Abstract
Meiosis is a complex developmental process that generates haploid cells from diploid progenitors. We measured messenger RNA (mRNA) abundance and protein production through the yeast meiotic sporulation program and found strong, stage-specific expression for most genes, achieved through control of both mRNA levels and translational efficiency. Monitoring of protein production timing revealed uncharacterized recombination factors and extensive organellar remodeling. Meiotic translation is also shifted toward noncanonical sites, including short open reading frames (ORFs) on unannnotated transcripts and upstream regions of known transcripts (uORFs). Ribosome occupancy at near-cognate uORFs was associated with more efficient ORF translation; in contrast, some AUG uORFs, often exposed by regulated 5′ leader extensions, acted competitively. This work reveals pervasive translational control in meiosis and helps to illuminate the molecular basis of the broad restructuring of meiotic cells.
Sexual reproduction is enabled by meiosis, a strongly conserved cell division that generates haploid progeny from a diploid precursor. Meiosis has been studied for over a century including extensive analyses in the budding yeast Saccharomyces cerevisiae [reviewed in (1, 2)], where it is linked to spore formation. These efforts have provided a wealth of knowledge about the movement and changes in organization of meiotic chromosomes. Far less is known about the molecular basis of the remodeling events that impact other aspects of meiotic cellular physiology. Pioneering microarray studies (3) provided a basic framework of molecular changes accompanying yeast meiotic progression but failed to capture many dynamic processes, in part because of extensive posttranscriptional regulation, including specific instances of functionally significant translational control [reviewed in (2); see also (4)]. Whether translational control plays a general role in meiotic protein production, however, is unclear.
Ribosome profiling, based on deep sequencing of ribosome-protected mRNA fragments, allows monitoring of translation with scale, speed, and accuracy that rivals approaches for following mRNA levels (5, 6). Applying this method to sporulating S. cerevisiae cells allowed us to follow the molecular events underlying meiosis with unprecedented depth.
A high-resolution atlas of meiotic mRNA abundance and new protein synthesis
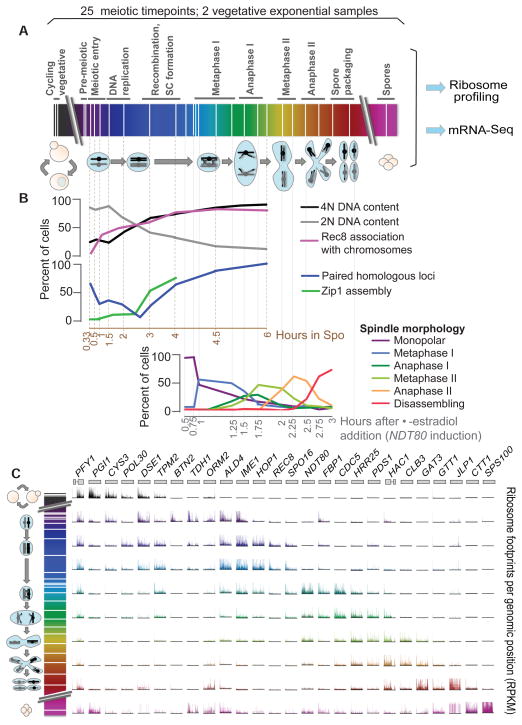
Our studies relied on three critical features: optimized meiotic synchrony, dense time points that oversampled meiotic transitions, and in-depth staging of each time point. We collected samples through two separate meiosis experiments (Fig. 1, A and B, and fig. S1A). The first used an optimized version of traditional synchronization procedures and focused on early meiotic stages. The second time course used an estrogen-activatable variant of the Ndt80 transcription factor (4, 7), which allowed synchronous progression through the meiosis I and II (MI and MII) chromosome segregation stages (4). Each time point was staged in detail (Fig. 1B and figs. S2 and S3), and we selected 25 of them, chosen for comprehensive meiotic coverage, along with two cycling vegetative samples, for ribosome profiling and mRNA sequencing (Fig. 1A and fig. S1A). Use of time points that oversampled meiotic stages allowed for synthesis of the data into a master time course (Fig. 1A and fig. S1B) and selective pooling, which collapsed meiotic progression into nine categories for some analyses (fig. S4).
Fig. 1.
Ribosome profiling through meiosis. (A) Time points (white lines) were taken through two overlapping time courses. Cartoon representations of meiotic stages are below. (B) A subset of staging controls. Positions of staging plots correspond to time points in (A). (C) Ribosome footprints across specific genes are shown for categories in fig. S4. Scales on the y axis are independent by gene.
Staging revealed a high degree of synchrony and provided a cytological framework to anchor expression data (Fig. 1B and figs. S2 and S3). Examination of ribosome footprints for specific genes showed that the sample synchrony was reflected in sharp, discrete translation patterns (Fig. 1C). The large majority (6134 out of 6708) of genes were translated at some point in meiosis, and most showed strong temporal regulation. In addition to a large shift in expression patterns between vegetative cells and cells entering meiosis, 66% of meiotically expressed genes varied by at least 10-fold in protein synthesis level through meiotic progression itself, a range that far exceeded measurement errors (Fig. 2A and fig. S5, A to D). These changes were due largely to the meiotic program itself rather than the nutrient deprivation conditions that accompany sporulation (fig. S6).
