Abstract
Pluripotent cells can be derived from different types of somatic cells by nuclear reprogramming through the ectopic expression of four transcription factors, Oct3/4, Sox2, Klf4, and c-Myc. It is however unclear whether post-mitotic neurons are susceptible to direct reprogramming. Here we show that postnatal cortical neurons, the vast majority of which are post-mitotic, are amenable to epigenetic reprogramming. However, ectopic expression of the four canonical reprogramming factors is not sufficient to reprogram postnatal neurons. Efficient reprogramming was only achieved after forced cell proliferation by p53 suppression. Additionally, overexpression of REST, a suppressor of neuronal gene activity, increased reprogramming efficiencies in combination with the reprogramming factors. Our findings indicate that terminally differentiated postnatal neurons are able to acquire the pluripotent state by direct epigenetic reprogramming, and this process is made more efficient through the suppression of lineage specific gene expression.
INTRODUCTION
The return of a somatic epigenome back to an embryonic-like pluripotent state can be achieved through the ectopic expression of the four transcription factors Oct4, Klf4, Sox2, and c-Myc (O, K, S, M), resulting in the formation of induced pluripotent stem (iPS) cells (1–3). The in vitro reprogramming of somatic cells into iPS cells has been achieved in a variety of terminally differentiated somatic cell types including pancreatic β cells (4), keratinocytes (5), hepatocytes (6), and B & T cells (7, 8).
While mounting evidence demonstrates that a diverse array of somatic cells can be reprogrammed, the generation of iPS cells from terminally differentiated post-mitotic cells has not been thoroughly studied due to the heterogeneous donor cell populations and ambiguous genetic markers for post-mitotic cells. Nuclear reprogramming of post-mitotic cells provides critical evidence for the equivalence of genomes from terminally differentiated and ES cells suggesting that the lack of developmental plasticity inherent in terminally differentiated cells is a result of their epigenetic states. In previous studies, the question of whether the epigenome of terminally differentiated post-mitotic olfactory neurons could be reprogrammed to pluripotency has been investigated by somatic cell nuclear transfer (SCNT) experiments (9, 10). However, since nuclear reprogramming by SCNT is a rapid process in which cell division is known not to be required, the underlying molecular mechanism as to how post-mitotic cells re-establish the pluripotent state and why they are less efficient for nuclear reprogramming remains unclear (11).
Mature neurons epitomize a terminally differentiated somatic cell type. Neurons exit the cell cycle, do not proliferate, and are not replaced by precursors in the event of their loss. Although recent studies have described neuronal regeneration from precursor cells located in the subventricular zone (12), this remains a localized phenomenon and not a general characteristic of neurons. Cell cycle regulators and proneural genes are thought to be unnecessary in terminally differentiated non-cycling mature neurons (13). To genetically identify terminally differentiated neurons, we isolated postnatal day 7 (P7) cortical neurons genetically marked by α-Calcium-Calmodulin-dependent Kinase II promoter-driven Cre (CamKII-Cre-159)-mediated activation of a LoxP-Stop-LoxP-enhanced green fluorescent protein (LSL-eGFP) reporter allele whose activity is restricted to terminally differentiated, mature neurons (14).
Here, we assessed the specificity of Cre-mediated recombination in cortical neurons cultured ex vivo and observed that the vast majority of these genetically marked neurons are post-mitotic. We observed that the ectopic expression of the four canonical reprogramming factors is not sufficient to generate iPS cells, to induce significant cell proliferation, or to suppress neuronal gene expression from eGFP+ cortical neurons. We demonstrate that accelerated cell proliferation by suppression of p53 activity coupled with transduction of the reprogramming factors results in efficient iPS formation from eGFP+ postnatal neurons. Furthermore, we find that suppression of neuronal gene activity by overexpression of the Repressor element-1 silencing transcription/neuron-restrictive silencer factor (REST/NRSF) increases reprogramming efficiency.
MATERIALS AND METHODS
Cell culture
Tail tip fibroblasts used to derive primary iPS lines were harvested from crosses between mice homozygous for a α-Calcium-Calmodulin-dependent Kinase II promoter-driven Cre allele (CamKII-Cre-159) (14) and mice carrying LoxP-Stop-LoxP-enhanced green fluorescent protein (LSL-eGFP) reporter allele. ES and established iPS cells were cultured on irradiated MEFs in DME containing 15% FBS, leukemia inhibiting factor (LIF), penicillin/streptomycin, L-glutamine, beta-mercaptoethanol and nonessential amino acids. For the primary postnatal neuronal culture, the highly contributed chimera mice at postnatal day 7 were anesthetized, forebrains were removed, and the cortical plates from the cerebral cortex were dissected and dissociated with Papain Dissociation System (Worthington Biochem. Co) according to the manufacture’s protocol. The dissociated neuronal cells were then plated onto poly-d-lysine/laminin coated plates and cultured in neuronal culture media. To examine the genomic integrity of neurons, we analyzed metaphase spreads of neuron-derived iPS cells for karyotypic abnormalities. Cells were arrested in metaphase with colcemid (0.5ug/mL) for three hours and metaphase spreads were prepared and stained with Geimsa. Analysis of twenty spreads from neuron-derived iPS cell lines showed no evidence of chromosome number alterations, chromosome breaks and chromosome gaps. Based on this analysis we conclude that both the neurons and neuron-derived iPS cells are karytypically normal (Supplemental figure 4b).
