Navigation requires animals to adjust ongoing movements in response to pertinent features of the environment and select between competing target cues. The neurobiological basis of navigational behavior in vertebrates is hard to analyze, partly because underlying neural circuits are experience dependent. Phototaxis in zebrafish is a hardwired navigational behavior [1, 2], performed at a stage when larvae swim using a small repertoire of stereotyped movements [3, 4, 5]. We established conditions to elicit robust phototaxis behavior and found that zebrafish larvae deploy directional orienting maneuvers and regulate forward swimming speed to navigate toward a target light. Using genetic analysis and targeted laser ablations, we show that retinal ON and OFF pathways play distinct roles during phototaxis. The retinal OFF pathway controls turn movements via retinotectal projections and establishes correct orientation by causing larvae to turn away from non-target areas. In contrast, the retinal ON pathway activates the serotonergic system to trigger rapid forward swimming toward the target. Computational simulation of phototaxis using an OFF-turn, ON-approach algorithm verifies that our model accounts for key features of phototaxis and provides a simple and robust mechanism for behavioral choice between competing targets.
Both positive and negative phototaxis behaviors have been reported in zebrafish [1, 2, 6, 7]. Larvae rapidly swim toward a low intensity target light spot which appears after the starting condition of uniform field illumination is extinguished (Figure 1A; Movie S1) showing maximal aggregation in the target area after approximately 60 seconds (Figure 1B). The speed of movement toward the target light is maximal when the target light is 10 fold less intense than pre-adapted illumination levels. At high target light intensities, larvae show negative phototaxis, moving slowly away from the illuminated spot (Figure 1C). Thus the rate and direction of phototaxis is determined by the relative intensities of the uniform illumination and the target light.
Figure 1.
Zebrafish larvae perform positive and negative phototaxis.
a Schematic showing the phototaxis conditions. Larvae were maintained under uniform illumination and tested for phototaxis by transitioning to a dark field with a single light spot. b Swim tracks for nine larvae during phototaxis superimposed over an image of the target arena showing the position of the target light (U/RTI=100/-1). Circles indicate starting positions. Scale bar 5 mm. c Phototaxis is associated with larvae rapidly orienting toward the target (red trace), reflected in a decreased Orientation to Target (OT) score (inset gives example of larvae oriented 45°). Low OT scores are maintained during target approach. Once larvae reach the target their orientation randomizes and mean OT returns to 90°. Mean distance from target is minimal after 60s of phototaxis (blue trace). n=6 groups (total 180 larvae). d The rate of phototaxis depends on the relative intensity of the target light and the light source to which larvae are pre-adapted. Larvae were pre-adapted to light intensities of 0.65 μW/cm2 (grey), 6.5 μW/cm2 (blue) or 65 μW/cm2 (orange) and maintained in uniform illumination of the same intensity in the testing arena. Relative target intensity = log10 ( (intensity in the center of the target spot light) / (pre-adapted intensity) ). We measured the mean larval distance to the target at 0.5 s intervals over the first 5 seconds of phototaxis, performed a linear fit to the data and calculated the rate of phototaxis by taking the gradient of the fitted line. n=4 groups per adaptation/target intensity pair. At the highest relative target light intensity, larvae showed a slow but significant rate of movement away from the target. * P < 0.05 for one-sample t-test versus 0, color indicates the pre-adaptation intensity for the point subject to statistical test. See also Figure S1 and Movie S1.