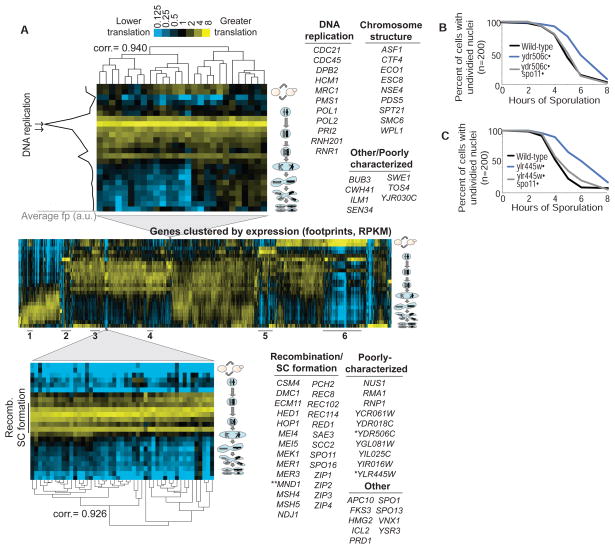
Fig. 2.
A global view of protein synthesis through sporulation. (A) Ribosome footprints (RPKMs) were summed over each yeast gene (columns) for all samples except steady-state spores (rows). The summed expression of each gene over time points was normalized for the time course, and genes were subjected to clustering. Several clusters are noted by lines below the chart: 1, Mitochondrial ribosome; 2, nutrient uptake and/or amino acid biosynthesis; 3, mitochondrial function; 4, proteasome; 5, redox and energy generation; and 6, ribosome and translation machinery. Meiotic progression is indicated pictorially to the right. (Top) A cluster containing genes responsible for DNA replication, with the gene identities to the right. To the left is the average footprint density across the cluster, with time points corresponding to bulk DNA replication represented by arrows. (Bottom) A cluster of genes associated with recombination and SC formation. The bar to the left shows the timing of these events as determined by staging controls. Single asterisks identify genes analyzed in (B) and (C) and fig. S7. Double asterisks indicate that both MND1 and the overlapping ORF, YGL182C, were identified in this cluster. (B) Wild-type, ydr506cΔ, and ydr506c spo11Δ cells were induced to sporulate. At indicated times, samples were scored for nuclear division. (C) Wild-type, ylr445wΔ, and ylr445wΔ spo11Δ cells were induced to sporulate and were treated as in (B).
Expression clustering to probe meiotic cell biology and gene function
Clustering of the time points by genome-wide protein synthesis patterns precisely recapitulated their order (figs. S1B and S6A). Thus, dynamic control of protein synthesis results in unique expression signatures throughout the meiotic program. Accordingly, grouping of all genes by protein synthesis pattern through meiosis revealed numerous multifaceted clusters (Fig. 2A).
Many clusters emerged from groups of functionally related genes. This was seen prominently for genes involved in translation, mitochondrial function, mitochondrial translation, nutrient uptake, proteasome function, and redox reactions (Fig. 2A, numbered in the middle panel, and tables S1 and S2). Furthermore, a tight cluster of 27 proteins that were synthesized at the onset of DNA replication was predominantly composed of critical DNA replication and chromosome structure factors [Fig. 2A, top, and table S2; (8)]. Similarly, genes involved in recombination and synaptonemal complex (SC) formation were expressed precisely when these processes occurred, and they emerged as a discrete group containing 46 genes from unbiased clustering of the full data set (Fig. 2A, bottom, and table S2). Notably, this cluster included the large majority of meiotic genes with characterized roles in double-strand–break formation, crossover-noncrossover choice, and SC structure [reviewed in (9–11)].
Several uncharacterized genes were found in the recombination/SC cluster, which suggested that they are involved in these intensely studied processes (1). Indeed, loss of either YDR506C or YLR445W delayed nuclear division, consistent with a role for these factors in prophase, when recombination and SC formation occur (Fig. 2, B and C, and fig. S7A). ydr506cΔ and ylr445wΔ cells showed distinct, specific defects in SC morphogenesis (fig. S7, B and C), and in both cases, the meiotic progression delay was largely alleviated when the recombination checkpoint was bypassed by deletion of SPO11 (Fig. 2, B and C, and fig. S7A). The strong delay in ylr445wΔ cells, however, was not fully dependent on Spo11 (Fig. 2C and fig. S7A), which implied that this gene has additional functions.
Evidence for cellular remodeling
Whereas our ability to observe precise temporal regulation allowed specific coclustering of some genes, there were prominent cases in which genes with a common function or localization showed highly disparate expression patterns. For example, we found tightly controlled but distinct patterns of expression among endoplasmic reticulum (ER) proteins, which suggested major ER remodeling events (fig. S8 and table S3). A strong down-regulation (relative to vegetative cells) of a set of ER genes, including ergosterol biosynthesis components, occurred before meiotic induction. After meiotic entry, a broad group of ER genes was induced, including glycosylation factors (table S3). Finally, after MI, a subset of folding factors, sphingolipid biosynthetic genes, and trafficking components were up-regulated. This last remodeling phase is accompanied by induction of the unfolded protein response (UPR) (12, 13) [Fig. 1C, see also Fig. 3F below].
Fig. 3.
