Abstract
Objectives
The scientific literature to date lacks population-based studies on the demographics, clinical features, and survival of patients with adenoid cystic carcinoma (ACC) of different anatomic sites.
Methods
We identified 5349 ACC cases in major salivary glands (N=1850), minor salivary glands (N=2077), breast (N=696), skin (N=291), lung and bronchus (N=203), female genital system (N=132), and eye and orbit (N=100) from the Surveillance, Epidemiology, and End Results (SEER) registry. Differences in demographics, clinical features, and survival of patients were assessed.
Results
ACC of the eye and orbit was associated with younger age at presentation (mean=49.9 years). ACC of the skin or breast tended to present with less aggressive prognostic features, while ACC of the lung and bronchus or eye and orbit tended to present with more aggressive prognostic features. In multivariate survival analysis of patients presenting with localized disease, patients with ACC of breast (HR=0.40) or skin (HR=0.40) had a significantly lower risk death than patients with ACC of major salivary glands, while patients with ACC of lung and bronchus (HR=3.72) or eye and orbit (HR=3.67) had a significantly higher risk. For patients presenting with regional disease, the only clear prognostic difference in multivariate analysis was that patients with ACC of skin did significantly better.
Conclusions
The demographics and clinical features of ACC differ by disease site. Site may be an important predictor of survival for patients presenting with localized disease but is less important for patients presenting with regional disease.
Keywords: Adenoid Cystic Carcinoma, SEER, Epidemiology, Site, Survival
Introduction
Adenoid cystic carcinoma (ACC) is a relatively rare malignancy that arises mostly in major and minor salivary glands. ACC accounts for only 1% of all head and neck malignancies.1 ACCs account for approximately one-quarter of all salivary gland carcinomas.2 ACC also occurs in other glandular areas, including breast, lung, cervix, skin, and lacrimal gland, and various other sites have also been reported. ACC is a slow-growing but highly invasive malignancy with a propensity for recurrence and delayed onset of distant metastasis. The prolonged clinical course of ACC is well known, and patients may survive for years even after recurrence. However, little is understood of the etiology of ACC.3
Interestingly, although ACCs at different anatomic sites share common histologic features and expression of the proto-oncogene c-KIT,4 the clinical features and outcomes of ACC appear to vary by site. For example, in large ACC studies,5,6 96% of ACC cases arising in the breast were localized, while only 57% of ACC cases arising in the head and neck were localized. Distant metastases and perineural invasion are rare for ACCs of the breast, while metastases and perineural invasion are common for ACCs of the salivary glands.4 The 10-year survival rates for patients with ACCs of the breast, major salivary glands, and lung have been reported to be >90%,4 71%,6 and 39%,7 respectively.
Because ACC is an uncommon malignancy, most published studies of ACC have been case reports or clinical series with sample sizes of fewer than 70 patients.8–10 There are a few published population-based studies of ACC5,11,12; however, these studies investigated only the clinical behaviors of ACCs at a single anatomic site and did not include comparisons between different sites. Therefore, we conducted a population-based study using Surveillance, Epidemiology, and End Results (SEER) program data to determine the demographics, clinical features, and survival of patients with ACC at different anatomic sites.
Materials and Methods
The SEER program databases contain information on cancer incidence and survival from specific geographic areas across the United States. We used the SEER 17 registries, which covers 28% of the US population and covers Atlanta, Connecticut, Detroit, Hawaii, Iowa, New Mexico, San Francisco-Oakland, Seattle-Puget Sound, Utah, Los Angeles, San Jose-Monterey, Rural Georgia, Alaska, Greater California, Kentucky, Louisiana, and New Jersey. Since the SEER program developed from 9 registries in 1973 to 17 registries in 2000, not all registries contributed cases since 1973. Atlanta, Connecticut, Detroit, Hawaii, Iowa, New Mexico, and San Francisco-Oakland were the original registries and contributed cases for the entire period from 1973 through 2008, while Seattle-Puget Sound and Atlanta contributed cases beginning in 1974 and 1975, respectively. Los Angeles, San Jose-Monterey, Rural Georgia and the Alaska Native Tumor Registry contributed cases beginning in 1992; and Greater California, Kentucky, Louisiana, and New Jersey contributed cases beginning in 2000. The data were extracted using SEER*Stat software, version 7.0.4 (NCI; Bethesda, Maryland), from the 1973–2008 databases, which were released in April 2011.
On the basis of the ICD-O-3 codes provided in the SEER database, we identified all cases of the following 4 histological tumor types, which are the most common types occurring in salivary glands: (1) ACC, “Adenoid cystic carcinoma 8200/3”; (2) mucoepidermoid carcinoma, “Mucoepidermoid carcinoma 8430/3”; (3) acinic cell carcinoma, “Acinar cell carcinoma 8550/3” and “Acinar cell cystadenocarcinoma 8551/3”; and (4) carcinoma-ex pleomorphic adenoma/malignant mixed tumor, “Carcinoma in pleomorphic adenoma 8941/3” and “Mixed tumor, malignant, NOS, 8940/3”.
A total of 5464 cases of ACC diagnosed during 1973–2008 with known patient age and tumor malignant behavior were identified. We categorized sites on the basis of the variable “site recode with Kaposi and mesothelioma” in the SEER database. Among all cases, 5349 cases arose in 7 sites with 100 or more cases and were included in the study. Sixty-three cases of ACC of unclassified site and 52 cases of ACC of unusual sites (23 digestive system, 18 male genital system, 7 soft tissue, 3 urinary system, and 1 endocrine system) were excluded. The sites with at least 100 cases each were grouped into 7 groups as follows: (1) major salivary glands; (2) minor salivary glands, including “lip”, “tongue”, “floor of mouth”, “ gum and other mouth”, “nasopharynx”, “oropharynx”, “hypopharynx”, “other oral cavity and pharynx”, “nose, nasal cavity and middle ear”, “larynx”, and “trachea”; (3) breast; (4) skin; (5) lung and bronchus; (6) female genital system; and (7) eye and orbit. We used “SEER Historic Stage A” to group patients according to tumor stage. Stage was defined according to the SEER program as follows: (1) localized, malignancy limited to the organ of origin; no spread beyond organ of origin; infiltration past basement membrane of epithelium into stroma of organ; (2) regional, tumor extension beyond the limits of the organ of origin but no distant disease; and (3) distant, tumor spread to areas of the body distant or remote from the primary tumor. The SEER staging system, which we utilized in this study, is somewhat independent on the nodal metastasis status of the tumor. For instance, a tumor at SEER regional stage may or may not have nodal metastasis, because tumor extension beyond the local organ may or may not be due to regional metastases. Nodal metastasis and tumor size information were not available until 1983. Information on tumor grade was not available for most cases. Specific data about perineural invasion are not available in the SEER database.
