Abstract
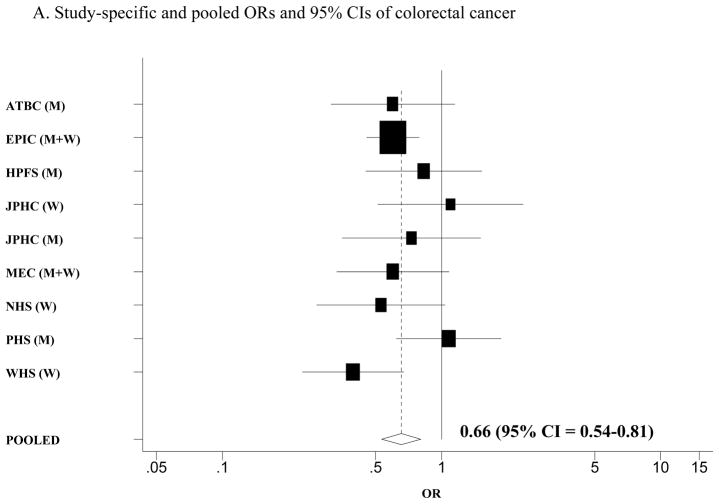
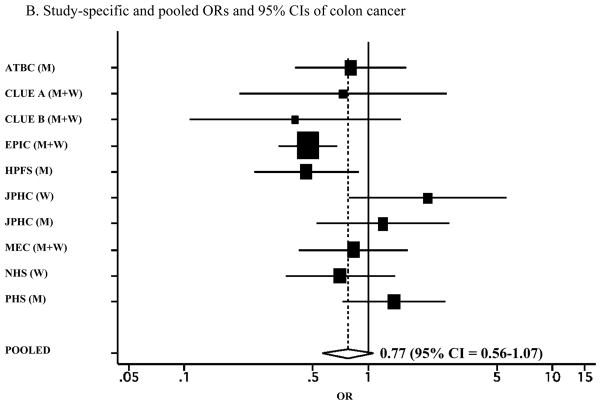
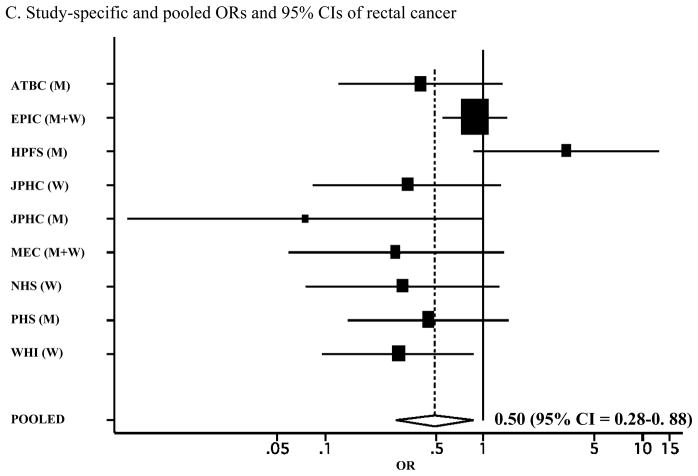
It remains unknown whether increased risk with low levels of vitamin D is present for colon and/or rectal cancer. To investigate the association between circulating vitamin D levels and colon and rectal cancer, we examined the associations between plasma levels of 25-hydroxyvitamin D (25(OH)D) and 1,25-dihydroxyvitamin D (1,25(OH)2D) and colon and rectal cancer in the Physicians’ Health Study and then conducted a meta-analysis of eight prospective studies of circulating levels of 25-hydroxyvitamin D (25(OH)D) and colon and rectal cancers, including the Physicians’ Health Study. Study-specific odds ratios (ORs) and 95% confidence intervals (CIs) were pooled using a random-effects model. A total of 1822 colon and 868 rectal cancers were included in the meta-analysis. We observed a significant inverse association for colorectal cancer (OR = 0.66; 95% CI = 0.54–0.81), comparing top versus bottom quantiles of circulating 25(OH)D levels. The inverse association was stronger for rectal cancer (OR = 0.50 for top versus bottom quantiles; 95% CI = 0.28–0.88) than colon cancer (OR = 0.77; 95% CI = 0.56–1.07; P for difference between colon and rectal cancer = 0.20). These data suggest an inverse association between circulating 25(OH)D levels and colorectal cancer, with a stronger association for rectal cancer.
Introduction
Vitamin D may decrease cancer risk by improving differentiation and apoptosis and decreasing proliferation, invasiveness, metastatic potential, and angiogenesis (1–4). Ultraviolet radiation is required for conversion of 7-dehydrocholesterol to vitamin D (cholecalciferol) in the skin, which is then hydroxylated to 25-hydroxylvitamin D (25(OH)D) in the liver. This 25(OH)D is converted to 1,25(OH)2D (the most active metabolite of vitamin D) by 1-α-hydroxylase (5). Low vitamin D status has been suggested to increase colorectal cancer (6–10). Some previous studies found a stronger inverse association for colon cancer compared to rectal cancer (8–9, 11), but another studies found that the association was particularly strong for rectal cancer (7, 12–14).