Widespread dynamic translational control in meiosis. (A) Log2 mRNA and footprints (RPKM) for a region containing SPS1 and SPS2 over pooled time points (fig. S4). (B) SPS1-3HA and SPS2-FLAG cells carrying an estrogen-inducible NDT80 allele were induced to sporulate. At 6 hours, β-estradiol was added. Samples from indicated times were subjected to Western blotting (WB) and Northern blotting (NB). (C) Log2 TE values for CLB3 and YPT1 for pooled time points (fig. S4). MI and MII are indicated by colored shading as boxes. (D) Cluster analysis of log2 TE through meiosis for pooled categories (fig. S4) for all genes. (E) Log2 TEs are plotted as in (C) for AMA1, RCR1, ORC1, and ZIP1. (F) Log2 TEs are plotted as in (C) for HAC1. Below, total RNA from the original time course (see fig. S1) was subjected to Northern blotting for HAC1.
Autophagy components also showed discrete patterns of expression, which suggested dynamic control of distinct autophagic processes during sporulation [fig. S9A and table S4; (14)]. ATG8, a gene central to many branches of autophagy (15), was highly expressed from early in the meiotic program, and its deletion caused an early and profound meiotic defect (fig. S9, B and C). By contrast, ATG32, a mitophagy-specific factor (16, 17), showed low expression until the meiotic divisions (fig. S9B). Delayed onset of mitophagy may ensure full mitochondrial function, which is needed to power early meiotic stages (18). Consistently, atg32Δ cells progressed normally past prophase, but showed delayed meiotic completion (fig. S9D).
Translational control in meiosis
Control of protein production reflects both regulation of mRNA levels and the efficiency with which these messages are translated into proteins. Measuring translation rates and mRNA levels allowed us to evaluate their relative contributions. Much transcriptional regulation was observed, but translational control also regulated the magnitude and timing of protein production in meiotic cells. An example of this interplay is provided by the adjacent SPS1 and SPS2 genes (Fig. 3A). mRNA for both genes accumulated late in prophase and persisted through the meiotic divisions, consistent with their transcriptional control by NDT80 (19). By contrast, SPS1, but not SPS2, was strongly translationally regulated, delaying Sps1 protein synthesis until MII (Fig. 3, A and B).
To quantitatively evaluate the role of translational control, we calculated relative translation efficiencies [TEs; ribosome footprint RPKM/mRNA RPKM; where RPKM is reads per kilobase of coding sequence per million mapped reads, as in (6)] for messages across our time course. Replicates indicated high TE reproducibility (error <20%), which allowed sensitive measurement of dynamic translational control [fig. S5, E and F; (6)]. This approach confirmed, both in timing and degree, the strong MI-specific translational repression that regulates the B-type cyclin, CLB3 [(4), Fig. 3C]. At least 10 genes showed a pattern of translational regulation highly similar to that of CLB3—including SPS1 (Fig. 3A), GIP1, and SPO20—which, like CLB3, have known roles only late in meiosis (20–22).
Genome-wide analysis revealed that meiotic translational regulation is both pervasive and nuanced (Fig. 3D). As seen for vegetative cells (6), meiotic cells showed strong basal differences in translation rates among genes (Fig. 3D). Globally, we observed a net decrease in translation, relative to vegetative cells in their exponential growth phase, that was most pronounced at the very earliest and latest time points (fig. S10). Further, gene-specific regulation was widely used to dynamically tune gene expression. For example, 24% of genes during the “core meiotic” period showed greater than threefold TE changes, a period during which net translation capacity appears stable (fig. S10). More than 200 genes in the full time course and 66 in the core meiotic period exhibited a dynamic range in TE that was comparable to the ~10-fold changes seen for GCN4, an archetype of strong translational regulation (23).
Changes in TE frequently correlated with timing of gene function (Fig. 3E). The DNA replication factor ORC1 (24), for example, showed strong translational repression at later meiotic stages when cells do not replicate DNA. Zip1, an SC component (25), specifically showed poor translation in vegetative cells and spores, consistent with the lack of SC in either state. Chitin deposition factor Rcr1 (26) is translated efficiently only at late time points, concomitant with new cell wall generation. Finally, HAC1, the central UPR mediator (12, 13), showed transient translational activation shortly upon transfer of cells to nutrient-limited conditions, followed by a later, stronger translational activation during the meiotic divisions, as cells are synthesizing new membrane and spore walls (Fig. 3F). HAC1 is regulated translationally through cytoplasmic splicing of its message (27). Consistently, HAC1 mRNA splicing mirrored TE measurements, both in timing and degree (Fig. 3F). The UPR has been heavily studied in yeast using harsh inhibitors of ER folding (e.g., dithiothreitol). This study reveals a novel physiological setting to follow UPR induction.
Noncanonical translation
Beyond translational control of canonical open reading frames (ORFs), we also observed a shift toward noncanonical translation during the meiotic program. Whereas vegetative cells exhibited ~5% of ribosome footprints mapping outside annotated ORFs, in meiotic cells, up to ~30% of footprints mapped outside of these regions (Fig. 4A). These footprints largely mapped to discrete novel translation sites with well-defined AUG starts and stop codon stops. We systematically annotated translation units by exploiting the strong peak in ribosome density seen at translation initiation sites to identify utilized start codons [fig. S11; (6)]. This strategy was sensitive and allowed de novo identification of start codons for most known ORFs (fig. S12A) and specific, strongly enriching for ORFs initiating at AUG (fig. S13).