Statistical analysis
The distributions of histological type, demographics, and clinical features were calculated using the frequency session of SEER*Stat 7.0.4. Differences in demographics and clinical features were assessed with chi-square test or t-test. Patients diagnosed with second or later primary ACC patients, not actively followed, and patients diagnosed by death certificate only or by autopsy only were excluded during survival analysis. Patients were stratified by tumor stage during the survival analysis. The 5-year, 10-year, 15-year, 20-year, and 25-year disease-specific survival rates were estimated using the survival session of SEER*Stat 7.0.4 using the Kaplan-Meier method. The survival time was calculated from the date of diagnosis to the date of death caused by ACC or December 31, 2008, the follow-up cut-off date. Patients who died of causes other than ACC and patients who were still alive at the time of the follow-up cut-off were censored. The log-rank test was utilized to assess differences between survival curves. The hazard ratios and their 95% confident intervals (CIs) were determined using univariate and multivariate Cox proportional hazards models and SAS statistical software, version 9.2 (SAS Institute Inc., Carey, NC).13 Year of diagnosis, age, sex, race, marital status, and treatment were adjusted when the relative risk was estimated. All the factors were treated as categorical variables except age, which was treated as a continuous variable. Tumor grade, tumor size, and lymph node metastasis were not included in multivariate analysis because the large amount of missing information on these variables would have resulted in a drastic decrease in our sample size. For all statistical tests, significance was considered to be achieved when P<.05.
Results
Table 1 shows the distribution by anatomic site of the 4 most common histological tumor types occurring in salivary glands: ACC, mucoepidermoid carcinoma, acinar cell carcinoma, and carcinoma ex pleomorphic adenoma/malignant mixed tumor. With the exception of acinar cell carcinoma, which is also a histological variant of prostate carcinoma, these cancers most commonly occurred in major and minor salivary glands. ACC demonstrated a relatively widespread distribution: 13% of cases occurred in the breast, 5% in the skin, 4% in the lung or bronchus, 2% in the female genital system, and 2% in the eye or orbit. ACC occurred with almost equal frequency within the parotid gland and submandibular gland, while other salivary gland carcinomas occurred with much greater frequency in the parotid gland.
Table 1.
Site Distribution of Adenoid Cystic Carcinomas, Mucoepidermoid Carcinomas, Acinar Cell Carcinomas, and Carcinoma Ex Pleomorphic Adenoma/Malignant Mixed Tumors
| Adenoid Cystic Carcinoma N = 5464 |
Mucoepidermoid Carcinoma N = 6751 |
Acinar Cell Carcinoma N = 17066 |
Carcinoma-ex /Malignant Mixed N = 912 |
|
|---|---|---|---|---|
| Tumor Site | No. (%) | No. (%) | No. (%) | No. (%) |
| Oral cavity & pharynx | 3208 (58.7) | 5837 (86.5) | 1822 (10.7) | 755 (82.8) |
| Major salivary gland | 1850 (33.9) | 3652 (54.1) | 1681 (9.8) | 673 (73.8) |
| Parotid gland | 907 (16.6) | 3107 (46.0) | 1611 (9.4) | 492 (53.9) |
| Submandibular gland | 783 (14.3) | 369 (5.5) | 39 (0.2) | 123 (13.5) |
| Sublingual gland | 57 (1.0) | 66 (1.0) | 1 (0.0) | 1 (0.1) |
| NOS or overlapping | 103 (1.9) | 110 (1.6) | 30 (0.2) | 54 (5.9) |
| Lip | 90 (1.7) | 139 (2.1) | 21 (0.1) | 7 (0.8) |
| Tongue | 217 (4.0) | 225 (3.3) | 2 (0.0) | 2 (0.2) |
| Floor of mouth | 94 (1.7) | 182 (2.7) | 7 (0.0) | 1 (0.1) |
| Gum & other mouth | 790 (14.5) | 1506 (22.3) | 103 (0.6) | 59 (6.5) |
| Nasopharynx | 109 (2.0) | 35 (0.5) | 2 (0.0) | 5 (0.5) |
| Oropharynx | 26 (0.5) | 58 (0.9) | 3 (0.0) | 2 (0.2) |
| Hypopharynx | 12 (0.2) | 21 (0.3) | 1 (0.0) | 0 (0.0) |
| Other oral cavity & pharynx | 20 (0.4) | 19 (0.3) | 2 (0.0) | 6 (0.7) |
| Digestive system | 23 (0.4) | 62 (0.9) | 324 (1.9) | 9 (1.0) |
| Esophagus | 16 (0.3) | 16 (0.2) | 2 (0.0) | 1 (0.1) |
| Pancreas | 1 (0.0) | 15 (0.2) | 280 (1.6) | 0 (0.0) |
| Anus, anal canal, & anorectum | 6 (0.1) | 16 (0.2) | 0 (0.0) | 0 (0.0) |
| Respiratory system | 922 (16.9) | 579 (8.6) | 1159 (6.8) | 26 (2.9) |
| Nose, nasal cavity, & middle ear | 525 (9.6) | 131 (1.9) | 17 (0.1) | 17 (1.9) |
| Larynx | 62 (1.1) | 43 (0.6) | 0 (0.0) | 2 (0.2) |
| Trachea | 132 (2.4) | 21 (0.3) | 1 (0.0) | 0 (0.0) |
| Lung & bronchus | 203 (3.7) | 384 (5.7) | 1141 (6.7) | 7 (0.8) |
