Abstract
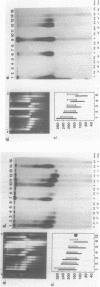
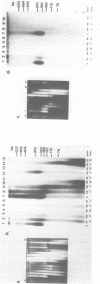
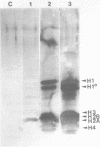
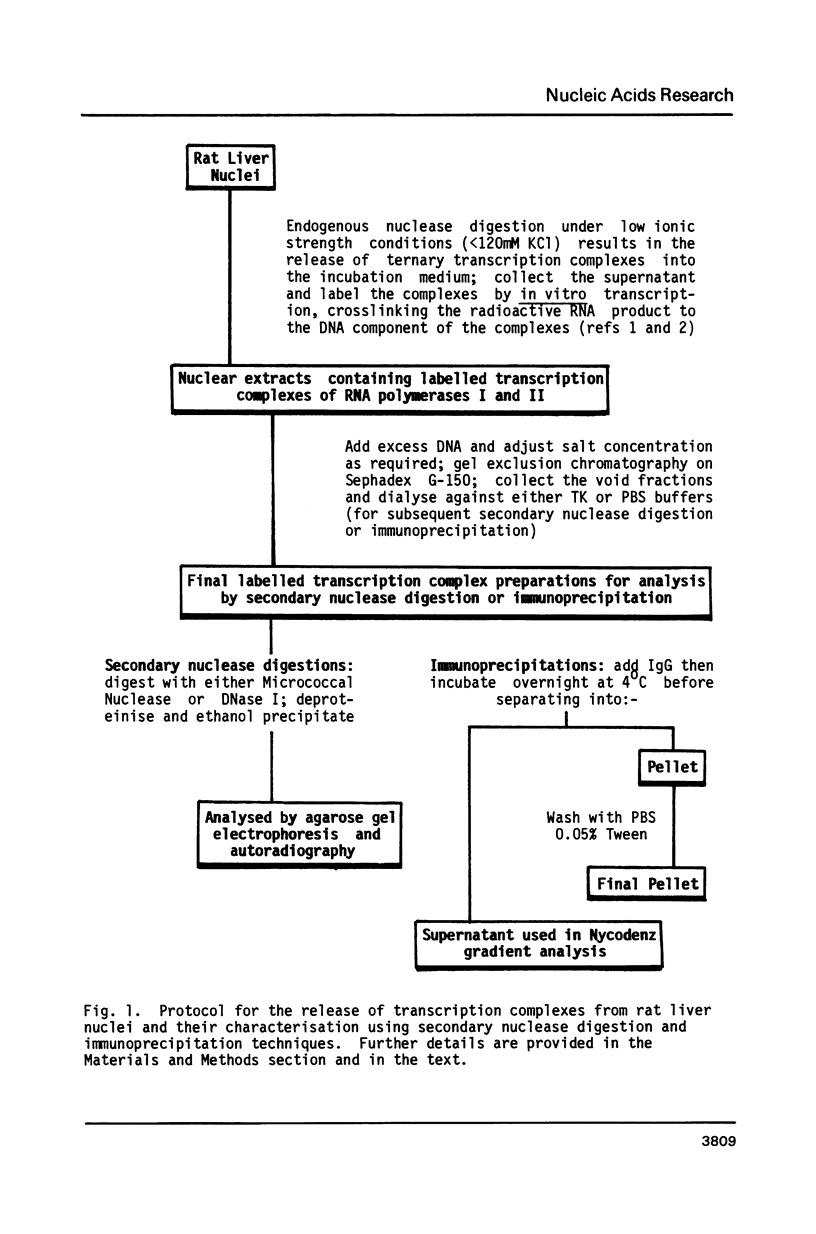
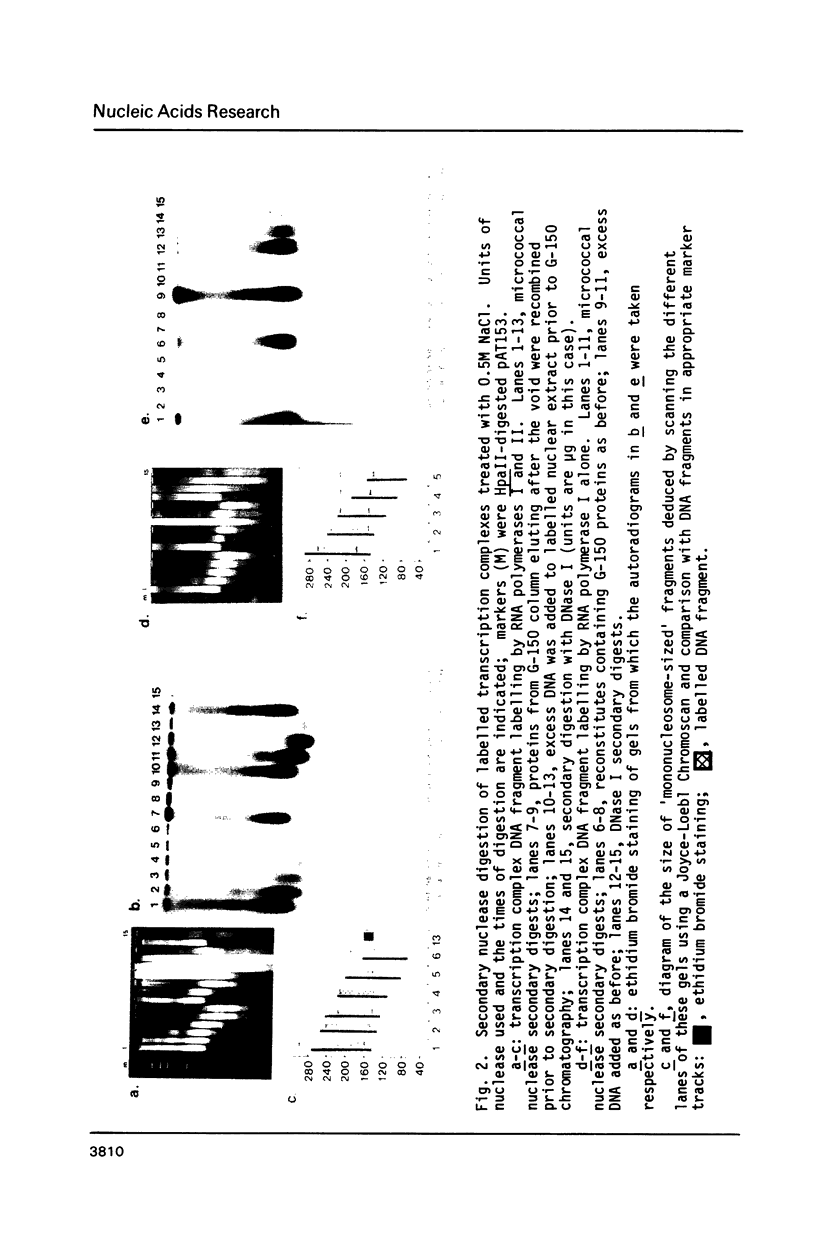
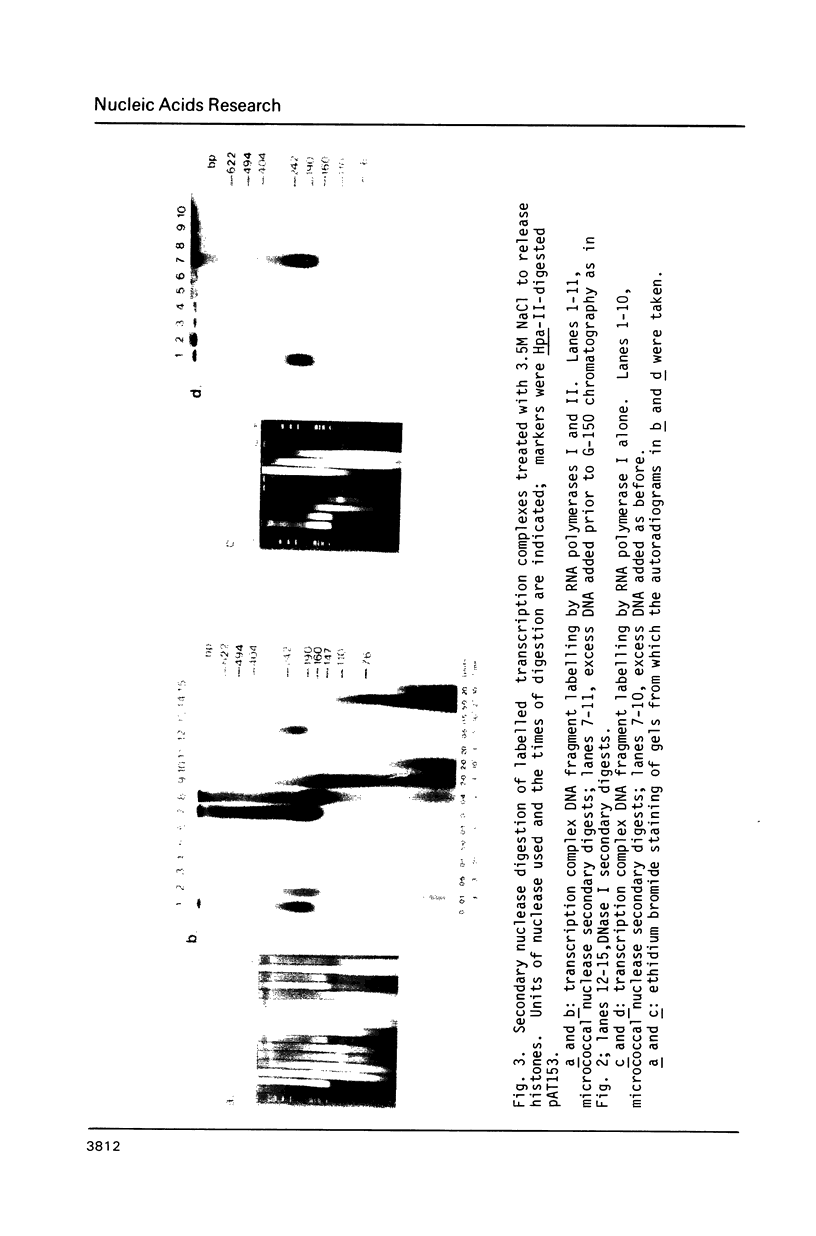
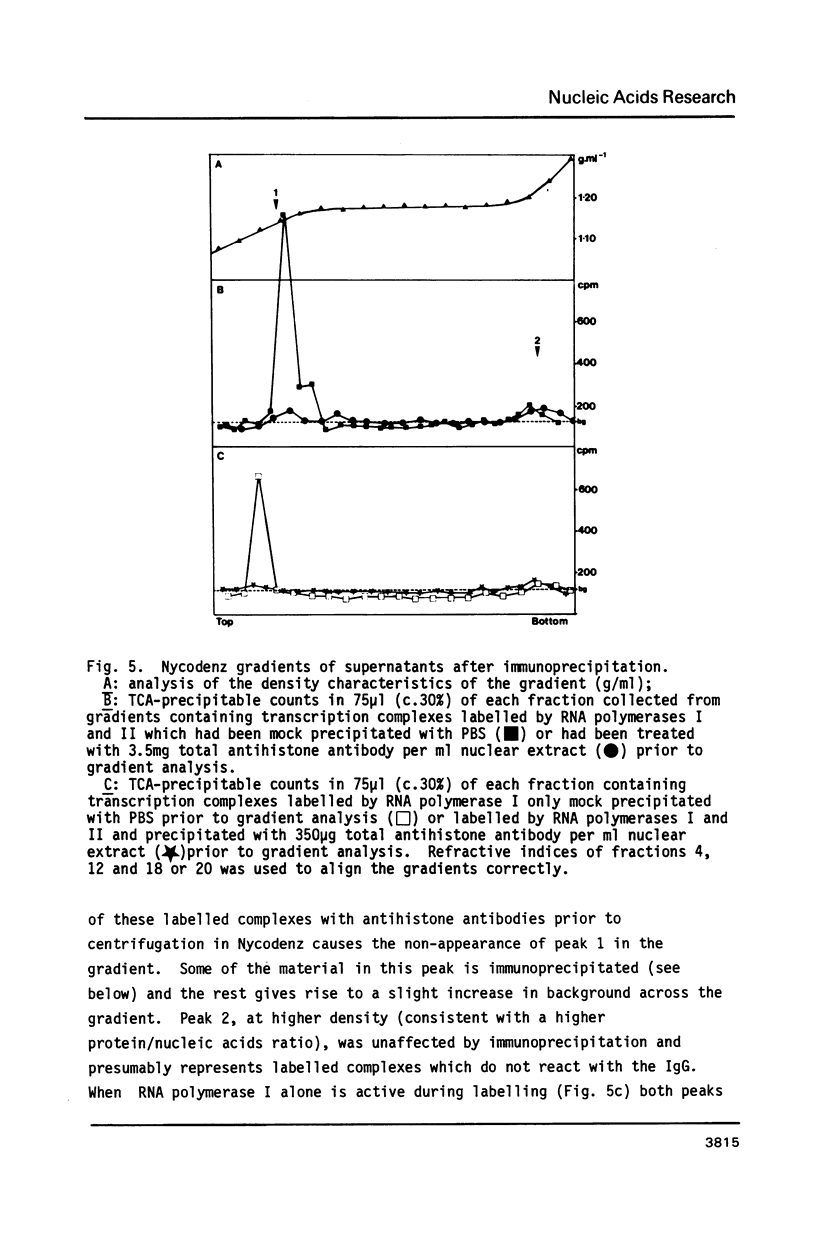
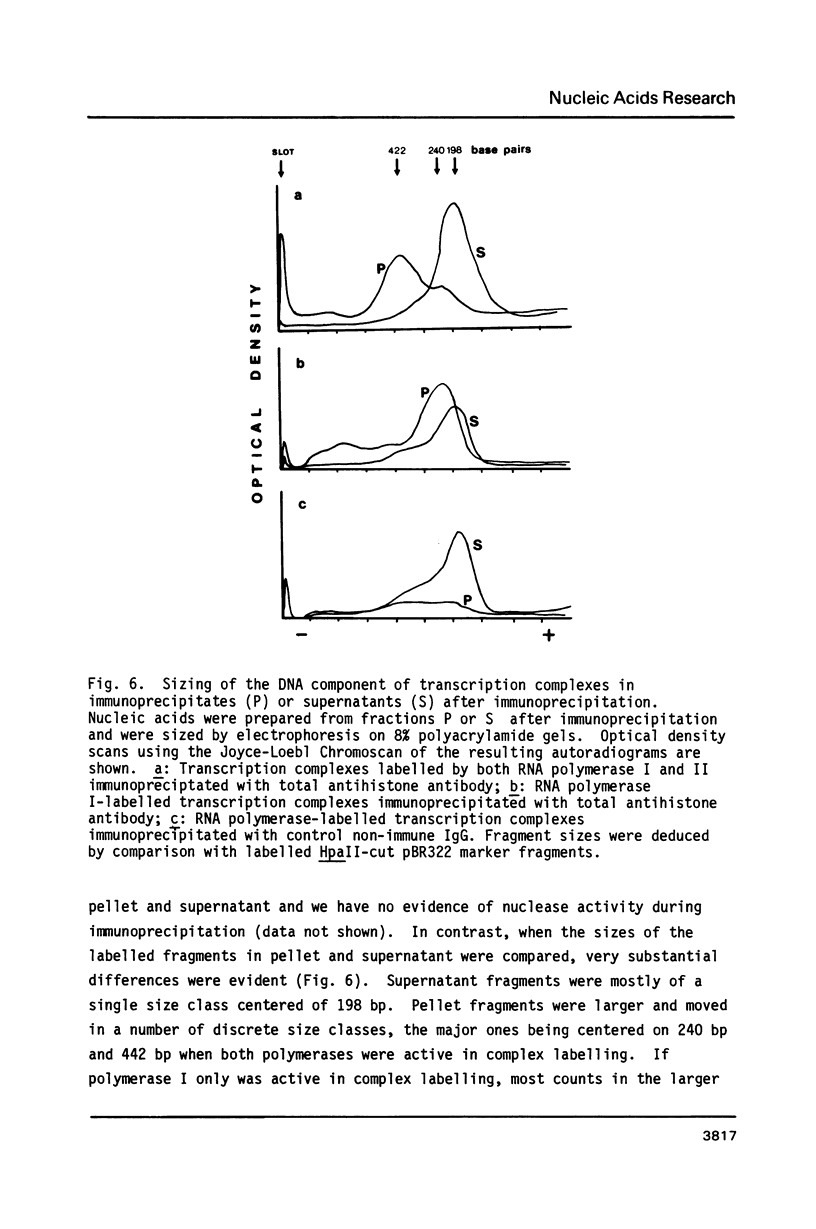
Using a psoralen crosslinking, radioactive labelling technique, we have previously been able to study ternary transcription complexes containing DNA-dependent RNA polymerases I and II which are released from rat liver nuclei by endogenous nuclease digestion [Sargan and Butterworth, refs 1 and 2]. Although the DNA component of these complexes was found to have a 'nucleosome-like' size profile and although the experimental conditions for autodigestion were designed to minimise histone rearrangement, it is necessary to provide further evidence that the periodicity of nuclease cutting around these transcription complexes is conferred by histones. Studies using secondary nuclease digestion of the released transcription complexes now show a digestion barrier characteristic of that conferred by nucleosomal histones which is lost if histones are removed from the complexes. Furthermore, antibodies raised against histones are effective in precipitating transcription complexes of RNA polymerase II and, to a lesser extent, of RNA polymerase I. The data suggest that, in rat hepatic tissue, transcription complexes are in very close proximity (within a few hundred base pairs) of histone-containing, nucleosome-like particles in vivo.
