur-C in mouse oocytes.25 We adopted this approach, because the maternal load of Aur-B is sufficient to prevent successful depletion via morpholinos (unpublished results), and because ZM447439, an often-employed Aur-B inhibitor, is not effective in intact Xenopus oocytes (our unpublished results).26
As a first assessment of the role of Aur-B in meiosis, the distribution of RFP-H2B was monitored in oocytes injected with nothing, mRNA encoding wild type (WT) Aur-B or mRNA encoding the K122R mutant, Aur-B122R. All three groups underwent GVBD (Fig. 2B), and the uninjected or WT Aur-B control oocytes emitted the first polar body and arrested normally at metaphase II (Fig. 2B). In contrast, Aur-B122R oocytes failed to emit the first polar body and, moreover, the side view of their chromosome array indicated that the chromosomes were not properly organized. The difference between WT Aur-B and Aur-B122R cannot be explained based on differences in expression, in that immunoblotting showed equivalent expression of the two constructs (Fig. 2B).
We next sought to characterize in detail the nature of the deficits associated with Aur-B suppression. Figure 2C summarizes the analyses of more than 200 oocytes (in 20 experiments) imaged live between 90–110 min after GVBD when control oocytes and WT Aur-B-injected oocytes assembled a bipolar spindle asymmetrically attached to the cortex (Fig. 2C, bipolar; left three panel depicts chromosomes and microtubules, while right three panel depicts chromosomes and Aur-A). We observed mostly two phenotypes in Aur-B122R-injected oocytes, short spindles or monopolar spindles, correlated with increasing Aur-B122R mRNA and increasing levels of Aur-B122R protein expression (Fig. 2C). By far the most dominant phenotype was a monopolar spindle with all chromosomes attached in a sphere-shaped configuration, similar to that found in Aur-B122R oocytes following overnight incubation (Fig. 2B). In some of these monopolar spindles, prominent aster-like microtubules can be seen (Fig. 2D). With lower concentrations of mRNA, a short spindle became prevalent. In the oocytes exhibiting short spindles, the two spindle poles (marked by the presence of Aur-A) appeared to partially overlap. The pole-to-pole distance (measured from the center of the poles) of these short spindles was 15–20 μm, which was about half the length of the normal bipolar spindles (Fig. 2C). The highest concentration of Aur-B122R mRNA (10 ng per oocyte) sometimes also produced scattered chromosomes at the cortex (not shown).
Aur-B regulates spindle bipolarity by inhibiting the microtubule-depolymerizing kinesin MCAK.
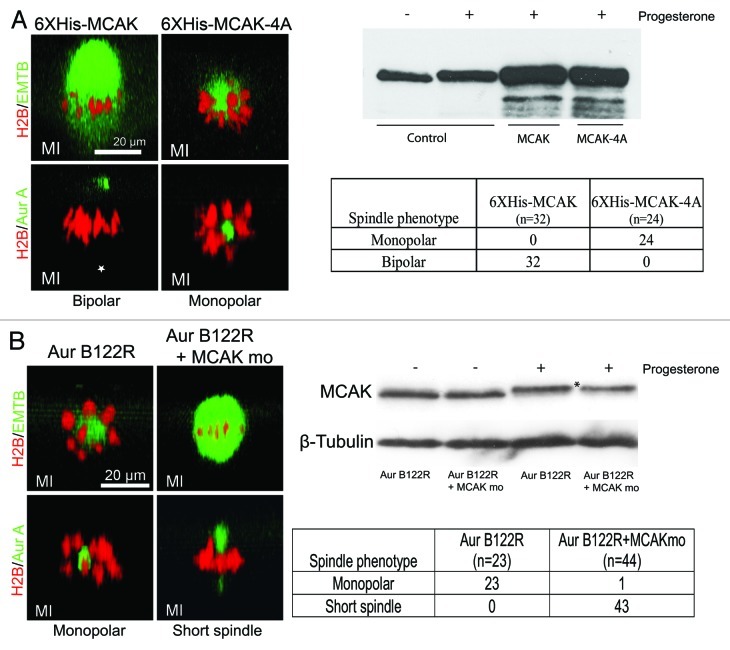
MCAK, a microtubule-depolymerizing kinesin, represents the prime candidate target for Aur-B in regulating spindle bipolarity in frog oocytes. It is present in high concentrations in mature Xenopus eggs13 and co-localizes with, and is phosphorylated and inhibited by, Aur-B.5,6 Furthermore, depletion of components of the CPC in Xenopus egg extracts leads to instability of chromosome-nucleated microtubule, and microtubule stability can be rescued by co-depletion of MCAK.7,10 To examine if MCAK has a role in spindle bipolarity in frog oocytes, we utilized purified WT MCAK as a control, and MCAK-4A, which is insensitive to Aur-B regulation due to substitution of alanine for all of the Aur‑B-phosphorylated residues.6 Oocytes injected with 20 ng purified WT MCAK assembled normal bipolar spindles (Fig. 3A). Strikingly, however, oocytes injected with 20 ng MCAK-4A protein exhibited monopolar meiosis I spindles (Fig. 3A), recapitulating the effect of injecting Aur-B122R mRNA (Fig. 2).