| Bones and joints | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Soft tissue including heart | 7 (0.1) | 4 (0.1) | 1 (0.0) | 4 (0.4) |
| Skin | 291 (5.3) | 49 (0.7) | 5 (0.0) | 53 (5.8) |
| Breast | 696 (12.7) | 5 (0.1) | 35 (0.2) | 8 (0.9) |
| Female genital system | 132 (2.4) | 15 (0.2) | 2 (0.0) | 17 (1.9) |
| Cervix uteri | 74 (1.4) | 13 (0.2) | 0 (0.0) | 0 (0.0) |
| Corpus & uterus, NOS | 1 (0.0) | 0 (0.0) | 1 (0.0) | 5 (0.5) |
| Ovary | 2 (0.0) | 2 (0.0) | 1 (0.0) | 12 (1.3) |
| Vagina | 5 (0.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Vulva | 50 (0.9) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Male genital system | 18 (0.3) | 0 (0.0) | 13659 (80.0) | 5 (0.5) |
| Prostate | 16 (0.3) | 0 (0.0) | 13659 (80.0) | 2 (0.2) |
| Urinary system | 3 (0.1) | 0 (0.0) | 27 (0.2) | 15 (1.6) |
| Eye & orbit | 100 (1.8) | 46 (0.7) | 0 (0.0) | 10 (1.1) |
| Lacrimal gland | 81 (1.5) | 21 (0.3) | 0 (0.0) | 9 (1.0) |
| Brain & other nervous system | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Endocrine system | 1 (0.0) | 35 (0.5) | 6 (0.0) | 1 (0.1) |
| Thyroid gland | 1 (0.0) | 26 (0.4) | 6 (0.0) | 1 (0.1) |
| Miscellaneous | 63 (1.2) | 119 (1.8) | 26 (0.2) | 9 (1.0) |
NOS, not otherwise specified.
Table 2 shows the distribution of ACC patients’ demographics and clinical features across the 7 sites included in this study. The majority of ACC cases occurred in patients aged 40–69 years. However, almost one-third of patients with eye and orbit ACC presented before 40 years of age, and almost half of women with genital system ACC presented at age 70 years or older. Patients with ACC of the eye and orbit were significantly younger at presentation than patients with ACC of the major salivary glands (mean age, 49.9 years versus 55.5 years; P=.001), while patients with tumors of the female genital system (65.0 years), skin (57.5 years), and breast (61.5 years) were significantly older (P≤.001, P<.001, and P<.001, respectively). For all sites except lung and bronchus, which had a significantly lower proportion of female cases (P<.001), more females than males were affected. The proportion of African Americans was significantly higher among patients with ACC of the female genital system or eye and orbit than among patients with ACC of major salivary glands (P<.001, and P=.001, respectively), while the percentage of African Americans was significantly lower among patients with ACC of the breast (P=.038).
Table 2.
Demographics and Clinic Features of Patients with Adenoid Cystic Carcinoma by Tumor Site
| Major Salivary Glands |
Minor Salivary Glands |
Breast | Skin | Lung & Bronchus |
Female Genital System |
Eye & Orbit |
|
|---|---|---|---|---|---|---|---|
| N = 1850 | N = 2077 | N = 696 | N = 291 | N = 203 | N = 132 | N = 100 | |
| Variablea | No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | No. (%) |
| Year of diagnosis | |||||||
| 1973–1981 | 311 (16.8) | 330 (15.9) | 148 (21.3) | 33 (11.3) | 45 (22.2) | 25 (18.9) | 22 (22.0) |
| 1982–1990 | 300 (16.2) | 374 (18.0) | 86 (12.4) | 52 (17.9) | 30 (14.8) | 15 (11.4) | 16 (16.0) |
| 1991–1999 | 398 (21.5) | 478 (23.0) | 136 (19.5) | 64 (22.0) | 35 (17.2) | 41 (31.1) | 13 (13.0) |
| 2000–2008 | 841 (45.5) | 895 (43.1) | 326 (46.8) | 142 (48.8) | 93 (45.8) | 51 (38.6) | 49 (49.0) |
| Age | |||||||
| ≤39 years | 337 (18.2) | 260 (12.5) | 28 (4.0) | 26 (8.9) | 13 (6.4) | 9 (6.8) | 29 (29.0) |
| 40–69 years | 1084 (58.6) | 1237 (59.5) | 466 (67.0) | 155 (53.3) | 128 (63.1) | 58 (43.9) | 53 (53.0) |
| ≥70 years | 429 (23.2) | 580 (28.0) | 202 (29.0) | 110 (37.8) | 62 (30.5) | 65 (49.3) | 18 (18.0) |
| Sex | |||||||
| Male | 754 (40.8) | 879 (42.3) | 11 (1.6) | 123 (42.3) | 110 (54.2) | 0 (0.0) | 43 (43.0) |
| Female | 1096 (59.2) | 1198 (57.7) | 685 (98.4) | 168 (57.7) | 93 (45.8) | 132 (100.0) | 57 (57.0) |
| Race | |||||||
| White | 1508 (81.8) | 1663 (80.7) | 606 (87.7) | 251 (89.0) | 171 (84.2) | 92 (69.7) | 70 (70.0) |
| Black | 164 (8.9) | 214 (10.4) | 46 (6.7) | 18 (6.4) | 17 (8.4) | 32 (24.2) | 18 (18.0) |
| Other | 172 (9.3) | 184 (8.9) | 39 (5.6) | 13 (4.6) | 15 (7.4) | 8 (6.1) | 12 (12.0) |
| Marital status | |||||||
| Single | 269 (15.0) | 268 (13.6) | 72 (10.6) | 37 (15.9) | 31 (15.6) | 24 (18.6) | 21 (22.1) |
| Married | 1178 (66.0) | 1256 (63.8) | 411 (60.6) | 137 (58.8) | 122 (61.3) | 44 (34.1) | 54 (56.8) |
| Sep/Div/Wid | 339 (19.0) | 446 (22.6) | 195 (28.8) | 59 (25.3) | 46 (23.1) | 61 (47.3) | 20 (21.1) |