Full text
PDF












Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baer B. W., Rhodes D. Eukaryotic RNA polymerase II binds to nucleosome cores from transcribed genes. Nature. 1983 Feb 10;301(5900):482–488. doi: 10.1038/301482a0. [DOI] [PubMed] [Google Scholar]
- Beard P. Mobility of histones on the chromosome of simian virus 40. Cell. 1978 Nov;15(3):955–967. doi: 10.1016/0092-8674(78)90279-9. [DOI] [PubMed] [Google Scholar]
- Beebee T. J., Butterworth P. H. Transcription of isolated nuclei and nucleoli by exogenous RNA polymerase A and B. Eur J Biochem. 1975 Feb 21;51(2):537–545. doi: 10.1111/j.1432-1033.1975.tb03954.x. [DOI] [PubMed] [Google Scholar]
- Bloom K. S., Anderson J. N. Conformation of ovalbumin and globin genes in chromatin during differential gene expression. J Biol Chem. 1979 Oct 25;254(20):10532–10539. [PubMed] [Google Scholar]
- Camerini-Otero R. D., Zasloff M. A. Nucleosomal packaging of the thymidine kinase gene of herpes simplex virus transferred into mouse cells: an actively expressed single-copy gene. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5079–5083. doi: 10.1073/pnas.77.9.5079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandler D. W., Gralla J. Specific binding and protection of form II SV40 deoxyribonucleic acid by ribonucleic acid polymerase II from wheat germ. Biochemistry. 1980 Apr 15;19(8):1604–1612. doi: 10.1021/bi00549a012. [DOI] [PubMed] [Google Scholar]
