Abstract
LIN28A and LIN28B, the mammalian homologs of lin-28, are implicated in malignant transformation in part because of their ability to promote degradation of the let-7 family of miRs. In the present study, we show that overexpression of Lin28b in vivo leads to an aggressive peripheral T-cell lymphoma (PTCL) characterized by widespread infiltration of parenchymal organs with malignant CD4+ cells. Similar to patients with PTCL, Lin28b-transgenic mice show signs of inflammation such as eosinophilia, increased C-reactive protein, release of inflammatory cytokines, and pleural effusion. The PTCLs that develop in Lin28b mice are derived from activated T cells and show decreased let-7 expression, increased Il6 expression, activation of NF-κB, and infiltration of B cells, all resulting in an inflammatory microenvironment. In addition, LIN28B is overexpressed 7.5-fold in PTCL patient samples compared with activated CD4+ cells. The results of the present study demonstrate for the first time that Lin28b can transform primary cells in vivo, identify a previously unsuspected link between Lin28b and PTCL, and provide a unique animal model for the study of PTCL biology and therapy.
Introduction
Lin28b is an RNA-binding protein that shares 69% amino acid sequence identity with Lin28a (formerly known as Lin28).1 The discovery of the Lin28a and Lin28b orthologs followed the identification of lin-28 as a heterochronic gene that regulates the development of Caenorhabditis elegans.2 Mutations in lin-28 prompt premature larval stage development.3 In mammals, Lin28a and Lin28b mRNA was noted to be enriched in embryonic stem cells, and, in conjunction with NANOG, OCT4, and SOX2, LIN28A has been used to transform human fibroblasts to induced pluripotent stem (iPS) cells.4 LIN28A replaced MYC as one of the original Yamanaka factors (MYC, NANOG, OCT4, and SOX2) that were used to generate iPS cells in part because of the concern that constitutive expression of MYC, a known protooncogene, may lead to malignant transformation of iPS cells.4 However, there is no animal model available with which to study the potential for Lin28a or Lin28b to transform primary cells in vivo.
Given that Lin28a and Lin28b expression is associated with undifferentiated cell states, a correlation with malignant transformation was suspected. Indeed, overexpression of LIN28A or LIN28B has been reported in approximately 15% of human malignancies encompassing a broad spectrum of tumor types5 and LIN28B expression is correlated with advanced tumors such as high-grade colon and ovarian cancers.6 In addition, LIN28B expression was shown to be increased dramatically (more than 1000-fold) in blast crisis chronic myeloid leukemia (CML) compared with chronic-phase CML.5
Overexpression of both LIN28A and LIN28B is associated with decreased levels of the let-7 family of miRs, and a role for LIN28 proteins has been demonstrated in miR biogenesis. By binding to the terminal loop of pre–let-7 miRs, LIN28A and LIN28B facilitate the recruitment of terminal uridylyl transferase 4 (TUT4), leading to the addition of uracil residues that results in inhibition of Dicer processing and decreased levels of mature let-7 miRs.7 Let-7 family members regulate the expression of genes such as MYC, HMGA2, and KRAS, further implicating LIN28B in the process of malignant transformation.8,9
Several studies have linked the expression of LIN28A or LIN28B and malignant transformation in vitro, suggesting that LIN28A and LIN28B might function as dominant-acting protooncogenes if expressed ectopically. For example, induced expression of LIN28B in MCF-7 breast cancer cells increased cell proliferation, whereas knockdown of LIN28B expression led to differentiation and growth inhibition of K562 CML cells1,5 and immortalized cell lines that express LIN28A or LIN28B proliferate in immunodeficient mice.5,10,11 However, expression of Lin28a in a transgenic mouse model did not result in malignant transformation, but instead led to developmental abnormalities including increased body and organ size and delayed puberty.12 In the present study, we tested the hypothesis that Lin28b is oncogenic in vivo by generating a mouse that constitutively expresses Lin28b in hematopoietic cells. We report that transgenic mice overexpressing Lin28b show aberrant T-cell development and that these mice develop a peripheral T-cell lymphoma (PTCL) that develops in the background of an inflammatory tumor microenvironment. Our results provide direct evidence that Lin28b is oncogenic in vivo, link the transforming ability of Lin28b to inflammation, and suggest that the pathway involving Lin28b and inflammation is a target for therapeutic intervention.
Methods
Transgenic mice
The murine Lin28b cDNA was inserted into the HS 21/45-vav vector containing 3′ and 5′ Vav-regulatory elements13 for targeted expression in hematopoietic tissues (Figure 1A), as described previously.14 The purified Vav-Lin28b construct was microinjected into zygotes from C57BL/6 mice. Mouse neoplasms were evaluated using classification systems from the Bethesda proposal for lymphoid neoplasms in mice15 and the 2008 World Health Organization classification for lymphoma in humans.16 All animal studies were approved by the National Cancer Institute Intramural Animal Care and Use Committee.
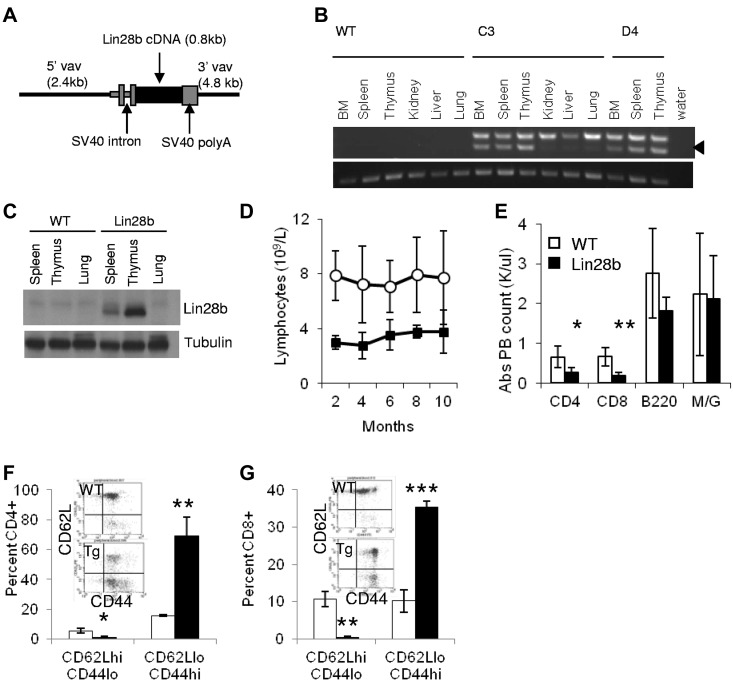
Figure 1.
Expression of Lin28b leads to lymphopenia in transgenic mice. (A) Schematic of Vav expression vector used to generate transgenic mice. (B) Expression of the Lin28b transgene. Top panel: spliced transgenic Lin28b mRNA (arrowhead); the upper band seen in all transgenic tissues is due to amplification of unspliced, contaminating genomic DNA. Bottom panel: Actb used as a loading control. (C) Western blot for Lin28b protein in hematopoietic tissues of Lin28b mice. (D) Serial lymphocyte counts in the peripheral blood of WT and Lin28b-transgenic mice (n = 11). P < .01 for each time point. (E-F) Leukocyte subsets in peripheral blood (E; n = 5) and spleen (F; n = 3). M/G indicates myeloid cells that stain for the antigens Mac1 and Gr-1. (G-H) Percent of naive (CD62L+CD44−) and effector memory (CD62L−CD44+) CD4 (G) and CD8 (H) cells in peripheral blood from WT or Lin28b mice (n = 3). *P < .05; **P < .01; ***P < .001. WT is indicated by open circles or white bars; Lin28b, black squares or bars.
Flow cytometry, immunohistochemistry, and immunoblots
Antibodies from BD Pharmingen and eBiosciences were used to stain cell-surface markers. Single-cell suspensions of 1 × 106 cells were prepared in HBSS with 2% FBS and incubated with antibodies conjugated to FITC, PE, or APC for 30 minutes on ice, washed with PBS, resuspended in 1 μg/mL of propidium iodide to assess viability, and analyzed on an FACScan (BD Pharmingen). Formalin-fixed, paraffin-embedded sections were stained with H&E, CD3 (MCA1477; AbD Serotec), B220 (553086; BD Pharmingen), CD31 (sc-1506; Santa Cruz Biotechnology), CD138 (281-2; BD Pharmingen), and NF-κB p65 phospho S276 (ab30623; Abcam). Stained slides were scanned in their entirety at 20× magnification using a ScanScope XT digital slide scanner (Aperio Technologies). Whole-slide image data files were generated at 0.5 μm/pixel resolution and stored in the Spectrum Image Management System for analysis with ImageScope Version 10.0 software (Aperio Technologies). Immunoblotting was performed per standard protocol with primary antibodies to the C-terminal region of Lin28b (AP1485b; Abgent), Syk (sc-1077; Santa Cruz Biotechnology), and Tubulin (clone 11H10; Cell Signaling Technology). For bromodeoxyuridine (BrdU) analysis, mice were injected IP with 1 mg of BrdU using the BrdU Flow kit (BD Pharmingen) and euthanized after 16 hours. 1 × 106 thymocytes were stained for CD4, CD8, CD44, CD25 and BrdU (anti–BrdU-APC) per the BrdU Flow kit manual (BD Pharmingen).
ELISA
Serum IgG was assessed with the mouse IgG ELISA Kit from Kamiya Biomedical Company (KT-402). Fifty microliters of serum was diluted 1:25 000-1:50 000 and analyzed per the manufacturer's instructions. C-reactive protein was detected using a CRP ELISA Test Kit (Life Diagnostics). Serum was diluted 1:100, followed by serial dilution of 1:2. Plates were read at 450 nm with a VICTOR X4 Multilabel Plate Reader (PerkinElmer).