Virus generation
Lentiviral preparation and infection with doxycycline (dox)-inducible lentiviruses encoding a polycistronic Oct4, Klf4, Sox2 and c-Myc expression cassette (pHAGE2-tetOminiCMV-STEMCCA) (15) or a constitutive reverse tetracycline transactivator (FUW-M2rtTA) were previously described (16). A p53 KD-lentiviral vector encoding a specific hairpin for the mouse p53 tumour suppressor gene previously described (17), and the mouse REST cDNA were cloned into the EcoRI cloning site of dox-inducible FUW lenti-viral vectors. For lentiviral vector infections of primary fibroblasts, cells were seeded in 6-well plates at a density of 1 × 105 cells per well and infected on three consecutive days. Medium changes were performed 12–24 h after infection. One day after the last infection, ES cell medium containing 2µg/ml dox was added. Fresh ES cell medium with dox was added every other day until iPS cell colonies developed. iPS cell colonies were picked, trypsinized, and grown on MEFs in standard ES cell conditions.
Flow cytometry
For BrdU analysis, we used a Click-iT EdU flow cytometry assay kit (Invitrogen). Cells were resuspended in FACS buffer for analysis on a FACS LSR cell sorter. For live GFP sorting, cells were sorted on a FACS Aria cell sorter
Blastocyst injections
Injections of iPS cells into B6×DBA F2 host blastocysts were carried out as previously described (18).
Alkaline phosphatase staining
Alkaline phosphatase staining was performed using an Alkaline Phosphatase substrate kit (Vector laboratories) according to manufacturer’s manual. For the number of AP+ colonies, equal numbers of cells were plated in the absence or presence of doxycycline on 100-mm dishes coated with gelatin. The number of doxycycline-independent iPS cell colonies that grew after the withdrawal of doxycyclin was determined. iPS cell pluripotency was further validated by immunofluorescence staining for Nanog.
Teratoma assays
iPS cells were collected and separated from feeders by sedimentation of iPS cell aggregates. Cells were washed, resuspended in 500 µl mouse ES cell medium and injected subcutaneously into SCID mice (Taconic). 4 weeks after injection tumors were removed from euthanized mice and fixed in formalin. Samples were paraffin imbedded, sectioned and analyzed on the basis of hematoxylin and eosin stainings.
Bisulfite sequencing
Bisulfite reactions were carried out according to the manufacturer’s instructions (Epitect Bisulfite Kit, Qiagen). 2–4 µl of bisulfite treated DNA was used in a standard PCR protocol to amplify Oct4 and Nanog promoter regions in mouse V6.5 ES cells, neurons and iPS cells. PCR products were cloned into pCR2.1 vectors (Invitrogen) and sequenced using the M13 forward primer. Primers used to amplify the Oct4 and Nanog promoter region were described previously (18).
Immunocytochemistry
Cells were fixed in 4% paraformaldehyde in PBS and immunostained according to standard protocols using the following primary antibodies: SSEA1 (Developmental Studies Hybridoma Bank); SOX2 (R&D Systems); OCT3/4 (Santa Cruz Biotechnology); mouse Nanog (polyclonal rabbit; Bethyl), NeuN (Chemicon), eGFP (Abcam) and appropriate fluorescent secondary antibodies (Invitrogen).
Southern blot analysis
15ug genomic DNA was digested with the indicated restriction enzymes. Electrophoresis and transfer was performed according to standard procedures. The blots were hybridized to radioactively labeled probes against c-Myc, Klf4 & REST. V6.5 ES cells were used as negative controls for determining background and endogenous bands.
Quantitative RT-PCR
Total RNA was isolated using an Rneasy Kit (QIAGEN). One microgram of DNase treated RNA was reverse transcribed using a First Strand Synthesis kit (Invitrogen). Quantitative RT-PCR analysis was performed in triplicate using 1/50 of the reverse transcription reaction in an ABI Prism 7000 (Applied Biosystems, Foster City, CA) with Platinum SYBR green qPCR SuperMix-UDG with ROX (Invitrogen). Gene expression analysis for ES and neuronal markers was performed by RT-PCR. PCR primer sequences are available upon request.
Statistical analysis
Results are given as mean ± S.E.M. Where appropriate, statistical analysis was performed with an analysis of variance (ANOVA) test. The null hypothesis was rejected at the P<0.05 level.
RESULTS
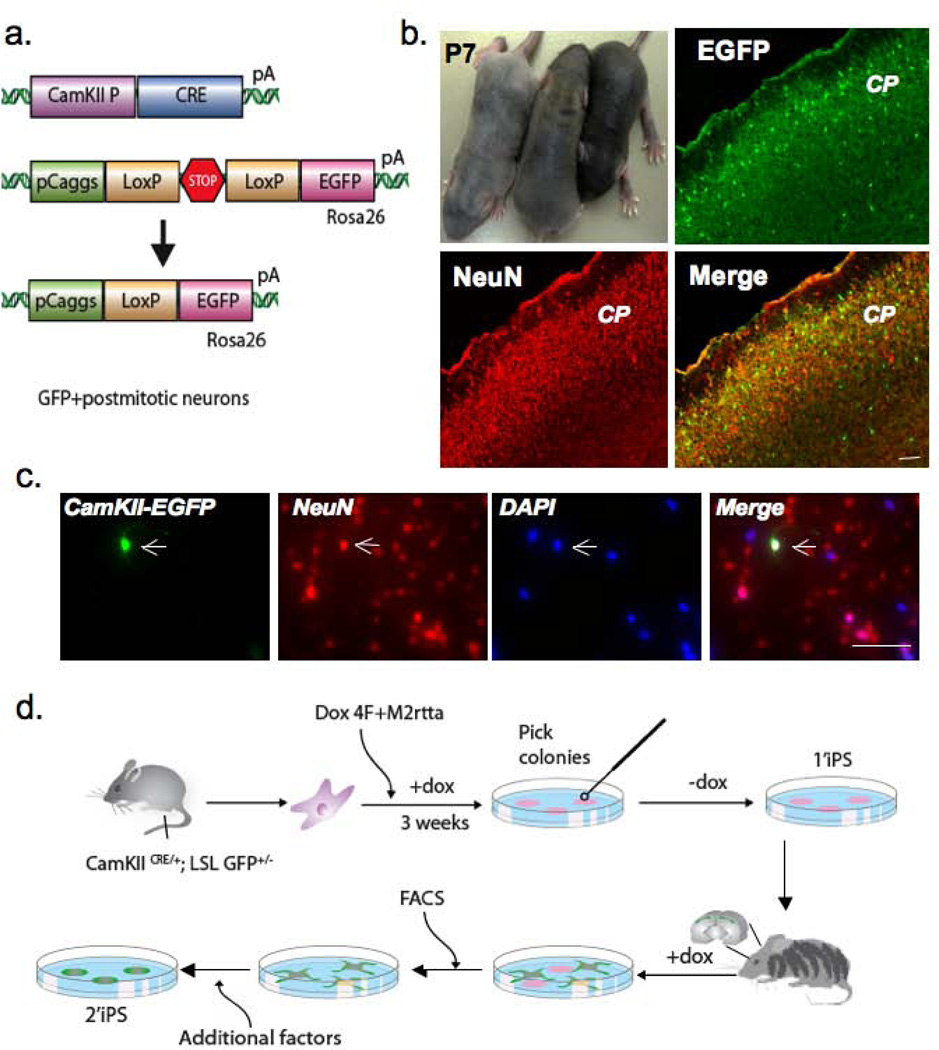
