Abstract
Bioactive lipids such as the prostaglandins have been reported to have various cytoprotective or toxic properties in acute and chronic neurological conditions. The roles of PGF2α and its receptor (FP) are not clear in the pathogenesis of ischemic brain injury. Considering that this G-protein coupled receptor has been linked to intracellular calcium regulation, we hypothesized that its blockade would be protective. We used FP antagonist (AL-8810) and FP receptor knockout (FP−/−) mice in in vivo and in vitro stroke models. Mice that were treated with AL-8810 had 35.7 ± 6.3% less neurologic dysfunction and 36.4 ± 6.0% smaller infarct volumes than did vehicle-treated mice after 48 hours of permanent middle cerebral artery occlusion (pMCAO); FP−/− mice also had improved outcomes after pMCAO. Blockade of the FP receptor also protected against oxygen-glucose deprivation (OGD)-induced cell death and reactive oxygen species formation in slice cultures. Finally, we found that an FP receptor agonist dose dependently increased intracellular Ca2+ levels in cultured neurons and established that FP-related Ca2+ signaling is related to ryanodine receptor signaling. These results indicate that the FP receptor is involved in cerebral ischemia-induced damage and could promote development of drugs for treatment of stroke and acute neurodegenerative disorders.
Keywords: cerebral ischemia, inflammation, receptor, stroke, oxygen glucose deprivation
Introduction
We and others have described that the bioactive lipids, such as the prostaglandins, might affect neuronal outcomes after a variety of brain insults, including hypoxia, inflammation, and excitotoxicity (Dore et al., 2003). Therefore, we have focused our research on the unique properties of the prostaglandins and their receptors (Doré, 2006). Cyclooxgenases are the rate limiting enzymes for the conversion of arachidonic acid to prostaglandin PGH2, which is metabolized by specific prostaglandin synthases to at least five structurally related bioactive lipid molecules, including PGE2, PGD2, PGF2α, PGI2, and thromboxane A2 (Doré, 2006). PGF2α, which is synthesized from PGH2 via PGF synthase, plays a major role in initiation of parturition, renal function, control of cerebral blood flow autoregulation in newborn piglets (Chemtob et al., 1990), contraction of arteries, and myocardial dysfunction (Takayama et al., 2005). Recently, a report suggested that FP signaling can facilitate idiopathic pulmonary fibrosis (Oga et al., 2009). Although the critical biological functions of PGF2α were reported to be mediated through activation of the FP receptor, the effect and mechanism of FP activation are not well understood in ischemic stroke, especially in regard to stroke-related Ca2+ signaling. Based on reports of the Prostaglandin F (FP) receptor’s presence in the central nervous system and its physiologic properties in regards to stroke and Ca2+ signaling (Abramovitz et al., 1994; Kitanaka et al., 1994; Saleem et al., 2009a), we hypothesize that activation of the FP receptor subsequent to injury contributes to excitotoxic and ischemic damage. Therefore, our goal is to investigate the role of the FP receptor in the brain and in neuronal cells and to further address relevant signaling pathways.
We have established the importance of the PGE2 EP1 receptor in ischemic models (Ahmad et al., 2008). Based on its similarity to the EP1 receptor in terms of amino acid sequence and cell signaling, we hypothesize that the FP receptor might contribute to excitotoxic and ischemic brain damage similar to that caused by the EP1 receptor. We speculate that the mechanism of injury might stem from dysregulation of Ca2+ homeostasis. Therefore, we studied the role of the FP receptor in a mouse model of permanent middle cerebral artery occlusion (pMCAO) and in ischemia-related Ca2+ signaling in cultured neurons.
Materials and Methods
Animals
All animal protocols were approved by the Institutional Animal Care and Use Committee. All mice were maintained and housed in the vivarium under controlled conditions (23 ± 2°C; 12 hours light/dark periods) with access to food and water ad libitum. Adult male WT mice and FP−/− C57BL/6 mice were used at 8–10 weeks of age (20–25 g) in this study.
pMCAO, neurologic function, and infarct size determination
The pMCAO procedure was carried out as previously described (Saleem et al., 2009c). Mice were anesthetized with 250 mg/kg Avertin (Sigma), administered intraperitoneally, and a 2.0-mm burr hole was made just over the middle cerebral artery, visible through the temporal bone. The main trunk of the distal part of middle cerebral artery was occluded with a bipolar coagulator. Successful occlusion was defined as ≥80% decrease in blood flow and was confirmed by laser-Doppler flowmetry. Body temperature was maintained at 36.5–37.5°C. To evaluate neurologic deficit in the mice, we applied a piece of adhesive tape to each paw and recorded the time required for the mouse to remove each piece. A cut-off time of 120 seconds was used. Mice were trained daily before surgery and subsequently tested 24 hours before and 24 and 48 hours after surgery. After the post-surgical test, mice were sacrificed for infarct volume analysis. Brains were harvested, sliced into five 2-mm thick coronal sections, mounted onto slides, and stained with 1% triphenyltetrazolium chloride (TTC). The infarct area was translated into a percentage of the contralateral hemisphere of the same brain with correction for swelling (Saleem et al., 2009c).
Mouse hippocampal organotypic slice cultures
Hippocampi from 7–8-day-old C57BL/6 mouse pups were dissected out, and 350-µm sections were obtained with a Vibratome and stored in Hibernate solution (BrainBits) bubbled with 95% O2/5% CO2 (Ahmad et al., 2008). Slices were transferred onto membrane inserts in plates containing Neurobasal medium with B27 and GlutaMAX-I (Invitrogen). On day 13, the medium was replaced with medium containing propidium iodide (PI; 4.5 µg/mL).
