Abstract
Background
Asthma and chronic obstructive pulmonary disease (COPD) display features of overlap in airway physiology and airway inflammation. Whether inflammatory phenotypes in airway disease describe similar mediator expression is unknown.
Objectives
To explore the relationship of airway inflammation and cytokine and chemokine expression in asthma and COPD.
Methods
Subjects with asthma and COPD (n = 54 and n = 49) were studied. Clinical characteristics and sputum were collected at entry into the study. A 2-step sputum processing method was performed for supernatant and cytospin preparation. Meso Scale Discovery and Luminex platforms were used to measure cytokines, chemokines and matrix metalloproteinase levels.
Results
Analytes sensitive to dithiothreitol (DTT) that had increased recovery in the 2-step sputum process were IL-1β, 4, 5, 10, 13, IFN-γ, TNFRI, GM-CSF, CCL2, 3, 4, 5, 13 and 17. There was a differential expression in IL-8, TNFRI and TNFRII between asthma and COPD [mean fold difference (95% CI): IL-8, 2.6 (1.3–5.4), p = 0.01; TNFRI, 2.1 (1.3–5.4), p = 0.03; TNFRII, 2.6 (1.2–5.6), p = 0.02]. In neutrophilic and eosinophilic airway inflammation, TNFα, TNFRI, TNFRII, IL-6, IL-8 and IL-5 could differentiate between these phenotypes. However, these phenotypes were unrelated to the diagnosis of asthma or COPD.
Conclusion
Recovery of sputum mediators sensitive to DTT can be improved using the described sputum processing technique. Within airway inflammatory sub-phenotypes there is a differential pattern of mediator expression that is independent of disease. Whether these inflammatory phenotypes in asthma and COPD confer distinct pathogeneses, therapeutic responses and clinical phenotypes needs to be further evaluated.
Key Words: COPD, Asthma, Airway inflammation, Cytokines
Introduction
Chronic obstructive pulmonary disease (COPD) and asthma represent a significant challenge to the respiratory physician in their management and diagnosis, with significant overlap in their clinical features, physiological parameters and airway inflammation [1, 2]. COPD is characterised by chronic irreversible airflow obstruction, structural changes such as small airway destruction, fibrosis and neutrophilic airway inflammation [3, 4, 5, 6]. However, up to 40% of COPD subjects display eosinophilic airway inflammation [7] which is associated with elevated levels of interleukin (IL)-5. Treatment with corticosteroid therapy reduces eosinophilic inflammation and IL-5 levels [8]. In contrast, asthma is characterised by variable airflow obstruction, airway hyper-responsiveness, a thickened basement membrane and eosinophilic airway inflammation [4]. Both non-eosinophilic and neutrophilic asthma have been described and these are hypothesised to confer steroid resistance [9, 10]. The use of induced sputum has led to further insights into the pulmonary inflammation of asthma and COPD [11, 12], allowing characterisations of pro-inflammatory cytokines and chemokines. Importantly, COPD and asthma have often been studied independently using limited mediator panels [8, 13]. In light of the increasing recognition of the heterogeneity of asthma and COPD and their potential overlap [2, 14], it is important to investigate whether there are different pulmonary mediator responses and whether airway inflammatory phenotypes confer similar cytokine and chemokine expression in both asthma and COPD.
In this study we sought to investigate the sputum cytokine and chemokine pattern in subjects with asthma and COPD using a novel sputum processing method and to compare the inflammatory response within and between diseases and in relation to sputum phenotypes.
Methods
Design
This was a prospective study examining sputum mediators of subjects with asthma and COPD. The study was approved by the Leicestershire, Rutland and Northamptonshire ethics committee, and all subjects gave informed written consent (REC 08/H0401/88).
Subjects
Subjects with a physician diagnosis of asthma or COPD attending the respiratory clinics at Glenfield Hospital, Leicester, UK, were invited to take part in the study. Subjects with a diagnosis of active pulmonary tuberculosis, other clinically relevant lung or medical illnesses or the inability to produce sputum following the induced sputum procedure were excluded.