During murine brain development, neuronal precursors in the cerebral cortex are generated in a proliferative area called the subventricular zone (SVZ). After their final cell division in the SVZ, post-mitotic neurons migrate to the cortical plate and form unique laminar structures. Post-mitotic cortical neurons are located in the layer of the cortical plates in the postnatal mouse brain. In order to examine the possibility that postnatal neurons can undergo direct epigenetic reprogramming, we generated mice that harbor both a LSL-eGFP reporter allele and a CamKII-Cre allele (Figure 1a). CamKII promoter activity is restricted to neurons with synaptic function leading to deletion of the LSL cassette and specific expression of eGFP in mature neurons of the adult forebrain and hippocampus (14). This system allows for the selective purification of mature postnatal neurons, and for the retrospective confirmation that the cell of origin of any resulting iPS clones was in fact a mature neuron.
Figure 1. Genetic marking of postnatal neurons.
(a) Strategy for genetic marking of postnatal neurons by CamKII-Cre-mediated activation of a loxP-stop-loxP-eGFP (LSL-eGFP) reporter allele. Cre expression controlled by the CamKII promoter excises the loxp-stop-loxp cassette and irreversibly activates eGFP (b) eGFP labeled postnatal neurons in the cortical plate (CP) of cerebral cortex co-stained with an antibody specific to the post-mitotic neuronal marker NeuN. Scale bars=200µm (c) Immunofluorescence for CamKII-eGFP+ (green) neurons in primary cortical culture double-labeled in red with the post-mitotic neuronal marker NeuN. Scale bars=200µm (d) Strategy for reprogramming mature postnatal neurons using a secondary reprogramming system with polycistronic dox-inducible lentiviral vectors.
Initially we attempted to prepare primary cortical neurons from 2 or 4 week old murine brains but found that neurons cannot be efficiently cultured from animals of this age. Therefore, we isolated primary cortical neurons from postnatal day 7 mice, which is the latest time point at which efficient ex vivo neuronal culture is possible, and examined their expression of eGFP. Figure 1b shows that most eGFP expression observed in the postnatal day 7 mouse forebrain was restricted to the cortical plate (CP) where post-mitotic neurons reside after migration from the SVZ. We also confirmed that the vast majority of eGFP marked cells in the CP are cortical neurons by co-staining with an antibody specific to NeuN, a marker protein restricted to post-mitotic neurons (19), (Figure 1b). Importantly, no eGFP+ cells were observed in the SVZ, where mitotic neural progenitors reside (data not shown). The differentiated state of eGFP+ cortical neurons in primary ex vivo cultures was confirmed by NeuN co-staining (Figure 1c).
Because of the low infection efficiency of postnatal neurons and the heterogeneity of cell types in the primary neuronal cultures, we used a secondary reprogramming system to examine the ability of terminally differentiated postnatal neurons to undergo epigenetic reprogramming by the ectopic expression of the O,K,S,M reprogramming factors (Figure 1d). In this system, somatic fibroblasts are initially transduced with doxycycline-inducible (dox) lentiviral vectors encoding the O,K,S,M reprogramming factors to induce pluripotency (7, 20). The resulting primary iPS cells become independent of ectopic O,K,S,M expression and upon withdrawal of dox can generate chimeras after blastocyst injection. Such chimeras contain populations of 'secondary' somatic cells carrying the identical proviral insertions that generated the primary iPS cells. It has previously been shown that subsequent treatment of such secondary somatic cells with dox can reactivate the reprogramming factors resulting in formation of secondary iPS cells with increased efficiencies (20).
We transduced CamKII-Cre/LSL-eGFP mouse tail tip fibroblasts with the polycistronic dox-inducible lentiviral vector harboring the O,K,S,M reprogramming factors (15, 21) along with a lentiviral vector constitutively expressing the reverse tetracycline transactivator (M2rtTA). After infection, dox was introduced into the culture medium and three weeks later iPS cell clones were isolated. These primary iPS cells were eGFP negative, consistent with the exclusion of postnatal neurons from the fibroblast preparation (Figure 2a). Ten iPS colonies were expanded in the absence of dox, indicating that these cultures had reactivated their endogenous pluripotency regulatory networks, consistent with bisulfite sequence analysis revealing complete demethylation of the endogenous Oct4 and Nanog gene promoters (Figure S1a). To verify the pluripotency of these iPS cell lines, we examined the expression of Oct4, Nanog, and SSEA-1 and found that all dox-independent iPS cell lines expressed these markers of pluripotency (Figure 2a). We further verified the developmental potency of the primary iPS cell lines by subcutaneous injection into immunodeficient (SCID) mice. Four weeks after injection, teratomas were readily visible and histological analysis confirmed the presence of cell types derived from all three embryonic germ layers validating the pluripotency of these primary iPS cells (Figure 2b).