| Tumor grade | |||||||
| Low (1 or 2) | 210 (62.3) | 243 (61.8) | 272 (80.2) | 20 (87.0) | 33 (75.0) | 12 (42.9) | 7 (41.2) |
| High (3 or 4) | 127 (37.7) | 150 (38.2) | 67 (19.8) | 3 (13.0) | 11 (25.0) | 16 (57.1) | 10 (58.8) |
| Tumor sizeb | |||||||
| ≤2 cm | 528 (42.8) | 422 (38.3) | 318 (63.1) | 91 (72.2) | 28 (27.4) | 17 (27.4) | 10 (22.2) |
| >2 cm | 705 (57.2) | 679 (61.7) | 186 (36.9) | 35 (27.8) | 74 (72.6) | 45 (72.6) | 35 (77.8) |
| Nodal metastasisb | |||||||
| None | 1049 (82.7) | 1111 (90.2) | 466 (95.5) | 146 (98.0) | 74 (61.2) | 79 (96.3) | 56 (100.0) |
| Yes | 219 (17.3) | 121 (9.8) | 22 (4.5) | 3 (2.0) | 47 (38.8) | 3 (3.7) | 0 (0.0) |
| Disease stage | |||||||
| Localized | 910 (51.8) | 635 (35.5) | 622 (90.5) | 173 (69.2) | 34 (26.8) | 77 (63.1) | 41 (44.5) |
| Regional | 638 (36.3) | 901 (50.4) | 55 (8.0) | 67 (26.8) | 59 (46.4) | 37 (30.3) | 41 (44.5) |
| Distant | 208 (11.9) | 252 (14.1) | 10 (1.5) | 10 (4.0) | 34 (26.8) | 8 (6.6) | 10 (11.0) |
| Treatment | |||||||
| Surgery + XRT | 1125 (62.5) | 968 (48.4) | 207 (30.4) | 37 (13.0) | 32 (16.7) | 35 (27.6) | 52 (54.7) |
| Neither | 60 (3.4) | 124 (6.2) | 11 (1.6) | 17 (5.9) | 39 (20.3) | 4 (3.1) | 3 (3.2) |
| XRT only | 51 (2.8) | 269 (13.4) | 2 (0.3) | 6 (2.1) | 41 (21.3) | 21 (16.5) | 2 (2.1) |
| Surgery only | 563 (31.3) | 640 (32.0) | 461 (67.7) | 226 (79.0) | 80 (41.7) | 67 (52.8) | 38 (40.0) |
Sep/Div/Wid, separated, divorced, or widowed; XRT, radiation therapy.
The unknown group for each variable was not shown.
SEER database only provides data from 1983–2008.
Information on tumor grade was available for very few cases. Over 80% of tumors in the breast and skin were low grade, while tumors of the female genital system and eye and orbit were typically high grade. Over 60% of ACCs of the skin or breast were ≤2 cm, while over 70% of ACCs of the lung and bronchus, female genital system, and eye and orbit were ≥2 cm. The mean tumor size at presentation was significantly smaller for ACC of the breast (2.2 cm) than for ACC of the major salivary glands (2.8 cm) (P<.001), while the mean tumor size was significantly larger for ACC of the female genital system (3.7 cm) and lung and bronchus (3.7 cm) (P= .012 and P<.001, respectively).
Nodal metastasis was not common for any site except lung and bronchus. Similarly, only ACC of the lung and bronchus was commonly associated with distant metastases at presentation (27%). Ninety-one percent of breast ACCs were localized at diagnosis, versus only 36% of minor salivary gland ACCs and only 27% of lung and bronchus ACCs. ACCs in breast, skin, lung and bronchus, and female genital system were more likely to be treated with surgery without radiation, while ACCs in major salivary glands, minor salivary glands, and eye and orbit were more likely to be treated with both surgery and radiation.
Because stage is such an important predictor of prognosis,6,11 we stratified tumors using the SEER Historic Stage A system (localized, regional, and distant stage) when we conducted the survival analysis. Table 3 shows disease-specific survival rates at 5, 10, 15, 20, and 25 years for patients with ACC. Figures 1 and 2 show Kaplan-Meier disease-specific survival curves for patients with localized disease and regional disease, respectively. Patients who presented with localized disease had favorable survival, and for all sites except eye and orbit, the disease-specific survival rates for patients with localized disease at presentation were above 50% even 25 years after diagnosis. Patients who presented with distant disease had low survival rates, and the disease-specific survival rate was only approximately 30% 10 years after diagnosis. In Kaplan-Meier disease-specific survival analysis of patients with localized disease, ACC of the skin and breast was associated with favorable survival compared to ACC of major salivary glands (log-rank P=.017 and P<.001, respectively), whereas ACC of lung and bronchus and eye and orbit was associated with worse survival (both log-rank P<.001) (Figure 1). Among patients with regional disease at diagnosis, the disease-specific survival rates were significantly better for patients with ACC of the skin than for ACC of major salivary glands (log-rank P=.003), whereas disease-specific survival rates were significantly worse for patients with ACC of minor salivary glands or lung and bronchus (log-rank P=.008 and .017, respectively) (Figure 2).
Table 3.