Figure 3. Aur-B regulates spindle bipolarity by suppressing MCAK. (A) MCAK-4A, but not WT MCAK, caused monopolar spindles in meiosis I: Oocytes were injected with RFP-H2B and eGFP-EMTB, or RFP-H2B and Alexa488 anti-Aur A, together with either 20 ng of 6XHis-MCAK or 20 ng of 6XHis-MCAK-4A. The oocytes were treated with progesterone and examined live between 90–110 min after GVBD (prometaphase to metaphase). Shown are typical confocal images (side view) of the bipolar spindles in MCAK oocytes and monopolar spindles in MCAK-4A oocytes, as summarized in the table. Note that the cytoplamic pole (*) of the control oocyte is not visible due to its depth and the relatively weak Aur-A signal. Also shown is a representative anti-MCAK immunoblot of extracts of oocytes injected with 20 ng of 6XHis-MCAK or 6XHis-MCAK-4A, compared with endogenous MCAK (control). Five oocytes were lysed in each group but only 1/5 of the extracts (equivalent to one oocyte) were analyzed. (B) MCAK mo rescued Aur-B122R monopolar phenotype: Oocytes were injected with Aur-B122R mRNA (10 ng per oocyte), together with the appropriate fluorescence probes (RFP-H2B, eGFP-EMTB or Alexa488 anti-Aur A). Half of the oocytes were further injected with MCAK mo (1 mM, 20 nL per oocyte). The oocytes were treated with progesterone and examined live between 90–110 min after GVBD (prometaphase to metaphase). Shown are typical confocal images (side view) of the monopolar spindles in Aur-B122R oocytes and short spindles in oocytes co-injected with Aur-B122R and MCAK mo, as summarized in the table. Also shown is a representative (out of a total of 5 independent experiments) anti-MCAK immunoblot of extracts of oocytes injected with Aur-B122R alone, or together with MCAK mo, following overnight incubation in the presence or absence of progesterone.
To further substantiate the functional relationship between Aur-B and MCAK in frog oocytes, we attempted to rescue Aur‑B122R monopolar phenotype by co-depletion of MCAK. Oocytes were injected with Aur-B122R mRNA (10 ng per oocyte) alone or together with translation-inhibiting morpholino oligos against MCAK (MCAK mo). These experiments indicated that injection of Aur-B122R alone produced mostly monopolar spindles (Fig. 3B, confocal images and table), but oocytes co-injected with Aur-B122R and MCAK mo exhibited mostly short spindles with overlapping Aur-A poles and with greatly increased microtubules, similar to the short spindle phenotype in oocytes injected with less Aur-B122R (Fig. 2C). Immunoblotting confirmed that MCAK mo caused 30.4% (±9.0% in 7 experiments) reduction of MCAK in the oocytes treated with progesterone (Fig. 3B, comparing the left most two lanes). In addition, progesterone also causes a slight upward shift in MCAK mobility in the gel, presumably reflecting hyperphosphorylation during oocyte maturation. These results demonstrated a functional role for Aur-B in regulating spindle bipolarity by suppressing endogenous MCAK in frog oocytes.