All COPD subjects had airflow obstruction with a post-bronchodilator forced expiratory volume in 1 s (FEV1)/forced vital capacity (FVC) ratio (FEV1/FVC) of less than 0.7, with which severity was classified according to Global Initiative for Obstructive Lung Disease (GOLD) criteria [6]. COPD subjects that demonstrated bronchodilator reversibility were not excluded from the study. A diagnosis of asthma was made by a respiratory physician with consistent symptomatology. Asthma severity was classified according to the Global Initiative for Asthma (GINA) treatment steps and described as mild (GINA I–II) or moderate to severe (GINA III–V) [15].
Measurements
All subjects had complete demographic data including age, duration of symptoms and full smoking history recorded. Spirometry and bronchodilator reversibility was performed on all subjects according to ATS/European Respiratory Society (ERS) guidelines [16]. Asthma subjects had atopy assessment with skin prick testing for Dermatophagoides pteronyssinus, dog, cat, Aspergillus fumigatus, tree and grass pollen and measurements of airway hyper-responsiveness using methacholine (Pc20) and exhaled nitric oxide (measured at 50 ml/s; NIOX; Aerocrine, Stockholm, Sweden).
Samples
Subjects provided spontaneous or induced sputum which was processed using a 2-step method with a Dulbecco's phosphate-buffered saline (D-PBS) wash step followed by a dithiothreitol (DTT) step and cytospins. In summary, selected and weighed sputum plugs were incubated with an 8× volume (sputum weight) of D-PBS, vortexed for 15 s and placed on a bench rocker on ice for 15 min. After centrifuging at 790 g for 10 min, the solute preparation was split in two: 4 volumes of the supernatant were removed (one half) and 300-μl aliquots were prepared and stored directly at −80°C; this was identified as the D-PBS supernatant. The remainder of the solute was incubated with a 4× volume (sputum weight) of 0.2% DTT for 15 min on a bench rocker on ice to disperse the cells. This sample was then filtered through a 50-μm nylon mesh filter and an aliquot removed for estimation of the total cell count. The remainder of the filtered sputum was centrifuged and the supernatant was removed and stored at −80°C (DTT supernatant). The remaining cell, D-PBS and mucus pellets were processed to form cytospins for sputum cell differential as previously described [17]. Comparison and validation of the effects of the additional D-PBS step with that of currently used sputum processing for cytospin preparation for the sputum total cell count and differential cell count was performed in 20 subjects with COPD and asthma. There were no significant differences in the cell differential counts between the methods with or without the additional D-PBS wash step [mean (SEM) neutrophil count 78% (5) vs. 80% (5); geometric mean (95% CI) eosinophil count 1.3% (0.7–2.7) vs. 1.0% (0.6–1.9), and total cell count 4.0 × 106 cells/g sputum (1.7–9.8) vs. 3.8 × 106 cells/g sputum (1.6–9.0)].
Meso Scale Discovery and Luminex Platform
The D-PBS sputum supernatant was analysed using the Meso Scale Discovery platform (MSD; USA) and Luminex platform (R&D Systems) for measurement of cytokine and chemokine mediators and matrix metalloproteinases (MMPs), respectively. Recovery of cytokine and chemokine mediators and MMPs using the MSD and Luminex platform was assessed by adding standard chemokine, cytokine and MMP spike to the sputum plug and to the buffer prior to processing. Following ERS recommendations [17] further analysis was carried out on those mediators that gave >80% recovery of spike from D-PBS-treated sputum compared to buffer controls.