NF-κB activation assay
Nuclear protein extracts were prepared using the Nuclear Extraction Kit protocol (SK-0001; Signosis) and the protein concentration was determined with the Bio-Rad DC Protein assay. Using the TransAM NF-κB p65 Transcription Factor ELISA (40096; Active Motif), nuclear extracts (5 μg) were assessed for binding to an NF-κB consensus site immobilized as an oligonucleotide (4 pmol/well) in a 96-well format with or without competitor (NF-κB consensus site oligonucleotide added in solution at 20 pmol/well).17
Gene-expression assays
Expression of the Lin28b transgene was confirmed with RT-PCR (for primers see supplemental Table 4, available on the Blood Web site; see the Supplemental Materials link at the top of the online article). Real-time PCR was performed with 1 μg of RNA isolated using TRIzol reagent (Invitrogen) and reverse transcribed with SuperScript III (Invitrogen). Expression of Myc (Mm00487804_m1), Il6 (Mm00446191_m1), Bcl6 (Mm00477633_m1), Icos (Mm00497600_m1), Il21 (Mm00517640_m1), Cxcl13 (Mm00444534_m1), Ccl7 (Mm00443113_m1), Ccnd2 (Mm00438070_m1), Hmga2 (Mm04183367_g1), and Kras (Mm00517492_m1) was measured using TaqMan primer-probe sets and the 7500 Fast Real-time PCR System (Applied Biosystems). Samples were normalized to 18S rRNA endogenous control (4308329) and calibrated to wild-type (WT) CD4+ splenocytes unless otherwise indicated. The splenocytes were isolated by positive selection with CD4 MicroBeads and LS columns (Miltenyi Biotec). TaqMan miRNA assays (Applied Biosystems) were used to assess let-7a (377), let-7f (382), and let-7g (2282) expression. cDNA was transcribed from 10 ng of RNA using the TaqMan MicroRNA Reverse Transcription Kit (Applied Biosystems). Sno202 (1232) or U6 snRNA (1973) was used as an endogenous control. Inflammatory Response and Autoimmunity PCR arrays (SA Biosciences) were used to examine cytokine and chemokine gene expression. The assay was performed with 1 μg of RNA that was transcribed with the RT2 First Strand Kit, and the data were analyzed with RT2 Profiler PCR Array Data Analysis software.
RNA isolation and small RNA library preparation for deep sequencing
Total RNA was isolated using the mirVana miRNA Isolation Kit (Ambion). The small RNA fraction (18-30 nt size range) was enriched from 10 μg of total RNA using 15% denaturing tris-borate-EDTA urea polyacrylamide gels. Purified small RNAs were ligated to proprietary 3′ and 5′ RNA adapters (Illumina) and used as templates for cDNA synthesis. cDNA amplification was performed using adapter-specific primers (12 PCR cycles), purified using tris-borate-EDTA-PAGE, and diluted to 8pM for cluster generation and sequencing on an Illumina GAII. Sequence tags were aligned against mature miR sequences from miRBase (download date: 05/20/2009) using standalone Blat Version 34 and default parameters with the exception of tileSize = 8. If a tag matched multiple targets, the tag was assigned to 1 randomly selected alternative match.
Tcrb gene rearrangements and candidate gene resequencing
Genomic DNA from spleen or lymph node samples was digested with HindIII or SstI and size fractionated by agarose gel electrophoresis. DNA was transferred to a nitrocellulose membrane and hybridized to a 32P-labeled probe that detected the constant region of the Tcrb gene.18 Notch1, Bcl11b, Idh1/2, Fbxw7, and Pten were examined using primers listed in supplemental Table 4 or as described previously.19 A cDNA template was used for Pten and genomic DNA was used for the others.
Transplantation assays
Recipient C57BL/6 mice bearing the CD45 allele Ly5.1 or Ly5.2 were lethally irradiated (1000 rad) and transplanted with 1 × 106 donor BM nucleated cells bearing Ly5.2 or Ly5.1, respectively. Engraftment of donor cells was determined by flow cytometric analyses on whole blood obtained from the tail vein as described previously.20 To assess tumorigenicity, 1 × 107 cells from lymph node or thymus of Lin28b mouse no. 6266 or WT thymocytes were injected subcutaneously in 200 μL of sterile PBS. Tumor growth and animal health were monitored daily.
Statistical analysis
The Student t test was used to determine statistical significance unless stated otherwise.
Results
Expression of a Lin28b transgene leads to lymphopenia
We previously reported that the expression of a NUP98-HOXD13 (NHD13) transgene in the hematopoietic compartment leads to impaired myeloid and lymphoid differentiation, which is typically followed by leukemic transformation, most commonly transformation to acute myeloid leukemia or pre-T-lymphoblastic leukemia/lymphoma (pre–T-LBL).2122 A gene-expression array identified Lin28b as one of the most highly overexpressed genes in the bone marrow and thymus of NHD13 mice (Slape C and Aplan PD, unpublished data). To determine whether Lin28b overexpression contributes to malignant transformation in vivo, we used Vav regulatory elements to target expression of Lin28b to the hematopoietic compartment (Figure 1A).13 The long isoform of Lin28b was used for these experiments because it is more similar to Lin28a than is the short isoform, which lacks the conserved N-terminal cold-shock domain.1 We obtained 11 potential founders, and bred 7 of these to WT mice. Of the 4 potential founders that produced progeny, founder lines C3, D4, and D10 transmitted the transgene; we chose to focus our studies on lines C3 and D4.
The Lin28b transgene was expressed in the BM, spleen, and thymus of mice from these 2 transgenic lines (Figure 1B-C). To distinguish mRNA expression from small amounts of contaminating genomic DNA, we used a forward primer that annealed to the 5′ SV40 region and a reverse primer that annealed to exon 2 of the Lin28b cDNA (Figure 1A). Expression of the transgene resulted in a splicing event that removed the SV40 intron to produce a 321-bp product (Figure 1B arrow). Lin28b sequences derived from either unspliced mRNA or contaminating genomic DNA were present in all transgenic tissues assayed (upper band, 418 bp). The transgenic Lin28b was expressed in the BM, spleen, and thymus; very faint bands in other tissues could represent small amounts of blood cells within these organs. In agreement with the mRNA expression results, the transgenic protein was also expressed in hematopoietic tissues (Figure 1C).
Peripheral blood complete blood counts were monitored every 2 months for a cohort of Lin28b-transgenic mice and littermate controls. The WBC count was decreased by nearly 2-fold in Lin28b-transgenic mice compared with age-matched WT control mice due to a 2.6-fold decrease in circulating lymphocytes (Table 1), whereas the absolute numbers of granulocytes, monocytes, RBCs, and platelets were similar. The lymphopenia persisted for the entire study and resulted from a decrease of both CD4 and CD8 single-positive (SP) T cells (Figure 1D-E). Surprisingly, there was a marked (3- to 4-fold) increase in the percentage of both CD4 and CD8 effector memory T cells (CD62L−CD44+) compared with WT controls (Figure 1F-G).
Table 1.
Transgenic Lin28b mice exhibit peripheral blood lymphopenia
| WBCs, 109/L | Lymphocytes, 109/L | Neutrophils, 109/L | Hemoglobin, g/dL | Platelets, 109/L | |
|---|---|---|---|---|---|
| WT | 10.70 ± 3.68 | 7.23 ± 2.79 | 2.84 ± 1.07 | 13.95 ± 0.64 | 910.27 ± 90.82 |
| Lin28b | 5.53 ± 1.59* | 2.78 ± 0.96* | 2.22 ± 0.63 | 13.64 ± 1.56 | 807.55 ± 222.24 |
n = 11 for each genotype. Error reported as SD.
P < .001.
Consistent with the similar numbers of myeloid and erythroid cells in the Lin28b and WT mice, there was no difference in the number of BM colony-forming units between the Lin28b and WT mice or in the replating potential of these colonies (supplemental Figure 1A-B). There was no difference in the proportion of B-cell precursors or common lymphoid progenitors in the BM of Lin28b mice compared with WT controls (supplemental Figure 1C-D).
To determine whether the lymphopenic phenotype was cell autonomous, BM from WT or Lin28b mice (which express Ly5.2) was transplanted into lethally irradiated WT recipient mice (which express Ly5.1). As shown in supplemental Figure 2, there was no difference in the absolute number of Mac1+/Gr1+ circulating myeloid cells between recipients of WT BM or Lin28b BM. Similar to findings with the Lin28b-transgenic mice, there was a trend toward decreased numbers of B220+ cells in the Lin28b recipients. However, there was a clear and consistent decrease in circulating CD4+ and CD8+ T cells, again consistent with findings from the Lin28b mice, indicating that the lymphopenia that develops in Lin28b mice was cell autonomous and not dependent on host-derived stromal cells.