Disease-Specific Survival for Patients with Adenoid Cystic Carcinoma by Anatomic Site and Stage at Presentation
| Variable | Major Salivary Glands |
Minor Salivary Glands |
Breast | Skin | Lung & Bronchus |
Female Genital System |
Eye & Orbit |
|---|---|---|---|---|---|---|---|
| Localized stage | |||||||
| No. | 809 | 552 | 542 | 144 | 22 | 63 | 38 |
| 5-year DSS (%) | 93.9 | 92.4 | 96.8 | 96.4 | 71.5 | 87.2 | 88.7 |
| 10-year DSS (%) | 88.1 | 85.5 | 94.0 | 93.7 | 71.5 | 76.8 | 58.7 |
| 15-year DSS (%) | 79.1 | 78.3 | 90.0 | 93.7 | N.A. | 66.6 | 52.2 |
| 20-year DSS (%) | 71.7 | 74.0 | 85.7 | 88.5 | N.A. | 66.6 | 44.7 |
| 25-year DSS (%) | 69.6 | 65.2 | 84.5 | 88.5 | N.A. | 66.6 | 44.7 |
| Regional stage | |||||||
| No. | 575 | 804 | 48 | 59 | 47 | 36 | 35 |
| 5-year DSS (%) | 78.9 | 76.2 | 78.5 | 97.8 | 72.0 | 62.6 | 84.9 |
| 10-year DSS (%) | 64.7 | 56.7 | 75.4 | 94.7 | 45.5 | 56.9 | 36.8 |
| 15-year DSS (%) | 55.4 | 47.3 | 70.0 | 81.1 | 15.2 | 56.9 | N.A. |
| 20-year DSS (%) | 49.1 | 37.5 | 64.6 | 54.1 | N.A. | 56.9 | N.A. |
| 25-year DSS (%) | 42.1 | 33.1 | 64.6 | 54.1 | N.A. | N.A. | N.A. |
| Distant stage | |||||||
| No. | 187 | 226 | 9 | 8 | 28 | 6 | 10 |
| 5-year DSS (%) | 43.3 | 55.4 | 15.9 | 54.7 | 34.7 | 80.0 | 40.0 |
| 10-year DSS (%) | 30.7 | 33.4 | 15.9 | 0.0 | N.A. | 26.7 | 26.7 |
| 15-year DSS (%) | 21.1 | 20.9 | 0.0 | 0.0 | N.A. | 26.7 | 26.7 |
| 20-year DSS (%) | 21.1 | 17.1 | 0.0 | 0.0 | N.A. | N.A. | 26.7 |
| 25-year DSS (%) | 21.1 | 8.6 | 0.0 | 0.0 | N.A. | N.A. | N.A. |
DSS, disease-specific survival; N.A., not enough intervals to produce statistics.
Figure 1.
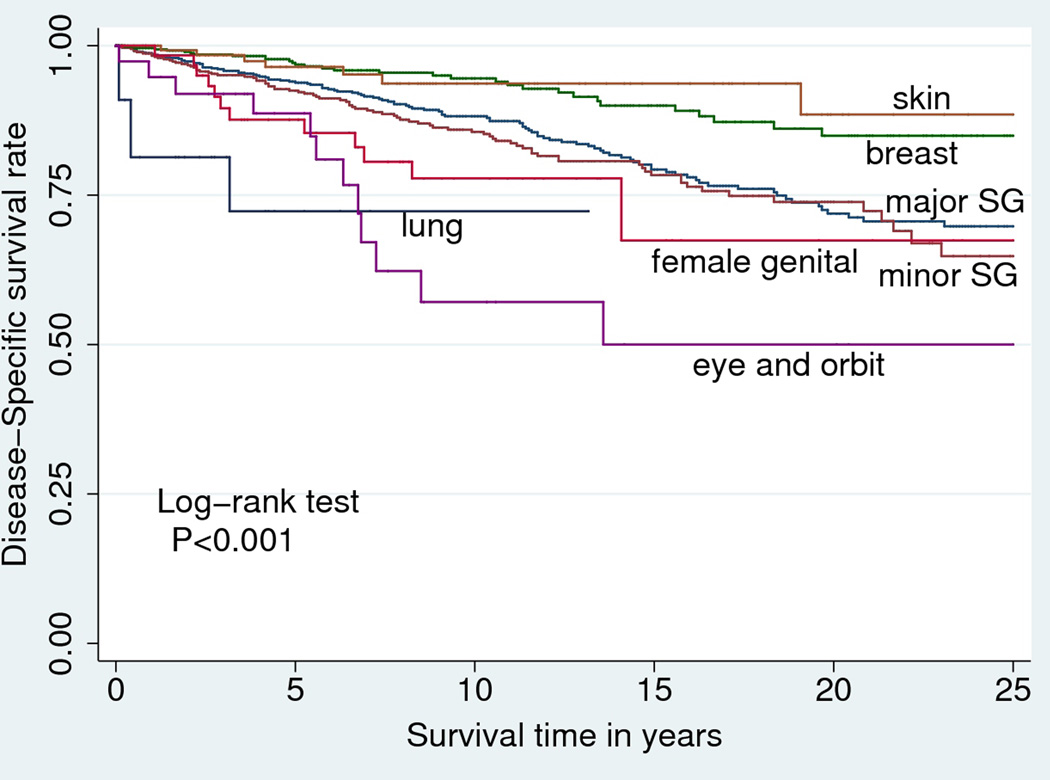
Kaplan-Meier survival curves for patients presenting with localized disease, by anatomic site.
Figure 2.

Kaplan-Meier survival curves for patients presenting with regional disease, by anatomic site.
Tables 4 and 5 show the results of univariate and multivariate Cox proportional hazards model analysis of ACCs diagnosed at localized and regional stage, respectively. Model 1 shows the hazard ratio of each site after adjustment for factors that remained in the model after stepwise selection. Model 2 is the full model, which shows the hazard ratio of each site after adjustment for year of diagnosis, age, sex, race, marital status, and treatment. Unfortunately, information on tumor size, tumor grade, and lymph node metastases was available for only a minority of patients. The crude hazard ratios and their P values (Table 4) show that localized tumors in the breast and skin were associated with approximately half the risk of death of localized tumors in the major salivary glands, while tumors in the lung and bronchus and eye and orbit were associated with 5 times and almost 3 times, respectively, the risk of death. No significant survival differences were observed between ACCs of the minor salivary glands or female genital system and ACCs of the major salivary glands. The HRs changed slightly after adjustment for other factors; however, the results were consistent with the results in the unadjusted model.