Figure 2. Generation of primary iPS cell lines.
(a) Immunostaining of primary iPS cells derived from CamKII-eGFP tail-tip fibroblasts for the pluripotency markers Oct4, Nanog, SSEA1; these cells were eGFP negative. Scale bars=200µm (b) Teratomas derived from the primary iPS cell line CK-iPS#7. (c) FACS analysis for eGFP and BrdU after 24 hours of BrdU incorporation in postnatal cortical neurons derived from CamKII-eGFP chimeric mice (left panel) compared with BrdU analysis in cultures after 5 day and 9 day cultures with dox (right panels).
We next injected three primary iPS clones (#3, #7, and #9) into blastocysts and generated chimeras comprising secondary somatic cells as well as host blastocyst-derived cells. Primary cortical neuron cultures were derived from the cortical plate of these chimeras’ forebrains at P7 and were immediately exposed to dox for one week after which time we purified eGFP+ cells by FACS to remove host blastocyst-derived cells and contaminating cell types (Figure 2c). Immunostaining confirmed reactivation of the polycistronic reprogramming cassette in eGFP+ secondary postnatal neurons after dox exposure (Figure S1b), with those derived from iPS line #7 showing the most robust activation (Figure S1c). We therefore used secondary postnatal neurons from iPS cell line #7 for subsequent experiments.
We initially confirmed that the eGFP+ secondary postnatal cortical neurons were post-mitotic after isolation and culture by 5-bromodeoxyuridine (BrdU) incorporation. Approximately, 2.43% of total cells in the neuronal culture were negative for BrdU incorporation and positive for EGFP with no appreciable double positive population, indicating their post-mitotic status (Figure 2c left panel). Upon addition of dox to the cultures, a small subset of EGFP+ cells in the secondary system neuronal culture began cycling in response to expression of the four reprogramming factors, approximately 0.43% and 0.57% on days 5 and 9 of dox exposure, respectively (Figure 2c middle and right panels, Sup. figure 2). Thus, these data show that the CamKII-Cre/LSL-eGFP system induces eGFP expression faithfully in the terminally differentiated post-mitotic neurons of the cerebral cortex and that few post-mitotic cortical neurons can re-enter the cell cycle upon exposure to the reprogramming factors using the dox-inducible secondary system.
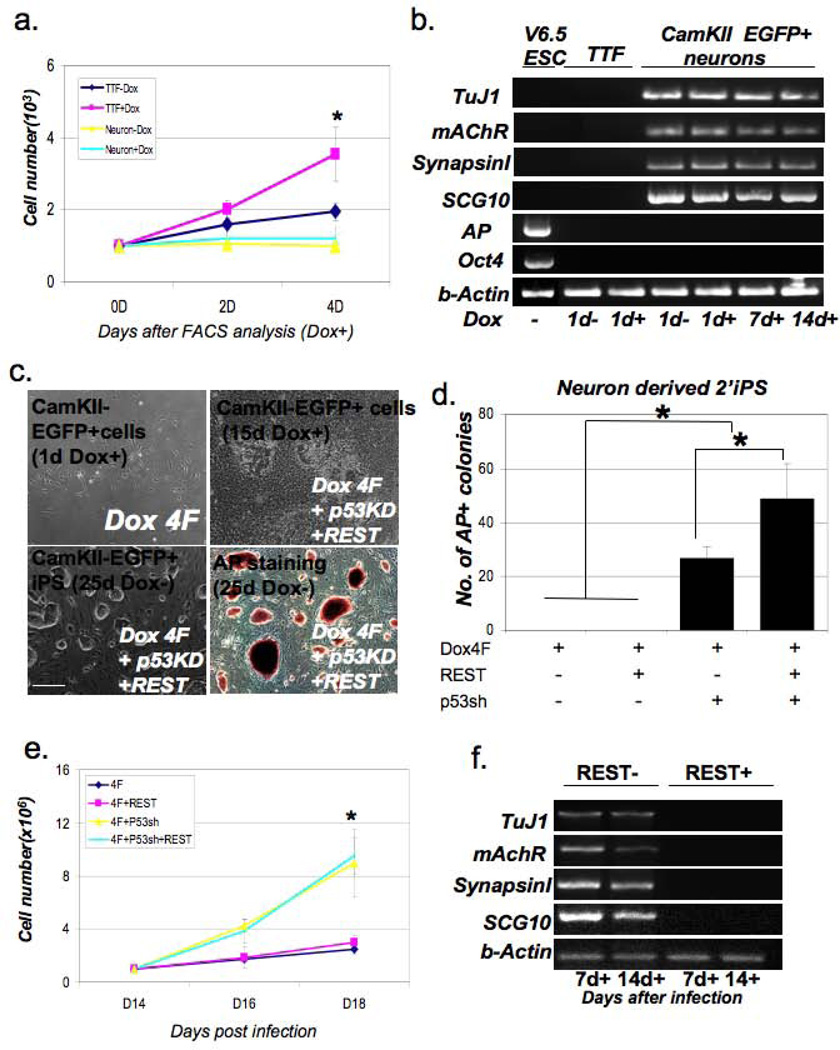
We assessed the reprogramming activity of eGFP+ secondary neurons during two weeks of dox treatment. Initially, secondary CamKII-eGFP+ neurons were treated for 9 days with dox, then FACS purified by gating on eGFP expression, re-plated and cultured in the presence or absence of dox. During this period, FACS-purified secondary neurons underwent little or no proliferation in either the presence or absence of dox, whereas secondary fibroblasts derived from the same mice underwent robust growth in response to dox treatment (Figure 3a). CamKII-eGFP+ postnatal neurons showed no repression of neuronal genes after 14 days of dox treatment (Figure 3b) and no induction of pluripotency associated markers such as Alkaline Phosphatase (AP) or endogenous Oct4 (Figure 3b). We maintained the eGFP+ secondary neurons up to 16 weeks with dox and observed very little proliferation or cell death in these cultures, and no induction of reprogramming (data not shown). These results suggest that the activity of the canonical O,K,S,M reprogramming factors is insufficient to mediate nuclear reprogramming in mature neurons. In contrast to CamKII-eGFP+ postnatal neurons, secondary fibroblasts were able to undergo reprogramming in the presence of dox alone (Figure S3a and b). We therefore considered that additional factors might be necessary for the reprogramming of postnatal neurons, possibly those acting to inhibit neuronal identity. It has been shown that terminally differentiated B lymphocytes can be reprogrammed efficiently after suppression of B cell identity through CEBP alpha overexpression (7), however mature pancreatic beta cells can be reprogrammed by the ectoptic expression of only four reprogramming factors (4). Thus, it remains unclear whether terminally differentiated cells can be efficiently reprogrammed to pluripotency by the four factors. For the reprogramming of postnatal neurons, we chose additional manipulations including p53 inhibition, which is known to accelerate epigenetic reprogramming, and REST overexpression. REST is a well-known negative regulator of neuronal genes that is expressed in non-neuron cells, but turned off in post-mitotic neurons, indicating that it acts as a negative regulator of neuronal identity. Therefore, we reasoned that overexpression of REST may aid in reprogramming through inhibition of neuronal gene expression which might then allow the four factors to establish the pluripotent state.