Lin28b mice thymocytes fail to develop normally
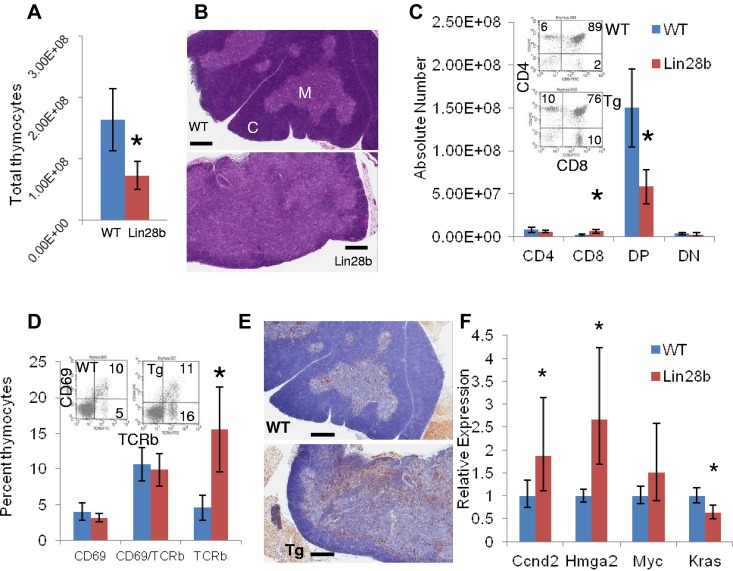
To determine the reason for the decreased number of circulating T cells, we compared Lin28b and WT thymi. Thymi from Lin28b mice had significantly fewer total thymocytes than did the WT control mice (7.27 × 107 ± 2.32 × 107 vs 16.3 × 107 ± 5.01 × 107, respectively) than did the WT control mice (Figure 2A). In addition, thymi from Lin28b mice had a narrower cortex and a more expansive medulla than those from WT mice, which correlated morphologically with a less distinct corticomedullary junction (Figure 2B). Furthermore, the number of CD8 SP cells was increased in the Lin28b thymi and the number of CD4+CD8+ (DP) cells was decreased (Figure 2C). To determine whether these were mature T cells, we assayed expression of CD69 and Tcrb, which demonstrated an increased proportion of mature T cells (CD69−TCRb+) in the transgenic thymi, suggesting that the increased number of CD8 SP cells might be due to a failure of the SP thymocytes to emigrate from the thymus (Figure 2D). In addition, there was no decrease in apoptosis of the CD4 or CD8 SP cells (data not shown) and in vitro survival of Lin28b thymocytes did not differ from WT thymocytes, indicating that increased survival of the SP cells was not a likely cause for the increased proportion of SP cells in the thymus (supplemental Figure 3A). Finally, we used BrdU incorporation to determine whether there was an increased proliferation of SP cells within the thymus. Although there was no increase in BrdU incorporation in the CD8 SP cells, there was a clear increase in CD4+ proliferation that may reflect the presence of activated, proliferative T cells in the Lin28b thymus (supplemental Figure 3B). Interestingly, there was an increased number of B220+ B cells infiltrating the Lin28b thymus (Figure 2E), again consistent with the presence of activated mature T cells.
Figure 2.
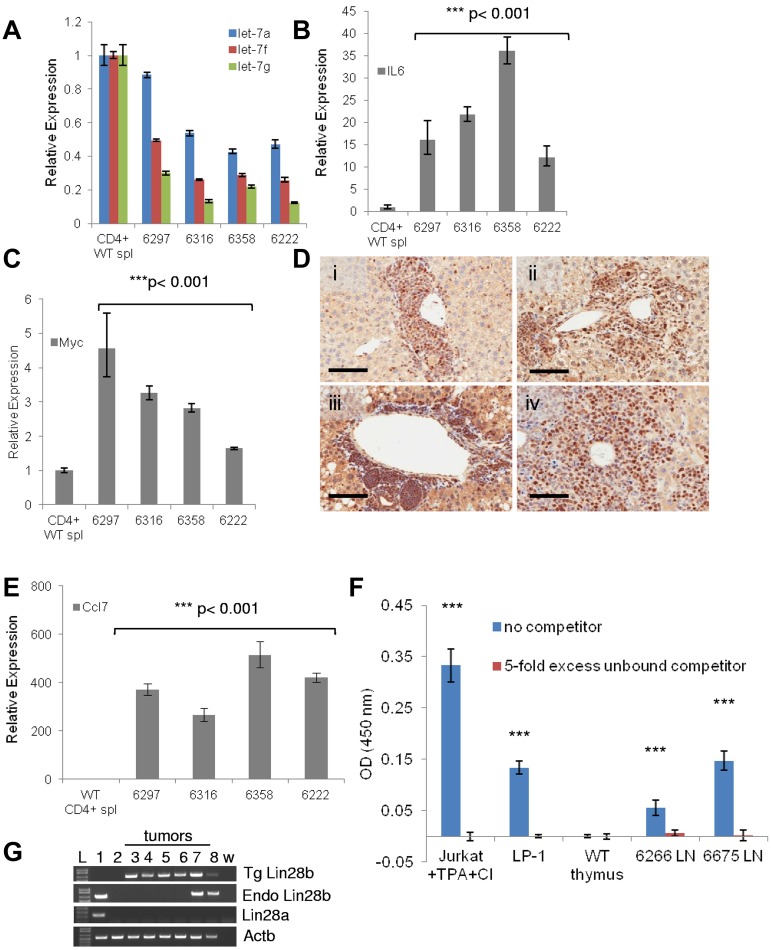
Developmental abnormalities in the Lin28b thymus. (A) Total thymocyte number in WT (blue) or Lin28b (red) thymi (n = 4). (B) H&E staining of WT or Lin28b whole thymus. M indicates medulla; and C, cortex. Scale bar indicates 400 μm. (C) Absolute numbers from the thymus with representative FACS plots showing percentages (inset; n = 4). (D) Retention of mature CD69−/Tcrb+ cells in the thymus (n = 3). Inset shows representative FACS. (E) B220 staining in the medulla of the thymus. Scale bar indicates 400 μm. *P < .05; ***P < .001. WT is indicated by blue; Lin28b, red. (F) Let-7 target gene expression in the thymi from mice 1-4 months of age (n = 6 per genotype). Samples were compared with the WT samples set at 1. *P < .05.
Given that Lin28b is known to regulate let-7 biogenesis, miRs from Lin28b and WT thymi were compared using deep sequencing. In this unbiased survey, 4 of the top 5 and 5 of the top 10 most differentially down-regulated miRs were let-7 family members, including let-7d, let-7g, let-7f, let-7i, and miR-98 (Table 2). These let-7 miRs represented some of the most abundant miRs in the thymus, together representing 9.1% of all miRs in the WT thymus, but only 2% of miRs in the Lin28b thymus. In addition to the let-7 family, miR-19a, miR-19b, miR-155, and miR-142-3p were also down-regulated in the Lin28b thymus. The most abundant, overexpressed miRs in the Lin28b thymus were miR-378, miR-99b, and miR-652. Similar to findings with the whole thymus, let-7 family members (let-7b, let-7d, let-7f, let-7g, let-7i, and miR-98) were down-regulated 3- to 4-fold in immature, double-negative thymocytes (supplemental Table 1). In addition, miR-181a, miR-181b, miR-181d, miR-20b, miR-128, and miR-106a were among the miRs down-regulated at least 2-fold. Among the abundantly overexpressed miRs were miR-342-3p and miR-150.
Table 2.
miR sequencing of the Lin28b thymus
| Whole thymus miRNA | Lin28b/WT ratio | Counts in WT thymus | miRNA | Lin28b/WT ratio | Counts in WT thymus |
|---|---|---|---|---|---|
| mmu-miR-137 | 0.148 | 25 | mmu-miR-298 | 7.96 | 39 |
| mmu-let-7g | 0.181 | 126 426 | mmu-miR-331-3p | 7.24 | 11 |
| mmu-miR-98 | 0.200 | 2060 | mmu-miR-139-3p | 7.19 | 195 |
| mmu-let-7i | 0.211 | 67 034 | mmu-miR-669c | 7.02 | 62 |
| mmu-let-7f | 0.219 | 399 526 | mmu-miR-382 | 6.81 | 20 |
| mmu-miR-19a | 0.268 | 2258 | mmu-miR-434-5p | 6.38 | 26 |
| mmu-miR-196b | 0.307 | 531 | mmu-miR-139-5p | 5.92 | 649 |
| mmu-miR-19b | 0.321 | 8974 | mmu-miR-129-5p | 5.31 | 37 |
| mmu-miR-7b | 0.328 | 528 | mmu-miR-485 | 5.25 | 9 |
| mmu-miR-292-3p | 0.341 | 38 | mmu-miR-700 | 5.20 | 13 |
| mmu-miR-190 | 0.343 | 73 | mmu-miR-542-5p | 5.01 | 17 |
| mmu-let-7d | 0.347 | 23 568 | mmu-miR-23b | 4.58 | 669 |
| mmu-miR-32 | 0.348 | 886 | mmu-miR-23a | 4.49 | 624 |
| mmu-miR-190b | 0.364 | 79 | mmu-miR-378 | 4.18 | 167 202 |
| mmu-miR-142-3p | 0.374 | 263 424 | mmu-miR-342-5p | 4.04 | 646 |
| mmu-miR-216a | 0.394 | 40 | mmu-miR-652 | 4.02 | 2595 |
| mmu-miR-211 | 0.403 | 108 | mmu-miR-99b | 4.01 | 6987 |
| mmu-miR-155 | 0.405 | 10 880 | mmu-miR-339-3p | 3.96 | 76 |
| mmu-miR-340-5p | 0.411 | 2971 | mmu-miR-296-3p | 3.92 | 140 |
| mmu-miR-335-5p | 0.415 | 29 | mmu-miR-151-3p | 3.58 | 1114 |
| mmu-miR-7a | 0.423 | 14 007 | mmu-miR-210 | 3.45 | 236 |
| mmu-miR-20a | 0.424 | 91 362 | mmu-miR-193b | 3.39 | 41 |
| mmu-miR-29b | 0.428 | 2938 | mmu-miR-582-3p | 3.31 | 7 |
| mmu-miR-217 | 0.432 | 75 | mmu-miR-541 | 3.26 | 503 |
| mmu-miR-350 | 0.441 | 254 | mmu-miR-214 | 3.16 | 29 |
Counts refer to numbers of individual sequence tags for the specified miR. miRs with at least 25 counts in 1 sample and a 2-fold or greater change are reported. Ratios were calculated using the percentage of totals and counts are from the WT thymus.