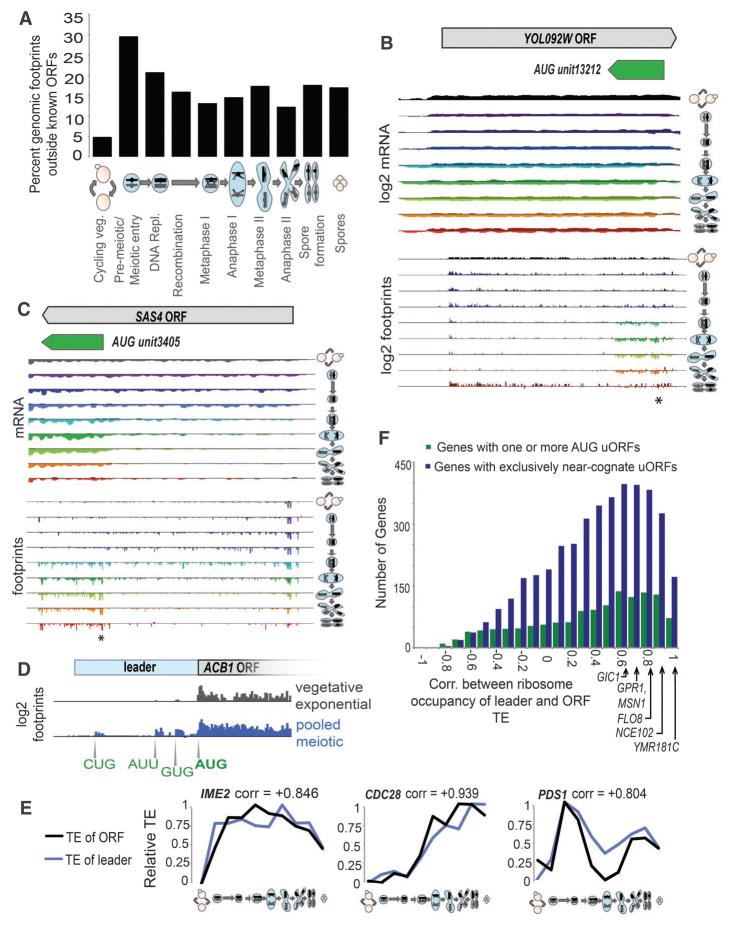
Fig. 4.
Noncanonical translation is pervasive in meiotic cells. (A) Footprints from pooled time points (see fig. S4) were mapped. The percentage of these footprints outside of known ORF annotations is plotted. (B) mRNA and ribosome occupancy profiles around YOL092W, with sense above the line for each time point and antisense below. Single asterisk denotes the sORF start site. The “AUG unit” (sORF) was annotated by the strategy shown in fig. S11. (C) The region around SAS4 is displayed as in (B), with truncated ORF start denoted by a single asterisk. (D) Ribosome occupancy profile for vegetative cells in exponential growth phase and meiotic cells over the leader of ACB1. (E) For pooled time points (see fig. S4), TEs are plotted for ORFs and for leaders (see SOM for a discussion of leader TE determination) for IME1, CDC28, and PDS1. Values are normalized to the same range for both plots. (F) Correlation coefficients [determined from plots as in (E)] were determined for each gene with uORFs for leaders with only near-cognate uORFs and at least one AUG uORF. The positions of six genes that support cap-independent translation (39) are noted.
Novel ORFs were found on noncanonical mRNAs, including transcripts antisense to known ORFs, alternate transcripts at canonical loci, and transcripts in regions thought to be intergenic (Fig. 4, B and C, and figs. S12A, S14, and S15A). We also identified instances of genome misannotation (e.g., fig. S15B). Many newly annotated ORFs were on stable transcripts, similar to those predicted as noncoding in a meiotic tiling array study [(28), fig. S14]. Our empirical strategy found translation of short ORFs (sORFs) (fig. S12A) that were well-expressed (fig. S12, B and C) and highly regulated (fig. S16) but below the cutoff of 80 to 100 amino acids used historically to computationally identify yeast ORFs.
Recent studies have identified cellular functions for short peptides (29, 30), although the function of these meiotic sORFs remains an open question. Minimally, our data suggest the export of many novel transcripts into the cytoplasm, allowing translation by ribosomes. Conversely, this data set facilitates identification of transcripts that act at the RNA level. For example, most antisense transcripts are poorly translated, including RME2 and RME3 (antisense to IME4 and ZIP2, respectively), which are known to act through direct cis-transcriptional interference of their sense counterpart [figs. S12C and S17, A, B, and D; (31, 32)]. By contrast, a transcript antisense to YFL012W that shows no transcriptional interference activity contained prominent regions of translation [(31), fig. S17, C and D].
uORFs in meiosis
The second major source of novel meiotic ribosome density was leader sequences [commonly called 5′ untranslated regions (5′UTRs)], situated upstream of the canonical ORFs (Fig. 4D and fig. S18A). We saw no general meiotic increase in footprints in 3′UTRs, which argues against a nonspecific increase in translational background noise. Examination of individual gene leaders revealed short footprint spans that started at either AUGs or near-cognate codons and generally spanned the region until the next stop codon (Fig. 4D and fig. S18A). Nearly 300 of such upstream ORFs (uORFs) have been identified in yeast under starvation conditions [(6), reviewed in (33)], but we found them to be far more common in meiosis.
