Abstract
Introduction:
Vitamin D may affect multiple health outcomes. If so, an effect on mortality is to be expected. Using pooled data from randomized controlled trials, we performed individual patient data (IPD) and trial level meta-analyses to assess mortality among participants randomized to either vitamin D alone or vitamin D with calcium.
Subjects and Methods:
Through a systematic literature search, we identified 24 randomized controlled trials reporting data on mortality in which vitamin D was given either alone or with calcium. From a total of 13 trials with more than 1000 participants each, eight trials were included in our IPD analysis. Using a stratified Cox regression model, we calculated risk of death during 3 yr of treatment in an intention-to-treat analysis. Also, we performed a trial level meta-analysis including data from all studies.
Results:
The IPD analysis yielded data on 70,528 randomized participants (86.8% females) with a median age of 70 (interquartile range, 62–77) yr. Vitamin D with or without calcium reduced mortality by 7% [hazard ratio, 0.93; 95% confidence interval (CI), 0.88–0.99]. However, vitamin D alone did not affect mortality, but risk of death was reduced if vitamin D was given with calcium (hazard ratio, 0.91; 95% CI, 0.84–0.98). The number needed to treat with vitamin D plus calcium for 3 yr to prevent one death was 151. Trial level meta-analysis (24 trials with 88,097 participants) showed similar results, i.e. mortality was reduced with vitamin D plus calcium (odds ratio, 0.94; 95% CI, 0.88–0.99), but not with vitamin D alone (odds ratio, 0.98; 95% CI, 0.91–1.06).
Conclusion:
Vitamin D with calcium reduces mortality in the elderly, whereas available data do not support an effect of vitamin D alone.
During the last decade, there has been increasing recognition of the potential role of vitamin D in a large number of tissues, where vitamin D receptors have been identified. Thus, maintenance of adequate vitamin D status may be necessary for many physiological functions, beyond that of the classical actions of vitamin D on bone and mineral metabolism. Cross-sectional and longitudinal studies have linked reduced vitamin D status with a number of cancers, adverse cardiovascular and immunological outcomes, and increased all-cause mortality, although some observational data have suggested either U-shaped or reverse J-shaped relationships (1–3).
A meta-analysis of randomized controlled trials of vitamin D supplementation published in 2007 found that vitamin D reduced all-cause mortality by 7% (relative risk, 0.93; 95% CI, 0.87–0.99) (4). However, three subsequent meta-analyses using updated searches and different study inclusion criteria failed to show a statistically significant effect of vitamin D alone on mortality (2, 5, 6), although two of the studies did show a reduced mortality if vitamin D was given together with calcium (5, 6). Study level meta-analyses using summary data may be adequate when estimating a single treatment effect or investigating study level characteristics but may lead to biased assessments and have limitations in explaining heterogeneity. To investigate whether treatment effects are related to patient characteristics, individual patient data (IPD) analyses offer advantages and have greater statistical power than study level meta-analyses (7).
We recently published results of an IPD level meta-analysis showing that vitamin D in doses of 10–20 μg/d given alone were not effective in fracture prevention, but when given with calcium reduced fracture incidence (8). In the present study, we aimed to investigate effects of vitamin D supplementation, given either alone or with calcium, on all-cause mortality using both IPD level and study level meta-analysis methodologies.
Materials and Methods
Searching and selection criteria
As previously detailed, we undertook a systematic literature search for publications between 1966 and 2008 in Medline, Embase, and the Cochrane Central Register of Controlled Trials on the effects of vitamin D treatment on the risk of fracture (8). No language restrictions were applied. We included only randomized (individual or cluster) controlled studies in which cholecalciferol (vitamin D3) or ergocalciferol (vitamin D2) was given in at least one intervention arm and with one arm without vitamin D. For the IPD analysis, we excluded studies with less than 1000 participants (nine trials) due to concerns that studies with fewer participants, although contributing additional cases, would also further reduce the mass of shared study variables for aggregated analysis. The search revealed 11 eligible studies (9–19). For the present analysis, we updated our search to January 2011 and included the MeSH terms: [death, mortality], thereby identifying two further eligible studies (20, 21). Five study groups were unwilling or unable to provide patient level data (9, 10, 12, 16, 17), leaving eight studies for IPD analysis (Table 1). All studies had incident fractures as the primary endpoint, whereas data on death were collected as secondary endpoints during follow-up (14, 15, 18–20) and/or by the use of national registers of vital statistics (11, 13, 15, 19, 21).
Table 1.
Characteristics of the eight randomized controlled trials included in the IPD analysis and 16 additional studies included in the trial level meta-analysis
| First author, year (Ref.) | Patient category; total no. of included participants (% females); median age (IQR) or mean ± sd; place of study | Randomization and duration | Study arms; no. of participants in each arm/total no. of deaths during the entire period of follow-upa (no. of deaths during the first 36 months of follow-upb) |
|---|---|---|---|
| Studies included in IPD analysis | |||
| Meyer, 2002 (11) | Nursing home residents; n = 1,144 (75.9%); age, 85 (81–90) yr; Norway | Q, 24 months, double-blind | Daily oral 10 μg D3, 569/246 (246), vs. placebo, 575/245 (245) |
| Larsen, 2004 (13) | Community-dwelling aged ≥66 yr; n = 9,605 (60.1%); age, 74 (70–79) yr; Denmark | C, 42 months, open-label | Daily oral 10 μg D3 + 1000 mg Ca, 4,957/832 (715), vs. no treatment, 4,648/839 (705) |
| Porthouse, 2005 (14) | GP patients with risk factors for fracture; n = 3,314 (100%); age, 76 (73–80) yr; UK | I, 18–42 (median, 22.5) months, open-label | Daily oral 20 μg D3 + 1000 mg Ca. 1,321/57 (57), vs. leaflet, 1,993/68 (67) |
| Grant (RECORD), 2005 (15) | Previous osteoporotic fracture; n = 5,292 (84.7%); age, 77 (73–81) yr; UK | I, 24–62 (median, 30.4) months, double-blind | Four groups with daily oral dose: 1) 20 μg D3 + 1000 mg Ca, 1,306/221 (161); 2) 20 μg D3, 1,343/217 (167); 3) 1000 mg Ca, 1,311/243 (198); 4) double placebo, 1,332/217 (172) |
| Smith, 2007 (18) | GP patients presenting for influenza vaccination; n = 9,440 (53.9%); age, 79 (77–83) yr; UK | I, up to 36 (median, 36.0) months, double-blind | Once a year im 300,000 IU D2, 4,727/316 (290), vs. placebo, 4,713/312 (291) (equal to an average D2 dose of 20.5 μg/d) |
| LaCroix (WHI), 2009 (19)c | Community-based postmenopausal women; n = 36,282 (100%); age, 62 (57–68) yr; USA | I, median, 85.4 months, double-blind | Daily oral 10 μg D3 + 1000 mg Ca, 18,176/755 (180), vs. placebo, 18,106/825 (220) |
| Sanders, 2010 (20)e | Community-dwelling aged ≥70 yr; n = 2,256 (100%); age, 76 (73–80) yr; Australia | I, up to 60 (median, 36) months, double-blind | Once a year oral 500,000 IU D3, 1,131/46 (39), vs. placebo, 1,125/54 (46) (equal to an average D3 dose of 34 μg/d) |
| Salovaara (OSTPRE-FPS), 2010 (21)d | Community-dwelling women aged 65–71 yr; n = 3,195 (100%); age, 67 (66–69) yr; Finland | I, up to 36 months; mean follow-up time, 3.01 (sd, 0.22) yr, open-label | Daily oral 20 μg D3 + 1000 mg Ca, 1,586/15 (15), vs. no treatment, 1,609/18 (18) |