Statistical Analysis
Statistical analysis was performed using PRISM version 4 (GraphPad Software, San Diego, Calif., USA) and SPSS version 16 (SPSS, Inc., Chicago, Ill., USA). Parametric and nonparametric data are presented as means (SEM) and medians (interquartile range) unless otherwise stated. Log-transformed data is presented as geometric means (95% CI). For comparison of unpaired or paired parametric or non-parametric groups, the Student t test, paired t test, Mann-Whitney test and Wilcoxon matched pairs test were used, respectively. For comparison of 3 groups or more for parametric and non-parametric variables the one-way analysis of variance (ANOVA) or Kruskal-Wallis test was used and the χ2 test was used for proportions. Sputum eosinophilia was defined as >2% eosinophils of non-squamous cells, and sputum neutrophilia was classified as >61% neutrophils of non-squamous cells [18]. p < 0.05 was considered statistically significant.
Results
One hundred and three subjects were recruited (asthma, n = 54; COPD, n = 49). The baseline demographics of all subjects are presented in table 1. Asthma severity showed that 13, 24, 30 and 33% of subjects were GINA I, II, III and IV/V. The GOLD classification for COPD severity identified 19, 33, 33 and 15% as having GOLD I, II, III and IV. The mean (SEM) post-bronchodilator FEV1 % predicted for the asthma and COPD subjects was 87% (4) and 54% (4), respectively. Sputum mediator analysis using the MSD and Luminex platforms was available in 37 asthmatic and 23 COPD subjects. The clinical characteristics of these subjects with available sputum mediator analysis are presented in table 2. Analytes with increased recovery in D-PBS-treated sputum compared to DTT-treated sputum were IL-1β, 4, 5, 10, 13, IFN-γ, TNFRI, GM-CSF, CCL2, 3, 4, 5, 13 and 17 (online suppl. fig. 1; for all online supplementary material, see www.karger.com/doi/10.1159/000330667). The mediators that were measured in the sputum and their lower and upper quantification limits are listed in online supplementary table 1.
Table 1.
Baseline demographics of asthma and COPD subjects
| COPD (n = 49) | Asthma (n = 54) | p value | |
|---|---|---|---|
| Males, n (%) | 36 (74) | 21 (39) | 0.07 |
| Mean age (range), years | 70 (42–87) | 52 (24–88) | <0.001 |
| Mean age of onset (range), years | 62 (35–79) | 26 (5–87) | <0.001 |
| Non-smokers, n (%) | 0 (0) | 36 (67) | <0.001 |
| Ex-smokers, n (%) | 11 (22) | 13 (24) | 0.99 |
| Current smokers, n (%) | 38 (78) | 5 (9) | <0.001 |
| Pack year history | 23 (2) | 4 (1) | <0.001 |
| Body mass index | 26.3 (0.7) | 27.0 (0.8) | 0.57 |
| Long acting beta agonist usage, % | 80 | 83 | 0.80 |
| Inhaled corticosteroid usage, % | 82 | 89 | 0.40 |
| Inhaled corticosteroid dosea, μg | 1,535 (99) | 1,150 (85) | 0.004 |
| Maintenance prednisolone, % | 8 | 15 | 0.37 |
| Mean prednisolone dose (range), mg | 8 (5–10) | 8 (5–10) | 0.65 |
| Airway hyper-responsiveness (Pc20)b | – | 1.1 (0.6–2.1) | – |
| FEV1/FVC (%)c | 49 (2) | 71 (1) | <0.001 |
| FEV1c, litres | 1.4 (0.1) | 2.6 (0.1) | <0.001 |
| Reversibility, ml | 122 (31) | 151 (29) | 0.51 |
| Reversibility, % | 9 (2) | 6 (1) | 0.12 |
| FEV1 % predictedc | 54 (4) | 87 (4) | <0.001 |
| FENOb, ppb | – | 34 (26–44) | – |
| Total sputum cell countb, × 106 cells/g | 4.0 (3.0–5.4) | 3.2 (2.4–4.2) | 0.24 |
| Sputum eosinophil countb, % | 1.2 (0.8–1.9) | 2.6 (1.6–4.2) | 0.02 |
| Sputum neutrophil count, % | 64 (4) | 57 (4) | 0.18 |
| Airway inflammatory phenotypes [18], % | |||
| Pauci-granular | 22 | 20 | 0.81 |
| Eosinophilic | 18 | 35 | 0.08 |
| Neutrophilic | 43 | 32 | 0.31 |
| Mixed-granulocytic | 16 | 13 | 0.78 |
Data are presented as means (SEM) unless otherwise stated.