Downstream targets of let-7 (Ccnd2, Hmga2, Myc, and Kras) were assessed in thymi from WT and Lin28b mice and Ccnd2 and Hmga2 were increased. As shown in Figure 2F, the standard error indicates greater variability in expression of these genes among the Lin28b samples compared with the WT samples; this variation may be because of differing degrees of abnormal T-cell development among the Lin28b mice.
Lin28b mice develop a disease that resembles human PTCL
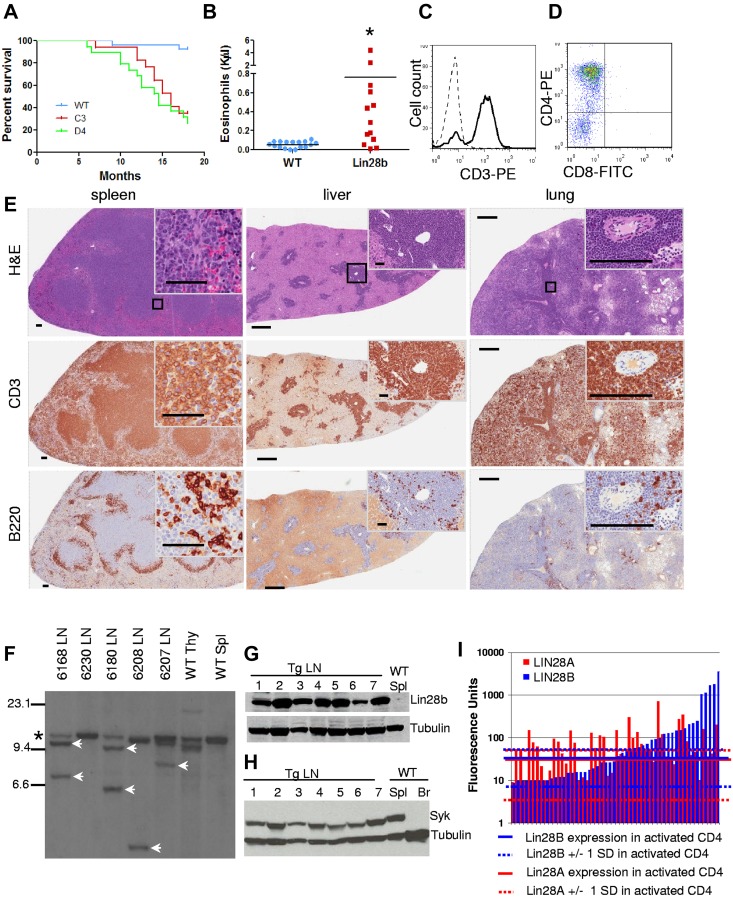
A cohort of Lin28b-transgenic mice and littermate controls from 2 different founder lines, C3 and D4, were observed for an 18-month study period. Survival curves demonstrated significantly decreased survival of both Lin28b-transgenic lines (Figure 3A). At approximately 10 months, Lin28b mice developed signs of disease such as hunched posture, ruffled fur, lethargy, and lymphadenopathy. Necropsy typically revealed lymphadenopathy, splenomegaly, and grossly infiltrated parenchymal organs and, less commonly, ascites or pleural effusions (Table 3). In contrast to the massively enlarged thymi characteristic of murine pre–T-LBL, Lin28b mice typically had a normal-sized thymus. Complete blood counts showed a variable degree of anemia, neutrophilia, and marked eosinophilia (Figure 3B and Table 3). Flow cytometry revealed a CD3+CD4+ population (Figure 3C-D), and the spleen, liver, and lungs were infiltrated with sheets of CD3+ T cells interspersed with B220+ B cells (Figure 3E). In addition, Southern blots demonstrated clonal Tcrb gene rearrangements (Figure 3F). These results indicate that Lin28b-transgenic mice develop an aggressive, fatal, T-cell malignancy.
Figure 3.
Lin28b-transgenic mice develop T-cell tumors. (A) Survival of Lin28b mice from C3 (n = 17) and D4 (n = 19) founders compared with WT littermate controls (n = 27). Median survival was 14.5 and 16 months for the D4 and C3 lines, respectively, P < .0001 compared with WT controls by log-rank test. (B) Eosinophil counts from PTCL mice with a mean age of 12 months compared with WT mice, also mean age 12 months. *P < .05. (C-D) CD3 staining of tumor cells (C; dashed line, isotype control; solid line, CD3) and CD4 positivity of tumor cells from mouse 6180 (D). (E) Immunohistochemistry of tissues from mouse 6180. Scale bars for the spleen indicate 500 μm (low magnification) and 50 μm (high magnification inset). Scale bars for the liver and lung indicate 500 μm (low magnification) and 200 μm (high magnification inset). Black square indicates area enlarged for inset. (F) Southern blot for Tcrb gene rearrangements. SstI-restricted genomic DNA, hybridized to a TCRb probe *Germline band; white arrows, nongermline-rearranged bands. (G) Immunoblot for Lin28b expression. Lanes 1-7, Lin28b tumor no. 6266, 6222, 6226, 6297, 6316, 6358, and 6549 with WT spleen as the negative control. (H) Immunoblot for Syk expression. Lanes 1 through 7, Lin28b tumors 6266, 6222, 6226, 6297, 6316, 6358, and 6569; lane 8, WT spleen (positive control); lane 9, WT brain (negative control). (I) LIN28A and LIN28B expression in patients with PTCL34 compared with mean values of activated CD4+ (aCD4) T cells (n = 4). Solid blue (LIN28B) and solid red (LIN28A) lines represent means for activated CD4+ T cells; dashed lines represents 1 SD from these means. Each bar represents 1 PTCL patient; data are presented on a logarithmic scale.
Table 3.
Lymphoid malignancy in Lin28b mice
| ID | Diagnosis | Sex | Founder | Age, mo | Gross findings | CBC WBC | NE | LY | PLT | Hb | IHC Pathology | CD3 | B220 | FACS CD3 | B220 | CD4 SP | CD8 SP | Tcrb Rgmt |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C3 | PTCL | M | C3 | 5 | L, LN, S | 21.40 | 16.38 | 3.97 | 177 | 14.0 | LN Lung |
+++ +++ |
+ + |
38 | 10 | 45 | 1 | + |
| 6179 | PTCL | F | C3 | 7 | S | 68.17 | 39.70 | 5.80 | 789 | 15.5 | LN | ND | ND | 45 | 3 | ND | ND | + |
| Lung | +++ | + | ||||||||||||||||
| 6180 | PTCL | F | C3 | 12 | H, L, LN, S | 5.52 | 2.36 | 2.68 | 587 | 13.4 | LN | +++ | ++ | 47 | 5 | 81 | 0.06 | + |
| Lung | +++ | + | ||||||||||||||||
| 6222 | PTCL | F | C3 | 14 | H, Pl. ef., LN, S | 6.40 | 4.88 | 0.92 | 605 | 11.7 | LN | +++ | ++ | 69 | 4 | 68 | 1 | + |
| Lung | ++ | + | ||||||||||||||||
| 6226 | PTCL | F | C3 | 17 | H, L, LN, S | 14.06 | 9.71 | 3.06 | 382 | 12.6 | Thymus/LN | +++ | ++ | 61 | 21 | 78 | 1 | + |
| Lung | ++ | ++ | ||||||||||||||||
| 6250 | PTCL | M | C3 | 18 | H, L, LN | 12.14 | 6.41 | 5.21 | 982 | 12.5 | Lung | ++ | + | 30 | 15 | 18 | 14 | - |
| 6252 | PTCL | M | C3 | 18 | LN, S | ND | Lung | ++ | ND | 51 | 11 | 14 | 41 | + | ||||
| LN | +++ | ND | ||||||||||||||||
| 6253 | PTCL | M | C3 | 14 | LN, S | 4.74 | 3.34 | 0.96 | 742 | 12.8 | LN | +++ | +++ | 35 | 57 | 49 | 2 | + |
| Lung | ++ | ++ | ||||||||||||||||
| 6292 | PTCL | F | C3 | 13 | LN | 6.32 | 2.10 | 3.94 | 886 | 14.5 | LN | + | ++ | 23 | 23 | 30 | 10 | - |
| Lung | ++ | ++ | ||||||||||||||||
| 6145 | PTCL | M | D4 | 18.5 | H, LN, S | 2.64 | 2.03 | 0.36 | 429 | 6.1 | Lung | ++ | ++ | 65 | 6 | 58 | 2 | + |
| LN | +++ | + | ||||||||||||||||
| 6206 | PTCL | M | D4 | 14.5 | LN, S | 1.56 | 0.85 | 0.46 | 787 | 8.3 | LN | +++ | + | 72 | 5 | 73 | 2 | + |
| Lung | +++ | + | ||||||||||||||||
| 6207 | PTCL | M | D4 | 10 | H | 4.36 | 2.89 | 1.19 | 919 | 8.8 | Lung | ++ | ++ | 27 | 6 | 28 | 0.25 | + |
| 6208 | PTCL | M | D4 | 10 | H, LN, S | 21.72 | 14.24 | 2.42 | 400 | 7.4 | LN | +++ | ++ | 86 | 2 | 91 | 1 | + |
| Lung | +++ | ++ | ||||||||||||||||
| 6214 | PTCL | F | D4 | 14.5 | L, LN, S | 29.32 | 11.85 | 11.31 | 258 | 11.2 | LN | +++ | ++ | 52 | 1 | 33 | 1 | + |
| Lung | +++ | ++ | ||||||||||||||||
| 6216 | PTCL | M | D4 | 16 | H, LN, S | 10.16 | 6.51 | 2.70 | 270 | 10.2 | LN | + | + | 5 | 28 | 82 | 1 | - |
| Lung | ++ | ++ | ||||||||||||||||
| 6219 | PTCL | F | D4 | 12.5 | Asc., H, LN, S | 19.7 | 7.78 | 9.44 | 520 | 7.5 | LN | +++ | + | 75 | 1 | 77 | 0.05 | + |
| Lung | ++ | ++ | ||||||||||||||||
| 6220 | PTCL | F | D4 | 12.5 | LN, S | 40.36 | 18.77 | 11.87 | 512 | 12.5 | Lung | +++ | ++ | 20 | 24 | 71 | 1 | + |
| 6230 | PTCL | F | D4 | L, LN, S | 8.66 | 6.38 | 1.82 | 88 | 7.9 | LN | ++ | + | 59 | 3 | 53 | 1 | + | |
| Lung | ++ | + | ||||||||||||||||
| 6235 | PTCL | M | D4 | 12 | H, LN, S | 18.18 | 7.10 | 9.33 | 316 | 11.3 | Lung | +++ | ++ | 62 | 10 | 37 | 7 | + |
| 6239 | Pre-T LBL | F | D4 | 18 | L, LN, S, T | 213.18 | 38.94 | 138.75 | 596 | 13.8 | LN | +++ | ND | 38 | 1 | 1 | 93 | + |
| Lung | ++ | ND | ||||||||||||||||
