Abstract
Background
The study was conducted to estimate the effect of depot medroxyprogesterone acetate (DMPA) and oral contraceptives (OC) containing 20 mcg ethinyl estradiol on serum B12 and whether observed changes impact bone mineral density (BMD).
Study Design
Serum B12 and BMD at the lumbar spine and femoral neck were measured on 703 women using OC, DMPA, or nonhormonal (NH) birth control at baseline and every 6 months thereafter for 3 years.
Results
OC and DMPA users experienced greater decreases in B12 than NH users (p<.001). A sharp decrease in B12 was observed during the first 6 months of hormonal contraceptive use (OC: 97 pg/mL and DMPA: 64 pg/mL) in contrast to 14 pg/mL among NH users (20%, 13% and 3% of their baseline values, respectively). Over the following 30 months, B12 levels of OC users remained almost flat while DMPA users had a further 22 units decrease. Very few women demonstrated B12 deficiency. Moreover, B12 levels were not associated with BMD.
Conclusion
Hormonal contraception causes B12 levels to decrease, but this does not appear to be clinically significant or affect BMD.
Keywords: Depot medroxyprogesterone acetate, Depo-Provera, oral contraceptive pills, contraception, birth control, bone density, vitamin B12
1. Introduction
Use of oral contraceptive pills has been associated with lower serum levels of B12 [1–15]. For example, Sütterlin et al [14] observed in a case-control study conducted in Western Europe significantly lower B12 levels in oral contraceptive (OC) users than in controls. A cross-sectional study from the same geographic region also observed similar results [15]. Other studies, however, have failed to find an association between OC and B12 levels making it difficult to determine if a relationship does exist [16–18]. Moreover, these studies were all cross-sectional in design and thus, a cause and effect relationship could not be examined. In addition, studies have not examined the effects of another popular form of birth control, depot medroxyprogesterone acetate (DMPA), on B12 levels. Thus, it is not known whether a progestin-only contraceptive would have similar effects as those reported with use of OC.
If hormonal contraceptive use does lead to lower B12 levels, this could have important clinical implications as several studies on postmenopausal women have shown an association between low B12 levels and low bone mineral density (BMD) [19–23]. A similar association was also shown in one study on adolescents, demonstrating that this may also occur during the reproductive years [24]. However, two other studies did not observe any association between B12 and BMD levels [25, 26]. Thus, it is unclear whether changes in B12 levels which may occur during contraceptive use would actually affect bone density.
To examine the interplays among hormonal contraceptive use, BMD and serum B12, a longitudinal study examining use of these two popular methods with an adequate follow-up period is essential. The purpose of this study was to estimate the effects of OC containing 20 mcg EE and 0.15 mg desogestrel as well as DMPA over 3 years on B12 level, and the role of B12 on the association between hormonal contraceptives and BMD using longitudinal data with a control group.
2. Methods
As part of a larger study, 805 non-Hispanic black, non-Hispanic white, and Hispanic women between 16 and 33 years of age were recruited between October 9, 2001, and September 14, 2004. The methods for the larger study are reported in detail elsewhere [27–29]. Briefly, recruitment was conducted to achieve a sample that was balanced by age group (16–24 years and 25–33 years), race (Black, White, Hispanic) and contraceptive method: nonhormonal (NH), OCs, and DMPA. Of the 805 women who signed a consent form for the larger study, 5 withdrew prior to completing their first visit and 97 were excluded due to abnormal laboratory or bone scan results. Thus, 703 women were invited to participate in the longitudinal study. Those excluded (n=102) did not differ from women included in the longitudinal study (n=703) on age, marital status, parity, or education (all p>.05). Written, informed consent was obtained from all participants and parental consent was obtained for participants <18 years of age. All procedures were approved by the Institutional Review Board at the University of Texas Medical Branch at Galveston.
Following counseling on the different types of contraception available and their efficacies, women were allowed to select one of three types of birth control: 245 chose OC (0.15 mg desogestrel + 20 mcg ethinyl estradiol taken for 21 days, followed by 2 days of placebo and 5 days of 10 mcg ethinyl estradiol); 240 chose DMPA; and 218 chose a NH method (barrier method: 53%; tubal ligation: 33%; copper T:10%; and partner had vasectomy: 4%). Both DMPA and OC were dispensed every 3 months. NH contraceptive users also attended clinic every 3 months and were offered a supply of condoms at each visit. All participants received free well-woman care and contraception during the study as well as monetary compensation. Those who did not return for scheduled visits were reminded by phone and certified letters.