- Colavito-Shepanski M., Gorovsky M. A. The histone content of Tetrahymena ribosomal gene-containing chromatin. J Biol Chem. 1983 May 10;258(9):5944–5954. [PubMed] [Google Scholar]
- Coupar B. E., Chesterton C. J. The mechanism by which heparin stimulates transcription in isolated rat liver nuclei. Polyribonucleotide elongation rates and the number of transcribing RNA polymerase molecules present. Eur J Biochem. 1977 Oct 3;79(2):525–533. doi: 10.1111/j.1432-1033.1977.tb11837.x. [DOI] [PubMed] [Google Scholar]
- Coupar B. E., Davies J. A., Chesterton C. J. Quantification of hepatic transcribing RNA polymerase molecules, polyribonucleotide elongation rates and messenger RNA complexity in fed and fasted rats. Eur J Biochem. 1978 Mar 15;84(2):611–623. doi: 10.1111/j.1432-1033.1978.tb12204.x. [DOI] [PubMed] [Google Scholar]
- Ferencz A., Seifart K. H. Comparative effect of heparin on RNA synthesis of isolated rat-liver nucleoli and purified RNA polymerase A. Eur J Biochem. 1975 May 6;53(2):605–613. doi: 10.1111/j.1432-1033.1975.tb04104.x. [DOI] [PubMed] [Google Scholar]
- Foe V. E., Wilkinson L. E., Laird C. D. Comparative organization of active transcription units in Oncopeltus fasciatus. Cell. 1976 Sep;9(1):131–146. doi: 10.1016/0092-8674(76)90059-3. [DOI] [PubMed] [Google Scholar]