Oxygen-glucose deprivation (OGD) and measurement of neuronal death and reactive oxygen species (ROS) formation
Anoxia was induced by subjecting slices to oxygen-glucose deprivation (OGD) for 1 hour in deoxygenated glucose-free Hibernate A solution and a 37°C chamber with anoxic gas (5% CO2, 95% N2) (Saleem et al., 2009b; Zhou et al., 2008). After OGD, slices were treated with 0.1−10 µM AL-8810 (Cayman Chemical Co.). Slices were imaged for PI fluorescence 24 hours after OGD. To obtain maximal PI fluorescence, they were stimulated for 24 hours with a lethal dose of NMDA (100 µM) and photographed. To measure cell death, mean PI fluorescence in the CA1 subregion of each slice was quantified. Sequential fluorescence was measured before OGD stimulation to determine basal levels of neuronal death (Fbasal); 24 hours after stimulation with OGD (FOGD); and after a final exposure to 100 µM NMDA overnight (Fmax). The percentage of cell death was calculated by the formula: (FOGD−Fbasal)/(Fmax−Fbasal) × 100%. During OGD, we exposed the slice cultures to 5 µM dihydroethidium, an indicator of ROS. After OGD, the medium was changed and slices were incubated for 2 hours. Dihydroethidium intensity in the CA1 region was then evaluated.
Neuronal cell cultures
Cerebral cortices were obtained from newborn pups (Zhen et al., In Press). The cortices were digested by 0.067% papain (Worthington) and 0.01% DNase and then triturated through pipette tips to dissociate the cells. Cells were plated onto 15-mm circular cover slips or in 24-well plates (0.5 × 106 cells per well) pre-coated with 0.01% poly-D-lysine and grown in Neurobasal medium, Glutamax, B27, and antibiotic-antimycotic solution as previously described (Saleem et al., 2008). In previous studies, our research group has demonstrated that this procedure produces cultures composed of 95% neurons and fewer than 5% astrocytes, based on MAP-2 immunostaining for neurons and GFAP staining for astrocytes (Echeverria et al., 2005). Experiments were performed after 10–15 days in vitro. For astrocyte cell cultures, the cortices were then dissociated by 0.25% trypsin+0.1% DNase digestion for 15 minutes. Cells (2X106) were plated onto 75-cm2 flasks pre-coated with 0.1% collagen in MEM medium supplemented with 10% horse serum, 2% Glutamax, and 1% antibiotic-antimycotic (Invitrogen). The cell cultures were kept at 37°C in a humidified incubator with 95% air/5% CO2. After 7–10 days, cells were subcultured into 24-well plates. Immunostaining with GFAP showed that these cultures were more than 98% astrocytes. All experiments were performed after 14–21 days in vitro.
OGD, cell viability assessment, and PGF2α ELISA in cultured cells
The cell cultures were exposed to OGD for 1 hour. First the neuronal or astrocyte cultures were rinsed with normal medium and equilibrated for 30 minutes at 37°C. Then they were rinsed with deoxygenated glucose-free MEM and transferred to an air-tight 37°C chamber. Anoxic gas (5% CO2, 95% N2) was flushed into the chamber. After 1 hour, cultures were transferred to a normal incubator for reoxygenation. Cell survival was quantified with the lactate dehydrogenase (LDH) assay kit (Sigma) as described previously with some modifications (Saleem et al., 2007). At 24 hours after OGD, cell lysate and medium were collected, and LDH assay mixture was added to each sample. Absorbance was measured at a wavelength of 490 nm with a spectrophotometer (Bio-Tek). The percent cell viability was calculated by the formula: (cell lysis LDH/(medium LDH + cell lysis LDH)) × 100%. After OGD, neuronal cultures were also double-stained with 1 µg/mL Hoechst 33342 and 2 µg/mL PI to determine living or dead cells. OGD-induced PGF2α production was determined by ELISA according to the manufacturer’s instructions (Cayman Chemical), and absorbance was measured at a wavelength of 420 nm. LDH and PGF2α ELISA experiments were repeated with three separate batches of cultures.
Ca2+ imaging
Intracellular Ca2+ imaging was performed as described in previous studies with some modifications (Kim et al., 2007). Cultured neurons were loaded with 3 µM fura-2AM in Tyrode solution [in mM: 138 NaCl, 2 CaCl2, 4 KCl, 1 MgCl2, 0.33 NaH2PO4, 10 HEPES, 10 glucose (pH=7.4)] at 37°C for 40 minutes. The excitation wavelengths were filtered through 340nm and 380-nm filters. Fluorescence intensity was collected with a cooled CCD digital camera (CoolSNAP, Photometrics) connected to a Nikon microscope. Intracellular Ca2+ concentration is presented as the ratio of collected fluorescence intensities of fura-2 excited at 340nm and 380nm, (Ratio340/380). The Ratio340/380 was recorded every 5 seconds.
Statistical analysis
Data are expressed as means ± S.E.M. Statistical analysis was performed by one-way ANOVA followed by Bonferroni post hoc test or Student’s t-test with Origin 8 (OriginLab). Significance was set at P<0.05.