Discussion
In this study we have combined live-cell imaging with selective inhibition of Aur-B activity to address the role of this kinase in Xenopus oocyte meiosis. We find that a major role of Aur-B is regulation of meiotic spindle bipolarity via suppression of MCAK activity. This role is supported by the results of specific inhibition of Aur-B; it is supported by contrast to results obtained with specific inhibition of Aur-A via function blocking antibody injection, which impairs meiotic spindle rotation, but not spindle bipolarity in frog oocytes;27 and it is supported by the fact that microinjection of Aur-B non-phosphorylatable, but not wild type MCAK recapitulate the effects of Aur-B inhibition on spindle bipolarity. Finally, a direct functional relationship between Aur-B and MCAK is supported by the partial rescue of Auo-B122R-mediated monopolar phenotype by simultaneous inhibition of MCAK translation during oocyte maturation. That a relatively small (30%) reduction of MCAK caused significant rescue (from monopolar to short bipolar spindles, with dramatic increase of microtubules) (Fig. 3B), and yet oocytes appeared to tolerate additional MCAK (wt) (Fig. 3A), has similarly been noted in studying spindle assembly in Xenopus egg extracts is surprising. Specifically, as little as 25% depletion of MCAK from Xenopus egg extracts is as efficient as 90% depletion in causing dramatic increase of microtubules and disrupting spindle morphology,28 and yet the extracts appear to tolerate excess purified MCAK (wt) in spindle assembly.13
How does Aur-B regulate spindle bipolarity in Xenopus oocytes? We propose that a major function of Aur-B during meiotic spindle assembly is to stabilize barrel array microtubules14,15 by local inhibition of MCAK.9,10 Therefore, inhibition of Aur-B (by overexpressing Aur-B122R) results in unconstraint MCAK activation, which, in turn, destabilizes barrel array microtubules, causing short spindles or monopolar spindles. Overexpression of MCAK-4A causes similarly unconstraint MCAK activation because MCAK-4A is not phosphorylated or inhibited by endogenous Aur-B. Consistent with the selective destabilization of barrel array microtubules in Aur-B122R monopolar oocytes, chromosome bivalents remain attached to a sphere of microtubules (presumably kinetochore microtubules) with a radius roughly half the length of a normal bipolar spindle (Fig. 2C), and polar array microtubules were also evident (Fig. 2D).
Our data do not directly address whether Aur-B is required for bipolar attachment of chromosome bivalents in Xenopus meiosis I, since the appearance of monopolar spindles would have masked this possible defect. Furthermore, generation of monopolar spindles required high levels of Aur-B overexpression relative to the level of endogenous Aur-B (Fig. 2C), which may not be readily achievable in mouse oocytes, providing a possible explanation as to why interfering with CPC function by overexpression of either a kinase deficient Aur-C or an Incenp mutant lacking Aurora kinase binding site,25 or by antisense Incenp morpholino oligos,29 does not lead to monopolar spindles. These interventions, however, cause chromosome misalignment in mouse oocytes, suggesting that CPC may regulate bipolar chromosome attachment in mouse oocytes.25,29 Inhibition of MCAK in mouse oocytes, either by a motor-dead, dominant-negative MCAK mutant or antisense morpholino oligos, also causes chromosome misalignment.30,31 Therefore, whereas unconstrained MCAK (MCAK-4A) destabilizes interpolar microtubules and causes monopolar spindles (Fig. 3A), hypoactive MCAK may cause defects in bipolar chromosome attachment.30,31 Further work will be required to determine how these MCAK functions are coordinated in vertebrate oocytes.
Materials and Methods
Plasmid DNA construction and mRNA synthesis.