At each 6 month visit, weight was measured with a digital scale accurate to the nearest 0.1 kg while women were wearing light indoor clothing. Height was measured using a wall-mounted stadiometer (Heightronic, Snoqualmie, WA) accurate to the nearest 0.001 m. BMI was calculated as weight (kg)/height (m2). At baseline and every 6 months thereafter for 36 months, BMD of the lumbar spine (L1–L4) and femoral neck was measured using dual-energy X-ray absorptiometry (DXA; Hologic QDR 4500W Elite fan-beam densitometer). A detailed description of how the bone densitometry was performed has been reported previously [27].
Serum levels of vitamin B12 were also measured at baseline and every 6 months thereafter. All blood samples were collected between 7:00 a.m. and 10:00 a.m. after an overnight fast. Assays were performed on the Siemens Advia Centaur Immunoassay system® (Siemans Healthcare Diagnostics Inc., Deerfield, IL) using the Bayer Advia Centaur direct chemiluminescent immunoassay and the manufacturer’s instructions, reagents and calibrators. Calibration was performed every 28 days per manufacturer’s recommendation along with daily quality control for any B12 samples processed. Total %-coefficients of variation ranged from 3.8% to 10.4%. Samples were assayed in batches at the University of Texas Medical Branch laboratory between March and November 2009 after the study was completed.
During the 36-month study period, 257 women were lost to follow-up, 137 women stated they desired a different contraceptive method and 123 women did not complete the study due to other reasons. Thus, 428 (NH 126, DMPA 145, OC 157), 294 (NH 86, DMPA 95, OC 113) and 186 (NH 51, DMPA 58, OC 77) completed 12, 24, and 36 months of follow-up, respectively. There were differences with regard to reasons for discontinuation among the three contraceptive groups: NH users were more likely to be lost to follow-up (NH 44%, DMPA 35%, OCP users 32%, p<.018), whereas DMPA users were more likely than their counterparts to seek a different contraceptive method (DMPA 36%, OC 15%, NH 6%, p<.001). Furthermore, OC users had a higher frequency of discontinuation due to pregnancy or a desire to become pregnant than DMPA users (7% compared with 2%, p<.006), but a similar rate to NH users (7% compared with 5%, p<.281).
2.1. Statistical analysis
One-way analysis of variance with Bonferroni correction for continuous variables and chi-square test for categorical variables were performed to compare the three contraceptive groups at baseline. We used longitudinal analyses to compare changes in vitamin B12 for each contraceptive method, along with their predictors over time. In addition, effect of B12 (exposure variable) as a continuous and categorical variable (quartile) on bone density at the spine and femoral neck (outcome variables) was also examined using the similar model after adjusting for baseline values of bone density. To accommodate the repeated measurements, the data were modeled with the use of a mixed effects regression procedure (xtmixed module; Stata Corporation, College Station, TX), which allowed us to obtain regression coefficients for various predictors while adjusting for the estimated errors for the repeated measurements. This class of model also allows inclusion of time-dependent covariates and accommodates subjects with incomplete data because of variation in number and spacing in observations over the period of follow-up, which frequently occurs in longitudinal studies. During the course of study, women who lost to follow-up, chose the contraceptive method other than that chosen at baseline, and discontinued the study for some other reasons were considered as missing observations in the longitudinal dataset. The primary outcomes were serum B12 levels and BMD at the spine and femoral neck. To examine the overall effect of method, race, and time, our models included contraceptive method (OC/DMPA/NH), race/ethnicity, and duration of contraceptive use (time) as main effects after adjusting for other covariates. Interaction terms (method x race/ethnicity; method x time) were then included in the model. To generate adjusted estimates, variables which were statistically significant at baseline by contraceptive methods were included in the final models. All analyses were performed using Stata 11 (Stata Corporation, College Station, TX).
3. Results
The mean age of the entire sample was 24 (SD: ± 5) years. Twenty-nine percent of the sample (n = 200) was non-Hispanic black, 36% (n = 256) were Hispanic (predominately Mexican American), and 35% (n = 247) were non-Hispanic white. The number of women in each racial/ethnic group and in each age category (16–24 y and 25–33 y) did not significantly differ by contraceptive method (Table 1). Furthermore, there were no significant differences between contraceptive groups in mean baseline height, weight, BMI, total fat mass, percent body fat, age at menarche, and previous use of birth control pills. Three differences were observed between groups at baseline: NH users had a higher mean parity, OC users were less likely to have used DMPA in the past, and DMPA users were more likely to smoke. No difference was observed among the three contraceptive groups with regard to mean serum vitamin B12 level or BMD of the spine or femoral neck at baseline.