Results
Blockade of FP receptor is protective in a stroke mouse model
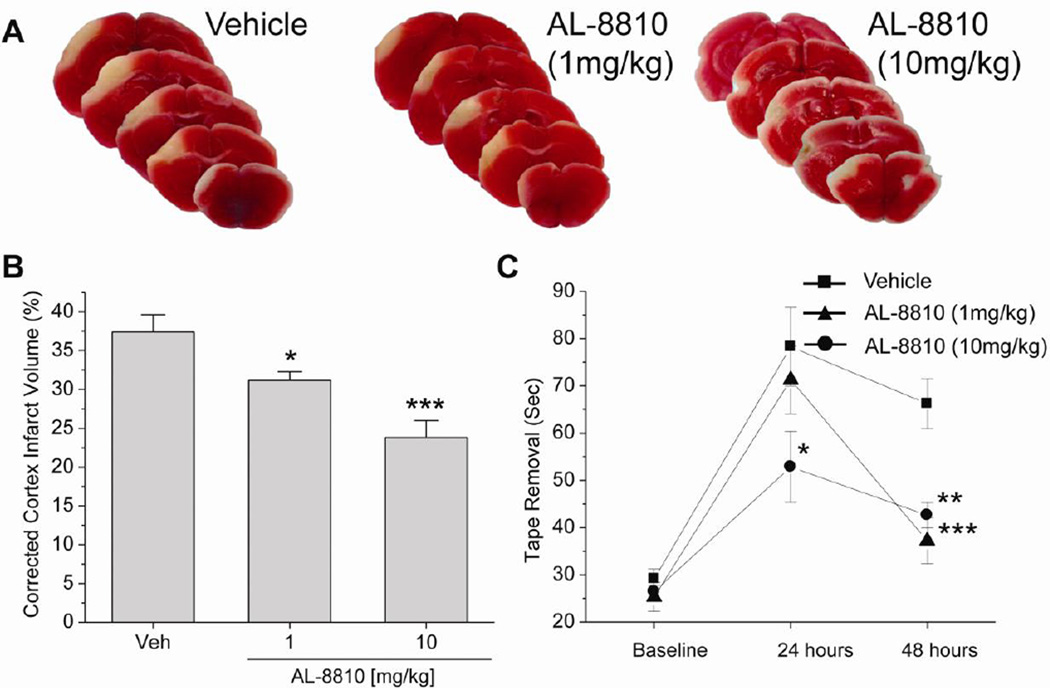
To determine the effect of FP receptor inhibition in stroke, we injected mice i.v. with 1 or 10 mg/kg of the FP antagonist AL-8810 (Cayman Chemical) immediately after pMCAO. The cortical infarct volume was significantly less in the AL-8810-treated mice than in the vehicle-treated group 48 hours after pMCAO (Fig. 1A, B; P<0.05 [1 mg/kg], P<0.001 [10 mg/kg]). Mice treated with AL-8810 also had significantly reduced tape-removal times compared with mice in the vehicle-treated group at 48 hours (Fig. 1C). Compared with vehicle, AL-8810 had no effect on body temperature, heart rate, mean blood pressure, glucose, or blood gases (pH, PaCO2, and PaO2) after 1 hour of treatment (n = 5 per group, Table 1). Similar results were observed in FP−/− mice. Genetic deletion of the FP receptor significantly reduced the cortical infarct volume at 48 hours after MCAO and enhanced the behavioral recovery after ischemia (Fig. 2). These data indicate that FP inhibition reduces anatomical and functional deficits after stroke.
Fig. 1.
Effects of FP receptor inhibition on ischemic brain injury and neurologic dysfunction after pMCAO in mice. (A) Representative photographs of infarcted brain slices from vehicle- and AL-8810-treated mice. (B) Cortical infarct volume, shown as a percent of total cortical volume and corrected for swelling, was significantly smaller in AL-8810-treated mice than in vehicle-treated mice after 48 hours. (C) The tape removal time in the sticky tape test assessed 48 hours after ischemia was significantly lower in AL-8810-treated mice than in vehicle-treated mice, indicating reduced neurologic dysfunction. *P<0.05, **P<0.01, ***P<0.001 (n = 10–13 mice).
Table 1.
Physiologic parameters in vehicle and AL-8810-treated mice (n = 5).
| Parameters | Vehicle | AL-8810 |
|---|---|---|
| Rectal temperature, °C | 36.8 ± 0.1 | 36.7 ± 0.1 |
| Heart rate, beats/min | 525 ± 20 | 529 ± 11 |
| Mean blood pressure, mm Hg | 86.2 ± 3.0 | 88.0 ± 3.5 |
| PaCO2, mm Hg | 38.9 ± 1.3 | 39.7 ± 1.6 |
| PaO2, mm Hg | 127 ± 8 | 125 ± 7 |
| pH | 7.34 ± 0.02 | 7.36 ± 0.02 |
| Glucose, mg/dL | 122 ± 9 | 126 ± 8 |
Fig. 2.
Effects of FP receptor deletion on ischemic brain injury and neurologic dysfunction after pMCAO. (A) Representative photographs of infracted brain slices. (B) Cortical infarct volume was significantly smaller in FP−/− mice than in wild-type (WT) mice after 48 hours. (C) The tape removal time was significantly lower in FP−/− mice than in WT mice at 24 and 48 hours. *P<0.05, **P<0.01 (n = 10–12 mice).