To examine whether circulating blood vitamin D levels were inversely associated with colon or rectal cancer separately, we examined the associations of plasma levels of 25(OH)D and 1,25(OH)2D in relation to risk of colon and rectal cancer in the Physicians’ Health Study and performed a meta-analysis of eight individual prospective studies (1822 colon and 868 rectal cancers and their matched controls), including the Physicians’ Health Study and three recently published studies (9–10, 12) that were not reviewed in a previous meta-analysis, which included four studies (15).
Materials and Methods
Physicians’ Health Study
The Physicians’ Health Study was a randomized, double-blind, placebo-controlled trial of aspirin and β-carotene for the primary prevention of cancer and cardiovascular disease among 22,071 US male physicians aged 40 to 84 enrolled in 1982 (16). Participants were excluded if they had a prior diagnosis of myocardial infarction, stroke, or transient ischemic attack, cancer (except non-melanoma skin cancer), renal or liver disease, peptic ulcer, or gout or used vitamin A or β-carotene supplements.
Blood samples were obtained during 1982 and 1984 (more than 70% of participants provided blood between September and November 1982) before randomization from 14,916 men. Blood collection kits were sent to all participants with instructions to have their blood drawn into the EDTA tubes. These samples were centrifuged, sent to Channing Laboratory, divided into aliquots, and stored at −82°C (later, at −140°C). Information on height, weight, physical activity, alcohol intake, multivitamin use, and smoking habits was collected by self-administered questionnaires at baseline. The frequency of intakes of whole milk, skim/low-fat milk, ice cream, hard cheese, and cold cereal with specified portions was obtained on the 18-week or 12-month questionnaire. Daily dairy calcium consumption was calculated using the U.S. food composition databases (17). This study was approved by the Institutional Review Board of the Brigham and Women’s Hospital.
Most cases were initially identified by annual follow-up questionnaires and the others were identified through mortality follow-up procedures. Initial reports were confirmed by review of medical records by the end-point committee. Among those who provided baseline blood samples, colorectal cases were ascertained through March 31, 2000, and controls were matched on age (±1 year for younger participants, up to ±5 years for older participants) and smoking status (never, past, current). A total of 229 colorectal cancer cases and 389 controls were included in the present analysis.
Plasma concentrations of 25(OH)D and 1,25(OH)2D were measured by radioimmunosorbent assay in the laboratory of Dr. Bruce Hollis (Medical University of South Carolina, Charleston, South Carolina). The median intraassay coefficient of variation (CV) from blinded quality-control samples was 13.8% for plasma 25(OH)D and 10.3% for plasma 1,25(OH)2D. Cases and their controls were analyzed in the same batch, and laboratory personnel were blinded to case, control, and quality-control status.
Identification and Selection of Studies for a Meta-Analysis of 25(OH)D and Colon and Rectal Cancer
We searched the PubMed database for the period from January 1966 to February 2010 using the terms ((vitamin D [MeSH] and “vitamin D” or “25-hydroxyvitamin D”) and (“cohort” or “case-control” or “case-cohort” or “epidemiology”) and (“cancer”)). The search was restricted to human studies published in English-language articles. We also examined references from these publications to identify further studies. We identified nine studies (7–14, 18) that examined serum or plasma 25(OH)D and colorectal cancer incidence in a prospective design (blood drawn prior to diagnosis of cancer). We contacted authors of three papers (10–12, 18) that did not report results for colon and rectal cancer separately. One study provided the odds ratios (ORs) and 95% confidence intervals (95% CIs) for rectal cancer, but not for colon cancer (10). Another study provided the ORs and 95% CIs for both colon and rectal cancers (12). Two reports from the CLUE cohort reported results for only colon cancer using the same dataset with different time periods (1975–1983 and 1984–1991)(11, 18). As a result, we included in this meta-analysis a total of seven published individual prospective studies of colon cancer (7–9, 11–14, 18) and seven studies of rectal cancer (7–10, 12–14), plus the present data from the Physicians’ Health Study.
Statistical Analyses
In the Physicians’ Health Study, to estimate ORs and 95% CIs, participants were categorized into quartiles based on plasma vitamin D metabolite levels among the controls. To remove the variation in vitamin D metabolites due to season of blood collection, we regressed plasma 25(OH)D or 1,25(OH)2D on the periodic function -sin(2ΠX/12)-cos(2ΠX/12), where X is the month of sample collection (19). The residuals from this model were added to the ethnic-specific plasma 25(OH)D means (Caucasian or non-Caucasian). We used a conditional logistic regression model to account for the matched case-control study design. To test for trend across quartiles, participants were assigned the median value of their quartile level. This variable was entered as a continuous term in the model, the coefficient for which was evaluated by the Wald test. In the multivariate analyses, we adjusted for season of blood collection (winter/spring, summer/autumn), ethnicity (Caucasian, non-Caucasian), fasting status (≤8, > 8 hours), body mass index (BMI; <23, 23–<25, 25–<27, ≥27 kg/m2), dairy calcium intake (≤146, 146<–340, 340<–580, ≥580 mg/d), and vigorous exercise (<1, 1–4 times/wk, ≥5 times/wk). The study was analyzed with the SAS 9.1 statistical package (SAS Institute, Cary, NC).