Full-length Xenopus Aur-B cDNA32 was PCR-amplified and subcloned into pSP64TM,33generating untagged x-Aur-B or WT Aur-B. A single amino acid substitution, codon AAG (Lys-122) to AGG (Arg), was introduced into WT Aur-B, resulting in Aur-B122R. Other plasmids used in this study have been described in our previous publications.16,34,35 All plasmids were transcribed following linearization using Ambion’s MessageMachine Kit according to manufacturer’s instructions. Messenger RNA (mRNA) was dissolved in water (1 mg/mL) and stored at -80°C. Each mRNA was titrated, but our typical starting concentration was 5 ng per oocyte. Rhodamine tubulin (Cytoskeleton Inc.) was divided in single use aliquots and stored at -80°C. Each aliquot was diluted using the buffer provided by the supplier to 2 mg per ml and between 10–20 nl was injected per oocyte.
Confocal fluorescence imaging of live Xenopus oocytes.
Sexually mature and oocyte-positive Xenopus laevis were purchased from NASCO and maintained and used according to Ottawa Hospital Research Institute’s animal care guidelines. The frogs were injected with pregnant mare serum gonadotropin (PMSG, Sigma, 50 IU/frog) 3–7 d prior to sacrifice and oocyte isolation. Unless otherwise indicated, manually defolliculated oocytes were used in this study. Oocytes were typically injected with mRNA and fluorescence probes the day before imaging experiments and incubated at 20°C in OCM [oocyte culture medium: 60% of L-15 medium (Sigma) supplemented with 1.07 g BSA per L, mixed with 40% autoclaved water to yield the appropriate isotonic solution for amphibian oocytes] plus gentamycin (Sigma, 5 mL/L). Progesterone (1 μM final concentration) was added with the aid of an automatic perfusion system, at least 6 h after mRNA injection. Oocytes exhibiting the white “maturation spot,” indicative of germinal vesicle breakdown (GVBD) (Fig. 1A) were individually transferred to new wells and further incubated in OCM until imaging time. Although individual oocytes vary, often considerably, in the timing of GVBD following the addition of progesterone, we found that they are remarkably synchronized from GVBD to first polar body emission.16,36 Typically, 120 min after GVBD, the oocyte initiates anaphase I. In all figures, time zero (00:00) corresponds to the start of the imaging of the particular oocyte, not GVBD time. Oocytes were imaged with a 60x oil objective on a Zeiss Axiovert with a BioRad 1024 laser scanning confocal imaging system equipped with a krypton argon ion laser. Green fluorophores (eGFP fusion proteins or alexa488-coupled antibodies) were excited with 488 nm laser line, coupled with 522/35 emission filter, and red fluorophores (RFP fusion proteins) were excited with 568 nm line, coupled with 605/32 emission filter. Time-lapse image series were collected at various time intervals (30 sec to 10 min depending on the experiments). Each time point volume was comprised of 15–30 image planes 0.5–3 μm apart (Z-step), at 0.3 μm per pixel (X and Y) (unless otherwise indicated). Image series were 3D-rendered using Volocity Visualization Software (Improvision). Most of the time series in this paper are 3-D images in transverse direction (“side view”), such that the readers are looking “side-on” at the plasma membrane. “Top view” refers to the direct view as seen through the microscope eye piece (Fig. 1A).
Proteins, antibodies and antibody-fluorophore conjugation.
Purified Xenopus MCAK and MCAK-4A (T95A, S110A, S177A and S196A), derived from baculovirus-mediated expression, as well as affinity purified antibodies against Xenopus MCAK, were generous gifts from Dr. Ryoma Ohi.6 We generated anti-Aur-A (against Aur-A N-terminal 123 amino acids) 19 and anti-Aur-B (against Aur-B N-terminal 128 amino acids) 16 antibodies using bacterially expressed GST-fusion proteins containing N-terminal sequences outside of the kinase domains. Unlike the kinase domains of the two proteins, which are related, these N-terminal sequences bear little sequence similarity and therefore the antibodies are highly specific to the respective target kinases, as evident in Figure 1B–E. Affinity purification was performed by absorbing the IgG fractions to the purified antigen immobilized on Sepharose beads. Coupling of affinity purified IgG to Alexa fluorophores (Molecular Probes) was according to manufacturer’s instruction. The conjugated antibodies were diluted to appropriate concentrations in PBS before injection. The following amounts were injected per oocyte of the various fluorescent antibodies: 10 nl of 0.05 mg/mg Alexa 488 anti-Aur A; 10 nl of 0.2 mg/ml Alexa 488 anti-Aur B or Alexa 594 anti-Aur B.