| Additional medium- and large-sized (>500 participants) studies included in trial level meta-analysis | |||
| Chapuy, 1994 (9) | Nursing home residents; n = 3,270 (100%); age, 84 ± 6 yr; France | I, up to 36 months, double-blind | Daily oral 20 μg D3 + 1200 mg Ca, 1,634/601, vs. placebo, 1,636/597 |
| Lips, 1996 (10) | Living independently, in apartments/homes for elderly persons aged ≥70 yr; n = 2,578 (74%); age, 80 ± 6 yr; The Netherlands | I, up to 42 months, double-blind | Daily oral 10 μg D3, 1,291/223, vs. placebo, 1,287/251 |
| Chapuy, 2002 (27) | Ambulatory institutionalized women; n = 583 (100%); age, 85 ± 7 yr; France | I, up to 24 months, double-blind | Daily oral 20 μg D3 + 1200 mg Ca, 393/71, vs. placebo, 190/45 |
| Trivedi, 2003 (12) | Community-dwelling aged ≥65 yr; n = 2,686 (24%); age, 75 ± 5 yr; UK | I, up to 60 months, double-blind | Every fourth month, oral 100,000 IU D3, 1,345/224, vs. placebo, 1,341/247 (equal to an average D3 dose of 21 μg/d) |
| Flicker, 2005 (28) | Nursing home residents; n = 625 (95%); age, 83 ± 8 yr; Australia | I, up to 24 months, double-blind | Daily oral 600 mg Ca + D2, initially 10,000 IU weekly followed by 25 μg/d, 313/76, vs. placebo, 312/85 |
| Law, 2006 (16)f | Nursing home residents; n = 2,641 (76%); mean age, 85 yr; UK | C, median, 10 (IQR, 7–14) months, open-label | Every third month, oral 100,000 IU D2, 1,252/247, vs. placebo, 1,389/229 (equal to an average D2 dose of 28 μg/d) |
| Lyons, 2007 (17) | Nursing home residents; n = 3,440 (76%); age, 84 ± 8 yr; UK | I, up to 36 months, double-blind | Every fourth month, oral 100,000 IU D2, 1,673/713, vs. placebo, 1,670/715 (equal to an average D2 dose of 21 μg/d) |
| Small-sized studies (<500 participants) included in trial level meta-analysis | |||
| Baeksgaard, 1998 (29) | Healthy women aged 58–67 yr; n = 240 (100%); mean age, 62 yr; Denmark | I, 24 months, double-blind | Daily oral 14 μg D3 + 1000 mg Ca, 160/1, vs. placebo, 80/1 |
| Krieg, 1999 (43) | Institutionalized women aged 62–98 yr; n = 248 (100%); age, NA; Switzerland | I, 24 months, open-label | Daily oral 22 μg D3 + 1000 mg Ca, 124/21, vs. no treatment, 124/26 |
| Komulainen, 1999 (44) | Early postmenopausal women; n = 458 (100%); mean age, 53 yr (Finland) | I, 60 months, open-label | Four treatment groups with daily oral: 1) HRT, 115/1; 2) 7.5 μg D3 (only 2.5 μg/d during the fifth year), 112/0; 3) HRT + D3 as above, 116/1; 4) placebo, 115/1 |
| Latham, 2003 (30) | Frail elderly aged >65 yr; n = 243 (53%); age, 79 ± 7 yr; New Zealand | I, 6-month follow-up, double-blind | Single oral dose 300,000 IU D3, 121/11, vs. placebo, 122/3 |
| Avenell, 2004 (45) | Aged >70 yr and a history of an osteoporotic fracture within the last 10 yr; n = 134 (82%); age, 77 ± 5 yr; UK | I, 12 months, open-label | Daily oral 20 μg D3 + 1000 mg Ca, 99/1, vs. no treatment, 35/1 |
| Harwood, 2004 (46) | Elderly with a recent hip fracture; n = 150 (100%); age, 81 (67–92) yr; UK | I, 12 months, open-label | Four study groups: 1) single injection of D2 300,000 IU, 38/7; 2) single injection D2 300,000 IU + 1000 mg/d Ca, 36/11; 3) oral D3 20 μg/d + 1000 mg/d Ca, 39/6; 4) no treatment, 37/5 |
| Meier, 2004 (47) | Healthy community dwelling aged 33–78 yr; n = 55 (65%); age, NA; Germany | I, 12 months, open-label | Daily oral 12.5 μg D3 + 500 mg Ca, 30/0, vs. no treatment, 25/1 |
| Brazier, 2005 (31) | Women >65 yr with plasma 25OHD <30 nmol/liter; n = 192 (100%); age, 75 ± 7 yr; France | I, 12 months, double-blind | Daily oral 20 μg D3 + 1000 mg Ca, 95/3, vs. placebo, 97/1 |
| Schleithoff, 2006 (32) | Patients with congestive heart failure; n = 123 (17%); median age, 55 yr; Germany | I, 9 months, double-blind | Daily oral 50 μg D3 + 500 mg Ca, 61/7, vs. 500 mg Ca, 62/6 |
I, Individually randomized; Q, quasi-randomized by birth date; C, cluster randomized; IQR, interquartile range; 25OHD, 25-hydroyvitamin D; GP, general practice; Ca, calcium supplement; NA, not available. To convert micrograms of vitamin D into international units, multiply by 40.
Total number of deaths during the entire follow-up period, i.e. not restricted to the 36 months of follow-up used in the IPD analysis.
Data only available for studies included in the IPD analysis.
Number of deaths is based on the WHI Investigator Data Set updated on September 12, 2005 (released on December 21, 2006), in which slightly more (n = 29) deaths have been recorded than in the original trial report.
Number of deaths is based on an updated data extraction (October, 2010) from the Finnish National Population Register in which slightly more (n = 5) deaths have been recorded than in the original trial report.
Number of deaths in the original trial report was based on subjects who died while actively participating, whereas the number of deaths stated in the table is based on treatment allocation independently of whether subjects participated actively or not in the study.
Adjustments to the number of participants (1762 vs. 1995) with outcomes, and denominators in study were made using an intraclass correlation coefficient of 0.026—see Sensitivity section for further explanations.
Analytical approach
We defined base models using unconditional logistic regression incorporating age and sex, which we expected a priori to contribute to variation in mortality. We also included data on incident major osteoporotic fracture during the study, i.e. fractures at the hip or spine because mortality is known to be increased after such fractures, and information on the use of postmenopausal hormone replacement therapy (HRT) and bisphosphonates. Analysis on mortality was performed using a stratified Cox regression model, with the clinical study as stratum, thereby ensuring that like was compared with like, because the inference is based on the randomization of patients within each trial. We added treatment allocation and interaction terms to this model to study effects of vitamin D vs. no vitamin D supplements on risk of death. Interaction terms studied included age, sex, and daily dose of vitamin D. In studies with intermittent (monthly or yearly) administration of vitamin D, an equivalent daily dose was calculated. In all analyses, observations were truncated after 36 months (1095 d) because few of the studies provided substantial data beyond this time point. However, to evaluate the effect of interventions during the entire duration of trials, we also performed logistic regression analyses with treatment allocation added to the base model. All analyses were performed using an intention-to-treat approach including all randomized participants.
Prespecified subanalyses
Three prespecified analyses were performed:
Because ergocalciferol may have a shorter half-life than cholecalciferol and lower systemic availability if injected im (22), we studied the effects of cholecalciferol separately and addressed the impact of vitamin D dose on mortality by regrouping the Smith et al. (18) study (average, 20.5 μg ergocalciferol/d) with 10-μg cholecalciferol studies.
Whether vitamin D was administered alone or with calcium supplements (CaD) and whether daily vs. intermittent administration affected results.
In the RECORD study, one of the treatment arms had calcium as stand-alone treatment, i.e. without concomitant vitamin D (15). A priori, participants randomized to the calcium-alone group were considered as belonging to the placebo/non-vitamin D group. However, because recent studies have suggested detrimental effects of calcium-alone treatment (23), analyses were also performed in which participants randomized to the calcium-alone group were removed from the analyses.