For the meta-analysis, we used a random effects model developed by DerSimonian and Laird (20). The individual study estimates were weighted by the inverse of their variance. We tested for heterogeneity between studies using the Q statistic (20). To test the null hypothesis that there is no difference between colon and rectal cancers, we use a contrast test (21). Pooled ORs and 95% CIs were calculated with the statistical software STATA 10.1 (StataCorp, College Station, TX). P<0.05 (two sided) was considered statistically significant.
Results
In the Physicians’ Health Study, plasma 25(OH)D levels were higher among men who took multivitamins, regularly exercised, and provided blood samples during summer/autumn. Plasma 1,25(OH)2D levels were higher among men who consumed moderate alcohol and provided blood samples during summer/autumn (data not shown). The median follow-up time from baseline to diagnosis of colorectal cancer was 8.9 years. We found no significant association between plasma 25(OH)D and overall colorectal cancer risk (Table 1). When we examined colon cancer and rectal cancer separately, a suggestive inverse association was observed for rectal cancer with a threshold effect (ORs for each subsequent quartile vs. 1st quartile of plasma 25(OH)D were 0.53, 0.42, 0.45; P for trend = 0.05), but not for colon cancer. We also observed a suggestive inverse trend for 1,25(OH)2D and risk of colorectal cancer but neither the point estimates or the P for trends were statistically significant for overall colorectal or site-specific cancers (Table 1).
Table 1.
Odds Ratio (OR) and 95% confidence intervals (CIs) of colorectal cancer according to plasma vitamin D metabolites in the Physicians’ Health Study
| Categories* | Colorectal cancer
|
Colon cancer
|
Rectal cancer
|
||||||
|---|---|---|---|---|---|---|---|---|---|
| Cases/controls (N) | Median | Simple matched OR† | Multivariate OR‡ | Cases/controls (N) | Multivariate OR‡ | Cases/controls (N) | Multivariate OR‡ | ||
| 25(OH)D (ng/mL) | |||||||||
| Quartile1 | 57/96 | 15.7 | 1.00 | 1.00 | 36/70 | 1.00 | 20/24 | 1.00 | |
| Quartile2 | 41/97 | 22.3 | 0.72 (0.43–1.20) | 0.71 (0.42–1.21) | 37/72 | 0.95 (0.52–1.74) | 15/26 | 0.53 (0.18–1.60) | |
| Quartile3 | 74/99 | 26.7 | 1.26 (0.78–2.02) | 1.24 (0.76–2.04) | 52/74 | 1.34 (0.75–2.39) | 9/28 | 0.42 (0.13–1.40) | |
| Quartile4 | 57/97 | 37.9 | 1.02 (0.62–1.69) | 1.08 (0.62–1.87) | 47/71 | 1.38 (0.73–2.64) | 13/24 | 0.45 (0.14–1.46) | |
| P for trend | 0.99 | 0.67 | 0.35 | 0.05 | |||||
| 1,25(OH)2D (pg/mL) | |||||||||
| Quartile1 | 66/93 | 25.5 | 1.00 | 1.00 | 49/68 | 1.00 | 20/24 | 1.00 | |
| Quartile2 | 60/96 | 31.2 | 0.87 (0.54–1.41) | 0.91 (0.55–1.50) | 40/71 | 0.83 (0.46–1.49) | 13/24 | 0.57 (0.20–1.60) | |
| Quartile3 | 53/96 | 34.7 | 0.79 (0.49–1.27) | 0.84 (0.51–1.38) | 47/71 | 0.96 (0.54–1.68) | 10/26 | 0.43 (0.13–1.39) | |
| Quartile4 | 45/94 | 41.1 | 0.66 (0.39–1.09) | 0.70 (0.41–1.18) | 33/71 | 0.64 (0.34–1.19) | 12/24 | 0.75 (0.27–2.09) | |
| P for trend | 0.14 | 0.24 | 0.22 | 0.72 | |||||
Among control participants, quartile cutoff points were determined by season (summer/autumn or winter/spring) and batches (two batches).
Matched on age (±1 year for younger participants, ±5 years for older participants) and smoking status (never, past, current).
Matched on age (±1 year for younger participants, ±5 years for older participants) and smoking status (never, past, current). Further adjusted for seasons (summer/autumn or winter/spring), fasting status (≤8, > 8 hours), race (Caucasian or non-Caucasian), vigorous exercise (<1, 1–4 times/wk, ≥5 times/wk), dairy calcium intake (≤146, 146<–340, 340<–580, ≥580 mg/d), and body mass index (<23, 23–<25, 25–<27, ≥27 kg/m2).