Acknowledgment
This work is supported by an operating grant from the Canadian Institute of Health Research (CIHR) to X.J.L. Julie Delcorde participated in this work as an NSERC summer student (2010). We thank Dr. Ryoma Ohi for purified MCAK proteins and for antibodies against MCAK and Dr. Aaron Straight for RFP-H2B plasmid.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Footnotes
Previously published online: www.landesbioscience.com/journals/cc/article/21016
Reference List
- 1.Tanaka TU, Rachidi N, Janke C, Pereira G, Galova M, Schiebel E, et al. Evidence that the Ipl1-Sli15 (Aurora kinase-INCENP) complex promotes chromosome bi-orientation by altering kinetochore-spindle pole connections. Cell. 2002;108:317–29. doi: 10.1016/S0092-8674(02)00633-5. [DOI] [PubMed] [Google Scholar]
- 2.Lampson MA, Renduchitala K, Khodjakov A, Kapoor TM. Correcting improper chromosome-spindle attachments during cell division. Nat Cell Biol. 2004;6:232–7. doi: 10.1038/ncb1102. [DOI] [PubMed] [Google Scholar]
- 3.Knowlton AL, Lan W, Stukenberg PT. Aurora B is enriched at merotelic attachment sites, where it regulates MCAK. Curr Biol. 2006;16:1705–10. doi: 10.1016/j.cub.2006.07.057. [DOI] [PubMed] [Google Scholar]
- 4.Andrews PD, Ovechkina Y, Morrice N, Wagenbach M, Duncan K, Wordeman L, et al. Aurora B regulates MCAK at the mitotic centromere. Dev Cell. 2004;6:253–68. doi: 10.1016/S1534-5807(04)00025-5. [DOI] [PubMed] [Google Scholar]
- 5.Lan W, Zhang X, Kline-Smith SL, Rosasco SE, Barrett-Wilt GA, Shabanowitz J, et al. Aurora B phosphorylates centromeric MCAK and regulates its localization and microtubule depolymerization activity. Curr Biol. 2004;14:273–86. doi: 10.1016/j.cub.2004.01.055. [DOI] [PubMed] [Google Scholar]
- 6.Ohi R, Sapra T, Howard J, Mitchison TJ. Differentiation of cytoplasmic and meiotic spindle assembly MCAK functions by Aurora B-dependent phosphorylation. Mol Biol Cell. 2004;15:2895–906. doi: 10.1091/mbc.E04-02-0082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gadea BB, Ruderman JV. Aurora kinase inhibitor ZM447439 blocks chromosome-induced spindle assembly, the completion of chromosome condensation, and the establishment of the spindle integrity checkpoint in Xenopus egg extracts. Mol Biol Cell. 2005;16:1305–18. doi: 10.1091/mbc.E04-10-0891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kelly AE, Sampath SC, Maniar TA, Woo EM, Chait BT, Funabiki H. Chromosomal enrichment and activation of the aurora B pathway are coupled to spatially regulate spindle assembly. Dev Cell. 2007;12:31–43. doi: 10.1016/j.devcel.2006.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gadea BB, Ruderman JV. Aurora B is required for mitotic chromatin-induced phosphorylation of Op18/Stathmin. Proc Natl Acad Sci USA. 2006;103:4493–8. doi: 10.1073/pnas.0600702103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sampath SC, Ohi R, Leismann O, Salic A, Pozniakovski A, Funabiki H. The chromosomal passenger complex is required for chromatin-induced microtubule stabilization and spindle assembly. Cell. 2004;118:187–202. doi: 10.1016/j.cell.2004.06.026. [DOI] [PubMed] [Google Scholar]
- 11.Gard DL. Microtubule organization during maturation of Xenopus oocytes: assembly and rotation of the meiotic spindles. Dev Biol. 1992;151:516–30. doi: 10.1016/0012-1606(92)90190-R. [DOI] [PubMed] [Google Scholar]