| 6178 | Pancytopenia | F | C3 | 12 | LN, S | 1.08 | 0.17 | 0.79 | 162 | 2.4 | LN | ND | ND | 19 | 37 | 23 | 8 | ND |
| Liver | - | - | ||||||||||||||||
| 6168* | PTCL | F | D10 | 10 | L, LN, S | 7.14 | 5.84 | 0.69 | 671 | 13.8 | LN | +++ | ++ | 90 | 6 | 88 | 0.23 | + |
| Lung | ++ | ++ | ||||||||||||||||
| 6266* | PTCL | F | D10 | 12 | L, LN, S | 10.90 | 5.20 | 4.79 | 514 | 11.9 | LN | +++ | + | 86 | 0.46 | 96 | 0.21 | + |
| Lung | +++ | ++ | ||||||||||||||||
| 6275*† | PTCL | F | D4 | 12 | Pl. ef. | 1.44 | 0.43 | 0.75 | 573 | 10.6 | Lung | +++ | ++ | 28 | 3 | 35 | 5 | ND |
| 6297* | PTCL | M | D10 | 13 | L, LN | 43.12 | 36.72 | 1.07 | 746 | 12.8 | LN | +++ | ++ | 87 | 5 | 60 | 1 | + |
| Lung | +++ | ++ | ||||||||||||||||
| 6316* | PTCL | F | C3 | 12 | LN, S | 8.20 | 3.69 | 2.92 | 410 | 12.1 | LN | +++ | + | 56 | 6 | 35 | 11 | + |
| Lung | ++ | ++ | ||||||||||||||||
| 6358* | PTCL | M | D4 | 12.5 | H, LN, S | 13.88 | 4.04 | 8.38 | 669 | 10.1 | LN | - | +++ | 76 | 9 | 43 | 1 | + |
| Lung | ++ | ++ | ||||||||||||||||
| 6348* | Pre-T LBL | M | C3 | 7 | T | ND | Thymus | +++ | + | 41 | 1 | 42 | 1 | + | ||||
| Spleen | ++ | +++ | ||||||||||||||||
| 6412* | PTCL | M | C3 | 12 | Asc., LN, H, S | ND | LN | +++ | ++ | ND | + | |||||||
| Lung | ++ | + | ||||||||||||||||
| 6502* | PTCL | F | D4 | 14 | H, LN, Pl. ef., S | ND | ND | 64 | 2 | 97 | 0.11 | ND |
Asc indicates ascites; Pl. ef., pleural effusion; H, hepatomegaly/invaded liver; L, invaded lung; LN, lymphadenopathy/lymph node; T, thymoma; and S, splenomegaly.
These mice were not part of the initial cohort but were used for other assays. Five mice (6143, 6148, 6182, 6211, and 6215) were found dead and are not included in this table. Three mice (6238, 6261, and 6284) were hunched and lethargic with extramedullary hematopoiesis in the spleen but without evidence of lymphoma.
The FACS data for this mouse is from BM.
All of the tumors evaluated expressed the Lin28b protein (Figure 3G), and cells from a Lin28b tumor injected into Scid mice formed subcutaneous tumors, providing further evidence of malignancy (supplemental Figure 4A-C). Similar to the donor cells, the tumors in the recipient mice were CD3+, invaded parenchymal organs (supplemental Figure 4C), and demonstrated clonal Tcrb gene rearrangements identical to that of the donor mouse (supplemental Figure 4D).
Because murine19,23,24 and human25 pre–T-LBL are associated with Notch1 mutations, we examined the Notch1 proline, glutamic acid, serine, threonine-rich (PEST) and the heterodimerization (HD) domains in 17 Lin28b T-cell tumors for evidence of mutation. In addition, we searched for evidence of the recently described 5′ interstitial Notch1 deletion mutation that leads to ligand-independent activation of Notch1.26 The only Notch1 mutation (supplemental Figure 5 and supplemental Table 2) was from mouse no. 6348, 1 of 2 atypical mice with a large thymus and pre–T-LBL (Table 3).
We screened additional genes associated with T-cell malignancy for mutation, including Bcl11b, Fbwx7, Pten, and Idh1/2. V(D)J-mediated Bcl11b deletion mutations are frequently observed in murine pre–T-LBL (Sakata et al27; and M.O., unpublished results) and Idh2 mutations were recently reported in patients with a subset of PTCL.28 Mutations in these genes were not detected in the Lin28b mice evaluated (supplemental Table 2).
Most of the Lin28b lymphomas did not fit any of the diagnostic categories in the Bethesda proposals for classification of lymphoid neoplasms in mice15; therefore, we used the World Health Organization classification.16 The majority of the Lin28b lymphomas had clinical, morphological, and immunophenotypical features consistent with PTCL. Lin28b tumors expressed Syk (Figure 3H), which is consistent with a report that almost all patients with PTCL aberrantly express Syk protein.29 PTCL can be further subdivided into anaplastic large cell lymphoma (ALCL), angioimmunoblastic T-cell lymphoma (AITL), and PTCL, not otherwise specified (PTCL-NOS).16,30,31 ALCL is characterized by large cells with eccentric nuclei that are CD25+ and CD30+16,31; the Lin28b tumors did not show these features (Figure 3E and Table 4). Although the Lin28b PTCL had some clinical features consistent with AITL, such as ascites, pleural effusions, anemia (Table 3), eosinophilia (Figure 3B), and hypergammaglobulinemia (3 of 4 mice evaluated had serum IgG levels between 1.42 and 1.82 mg/mL, which was > 1 SD above their WT littermates at 0.91 ± 0.38 mg/mL), they did not display the prominent high endothelial venules that are a diagnostic feature of AITL.16,32,33 Interestingly, analysis of a publicly available dataset (http://www.ncbi.nlm.nih.gov/geo)34 demonstrated LIN28B (7.5-fold; P = .0012) overexpression in PTCL-NOS patients compared with activated CD4+ cells (Figure 3I). In sum, 42% (21 of 50) of PTCL-NOS and 27% (10 of 37) of AITL patients expressed LIN28B mRNA levels greater than 1 SD above the mean of control CD4+ cells Therefore, Lin28b mice developed a disease that resembled PTCL-NOS, and LIN28B is overexpressed in a large number of PTCL-NOS patients.
Table 4.
Expanded immunophenotype of lymph nodes from Lin28b mice with PTCL
| Mouse | CD3 | CD4 | CD8 | Tcrb | CD4+CD25+ | CD4+PD1+ | CD4+Cxcr5+ | CD4+CD44−CD62L+ | CD4+CD44+CD62L− |
|---|---|---|---|---|---|---|---|---|---|
| 239 | + | + | − | + | − | + | − | − | + |
| 6297 | + | + | − | + | − | + | − | − | + |
| 6409 | − | + | − | + | ND | + | − | − | + |
| 6502 | + | + | − | + | − | + | − | − | + |
| 6549 | + | + | − | + | − | + | − | − | + |
Any sample with greater than 20% positivity was considered positive.
ND indicates not determined.
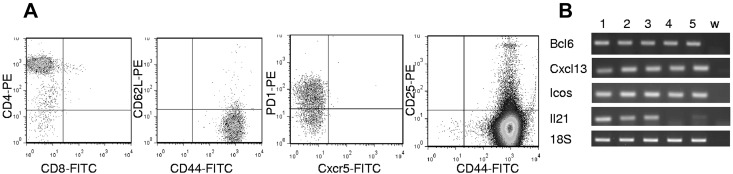
A recent effort to subclassify PTCL is based on cell of origin,31,34 using a combination of cytokine expression, immunophenotype, and histology. We determined that the malignant T cells from Lin28b mice are typically CD3+CD4+CD8−Tcrb+CD25−CD62L−CD44+PD1+CXCR5− (Figure 4A and Tables 3 and 4). The immunophenotype of the Lin28b PTCLs closely matched that of T-follicular helper cells (TFH) cells, because they were CD4+CD62L−CD44+CD25−PD1+. The Lin28b PTCLs had additional TFH cell features, including expression of Bcl6, Cxcl13, Icos, and Il21, all markers for TFH cells (Figure 4B). Consistent with the expression of Cxcl13 (a B-cell chemoattractant),35 many of the samples showed patchy infiltration with either B220+ B cells or CD138+ plasma cells (supplemental Figure 6).