FP receptor inhibition reduces neuronal damage and ROS formation following OGD
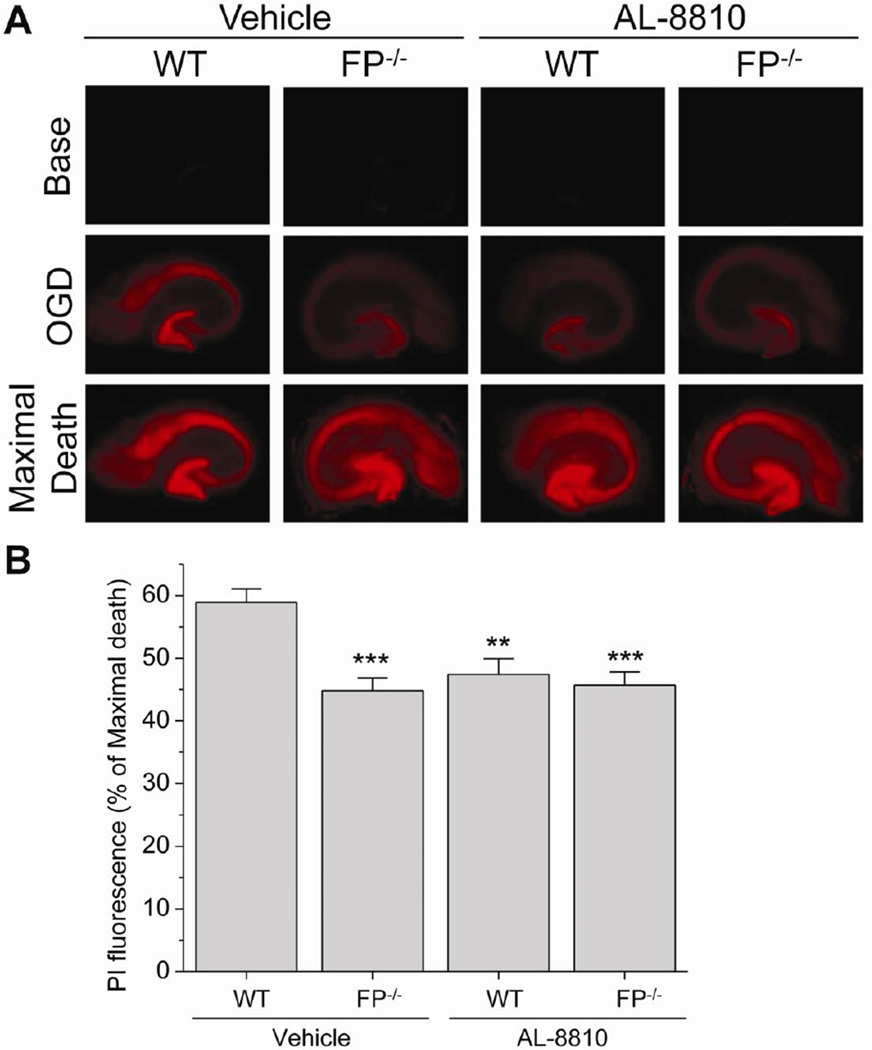
To study the effect of FP receptor in an in vitro model of ischemia, mouse hippocampal organotypic slice cultures were exposed to OGD. Cell death in the CA1 region was measured by PI labeling (Fig. 3). Slices were treated with 0.1, 1, or 10 µM AL-8810 after OGD, and PI fluorescence intensity was measured 24 hours later. Our data show that 10 µM AL-8810 reduced OGD-induced cell death compared with that of the vehicle-treated group (P<0.01; Fig. 3). Cell death after OGD was also significantly lower in slices from FP−/− mice than in those from WT mice (P<0.001; Fig. 4). We further addressed the selectivity of the antagonist in FP−/− slice cultures. OGD-induced cell death did not differ between FP−/− slice cultures treated with 10 µM AL-8810 and those treated with vehicle (Fig. 4).
Fig. 3.
Effects of FP receptor inhibition on OGD-induced cell death in cultured hippocampal slices. (A) Fluorescence images (4X magnification) show PI staining in hippocampal slices subjected to OGD for 1 hour. Images were taken before OGD (Base), 24 hours after OGD (OGD), and 24 hours after NMDA incubation (Maximal Death). (B) After OGD insult, the slice cultures were treated with AL-8810. The neuronal damage was significantly lower in 10 µM AL-8810-treated slices than in vehicle-treated slices after OGD-induced injury. **P<0.01 (n = 19–23 slices).
Fig. 4.
Effects of FP receptor deletion on OGD-induced cell death in cultured hippocampal slices. (A) Fluorescence images show PI staining in hippocampal slices subjected to OGD. (B) After OGD, neuronal cell death in the CA1 region measured as PI intensity was significantly lower in hippocampal slices from FP−/− mice than in those from WT mice. Cell death in cultured FP−/− slices was not changed by 10 µM AL-8810 compared with vehicle treatment. **P<0.01, ***P<0.001 (n = 21–23 slices).
To determine which cell type(s) OGD affects, we studied neuronal and astrocyte cultures individually. OGD significantly increased cell death in neuronal cell cultures (P<0.001, Fig. 5A), but not in astrocyte cell cultures (data not shown). Treatment with 10 µM AL-8810 significantly reduced neuronal cell death compared with vehicle treatment after OGD-induced injury (P<0.05, Fig. 5A). To show the living or dead neuronal cells, the culture neurons were double stained with the Hoechst 33342 and PI after OGD (Fig. 5B). The numbers of PI positive neurons were decreased by treatment with AL-8810 (Fig. 5B). PGF2α production was significantly increased after OGD in cultured neurons (P<0.05, P<0.01, Fig. 5C), but not in cultured astrocytes (data not shown). In addition, OGD-induced PGF2α production was significantly reduced by an NMDA receptor inhibitor (10 µM MK-801 in cultured neurons), but not by an AMPA receptor inhibitor (10 µM NBQX) (P<0.05, Fig. 5D). Examined together, these results indicate that the FP receptor contributes to acute ischemia-induced neuronal death.