We annotated 10,226 meiotic uORFs, present in the leaders of 3026 genes (fig. S11). These uORFs contained a density of ribosome footprints far greater than the ribosome footprint density in non-uORF leader regions, which suggested that our annotation approach was thorough and specific (fig. S19, A and B). Ribosome occupancy at uORFs was higher in meiotic than vegetative cells (Fig. 4D and figs. S18A and S19, B and C), and most of this effect derives from the meiotic program itself rather than the starvation conditions that accompany sporulation (fig. S19D). As expected, AUG, when present, was efficiently used for uORF translation initiation. The near-cognate codons that showed most efficient initiation, UUG and CUG (fig. S18, B and C), have also been shown to be most efficient in mammalian cells and in vitro (5, 34).
uORFs have been implicated in translational regulation, although no universal functional role has emerged. uORFs that have been well-characterized through reporter studies show diverse effects: enhancing, decreasing, or having little impact on downstream ORF translation [reviewed in (33)]. Three features of our study ideally positioned us to evaluate the role of uORFs in translation. First, we annotated many uORFs, which allowed us to distill general principles. Second, we collected data for each time point on mRNA abundance and rates of translation, which provided instantaneous quantification of TE for each downstream ORF, whereas traditional approaches require TE inference by steady-state protein abundance. Finally, our analysis of numerous sequential points through a dynamic process permitted us to detect temporal trends. Comparison of ribosome occupancy of leaders and TE of their corresponding downstream ORFs over 10 time points (see fig. S4) typically revealed a strong positive correlation (Fig. 4, E and F). However, a subset of leaders containing at least one AUG uORF showed a negative correlation, which suggested a competitive relation between uORF and ORF translation in these cases (Fig. 4F and fig. S20; see Fig. 5E below).
Fig. 5.
Regulated transcript extensions expose novel regulatory uORFs. (A) mRNA and ribosome occupancy profiles around SUP35. (B) ORC1 region displayed as in (A). (C) Total RNA from the original time course (see fig. S1) was subjected to Northern blotting for ORC1. (D) Analysis as in Fig. 4E for ORC1, SUP35, NDJ1, RED1, NDC80, and POP4. (E) Analysis as in Fig. 4F for genes with regulated leader extension (table S5).
Leader extensions and competitive uORFs
For some messages, we found that enriched footprint occupancy of leaders was caused by a programmed change in the transcript length during meiosis. Systematic analysis identified 192 genes with regulated leader length (Fig. 5, A to C; fig. S21; and table S5). For example, ORC1 showed an extended leader after prophase. This extension revealed a number of well-translated uORFs (Fig. 5B) and was accompanied by a concurrent decrease in translation of the ORC1 coding region (Figs. 3E and 5, B and D). Of genes with regulated leaders, a prominent subset showed a similar inverse relation, often corresponding well with known gene function. Orc1 and Ndj1, for example, have no characterized function late in meiosis (24, 35), and RED1, a key meiotic prophase factor (36), is translationally repressed exclusively in vegetative cells (Fig. 5D).
For genes with leader extensions containing one or more AUG uORF, at least half showed a strong negative correlation between the ribosome occupancy of the leader and TE of the ORF (Fig. 5E). By contrast, for leaders containing uORFs starting only with near-cognate, non-AUG codons, this correlation was strongly positive (Fig. 5E). Regulated leaders have been observed in budding yeast and mammalian cells, with longer forms often associated with poor ORF translation (37, 38). Here we have observed a far broader and more nuanced role for leader extensions in providing temporal translational control to many meiotic genes.
More generally, our analyses point to disparate roles for AUG and near-cognate uORFs (Figs. 4F and 5E). A fraction of AUG uORFs appear to competitively down-modulate ORF expression. By contrast, near-cognate uORFs are more common and show a generally strong positive correlation with expression of their downstream ORF, which may allow cells to divert limited resources to an important subset of messages. Whether uORFs directly prime translation of their downstream ORF is unclear. Nonetheless, genes with the strongest positive correlation between leader ribosome occupancy and ORF TE are highly enriched for known function in sporulation (table S6), which suggests physiological relevance of this regulation. The broad monitoring of gene expression by genomics has underscored the importance of quantitative modulation, beyond a model of binary on-off control. MicroRNAs provide a prominent example of developmental control through subtle regulation of broad sets of genes. uORFs may similarly allow condition-specific tuning of protein synthesis for a large portion of the genome.
The preponderance of uORFs suggests a shift of the translation initiation mechanism in meiotic cells from the predominant mechanism in which the initiation factors recognize the mRNA cap and the initiation complex scans the message for the first AUG to commence translation [reviewed in (23)]. A link between alternative translation initiation mechanisms and the use of uORFs is suggested by analysis of messages that were shown to support cap-independent translation in nitrogen-starved yeast cells (YMR181C, GPR1, BOI1, FLO8, NCE102, MSN1, and GIC1) (39). We found that all had leaders with well-translated near-cognate uORFs and a strong positive correlation between leader ribosome occupancy and ORF translation (Fig. 4F; fig. S22, excluding BOI1 as it has a complicating leader extension; table S5).
Perspective
We find that even in the extensively studied yeast, S. cerevisiae, genome coding has a complexity not captured by existing annotations. Ribosome profiling also captured a layer of regulation that is invisible to mRNA measurements and which revealed extensive and dynamic translational regulation of canonical ORFs. Transcription studies have enabled the identification of cis- and trans- transcriptional elements that control diverse cellular processes, whereas a similarly broad understanding of the importance and mechanisms of translational control remains elusive. This data set provides a valuable foundation for identifying such cis- and trans-translational regulators.