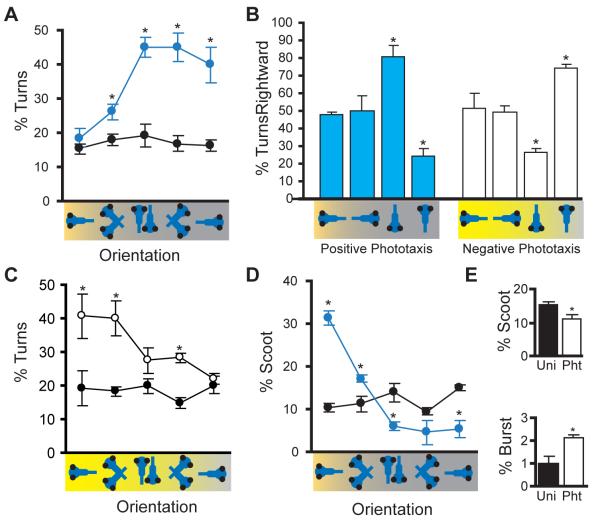
Routine turns (‘turns’) and slow swims (‘scoots’) are the main modes of patterned movement during unstimulated swimming [3, 5]. As turn maneuvers could be used to steer larvae toward a target, we measured the frequency with which turns are initiated during positive phototaxis. Distance from the target light only slightly modulated turn movement frequency (Figure S1A). In contrast, turn frequency was strongly influenced by the orientation of larvae relative to the target. Larvae oriented perpendicular to or away from the target, showed a more than two-fold increase in the frequency of turn initiations (Figure 2A) and deployed larger magnitude turns than seen under uniform illumination (Figure S2A). Under baseline conditions turns are initiated with equal frequency to the left and right. However, during positive phototaxis, turns are preferentially directed toward the light: larvae facing the target light with their right eye preferentially turn rightward, whereas larvae facing the target light with their left eye initiate mostly leftward turns (Figure 2B). A distinct pattern of turn modulation was observed during negative phototaxis where turns were deployed most frequently when larvae were oriented toward the intense light spot (Figure 2C), these turns also being larger in amplitude (Figure S2A). During negative phototaxis, turns were directed away from the target light (Figure 2B). We conclude that turn maneuvers are modulated to allow larval zebrafish to control their orientation during phototaxis.
Figure 2.
Motor control during phototaxis.
a Larvae oriented away from the target during positive phototaxis show high rates of turn movements. Graph shows the frequency of turn initiations during uniform illumination (black) and for the first 5 seconds of positive phototaxis (blue, U/RTI=100/-1) for subsets of larvae in the schematized orientations (n=4 groups). In all figures, the x-axis shows orientation groups relative to a target light to the left. * P < 0.05 versus corresponding orientation during uniform illumination. b Turn movements are directed toward weak target lights and away from intense target lights. Bars show the percentage of turns initiated in a rightward direction: 50% represents no directional bias. During positive phototaxis (blue bars, n = 5 groups, U/RTI=100/-1) and negative phototaxis (white bars, n = 9 groups, U/RTI=20/+2) larvae oriented toward or away from the target show no bias, but larvae oriented perpendicularly to the target show significant directionality of turn movements toward or away from the target respectively. * P < 0.01 for one sample t-test against 50%. c During negative phototaxis, larvae facing an intense target light increase the frequency of turn movements. Turn frequency during uniform illumination (black) and during the first 5 seconds of negative phototaxis (open circles, U/RTI=15/+2) for larvae in the schematized orientations (n=6 groups). * P < 0.05 versus corresponding orientation during uniform illumination. d Larvae oriented toward the target produce high levels of scoot movements during positive phototaxis. The initiation frequency of scoot movements is shown during uniform illumination (black) and for subsets of larvae in the schematized orientations (blue, n=4 groups, U/RTI=100/-1). * P < 0.05 versus corresponding orientation during uniform illumination. e. During negative phototaxis (U/RTI=15/+2), larvae (n=6 groups) show a small reduction in the initiation frequency of scoot movements (‘Pht’, white bar) compared to uniform illumination (‘Uni’, black bar). Burst movements show a small increase above baseline (two-way ANOVA, main effect of test condition F(1,50)=11.2, P=0.002), but rates remain very low and are not modulated by orientation (no main effect of orientation F(4,50)=1.67, P=0.17 or interaction between test condition and orientation F(4,50)=0.59, P=0.67). * P < 0.05. See also Figure S2.
Scoots and burst swims (‘bursts’) are maneuvers which propel larvae forward [3, 5, 8] and could be used to approach the target light. During positive phototaxis larvae directly facing the target showed a rate of scoot initiations elevated almost 3-fold above baseline (Figure 2D) and an increase in burst swim initiations (Figure S2C). In negative phototaxis, scoots were reduced and burst swim frequency remained extremely low (Figure 2E). Neither the frequency of scoots or bursts was modulated by orientation. Thus larvae swim quickly forward during positive phototaxis but not during negative phototaxis. We focused on analyzing neural mechanisms of positive phototaxis, where movement is more strongly modulated. During phototaxis the testing arena is dark except for the illuminated target spot, thus larval movement may produce increases or decreases in light intensity across each retina. Separate populations of ON and OFF retinal ganglion cells relay information about increments and decrements in light intensity to the brain [9]. We therefore asked whether ON and OFF pathways are used for navigation during phototaxis.