BDP equivalent.
Geometric mean (95% CI).
Post-bronchodilator.
Table 2.
Clinical characteristics of subjects with measured sputum mediators
| COPD (n = 23) | Asthma (n = 37) | |
|---|---|---|
| Males, n (%) | 17 (74) | 17 (46) |
| Mean age (range), years | 71 (52–86) | 55 (24–88) |
| FEV1/FVC (%)a litres | 51 (3) | 69 (2) |
| FEV1a, litres | 1.4 (0.1) | 2.5 (0.1) |
| FEV1 % predicteda | 56 (4) | 86 (4) |
| Reversibility, ml | 154 (63) | 151 (38) |
| Reversibility, % | 10 (4) | 6 (1) |
| Total sputum cell countb, ×106cells/g | 5.1 (3.3–7.7) | 3.8 (2.8–5.1) |
| Sputum neutrophil count, % | 71 (5) | 56 (4) |
| Sputum eosinophil countb, % | 1.0 (0.5–1.8) | 3.1 (1.7–5.6) |
Data are presented as means (SEM) unless otherwise stated.
Post-bronchodilator.
Geometric mean (95% CI).
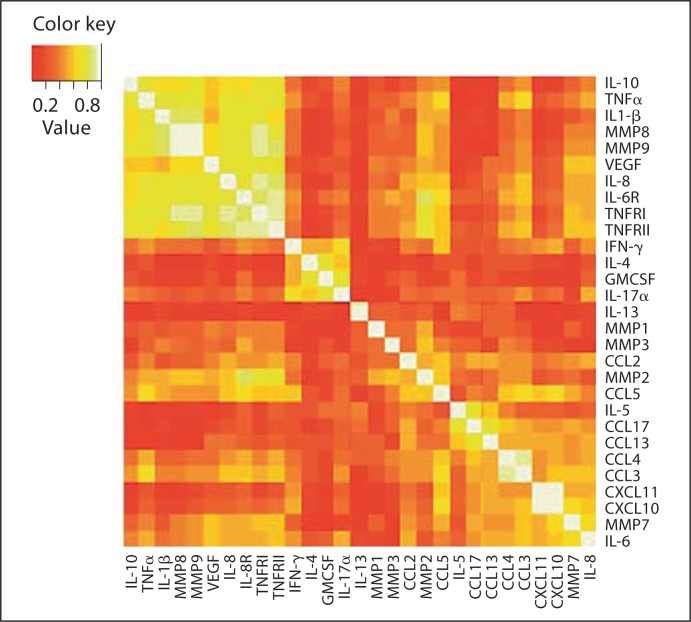
Correlations of all measured sputum markers are presented as a heat map illustration in figure 1. Sputum markers that are closely correlated are identified as intense bright colours (white: r = 1.0). There was a strong correlation identified between TNFRI, TNFRII and IL-6R (r = 0.93, r = 0.90 and r = 0.90); MMP8, MMP9 and IL-1β (r = 0.94, r = 0.91 and r = 0.87), and CXCL10 with CXCL11 (r = 0.92).
Fig. 1.
Heat map demonstrating correlations of all sputum markers measured. Perfect correlation (r = 1.0) is identified by white colour.