This study also gives a holistic view of the metabolic and cellular reorganization, seen through the yeast meiotic program that extends beyond a traditional chromosome-centric picture. Previous studies suggested that meiotic transcriptional control was limited to a few discrete waves (3, 40). Our data reveal multifaceted control of protein production, enabled by the tightly timed induction of many translational and transcriptional programs, including those driving translation factors, the proteasome, and the UPR. Indeed, the view of such responses as environmentally controlled stress pathways may reflect the historical context of their discovery rather than their sole physiological role.
Supplementary Material
Acknowledgments
We thank E. Ünal, A. Amon, and members of the Weissman laboratory for critical reading of this manuscript; C. Chu for sequencing assistance; J. Dunn for protocol development assistance; F. Van Werven for the MATa/a strain; and F. Klein for the the Zip1 antibody. G.A.B. is supported by American Cancer Society Postdoctoral Fellowship 117945-PF-09-136-01-RMC. M.Y. is supported by a Clore Fellowship. This work was supported by a grant from the U.S.-Israel Bi-national Science Foundation (N.F. and A.R.), an NIH P01 award (AG10770; N.T.I.), a Ruth L. Kirschstein National Research Service Award (GM080853; N.T.I.), and Howard Hughes Medical Institute funding (J.S.W. and A.R.). A patent on the ribosome profiling approach has been assigned to the University of California. Supporting files are in the SOM and at www.ncbi.nlm.nih.gov/geo/ with series accession number GSE34082.
Footnotes
Note added in proof: YDR506C and YLR445W are now named GMC1 and GMC2, respectively.
www.sciencemag.org/cgi/content/full/science.1215110/DC1
Materials and Methods
References and Notes
- 1.Zickler D, Kleckner N. Meiotic chromosomes: Integrating structure and function. Annu Rev Genet. 1999;33:603. doi: 10.1146/annurev.genet.33.1.603. [DOI] [PubMed] [Google Scholar]
- 2.Marston AL, Amon A. Meiosis: Cell-cycle controls shuffle and deal. Nat Rev Mol Cell Biol. 2004;5:983. doi: 10.1038/nrm1526. [DOI] [PubMed] [Google Scholar]
- 3.Chu S, et al. The transcriptional program of sporulation in budding yeast. Science. 1998;282:699. doi: 10.1126/science.282.5389.699. [DOI] [PubMed] [Google Scholar]
- 4.Carlile TM, Amon A. Meiosis I is established through division-specific translational control of a cyclin. Cell. 2008;133:280. doi: 10.1016/j.cell.2008.02.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ingolia NT, Lareau LF, Weissman JS. Ribosome profiling of mouse embryonic stem cells reveals the complexity and dynamics of mammalian proteomes. Cell. 2011;147:789. doi: 10.1016/j.cell.2011.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ingolia NT, Ghaemmaghami S, Newman JR, Weissman JS. Genome-wide analysis in vivo of translation with nucleotide resolution using ribosome profiling. Science. 2009;324:218. doi: 10.1126/science.1168978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Benjamin KR, Zhang C, Shokat KM, Herskowitz I. Control of landmark events in meiosis by the CDK Cdc28 and the meiosis-specific kinase Ime2. Genes Dev. 2003;17:1524. doi: 10.1101/gad.1101503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Spellman PT, et al. Comprehensive identification of cell cycle-regulated genes of the yeast Saccharomyces cerevisiae by microarray hybridization. Mol Biol Cell. 1998;9:3273. doi: 10.1091/mbc.9.12.3273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lynn A, Soucek R, Börner GV. ZMM proteins during meiosis: Crossover artists at work. Chromosome Res. 2007;15:591. doi: 10.1007/s10577-007-1150-1. [DOI] [PubMed] [Google Scholar]
- 10.Keeney S, Neale MJ. Initiation of meiotic recombination by formation of DNA double-strand breaks: Mechanism and regulation. Biochem Soc Trans. 2006;34:523. doi: 10.1042/BST0340523. [DOI] [PubMed] [Google Scholar]
- 11.Page SL, Hawley RS. The genetics and molecular biology of the synaptonemal complex. Annu Rev Cell Dev Biol. 2004;20:525. doi: 10.1146/annurev.cellbio.19.111301.155141. [DOI] [PubMed] [Google Scholar]
- 12.Cox JS, Walter P. A novel mechanism for regulating activity of a transcription factor that controls the unfolded protein response. Cell. 1996;87:391. doi: 10.1016/S0092-8674(00)81360-4. [DOI] [PubMed] [Google Scholar]
- 13.Mori K, Kawahara T, Yoshida H, Yanagi H, Yura T. Signalling from endoplasmic reticulum to nucleus: Transcription factor with a basic-leucine zipper motif is required for the unfolded protein-response pathway. Genes Cells. 1996;1:803. doi: 10.1046/j.1365-2443.1996.d01-274.x. [DOI] [PubMed] [Google Scholar]
- 14.Piekarska I, Kucharczyk R, Mickowska B, Rytka J, Rempola B. Mutants of the Saccharomyces cerevisiae VPS genes CCZ1 and YPT7 are blocked in different stages of sporulation. Eur J Cell Biol. 2010;89:780. doi: 10.1016/j.ejcb.2010.06.009. [DOI] [PubMed] [Google Scholar]