Scoot frequency is maximal when larvae face the target light with the darkest area of the testing arena behind them. We therefore presented larvae with targets simultaneously illuminated at opposite ends of the testing arena, hypothesizing that if light ahead is the optimal cue, scoot frequency would be elevated when larvae faced either light, but if dark behind is the stimulus, larvae oriented perpendicular to the two targets would show the greatest rate of scoots. Scoot initiations were maximal when larvae faced either of the two targets (Figure 3A) suggesting that target approach is triggered by the retinal ON pathway. To verify this, we examined whether the modulation of scoots is impaired in no optokinetic response c (nrc) mutant larvae which have a selective disruption of the retinal ON pathway[10, 11]. nrc wildtype sibling larvae behaved like wildtype larvae, and increased the frequency of scoots above baseline levels when facing the target light (Figure 3B). Mutants failed to potentiate scoots (Figure 3C) confirming that the retinal ON pathway drives forward swimming during phototaxis. Locomotor activity in zebrafish and other vertebrates is regulated by the serotonergic system [12, 13]. Neither the selective serotonin reuptake inhibitor fluoxetine, nor the non-selective 5HT receptor antagonist methysergide significantly altered the frequency of scoot initiations under uniform illumination (insets in Figure 3D, E). However, during phototaxis, treatment with fluoxetine increased the rate of scoots selectively for larvae oriented toward the target (Figure 3D). Conversely, after treatment with methysergide, larvae oriented toward the target failed to potentiate scoots (Figure 3E). These experiments show that serotonin signaling is a key part of the neuronal pathway through which retinal ON signaling drives approach movements during phototaxis.
Figure 3.
The retinal ON pathway triggers scoots
a Initiation frequencies of scoots when larvae are tested with a single target (1, blue circles, U/RTI=100/-1, n=6 groups) or two equally intense lights on opposite sides of the testing arena (1+2, green circles, n=6 groups). Dotted line shows the rate of scoot initiations under uniform illumination (n=6 groups). As orientation relative to the target differs for the two lights, absolute orientation of the larvae was used and recordings restricted to a 15×15 mm patch in the center of the testing arena. * P < 0.05 verses uniform illumination. b Wildtype nrc siblings show increased scoot movements when oriented toward the target and reduced levels of scoot movements when facing away from the target. * P < 0.05 for schematized orientation during phototaxis (grey bars) compared to uniform illumination (black bars). In contrast, c nrc mutants show similar levels of scoot movements during uniform illumination (black bar) and when facing the target light during phototaxis (grey bars). d The SSRI fluoxetine (30 μM, green) increases the rate of scoot initiations compared to larvae treated with vehicle DMSO alone (black circles). This increase was specific to larvae facing the target light during phototaxis (U/RTI=100/-2, n=3 groups each treatment), and not apparent during baseline movement under uniform illumination (inset). Drug was added 15 minutes before testing. * P < 0.05 compared to same orientation in DMSO treated groups. e Methysergide (80 μM, red) a non-selective serotonin receptor antagonist, suppresses the increase in scoot initiations seen for vehicle larvae (black) facing the target during phototaxis (U/RTI=100/-1, n=5 groups per condition). Methysergide does not impair locomotor activity under uniform illumination (inset). Drug was added 120 minutes before testing. * P < 0.01 compared to same orientation in DMSO groups.
In mammals the superior colliculus is required for orienting responses to spatially localized stimuli [14, 15]. The homologous structure in fish is the optic tectum, which in zebrafish is the primary target of retinal ganglion cells [16]. Retinal ganglion cell projections are completely crossed in zebrafish larvae. We therefore disrupted a visual hemifield by severing one optic nerve, or laser ablating a single tectal lobe (Figure S3A), visualized using the Ath5:GFP transgenic line [17]. As a control for mounting and laser surgery, we ablated visual region AF-7 [16, 18]. No bias in turn directionality was evident under uniform illumination after either tectal ablation or unilateral optic nerve section (Figure S3B, C), demonstrating that the absence of a signal from the operated eye does not constitutively influence turn direction.