Fig. 5.
OGD-induced cell death and PGF2α production in cultured neurons. Cultured neurons and astrocytes were subjected to OGD for 1 hour; 24 hours later, the LDH assay was performed to assess cell death. (A) Cell death was significantly higher in neuronal cell cultures exposed to OGD than in those exposed to normal growing conditions (control). Neuronal cell death was significantly lower in cultures treated with 10 µM AL-8810 than in vehicle-treated cultures. ***P<0.001 vs. control, #P<0.05 vs. vehicle-treated group. (B) Representative images of the Hoechst 33342 and PI double staining (a: phase contrast, b: sham, c: OGD+vehicle, d: OGD+10 µM AL8810, Bar = 50 µm). Images display typical results from three independent experiments. (C) To study the effect of ischemia on PGF2α production, cultured neurons and astrocytes were subjected to OGD for 1 hour. In cultured neurons, PGF2α production was significantly increased at 2 hours and 6 hours after OGD. *P<0.05, **P<0.01. (D) OGD-induced PGF2α production was significantly decreased by 10 µM MK-801, but not by 10 µM NBQX. *P<0.05.
After stroke, increased glutamate release, intracellular Ca2+ accumulation, and formation of ROS lead to oxidative stress and cell death. Therefore, we examined ROS formation and oxidative injury after OGD. During OGD, we treated the tissue slices with an ROS indicator, dihydroethidium. Dihydroethidium intensity data indicate that 10 µM AL-8810 significantly reduced OGD-induced ROS formation in hippocampal slice cultures (P<0.001; Fig. 6A). Slices from FP−/− mice also had reduced OGD-induced ROS formation compared with those from WT mice. OGD-induced ROS formation was unchanged in AL-8810-treated FP−/− slices compared with the vehicle-treated FP−/− group (Fig. 6).
Fig. 6.
Effects of FP receptor inhibition on OGD-induced ROS formation in hippocampal slices. (A) Dihydroethidium (DHE) fluorescence revealed that AL-8810 (10 µM) significantly reduced OGD-induced ROS formation in hippocampal slice cultures. (B) OGD-induced ROS formation was significantly lower in hippocampal slices from FP−/− mice than in those from WT mice. AL-8810 had no additional effect on slices from FP−/− mice. ***P<0.001 (n = 20–22 slices).
FP receptor mechanism in stroke-related Ca2+ signaling
Next, we investigated the FP-associated Ca2+ response in cortical neuronal cell cultures. When cultured neurons were exposed to FP receptor agonist fluprostenol (Cayman Chemical) for 1 minute, [Ca2+]i increased in a concentration-dependent manner (Fig. 7A, B). Pretreatment with 10 µM of the inhibitor AL-8810 blocked this response (P<0.01; Fig. 7C, D). We studied the relationship between the FP receptor and the ryanodine receptor. When cells were perfused with 50 µM ryanodine, a ryanodine receptor antagonist, the FP agonist-induced Ca2+ response was almost entirely blocked (P<0.001; Fig. 7C, D). In addition, we treated cultured neurons with 1 µM fluprostenol and measured cell death after 24 hours. Fluprostenol increased the neuronal cell death (P<0.01) and AL-8810 reduced it significantly (P70003C;0.01; Fig. 7E). Finally, to study OGD-induced Ca2+ increase and the effect of FP inhibition, we compared the Ca2+ level before and after OGD. OGD-induced Ca2+ increase was significantly reduced by treatment with AL-8810 (P<0.01; Fig. 7F).
Fig. 7.
Effects of FP receptor inhibition and ryanodine receptor inhibition on FP agonist-induced Ca2+ response in cultured neurons. (A) Ca2+ response was evoked by 1-minute exposure to 1, 10, 100, and 1000 nM fluprostenol--an FP receptor agonist. (B) Mean change in Ratio340/380 (n = 17–19 cells). (C) Cultured neurons were superfused with 10 µM AL-8810 or 50 µM ryanodine solution for 10 minutes before treatment of 100 nM fluprostenol. Pretreatment with 10 µM AL-8810 almost completely inhibited the FP agonist-induced Ca2+ response. The fluprostenol-induced Ca2+ response was also inhibited by pretreatment with 50 µM ryanodine, a ryanodine receptor antagonist. (D) Mean change in Ratio340/380; ***P<0.001 (n = 18–19 cells). (E) Treatment with 1 µM fluprostenol increases the neuronal cell death and 10 µM AL-8810 significantly attenuated the induced neuronal cell death. **P<0.01 vs. control, #P<0.05 vs. vehicle-treated group. (F) OGD-induced Ca2+ response was significantly reduced by 10 µM AL-8810 **P<0.01 (n = 17–18 cells).
Discussion
In our previous study, the FP receptor significantly reduced infarct volume in a transient MCAO mouse model (Saleem et al., 2009a). In this study, we showed that the inhibitor of the FP receptor significantly improved the outcome in mice after ischemia in terms of neurobehavioral function and infarct volume. In a previous study, OGD resulted in expression of apoptotic and necrotic cell death phenotypes, especially in neurons (Newcomb-Fernandez et al., 2001). We also demonstrated that FP receptor inhibition was protective against OGD-induced cell death and ROS formation in hippocampal slice cultures. Examined together, our results indicate that FP inhibition may be beneficial in protecting neurons from stroke- and neurotoxicity-induced insults.