We conducted a meta-analysis of the associations between circulating 25(OH)D levels and colorectal cancer in eight prospective studies (Figure 1A). The pooled multivariate OR (95% CI) was 0.66 (0.54–0.81) for colorectal cancer risk comparing the highest quantile with the lowest quantile of circulating 25(OH)D levels. We specifically evaluated the associations separately for colon and rectal cancer in a meta-analysis of the previous studies plus the present data from the Physicians’ Health Study (Table 2). We found a stronger inverse association for rectal cancer than colon cancer, although a formal test for difference between colon and rectal cancers was not statistically significant (P for difference = 0.20). The pooled multivariate OR was 0.77 (95% CI = 0.56–1.07; P for heterogeneity between studies = 0.04; Figure 1B) for colon cancer and 0.50 (95% CI = 0.28–0.88; P for heterogeneity between studies = 0.04; Figure 1C) for rectal cancer, comparing the highest quantile with the lowest quantile of circulating 25(OH)D levels. When we pooled the estimates from the three published studies that examined 1,25(OH)2D levels (7, 14, 18) plus the Physicians’ Health Study, the pooled multivariate OR (95% CI) comparing the highest quantile with the lowest quantile of circulating 1,25(OH)2D levels was 1.01 (0.59–1.73) for colorectal cancer and 0.88 (0.57–1.35) for colon cancer.
Figure 1.
A. Study-specific and pooled ORs and 95% CIs of colorectal cancer comparing the highest with lowest quantiles of blood 25(OH)D levels. B. Study-specific and pooled ORs and 95% CIs of colon cancer comparing the highest with lowest quantiles of blood 25(OH)D levels. C. Study-specific and pooled ORs and 95% CIs of rectal cancer comparing the highest with lowest quantiles of blood 25(OH)D levels. The black squares indicate the study-specific odds ratios; the horizontal lines represent the 95% CIs. The area of the black squares reflects the study-specific weights (inverse of the variance). The dashed line represents the pooled OR and the diamond represents the 95% CI for the pooled OR. ATBC = Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study; CLUE = from the campaign slogan, “Give us a CLUE to cancer” (A = 1975–1983; B = 1984–1991); EPIC = European Prospective Investigation into Cancer and Nutrition; HPFS = Health Professionals Follow-up Study; JPHC = Japan Public Health Center Study; MEC = multiethnic cohort study; NHS = Nurses’ Health Study; PHS = Physicians’ Health study; WHI = Women’s Health Initiative; M = men; W= women
Table 2.
Included prospective studies of circulating levels of 25(OH)D and risk of colon and rectal cancers
| Study name* | First author, year (Ref no.) | Country (Sex)† | Age at blood donation in controls (years) | Study dates (Follow-up) | 25(OH)D, mean or median | Colon | Rectum | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||||
| 25(OH)D levels in the top vs. bottom quantiles | No. of cases/controls | OR (95% CI) in the quantiles ‡ | 25(OH)D levels in the top vs. bottom quantiles | No. of cases/controls | OR (95% CI) in the quantiles ‡ | ||||||
| CLUE A§ | Garland, 1989 (11) | US (M, W) | 74.7% in 45–74 year | 1975–1983 | 30.5 in cases and 33.3 ng/mL in controls, mean | 42 vs. 19 ng/mL, cutoff | 34/67 | Q1–Q5 | N/A | N/A | N/A |
| 1.0 (ref), 0.48, 0.25, 0.21, 0.73 (0.20–2.66) | |||||||||||
| CLUE B§ | Braun, 1995 (18) | US (M, W) | 55, median | 1984–1991 | 23.6 in cases and 23.2 ng/mL in controls, mean | 30.1 vs. 17.2 ng/mL, cutoff | 57/114 | Q1–Q5 | N/A | N/A | N/A |
| 1.0 (ref), 0.3, 0.5, 0.7, 0.4 (0.1–1.4) | |||||||||||
| P-trend = 0.57 | |||||||||||