Sensitivity
To assess whether our conclusions would have been modified by failure to include one or more individual studies, we performed an influence analysis in which studies were removed one by one. To evaluate whether the lack of data from trials not included in the IPD analysis affected results, we performed a trial level meta-analysis including all available studies (n = 24) as detailed in Table 1. Number of deaths by treatment allocation up to 3 yr in the study by Chapuy et al. (9) was reported by personal communication (Prof. P. Meunier, July 2010). In the cluster randomized trial by Law et al. (16), adjustments to the number of participants with outcomes and denominators were made using an intraclass correlation coefficient of 0.026 derived from Dyer et al. (24) using methods described by Higgins and Green (25). All meta-analyses were performed stratified by whether vitamin D was administered alone or in combination with calcium and without the assumption of a common among-study variance component across subgroups. The manuscript was prepared in accordance with the PRISMA guidelines (26). Analyses were undertaken using SPSS version 15 (SPSS Inc., Chicago, IL) and Comprehensive Meta-analysis version 2.2 (Biostat, Englewood, NJ). All tests were two-tailed. We considered P < 0.05 as significant.
Results
IPD analyses
Our IPD analysis included 70,528 randomized participants (86.8% females) aged 48–103 (median, 70; interquartile range, 62–77) yr. A total of 27,345 were randomized to CaD and 7,771 to vitamin D alone, whereas 35,412 received placebo/no vitamin D.
Defining the base model
During the 36 months of follow-up, 3832 (5.4%) study participants died, and 1139 (1.6%) sustained an osteoporotic fracture at the hip or spine. At baseline, 970 (1.4%) reported the use of bisphosphonates. Among females, 20,402 (33.9%) were treated with HRT. This was mainly due to the high proportion of HRT users in the Women's Health Initiative (WHI) (50.7%) and the OSTPRE-FPS (48.5%) studies, whereas the frequency of HRT use in the other studies was low (2.3%). After mutual adjustments, the risk of death was significantly associated with increasing age [hazard ratio (HR) per decade, 1.87; 95% CI, 1.78–1.96], male sex (HR, 1.52; 95% CI, 1.41–1.64), and an incident major osteoporotic fracture during follow-up (HR, 1.47; 95% CI, 1.27–1.69), whereas mortality was not significantly affected by use of HRT (HR, 0.88; 95% CI, 0.75–1.04) or bisphosphonates (HR, 0.60; 95% CI, 0.32–1.10).
Differences in mortality rates were not fully explained by differences in age, sex, number of incident major osteoporotic fractures, or treatment with bisphosphonates or HRT. Accordingly, analyses were performed adjusted for age, sex, incident major osteoporotic fractures, and use of bisphosphonates or HRT and stratified by study.
Effects of vitamin D supplementation with or without calcium on risk of death
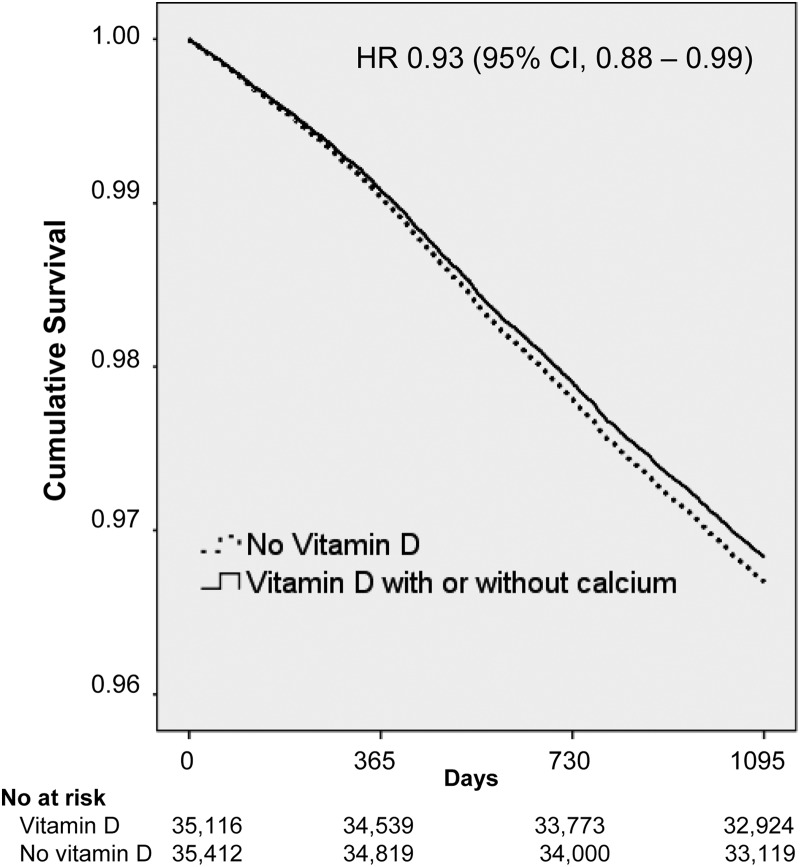
Within the group of participants randomized to vitamin D with or without calcium (n = 35,116), 1,870 (5.3%) died during follow-up, whereas death occurred in 1,962 (5.5%) of the participants not allocated to vitamin D (HRcrude, 0.94; 95% CI, 0.88–1.00). After adjustments, risk of death was reduced by 7% (HR, 0.93; 95% CI, 0.88–0.99) during the 3 yr of follow-up in participants randomized to vitamin D with or without calcium (Fig. 1). Visual inspection of the survival curve (Fig. 1) indicates that risk started to diverge quite quickly. However, although risk estimates were reduced in the group of vitamin D-treated participants, risk of death did not differ significantly between groups after 12 months (HR, 0.96; 95% CI, 0.86–1.08) or 24 months (HR, 0.94; 95% CI, 0.87–1.02) of treatment.
Fig. 1.
IPD analysis on survival in participants randomized to vitamin D with or without calcium supplements compared with placebo/no supplements. Data represent adjusted HR (95% CI).
Statistically significant interaction terms were whether vitamin D was coadministrated with calcium (P < 0.01), whereas no significant interactions were found for sex (P = 0.83), age (P = 0.08), route of vitamin D administration (P = 0.65), dosing interval (P = 0.98), or treatment at baseline with HRT (P = 0.46) or bisphosphonates (P = 0.16).
Mortality rates differed significantly between studies investigating the effects of vitamin D without calcium and studies on CaD (Table 1). On average, in the groups of untreated/placebo-treated participants, 4.4% died in the CaD trials, whereas 9.7% died in the studies on vitamin D alone (P < 0.01).
Effects of concomitant calcium administration
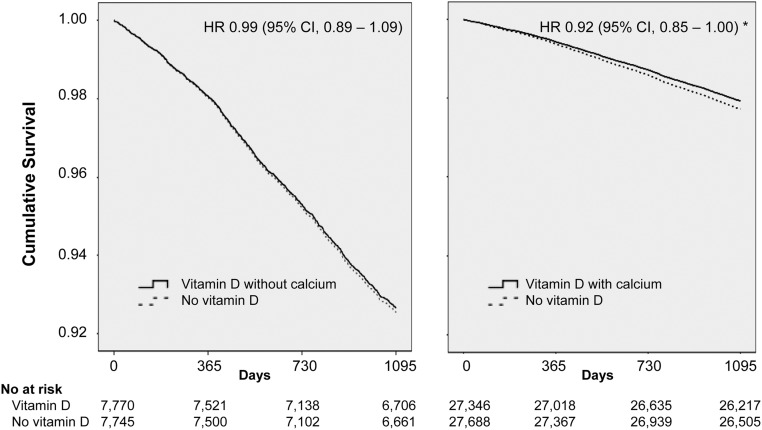
Restricting the analysis to studies with CaD supplementation (13–15, 19, 21), risk of death was significantly reduced among participants randomized to CaD (HR, 0.91; 95% CI, 0.84–0.98). Exclusion of participants from the placebo/non-vitamin D group who were treated with calcium alone (n = 1311) did not change the results (HR, 0.92; 95% CI, 0.85–1.00; P < 0.05) (Fig. 2).