- Gariglio P., Bellard M., Chambon P. Clustering of RNA polymerase B molecules in the 5' moiety of the adult beta-globin gene of hen erythrocytes. Nucleic Acids Res. 1981 Jun 11;9(11):2589–2598. doi: 10.1093/nar/9.11.2589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giri C. P., Gorovsky M. A. DNase I sensitivity of ribosomal genes in isolated nucleosome core particles. Nucleic Acids Res. 1980 Jan 11;8(1):197–214. doi: 10.1093/nar/8.1.197-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glikin G. C., Ruberti I., Worcel A. Chromatin assembly in Xenopus oocytes: in vitro studies. Cell. 1984 May;37(1):33–41. doi: 10.1016/0092-8674(84)90298-8. [DOI] [PubMed] [Google Scholar]
- Gould H. J., Cowling G. J., Harborne N. R., Allan J. An examination of models for chromatin transcription. Nucleic Acids Res. 1980 Nov 25;8(22):5255–5266. doi: 10.1093/nar/8.22.5255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodo H. G., 3rd, Sahasrabuddhe C. G., Plishker M. F., Saunders G. F. Use of RNA polymerase as an enzymatic probe of nucleosomal structure. Nucleic Acids Res. 1980 Sep 11;8(17):3851–3864. doi: 10.1093/nar/8.17.3851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Levy A., Noll M. Chromatin fine structure of active and repressed genes. Nature. 1981 Jan 15;289(5794):198–203. doi: 10.1038/289198a0. [DOI] [PubMed] [Google Scholar]
- Lilley D. M., Jacobs M. F., Houghton M. The nature of the interaction of nucleosomes with a eukaryotic RNA polymerase II. Nucleic Acids Res. 1979 Sep 25;7(2):377–399. doi: 10.1093/nar/7.2.