| ATBC§ | Tangrea, 1997 (14) | Finland (M) | 60, median | 1985–1993 | 12.1 in cases and 13.8 ng/mL in controls, mean | 19.3 vs. 9.8 ng/mL, cutoff | 91/181 | Q1–Q4 | 19.2 vs. 9.8 ng/mL, cutoff | 55/109 | Q1–Q4 |
| 1.0 (ref), 0.6, 0.8, 0.8 (0.4–1.6) | 1.0 (ref), 0.9, 0.8, 0.4 (0.1–1.1) | ||||||||||
| P-trend = 0.69 | P-trend = 0.06 | ||||||||||
| NHS|| | Feskanich, 2004 (7)¶ | US (W) | 60.0, mean | 1989–2000 | 23.6 in cases and 24.3 ng/mL in controls in lab1, 27.0 in cases and 30.3 ng/mL in controls in lab2, mean | 28.7 vs. 18.9 ng/mL in lab1, 35.9 vs. 23.6 ng/mL in lab 2, cutoff | 149/295 | Q1–Q4 | 27.2 vs. 20.5 ng/mL in lab1, 33.5 vs. 26.1 ng/mL in lab 2, cutoff | 44/88 | Q1–Q3 |
| 1.0 (ref), 1.03, 0.54, 0.70 (0.35–1.38) | 1.0 (ref), 0.52, 0.31 (0.08–1.31) | ||||||||||
| P-trend = 0.17 | P-trend = 0.03 | ||||||||||
| HPFS|| | Wu, 2007 (8) | US (M) | 66.1, mean | 1993–2002 | 28.7 in cases and 29.4 ng/mL in controls, mean | 38.8 vs. 19.3 ng/mL, median | 139/276 | Q1–Q4 | 37.4 vs. 21.2 ng/mL, median | 40/80 | Q1–Q3 |
| 1.0 (ref), 0.74, 0.29, 0.46 (0.24–0.89) | 1.0 (ref), 1.74, 3.32 (0.87–12.69) | ||||||||||
| P-trend = 0.005 | P-trend = 0.08 | ||||||||||
| WHI§ | Wactawski-Wende, 2006 (10) **,†† | US (W) | 50–79, range | 1993–2005 | N/A | N/A | N/A | N/A | 23.4 vs. 12.4 ng/mL, cutoff | 68/68 | Q1–Q4 |
| 3.40 (1.13–10.21) ‡‡, 1.08, 3.41, 1.00 (ref) | |||||||||||
| P-trend = 0.22 | |||||||||||
| JPHS|| | Otani, 2007 (13) | Japan (M) | 56.9, mean | 1990–2003 | 27.3 in cases and 27.6 ng/mL in controls, median | 35.6 vs. 19.9 ng/mL, median | 119/237 | Q1–Q4 | 35.6 vs. 19.9 ng/mL, median | 44/87 | Q1–Q4 |
| 1.0 (ref), 0.98, 1.0, 1.2 (0.51–2.7) | 1.0 (ref), 0.17, 0.25, 0.075 (0.0057–0.99) | ||||||||||
| P-trend = 0.70 | P-trend = 0.06 | ||||||||||
| Japan (W) | 56.4, mean | 1990–2003 | 22.5 in cases and 22.3 ng/mL in controls, median | 31.4 vs. 16.6 ng/mL, median | 106/195 | Q1–Q4 | 31.4 vs. 16.6 ng/mL, median | 54/102 | Q1–Q4 | ||
| 1.0 (ref), 1.7, 2.1, 2.1 (0.78–5.6) | 1.0 (ref), 0.26, 0.46, 0.33 (0.084–1.3) | ||||||||||
| P-trend = 0.12 | P-trend = 0.17 | ||||||||||
| EPIC||,§ | Jenab, 2010 (9)†† | Europe (M, W) | 58.7 for colon, 58.0 for rectum, mean | 1992–2003 | 20.7 in cases and 22.9 ng/mL in controls for colon, 22.0 in cases and 22.0 ng/mL in controls for rectum, geometric mean | 39.7 vs. 13.2 ng/mL, median | 785/785 | Q1–Q5 | 41.7 vs. 12.8 ng/mL, median | 463/463 | Q1–Q5 |
| 1.0 (ref),. 0.77, 0.65, 0.66, 0.47 (0.33–0.68) | 1.0 (ref), 1.29, 1.06, 1.18, 0.89 (0.56–1.43) | ||||||||||
| P-trend<0.001 | P-trend=0.647 | ||||||||||
| MEC|| | Woolcott, 2010 (12)§§ | US (M,W) | 69.2, mean | 2001–2006 | 23.2 in cases and 25.0 ng/mL in controls, mean | 32.8 vs. 16.8 ng/mL, cutoff | 170/319 | Q1–Q5 | 32.8 vs. 16.8 ng/mL, cutoff | 43/83 | Q1–Q5 |
| 1.0 (ref), 0.82, 0.70, 0.73, 0.83 (0.42–1.63) | 1.0 (ref), 0.55, 0.34, 0.28, 0.28 (0.06–1.38) | ||||||||||
| P-trend = 0.17 | P-trend = 0.02 | ||||||||||
| PHS|| | Lee (present study) | US (M) | 56.6, mean | 1982–2000 | 26.6 in cases and 25.6 ng/mL in controls, mean | 36.8 vs. 14.9 ng/mL, median | 172/287 | Q1–Q4 | 39.2 vs. 17.9 ng/mL, median | 57/102 | Q1–Q4 |
| 1.0 (ref), 0.95, 1.34, 1.38 (0.73–2.64) | 1.0 (ref), 0.53, 0.42, 0.45 (0.14–1.46) | ||||||||||
| P-trend = 0.35 | P-trend = 0.05 | ||||||||||
ATBC = Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study; CLUE = from the campaign slogan, “Give us a CLUE to cancer”; EPIC = European Prospective Investigation into Cancer and Nutrition; HPFS = Health Professionals Follow-up Study; JPHC = Japan Public Health Center Study; MEC = Multiethnic Cohort Study; NHS = Nurses’ Health Study; PHS = Physicians’ Health study; WHI = Women’s Health Initiative
M = men; W= women
Q1–Q5 = quintiles; Q1–Q4 = quartiles; Q1–Q3 = tertiles
Serum specimen was used.
Plasma specimen was used.