Surprisingly, during phototaxis, turns were normal when the target light was in the blind visual field but made in the wrong direction when the target was in the intact visual field. The left optic nerve and its target, the right tectum, carry information arising from the left visual field - yet after lesion of the left optic nerve or the right tectum, larvae successfully turned toward targets in the blind left visual field, but paradoxically oriented away from targets in the intact right visual field (Figure 4A,B).
Figure 4.
The retinotectal OFF pathway triggers turns allowing target selection
a Percentage of turns initiated in a rightward direction after Mock operation (‘Mock’, n=9 groups), unilateral section of right optic nerve (‘R Opn’, n=5 groups) or unilateral section of left optic nerve (‘L OpN’, n=6 groups). Turn direction was separately assessed for larvae facing the target with left or right eye as indicated. * P < 0.01 against corresponding orientation in mock. U/RTI=100/-1 for all groups. b Percentage of turns initiated in a rightward direction after laser ablation of right arborization field 7 (‘R A7’, n=11 larvae), right optic tectum (‘R Tec’, n=14 larvae) or sham operation (‘Mock’, n=10 larvae). Turns show significant directional bias for all orientations in all groups (P < 0.05 for one sample t-test versus 50%). * P < 0.001. c Modulation of turn frequency during phototaxis in the nrc retinal ON pathway mutant. Larvae (n=20) oriented either perpendicular to or away from the target show a significant increase in the frequency of turn initiations during phototaxis (grey bars, U/RTI=300/-1 ) relative to uniform illumination (black bar). * P < 0.05. Moreover, d turns initiated by mutants show a robust directional bias toward the target light. One sample t-test, * P < 0.01 versus 50%. Mutants showed the same overall preference for turning toward the target light as sibling larvae (target bias in siblings 77.4 % ± 8.6%, mutants 83.3 % ± 16.7 %, t-test P=0.76). e Schematic illustrating information flow through retinotectal ON and OFF pathways when the target light is in the blind visual field. The OFF pathway model enables larvae to orient toward the target light by turning away from the eye perceiving a light decrement. f Simulation of zebrafish-like agents whose movement is controlled by an OFF-turn, ON-approach algorithm (blue traces) demonstrates that this is a plausible navigational strategy to implement phototaxis. g When a second target was simultaneously presented in the model, zebrafish-like agents efficiently selected a single navigational target, swimming directly toward it. h Zebrafish larvae show choice behavior in a two target test, swimming toward one target and ignoring the second. Traces show the swim paths of individual larvae starting in the center of the testing arena and followed for 15s of phototaxis (or until they reached a target spot). i Quantification behavior in two choice test. The graph shows the distribution of trajectories taken by larvae presented with a single target light at the top of the testing arena (n=27 larvae), on the right side of the arena (n=21 larvae) or in both positions simultaneously (n=16 larvae). The area of each circle indicates the proportion of larvae following the trajectory indicated on the Y-Axis. There were no instances were larvae swam in trajectories outside the range illustrated. Trajectories were measured over the first 5s of phototaxis with recordings made at 25 frames per second. See also Figure S3.
To resolve this paradox, we hypothesized that the retinal OFF pathway drives turns away from the eye experiencing the greater reduction in light intensity. Decrements in light intensity occur during the initial transition from uniform illumination to phototaxis conditions and when larvae inadvertently turn off their trajectory toward the target. Supporting this, we found that the frequency of turns was greater when the intact eye faced the dark side of the testing arena (35.3 % ± 2.9 %) than when it faced the target light (26.1 % ± 3.2 % ; n=11 groups, paired t-test P=0.003). This hypothesis also explains our observation that larvae positioned further away from the target light generate more turns (Figure S1): these larvae are exposed to the greatest reduction in light intensity during the transition to phototaxis. As an independent line of evidence, we measured turns in the nrc mutant. Anatomical and electroretinographic correlates of the OFF pathway are spared in nrc [19]. We found that nrc mutants show a vigorous increase in turn initiations during phototaxis (Figure 4C) and also accurately turn toward the target light (Figure 4D). These results confirm that orientation during phototaxis is achieved by using the OFF retinotectal pathway to avoid dark areas of the testing arena (Figure 4E).