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maniatis T., Jeffrey A., van deSande H. Chain length determination of small double- and single-stranded DNA molecules by polyacrylamide gel electrophoresis. Biochemistry. 1975 Aug 26;14(17):3787–3794. doi: 10.1021/bi00688a010. [DOI] [PubMed] [Google Scholar]
- Mathis D. J., Gorovsky M. A. Subunit structure of rDNA-containing chromatin. Biochemistry. 1976 Feb 24;15(4):750–755. doi: 10.1021/bi00649a005. [DOI] [PubMed] [Google Scholar]
- Mathis D., Oudet P., Chambon P. Structure of transcribing chromatin. Prog Nucleic Acid Res Mol Biol. 1980;24:1–55. doi: 10.1016/s0079-6603(08)60670-4. [DOI] [PubMed] [Google Scholar]
- Ness P. J., Labhart P., Banz E., Koller T., Parish R. W. Chromatin structure along the ribosomal DNA of Dictyostelium. Regional differences and changes accompanying cell differentiation. J Mol Biol. 1983 May 25;166(3):361–381. doi: 10.1016/s0022-2836(83)80090-4. [DOI] [PubMed] [Google Scholar]
- Piper P. W., Celis J., Kaltoft K., Leer J. C., Nielsen O. F., Westergaard O. Tetrahymena ribosomal RNA gene chromatin is digested by micrococcal nuclease at sites which have the same regular spacing on the DNA as corresponding sites in the bulk nuclear chromatin. Nucleic Acids Res. 1976 Feb;3(2):493–505. doi: 10.1093/nar/3.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prior C. P., Cantor C. R., Johnson E. M., Littau V. C., Allfrey V. G. Reversible changes in nucleosome structure and histone H3 accessibility in transcriptionally active and inactive states of rDNA chromatin. Cell. 1983 Oct;34(3):1033–1042. doi: 10.1016/0092-8674(83)90561-5. [DOI] [PubMed] [Google Scholar]