- 15.Xie Z, Klionsky DJ. Autophagosome formation: Core machinery and adaptations. Nat Cell Biol. 2007;9:1102. doi: 10.1038/ncb1007-1102. [DOI] [PubMed] [Google Scholar]
- 16.Kanki T, Wang K, Cao Y, Baba M, Klionsky DJ. Atg32 is a mitochondrial protein that confers selectivity during mitophagy. Dev Cell. 2009;17:98. doi: 10.1016/j.devcel.2009.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Okamoto K, Kondo-Okamoto N, Ohsumi Y. Mitochondria-anchored receptor Atg32 mediates degradation of mitochondria via selective autophagy. Dev Cell. 2009;17:87. doi: 10.1016/j.devcel.2009.06.013. [DOI] [PubMed] [Google Scholar]
- 18.Jambhekar A, Amon A. Control of meiosis by respiration. Curr Biol. 2008;18:969. doi: 10.1016/j.cub.2008.05.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chu S, Herskowitz I. Gametogenesis in yeast is regulated by a transcriptional cascade dependent on Ndt80. Mol Cell. 1998;1:685. doi: 10.1016/S1097-2765(00)80068-4. [DOI] [PubMed] [Google Scholar]
- 20.Neiman AM. Prospore membrane formation defines a developmentally regulated branch of the secretory pathway in yeast. J Cell Biol. 1998;140:29. doi: 10.1083/jcb.140.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tachikawa H, Bloecher A, Tatchell K, Neiman AM. A Gip1p-Glc7p phosphatase complex regulates septin organization and spore wall formation. J Cell Biol. 2001;155:797. doi: 10.1083/jcb.200107008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Friesen H, Lunz R, Doyle S, Segall J. Mutation of the SPS1-encoded protein kinase of Saccharomyces cerevisiae leads to defects in transcription and morphology during spore formation. Genes Dev. 1994;8:2162. doi: 10.1101/gad.8.18.2162. [DOI] [PubMed] [Google Scholar]
- 23.Sonenberg N, Hinnebusch AG. Regulation of translation initiation in eukaryotes: Mechanisms and biological targets. Cell. 2009;136:731. doi: 10.1016/j.cell.2009.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bell SP, Kobayashi R, Stillman B. Yeast origin recognition complex functions in transcription silencing and DNA replication. Science. 1993;262:1844. doi: 10.1126/science.8266072. [DOI] [PubMed] [Google Scholar]
- 25.Sym M, Engebrecht JA, Roeder GS. ZIP1 is a synaptonemal complex protein required for meiotic chromosome synapsis. Cell. 1993;72:365. doi: 10.1016/0092-8674(93)90114-6. [DOI] [PubMed] [Google Scholar]
- 26.Imai K, Noda Y, Adachi H, Yoda K. A novel endoplasmic reticulum membrane protein Rcr1 regulates chitin deposition in the cell wall of Saccharomyces cerevisiae. J Biol Chem. 2005;280:8275. doi: 10.1074/jbc.M409428200. [DOI] [PubMed] [Google Scholar]
- 27.Sidrauski C, Cox JS, Walter P. tRNA ligase is required for regulated mRNA splicing in the unfolded protein response. Cell. 1996;87:405. doi: 10.1016/S0092-8674(00)81361-6. [DOI] [PubMed] [Google Scholar]
- 28.Lardenois A, et al. Execution of the meiotic noncoding RNA expression program and the onset of gametogenesis in yeast require the conserved exosome subunit Rrp6. Proc Natl Acad Sci U S A. 2011;108:1058. doi: 10.1073/pnas.1016459108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kondo T, et al. Small peptides switch the transcriptional activity of Shavenbaby during Drosophila embryogenesis. Science. 2010;329:336. doi: 10.1126/science.1188158. [DOI] [PubMed] [Google Scholar]
- 30.Xu Y, Ganem D. Making sense of antisense: Seemingly noncoding RNAs antisense to the master regulator of Kaposi’s sarcoma-associated herpesvirus lytic replication do not regulate that transcript but serve as mRNAs encoding small peptides. J Virol. 2010;84:5465. doi: 10.1128/JVI.02705-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gelfand B, et al. Regulated antisense transcription controls expression of cell-type-specific genes in yeast. Mol Cell Biol. 2011;31:1701. doi: 10.1128/MCB.01071-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hongay CF, Grisafi PL, Galitski T, Fink GR. Antisense transcription controls cell fate in Saccharomyces cerevisiae. Cell. 2006;127:735. doi: 10.1016/j.cell.2006.09.038. [DOI] [PubMed] [Google Scholar]
- 33.Hood HM, Neafsey DE, Galagan J, Sachs MS. Evolutionary roles of upstream open reading frames in mediating gene regulation in fungi. Annu Rev Microbiol. 2009;63:385. doi: 10.1146/annurev.micro.62.081307.162835. [DOI] [PubMed] [Google Scholar]