Estimates of quantile cutoff were obtained from the investigator of the Nurses’ Health Study
Estimates for rectal cancer were obtained from the Publications and Presentations committee of the Women’s Health Initiative
The conversion factor for 25(OH)D from nmol/L to ng/mL is 0.401
For top vs. bottom quartiles, OR (95% CI) = 0.29 (0.098–0.89)
Estimates for colon and rectal cancers were obtained from the investigator of the Multiethnic Cohort Study
Discussion
In the Physicians’ Health Study, we found an inverse trend of circulating blood 25(OH)D levels with rectal cancer risk, which was in line with the results from some of the previous studies that reported a significant inverse association or trend for rectal cancer (7, 12–14). By combining results from the Physicians’ Health Study and the previous seven prospective studies, a meta-analysis of eight prospective studies support a stronger inverse association of circulating blood 25(OH)D levels with rectal cancer risk than with colon cancer risk. Statistically non-significant difference warrants more prospective studies of circulating vitamin D levels and colon and rectal cancers and additionally distal and proximal cancers.
Etiologic differences between colon and rectal cancer, such as differences in the associations with life style modifiers, prevalence of specific gene mutations, and expression levels of specific genes, are commonly described (22–24). In a clinical study that examined vitamin D deficiency among 221 colon cancer and 94 rectal cancer patients, severe vitamin D deficiency (defined as 15 ng/mL) was more common among rectal cancer patients than colon cancer patients (25). Several studies reported difference in associations for vitamin D receptor polymorphisms and calcium or vitamin D by tumor sites. The SS genotype of poly(A) mononucleotide repeat and BB genotype of BsmI were associated with a reduced risk of rectal cancer, not with colon cancer in the presence of low calcium and low energy (26). There is evidence that the associations for calcium and vitamin D were stronger for more distal and rectal tumors than proximal colon tumors (26–27). Also, in a study of molecular marker in rectal tumors, the associations of vitamin D intake and vitamin D receptor polymorphism with TP53 mutation status were present in rectal tumors (28). These studies further suggest a possible difference in vitamin D-related mechanisms in the pathogenesis of rectal cancer compared with colon cancer.
The lack of a significant inverse association in the Physicians’ Health Study between circulating 25(OH)D levels and colon cancer may be because the association of 25(OH)D is weaker for colon cancer or because most of our blood samples (>70 %) were collected during summer/autumn, which may not fully capture differences in levels throughout the year. One study that examined the associations across seasons found that an inverse association was limited to those whose blood samples were collected during winter (8). Also, we cannot rule out the possibility of laboratory measurement error or misclassification of one-time baseline measurement of 25(OH)D levels in the Physicians’ Health Study because of the possible changes in vitamin D levels influenced by lifestyle factor modification during the follow-up period or the influence of multivitamin intervention arm from the trial, which started in 1997 and is scheduled to end by 2012.
Only three prospective studies examined the association of circulating 1,25(OH)2D levels with risk of colorectal cancer or colon cancer (7, 14, 18), and our meta-analysis, including the Physicians’ Health Study, suggests no association. This may be because 1,25(OH)2D is more tightly regulated largely by renal 1-α-hydroxylase activity (29).
Vitamin D may reduce the risk of rectal cancer through regulation of progression and differentiation (1) and inhibition of angiogenesis (3). In animal studies, vitamin D (the vitamin D3 analogue; EB 1089) improved tumor control by radiation treatment, possibly by promoting apoptosis (4). However, because vitamin D status is largely dependent on sun exposure, there was limited evidence of inverse associations between total or dietary vitamin D intake and colorectal cancer (30). In contrast, serum or plasma concentrations of 25(OH)D are regarded as a good biomarker of vitamin D status integrating intake through foods, supplements, and exposure to ultraviolet light.
Each study had only a single measure of plasma vitamin D metabolite levels, which did not allow us to examine changes across time. However, a single measure of vitamin D metabolites has shown to be a useful marker to reflect long-term vitamin D status (31–33). We could not investigate the effects of changes in vitamin D, or in other variables during follow-up because only baseline measures were available. However, a significant trend for rectal cancer may suggest that misclassification might not be too serious to exclude the association we observed. Also, we cannot rule out the possible residual confounding because our study did not allow us to adjust for cumulative information on covariates across follow-up time. However, we found no appreciable change in the associations after adjusting for various measured risk factors, suggesting that residual confounding by risk factors may not explain the association observed in the Physicians’ Health Study. Heterogeneity across studies in a meta-analysis may be due to differences in laboratory measurement methods, follow-up periods, seasons of blood draw, exposure to sun light, ethnicity, and prevalence of other interaction factors and unknown/unmeasured confounding factors. Important strengths of our study and meta-analysis include the prospective design in which blood samples were collected before cancer diagnosis.
Taken together, these prospective results support the hypothesis that vitamin D is inversely related to colorectal cancer risk, and that vitamin D is more strongly associated with reduced risk of rectal cancer than colon cancer.