Table 1.
Sample characteristics according to contraceptive method selected at baseline
| Characteristic | OC (n = 245) | DMPA (n = 240) | NH (n = 218) |
|---|---|---|---|
| Age, % | |||
| 16–24 years | 54.3 | 56.7 | 45.9 |
| 25–33 years | 45.7 | 43.3 | 54.1 |
| Race, % | |||
| Black | 29.8 | 30.0 | 25.2 |
| White | 33.5 | 34.2 | 38.1 |
| Hispanic | 36.7 | 35.8 | 36.7 |
| Current smoker, % | 23.3§ | 36.3† | 22.0 |
| Weight-bearing exercise >120 min/wk,% | 43.2 | 36.3 | 37.5 |
| Mean (SD) | |||
| Weight, kg | 73.3 (17.7) | 71.8 (19.2) | 73.2 (18.6) |
| BMI, kg/m2 | 27.9 (6.4) | 27.2 (6.9) | 28.3 (7.0) |
| Age at menarche, years | 12.2 (1.5) | 12.5 (1.7) | 12.2 (1.6) |
| Previous use of pill (months) | 21.9 (31.8) | 16.9 (29.6) | 17.7 (28.6) |
| Previous use of DMPA injection (#) | 1.4 (3.5)‡ | 3.5 (6.5)§ † | 2.6 (5.2) |
| Alcohol use, g/day | 1.6 (8.7) | 1.2 (6.1) | 2.3 (14.5) |
| Parity | 0.9 (1.1)‡ | 1.1 (1.2)† | 1.6 (1.5) |
| Serum B12 level | 476.0 (191.0) | 495.8. (190.0) | 492.4 (229.7) |
| Spine BMD, g/cm2, mean (SE) | 1.065 (0.109) | 1.055 (0.112) | 1.051 (0.100) |
| Femoral neck BMD, g/cm2, mean (SE) | 0.921 (0.124) | 0.912 (0.132) | 0.900 (0.114) |
OC = oral contraceptive; DMPA = depot medroxyprogesterone acetate; NH = nonhormonal contraception; BMI = body mass index; SD = standard deviation
One-way analysis of variance with Bonferroni correction was used for continuous variables and chi-square tests were used for categorical variables. To identify specific pairwise differences for categorical variables, we created separate 2x2 tables for each of the pairs and used chi square tests. To adjust for multiple comparison, p<.017 (.05/3) was used to indicate the statistical significance between any two contraceptive groups.
Significant difference was found between OC and DMPA after bonferroni adjustment.
Significant difference was found between DMPA and NH contraception after Bonferroni adjustment.
Significant difference was found between OC and NH contraception after Bonferroni adjustment.
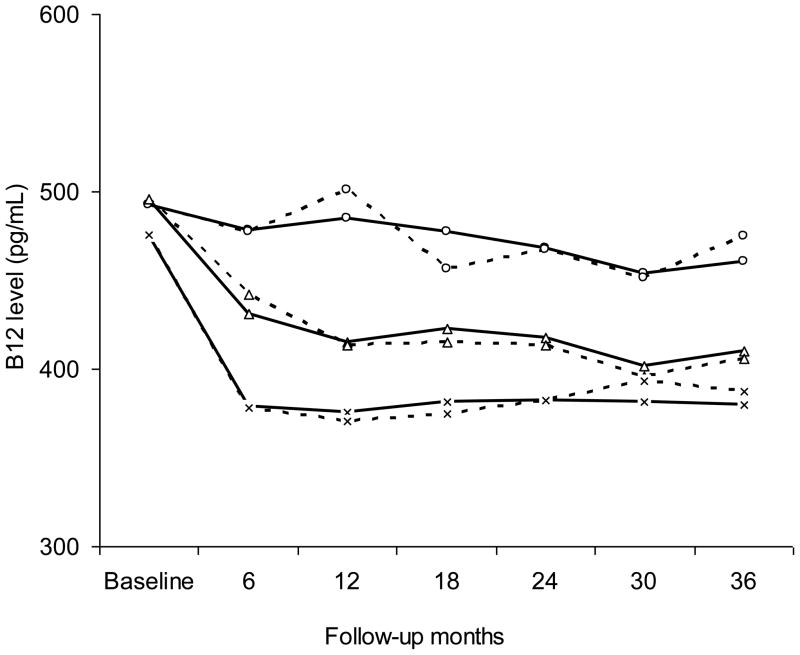
Over 3 years, all three groups experienced a decrease in mean B12. These patterns differed by type of contraception. Among both OC and DMPA users, a marked decrease in mean B12 was observed during the first 6 months of use (OC: 97 pg/mL and DMPA: 64 pg/mL) (20% and 13% of their baseline values, respectively). In contrast, NH users had a mean 14 pg/mL (3% of baseline value) decrease during the first 6 months of follow-up.