Next we asked whether a mechanism where larvae move forward after light increments, and turn away from light decrements, is a plausible strategy for phototaxis. Simulation confirmed that larvae successfully navigate toward a target light using such a mechanism (Figure 4F). Intriguingly, when we added a second target light to the simulation, navigational paths were directed toward one or the other of the targets (Figure 4G). This prompted us to test whether larvae can choose a target when presented with competing cues. When presented with two spatially separated targets of the same intensity, larvae in a central starting zone selected and swam toward one target (Figure 4H, I). The same OFF-turn, ON-approach mechanism that enables navigation toward a single target also accounts for behavioral choice between competing cues.
Discussion
Animals navigate using a variety of strategies [20]. During phototaxis zebrafish larvae navigate toward a target by deploying distinct stereotyped movements, turns versus scoot or burst movements, depending on their orientation. Phototaxis is a tunable behavior, as target lights of similar or weaker intensity than the preadaptation level elicit approach, while larvae turn away from target lights significantly more intense than the preadaptation level. This finding may resolve why both dark and light preference have been described in adult zebrafish [6, 7]. It has been proposed that a common strategy for young organisms is to approach low intensity stimuli and withdraw from high intensity stimuli [21]. Our findings concord well with this idea. Such simple rules offer robust solutions to the problem of hardwiring organisms to face variable environmental challenges.
In mammals removal of one superior colliculus impairs orienting responses toward stimuli in the contralateral visual hemifield [14, 15]. Yet here we show that unilateral ablation of the homologous structure in fish, the optic tectum, disrupts orienting toward target lights in the ipsilateral visual field. This contradiction is resolved by our hypothesis that orienting turns are triggered by OFF signals in the eye facing the dark side of the testing arena. When the target light is contralateral to the ablated tectum, OFF signals from the dark side of the testing arena activate the intact tectum enabling larvae to turn toward the target. A corollary is that during phototaxis, each tectum initiates ipsiversive turns. Supporting this, in prey catching behavior in larvae, the tectum activates motor responses primarily via a pair of ipsilaterally projecting reticulospinal neurons [22]. The hypothesis is also consistent with our previous finding that the sudden onset of uniform darkness induces a transient period in which larvae repeatedly execute turn maneuvers [5]. In phototaxis experiments, the sudden onset of darkness is accompanied by the presentation of a target light resulting in an unequal decrement in light intensity at each eye and enabling the larva to orient away from the eye experiencing the greater reduction in light intensity. This mechanism also enables the larva to remain properly oriented as it swims toward the target - if it turns away from the target the larger decrement in light intensity at one eye will trigger a corrective contraversive movement. Thus the retinal OFF pathway is involved in steering during phototaxis. Conversely, we propose that the ON pathway controls the rate of approach. When directly facing the target light, a forward scoot movement produces an increase in light intensity on each retina. This signal triggers another scoot forward allowing the larva to rapidly swim toward the target light. The loss of the retinal ON pathway in nrc mutants disrupts this positive feedback loop, and larvae approach the target only at baseline movement rates. We show that the serotonergic system plays a key role in enabling rapid forward movement. No direct connections between retinal ganglion cells and serotonergic neurons have been described in zebrafish and further experiments will be required to identify the pattern of connectivity underlying approach movements. Modeling of an OFF-turn, ON-approach strategy showed that it might enable larvae to choose between competing target cues. This is not a universal feature of navigational strategies. For instance, during phototaxis Drosophila move forward when stimulation of the eyes is balanced [23], a strategy that results in orienting between targets in the two light test [20]. We found that zebrafish larvae can indeed successfully select a target light in a two target test. Thus this navigational strategy provides a simple solution to the problem of behavioral choice when multiple targets are present in the visual world.
Here we show that opposing visual channels control distinct maneuvers employed for phototaxis in zebrafish larvae: the ON pathway controls the rate of approach by activating scoot maneuvers while the OFF pathway deploys turns to enable steering. The control of turns shows an intriguing similarity to the pathway for food-seeking behavior in C. elegans, where loss of an odorant activates interneurons which facilitate turn movements [24]. The olfactory circuit which mediates this behavior in C. elegans shows striking molecular and cellular parallels to the vertebrate OFF bipolar cell pathway. Our evidence extends this analogy by showing that a vertebrate OFF pathway also specifically controls turning movements in response to stimulus withdrawal.