Fig. 2.
IPD analysis on survival in participants randomized to vitamin D alone (left) or vitamin D with calcium (right) vs. placebo/no supplements. For the RECORD study, participants treated with calcium without concomitant vitamin D (n = 1311) were excluded, whereas the placebo group (n = 1332) was included in both analyses. Data represent adjusted HR (95% CI). *, P < 0.05.
On the other hand, restricting the analysis to the four studies on vitamin D alone (11, 15, 18, 20), risk of death did not differ according to treatment allocation (HR, 0.96; 95% CI, 0.87–1.06). Removal of the participants treated with calcium alone did not change the results (Fig. 2). Neither did vitamin D affect mortality if analyses were restricted to include only studies using cholecalciferol (HR, 0.94; 95% CI, 0.83–1.06).
Dose-effect relationships
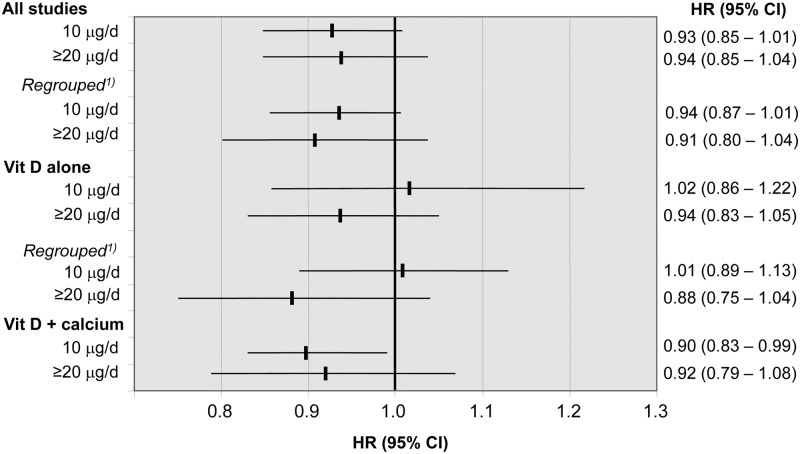
As shown in Fig. 3, no statistically significant dose-effect relationships were evident. Most estimates showed a reduced risk, but mortality was only significantly reduced in participants treated with a daily dose of 10 μg vitamin D in combination with calcium (HR, 0.90; 95% CI, 0.83–0.99).
Fig. 3.
IPD analysis on dose-effect relationships of vitamin D treatment on risk of death. Note that none of the included studies used a daily dose between 10 and 20 μg. All studies and stratified by whether vitamin D was provided alone (Vit D alone) or in combination with calcium (Vit D + calcium). Data represent adjusted HR (95% CI). The Smith study using an average daily dose of 20 μg vitamin D2 was regrouped into the low-dose (10 μg/d) group.
Daily vs. intermittent administration
Two studies provided data on intermittent (once-a-year) administration of either ergocalciferol (18) or cholecalciferol (20), which did not affect mortality (HR, 0.97; 95% CI, 0.83–1.12), whereas mortality was significantly decreased in participants randomized to receive daily supplements (HR, 0.93; 95% CI, 0.86–0.99). However, daily treatment with vitamin D without calcium did not affect mortality (HR, 0.95; 95% CI, 0.84–1.08), whereas mortality was reduced in participants randomized to daily CaD (HR, 0.91; 95% CI, 0.84–0.98).
Sensitivity analyses
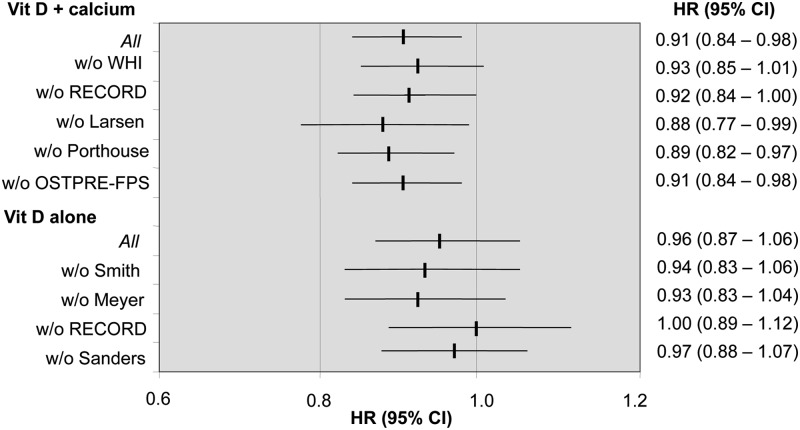
Influence analyses showed only minor effects of exclusion of studies one by one (Fig. 4). Removal of the cluster randomized study by Larsen et al. (13) from the analysis on CaD trials did not change the results (HR, 0.88; 95% CI, 0.77–0.99).
Fig. 4.
Sensitivity analysis: influence of removing individual studies from the IPD analysis stratified by whether vitamin D (Vit D) was provided alone or in combination with calcium (Vit D + calcium). Data represent adjusted HR (95% CI). w/o, Without.
In analyses not accounting for whether calcium was coadministrated, mortality was borderline significantly reduced if analyses were restricted to only double-blind studies (Pinteraction = 0.05; HR, 0.92; 95% CI, 0.85–1.00) (11, 15, 18–20) and significantly reduced if analyses were further restricted to include only individually randomized double-blind studies (Pinteraction = 0.02; HR, 0.89; 95% CI, 0.81–0.98) (15, 18–20). Within the group of individually randomized double-blind studies, mortality was significantly reduced in response to CaD (HR, 0.83; 95% CI, 0.73–0.95) (15, 19), but not in response to vitamin D alone (HR, 0.96; 95% CI, 0.85–1.09) (15, 18, 20). Restricting analyses to include data from only open-label trials showed no significant effects on mortality (HR, 0.95; 95% CI, 0.86–1.05) (13, 14, 21).
Analyses not accounting for study duration
Although most included studies only provided data for a 3-yr intervention period, some studies had a longer duration (Table 1). Including all deaths occurring during studies (n = 5526), logistic regression analysis (adjusted for indices included in the base model) showed no statistical significant effects on mortality in participants randomized to vitamin D with or without calcium (OR, 0.96; 95% CI, 0.90–1.01). However, stratification by coadministration of calcium (Pinteraction <0.01) showed a reduced mortality in the CaD trials (OR, 0.93; 95% CI, 0.87–0.99), but not if vitamin D was given alone (OR, 0.98; 95% CI, 0.89–1.09).
Absolute risk reduction and numbers needed to treat
Because vitamin D alone was not shown to reduce mortality significantly, we did this analysis only for the CaD studies. Over 3 yr of follow-up, the absolute risk reduction was 0.66% (untreated event rate, 16 per 1000 person years), i.e. 151 persons should be treated for 3 yr to prevent one death.