- 34.Kolitz SE, Takacs JE, Lorsch JR. Kinetic and thermodynamic analysis of the role of start codon/anticodon base pairing during eukaryotic translation initiation. RNA. 2009;15:138. doi: 10.1261/rna.1318509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wu HY, Burgess SM. Two distinct surveillance mechanisms monitor meiotic chromosome metabolism in budding yeast. Curr Biol. 2006;16:2473. doi: 10.1016/j.cub.2006.10.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rockmill B, Roeder GS. RED1: A yeast gene required for the segregation of chromosomes during the reductional division of meiosis. Proc Natl Acad Sci U S A. 1988;85:6057. doi: 10.1073/pnas.85.16.6057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Badhai J, Schuster J, Gidlöf O, Dahl N. 5′UTR variants of ribosomal protein S19 transcript determine translational efficiency: Implications for Diamond-Blackfan anemia and tissue variability. PLoS ONE. 2011;6:e17672. doi: 10.1371/journal.pone.0017672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Law GL, Bickel KS, MacKay VL, Morris DR. The undertranslated transcriptome reveals widespread translational silencing by alternative 5′ transcript leaders. Genome Biol. 2005;6:R111. doi: 10.1186/gb-2005-6-13-r111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gilbert WV, Zhou K, Butler TK, Doudna JA. Cap-independent translation is required for starvation-induced differentiation in yeast. Science. 2007;317:1224. doi: 10.1126/science.1144467. [DOI] [PubMed] [Google Scholar]
- 40.Mitchell AP. Control of meiotic gene expression in Saccharomyces cerevisiae. Microbiol Rev. 1994;58:56. doi: 10.1128/mr.58.1.56-70.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Longtine MS, et al. Additional modules for versatile and economical PCR-based gene deletion and modification in Saccharomyces cerevisiae. Yeast. 1998;14:953. doi: 10.1002/(SICI)1097-0061(199807)14:10<953::AID-YEA293>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 42.Brar GA, Hochwagen A, Ee LS, Amon A. The multiple roles of cohesin in meiotic chromosome morphogenesis and pairing. Mol Biol Cell. 2009;20:1030. doi: 10.1091/mbc.E08-06-0637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Visintin R, et al. The phosphatase Cdc14 triggers mitotic exit by reversal of Cdk-dependent phosphorylation. Mol Cell. 1998;2:709. doi: 10.1016/S1097-2765(00)80286-5. [DOI] [PubMed] [Google Scholar]
- 44.Loidl J, Klein F, Engebrecht J. Genetic and morphological approaches for the analysis of meiotic chromosomes in yeast. Methods Cell Biol. 1998;53:257. doi: 10.1016/S0091-679X(08)60882-1. [DOI] [PubMed] [Google Scholar]
- 45.Nairz K, Klein F. mre11S—a yeast mutation that blocks double-strand-break processing and permits nonhomologous synapsis in meiosis. Genes Dev. 1997;11:2272. doi: 10.1101/gad.11.17.2272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yassour M, et al. Strand-specific RNA sequencing reveals extensive regulated long antisense transcripts that are conserved across yeast species. Genome Biol. 2010;11:R87. doi: 10.1186/gb-2010-11-8-r87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.de Hoon MJ, Imoto S, Nolan J, Miyano S. Open source clustering software. Bioinformatics. 2004;20:1453. doi: 10.1093/bioinformatics/bth078. [DOI] [PubMed] [Google Scholar]
- 48.Eisen MB, Spellman PT, Brown PO, Botstein D. Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci U S A. 1998;95:14863. doi: 10.1073/pnas.95.25.14863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Saldanha AJ. Java Treeview—extensible visualization of microarray data. Bioinformatics. 2004;20:3246. doi: 10.1093/bioinformatics/bth349. [DOI] [PubMed] [Google Scholar]
- 50.Homann OR, Johnson AD. MochiView: Versatile software for genome browsing and DNA motif analysis. BMC Biol. 2010;8:49. doi: 10.1186/1741-7007-8-49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yassour M, et al. Ab initio construction of a eukaryotic transcriptome by massively parallel mRNA sequencing. Proc Natl Acad Sci U S A. 2009;106:3264. doi: 10.1073/pnas.0812841106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Moll T, Tebb G, Surana U, Robitsch H, Nasmyth K. The role of phosphorylation and the CDC28 protein kinase in cell cycle-regulated nuclear import of the S. cerevisiae transcription factor SWI5. Cell. 1991;66:743. doi: 10.1016/0092-8674(91)90118-I. [DOI] [PubMed] [Google Scholar]
- 53.Cohen-Fix O, Peters JM, Kirschner MW, Koshland D. Anaphase initiation in Saccharomyces cerevisiae is controlled by the APC-dependent degradation of the anaphase inhibitor Pds1p. Genes Dev. 1996;10:3081. doi: 10.1101/gad.10.24.3081. [DOI] [PubMed] [Google Scholar]
- 54.Hochwagen A, Amon A. Checking your breaks: Surveillance mechanisms of meiotic recombination. Curr Biol. 2006;16:R217. doi: 10.1016/j.cub.2006.03.009. [DOI] [PubMed] [Google Scholar]
- 55.Keeney S, Giroux CN, Kleckner N. Meiosis-specific DNA double-strand breaks are catalyzed by Spo11, a member of a widely conserved protein family. Cell. 1997;88:375. doi: 10.1016/S0092-8674(00)81876-0. [DOI] [PubMed] [Google Scholar]
- 56.Bergerat A, et al. An atypical topoisomerase II from Archaea with implications for meiotic recombination. Nature. 1997;386:414. doi: 10.1038/386414a0. [DOI] [PubMed] [Google Scholar]
- 57.He F, et al. Genome-wide analysis of mRNAs regulated by the nonsense-mediated and 5′ to 3′ mRNA decay pathways in yeast. Mol Cell. 2003;12:1439. doi: 10.1016/S1097-2765(03)00446-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.