Acknowledgments
The authors thank the Publications and Presentations committee of the Women’s Health Initiative and Christy Woolcott in the Multiethnic Cohort Study for providing estimates for a meta-analysis. The authors also thank Haiyan Zhang for her assistance in data management and Diane Feskanich in the Nurses’ Health Study for providing estimates. This study was supported by Grant Numbers CA42182, CA34944, CA40360, CA 137178 and CA097193 from the National Cancer Institute, by Grant Numbers HL26490 and HL34595 from the National Heart, Lung, and Blood Institute and by Prevent Cancer Foundation fellowship grant (JEL). Dr. Chan is a Damon Runyon Clinical Investigator. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Heart, Lung, and Blood Institute. Dr. Hollis is a consultant for DiaSorin Corp., the maker of the 25(OH)D radioimmunoassay kits used in the study. The sponsors had no role in the study design, the data collection and analysis, interpretation of the results, the preparation of the manuscript, or the decision to submit the manuscript for publication.
References
- 1.Lamprecht SA, Lipkin M. Cellular mechanisms of calcium and vitamin D in the inhibition of colorectal carcinogenesis. Ann N Y Acad Sci. 2001;952:73–87. doi: 10.1111/j.1749-6632.2001.tb02729.x. [DOI] [PubMed] [Google Scholar]
- 2.Evans SR, Shchepotin EI, Young H, Rochon J, Uskokovic M, Shchepotin IB. 1,25-dihydroxyvitamin D3 synthetic analogs inhibit spontaneous metastases in a 1,2-dimethylhydrazine-induced colon carcinogenesis model. Int J Oncol. 2000;16:1249–54. doi: 10.3892/ijo.16.6.1249. [DOI] [PubMed] [Google Scholar]
- 3.Iseki K, Tatsuta M, Uehara H, Iishi H, Yano H, Sakai N, et al. Inhibition of angiogenesis as a mechanism for inhibition by 1alpha-hydroxyvitamin D3 and 1,25-dihydroxyvitamin D3 of colon carcinogenesis induced by azoxymethane in Wistar rats. Int J Cancer. 1999;81:730–3. doi: 10.1002/(sici)1097-0215(19990531)81:5<730::aid-ijc11>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- 4.Sundaram S, Sea A, Feldman S, Strawbridge R, Hoopes PJ, Demidenko E, et al. The combination of a potent vitamin D3 analog, EB 1089, with ionizing radiation reduces tumor growth and induces apoptosis of MCF-7 breast tumor xenografts in nude mice. Clin Cancer Res. 2003;9:2350–6. [PubMed] [Google Scholar]
- 5.Hollis BW, Kamerud JQ, Selvaag SR, Lorenz JD, Napoli JL. Determination of vitamin D status by radioimmunoassay with an 125I-labeled tracer. Clin Chem. 1993;39:529–33. [PubMed] [Google Scholar]
- 6.Giovannucci E. The epidemiology of vitamin D and colorectal cancer: recent findings. Curr Opin Gastroenterol. 2006;22:24–9. doi: 10.1097/01.mog.0000196150.36701.c2. [DOI] [PubMed] [Google Scholar]
- 7.Feskanich D, Ma J, Fuchs CS, Kirkner GJ, Hankinson SE, Hollis BW, et al. Plasma vitamin D metabolites and risk of colorectal cancer in women. Cancer Epidemiol Biomarkers Prev. 2004;13:1502–8. [PubMed] [Google Scholar]
- 8.Wu K, Feskanich D, Fuchs CS, Willett WC, Hollis BW, Giovannucci EL. A nested case control study of plasma 25-hydroxyvitamin D concentrations and risk of colorectal cancer. J Natl Cancer Inst. 2007;99:1120–9. doi: 10.1093/jnci/djm038. [DOI] [PubMed] [Google Scholar]
- 9.Jenab M, Bueno-de-Mesquita HB, Ferrari P, van Duijnhoven FJ, Norat T, Pischon T, et al. Association between pre-diagnostic circulating vitamin D concentration and risk of colorectal cancer in European populations:a nested case-control study. Bmj. 2010;340:b5500. doi: 10.1136/bmj.b5500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wactawski-Wende J, Kotchen JM, Anderson GL, Assaf AR, Brunner RL, O’Sullivan MJ, et al. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med. 2006;354:684–96. doi: 10.1056/NEJMoa055222. [DOI] [PubMed] [Google Scholar]
- 11.Garland CF, Comstock GW, Garland FC, Helsing KJ, Shaw EK, Gorham ED. Serum 25-hydroxyvitamin D and colon cancer: eight-year prospective study. Lancet. 1989;2:1176–8. doi: 10.1016/s0140-6736(89)91789-3. [DOI] [PubMed] [Google Scholar]
- 12.Woolcott CG, Wilkens LR, Nomura AM, Horst RL, Goodman MT, Murphy SP, et al. Plasma 25-hydroxyvitamin D levels and the risk of colorectal cancer: the multiethnic cohort study. Cancer Epidemiol Biomarkers Prev. 2010;19:130–4. doi: 10.1158/1055-9965.EPI-09-0475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Otani T, Iwasaki M, Sasazuki S, Inoue M, Tsugane S. Plasma vitamin D and risk of colorectal cancer: the Japan Public Health Center-Based Prospective Study. Br J Cancer. 2007;97:446–51. doi: 10.1038/sj.bjc.6603892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tangrea J, Helzlsouer K, Pietinen P, Taylor P, Hollis B, Virtamo J, et al. Serum levels of vitamin D metabolites and the subsequent risk of colon and rectal cancer in Finnish men. Cancer Causes Control. 1997;8:615–25. doi: 10.1023/a:1018450531136. [DOI] [PubMed] [Google Scholar]