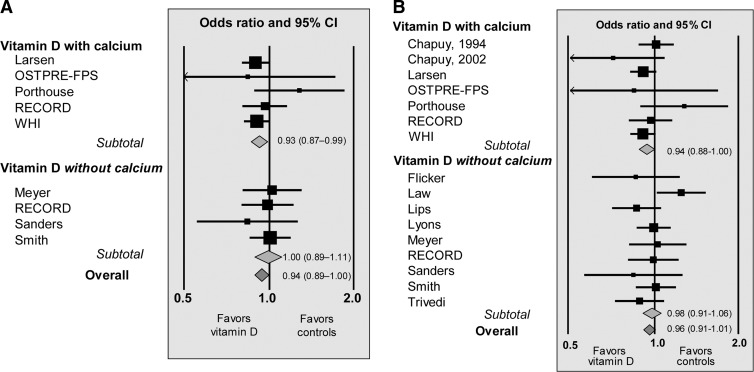
Trial level meta-analysis
As shown in Fig. 5A, trial level meta-analysis of included studies from the IPD analysis showed results similar to the IPD analysis with a reduced risk of death in participants randomized to CaD (HR, 0.93; 95% CI, 0.87–0.99). Extending the analysis to include additional data from large- and medium-sized studies not included in the IPD analysis (Table 1) showed similar results (Fig. 5B). Finally, results were not changed by extending the analysis to include data from all studies shown in Table 1. This analysis included data on a total of 88,097 randomized participants, among whom 28,212 were included in 12 trials with vitamin D alone and 59,885 were included in 13 trials on CaD. Overall, mortality was significantly reduced (P = 0.04) in participants randomized to treatment with vitamin D with or without calcium (OR, 0.95; 95% CI, 0.91–1.00). Stratification by coadministration of calcium showed reduced mortality in participants randomized to CaD (OR, 0.94; 95% CI, 0.88–0.99), but not in participants treated with vitamin D alone. Influence analyses with removal of studies one by one did not change risk estimates to any major degree, although mortality was no longer significantly reduced in response to CaD if the Larsen et al. (13) trial (HR, 0.94; 95% CI, 0.86–1.03) or the WHI (19) study (HR, 0.95; 95% CI, 0.87–1.03) was removed from analyses (Supplemental Fig. 1, published on The Endocrine Society's Journals Online web site at http://jcem.endojournals.org). Removal of the cluster randomized trial by Law et al. (16) showed results similar to the overall analysis with no effect of vitamin D alone (Supplemental Fig. 1). Restricting the trial level analysis to include only individually randomized double-blind studies (9, 10, 12, 15, 17–20, 27–32), mortality was reduced overall (OR, 0.94; 95% CI, 0.89–0.99). Stratification by coadministration of calcium showed a significantly (P < 0.05) reduced mortality in response to CaD (OR, 0.93; 95% CI, 0.86–1.00), but not with vitamin D alone (OR, 0.95; 95% CI, 0.89–1.03).
Fig. 5.
Trial level meta-analyses (random effect model) of studies included in the IPD analysis (A) and meta-analysis including additional large- and medium-sized studies (B). For studies included in the IPD, all deaths occurring during trials were accounted for as detailed in Table 1. 1)No between-study heterogeneity was evident for studies on vitamin D with calcium (P = 0.46; I2 = 0%), vitamin D without calcium (P = 0.86; I2 = 0%) or the overall effect (P = 0.69; I2 = 0%). Influence analyses showed no statistically significant effect of treatment if the WHI study (19) (OR, 0.94; 95% CI, 0.86–1.03) or the Larsen et al. (13) study (OR, 0.94; 95% CI, 0.86–1.02) was removed from the analysis. 2)No between-study heterogeneity was evident for studies on vitamin D with calcium (P = 0.37; I2 = 8%), vitamin D without calcium (P = 0.30; I2 = 16%), or the overall effect (P = 0.30; I2 = 13%). Influence analyses showed no effects of removal of studies one by one, except for removal of the Porthouse et al. (14) study, leaving a statistically significant (P = 0.02) risk estimate for the remaining seven studies (OR, 0.92; 95% CI, 0.87–0.99). Effect size changed slightly by removal of the cluster randomized studies by Larsen et al. (13) (OR, 0.95; 95% CI, 0.88–1.04) and by Law et al. (16) (OR, 0.96; 95% CI, 0.89–1.03).
Discussion
In a pooled analysis, including data at the level of the individual from more than 70,000 elderly participants, mortality was significantly reduced during 3 yr of treatment with CaD, whereas vitamin D alone did not affect mortality. These findings were further supported by a trial level meta-analysis.
Contrast with previous reviews of vitamin D with or without calcium, and calcium alone on mortality
In the 2007 trial level meta-analysis of vitamin D with or without calcium and mortality, including 18 trials without size limit with a total of 57,311 participants, Autier and Gandini (4) found a risk ratio of 0.93 (95% CI, 0.87–0.99). The results did not appear to be influenced by whether calcium was provided or not, dose of vitamin D, or length of follow-up. Our IPD analysis included four large trials not included in the Autier and Gandini review (4). Chung et al. (2) updated the Autier and Gandini review (4) but used different inclusion criteria and found a risk ratio of 0.97 (95% CI, 0.92–1.02) for vitamin D alone (13,833 participants, four trials) and a risk ratio of 0.93 (95% CI, 0.86–1.01) for CaD (44,688 participants, 11 trials). Thus, although their results were not statistically significant for any comparison, the trends were similar to those found here. In accordance with our results, in the 2009 Cochrane review of vitamin D for the prevention of fractures only, the risk ratio for mortality was 1.01 (95% CI, 0.95–1.06) for vitamin D trials with statistical evidence of heterogeneity (Pheterogeneity = 0.04; I2 = 68%) and 0.94 (95% CI, 0.89–0.99) for trials with CaD, with little statistical evidence for heterogeneity of effect (Pheterogeneity = 0.70; I2 = 0%) (5).
Contrast between vitamin D plus calcium and vitamin D alone results
Reduced mortality with CaD, but not with vitamin D alone could suggest that the reduced mortality is due to an effect of calcium rather than vitamin D. However, two recent IPD analyses and trial level meta-analyses by Bolland et al. (23, 33) show an increased risk of myocardial infarction with calcium, with or without vitamin D. So how can CaD reduce mortality when vitamin D alone appears not to be an influence and calcium alone may increase cardiovascular morbidity? There are several possible explanations. First, the reported 30% increase in myocardial infarction from calcium supplementation would have only a small effect on overall mortality (23). For example, in the United Kingdom, myocardial infarction is responsible for 6.4% of all deaths in women over 75 yr of age; thus, a 30% increase in mortality would have a very small effect on overall mortality (19). Secondly, competing risks for disease may be another explanation. Calcium supplementation could reduce recurrence of colorectal adenomas and thus colorectal cancer and mortality (34), an effect that may be strengthened if vitamin D is also provided (35). Calcium and vitamin D may reduce the risk of cancer, particularly breast and colorectal cancer (36). Calcium supplementation has some effect in preventing osteoporotic fractures, although probably not hip fractures, the most important direct cause of osteoporosis mortality (37). However, because we adjusted for incident hip and spine fractures, the reduced mortality in our analysis is most likely not attributable to a lower risk of fracture (and deaths after fractures). Thirdly, CaD may have effects when given together greater than their individual effects. Several studies have shown associations between high plasma levels of PTH and the risk of different (including cardiovascular) diseases and mortality (38, 39). Calcium, as well as vitamin D, is known to lower PTH levels. The doses of vitamin D tested in the vitamin D-alone studies were possibly too low to affect PTH levels, whereas PTH levels were lowered in response to CaD. Because PTH levels were not measured systematically in the included trials, we were unable to test this hypothesis. Fourthly, the vitamin D trials may have selected doses and methods of administration that were not adequate; in addition, the participants included in the vitamin D-alone trials were older than those included in the CaD trials. Our data do not exclude that treatment (vitamin D with or without calcium) is only effective in the early stages of a disease. Finally, differences in the ascertainment of events between the Bolland et al. (23, 33) meta-analyses and the robust ascertainment of death might be another explanation.
Hazard rates started to diverge quite quickly in our IPD analysis, as shown in Figs. 1 and 2. Unfortunately, we were unable to evaluate the causes of death in response to the use of supplements. Epidemiological studies have suggested that vitamin D and calcium may affect many different organ systems and health outcomes, including infections, cardiovascular health, autoimmune diseases, and malignancies (3). Our analyses suggest that supplementation with CaD starts to affect general health early, which is sustained during long-term treatment. Our data do not allow for conclusions on whether this is due to an immediate and sustained effect or whether harm/benefit ratios for certain health outcomes change over time.