Figure 4.
Malignant cells from Lin28b lymph nodes resemble TFH cells. (A) FACS plots describing tumor cell phenotype from mouse 6502. Analyses for CD44/CD62L, CXCR5/PD1, and CD44/CD25 were gated on the CD4+ population. (B) Expression of Bcl6, Cxcl13, Icos, and Il21 in Lin28b tumors. Lanes 1 through 6, WT CD4+ splenocytes, 6502 LN, 6222 LN, 6208 LN, 6297 LN, and w (water).
Lin28b inflammatory pathway functions in vivo
A positive feedback loop involving LIN28B, let-7, IL6, and NF-κB that links inflammation and cancer was identified recently using a mammary epithelial cell line.3610 In this pathway, Lin28b expression leads to decreased expression of let-7 family members, which leads to increased IL6 expression and subsequent NF-κB activation. Activated NF-κB leads to transcription of Lin28b, resulting in an autoregulatory loop. We assayed the genes and miRs involved in this loop. Let-7f and let-7g levels were 2- to 8-fold lower in the Lin28b PTCL samples (Figure 5A). Consistent with the miR deep-sequencing results (Table 2), let-7a levels were less markedly decreased. Il6 was markedly overexpressed in the PTCL samples, as was Myc, another known target for let-7 family members (Figure 5B-C). The PTCL samples showed strong nuclear staining for NF-κB p65 (Figure 5D), indicating activation of the NF-κB pathway. We assayed expression of Ccl7, a known NF-κB target,37 and, as shown in Figure 5E, Ccl7 levels were increased 200- to 600-fold, consistent with NF-κB activation. NF-κB activation was also confirmed in the Lin28b tumors with a binding assay (Figure 5F).17,38 Although endogenous Lin28b was expressed in 2 of 6 tumors tested, Lin28a was not expressed (Figure 5G), suggesting that its expression is not correlated with NF-κB activation in this model. Furthermore, several proinflammatory chemokines, cytokines, and complement, such as Ccl11, Ccl20, Ccl8, Cxcr5, Cxcl1, Il7, and C4b, which are involved in the recruitment of immune cells, were increased in the Lin28b PTCL samples (supplemental Table 3), further supporting the presence of an inflammatory tumor microenvironment. Finally, C-reactive protein, a serum marker for inflammation, was elevated in 3 of 5 clinically healthy Lin28b mice that showed no signs of illness (supplemental Figure 7), suggesting that systemic inflammation may precede the onset of PTCL.
Figure 5.
A Lin28b-regulated inflammatory pathway in vivo. (A) Expression of let-7a, let-7f, and let-7g (determined by TaqMan assays) in tumors from mice with PTCL compared with CD4+ WT splenocytes. Values shown are means ± SEM. All P values for the tumor samples were < .001 except 6297 for let-7a, which was P < .01. (B-C) Comparison as in panel A for Il6 (B) and Myc (C) mRNA. Values shown are means ± SEM. (D) Immunohistochemistry demonstrates nuclear NF-κB (p65) staining in the infiltrated livers of mice with PTCL: 6168 (i), 6226 (ii), 6297 (iii), and 6358 (iv). Scale bar indicates 100 μm. (E) Expression of the NF-κB target gene Ccl7 in PTCL tumors. Values shown are means ± SEM. For all RNA-expression experiments, the data were normalized to WT CD4+ splenocytes, which is set at 1. (F) Measurement of activated NF-κB in nuclear extracts from Lin28b tumors. Jurkat and LP-138 nuclear extracts were used as positive controls. The optical density 450 nm reflects binding of NF-κB p65 to the immobilized oligonucleotide (blue bars); the red bars reflect competition by a 5-fold excess of nonimmobilized oligonucleotide. TPA indicates 12-O-tetradecanoylphorbol 13-acetate; CI, calcium ionophore; and LN, lymph node. ***P < .001 compared with WT thymus. (G) Expression levels of Lin28a and Lin28b in tumors from transgenic Lin28b mice with PTCL. L-DNA ladder: mouse embryonic stem cell line (1); WT CD4+ splenocytes (2); 6222 (3); 6297 (4); 6316 (5); 6358 (6); 6502 (7); 6266 (8). w indicates water control; Tg, transgenic Lin28b; and Endo, endogenous Lin28b.
Transplantation of WT BM corrects the lymphopenia in Lin28b mice
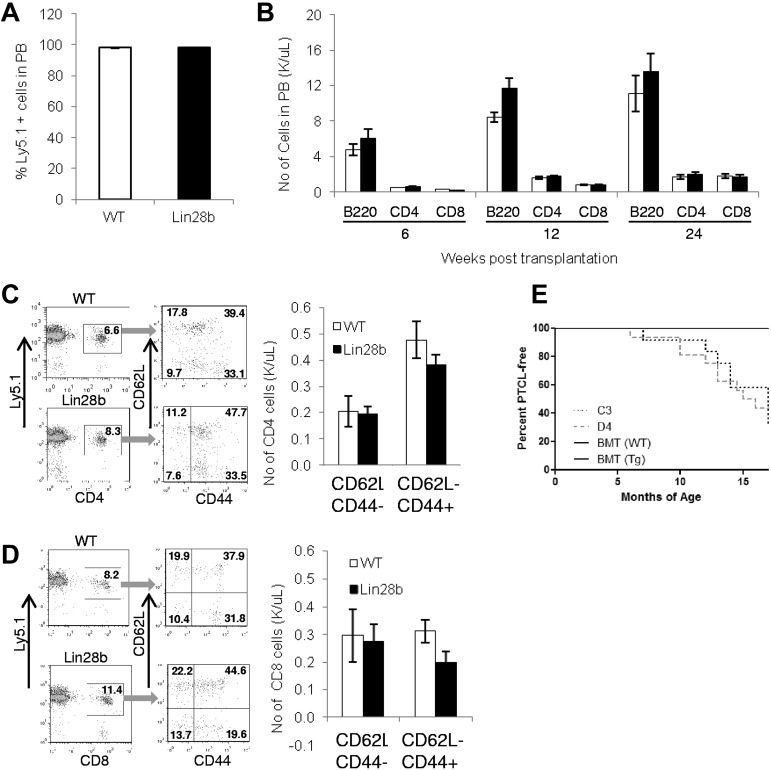
To determine whether the lymphopenia seen in Lin28b mice could be corrected, Lin28b and WT control mice (both of which express the Ly5.2 allele of Ly5) were lethally irradiated, ablating the myeloid and lymphoid compartments, and transplanted with WT BM (which expresses the Ly5.1 allele of Ly5). Figure 6A demonstrates that both WT and Lin28b recipients have successfully engrafted, with the majority of cells in the peripheral blood being of donor (Ly5.1) origin. There was no difference in absolute numbers of CD4, CD8, or B220 positive cells in the peripheral blood of the Lin28b mice compared with the WT controls, and this effect persisted for at least 24 weeks (Figure 6B). As opposed to nontransplanted Lin28b mice, which show a marked skewing of peripheral T cells away from naive cells and toward memory cells (Figure 1G-H), the Lin28b mice transplanted with WT BM showed no difference in the proportions of naive and memory T cells compared with transplanted control mice (Figure 6C-D). These Lin28b transplant recipients are now 18 months old, and none has yet developed PTCL, suggesting that replacement of the Lin28b-derived hematopoietic system with WT hematopoietic cells prevents the development of PTCL (Figure 6E).
Figure 6.
Transplantation with WT BM rescues lymphopenia. (A) Donor WT cells (Ly5.1) were transplanted into either WT or Lin28b mice and an engraftment assay was performed at 24 weeks after transplantation (n = 6 Lin28b recipients, n = 5 WT recipients). (B) Lymphocyte engraftment after transplantation at the indicated time points. White bars indicate WT; and black bars, Lin28b. (C) Quantification of naive (CD62L+CD44−), central memory (CD62L+CD44+), and effector memory (CD62L−CD44+) CD4+ cells in the peripheral circulation. (D) Quantification of naive, central memory, and effector memory CD8+ cells. White bars indicate WT; and black bars, Lin28b. (E) Percentage of WT (n = 3) or Lin28b (n = 3) recipients free of PTCL after transplantation of WT BM cells (C3, n = 12; D4, n = 16). Mice that died of causes other than PTCL were not included in this analysis (WT, n = 2; Lin28b, n = 3; C3, n = 5; D4, n = 3).
Discussion
Overexpression of LIN28A and LIN28B has been linked to several human malignancies, particularly those of germ cell, colon, or hematopoietic origin.5,6 Given that LIN28A and LIN28B regulate multiple miRs, which in turn regulate multiple mRNAs, these proteins are located at a nodal point for global regulation of genetic programs. Because of these links to human malignancy, we expressed Lin28b in the hematopoietic compartment of mice. Lin28b mice developed a range of clonal T-cell malignancies, most commonly a malignancy that closely resembles human PTCL-NOS. This observation not only supports the previously published associations between Lin28b expression and malignancy, but also provides direct evidence that constitutive expression of Lin28b can be oncogenic in vivo.