Airway Inflammation in Asthma and COPD
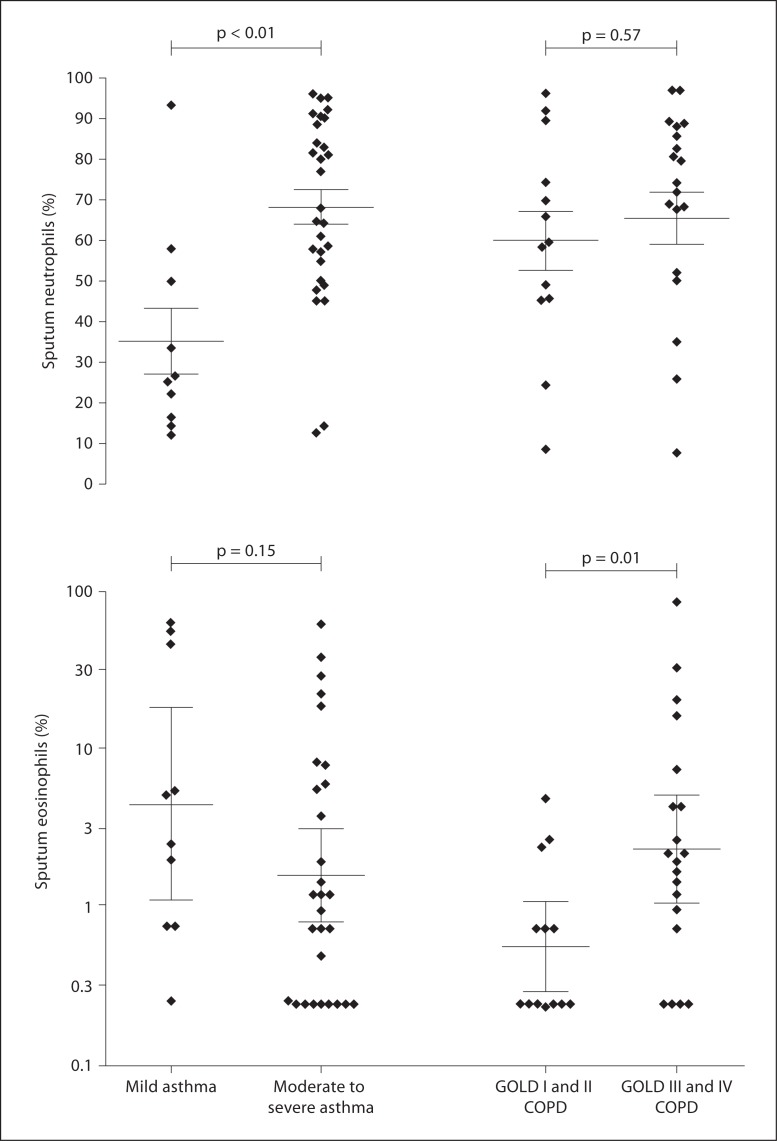
Airway eosinophilic and neutrophilic inflammation was present in 48 and 35% and in 45 and 59% of asthma and COPD subjects, respectively. Sputum eosinophils were higher in asthma compared to COPD subjects (2.6 vs. 1.2, fold difference 0.3; 95% CI fold difference 0.1–0.6, p = 0.02). There was no difference in sputum neutrophil counts between asthma and COPD (57 vs. 64%, mean difference −7; 95% CI −18 to 3, p = 0.18). Sputum neutrophils but not eosinophils were significantly lower in mild asthma compared to moderate/severe asthma (mean neutrophils 35 vs. 69%, mean difference 34; 95% CI −51 to −16, p = 0.0004; and geometric mean eosinophils 4.5 vs. 1.6%, fold difference 0.4; 95% CI −0.2 to 1.0, p = 0.15). Sputum eosinophils but not neutrophils were significantly lower in mild/moderate COPD compared to severe COPD (geometric mean eosinophils 0.6 vs. 2.4%, fold difference −0.6; 95% CI −1.1 to −0.1, p = 0.01; and mean neutrophils 60 vs. 65%, mean difference −6; 95% CI −26 to 14, p = 0.0004) (fig. 2).