Experimental Procedures
Behavioral experiments were conducted on zebrafish larvae ages 5-7 days post fertilization. Larvae were first pre-adapted to light of the same intensity as in the testing arena. An LED array mounted above the arena provided uniform illumination while a single LED mounted below illuminated a target spot on one edge of the arena during phototaxis. The spot remained continuously illuminated for 5 seconds during phototaxis trials. As the rate and direction of phototaxis depends on the relative intensity of the uniform illumination field and the target light, for each experiment we specify both the intensity of the uniform field and the log10 of the relative target intensity (U/RTI). Thus where the uniform field is 100 μW/cm2 and the target intensity is 10 μW/cm2, the U/RTI = 100/-1. Video was captured using a Motionpro high-speed camera (Redlake, Tucson, AZ, USA) at 1000 frames per second. Behavioral analysis was carried out using the Flote analysis package [5, 25]. Cartoons schematize orientation relative to the target light. Each orientation depicted includes angles 22.5° on either side, covering a sector of 45°. In figures showing larvae tested under uniform illumination conditions, the orientation groups are relative to the position of the target during phototaxis trials. A detailed description of experimental procedures and behavioral analyses is provided in the online supplement.
Supplementary Material
Acknowledgements
This work was supported by an NRSA postdoctoral fellowship to H.B. and National Institutes of Health grants (MH075691 and HD37975) to M.G.
References
- 1.Brockerhoff S, Hurley J, Janssen-Bienhold U, Neuhauss S, Driever W, Dowling J. A Behavioral Screen for Isolating Zebrafish Mutants with Visual System Defects. Proceedings of the National Academy of Sciences. 1995;92:10545–10549. doi: 10.1073/pnas.92.23.10545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Orger M, Baier H. Channeling of red and green cone inputs to the zebrafish optomotor response. Visual Neuroscience. 2005;22:275–281. doi: 10.1017/S0952523805223039. [DOI] [PubMed] [Google Scholar]
- 3.Budick S, O’Malley D. The behavioral repertoire of larval zebrafish: swimming, escaping and prey capture. J Exp Biol. 2000;203:2565–2579. doi: 10.1242/jeb.203.17.2565. [DOI] [PubMed] [Google Scholar]
- 4.McElligott M, O’Malley D. Prey Tracking by Larval Zebrafish: Axial Kinematics and Visual Control. Brain Behav Evol. 2005;66:177–196. doi: 10.1159/000087158. [DOI] [PubMed] [Google Scholar]
- 5.Burgess H, Granato M. Modulation of locomotor activity in larval zebrafish during light adaptation. Journal of Experimental Biology. 2007;210:2526–2539. doi: 10.1242/jeb.003939. [DOI] [PubMed] [Google Scholar]
- 6.Serra E, Medalha C, Mattioli R. Natural preference of zebrafish (Danio rerio) for a dark environment. Brazilian Journal of Medical and Biological Research. 1999;32:1551–1553. doi: 10.1590/s0100-879x1999001200016. [DOI] [PubMed] [Google Scholar]
- 7.Gerlai R, Lahav M, Guo S, Rosenthal A. Drinks like a fish: zebra fish (Danio rerio) as a behavior genetic model to study alcohol effects. Pharmacol Biochem Behav. 2000;67:773–782. doi: 10.1016/s0091-3057(00)00422-6. [DOI] [PubMed] [Google Scholar]
- 8.Muller U, van Leeuwen J. Swimming of larval zebrafish: ontogeny of body waves and implications for locomotory development. Journal of Experimental Biology. 2004;207:853–868. doi: 10.1242/jeb.00821. [DOI] [PubMed] [Google Scholar]
- 9.Schiller PH, Sandell JH, Maunsell JHR. Functions of the ON and OFF channels of the visual system. Nature. 1986;322:824–825. doi: 10.1038/322824a0. [DOI] [PubMed] [Google Scholar]