- 12.Brunet S, Maria AS, Guillaud P, Dujardin D, Kubiak JZ, Maro B. Kinetochore fibers are not involved in the formation of the first meiotic spindle in mouse oocytes, but control the exit from the first meiotic M phase. J Cell Biol. 1999;146:1–12. doi: 10.1083/jcb.146.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ohi R, Burbank K, Liu Q, Mitchison TJ. Nonredundant functions of Kinesin-13s during meiotic spindle assembly. Curr Biol. 2007;17:953–9. doi: 10.1016/j.cub.2007.04.057. [DOI] [PubMed] [Google Scholar]
- 14.Houghtaling BR, Yang G, Matov A, Danuser G, Kapoor TM. Op18 reveals the contribution of nonkinetochore microtubules to the dynamic organization of the vertebrate meiotic spindle. Proc Natl Acad Sci USA. 2009;106:15338–43. doi: 10.1073/pnas.0902317106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yang G, Cameron LA, Maddox PS, Salmon ED, Danuser G. Regional variation of microtubule flux reveals microtubule organization in the metaphase meiotic spindle. J Cell Biol. 2008;182:631–9. doi: 10.1083/jcb.200801105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang X, Ma C, Miller AL, Katbi HA, Bement WM, Liu XJ. Polar body emission requires a RhoA contractile ring and Cdc42-mediated membrane protrusion. Dev Cell. 2008;15:386–400. doi: 10.1016/j.devcel.2008.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Leblanc J, Zhang X, McKee D, Wang ZB, Li R, Ma C, et al. The Small GTPase Cdc42 Promotes Membrane Protrusion during Polar Body Emission via ARP2-Nucleated Actin Polymerization. Mol Hum Reprod. 2011;17:305–16. doi: 10.1093/molehr/gar026. [DOI] [PubMed] [Google Scholar]
- 18.Bement WM, Sokac AM, Mandato CA. Four-dimensional imaging of cytoskeletal dynamics in Xenopus oocytes and eggs. Differentiation. 2003;71:518–27. doi: 10.1111/j.1432-0436.2003.07109005.x. [DOI] [PubMed] [Google Scholar]
- 19.Ma C, Cummings C, Liu XJ. Biphasic activation of Aurora-A kinase during the meiosis I- meiosis II transition in Xenopus oocytes. Mol Cell Biol. 2003;23:1703–16. doi: 10.1128/MCB.23.5.1703-1716.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yao LJ, Zhong ZS, Zhang LS, Chen DY, Schatten H, Sun QY. Aurora-A is a critical regulator of microtubule assembly and nuclear activity in mouse oocytes, fertilized eggs, and early embryos. Biol Reprod. 2004;70:1392–9. doi: 10.1095/biolreprod.103.025155. [DOI] [PubMed] [Google Scholar]
- 21.Yamamoto TM, Lewellyn AL, Maller JL. Regulation of the Aurora B chromosome passenger protein complex during oocyte maturation in Xenopus laevis. Mol Cell Biol. 2008;28:4196–203. doi: 10.1128/MCB.00169-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schuh M, Ellenberg J. Self-organization of MTOCs replaces centrosome function during acentrosomal spindle assembly in live mouse oocytes. Cell. 2007;130:484–98. doi: 10.1016/j.cell.2007.06.025. [DOI] [PubMed] [Google Scholar]