- Reeves R. Structure of Xenopus ribosomal gene chromatin during changes in genomic transcription rates. Cold Spring Harb Symp Quant Biol. 1978;42(Pt 2):709–722. doi: 10.1101/sqb.1978.042.01.073. [DOI] [PubMed] [Google Scholar]
- Ryoji M., Worcel A. Chromatin assembly in Xenopus oocytes: in vivo studies. Cell. 1984 May;37(1):21–32. doi: 10.1016/0092-8674(84)90297-6. [DOI] [PubMed] [Google Scholar]
- Sargan D. R., Butterworth P. H. Eukaryotic ternary transcription complexes. I. The release of ternary transcription complexes of RNA polymerases I and II by the endogenous nucleases of rat liver nuclei. Nucleic Acids Res. 1982 Aug 11;10(15):4641–4653. doi: 10.1093/nar/10.15.4641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sargan D. R., Butterworth P. H. Eukaryotic ternary transcription complexes. II. An approach to the determination of chromatin conformation at the site of transcription. Nucleic Acids Res. 1982 Aug 11;10(15):4655–4669. doi: 10.1093/nar/10.15.4655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha S. N., Hellwig R. J., Allison D. P., Niyogi S. K. Conversion of simian virus 40 DNA to ordered nucleoprotein structures by extracts that direct accurate initiation by eukaryotic RNA polymerase II. Nucleic Acids Res. 1982 Sep 25;10(18):5533–5552. doi: 10.1093/nar/10.18.5533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sollner-Webb B., Felsenfeld G. A comparison of the digestion of nuclei and chromatin by staphylococcal nuclease. Biochemistry. 1975 Jul;14(13):2915–2920. doi: 10.1021/bi00684a019. [DOI] [PubMed] [Google Scholar]