The mean B12 level of OC users remained almost flat over the next 30 months while DMPA users experienced a further decrease of 22 pg/mL and NH users had a 17 pg/mL decrease during this same period of time. When the entire 36 months of follow-up was examined, it was observed that OC users had a greater decrease in mean B12 levels than DMPA users (p=.004). However, both DMPA and OC users experienced greater decreases in mean B12 level than NH users (p<.001; Table 2, Fig. 1).
Table 2.
| NH | DMPA | OC | p value | ||||
|---|---|---|---|---|---|---|---|
|
| |||||||
| N | Mean | N | Mean | N | Mean | ||
| Baseline | 215 | 492.4 | 238 | 495.8 | 238 | 476.0 | NH vs. OC: <.001 |
| 6 mo | 146 | 478.1 | 171 | 431.5 | 162 | 378.8 | |
| 12 mo | 128 | 484.5 | 133 | 415.3 | 140 | 375.4 | NH vs DMPA: <.001 |
| 18 mo | 131 | 477.6 | 94 | 422.5 | 114 | 381.2 | |
| 24 mo | 112 | 468.2 | 69 | 417.5 | 104 | 382.5 | OC vs. DMPA: =.004 |
| 30 mo | 89 | 454.2 | 55 | 401.8 | 81 | 381.6 | |
| 36 mo | 79 | 460.9 | 31 | 409.9 | 67 | 379.9 | |
OC = oral contraceptive; DMPA = depot medroxyprogesterone acetate; NH = nonhormonal contraception.
Adjusted by baseline value of B12, BMI weight status [(time varying variable; normal weight (BMI: <25 kg/m2), overweight (BMI: 25–29.9 kg/m2), and obese (BMI: 30 or above kg/m2)], age (16–24 years vs. 25–33 years), race/ethnicity (white, black, and Hispanic), parity (continuous), smoking status (current smoker vs. not current smoker), and previous use of DMPA.
Mixed-model regression analyses were used for the adjustment.
B12 level was not available for some participants although other follow-up data were available (12, 0, 11, 4, 2, 4, and 2 at baseline and other follow-up visits, respectively)
Fig. 1.
Modeled mean change of B12 from baseline across 36 months by contraceptive method. Empty circles: NH contraceptives; X shape: OC, and empty triangles: DMPA Solid lines represent the estimated mean changes, and dotted lines represent the unadjusted values.
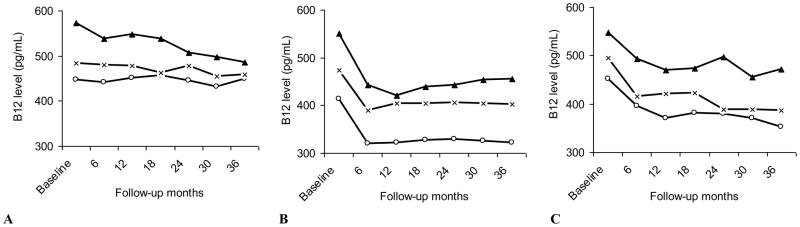
Racial differences were also observed as non-Hispanic black and Hispanic women had higher levels of mean B12 at baseline and after 36 months of use than white women, irrespective of contraceptive methods. Black women had a higher mean B12 level at baseline compared to white and Hispanic women (white; 437 pg/mL, black: 556 pg/mL, and Hispanic: 484pg/mL; p<.001 for both) while Hispanic women had significantly higher B12 levels than whites (p<.01) (Fig. 2). Similar significant differences in mean B12 levels by ethnicity were also observed after 36 months, irrespective of contraceptive use. Women of all racial/ethnic groups who had a higher B12 level at baseline were more likely to have a higher level after 36 months. No other covariates were significantly associated with B12 levels over 36 months of contraceptive use.
Fig. 2.
Modeled mean change of B12 level from baseline across 36 months by race/ethnicity and contraceptive method: (A) B12 level – NH method; (B) B12 – level OC; and (C) B12 – level DMPA.