Figure 3. Direct reprogramming of postnatal CamKII-eGFP+ neurons.
(a) Growth curves for secondary fibroblasts and secondary CamKII-eEGFP+ neurons derived from CK-iPS#7 chimeras in the presence or absence of dox. The secondary CamKII-eGFP+ neurons were FACS purified and re-plated after 9 days of dox treatment, whereas secondary fibroblasts derived from the same mice after 9-day dox treatment were dissociated and equal numbers of cells were plated. Cell numbers were counted 2 and 4 days after plating. Data represent mean ± SEM; three independent experiments were performed. ANOVA test, *P < 0.05. (b) Quantitative RT-PCR analysis of neuronal and ES cell specific gene expression during the reprogramming process. (c) FACS purified CamKII-eGFP+ cells were co-infected with a lentivirus expressing a p53shRNA and a lentivirus expressing REST. Cultures were stained for Alkaline Phosphatase activity 3 weeks after infection. Scale bars=200µm (d) The number of Alkaline Phosphatase (AP) positive colonies from FACS-purified CamKII-eGFP+ cells reprogrammed with p53sh and REST 3 weeks after dox treatment. Equal numbers of cells were plated in the presence of doxycycline and the number of AP+ colonies that grew after the withdrawal of doxycyclin was determined 21 days later. 15 individual AP colonies from each group were validated by immunofluorescence staining for Nanog. Data represent mean ± SEM; six independent experiments were performed with three different primary iPS derived eGFP+ cells; ANOVA test, *P < 0.05. (e) Growth curves for CamKII-eGFP+ neurons infected by the additional factors on dox. Equal numbers of FACS purified cells were infected by the additional lentivirial vectors encoding REST and p53sh RNA one day after plating and cultured in the presence of dox. The cell number was determined 2 weeks later. Data represent mean ± SEM; three independent experiments were performed; (f) RT-PCR analysis of neuronal gene expression during reprogramming of eGFP+ neurons by O,K,S,M and p53 shRNA expression in the presence or absence of REST overexpression.
We asked whether reprogramming could be induced either through accelerated proliferation by inhibition of the p53 tumor suppressor or inhibition of neuronal gene activity by overexpression of REST. Lentiviruses encoding a p53 shRNA construct, and/or REST were introduced into CamKII-eGFP+ cells one day after sorting. We found that overexpression of O,K,S,M reprogramming factors alone in secondary neurons had no effect on the reprogramming process; however, overexpression of the O,K,S,M reprogramming factors in combination with inhibition of p53 and overexpression of REST resulted in efficient iPS cell formation (Figure 3c and 3d). Upon further examination, we found that overexpression of REST alone with O,K,S,M reprogramming factors was insufficient for iPS formation whereas inhibition of p53 alone with O,K,S,M reprogramming factors was sufficient for iPS cell formation (Figure 3d). Overexpression of REST in CamKII-eGFP+ cells infected with p53 shRNA resulted in an approximate two-fold increase in the number of AP+ colonies (Figure 3d). This observation indicates that (a) increased proliferation driven by inhibition of p53 in combination with the O,K,S,M reprogramming factors is required for the reprogramming of mature postnatal neurons and that (b) REST activity can significantly enhance the efficiency of this process, presumably by suppression of neuronal genes. REST overexpression in the secondary fibroblasts did not significantly affect the reprogramming efficiency as expected based on its known role as a suppressor of neuronal identity, while p53 inhibition accelerated reprogramming in secondary fibroblasts as previously reported (17), (Figure S2a).
Consistent with this idea, analysis of cell proliferation during the reprogramming period confirmed that p53 inhibition coupled with O,K,S,M activity was sufficient to induce proliferation in eGFP+ secondary neurons (Figure 3e), and neuronal gene expression was efficiently repressed by the overexpression of REST during reprogramming (Figure 3f). Taken together, these results demonstrate that inhibition of lineage specific gene expression can positively influence the efficiency of the reprogramming process, but only in the context of active proliferation.