- Stacks P. C., Schumaker V. N. Nucleosome dissociation and transfer in concentrated salt solutions. Nucleic Acids Res. 1979 Dec 20;7(8):2457–2467. doi: 10.1093/nar/7.8.2457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stollar B. D., Ward M. Rabbit antibodies to histone fractions as specific reagents for preparative and comparative studies. J Biol Chem. 1970 Mar 25;245(6):1261–1266. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weisbrod S., Weintraub H. Isolation of actively transcribed nucleosomes using immobilized HMG 14 and 17 and an analysis of alpha-globin chromatin. Cell. 1981 Feb;23(2):391–400. doi: 10.1016/0092-8674(81)90134-3. [DOI] [PubMed] [Google Scholar]
- Woodcock C. L., Frado L. L., Hatch C. L., Ricciardiello L. Fine structure of active ribosomal genes. Chromosoma. 1976 Oct 12;58(1):33–39. doi: 10.1007/BF00293438. [DOI] [PubMed] [Google Scholar]
- Wu C., Wong Y. C., Elgin S. C. The chromatin structure of specific genes: II. Disruption of chromatin structure during gene activity. Cell. 1979 Apr;16(4):807–814. doi: 10.1016/0092-8674(79)90096-5. [DOI] [PubMed] [Google Scholar]
- von Holt C., Brandt W. F. Fractionation of histones on molecular sieve matrices. Methods Cell Biol. 1977;16:205–225. doi: 10.1016/s0091-679x(08)60101-6. [DOI] [PubMed] [Google Scholar]