Empty circles: white women; solid triangles: black women; X shape: Hispanic women.
Adjusted by baseline value of B12, BMI weight status [(time varying variable; normal weight (BMI: <25 kg/m2), overweight (BMI: 25–29.9 kg/m2), and obese (BMI: 30 or above kg/m2)], age (16–24 years vs. 25–33 years), race/ethnicity (white, black, and Hispanic), parity (continuous), smoking status (current smoker vs. not current smoker), and previous use of DMPA.
We also examined the effect of B12 on BMD changes at the lumbar spine and femoral neck over 3 years using the mixed model regression analysis after adjusting for age, whole body lean mass, duration of use, contraceptive methods, race/ethnicity, parity, smoking status, and previous use of DMPA. No significant associations were observed between mean B12 levels (continuous data) and mean BMD at the spine (p=.107) or femoral neck (p=.877) after adjusting for different covariates.
Very few participants had abnormally low (<180 pg/mL) levels of B12 (NH 1, DMPA 2, OC 0) at baseline which remained low at subsequent visits. Overall, 4 NH users (2% of total NH users at baseline) developed an abnormally low level of B12 during the follow-up period. Two became normal at a subsequent visit, while the other two had abnormally low levels at 30 and 36 months only and then were lost to follow-up. Study stopped at 36 months. Two DMPA users (1% of total DMPA users) had abnormally low B12 levels at baseline, of which one became normal at a later visit and the other was lost to follow-up after an abnormal result at 18 months. With regards to OC users, 9 (4% of total OC users) had abnormally low B12 levels during the follow-up period of which 4 became normal at a later visit, 4 were lost to follow-up, and one remained abnormally low over the 36 months of follow-up.
4. Discussion
In this study, we observed that use of both OC and DMPA resulted in lower mean serum levels of vitamin B12. However, levels still remained in the normal range for almost all users. By and large, these findings agree with previous cross-sectional and case-control studies on the effect of OC on serum B12 levels [1–15]. This longitudinal study adds to the literature by demonstrating a cause and effect relationship between OC use and serum B12 levels. In addition, we observed lower B12 levels among DMPA users, which has not been previously reported in the literature.
It is important to note that even though B12 levels decreased, very few OC or DMPA users demonstrated abnormally low B12 levels during the 3-year follow-up period. We observed only 4% of OC users had an abnormal B12 level during follow-up, of which almost half demonstrated a return to normal at a subsequent visit. This is much lower than reported in the case-control study conducted by Sütterlin et al. [14], which similar to our study, also examined third generation OC users. In that study, 13% of OC users had frank B12 deficiency (<180 pg/mL) while another 15% demonstrated subnormal (180–223 pg/mL) B12 levels, although none in either group developed clinical symptoms. It is possible that dietary differences in the populations studied could have contributed to differences in findings between their study and ours. Differences in assay methods (intrinsic factor-coated micro-particle enzyme immunoassay vs. direct chemiluminescent immunoassay in our study) and types of OC used by participants might also have played some role.
Our findings among DMPA users were even more reassuring. We observed frank B12 deficiency (<180 pg/mL) in <1% of DMPA users during the follow-up interval, which was comparable to our control group of NH users. Furthermore, none showed any clinical signs or symptoms, so the clinical significance of this laboratory abnormality is uncertain. Overall, these data do not justify routine measurement of B12 in OC and DMPA users.
The mechanism by which hormonal contraception causes a decrease in serum B12 has yet to be elucidated. One possible mechanism is a deficiency in the level of serum B12 binders resulting in a false low B12 level in OC users [8, 11, 15, 30–32]. However, several studies [1, 6] have shown no difference in the level of mean unsaturated B12 binders between OC users and nonusers, suggesting that absorption is not affected and that redistribution of B12 throughout the body could be responsible [1]. However, the mechanism of this redistribution is unknown. Furthermore, the mechanism of suppression of B12 levels among DMPA users, which was less severe than that observed among OC users in our study, has not been determined.
Our finding that the level of B12 was not associated with BMD, irrespective of contraceptive use, is in contrast to the findings of several cross-sectional studies [19–24]. All but one of these prior studies, however, focused on postmenopausal women, who may react differently. The one study which did include reproductive-age women was limited to European adolescents who adhered to a specific diet plan (one group consumed a macrobiotic diet for up to 6 years followed by a vegetarian/omnivorous diet while the other group ate an omnivorous diet throughout their life). Furthermore, it differed from the current study in that it was cross-sectional in design and included males as well as females. These differences could explain the variation in findings between their study and ours.