Table 4.
Survival Analysis for Patients Presenting with Localized Disease
| Site | Unadjusted Model: Crude Hazard Ratio (95% CI) |
P |
aModel 1: Hazard Ratio (95% CI) |
P |
bModel 2: Hazard Ratio (95% CI) |
P |
|---|---|---|---|---|---|---|
| Major salivary glands | 1.00 | Ref. | 1.00 | Ref. | 1.00 | Ref. |
| Minor salivary glands | 1.12 (0.85–1.49) | .415 | 0.90 (0.67–1.21) | .495 | 0.87 (0.64–1.18) | .360 |
| Breast | 0.47 (0.32–0.69) | <.001 | 0.40 (0.26–0.60) | <.001 | 0.43 (0.28–0.66) | <.001 |
| Skin | 0.46 (0.22–0.93) | .032 | 0.40 (0.19–0.82) | .013 | 0.47 (0.22–0.98) | .043 |
| Lung & bronchus | 4.89 (1.98–12.05) | .001 | 3.72 (1.50–9.19) | .005 | 3.85 (1.51–9.82) | .005 |
| Female genital system | 1.49 (0.84–2.65) | .172 | 1.05 (0.56–1.94) | .889 | 1.03 (0.54–1.96) | .925 |
| Eye & orbit | 2.60 (1.44–4.72) | .002 | 3.67 (2.02–6.68) | <.001 | 3.79 (2.06–6.95) | .001 |
CI, confidence interval; Ref., reference group.
Adjusted by age (continuous variable) and treatment.
Adjusted by year of diagnosis, age (continuous variable), sex, race, marital status, and treatment.
Table 5.
Survival Analysis for Patients Presenting with Regional Disease
| Site | Unadjusted Model: Crude Hazard Ratio (95% CI) |
P |
aModel 1: Hazard Ratio (95% CI) |
P |
bModel 2: Hazard Ratio (95% CI) |
P |
|---|---|---|---|---|---|---|
| Major salivary glands | 1.00 | Ref. | 1.00 | Ref. | 1.00 | Ref. |
| Minor salivary glands | 1.30 (1.08–1.57) | .006 | 1.08 (0.89–1.32) | .424 | 1.10 (0.90–1.34) | .344 |
| Breast | 0.85 (0.46–1.56) | .602 | 1.04 (0.54–1.99) | .915 | 1.06 (0.55–2.04) | .856 |
| Skin | 0.29 (0.12–0.70) | .006 | 0.26 (0.09–0.69) | .007 | 0.26 (0.10–0.70) | .008 |
| Lung & bronchus | 1.84 (1.12–3.04) | .016 | 1.49 (0.88–2.52) | .135 | 1.54 (0.91–2.62) | .110 |
| Female genital system | 1.76 (0.99–3.09) | .051 | 1.35 (0.72–2.55) | .352 | 1.34 (0.71–2.56) | .369 |
| Eye & orbit | 1.38 (0.75–2.54) | .301 | 1.50 (0.79–2.85) | .214 | 1.65 (0.86–3.14) | .130 |
CI, confidence interval; Ref., reference group.
Adjusted by age (continuous variable), sex, marital status, and treatment.
Adjusted by year of diagnosis, age (continuous variable), sex, race, marital status, and treatment.
Table 5 shows that among patients presenting with regional disease, patients with tumors of the skin had only one-third the risk of death of patients with ACCs of the major salivary glands, while patients with tumors of the minor salivary glands had a 30% increased risk of death and patients with tumors of the lung and bronchus had an 84% increased risk of death. After adjustment for other factors, only patients with ACC of the skin had a significantly different risk (75% reduced) compared to the risk in patients with ACC of the major salivary glands. Because of the small sample size of patients with ACC presenting with distant disease, we were not able to conduct multivariate analysis of such patients.
In an attempt to also adjust for tumor size, tumor grade, and lymph node metastases (variables unavailable for most patients), we also conducted survival analysis among patients with ACCs at the 3 most common sites: major salivary glands (reference group), minor salivary glands, and breast. During Cox proportional hazards model analysis, we took into consideration all demographics and clinical features, including tumor grade, tumor size, and lymph node metastases (nodal metastases data were excluded when we analyzed the survival of patients presenting with localized stage, i.e., all nodes negative). The effect model after stepwise selection showed that for patients with ACC presenting with localized disease, the hazard ratios for minor salivary glands and breast were 0.94 (95% CI, 0.49–1.82, P=.864) and 0.21 (95% CI, 0.09–0.51, P<.001), respectively. For patients with ACCs presenting with regional disease, the hazard ratios for minor salivary glands and breast were 0.87 (95% CI, 0.54–1.41, P=.584) and 1.29 (95% CI, 0.53–3.14, P=.574), respectively.
Discussion
Our findings show that as compared to the other 3 common histological types of salivary gland carcinoma, ACC arise at more widespread sites across the body. There are a larger proportion of ACC cases occurring at sites outside the major and minor salivary glands (especially the breast) compared with other histological types (exception being many prostate carcinomas classified as acinar cell carcinomas). The demographics, clinical features, and survival of patients with ACC differ by primary tumor site. As expected, stage (as classified by the SEER local/regional/distant staging system) appeared to be an important predictor of survival, though we were unable to assess the TNM staging system within this cohort. Furthermore, site appeared to be an important predictor of survival for patients presenting with localized ACC but was less important for patients presenting with more advanced disease.