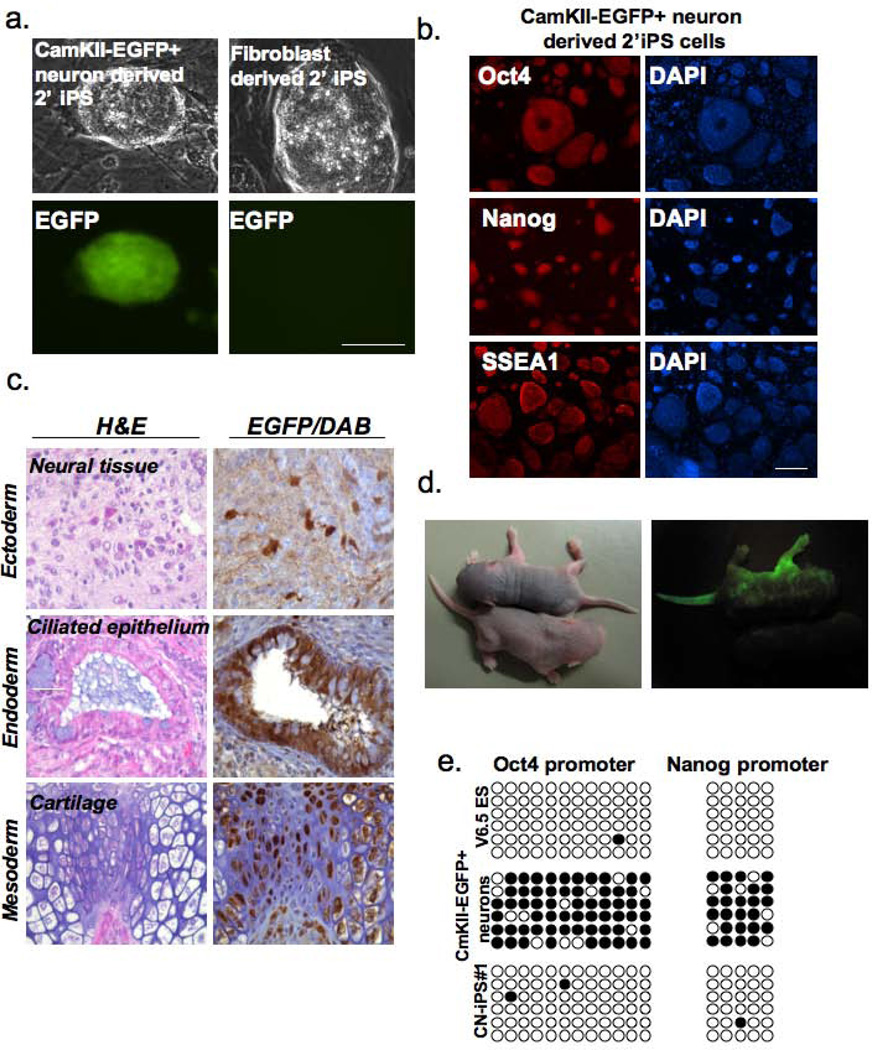
The neuronal origin of the iPS cells resulting from these experiments was confirmed by the expression of eGFP (Figure 4a). The pluripotent state of both eGFP+ secondary neuron-derived iPS cells as well as secondary fibroblast-derived iPS cells was confirmed by their expression of pluripotency markers Oct4, Nanog, and SSEA1, by loss of methylation on the Oct4 and Nanog gene promoters in the absence of dox, by their ability to form differentiated teratomas upon subcutaneous injection into SCID mice, and by their ability to contribute to chimeras upon injection into blastocysts (Figure 4b–e). Neuron-derived secondary iPS cells formed chimeras with similar efficiencies as the primary iPS cells derived from tail-tip fibroblasts or secondary iPS cells derived from secondary tail-tip fibroblasts (Figure 4d). We obtained four chimeric mice from neuron-derived iPS cells, but we were not able to test germ-line for transmission of these chimeras due to early formation of tumors. This accelerated tumorigenesis is likely due to p53 loss rather than an intrinsic property of the iPSCs (17, 22). Southern blot analysis showed that an eGFP+ secondary neuron-derived iPS cell carried four copies of the O,K,S,M lentiviral vector and one copy of the REST lentiviral vector (Figure S4a). Therefore, our results are consistent with the notion that forced cell proliferation is required for the formation of iPS cells from postnatal neurons in response to O,K,S,M expression and that reprogramming is enhanced by inhibition of neuronal gene activity by the Repressor element-1 silencing transcription/neuron-restrictive silencer factor (REST/NRSF).
Figure 4. Developmental potential of secondary iPS cells derived from CamKII-eGFP+ postnatal neurons.
(a) CamKII-eGFP neuron-derived secondary iPS cells are GFP positive, while tail tip fibroblast-derived secondary iPS cells are GFP negative. Scale bars=200µm (b) Immunostaining of CamKII-eGFP neuron-derived secondary iPSCs for the pluripotency markers Oct4, Nanog and SSEA1. Scale bars=200µm (c) Teratomas derived from the secondary iPS cell line shown in (4b). (d) Generation of chimeras from CamKII-eGFP secondary iPS cells by blastocyst injection. The upper chimera exhibits 70–80% chimerism and the lower chimera approximately 10% as judged by GFP fluorescence. (e) Bisulfite sequencing of Oct4 and Nanog gene promoters showing the methylation state of CamKII-eGFP+ secondary iPS cells. Open circles indicate unmethylated and filled circles methylated CpG dinucleotides.
DISCUSSION
In vitro reprogramming by the ectopic expression of defined factors has been achieved in a variety of somatic cell types. However it is not clear from these studies whether reprogramming can be achieved in terminally differentiated neurons. Here, we report that the genome of postnatal neurons from the cerebral cortex that have undergone epigenetic changes associated with terminal differentiation remains amenable to direct epigenetic reprogramming.