- 10.Allwardt BA, Lall AB, Brockerhoff SE, Dowling JE. Synapse Formation Is Arrested in Retinal Photoreceptors of the Zebrafish nrc Mutant. J. Neurosci. 2001;21:2330–2342. doi: 10.1523/JNEUROSCI.21-07-02330.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Van Epps HA, Hayashi M, Lucast L, Stearns GW, Hurley JB, De Camilli P, Brockerhoff SE. The zebrafish nrc mutant reveals a role for the polyphosphoinositide phosphatase synaptojanin 1 in cone photoreceptor ribbon anchoring. J Neurosci. 2004;24:8641–8650. doi: 10.1523/JNEUROSCI.2892-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brocco M, Dekeyne A, Veiga S, Girardon S, Millan M. Induction of hyperlocomotion in mice exposed to a novel environment by inhibition of serotonin reuptake. A pharmacological characterization of diverse classes of antidepressant agents. Pharmacol Biochem Behav. 2002;71:667–680. doi: 10.1016/s0091-3057(01)00701-8. [DOI] [PubMed] [Google Scholar]
- 13.Brustein E, Chong M, Holmqvist B, Drapeau P. Serotonin patterns locomotor network activity in the developing zebrafish by modulating quiescent periods. J Neurobiol. 2003;57:303–322. doi: 10.1002/neu.10292. [DOI] [PubMed] [Google Scholar]
- 14.Sprague JM, Meikle TH. The Role of the Superior Colliculus in Visually Guided Behavior. Experimental Neurology. 1965;11:115–146. doi: 10.1016/0014-4886(65)90026-9. [DOI] [PubMed] [Google Scholar]
- 15.Schneider GE. Two Visual Systems: Brain mechanisms for visual discrimination and Localization are dissociated by tectal and cortical lesions. Science. 1969;163:895–902. doi: 10.1126/science.163.3870.895. [DOI] [PubMed] [Google Scholar]
- 16.Burrill JD, Easter SS., Jr. Development of the retinofugal projections in the embryonic and larval zebrafish (Brachydanio rerio) J Comp Neurol. 1994;346:583–600. doi: 10.1002/cne.903460410. [DOI] [PubMed] [Google Scholar]
- 17.Masai I, Lele Z, Yamaguchi M, Komori A, Nakata A, Nishiwaki Y, Wada H, Tanaka H, Nojima Y, Hammerschmidt M, et al. N-cadherin mediates retinal lamination, maintenance of forebrain compartments and patterning of retinal neurites. Development. 2003;130:2479–2494. doi: 10.1242/dev.00465. [DOI] [PubMed] [Google Scholar]
- 18.Roeser T, Baier H. Visuomotor behaviors in larval zebrafish after GFP-guided laser ablation of the optic tectum. J Neurosci. 2003;23:3726–3734. doi: 10.1523/JNEUROSCI.23-09-03726.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Emran F, Rihel J, Adolph AR, Wong KY, Kraves S, Dowling JE. OFF ganglion cells cannot drive the optokinetic reflex in zebrafish. Proc Natl Acad Sci U S A. 2007;104:19126–19131. doi: 10.1073/pnas.0709337104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fraenkel G, Gunn D. The Orientation of Animals, Kineses, Taxes and Compass Reactions. Dover Publications; 1961. [Google Scholar]
- 21.Schneirla T. An evolutionary and developmental theory of biphasic processes underlying approach and withdrawal. Nebraska Symposium on Motivation. 1959;7:1–42. [Google Scholar]
- 22.Gahtan E, Tanger P, Baier H. Visual Prey Capture in Larval Zebrafish Is Controlled by Identified Reticulospinal Neurons Downstream of the Tectum. Journal of Neuroscience. 2005;25:9294–9303. doi: 10.1523/JNEUROSCI.2678-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hotta Y, Benzer S. Genetic Dissection of the Drosophila Nervous System by means of Mosaics. Proceedings of the National Academy of Sciences. 1970;67:1156–1163. doi: 10.1073/pnas.67.3.1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chalasani SH, Chronis N, Tsunozaki M, Gray JM, Ramot D, Goodman MB, Bargmann CI. Dissecting a circuit for olfactory behaviour in Caenorhabditis elegans. Nature. 2007;450:63–70. doi: 10.1038/nature06292. [DOI] [PubMed] [Google Scholar]
- 25.Burgess H, Granato M. Sensorimotor Gating in Larval Zebrafish. Journal of Neuroscience. 2007;27:4984. doi: 10.1523/JNEUROSCI.0615-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.