Limitations of our analysis
Although it contributes a systematic appraisal of important confounders that may contribute to the difference in survival effects found in clinical trials, the present analysis has important limitations.
Overall, despite a large sample size, the IPD analysis cannot correct potential flaws of the original studies, e.g. in design and compliance with medication. Additional limitations are imposed by the limited set of variables that had been addressed in a fairly consistent way across studies. In general, large CaD trials have been conducted with limited funding, and this imposed limitations on the number of variables that could be collected and the degree of adjudication on exposures and outcomes performed. Although vitamin D can be split into two dose groups (about 10 μg/d and about 20 μg/d, respectively), and some studies gave calcium whereas others did not, the pooled study population does not create a true factorial design of four study arms well matched for age, sex, and risk factors. For example, only the RECORD study (15)—a tertiary prevention study for osteoporosis, because all patients had prior fractures—used daily oral vitamin D at the 20 μg/d dose without calcium. It is possible that the lack of effect in the patient level analysis regarding vitamin D given alone is influenced more by the design of the trials than by the active intervention itself. Of the four studies, the Smith et al. (18) and Sanders et al. (20) studies (once yearly high-dose treatment) may have been influenced by too high and long a dosing interval, the RECORD study (15) by the use of calcium in the control arm and poorer compliance, and the Meyer study (11) by the low dose of vitamin D. Our findings showed a significantly reduced mortality if CaD was administrated daily, whereas no effect was evident if vitamin D was administrated intermittently. However, because the test on interaction between daily vs. intermittent administration was insignificant, our data do not exclude that the lack of an effect may be due to an insufficient statistical power, e.g. too few participants in the group receiving vitamin D intermittently. Several of the risk estimates on the effects of vitamin D alone were below 1.00, with an upper limit of the CI only slightly above 1.00, which may indicate potential beneficial effects of vitamin D alone. However, because the results did not reach strict statistical significance, we think that our results should only be interpreted in such a way that they should encourage further exploration of whether such effects are present.
In the IPD analysis, information on adherence, self-administered CaD supplements, and baseline dietary calcium and vitamin D intake was not consistently available, so this could not be accurately addressed. Accordingly, we were not able to perform a per-protocol analysis. However, our intention-to-treat analytical approach probably offers a more realistic measure of what can be achieved by applying such measures in the general population of elderly participants at risk of fracture than more intensive studies. In accordance, we chose to include all randomized studies in our primary analyses independently of the nature of randomization. Although cluster randomized studies may be prone, for example, to selection bias (40), the less rigorous design may more accurately reflect the real-world circumstances because study participants are normally not required to fulfill a number of individual criteria for participation. Nevertheless, analyses including only individually randomized controlled studies showed results similar to the overall analyses, indicating that inclusion of non-individually randomized studies did not change the overall findings. An additional limitation in the IPD analysis was the restricted time frame of 3 yr. Nevertheless, extending our IPD analysis to include all deaths occurring during the entire duration of the eight trials did not change the overall results, which was further supported by the trial level meta-analysis for all deaths during the entire duration of the trials.
Within the CaD trials, a significantly reduced mortality was only shown for the low dose of vitamin D. Our analyses did not indicate that a higher dose (≥20 μg/d) is inferior to a lower daily dose. Rather, most risk estimates on a daily dose of at least 20 μg of vitamin D alone were lower than with a daily dose of 10 μg. In addition to differences in average age between studies on effects of CaD or vitamin D alone, it should be emphasized that the primary endpoint in all studies was fracture prevention and that the different studies included different groups of participants. In some studies, effects of supplements were studied in the general population of elderly participants (13, 18–21), whereas other studies investigated supplements for secondary (11, 14) or tertiary (15) fracture prophylaxis. Vitamin D status before study entry may have differed between studies, and effects of vitamin D supplementation with or without calcium may depend on vitamin D status (and daily calcium intake). We cannot exclude the possibility that such characteristics, inherited in the design of the original studies, may affect mortality and may have influenced effects of vitamin D (and calcium). Epidemiological studies have suggested that mortality may be increased in participants with low vitamin D levels, although high levels could be harmful as well (41). It remains unknown whether this is due to the biological effect of vitamin D or due to confounders that it may not be possible to adjust fully for in epidemiological studies. Because plasma 25-hydroxyvitamin D levels were not measured and calcium intake was not assessed consistently at baseline or during follow-up in the trials included, unfortunately our data do not allow for further clarification on this matter.
In our study, we adjusted for incident fractures during trials to assess effects beyond an effect due to a reduced mortality caused by fewer fractures in response to treatment. Interestingly, antiresorptive treatments with bisphosphonates, strontium ranelate, or denosumab have been shown to reduce mortality, often to a greater extent than is explained by the reduction in fracture risk (42). Further studies are needed to investigate possible interrelationships between these findings.
In conclusion, our IPD analysis and trial level meta-analysis consistently showed reduced mortality in elderly participants randomized to vitamin D supplements in combination with calcium, whereas the analyses based on existing interventions and trial populations did not support an effect of vitamin D alone. Accordingly, calcium with vitamin D supplementation to elderly participants is overall not harmful to survival, and may have beneficial effects on general health.
Supplementary Material
Acknowledgments
We are grateful to the authors of the original trial reports, including Guro B. Smedshaug, Elisabeth Kvaavik, Jan A. Falch, Aage Tverdal, Jan I. Pedersen, Erik Roy Larsen, Anders Foldspang, Geoff Nicholson, Amanda Stuart, Elizabeth Williamson, Graeme MacLennan, Alison M. McDonald, Gladys C. McPherson, Adrian M. Grant, Marion K. Campbell, Helen Raphael, Sarah Crozier, Paul Maslin, Marjo Tuppurainen, Matti Karkkainen, Toni Rikkonen, Lorenzo Sandini, Joonas Sirola, Risto Honkanen, Esko Alhava, and Heikki Kroger. The authors thank the WHI investigators and staff for their dedication, and the study participants for making the program possible. A full listing of WHI investigators can be found at: https://cleo.whi.org/researchers/Documents%20%20Write%20a%20Paper/WHI%20Investigator%20Short%20List.pdf.
The WHI program is funded by the National Heart, Lung, and Blood Institute, National Institutes of Health, U.S. Department of Health and Human Services through contracts N01WH22110, 24152, 32100-2, 32105-6, 32108-9, 32111-13, 32115, 32118-32119, 32122, 42107-26, 42129-32, and 44221. K.M.S. acknowledges funding through the National Health and Medical Research Council, Australia, with supplemental funding from the Australian Commonwealth Department of Health and Ageing. L.R. acknowledges personal funding (Grant 09-070940) from the Danish Council for Independent Research in Medical Sciences. A.A. acknowledges personal funding from the UK Medical Research Council and the Chief Scientist Office of the Scottish Government Health Directorates. L.M. acknowledges support from the Danish Osteoporosis Association, The Municipality of the City of Randers, Randers Central Hospital, Aarhus County, Denmark, and The Pharmacy Association of 1991. The Health Services Research Unit, University of Aberdeen, is funded by the Chief Scientist Office of the Scottish Government Health Directorates. No funding source had influence on the present paper.
Author Contributions: L.R. is guarantor, had full access to all the data in the study, and takes responsibility for the management of the data received from the primary trials and the accuracy of the data analysis. L.R., B.A., A.A., T.M., K.B., H.E.M., J.A.R., L.M., and R.M.F. conceived and designed the study. L.R. and B.A. acquired and analyzed the data. L.R., B.A., T.M., A.A., L.M., and R.M.F. wrote the manuscript. All authors contributed to revising the manuscript critically and approved the final version, as well as contributing to the interpretation of the joint results and to the design of their individual trials.