Stroke is now ranked as the number 4 cause of death in the United States, desperately needing novel and effective treatments. Many pathophysiologic mechanisms are activated after stroke and brain injury (Moskowitz et al., 2010). These mechanisms are divided into glutamate-induced excitotoxicity, formation of ROS, and apoptotic-like cell death pathways (Besancon et al., 2008). Ca2+ dysregulation links many of these multifactorial signals (Berliocchi et al., 2005). Therefore, blockade of Ca2+ imbalance-related receptors, transporters, and intracellular signals reveals neuroprotective targets for stroke and neurodegeneration (Simard et al., 2006). After a stroke, loss of cerebral blood flow rapidly triggers energy deficits and neuronal depolarization that cause the release of large amounts of glutamate into the extracellular space. Over-activation of NMDA and AMPA receptors subsequently cause a generalized ionic imbalance within neurons, especially of Ca2+. The Ca2+ overload is understood to induce a wide range of cell death enzymes, including ATPases, proteases, lipases, and DNAses (Besancon et al., 2008). Furthermore, during stroke, the excess glutamate and hyperactivation of its receptors result in intracellular activation of phospholipid enzymes, phospholipid hydrolysis, and arachidonic acid release (Muralikrishna Adibhatla and Hatcher, 2006). Although blockade of glutamate receptors has been shown to be neuroprotective in preclinical studies, in clinical trials the results have been disappointing (Wahlgren and Ahmed, 2004). Studies have consistently shown that the NMDA-induced Ca2+ imbalance is related to a failure to remove Ca2+ from the cell (Bano and Nicotera, 2007; Kawano et al., 2006). After cerebral ischemia, the loss of Na+/K+-ATPase activity in the plasma membrane can degrade the transmembrane Na+ gradients necessary for Na+/Ca2+ exchange function and lead to Ca2+ accumulation.
Reports have suggested that prostaglandins perform diverse functions in neurodegenerative disorders through their G-protein–coupled receptors (Doré, 2006). The FP receptor mRNA is expressed in rat whole brain (Kitanaka et al., 1994), and it has been reported that the FP receptor is expressed in cortical cultures enriched in neurons (Carlson, 2003). Activation of the FP receptor initiates several events, including stimulation of the inositol 1,4,5-trisphosphate receptor-related Ca2+ signaling pathway and store-operated Ca2+ entry (Snetkov et al., 2006). One study demonstrated that FP receptor activation by PGF2α in rodent cardiomyocytes increased the expression of early growth response factor-1 by activation of a Ras/Raf/mitogen-activated protein kinase signaling cascade (Xu et al., 2008). The amino acid sequence of the FP receptor has high sequence homology with that of the EP1 receptor, and the FP receptor shares the same branch of the phylogenetic tree with the EP1 and TP receptors. Interestingly, we have previously shown that the EP1 receptor in mice augments brain damage after excitotoxicity and focal cerebral ischemia (Ahmad et al., 2008). Although activation of the FP receptor initiates several events, including Ca2+ signaling, its role is not clear in stroke-related Ca2+ signaling. As previously described, activated EP1 can reduce Na+/Ca2+ exchange activity and cause dysregulation of Ca2+ homeostasis following ischemia (Kawano et al., 2006). Therefore, we also studied NMDA-induced Ca2+ response to determine the mechanism of FP receptor activity in NMDA neurotoxicity. NMDA-induced Ca2+ responses were not different between cultured neurons from WT mice and those from FP−/− mice (data not shown). However, we demonstrated that OGD increases PGF2α production, that activation of the FP receptor induces a Ca2+ response in cultured neurons, and that this response is connected to the ryanodine receptor pathway. In addition, the treatment of FP receptor agonist in cultured neurons increases cell death and FP receptor blockade decreases OGD-induced Ca2+ response. These data indicate that the protective effect of FP receptor inhibition and its mechanism in stroke-related Ca2+ signaling differs from that of the EP1 receptor-related Ca2+ pathway. In addition, there is direct evidence of endoplasmic reticulum (ER) stress after brain ischemia/reperfusion (Hayashi et al., 2005). Disruption of ER Ca2+ homeostasis plays an important role in induction of ER stress. Therefore, the FP receptor may affect ER Ca2+ homeostasis via ryanodine receptor signaling with ER Ca2+ release, a possibility that requires further study.
Ischemia leads to the activation of ROS-generating enzymatic systems (Moskowitz et al., 2010). The increase in [Ca2+]i promotes ROS formation by the protein kinase C pathway, nitric oxide pathway, and mitochondrial depolarization in ischemia (Brennan et al., 2009). We found that OGD-induced ROS formation was decreased by FP receptor depletion or FP receptor inhibitor. The results indicate that inhibiting the FP receptor can protect neurons in early periods of ischemia by reducing Ca2+ overload and ROS formation.
Conclusion
In this study, we used AL-8810 to evaluate the role of FP receptor in ischemic damage. AL-8810 reduced OGD-induced neuronal cell death in slice cultures from WT mice but had no effect on cell death or ROS production in slices from FP−/− mice, indicating that the drug is selective for FP. However, potent and selective FP antagonists should be developed for more pharmacologic studies and therapeutic applications. Our findings provide novel targets for intervention and treatment of cerebral ischemia and help to elucidate the therapeutic potential and role of the FP receptor in acute stroke.
Highlights.