We purified and characterized postnatal cortical neurons employing a Lox-Stop-Lox-eGFP::CamKII-Cre transgenic mouse strain for genetic marking. Using BrdU incorporation coupled with flow cytometric analysis, we determined that the vast majority of the marked cells in postnatal cortical cultures derived from these mice were post-mitotic and that about 0.57% of eGFP+ cells became mitotic in response to expression of the four reprogramming factors. Our data also showed that 0.015% of the marked cells were BrdU positive in the absence of reprogramming factor activity, raising the possibility that either a very small number of CamKII-eGFP+ neurons continue to cycle or that non-neuronal cells had been labeled by transgenic expression. This possibility seems unlikely as no iPS colonies could be generated from cortical neurons through expression of the four canonical reprogramming factors alone. To resolve this issue, it would be desirable to prepare neuronal cultures from adult brain. However, at present, no in vitro neuronal culture method for adult neurons exists. Our result suggests that P7 postnatal neurons are amenable to epigenetic reprogramming only after entering a proliferative state mediated by p53 inhibition along with expression of the four canonical reprogramming factors.
We demonstrate that overexpression of the O,K,S,M reprogramming factors is not sufficient to reprogram the genome of postnatal neurons, while most somatic cell types analyzed to date have been amenable to reprogramming by the O, K, S, M factors alone. Recent studies have shown that inhibition of p53 increases the apparent efficiency of generating iPS cells (17, 22–25). Importantly, Hanna et al. (2009) reported that almost all pro-B cells are capable of becoming iPSCs and that p53 inhibition accelerates the process via increasing the rate of cellular proliferation. Thus our findings support a model in which inhibition of p53 improves reprogramming primarily by accelerating the cell cycle.
Additionally, we found that overexpression of REST/NRSF increased the efficiency of the neuronal cell reprogramming without increasing cell proliferation. It has been shown previously that repression of lineage specifying genes can facilitate the reprogramming process (26). Given that REST inhibits neuronal gene expression (27), our data suggest that iPS cell formation not only requires reactivation of pluripotency genes, but can be enhanced by additional repression of lineage specific genes. However, there is also the possibility that REST/NRSF directly participates in the reprogramming process as REST can be directly activated by Wnt signaling in human cancer cell lines and in spinal cord neurons (28, 29). Since Wnt signaling is closely connected to the core circuitry of pluripotency and the reprogramming process (30), overexpression of REST in neurons could directly potentiate the reprogramming process. Thus, it will be of interest to understand the underlying molecular mechanism of REST/NRSF effects on the reprogramming process in the future. Eventually, understanding the molecular barriers underlying nuclear reprogramming in postnatal neurons would provide more efficient reprogramming strategies and is critical for pluripotent cell-based regenerative medicine.
CONCLUSION
In this study we report that terminally differentiated postnatal neurons can be reprogrammed to a pluripotent state by direct reprogramming, but that the canonical four reprogramming factors are not sufficient to induce this process. Rather, inhibition of p53 is required to reprogram postnatal neurons, and REST inhibition can increase the efficiency of this process. Although reprogramming of a variety of somatic cells has been shown, this is the first study to investigate nuclear reprogramming of terminally differentiated post-mitotic cells by direct epigenetic reprogramming, and thus this study provides intriguing data proving that the epigenetic state associated with terminally differentiated cells remain amenable to reprogramming.
Supplementary Material
ACKNOWLEDGMENTS
We thank R. Flannery for veterinary assistance, and B. Carey, D. Hockemeyer, F.Soldner, G. Kemske for comments and G. Welstead for technical assistance and comments. Additionally, we thank T. Dicesare for graphics support. This work was supported by grants from the National Institute of Health (NIH HD045022 and NIH 5R37CA084198).
Footnotes
Author contributions: J.K.: conception and design, provision of study material, collection and/or assembly of data, data analysis and interpretation, manuscript writing; C.L.: provision of study material, data analysis and interpretation, manuscript writing; O.K.: provision of study material, collection and/or assembly of data; J.H.: provision of study materials, collection and/or assembly of data; J.C.: provision of study materials, collection and/or assembly of data; M.L.: provision of study materials, collection and/or assembly of data conception and design, manuscript writing; S.W.: provision of study materials, collection and/or assembly of data conception and design; D.F.: provision of study materials, manuscript writing; E.S.: provision of study materials, collection and/or assembly of data conception and design; Q.G.: provision of study materials, collection and/or assembly of data ; D.F.: provision of study materials; D.M.: provision of study materials; R.J.: conception and design, data analysis and interpretation, manuscript writing.
DISCLOSURE OF POTENTIAL CONFLICTS OF INTEREST
The authors indicate no potential conflicts of interest.