- 23.Erhardt S, Mellone BG, Betts CM, Zhang W, Karpen GH, Straight AF. Genome-wide analysis reveals a cell cycle-dependent mechanism controlling centromere propagation. J Cell Biol. 2008;183:805–18. doi: 10.1083/jcb.200806038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Honda R, Körner R, Nigg EA. Exploring the functional interactions between Aurora B, INCENP, and survivin in mitosis. Mol Biol Cell. 2003;14:3325–41. doi: 10.1091/mbc.E02-11-0769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yang KT, Li SK, Chang CC, Tang CJ, Lin YN, Lee SC, et al. Aurora-C kinase deficiency causes cytokinesis failure in meiosis I and production of large polyploid oocytes in mice. Mol Biol Cell. 2010;21:2371–83. doi: 10.1091/mbc.E10-02-0170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Keady BT, Kuo P, Martínez SE, Yuan L, Hake LE. MAPK interacts with XGef and is required for CPEB activation during meiosis in Xenopus oocytes. J Cell Sci. 2007;120:1093–103. doi: 10.1242/jcs.03416. [DOI] [PubMed] [Google Scholar]
- 27.Castro A, Mandart E, Lorca T, Galas S. Involvement of Aurora A kinase during meiosis I-II transition in Xenopus oocytes. J Biol Chem. 2003;278:2236–41. doi: 10.1074/jbc.M207894200. [DOI] [PubMed] [Google Scholar]
- 28.Walczak CE, Mitchison TJ, Desai A. XKCM1: a Xenopus kinesin-related protein that regulates microtubule dynamics during mitotic spindle assembly. Cell. 1996;84:37–47. doi: 10.1016/S0092-8674(00)80991-5. [DOI] [PubMed] [Google Scholar]
- 29.Sharif B, Na J, Lykke-Hartmann K, McLaughlin SH, Laue E, Glover DM, et al. The chromosome passenger complex is required for fidelity of chromosome transmission and cytokinesis in meiosis of mouse oocytes. J Cell Sci. 2010;123:4292–300. doi: 10.1242/jcs.067447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Illingworth C, Pirmadjid N, Serhal P, Howe K, Fitzharris G. MCAK regulates chromosome alignment but is not necessary for preventing aneuploidy in mouse oocyte meiosis I. Development. 2010;137:2133–8. doi: 10.1242/dev.048306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vogt E, Sanhaji M, Klein W, Seidel T, Wordeman L, Eichenlaub-Ritter U. MCAK is present at centromeres, midspindle and chiasmata and involved in silencing of the spindle assembly checkpoint in mammalian oocytes. Mol Hum Reprod. 2010;16:665–84. doi: 10.1093/molehr/gaq025. [DOI] [PubMed] [Google Scholar]
- 32.Bolton MA, Lan W, Powers SE, McCleland ML, Kuang J, Stukenberg PT. Aurora B kinase exists in a complex with survivin and INCENP and its kinase activity is stimulated by survivin binding and phosphorylation. Mol Biol Cell. 2002;13:3064–77. doi: 10.1091/mbc.E02-02-0092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Booth RA, Cummings C, Tiberi M, Liu XJ. GIPC participates in G protein signaling downstream of insulin-like growth factor 1 receptor. J Biol Chem. 2002;277:6719–25. doi: 10.1074/jbc.M108033200. [DOI] [PubMed] [Google Scholar]
- 34.von Dassow G, Verbrugghe KJ, Miller AL, Sider JR, Bement WM. Action at a distance during cytokinesis. J Cell Biol. 2009;187:831–45. doi: 10.1083/jcb.200907090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Miller AL, Bement WM. Regulation of cytokinesis by Rho GTPase flux. Nat Cell Biol. 2009;11:71–7. doi: 10.1038/ncb1814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ma C, Benink HA, Cheng D, Montplaisir V, Wang L, Xi Y, et al. Cdc42 activation couples spindle positioning to first polar body formation in oocyte maturation. Curr Biol. 2006;16:214–20. doi: 10.1016/j.cub.2005.11.067. [DOI] [PMC free article] [PubMed] [Google Scholar]