Infarct volumes and behavioral deficits were reduced in FP antagonist AL8810 mice
Similar results in FP knockout mice after permanent ischemic stroke
FP receptor blockade protects against oxygen glucose deprivation cell death in organotypic slices
Neuronal death is less in cells treated with AL8810 not in cultured astrocytes
FP receptor agonist increases intracellular calcium levels of cultured neurons
Acknowledgements
The authors thank all Doré lab members for their insightful contributions for this manuscript. This research was supported by grants from the National Institutes of Health NS046400 and AG022971 (SD), a fellowship from the Korea Research Foundation, KRF-2007-357-E00016, and by a postdoctoral grant from the American Heart Association, 09POST2060557 (YTK).
Abbreviations
- Ca2+
calcium
- COX
cyclooxygenase
- FP receptor
PGF2αreceptor
- FP−/−
FP receptor knockout
- pMCAO
permanent middle cerebral artery occlusion
- OGD
oxygen-glucose deprivation
- WT
wildtype
- HBSS
Hanks’ buffered saline solution
- LDH
lactate dehydrogenase
- ER
endoplasmic reticulum.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosures
None.
References
- Abramovitz M, Boie Y, Nguyen T, Rushmore TH, Bayne MA, Metters KM, Slipetz DM, Grygorczyk R. Cloning and expression of a cDNA for the human prostanoid FP receptor. J Biol Chem. 1994;269:2632–2636. [PubMed] [Google Scholar]
- Ahmad AS, Kim YT, Ahmad M, Maruyama T, Doré S. Selective blockade of PGE2 EP1 receptor protects brain against experimental ischemia and excitotoxicity, and hippocampal slice cultures against oxygen-glucose deprivation. Neurotox Res. 2008;14:343–351. doi: 10.1007/BF03033858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bano D, Nicotera P. Ca2+ signals and neuronal death in brain ischemia. Stroke. 2007;38:674–676. doi: 10.1161/01.STR.0000256294.46009.29. [DOI] [PubMed] [Google Scholar]
- Berliocchi L, Bano D, Nicotera P. Ca2+ signals and death programmes in neurons. Philos Trans R Soc Lond B Biol Sci. 2005;360:2255–2258. doi: 10.1098/rstb.2005.1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Besancon E, Guo S, Lok J, Tymianski M, Lo EH. Beyond NMDA and AMPA glutamate receptors: emerging mechanisms for ionic imbalance and cell death in stroke. Trends Pharmacol Sci. 2008;29:268–275. doi: 10.1016/j.tips.2008.02.003. [DOI] [PubMed] [Google Scholar]
- Brennan AM, Suh SW, Won SJ, Narasimhan P, Kauppinen TM, Lee H, Edling Y, Chan PH, Swanson RA. NADPH oxidase is the primary source of superoxide induced by NMDA receptor activation. Nat Neurosci. 2009;12:857–863. doi: 10.1038/nn.2334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlson NG. Neuroprotection of cultured cortical neurons mediated by the cyclooxygenase-2 inhibitor APHS can be reversed by a prostanoid. J Neurosci Res. 2003;71:79–88. doi: 10.1002/jnr.10465. [DOI] [PubMed] [Google Scholar]
- Chemtob S, Beharry K, Rex J, Varma DR, Aranda JV. Prostanoids determine the range of cerebral blood flow autoregulation of newborn piglets. Stroke. 1990;21:777–784. doi: 10.1161/01.str.21.5.777. [DOI] [PubMed] [Google Scholar]
- Doré S. GPCR antagonists as an alternative to COX-2 inhibitors: a case for the PGE2 EP1 receptor. Trends Pharmacol Sci. 2006;27:458–460. doi: 10.1016/j.tips.2006.07.001. [DOI] [PubMed] [Google Scholar]
- Dore S, Otsuka T, Mito T, Sugo N, Hand T, Wu L, Hurn PD, Traystman RJ, Andreasson K. Neuronal overexpression of cyclooxygenase-2 increases cerebral infarction. Ann Neurol. 2003;54:155–162. doi: 10.1002/ana.10612. [DOI] [PubMed] [Google Scholar]
- Echeverria V, Greenberg DL, Dore S. Expression of prostaglandin E2 synthases in mouse postnatal cortical neurons. Ann N Y Acad Sci. 2005;1053:460–471. doi: 10.1111/j.1749-6632.2005.tb00056.x. [DOI] [PubMed] [Google Scholar]
- Hayashi T, Saito A, Okuno S, Ferrand-Drake M, Dodd RL, Chan PH. Damage to the endoplasmic reticulum and activation of apoptotic machinery by oxidative stress in ischemic neurons. J Cereb Blood Flow Metab. 2005;25:41–53. doi: 10.1038/sj.jcbfm.9600005. [DOI] [PubMed] [Google Scholar]
- Kawano T, Anrather J, Zhou P, Park L, Wang G, Frys KA, Kunz A, Cho S, Orio M, Iadecola C. Prostaglandin E2 EP1 receptors: downstream effectors of COX-2 neurotoxicity. Nat Med. 2006;12:225–229. doi: 10.1038/nm1362. [DOI] [PubMed] [Google Scholar]
- Kim YT, Namkung YL, Kwak J, Suh CK. Involvement of Na+-Ca2+ exchanger on metabotropic glutamate receptor 1-mediated [Ca2+]i transients in rat cerebellar Purkinje neurons. Neuroscience. 2007;146:170–177. doi: 10.1016/j.neuroscience.2007.01.040. [DOI] [PubMed] [Google Scholar]