Fig. 2.
Scatter plots presenting differential sputum neutrophil counts (a) and differential sputum eosinophil counts (b) in asthma and COPD subjects grouped according to severity of disease. Horizontal and error bars are set at the mean and SEM (a) and the geometric mean (95% CI) (b).
Sputum Mediators in Asthma and COPD
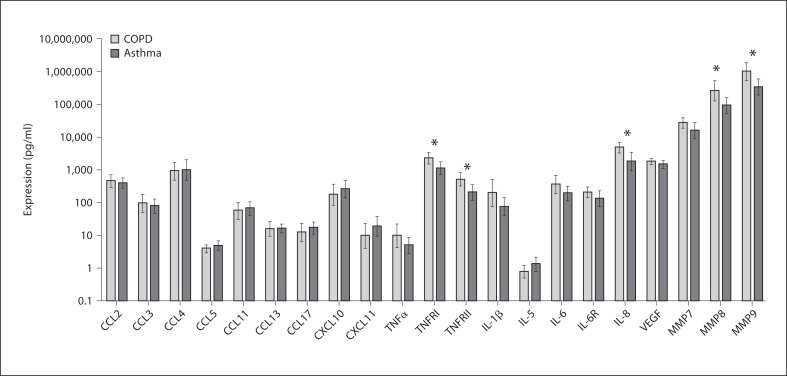
Sputum mediator profiling between asthma and COPD showed increased levels of IL-8, TNFRI and TNFRII in COPD compared to asthma subjects [mean fold difference (95% CI): IL-8, 2.6 (1.3–5.4), p = 0.01; TNFRI, 2.1 (1.3–5.4), p = 0.03, and TNFRII, 2.6 (1.2– 5.6), p = 0.02] and elevated MMP8 and 9 in COPD subjects [mean fold difference (95% CI) 2.7 (1.1–6.6), p = 0.03, and 3.0 (1.3–7.0), p = 0.01, respectively] (fig. 3; online suppl. table 2). No differences in sputum mediator expression could be determined in COPD or asthma subjects according to smoking status. The effect of inhaled corticosteroids (ICS) upon mediator expression was assessed in subjects with asthma and COPD; this was performed by subdividing subjects into high dose ICS (≥1,600 μg beclomethasone equivalent), moderate dose ICS (>400 and ≤800 μg beclomethasone equivalent) and low dose ICS (≤400 μg beclomethasone equivalent). In all subjects, significantly different mediator expression was found in CXCL10 and CXCL11 only, although this was not in a dose-dependant way. The geometric mean (95% CI) CXCL10 and CXCL11 mediator expression in high dose ICS, moderate dose ICS and low dose ICS was as follows: CXCL10, 214 (125–364) versus 562 (325–972) versus 86 (25–300), p = 0.020, and CXCL11 9 (3–24) versus 40 (16–101) versus 4 (2–12), p = 0.043. These findings were true for asthma and COPD subjects.
Fig. 3.
Bar chart representing measured sputum cytokines and chemokines in asthma and COPD subjects. Mediators that are marked by an asterisk are significantly different (p < 0.05). Horizontal and error bars are set at the geometric mean and 95% CI.