Our observations that non-Hispanic white women had the lowest mean B12 levels (437 pg/mL) while non-Hispanic blacks (556 pg/mL) had the highest levels are consistent with a large national study (460 pg/mL, 566 pg/mL and 484 pg/mL, respectively) of 20–39 year old women [33]. Other studies with smaller sample size have also noted similar disparities [34–37]. The reason for this disparity, however, has yet to be elucidated. Elevated serum transcobalamins (B12 binding protein) and higher unsaturated cobalamin binding capacities in non-Hispanic blacks compared to whites have been suggested as possible reasons [36, 38, 39]. Furthermore, it has been speculated that genetic factors, such as differences in transcobalamin II gene frequencies (40), might be responsible for the elevated transcobalamin levels in blacks [37, 38].
This study has several limitations. First, we did not randomly assign women to a contraceptive method because the three types under study have different efficacies and randomization could have led to unintended pregnancies. Second, we studied only one formulation of OC, so our findings cannot be generalized to other types of oral contraceptive pills with different amounts of estrogen or other progestins. Together, these limitations could impact the overall generalizability of our findings, and selection bias cannot be ruled out. Finally, we are unable to shed light on the mechanism by which OC and DMPA altered B12 levels.
Overall, this longitudinal study demonstrated that women receiving OC/DMPA may experience a suppression of their B12 level, but this decrease is rarely clinically significant and does not warrant regular measurement of B12 levels among users. Thus, data from this longitudinal study are overall reassuring about the effects of these two contraceptives on serum B12.
Acknowledgments
Dr. Berenson’s work was supported by the Eunice Kennedy Shriver National Institute of Child Health & Human Development (R01HD039883, PI: Berenson and K24HD043659, a Midcareer Investigator Award In Patient-Oriented Research Award PI: Berenson), and the General Clinical Research Centers program (5M01RR000073-43-8696), National Center for Research Resources, National Institutes of Health (NIH). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NICHD or the NIH.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Wertalik LF, Metz EN, LoBuglio AF, Balcerzak SP. Decreased serum B 12 levels with oral contraceptive use. JAMA. 1972;221:1371–4. [PubMed] [Google Scholar]
- 2.Smith JL, Goldsmith GA, Lawrence JD. Effects of oral contraceptive steroids on vitamin and lipid levels in serum. Am J Clin Nutr. 1975;28:371–6. doi: 10.1093/ajcn/28.4.371. [DOI] [PubMed] [Google Scholar]
- 3.Anderson KE, Bodansky O, Kappas A. Effects of oral contraceptives on vitamin metabolism. Adv Clin Chem. 1976;18:247–87. doi: 10.1016/s0065-2423(08)60300-5. [DOI] [PubMed] [Google Scholar]
- 4.Briggs M, Briggs M. Endocrine effects on serum-vitamin-B 12. Lancet. 1972;2(7785):1037. doi: 10.1016/s0140-6736(72)92457-9. [DOI] [PubMed] [Google Scholar]
- 5.Briggs M, Briggs MH. Changes in biochemical indices of vitamin nutrition in women using oral contraceptives during treatment with “Surbex 500”. Curr Med Res Opin. 1974–1975;2:626–30. doi: 10.1185/03007997409111874. [DOI] [PubMed] [Google Scholar]
- 6.Costanzi JJ, Young BK, Carmel R. Serum vitamin B12 and B12-binding protein levels associated with oral contraceptives. Tex Rep Biol Med. 1978;36:69–77. [PubMed] [Google Scholar]
- 7.Green TJ, Houghton LA, Donovan U, Gibson RS, O'Connor DL. Oral contraceptives did not affect biochemical folate indexes and homocysteine concentrations in adolescent females. J Am Diet Assoc. 1998;98:49–55. doi: 10.1016/S0002-8223(98)00014-5. [DOI] [PubMed] [Google Scholar]