- 15.Yin L, Grandi N, Raum E, Haug U, Arndt V, Brenner H. Meta-analysis: longitudinal studies of serum vitamin D and colorectal cancer risk. Aliment Pharmacol Ther. 2009;30:113–25. doi: 10.1111/j.1365-2036.2009.04022.x. [DOI] [PubMed] [Google Scholar]
- 16.Final report on the aspirin component of the ongoing Physicians’ Health Study. Steering Committee of the Physicians’ Health Study Research Group. N Engl J Med. 1989;321:129–35. doi: 10.1056/NEJM198907203210301. [DOI] [PubMed] [Google Scholar]
- 17.U.S. Department of Agriculture, Agriculture Research Service. USDA Nutrient Data Laboratory. USDA Nutrient Database for Standard Reference, Release 13. Washington, DC: Department of Agriculture; 1999. [Google Scholar]
- 18.Braun MM, Helzlsouer KJ, Hollis BW, Comstock GW. Colon cancer and serum vitamin D metabolite levels 10–17 years prior to diagnosis. Am J Epidemiol. 1995;142:608–11. doi: 10.1093/oxfordjournals.aje.a117682. [DOI] [PubMed] [Google Scholar]
- 19.Bliss CI. Periodic Regression in Biology and Climatology. New Haven: Conn. Agricultural Exp. Sta; 1958. [Google Scholar]
- 20.DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clin Trials. 1986;7:177–88. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 21.Anderson TW. Introduction to multivariate statistics. New York, NY: John Wiley Sons; 1984. [Google Scholar]
- 22.Iacopetta B. Are there two sides to colorectal cancer? Int J Cancer. 2002;101:403–8. doi: 10.1002/ijc.10635. [DOI] [PubMed] [Google Scholar]
- 23.Wei EK, Giovannucci E, Wu K, Rosner B, Fuchs CS, Willett WC, et al. Comparison of risk factors for colon and rectal cancer. Int J Cancer. 2004;108:433–42. doi: 10.1002/ijc.11540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dimberg J, Samuelsson A, Hugander A, Soderkvist P. Differential expression of cyclooxygenase 2 in human colorectal cancer. Gut. 1999;45:730–2. doi: 10.1136/gut.45.5.730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fakih MG, Trump DL, Johnson CS, Tian L, Muindi J, Sunga AY. Chemotherapy is linked to severe vitamin D deficiency in patients with colorectal cancer. Int J Colorectal Dis. 2009;24:219–24. doi: 10.1007/s00384-008-0593-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Slattery ML, Neuhausen SL, Hoffman M, Caan B, Curtin K, Ma KN, et al. Dietary calcium, vitamin D, VDR genotypes and colorectal cancer. Int J Cancer. 2004;111:750–6. doi: 10.1002/ijc.20330. [DOI] [PubMed] [Google Scholar]
- 27.Huncharek M, Muscat J, Kupelnick B. Colorectal cancer risk and dietary intake of calcium, vitamin D, and dairy products: a meta-analysis of 26,335 cases from 60 observational studies. Nutr Cancer. 2009;61:47–69. doi: 10.1080/01635580802395733. [DOI] [PubMed] [Google Scholar]
- 28.Slattery ML, Wolff RK, Herrick JS, Caan BJ, Samowitz W. Calcium, vitamin D, VDR genotypes, and epigenetic and genetic changes in rectal tumors. Nutr Cancer. 2010;62:436–42. doi: 10.1080/01635580903441204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fraser DR. Biochemical and clinical aspects of vitamin D function. Br Med Bull. 1981;37:37–42. doi: 10.1093/oxfordjournals.bmb.a071673. [DOI] [PubMed] [Google Scholar]
- 30.IARC. Vitamin D and cancer. Lyon: International Agency for Research on Cancer; 2008. Nov 25, [Google Scholar]
- 31.Hofmann JN, Yu K, Horst RL, Hayes RB, Purdue MP. Long-term variation in serum 25-hydroxyvitamin D concentration among participants in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Cancer Epidemiol Biomarkers Prev. 2010;19:927–31. doi: 10.1158/1055-9965.EPI-09-1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Platz EA, Leitzmann MF, Hollis BW, Willett WC, Giovannucci E. Plasma 1,25-dihydroxy- and 25-hydroxyvitamin D and subsequent risk of prostate cancer. Cancer Causes Control. 2004;15:255–65. doi: 10.1023/B:CACO.0000024245.24880.8a. [DOI] [PubMed] [Google Scholar]
- 33.Kotsopoulos J, Tworoger SS, Campos H, Chung FL, Clevenger CV, Franke AA, et al. Reproducibility of plasma and urine biomarkers among premenopausal and postmenopausal women from the Nurses’ Health Studies. Cancer Epidemiol Biomarkers Prev. 2010;19:938–46. doi: 10.1158/1055-9965.EPI-09-1318. [DOI] [PMC free article] [PubMed] [Google Scholar]