Sputum Mediators in Airway Inflammatory Phenotypes
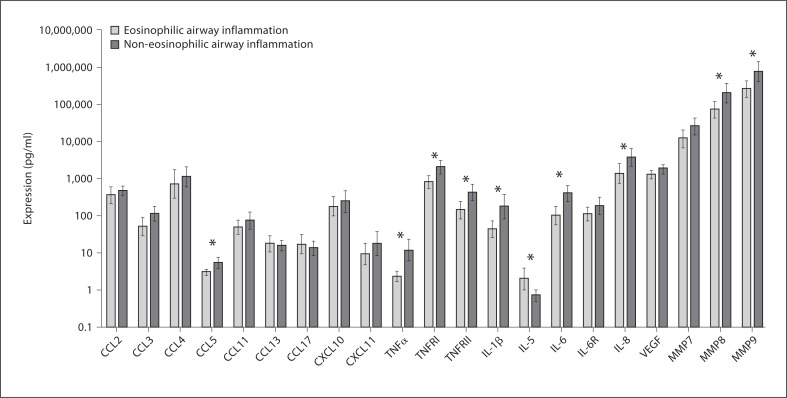
Differential mediator expression was present between eosinophilic and non-eosinophilic airway inflammation (fig. 4). There was no difference in mediator expression between subjects with asthma and subjects COPD after dichotomising for eosinophilic or neutrophilic airway inflammation (online suppl. fig. 2).
Fig. 4.
Bar chart comparing measured sputum mediators in eosinophilic airway inflammation and non-eosinophilic airway inflammation. Mediators that are marked by an asterisk are significantly different (p < 0.05). Horizontal and error bars are set at the geometric mean and 95% CI.
Discussion
In this study we have shown that sputum cellular patterns of airway inflammation are distinct across disease severity in asthma and COPD. However, sputum cell profiles are similar between severe asthma and COPD. Likewise, the sputum mediator profiles are largely similar between asthma and COPD, with a few mediators increased in COPD. Importantly, differential mediator expression is more marked between eosinophilic and non-eosinophilic sub-phenotypes, which is independent of disease.
Mediator expression between COPD and asthma has confirmed elevated levels of the pro-inflammatory mediators IL-6 and IL-8 in COPD [19, 20], whilst there is increased expression of Th2 cytokines such as IL-4, IL-5 and IL-13 in asthma and allergic disease [21, 22]. Bronchial biopsies studies have shown predominately neutrophilic inflammation in COPD and eosinophilic inflammation in asthma [5, 23]. However an overlap in cellular inflammation exists [24]; indeed we have shown a similar overlap in airway cellular inflammation with little difference in airway inflammation and in particular airway inflammatory phenotypes between moderate-to-severe asthma and COPD. It has been recognised that in persistent severe asthma there is often neutrophilic airway inflammation with elevated levels of IL-8 [25] and this type of inflammation confers steroid non-responsiveness [10, 26], albeit this maybe confounded itself by the effects of inhaled and oral corticosteroids [27], which have been shown to alter mediator expression [28]. Here we have shown that the neutrophilic airway inflammation that is present in severe asthma is similar to the degree of neutrophilic inflammation that exists in COPD. The identification of airway inflammatory phenotypes has been effectively used to reduce exacerbations in asthma and COPD [29, 30], whilst cluster analysis has determined airway inflammatory phenotypes within clinical phenotypes of asthma [14].
We have also validated a sputum processing method that improves the recovery of previously difficult to measure cytokines [13]. The use of multiplex platforms has provided the opportunity to assess inflammatory mediators in small volumes. In our study the initial incubation of selected sputum plugs with D-PBS followed by standard sputum processing techniques improved the recovery of cytokines including previously difficult to measure Th2 cytokines [8] and improved the recovery of several cytokines satisfying the European Respiratory Guidelines on mediator recovery [17].
In our study we have shown that the chemokine and cytokine expressions in asthma and COPD are broadly similar, with a few mediators increased in COPD. Indeed the differential expression of mediators between eosinophilic and non-eosinophilic airway inflammation is more marked and we could not demonstrate any differential expression between asthma and COPD after stratification for inflammatory airway cellular phenotypes. This mediator expression between airway inflammatory phenotypes has been demonstrated separately in COPD [8] and asthma [10]. Although it has been widely perceived that asthma and COPD are distinct entities [31], the observation of similarities in their clinical phenotypes [2], and airway inflammometry [1], increasingly suggests that one needs to view these airway inflammatory diseases together as a spectrum of disease with similar clinical symptoms, airway inflammation and physiology [32, 33]. A phenotypic approach can propagate the development of targeted treatment strategies including the development of monoclonal antibody therapies [34] and further the understanding in the pathogenesis of the disease [11, 12, 35, 36].