- 8.Hjelt K, Brynskov J, Hippe E, Lundström P, Munck O. Oral contraceptives and the cobalamin (vitamin B12) metabolism. Acta Obstet Gynecol Scand. 1985;64:59–63. doi: 10.3109/00016348509154689. [DOI] [PubMed] [Google Scholar]
- 9.Shojania AM. Effect of oral contraceptives on vitamin-B12 metabolism. Lancet. 1971;2(7730):932. doi: 10.1016/s0140-6736(71)92552-9. [DOI] [PubMed] [Google Scholar]
- 10.Shojania AM, Hornady GJ, Scaletta D. The effect of oral contraceptives on folate metabolism. III. Plasma clearance and urinary folate excretion. J Lab Clin Med. 1975;85:185–90. [PubMed] [Google Scholar]
- 11.Shojania AM, Wylie B. The effect of oral contraceptives on vitamin B12 metabolism. Am J Obstet Gynecol. 1979;135:129–34. [PubMed] [Google Scholar]
- 12.Brattstrom L, Israelsson B, Olsson A, Andersson A, Hultberg B. Plasma homocysteine in women on oral oestrogen-containing contraceptives and in men with oestrogen-treated prostatic carcinoma. Scand J Clin Lab Invest. 1992;52:283–7. doi: 10.1080/00365519209088360. [DOI] [PubMed] [Google Scholar]
- 13.Mountifield JA. Serum vitamin b(12) and folate levels in women taking oral contraceptives. Can Fam Physician. 1986;32:862–5. [PMC free article] [PubMed] [Google Scholar]
- 14.Sütterlin MW, Bussen SS, Rieger L, Dietl J, Steck T. Serum folate and Vitamin B12 levels in women using modern oral contraceptives (OC) containing 20 microg ethinyl estradiol. Eur J Obstet Gynecol Reprod Biol. 2003;107:57–61. doi: 10.1016/s0301-2115(02)00371-8. [DOI] [PubMed] [Google Scholar]
- 15.Lussana F, Zighetti ML, Bucciarelli P, Cugno M, Cattaneo M. Blood levels of homocysteine, folate, vitamin B6 and B12 in women using oral contraceptives compared to non-users. Thromb Res. 2003;112:37–41. doi: 10.1016/j.thromres.2003.11.007. [DOI] [PubMed] [Google Scholar]
- 16.Prasad AS, Lei KY, Moghissi KS, Stryker JC, Oberleas D. Effect of oral contraceptives on nutrients. III. Vitamins B6, B12, and folic acid. Am J Obstet Gynecol. 1976;125:1063–9. doi: 10.1016/0002-9378(76)90809-7. [DOI] [PubMed] [Google Scholar]
- 17.Grace E, Emans SJ, Drum DE. Hematologic abnormalities in adolescents who take oral contraceptive pills. J Pediatr. 1982;101:771–4. doi: 10.1016/s0022-3476(82)80317-x. [DOI] [PubMed] [Google Scholar]
- 18.Steegers-Theunissen RP, Boers GH, Steegers EA, Trijbels FJ, Thomas CM, Eskes TK. Effects of sub-50 oral contraceptives on homocysteine metabolism: a preliminary study. Contraception. 1992;45:129–39. doi: 10.1016/0010-7824(92)90047-w. [DOI] [PubMed] [Google Scholar]
- 19.Stone KL, Bauer DC, Sellmeyer D, Cummings SR. Low serum vitamin B-12 levels are associated with increased hip bone loss in older women: a prospective study. J Clin Endocrinol Metab. 2004;89:1217–21. doi: 10.1210/jc.2003-030074. [DOI] [PubMed] [Google Scholar]
- 20.Tucker KL, Hannan MT, Qiao N, et al. Low plasma vitamin B12 associated with lower bone mineral density: the Framingham osteoporosis study. J Bone Miner Res. 2005;20:152–8. doi: 10.1359/JBMR.041018. [DOI] [PubMed] [Google Scholar]
- 21.Dhonukshe-Rutten RA, Lips M, de Jong N, et al. Vitamin B-12 status is associated with bone mineral content and bone mineral density in frail elderly women but not in men. J Nutr. 2003;133:801–7. doi: 10.1093/jn/133.3.801. [DOI] [PubMed] [Google Scholar]
- 22.Morris MS, Jacques PF, Selhub J. Relation between homocysteine and B-vitamin status indicators and bone mineral density in older Americans. Bone. 2005;37:234–42. doi: 10.1016/j.bone.2005.04.017. [DOI] [PubMed] [Google Scholar]
- 23.Bozkurt N, Erdem M, Yilmaz E, et al. The relationship of homocysteine, B12 and folic acid with the bone mineral density of the femur and lumbar spine in Turkish postmenopausal women. Arch Gynecol Obstet. 2009;280:381–7. doi: 10.1007/s00404-009-0936-0. [DOI] [PubMed] [Google Scholar]