PTCLs represent a poorly understood group of mature T-cell malignancies that fall into 3 principal subcategories. Two of these diagnostic categories are AITL and ALCL; these diseases are fairly uniform in their clinical, morphologic, and immunophenotypic features.39 However, the most common subtype of PTCL is PTCL-NOS,39 a category that represents a heterogeneous group of mature, nodal, T-cell malignancies that do not share the features of AITL or ALCL. Patients with PTCL-NOS typically show peripheral lymphadenopathy and widespread dissemination of malignant T cells, with infiltration of the liver, spleen, and BM at diagnosis.16 The Lin28b-transgenic mice in the present study had a similar clinical presentation, with lethargy, diffuse lymphadenopathy, and widespread organ infiltration. Although the lymphomas that developed in the Lin28b-transgenic mice have some features in common with AITL, the defining characteristics of AITL are a polymorphous mixed infiltrate of malignant T cells and reactive inflammatory cells associated with a prominent proliferation of HEVs,16 neither of which was present in the PTCL that developed in the Lin28b mice.
Although the cell of origin for most PTCL-NOS patients has not been determined, several recent studies have identified a subset of PTCL-NOS patients who express TFH markers such as PD1, CXCL13, ICOS, and BCL6, but not CXCR5, and suggested that these tumors form a new subclass of PTCL-NOS.40,41 Similar to these patients, the PTCL that developed in Lin28b mice in the present study seemed to originate from TFH cells. The Lin28b PTCLs expressed a set of genes associated with TFH cells, including Bcl6, Icos, Cxcl13, Pdcd1, and Cxcr5. Although Lin28b PTCLs were Cxcr5−, and TFH cells are typically Cxcr5+,42,43 Cxcr5 is not absolutely required for TFH function, because Cxcr5−/− mice had only a 2-fold decrease in germinal center B cells, and similar levels of IgG compared with WT controls.44 Therefore, despite the lack of Cxcr5 expression, the Lin28b PTCL have many features consistent with TFH cells, making TFH cells the likely cell of origin for the Lin28b tumors. Interestingly, analysis of publicly available gene expression data demonstrated that PTCL-NOS patients show a marked elevation of LIN28B expression (7.5-fold). Our present findings indicate that overexpression of Lin28b leads to an aggressive, clonal, lethal T-cell malignancy that resembles human PTCL-NOS.
Before the onset of PTCL, clinically healthy Lin28b mice showed evidence for aberrant T-cell development, with a T-cell lymphopenia and increased proportions of mature T cells in the thymus. Although the precise cause for these T-cell abnormalities and how they relate to development of PTCL remain unknown, the theme of aberrant hematopoietic development preceding malignant transformation is well recognized.18 An increase in direct targets of let-7, including Ccnd2 and Hmga2, was also detected in the thymus.45 Although we did not observe a significant change in Myc, this may be explained by an in vivo homeostatic mechanism that compensates for decreases in let-7. Perhaps an in vitro study of isolated, purified, defined cell populations may yield significant increases in Myc. Although the lymphopenic phenotype is seen in virtually all Lin28b mice, the penetrance of the PTCL is incomplete, suggesting that Lin28b expression is not sufficient for the development of PTCL and that additional, complementary mutations are required for malignant transformation.
An epidemiologic link between inflammation and cancer, particularly many of the common epithelial cancers, such as those of the lung and gastrointestinal tract, is well recognized.46,47 It has been proposed that chronic inflammation leads to mutations through the action of reactive oxygen species typically associated with the inflammatory response.48 Recently, a feedback loop involving LIN28B, let-7 family members, IL6, and NF-κB that provides a link between inflammation and cancer has been elucidated. Let-7 family members were decreased in both the thymi and PTCL from Lin28b mice. In addition, Il6 and Myc were overexpressed in the Lin28b PTCL, NF-κB was activated, and overexpression of numerous proinflammatory cytokines was noted, providing evidence that overexpression of Lin28b in T cells leads to a proinflammatory environment in vivo. NF-κB activation has been reported previously as a common finding in patients with PTCL-NOS49; the observation that LIN28B is overexpressed in a high percentage of PTCL-NOS patients provides a possible mechanism for this activation.
PTCL-NOS is an aggressive disease with a poor prognosis; the 5-year survival is only 20%-30%. Given that there are no well-characterized PTCL-NOS cell lines available, it seems likely that the Lin28b in vivo model presented herein will be useful for preclinical studies of new therapeutic approaches. Allogeneic hematopoietic stem cell transplantation has been shown recently to be a promising therapeutic approach for some patients with PTCL-NOS.50 None of the Lin28b mice transplanted with WT BM in the present study have developed PTCL, although this was only a small group of 6 mice.
In the present study, we used expression of Lin28b in mature CD4+ T cells to induce an aggressive, clonal T-cell malignancy in mice that closely resembles human PTCL-NOS in terms of clinical presentation, morphology, immunophenotype, and gene expression. The PTCL that develops in Lin28b mice develops in a proinflammatory background and is linked to the activity of a previously described inflammatory pathway that links LIN28B overexpression with NF-κB activation. LIN28B (and LIN28A) are overexpressed in a subset of PTCL patients, suggesting that these proteins are attractive, previously unsuspected targets for directed therapy of this disease.
Supplementary Material
Acknowledgments
The authors thank Jerry Adams for the gift of the Vav plasmid; Jan Linkenhoker and Maria Jorge for excellent veterinary care and animal husbandry; Yulia Demchenko for providing technical insight and a positive control (LP-1) sample for the NF-κB assay; Ashish Lal, Dave Caudell, Mark Simpson, Michael Kuehl, Kevin Gardner, and members of the Aplan laboratory for helpful discussions; and Emily Leibovitch and Shelley Hoover for technical assistance.
This research was supported by the Intramural Research Program of the National Cancer Institute, National Institutes of Health (Bethesda, MD).
Footnotes
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: S.H.B. designed and performed the research, analyzed the data, and wrote the manuscript; M.O., Y.J.C., and C.S. designed and performed the research and analyzed the data; S.B. and P.M. analyzed the data; P.F., M.P., and R.L.W. designed and performed the research; and P.D.A. designed and analyzed the data and wrote the final draft of manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Peter D. Aplan, Genetics Branch, Center for Cancer Research, National Cancer Institute, National Institutes of Health, 41 Center Dr, Bethesda, MD 20892; e-mail: aplanp@mail.nih.gov.
References
- 1.Guo Y, Chen Y, Ito H, et al. Identification and characterization of lin-28 homolog B (LIN28B) in human hepatocellular carcinoma. Gene. 2006;384:51–61. doi: 10.1016/j.gene.2006.07.011. [DOI] [PubMed] [Google Scholar]
- 2.Moss EG, Lee RC, Ambros V. The cold shock domain protein LIN-28 controls developmental timing in C. elegans and is regulated by the lin-4 RNA. Cell. 1997;88(5):637–646. doi: 10.1016/s0092-8674(00)81906-6. [DOI] [PubMed] [Google Scholar]
- 3.Ambros V, Horvitz HR. Heterochronic mutants of the nematode Caenorhabditis elegans. Science. 1984;226(4673):409–416. doi: 10.1126/science.6494891. [DOI] [PubMed] [Google Scholar]
- 4.Yu J, Vodyanik MA, Smuga-Otto K, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318(5858):1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- 5.Viswanathan SR, Powers JT, Einhorn W, et al. Lin28 promotes transformation and is associated with advanced human malignancies. Nat Genet. 2009;41(7):843–848. doi: 10.1038/ng.392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.King C, Cuatrecasas M, Castells A, Sepulveda A, Lee JS, Rustgi AK. Lin28b promotes colon cancer progression and metastasis. Cancer Res. 2011;71(12):4260–4268. doi: 10.1158/0008-5472.CAN-10-4637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Heo I, Joo C, Kim YK, et al. TUT4 in concert with Lin28 suppresses microRNA biogenesis through pre-microRNA uridylation. Cell. 2009;138(4):696–708. doi: 10.1016/j.cell.2009.08.002. [DOI] [PubMed] [Google Scholar]
- 8.Chang TC, Zeitels LR, Hwang HW, et al. Lin-28B transactivation is necessary for Myc-mediated let-7 repression and proliferation. Proc Natl Acad Sci U S A. 2009;106(9):3384–3389. doi: 10.1073/pnas.0808300106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Roush S, Slack FJ. The let-7 family of microRNAs. Trends Cell Biol. 2008;18(10):505–516. doi: 10.1016/j.tcb.2008.07.007. [DOI] [PubMed] [Google Scholar]
- 10.Iliopoulos D, Hirsch HA, Struhl K. An epigenetic switch involving NF-kappaB, Lin28, Let-7 MicroRNA, and IL6 links inflammation to cell transformation. Cell. 2009;139(4):693–706. doi: 10.1016/j.cell.2009.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang YC, Chen YL, Yuan RH, et al. Lin-28B expression promotes transformation and invasion in human hepatocellular carcinoma. Carcinogenesis. 2010;31(9):1516–1522. doi: 10.1093/carcin/bgq107. [DOI] [PubMed] [Google Scholar]