Disclosure Summary: L.R. has received speakers' fees from Bristol-Myers Squibb and Eli Lilly. A.A. had study drugs for clinical trials of vitamin D and calcium supplementation supplied by Shire Pharmaceuticals and Nycomed. T.M. has served on advisory boards for Shire and has received lecture fees from Shire and Strakan Pharmaceuticals. L.M. had free study drugs of vitamin D and calcium supplied by Nycomed DAK. R.M.F. has served as an adviser to Nycomed, Shire, and Prostrakan, all of whom market calcium and vitamin D supplements. H.E.M. participates in a clinical trial of vitamin D supplementation sponsored by the University of Oslo and The Norwegian Women's Public Health Association. The study is also supported by Fürst Medisinsk Laboratorium and Nycomed, including free study drugs. J.E.M. and colleagues at Brigham and Women's Hospital, Harvard Medical School, are recipients of funding from the National Institutes of Health to conduct the VITamin D and OmegA-3 TriaL (VITAL), a large-scale randomized trial of vitamin D and omega-3 in the prevention of cancer and cardiovascular disease. C.C. has undertaken lecturing and consulting for Shire Pharmaceuticals and Nycomed. F.A. has had study drugs for clinical trials of vitamin D and calcium supplementation supplied by Shire Pharmaceuticals and Nycomed, and has undertaken lecturing and consulting for the same companies. K.B. has served on an advisory board for Osteologic, Servier and Amgen, received research grants from MSD, and served on speakers bureau for Servier, Amgen, and GSK. B.A. has received consultancy fees from Nycomed, Amgen, and Novartis; research grants from Roche and Amgen; and speakers' fees from Servier, Eli Lilly, and MSD. D.T. had study drugs for clinical trials of vitamin D and calcium supplementation supplied by Shire Pharmaceuticals. There were no conflicts of interest for E.T.J., H.E.S., K.S., J.A.R., K.M.S., and R.D.J.
Footnotes
- CaD
- Calcium with vitamin D
- CI
- confidence interval
- HR
- hazard ratio
- HRT
- hormone replacement therapy
- IPD
- individual patient data
- OR
- odds ratio.
References
- 1. Freedman DM, Looker AC, Abnet CC, Linet MS, Graubard BI. 2010. Serum 25-hydroxyvitamin D and cancer mortality in the NHANES III Study (1988–2006). Cancer Res 70:8587–8597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Chung M, Balk EM, Brendel M, Ip S, Lau J, Lee J, Lichtenstein A, Patel K, Raman G, Tatsioni A, Terasawa T, Trikalinos TA. 2009. Vitamin D and calcium: a systematic review of health outcomes. Evid Rep Technol Assess (Full Rep) 183:1–420 [PMC free article] [PubMed] [Google Scholar]
- 3. 2011 Dietary reference intakes for calcium and vitamin D. Washington, DC: Institute of Medicine [Google Scholar]
- 4. Autier P, Gandini S. 2007. Vitamin D supplementation and total mortality: a meta-analysis of randomized controlled trials. Arch Intern Med 167:1730–1737 [DOI] [PubMed] [Google Scholar]
- 5. Avenell A, Gillespie WJ, Gillespie LD, O'Connell D. 2009. Vitamin D and vitamin D analogues for preventing fractures associated with involutional and post-menopausal osteoporosis. Cochrane Database Syst Rev 2:CD000227. [DOI] [PubMed] [Google Scholar]
- 6. Bjelakovic G, Gluud LL, Nikolova D, Whitfield K, Wetterslev J, Simonetti RG, Bjelakovic M, Gluud C. 2011. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst Rev 7:CD007470. [DOI] [PubMed] [Google Scholar]
- 7. Lambert PC, Sutton AJ, Abrams KR, Jones DR. 2002. A comparison of summary patient-level covariates in meta-regression with individual patient data meta-analysis. J Clin Epidemiol 55:86–94 [DOI] [PubMed] [Google Scholar]
- 8. D.I.P.A.R.T. group, Abrahamsen B, Masud T, Avenell A, Anderson F, Meyer HE, Cooper C, Smith H, LaCroix AZ, Torgerson D, Johansen A, Jackson R, Rejnmark L, Wactawski-Wende J, Brixen K, Mosekilde L, Robbins JA, Francis RM. 2010. Patient level pooled analysis of 68,500 patients from seven major vitamin D fracture trials in US and Europe. BMJ 340:b5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Chapuy MC, Arlot ME, Delmas PD, Meunier PJ. 1994. Effect of calcium and cholecalciferol treatment for three years on hip fractures in elderly women. BMJ 308:1081–1082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lips P, Graafmans WC, Ooms ME, Bezemer PD, Bouter LM. 1996. Vitamin D supplementation and fracture incidence in elderly persons. A randomized, placebo-controlled clinical trial. Ann Intern Med 124:400–406 [DOI] [PubMed] [Google Scholar]
- 11. Meyer HE, Smedshaug GB, Kvaavik E, Falch JA, Tverdal A, Pedersen JI. 2002. Can vitamin D supplementation reduce the risk of fracture in the elderly? A randomized controlled trial. J Bone Miner Res 17:709–715 [DOI] [PubMed] [Google Scholar]
- 12. Trivedi DP, Doll R, Khaw KT. 2003. Effect of four-monthly oral vitamin D3 (cholecalciferol) supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. BMJ 326:469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Larsen ER, Mosekilde L, Foldspang A. 2004. Vitamin D and calcium supplementation prevents osteoporotic fractures in elderly community dwelling residents: a pragmatic population-based 3-year intervention study. J Bone Miner Res 19:370–378 [DOI] [PubMed] [Google Scholar]
- 14. Porthouse J, Cockayne S, King C, Saxon L, Steele E, Aspray T, Baverstock M, Birks Y, Dumville J, Francis R, Iglesias C, Puffer S, Sutcliffe A, Watt I, Torgerson DJ. 2005. Randomised controlled trial of calcium and supplementation with cholecalciferol (vitamin D3) for prevention of fractures in primary care. BMJ 330:1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Grant AM, Avenell A, Campbell MK, McDonald AM, MacLennan GS, McPherson GC, Anderson FH, Cooper C, Francis RM, Donaldson C, Gillespie WJ, Robinson CM, Torgerson DJ, Wallace WA. 2005. Oral vitamin D3 and calcium for secondary prevention of low-trauma fractures in elderly people (Randomised Evaluation of Calcium Or vitamin D, RECORD): a randomised placebo-controlled trial. Lancet 365:1621–1628 [DOI] [PubMed] [Google Scholar]
- 16. Law M, Withers H, Morris J, Anderson F. 2006. Vitamin D supplementation and the prevention of fractures and falls: results of a randomised trial in elderly people in residential accommodation. Age Ageing 35:482–486 [DOI] [PubMed] [Google Scholar]
- 17. Lyons RA, Johansen A, Brophy S, Newcombe RG, Phillips CJ, Lervy B, Evans R, Wareham K, Stone MD. 2007. Preventing fractures among older people living in institutional care: a pragmatic randomised double blind placebo controlled trial of vitamin D supplementation. Osteoporos Int 18:811–818 [DOI] [PubMed] [Google Scholar]
- 18. Smith H, Anderson F, Raphael H, Maslin P, Crozier S, Cooper C. 2007. Effect of annual intramuscular vitamin D on fracture risk in elderly men and women—a population-based, randomized, double-blind, placebo-controlled trial. Rheumatology (Oxford) 46:1852–1857 [DOI] [PubMed] [Google Scholar]
- 19. LaCroix AZ, Kotchen J, Anderson G, Brzyski R, Cauley JA, Cummings SR, Gass M, Johnson KC, Ko M, Larson J, Manson JE, Stefanick ML, Wactawski-Wende J. 2009. Calcium plus vitamin D supplementation and mortality in postmenopausal women: the Women's Health Initiative Calcium-Vitamin D Randomized Controlled Trial. J Gerontol A Biol Sci Med Sci 64:559–567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Sanders KM, Stuart AL, Williamson EJ, Simpson JA, Kotowicz MA, Young D, Nicholson GC. 2010. Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. JAMA 303:1815–1822 [DOI] [PubMed] [Google Scholar]