Our analysis by site showed that ACC occurs predominantly among people over 55 years old, except the eye and orbit, for which the mean age at diagnosis was 49.9 years. An even younger mean age at diagnosis, 39.5 years, was reported in a retrospective case series of 20 patients with ACC of the lacrimal gland treated at The University of Texas MD Anderson Cancer Center between 1952 and 2002,14 consistent with results of other small retrospective case series.15–17 We also found that the proportion of patients who were African American was significantly higher among women with ACC of the genital system than among patients with ACC of other sites. Interestingly, 90% of the female genital system ACCs in African American women occurred in the cervix, compared with only 46% of those in white women. A high proportion of African American women was previously reported in a case series of cervical ACC.18 With the exception of lung and bronchus, ACC was more common in women than men for all sites. A slight male predominance in lung and bronchus ACC patients has been reported by others,19 and in most studies, a slight female predominance for ACC of other sites has also been reported.4,6,14 Because of the rarity of ACC, sample size might be an issue in the evaluation of age, race, or sex distributions, but as our study is based on a relatively large population, these demographics may be more reliable.
A European population-based study of ACCs of major and minor salivary glands also found that primary site was an important predictor of prognosis: ACCs in the nasal cavity, pharynx, and larynx were associated with an increased risk of death compared to ACCs in major salivary glands.6 In the same study, localized ACCs in the oral cavity were associated with a better outcome than localized ACCs in the major salivary glands, while for ACCs of more advanced stages, differences in survival were not apparent.6 These findings were somewhat consistent with our finding that survival differences were obvious among localized ACCs but less apparent among more advanced ACCs. Among patients with ACCs presenting with regional disease, only those with tumors of the skin had a significantly reduced risk of death. This may be an artifact of the SEER staging system, in which regional disease includes some cases with local extension without nodal metastases. As demonstrated in Table 2, ACCs of the skin and of the eye and orbit had the lowest incidences of nodal metastases; consequently, it is likely that most patients with “regional” ACC of the skin have their disease staged as regional because of local extension, which may be easier to treat surgically than regional ACC of other sites. Patients with localized ACCs at presentation generally had better survival than patients with regional or distant disease. However, the TNM staging system would permit more precise stratification of ACCs to allow comparisons. Furthermore while the TNM staging system is more commonly used among clinicians and healthcare professionals, the SEER database only provides the applicable TNM staging information for patients diagnosed after 2004, but the local/regional/distant staging information is provided for most of the patients diagnosed during the years, 1973–2008. Given that the ACC is a rare malignancy, we chose to use local/regional/distant staging system to allow the analysis performed here would have adequate sample size and power.
Regardless, our study suggested that ACCs of the breast and skin have excellent clinical behavior. At presentation, the vast majority of them were localized, smaller than 2 cm, of lower grade, and without nodal metastasis. The excellent clinical behavior of breast ACC has also been shown in a rare cancer network study from Switzerland,20 in which the median tumor size was 2.0 cm and approximately 90% of patients presented with T1 or T2 disease. Our subgroup multivariate survival analysis (including tumor size and tumor grade) suggested that patients with localized ACC of the breast have better survival than patients with localized ACC of major salivary glands even after adjustment for clinical features. It is possible that factors such as variations in anatomy, pathology, and molecular characteristics contribute to these survival differences among sites.21
Unfortunately, we could not include tumor size, tumor grade, and nodal metastasis in our multivariate analysis for other sites because the missing information decreased our sample size and power, especially in less common sites, to such a degree that analysis was meaningless. However, the results we obtained for the 3 most common sites when we included size, grade, and nodal metastasis in the analysis were concordant with the results of our more limited multivariate analysis with the full sample set, and this to a large extent confirmed the validity of our survival analysis of all 7 sites. While information on surgical margins and perineural invasion is not available in the SEER database, future SEER studies will have more information on the clinical tumor features for each case, and thus adjustment for tumor size, tumor grade, and lymph node metastases will become feasible in such site-comparative analyses.
Our survival analysis was also limited by the rather small number of ACCs of the skin, lung and bronchus, female genital system, and eye and orbit. Additionally, site and histology misclassification are possible. However, the SEER registry is considered to be the largest population-based cancer registry in the United States, and to our knowledge our present study is the largest to date of ACCs of these less common sites. Although our results should be interpreted with caution, our study is important because it presents findings for ACCs of less common sites within a population database and is the first to specifically provide insights into differences in demographics, clinical features, and survival for ACCs of different anatomic sites. Therefore, our findings can provide guidance in ACC detection, therapy, and prognosis and serve as inspiration for future studies to explore the determinants of the distinct evolution and clinical behavior of ACCs at each site.
Conclusions
The demographics and clinical features of ACC differ by anatomic site. Patients who present with localized ACCs generally have better survival than those who present with regional or distant ACCs; thus, early detection could enhance survival and possibly quality of life for ACC patients. Anatomic site may be an important predictor of survival for patients who present with localized disease but is less important for patients who present with regional disease. However, limitations in the SEER database prevent adequate control for major prognostic confounders such as grade, size, lymph node metastases, perineural invasion, and surgical margins.
Acknowledgements
The authors thank Ms. Stephanie P. Deming for manuscript editing.
Funding: This work was supported by the National Institutes of Health through grant U01 DE019765-01 (to Dr. El-Naggar; Dr. Sturgis is project 2 leader) and MD Anderson’s Cancer Center Support Grant, CA016672, and by Halliburton Employees Fellow in Cancer Prevention funds (to Dr. Xu, fellow).
Abbreviations
- ACC
adenoid cystic carcinoma
- CI
confidence interval
- DSS
disease-specific survival
- HR
hazard ratio
- SEER
Surveillance
- Epidemiology
End Results
- Sep/Div/Wid
separated, divorced, or widowed
Footnotes
Conflict of interest disclosures: The authors made no disclosures.