- 12.Zhu H, Shah S, Shyh-Chang N, et al. Lin28a transgenic mice manifest size and puberty phenotypes identified in human genetic association studies. Nat Genet. 2010;42(7):626–630. doi: 10.1038/ng.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ogilvy S, Metcalf D, Gibson L, Bath ML, Harris AW, Adams JM. Promoter elements of vav drive transgene expression in vivo throughout the hematopoietic compartment. Blood. 1999;94(6):1855–1863. [PubMed] [Google Scholar]
- 14.Caudell D, Zhang Z, Chung YJ, Aplan PD. Expression of a CALM-AF10 fusion gene leads to Hoxa cluster overexpression and acute leukemia in transgenic mice. Cancer Res. 2007;67(17):8022–8031. doi: 10.1158/0008-5472.CAN-06-3749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Morse HC, Anver MR, 3rd, Fredrickson TN, et al. Bethesda proposals for classification of lymphoid neoplasms in mice. Blood. 2002;100(1):246–258. doi: 10.1182/blood.v100.1.246. [DOI] [PubMed] [Google Scholar]
- 16.Swerdlow S, Campo E, Harris NL, editors. International Agency for Research on Cancer. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissue. Geneva, Switzerland: World Health Organization; 2008. [Google Scholar]
- 17.Huang YH, Sojka DK, Fowell DJ. Cutting edge: regulatory T cells selectively attenuate, not terminate, T cell signaling by disrupting NF-kappa B nuclear accumulation in CD4 T cells. J Immunol. 2012;188(3):947–951. doi: 10.4049/jimmunol.1101027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Aplan PD, Jones CA, Chervinsky DS, et al. An scl gene product lacking the transactivation domain induces bony abnormalities and cooperates with LMO1 to generate T-cell malignancies in transgenic mice. EMBO J. 1997;16(9):2408–2419. doi: 10.1093/emboj/16.9.2408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lin YW, Nichols RA, Letterio JJ, Aplan PD. Notch1 mutations are important for leukemic transformation in murine models of precursor-T leukemia/lymphoma. Blood. 2006;107(6):2540–2543. doi: 10.1182/blood-2005-07-3013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chung YJ, Choi CW, Slape C, Fry T, Aplan PD. Transplantation of a myelodysplastic syndrome by a long-term repopulating hematopoietic cell. Proc Natl Acad Sci U S A. 2008;105(37):14088–14093. doi: 10.1073/pnas.0804507105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lin YW, Slape C, Zhang Z, Aplan PD. NUP98-HOXD13 transgenic mice develop a highly penetrant, severe myelodysplastic syndrome that progresses to acute leukemia. Blood. 2005;106(1):287–295. doi: 10.1182/blood-2004-12-4794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Choi CW, Chung YJ, Slape C, Aplan PD. A NUP98-HOXD13 fusion gene impairs differentiation of B and T lymphocytes and leads to expansion of thymocytes with partial TCRB gene rearrangement. J Immunol. 2009;183(10):6227–6235. doi: 10.4049/jimmunol.0901121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pear WS, Aster JC, Scott ML, et al. Exclusive development of T cell neoplasms in mice transplanted with bone marrow expressing activated Notch alleles. J Exp Med. 1996;183(5):2283–2291. doi: 10.1084/jem.183.5.2283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.O'Neil J, Calvo J, McKenna K, et al. Activating Notch1 mutations in mouse models of T-ALL. Blood. 2006;107(2):781–785. doi: 10.1182/blood-2005-06-2553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Weng AP, Ferrando AA, Lee W, et al. Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science. 2004;306(5694):269–271. doi: 10.1126/science.1102160. [DOI] [PubMed] [Google Scholar]
- 26.Jeannet R, Mastio J, Macias-Garcia A, et al. Oncogenic activation of the Notch1 gene by deletion of its promoter in Ikaros-deficient T-ALL. Blood. 2010;116(25):5443–5454. doi: 10.1182/blood-2010-05-286658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sakata J, Inoue J, Ohi H, et al. Involvement of V(D)J recombinase in the generation of intragenic deletions in the Rit1/Bcl11b tumor suppressor gene in gamma-ray-induced thymic lymphomas and in normal thymus of the mouse. Carcinogenesis. 2004;25(6):1069–1075. doi: 10.1093/carcin/bgh094. [DOI] [PubMed] [Google Scholar]
- 28.Cairns RA, Iqbal J, Lemonnier F, et al. IDH2 mutations are frequent in angioimmunoblastic T-cell lymphoma. Blood. 2012;119(8):1901–1903. doi: 10.1182/blood-2011-11-391748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Feldman AL, Sun DX, Law ME, et al. Overexpression of Syk tyrosine kinase in peripheral T-cell lymphomas. Leukemia. 2008;22(6):1139–1143. doi: 10.1038/leu.2008.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.de Leval L, Bisig B, Thielen C, Boniver J, Gaulard P. Molecular classification of T-cell lymphomas. Crit Rev Oncol Hematol. 2009;72(2):125–143. doi: 10.1016/j.critrevonc.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 31.Piccaluga PP, Agostinelli C, Tripodo C, et al. Peripheral T-cell lymphoma classification: the matter of cellular derivation. Expert Rev Hematol. 2011;4(4):415–425. doi: 10.1586/ehm.11.37. [DOI] [PubMed] [Google Scholar]
- 32.de Leval L, Gisselbrecht C, Gaulard P. Advances in the understanding and management of angioimmunoblastic T-cell lymphoma. Br J Haematol. 2010;148(5):673–689. doi: 10.1111/j.1365-2141.2009.08003.x. [DOI] [PubMed] [Google Scholar]
- 33.Dogan A, Attygalle AD, Kyriakou C. Angioimmunoblastic T-cell lymphoma. Br J Haematol. 2003;121(5):681–691. doi: 10.1046/j.1365-2141.2003.04335.x. [DOI] [PubMed] [Google Scholar]
- 34.Iqbal J, Weisenburger DD, Greiner TC, et al. Molecular signatures to improve diagnosis in peripheral T-cell lymphoma and prognostication in angioimmunoblastic T-cell lymphoma. Blood. 2010;115(5):1026–1036. doi: 10.1182/blood-2009-06-227579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Legler DF, Loetscher M, Roos RS, Clark-Lewis I, Baggiolini M, Moser B. B cell-attracting chemokine 1, a human CXC chemokine expressed in lymphoid tissues, selectively attracts B lymphocytes via BLR1/CXCR5. J Exp Med. 1998;187(4):655–660. doi: 10.1084/jem.187.4.655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Iliopoulos D, Jaeger SA, Hirsch HA, Bulyk ML, Struhl K. STAT3 activation of miR-21 and miR-181b-1 via PTEN and CYLD are part of the epigenetic switch linking inflammation to cancer. Mol Cell. 2010;39(4):493–506. doi: 10.1016/j.molcel.2010.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Thiefes A, Wolter S, Mushinski JF, et al. Simultaneous blockade of NFkappaB, JNK, and p38 MAPK by a kinase-inactive mutant of the protein kinase TAK1 sensitizes cells to apoptosis and affects a distinct spectrum of tumor necrosis factor [corrected] target genes. J Biol Chem. 2005;280(30):27728–27741. doi: 10.1074/jbc.M411657200. [DOI] [PubMed] [Google Scholar]
- 38.Demchenko YN, Glebov OK, Zingone A, Keats JJ, Bergsagel PL, Kuehl WM. Classical and/or alternative NF-kappaB pathway activation in multiple myeloma. Blood. 2010;115(17):3541–3552. doi: 10.1182/blood-2009-09-243535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Foss FM, Zinzani PL, Vose JM, Gascoyne RD, Rosen ST, Tobinai K. Peripheral T-cell lymphoma. Blood. 2011;117(25):6756–6767. doi: 10.1182/blood-2010-05-231548. [DOI] [PubMed] [Google Scholar]
- 40.Rodríguez-Pinilla SM, Atienza L, Murillo C, et al. Peripheral T-cell lymphoma with follicular T-cell markers. Am J Surg Pathol. 2008;32(12):1787–1799. doi: 10.1097/PAS.0b013e31817f123e. [DOI] [PubMed] [Google Scholar]
- 41.Huang Y, Moreau A, Dupuis J, et al. Peripheral T-cell lymphomas with a follicular growth pattern are derived from follicular helper T cells (TFH) and may show overlapping features with angioimmunoblastic T-cell lymphomas. Am J Surg Pathol. 2009;33(5):682–690. doi: 10.1097/PAS.0b013e3181971591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.King C, Tangye SG, Mackay CR. T follicular helper (TFH) cells in normal and dysregulated immune responses. Annu Rev Immunol. 2008:26741–766. doi: 10.1146/annurev.immunol.26.021607.090344. [DOI] [PubMed] [Google Scholar]
- 43.Yu D, Vinuesa CG. The elusive identity of T follicular helper cells. Trends Immunol. 2010;31(10):377–383. doi: 10.1016/j.it.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 44.Arnold CN, Campbell DJ, Lipp M, Butcher EC. The germinal center response is impaired in the absence of T cell-expressed CXCR5. Eur J Immunol. 2007;37(1):100–109. doi: 10.1002/eji.200636486. [DOI] [PubMed] [Google Scholar]
- 45.Johnson CD, Esquela-Kerscher A, Stefani G, et al. The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Res. 2007;67(16):7713–7722. doi: 10.1158/0008-5472.CAN-07-1083. [DOI] [PubMed] [Google Scholar]
- 46.Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140(6):883–899. doi: 10.1016/j.cell.2010.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kamp DW, Shacter E, Weitzman SA. Chronic inflammation and cancer: the role of the mitochondria. Oncology. 2011;25(5):400–413. [PubMed] [Google Scholar]
- 48.Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436–444. doi: 10.1038/nature07205. [DOI] [PubMed] [Google Scholar]
- 49.Martínez-Delgado B, Cuadros M, Honrado E, et al. Differential expression of NF-kappaB pathway genes among peripheral T-cell lymphomas. Leukemia. 2005;19(12):2254–2263. doi: 10.1038/sj.leu.2403960. [DOI] [PubMed] [Google Scholar]
- 50.Le Gouill S, Milpied N, Buzyn A, et al. Graft-versus-lymphoma effect for aggressive T-cell lymphomas in adults: a study by the Societe Francaise de Greffe de Moelle et de Therapie Cellulaire. J Clin Oncol. 2008;26(14):2264–2271. doi: 10.1200/JCO.2007.14.1366. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.