- 21. Salovaara K, Tuppurainen M, Kärkkäinen M, Rikkonen T, Sandini L, Sirola J, Honkanen R, Alhava E, Kröger H. 2010. Effect of vitamin D(3) and calcium on fracture risk in 65- to 71-year-old women: a population-based 3-year randomized, controlled trial–the OSTPRE-FPS. J Bone Miner Res 25:1487–1495 [DOI] [PubMed] [Google Scholar]
- 22. Romagnoli E, Mascia ML, Cipriani C, Fassino V, Mazzei F, D'Erasmo E, Carnevale V, Scillitani A, Minisola S. 2008. Short and long-term variations in serum calciotropic hormones after a single very large dose of ergocalciferol (vitamin D2) or cholecalciferol (vitamin D3) in the elderly. J Clin Endocrinol Metab 93:3015–3020 [DOI] [PubMed] [Google Scholar]
- 23. Bolland MJ, Avenell A, Baron JA, Grey A, MacLennan GS, Gamble GD, Reid IR. 2010. Effect of calcium supplements on risk of myocardial infarction and cardiovascular events: meta-analysis. BMJ 341:c3691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Dyer CA, Taylor GJ, Reed M, Dyer CA, Robertson DR, Harrington R. 2004. Falls prevention in residential care homes: a randomised controlled trial. Age Ageing 33:596–602 [DOI] [PubMed] [Google Scholar]
- 25. Higgins JPT, Green S. 2006. Cluster-randomized trials. Cochrane Handbook for Systematic Reviews of Interventions 4.2.6, Section 8.11.2. Issue 4. Chichester, UK: The Cochrane Collaboration [Google Scholar]
- 26. Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group 2009. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Chapuy MC, Pamphile R, Paris E, Kempf C, Schlichting M, Arnaud S, Garnero P, Meunier PJ. 2002. Combined calcium and vitamin D3 supplementation in elderly women: confirmation of reversal of secondary hyperparathyroidism and hip fracture risk: the Decalyos II study. Osteoporos Int 13:257–264 [DOI] [PubMed] [Google Scholar]
- 28. Flicker L, MacInnis RJ, Stein MS, Scherer SC, Mead KE, Nowson CA, Thomas J, Lowndes C, Hopper JL, Wark JD. 2005. Should older people in residential care receive vitamin D to prevent falls? Results of a randomized trial. J Am Geriatr Soc 53:1881–1888 [DOI] [PubMed] [Google Scholar]
- 29. Baeksgaard L, Andersen KP, Hyldstrup L. 1998. Calcium and vitamin D supplementation increases spinal BMD in healthy, postmenopausal women. Osteoporos Int 8:255–260 [DOI] [PubMed] [Google Scholar]
- 30. Latham NK, Anderson CS, Lee A, Bennett DA, Moseley A, Cameron ID. 2003. A randomized, controlled trial of quadriceps resistance exercise and vitamin D in frail older people: the Frailty Interventions Trial in Elderly Subjects (FITNESS). J Am Geriatr Soc 51:291–299 [DOI] [PubMed] [Google Scholar]
- 31. Brazier M, Grados F, Kamel S, Mathieu M, Morel A, Maamer M, Sebert JL, Fardellone P. 2005. Clinical and laboratory safety of one year's use of a combination calcium + vitamin D tablet in ambulatory elderly women with vitamin D insufficiency: results of a multicenter, randomized, double-blind, placebo-controlled study. Clin Ther 27:1885–1893 [DOI] [PubMed] [Google Scholar]
- 32. Schleithoff SS, Zittermann A, Tenderich G, Berthold HK, Stehle P, Koerfer R. 2006. Vitamin D supplementation improves cytokine profiles in patients with congestive heart failure: a double-blind, randomized, placebo-controlled trial. Am J Clin Nutr 83:754–759 [DOI] [PubMed] [Google Scholar]
- 33. Bolland MJ, Grey A, Avenell A, Gamble GD, Reid IR. 2011. Calcium supplements with or without vitamin D and risk of cardiovascular events: reanalysis of the Women's Health Initiative limited access dataset and meta-analysis. BMJ 342:d2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Weingarten MA, Zalmanovici A, Yaphe J. 2008. Dietary calcium supplementation for preventing colorectal cancer and adenomatous polyps. Cochrane Database Syst Rev 1:CD003548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Jacobs ET, Jurutka PW, Martínez ME, Alberts DS. 2009. Vitamin D, calcium, and colorectal neoplasia: new insights on mechanisms of action. Cancer Prev Res (Phila) 2:197–199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Bolland MJ, Grey A, Gamble GD, Reid IR. 2011. Calcium and vitamin D supplements and health outcomes: a reanalysis of the Women's Health Initiative (WHI) limited-access data set. Am J Clin Nutr 94:1144–1149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Reid IR, Bolland MJ, Grey A. 2008. Effect of calcium supplementation on hip fractures. Osteoporos Int 19:1119–1123 [DOI] [PubMed] [Google Scholar]
- 38. Hagström E, Hellman P, Larsson TE, Ingelsson E, Berglund L, Sundström J, Melhus H, Held C, Lind L, Michaëlsson K, Arnlöv J. 2009. Plasma parathyroid hormone and the risk of cardiovascular mortality in the community. Circulation 119:2765–2771 [DOI] [PubMed] [Google Scholar]
- 39. Chen JS, Sambrook PN, March L, Cameron ID, Cumming RG, Simpson JM, Seibel MJ. 2008. Hypovitaminosis D and parathyroid hormone response in the elderly: effects on bone turnover and mortality. Clin Endocrinol (Oxf) 68:290–298 [DOI] [PubMed] [Google Scholar]
- 40. Torgerson DJ. 2001. Contamination in trials: is cluster randomisation the answer? BMJ 322:355–357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Melamed ML, Michos ED, Post W, Astor B. 2008. 25-Hydroxyvitamin D levels and the risk of mortality in the general population. Arch Intern Med 168:1629–1637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Bolland MJ, Grey AB, Gamble GD, Reid IR. 2010. Effect of osteoporosis treatment on mortality: a meta-analysis. J Clin Endocrinol Metab 95:1174–1181 [DOI] [PubMed] [Google Scholar]
- 43. Krieg MA, Jacquet AF, Bremgartner M, Cuttelod S, Thiébaud D, Burckhardt P. 1999. Effect of supplementation with vitamin D3 and calcium on quantitative ultrasound of bone in elderly institutionalized women: a longitudinal study. Osteoporos Int 9:483–488 [DOI] [PubMed] [Google Scholar]
- 44. Komulainen M, Kröger H, Tuppurainen MT, Heikkinen AM, Alhava E, Honkanen R, Jurvelin J, Saarikoski S. 1999. Prevention of femoral and lumbar bone loss with hormone replacement therapy and vitamin D3 in early postmenopausal women: a population-based 5-year randomized trial. J Clin Endocrinol Metab 84:546–552 [DOI] [PubMed] [Google Scholar]
- 45. Avenell A, Grant AM, McGee M, McPherson G, Campbell MK, McGee MA, RECORD Trial Management Group 2004. The effects of an open design on trial participant recruitment, compliance and retention—a randomized controlled trial comparison with a blinded, placebo-controlled design. Clin Trials 1:490–498 [DOI] [PubMed] [Google Scholar]
- 46. Harwood RH, Sahota O, Gaynor K, Masud T, Hosking DJ. 2004. A randomised, controlled comparison of different calcium and vitamin D supplementation regimens in elderly women after hip fracture: the Nottingham Neck of Femur (NONOF) Study. Age Ageing 33:45–51 [DOI] [PubMed] [Google Scholar]
- 47. Meier C, Woitge HW, Witte K, Lemmer B, Seibel MJ. 2004. Supplementation with oral vitamin D3 and calcium during winter prevents seasonal bone loss: a randomized controlled open-label prospective trial. J Bone Miner Res 19:1221–1230 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.