- 24.Dhonukshe-Rutten RA, van Dusseldorp M, Schneede J, de Groot LC, van Staveren WA. Low bone mineral density and bone mineral content are associated with low cobalamin status in adolescents. Eur J Nutr. 2005;44:341–7. doi: 10.1007/s00394-004-0531-x. [DOI] [PubMed] [Google Scholar]
- 25.Golbahar J, Hamidi A, Aminzadeh MA, Omrani GR. Association of plasma folate, plasma total homocysteine, but not methylenetetrahydrofolate reductase C667T polymorphism, with bone mineral density in postmenopausal Iranian women: a cross- sectional study. Bone. 2004;35:760–5. doi: 10.1016/j.bone.2004.04.018. [DOI] [PubMed] [Google Scholar]
- 26.Cagnacci A, Baldassari F, Rivolta G, Arangino S, Volpe A. Relation of homocysteine, folate, and vitamin B12 to bone mineral density of postmenopausal women. Bone. 2003;33:956–9. doi: 10.1016/j.bone.2003.07.001. [DOI] [PubMed] [Google Scholar]
- 27.Berenson AB, Rahman M, Breitkopf CR, Bi LX. Effects of depot medroxyprogesterone acetate and 20-microgram oral contraceptives on bone mineral density. Obstet Gynecol. 2008;112:788–99. doi: 10.1097/AOG.0b013e3181875b78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Berenson AB, Rahman M, Wilkinson G. Effect of injectable and oral contraceptives on serum lipids. Obstet Gynecol. 2009;114:786–94. doi: 10.1097/AOG.0b013e3181b76bea. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Berenson AB, van den Berg P, Williams KJ, Rahman M. Effects of injectable and oral contraceptives on glucose and insulin levels. Obstet Gynecol. 2011;117:41–7. doi: 10.1097/AOG.0b013e318202ac23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shojania AM. Oral contraceptives: effect of folate and vitamin B12 metabolism. Can Med Assoc J. 1982;126:244–7. [PMC free article] [PubMed] [Google Scholar]
- 31.Snow CF. Laboratory diagnosis of vitamin B12 and folate deficiency. Arch Intern Med. 1999;159:1289 –98. doi: 10.1001/archinte.159.12.1289. [DOI] [PubMed] [Google Scholar]
- 32.Gardyn J, Mittelman M, Zlotnik J, Sela BA, Cohen AM. Oral contraceptives can cause falsely low vitamin B12 levels. Acta Haematol. 2000;104:22– 4. doi: 10.1159/000041064. [DOI] [PubMed] [Google Scholar]
- 33.Wright JD, Bialostosky K, Gunter EW, et al. Blood folate and vitamin B12: United States, 1988–94. Vital Health Stat. 1998;11(243):1–78. [PubMed] [Google Scholar]
- 34.Stabler SP, Allen RH, Fried LP, et al. Racial differences in prevalence of cobalamin and folate deficiencies in disabled elderly women. Am J Clin Nutr. 1999;70:911–9. doi: 10.1093/ajcn/70.5.911. [DOI] [PubMed] [Google Scholar]
- 35.Kwee HG, Bowman HS, Wells LW. A Racial Difference in Serum Vitamin B12 Levels. J Nucl Med. 1985;26:790–2. [PubMed] [Google Scholar]
- 36.Carmel R. Ethnic and racial factors in cobalamin metabolism and its disorders. Semin Hematol. 1999;36(1):88–100. [PubMed] [Google Scholar]
- 37.Saxena S, Carmel R. Racial differences in vitamin B12 levels in the United States. Am J Clin Pathol. 1987;88:95–7. doi: 10.1093/ajcp/88.1.95. [DOI] [PubMed] [Google Scholar]
- 38.Fernandes-Costa F, Metz J. A comparison of serum transcobalamin levels in white and black subjects. Am J Clin Nutr. 1982;35:83–6. doi: 10.1093/ajcn/35.1.83. [DOI] [PubMed] [Google Scholar]
- 39.Fleming AF, Ogunfunmilade YA, Cannel R. Serum vitamin B12 levels and vitamin B12 binding proteins of serum and saliva of healthy Nigerians and Europeans. Am J Clin Nutr. 1978;3l:1732–8. doi: 10.1093/ajcn/31.10.1732. [DOI] [PubMed] [Google Scholar]
- 40.Porck HJ, Fleming AF, Frants RR. Distribution of genetic variants of transcobalamin II in Nigerian black populations. Am J Hum Genet. 1984;36:710–7. [PMC free article] [PubMed] [Google Scholar]