References
- 1.Dodd RL, Slevin NJ. Salivary gland adenoid cystic carcinoma: a review of chemotherapy and molecular therapies. Oral Oncol. 2006;42:759–769. doi: 10.1016/j.oraloncology.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 2.Bjørndal K, Krogdahl A, Therkildsen MH, et al. Salivary gland carcinoma in Denmark 1990–2005: a national study of incidence, site and histology. Results of the Danish Head and Neck Cancer Group (DAHANCA) Oral Oncol. 2011;47:677–682. doi: 10.1016/j.oraloncology.2011.04.020. [DOI] [PubMed] [Google Scholar]
- 3.Gondivkar SM, Gadbail AR, Chole R, Parikh RV. Adenoid cystic carcinoma: a rare clinical entity and literature review. Oral Oncol. 2011;47:231–236. doi: 10.1016/j.oraloncology.2011.01.009. [DOI] [PubMed] [Google Scholar]
- 4.Marchiò C, Weigelt B, Reis-Filho JS. Adenoid cystic carcinomas of the breast and salivary glands (or ‘The strange case of Dr Jekyll and Mr Hyde’ of the exocrine gland carcinomas) J Clin Pathol. 2010;63:220–228. doi: 10.1136/jcp.2009.073908. [DOI] [PubMed] [Google Scholar]
- 5.Ghabach B, Anderson WF, Curtis RE, Huycke MM, Lavigne JA, Dores GM. Adenoid cystic carcinoma of the breast in the United States (1977 to 2006): a population-based cohort study. Breast Cancer Res. 2010;12:R54. doi: 10.1186/bcr2613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ciccolallo L, Licitra L, Cantú G, Gatta G EUROCARE Working Group. Survival from salivary glands adenoid cystic carcinoma in European populations. Oral Oncol. 2009;45:669–674. doi: 10.1016/j.oraloncology.2008.10.010. [DOI] [PubMed] [Google Scholar]
- 7.Molina JR, Aubry MC, Lewis JE, et al. Primary salivary gland-type lung cancer: spectrum of clinical presentation, histopathologic and prognostic factors. Cancer. 2007;110:2253–2259. doi: 10.1002/cncr.23048. [DOI] [PubMed] [Google Scholar]
- 8.Popa I, Welt L, Constantin G, et al. Adenoid cystic carcinoma of the breast--two case reports. Chirurgia (Bucur) 2010;105:827–830. [PubMed] [Google Scholar]
- 9.Sharma K, Rathi AK, Khurana N, et al. A retrospective study of 18 cases of adenoid cystic cancer at a tertiary care centre in Delhi. Indian J Cancer. 2010;47:424–429. doi: 10.4103/0019-509X.73571. [DOI] [PubMed] [Google Scholar]
- 10.Ozdemir C, Karacetin D, Tuna S, Karadeniz A. Treatment and clinicopathologic predictors for adenoid cystic carcinomas of the head and neck. J BUON. 2011;16:123–126. [PubMed] [Google Scholar]
- 11.Lloyd S, Yu JB, Wilson LD, Decker RH. Determinants and patterns of survival in adenoid cystic carcinoma of the head and neck, including an analysis of adjuvant radiation therapy. Am J Clin Oncol. 2011;34:76–81. doi: 10.1097/COC.0b013e3181d26d45. [DOI] [PubMed] [Google Scholar]
- 12.Coates JM, Martinez SR, Bold RJ, Chen SL. Adjuvant radiation therapy is associated with improved survival for adenoid cystic carcinoma of the breast. J Surg Oncol. 2010;102:342–347. doi: 10.1002/jso.21638. [DOI] [PubMed] [Google Scholar]
- 13.SAS Institute. (2009), Inc. SAS _ Version 9.2. Cary, NC: SAS Institute Inc.; 2009. [Google Scholar]
- 14.Esmaeli B, Ahmadi MA, Youssef A, El-Naggar A. Outcomes in patients with adenoid cystic carcinoma of the lacrimal gland. Ophthal Plast Reconstr Surg. 2004;20:22–26. doi: 10.1097/01.IOP.0000105518.72611.4F. [DOI] [PubMed] [Google Scholar]
- 15.Galliani CA, Faught PR, Ellis FD. Adenoid cystic carcinoma of the lacrimal gland in a six-year-old girl. Pediatr Pathol. 1993;13:559–565. doi: 10.3109/15513819309048243. [DOI] [PubMed] [Google Scholar]
- 16.Dagher G, Anderson RL, Ossoinig KC, Baker JD. Adenoid cystic carcinoma of the lacrimal gland in a child. Arch Ophthalmol. 1980;98:1098–1100. doi: 10.1001/archopht.1980.01020031088015. [DOI] [PubMed] [Google Scholar]
- 17.Lorenz B, Stefani FH, Theopold M. [Local excision of an adenoid cystic carcinoma of the lacrimal gland in a 12-year-old girl: case report (author's transl)] Klin Monbl Augenheilkd. 1982;180:164–165. doi: 10.1055/s-2008-1055041. [DOI] [PubMed] [Google Scholar]
- 18.Ferry JA, Scully RE. “Adenoid cystic" carcinoma and adenoid basal carcinoma of the uterine cervix. A study of 28 cases. Am J Surg Pathol. 1988;12:134–144. doi: 10.1097/00000478-198802000-00007. [DOI] [PubMed] [Google Scholar]
- 19.Moran CA, Suster S, Koss MN. Primary adenoid cystic carcinoma of the lung. A clinicopathologic and immunohistochemical study of 16 cases. Cancer. 1994;73:1390–1397. doi: 10.1002/1097-0142(19940301)73:5<1390::aid-cncr2820730513>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- 20.Khanfir K, Kallel A, Villette S, Jeanneret Sozzi W. Management of adenoid cystic carcinoma of the breast: a Rare Cancer Network study. Int J Radiat Oncol Biol Phys. 2011 May 11; doi: 10.1016/j.ijrobp.2010.12.008. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 21.Lawrence JB, Mazur MT. Adenoid cystic carcinoma: a comparative pathologic study of tumors in salivary gland, breast, lung, and cervix. Hum Pathol. 1982;13:916–924. doi: 10.1016/s0046-8177(82)80052-x. [DOI] [PubMed] [Google Scholar]