- Kitanaka J, Hasimoto H, Sugimoto Y, Negishi M, Aino H, Gotoh M, Ichikawa A, Baba A. Cloning and expression of a cDNA for rat prostaglandin F2 alpha receptor. Prostaglandins. 1994;48:31–41. doi: 10.1016/0090-6980(94)90094-9. [DOI] [PubMed] [Google Scholar]
- Moskowitz MA, Lo EH, Iadecola C. The science of stroke: mechanisms in search of treatments. Neuron. 2010;67:181–198. doi: 10.1016/j.neuron.2010.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muralikrishna Adibhatla R, Hatcher JF. Phospholipase A2, reactive oxygen species, and lipid peroxidation in cerebral ischemia. Free Radic Biol Med. 2006;40:376–387. doi: 10.1016/j.freeradbiomed.2005.08.044. [DOI] [PubMed] [Google Scholar]
- Newcomb-Fernandez JK, Zhao X, Pike BR, Wang KK, Kampfl A, Beer R, DeFord SM, Hayes RL. Concurrent assessment of calpain and caspase-3 activation after oxygen-glucose deprivation in primary septo-hippocampal cultures. J Cereb Blood Flow Metab. 2001;21:1281–1294. doi: 10.1097/00004647-200111000-00004. [DOI] [PubMed] [Google Scholar]
- Oga T, Matsuoka T, Yao C, Nonomura K, Kitaoka S, Sakata D, Kita Y, Tanizawa K, Taguchi Y, Chin K, Mishima M, Shimizu T, Narumiya S. Prostaglandin F(2alpha) receptor signaling facilitates bleomycin-induced pulmonary fibrosis independently of transforming growth factor-beta. Nat Med. 2009;15:1426–1430. doi: 10.1038/nm.2066. [DOI] [PubMed] [Google Scholar]
- Saleem S, Ahmad AS, Maruyama T, Narumiya S, Dore S. PGF(2alpha) FP receptor contributes to brain damage following transient focal brain ischemia. Neurotox Res. 2009a;15:62–70. doi: 10.1007/s12640-009-9007-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saleem S, Kim YT, Maruyama T, Narumiya S, Doré S. Reduced acute brain injury in PGE2 EP3 receptor-deficient mice after cerebral ischemia. J Neuroimmunol. 2009b;208:87–93. doi: 10.1016/j.jneuroim.2009.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saleem S, Li RC, Wei G, Dore S. Effects of EP1 receptor on cerebral blood flow in the middle cerebral artery occlusion model of stroke in mice. J Neurosci Res. 2007;85:2433–2440. doi: 10.1002/jnr.21399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saleem S, Shah ZA, Urade Y, Doré S. Lipocalin-prostaglandin D synthase is a critical beneficial factor in transient and permanent focal cerebral ischemia. Neuroscience. 2009c;160:248–254. doi: 10.1016/j.neuroscience.2009.02.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saleem S, Zhuang H, Biswal S, Christen Y, Dore S. Ginkgo biloba extract neuroprotective action is dependent on heme oxygenase 1 in ischemic reperfusion brain injury. Stroke. 2008;39:3389–3396. doi: 10.1161/STROKEAHA.108.523480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simard JM, Chen M, Tarasov KV, Bhatta S, Ivanova S, Melnitchenko L, Tsymbalyuk N, West GA, Gerzanich V. Newly expressed SUR1-regulated NC(Ca-ATP) channel mediates cerebral edema after ischemic stroke. Nat Med. 2006;12:433–440. doi: 10.1038/nm1390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snetkov VA, Knock GA, Baxter L, Thomas GD, Ward JP, Aaronson PI. Mechanisms of the prostaglandin F2alpha-induced rise in [Ca2+]i in rat intrapulmonary arteries. J Physiol. 2006;571:147–163. doi: 10.1113/jphysiol.2005.101394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takayama K, Yuhki K, Ono K, Fujino T, Hara A, Yamada T, Kuriyama S, Karibe H, Okada Y, Takahata O, Taniguchi T, Iijima T, Iwasaki H, Narumiya S, Ushikubi F. Thromboxane A2 and prostaglandin F2alpha mediate inflammatory tachycardia. Nat Med. 2005;11:562–566. doi: 10.1038/nm1231. [DOI] [PubMed] [Google Scholar]
- Wahlgren NG, Ahmed N. Neuroprotection in cerebral ischaemia: facts and fancies--the need for new approaches. Cerebrovasc Dis. 2004;17(Suppl 1):153–166. doi: 10.1159/000074808. [DOI] [PubMed] [Google Scholar]
- Xu W, Chou CL, Sun H, Fujino H, Chen QM, Regan JW. FP prostanoid receptor-mediated induction of the expression of early growth response factor-1 by activation of a Ras/Raf/mitogen-activated protein kinase signaling cascade. Mol Pharmacol. 2008;73:111–118. doi: 10.1124/mol.107.038778. [DOI] [PubMed] [Google Scholar]
- Zhen G, Kim YT, Li RC, Yocum J, Kapoor N, Langer J, Dobrowolski P, Maruyama T, Narumiya S, Dore S. PGE(2) EP1 receptor exacerbated neurotoxicity in a mouse model of cerebral ischemia and Alzheimer's disease. Neurobiol Aging. doi: 10.1016/j.neurobiolaging.2011.09.017. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P, Qian L, Chou T, Iadecola C. Neuroprotection by PGE2 receptor EP1 inhibition involves the PTEN/AKT pathway. Neurobiol Dis. 2008;29:543–551. doi: 10.1016/j.nbd.2007.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]