REFERENCES
- 1.Takahashi KS, Yamanaka S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell. 2006;126(4):663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 2.Maherali N, Sridharan R, et al. Directly Reprogrammed Fibroblasts Show Global Epigenetic Remodeling and Widespread Tissue Contribution. Cell Stem Cell. 2007;1(1):55–70. doi: 10.1016/j.stem.2007.05.014. [DOI] [PubMed] [Google Scholar]
- 3.Wernig M, Meissner A, et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007;448(7151):318–324. doi: 10.1038/nature05944. [DOI] [PubMed] [Google Scholar]
- 4.Stadtfeld M, Brennand K, et al. Reprogramming of Pancreatic 2 Cells into Induced Pluripotent Stem Cells. 2008;18(12):890–894. doi: 10.1016/j.cub.2008.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aasen T, Raya A, et al. Efficient and rapid generation of induced pluripotent stem cells from human keratinocytes. Nat Biotech. 2008;26(11):1276–1284. doi: 10.1038/nbt.1503. [DOI] [PubMed] [Google Scholar]
- 6.Aoi T, Yae K, et al. Generation of Pluripotent Stem Cells from Adult Mouse Liver and Stomach Cells. Science. 2008;321(5889):699–702. doi: 10.1126/science.1154884. [DOI] [PubMed] [Google Scholar]
- 7.Hanna J, Markoulaki S, et al. Direct Reprogramming of Terminally Differentiated Mature B Lymphocytes to Pluripotency. Cell. 2008;133(2):250–264. doi: 10.1016/j.cell.2008.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Eminli S, Foudi A, et al. Differentiation stage determines potential of hematopoietic cells for reprogramming into induced pluripotent stem cells. Nat Genet. 2009;41(9):968–976. doi: 10.1038/ng.428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eggan K, Baldwin K, et al. Mice cloned from olfactory sensory neurons. Nature. 2004;428(6978):44–49. doi: 10.1038/nature02375. [DOI] [PubMed] [Google Scholar]
- 10.Li J, Ishii T, et al. Odorant receptor gene choice is reset by nuclear transfer from mouse olfactory sensory neurons. Nature. 2004;428(6981):393–399. doi: 10.1038/nature02433. [DOI] [PubMed] [Google Scholar]
- 11.Simonsson S, Gurdon J. DNA demethylation is necessary for the epigenetic reprogramming of somatic cell nuclei. Nat Cell Biol. 2004;6(10):984–990. doi: 10.1038/ncb1176. [DOI] [PubMed] [Google Scholar]
- 12.Doetsch F, Caillé I, et al. Subventricular Zone Astrocytes Are Neural Stem Cells in the Adult Mammalian Brain. Cell. 1999;97(6):703–716. doi: 10.1016/s0092-8674(00)80783-7. [DOI] [PubMed] [Google Scholar]
- 13.Ohnuma S-i, Harris WA. Neurogenesis and the Cell Cycle. Neuron. 2003;40(2):199–208. doi: 10.1016/s0896-6273(03)00632-9. [DOI] [PubMed] [Google Scholar]
- 14.Minichiello L, Korte M, et al. Essential Role for TrkB Receptors in Hippocampus-Mediated Learning. Neuron. 1999;24(2):401–414. doi: 10.1016/s0896-6273(00)80853-3. [DOI] [PubMed] [Google Scholar]
- 15.Sommer CA, Stadtfeld M, et al. Induced Pluripotent Stem Cell Generation Using a Single Lentiviral Stem Cell Cassette. STEM CELLS. 2009;27(3):543–549. doi: 10.1634/stemcells.2008-1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Brambrink T, Foreman R, et al. Sequential Expression of Pluripotency Markers during Direct Reprogramming of Mouse Somatic Cells. Cell Stem Cell. 2008;2(2):151–159. doi: 10.1016/j.stem.2008.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hanna J, Saha K, et al. Direct cell reprogramming is a stochastic process amenable to acceleration. Nature. 2009;462(7273):595–601. doi: 10.1038/nature08592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wernig M, Meissner A, et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007;448(7151):318–324. doi: 10.1038/nature05944. [DOI] [PubMed] [Google Scholar]
- 19.Mullen RJ, Buck CR, et al. NeuN, a neuronal specific nuclear protein in vertebrates. Development. 1992;116(1):201–211. doi: 10.1242/dev.116.1.201. [DOI] [PubMed] [Google Scholar]
- 20.Wernig M, Lengner CJ, et al. A drug-inducible transgenic system for direct reprogramming of multiple somatic cell types. Nat Biotech. 2008;26(8):916–924. doi: 10.1038/nbt1483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sommer CA, Sommer AG, et al. Excision of Reprogramming Transgenes Improves the Differentiation Potential of iPS Cells Generated with a Single Excisable Vector. STEM CELLS. 2010;28(1):64–74. doi: 10.1002/stem.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hong H, Takahashi K, et al. Suppression of induced pluripotent stem cell generation by the p53-p21 pathway. Nature. 2009;460(7259):1132–1135. doi: 10.1038/nature08235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kawamura T, Suzuki J, et al. Linking the p53 tumour suppressor pathway to somatic cell reprogramming. Nature. 2009;460(7259):1140–1144. doi: 10.1038/nature08311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li H, Collado M, et al. The Ink4/Arf locus is a barrier for iPS cell reprogramming. Nature. 2009;460(7259):1136–1139. doi: 10.1038/nature08290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marion RM, Strati K, et al. A p53-mediated DNA damage response limits reprogramming to ensure iPS cell genomic integrity. Nature. 2009;460(7259):1149–1153. doi: 10.1038/nature08287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mikkelsen TS, Hanna J, et al. Dissecting direct reprogramming through integrative genomic analysis. Nature. 2008;454(7200):49–55. doi: 10.1038/nature07056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schoenherr CJ, Anderson DJ. Silencing is golden: negative regulation in the control of neuronal gene transcription. Current Opinion in Neurobiology. 1995;5(5):566–571. doi: 10.1016/0959-4388(95)80060-3. [DOI] [PubMed] [Google Scholar]
- 28.Willert K, Shibamoto S, et al. Wnt-induced dephosphorylation of Axin releases b-catenin from the Axin complex. Genes Dev. 1999;13:1768–1773. doi: 10.1101/gad.13.14.1768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nishihara S, Tsuda L, et al. The canonical Wnt pathway directly regulates NRSF/REST expression in chick spinal cord. Biochemical and Biophysical Research Communications. 2003;311(1):55–63. doi: 10.1016/j.bbrc.2003.09.158. [DOI] [PubMed] [Google Scholar]
- 30.Marson A, Foreman R, et al. Wnt Signaling Promotes Reprogramming of Somatic Cells to Pluripotency. Cell Stem Cell. 2008;3(2):132–135. doi: 10.1016/j.stem.2008.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.