A limitation in this study is that we used subjects with asthma and COPD and did not compare the mediator response with relation to healthy controls, and in particular smoking and non-smoking controls. However we have been able to investigate asthma and COPD across all severities and applied our analysis to a widely observed clinical spectrum of disease; smoking healthy controls have previously demonstrated mediator expression and airway inflammation similar to subjects with COPD [23]. In this study we did not fully characterise the airway disease phenotype using computed tomography or gas diffusion, and this and the effect of treatment could affect the inflammatory profiles seen. Serum and bronchoalveolar lavage cytokine expression with radiological characterisation of COPD have previously shown that mediator association is independent of lung function [37] and Th2 cytokine expression is higher in subjects with radiological emphysema [38, 39]. Thus, it is interesting to note that similar mediator expression was seen within inflammatory groups in different disease expressions.
Although we were unable to do so in this study, inspection of mediators that are released, expressed on the cell surface or act as a surrogate measure of cell activity would provide further characterisation of eosinophilic and neutrophilic airway inflammatory patterns in COPD and asthma above that already shown using sputum mediator analysis in this study.
In conclusion, we have described and validated a novel sputum processing method which improves recovery and detection of several cytokines and chemokines, including that of previously difficult to measure Th2 cytokines. We have demonstrated that there are limited differences in the expression of sputum cytokines and chemokines between asthma and COPD, and there is greater differential expression between eosinophilic and non-eosinophilic sub-phenotypes, which may be independent of disease. Whether these airway mediator phenotypes confer a particular pathogenesis and clinical phenotype, and subsequent responses to treatment, needs to be further investigated in larger cohorts of subjects with airway disease.
Sources of Funding
This study was funded by AstraZeneca, the European Regional Development Fund (ERDF 05567) and a grant from a Wellcome Senior Clinical Fellowship (C.E.B.).
Financial Disclosure and Conflicts of Interest
M.B., S.S., S.M., B.H., M.S., C.R., V.M. and D.P. have no financial interests to disclose. M.M., M.J., A.L., P.D., P.R. and P.N. are/were employees of AstraZeneca at the time of data collection and analysis. C.E.B. has received grant support and consultancy fees from AstraZeneca, MedImmune, Roche and GlaxoSmithKline.
Supplementary Material
Supplementary Material
Acknowledgements
The authors would like to thank all of the volunteers that took part in this study and the following people for their assistance in patient characterisation and laboratory support: M. Bourne, H. Pateman, A. Singapuri, S. Terry, M. Pancholi, and W. Monteiro.
References
- 1.Siddiqui S, Brightling CE. Airways disease: phenotyping heterogeneity using measures of airway inflammation. Allergy Asthma Clin Immunol. 2007;3:60–69. doi: 10.1186/1710-1492-3-2-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gibson PG, Simpson JL. The overlap syndrome of asthma and COPD: what are its features and how important is it? Thorax. 2009;64:728–735. doi: 10.1136/thx.2008.108027. [DOI] [PubMed] [Google Scholar]
- 3.Gomez FP, Rodriguez-Roisin R. Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines for chronic obstructive pulmonary disease. Curr Opin Pulm Med. 2002;8:81–86. doi: 10.1097/00063198-200203000-00001. [DOI] [PubMed] [Google Scholar]
- 4.Jeffery PK. Structural and inflammatory changes in COPD: a comparison with asthma. Thorax. 1998;53:129–136. doi: 10.1136/thx.53.2.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Saetta M, Di SA, Maestrelli P, Ferraresso A, Drigo R, Potena A, et al. Activated T-lymphocytes and macrophages in bronchial mucosa of subjects with chronic bronchitis. Am Rev Respir Dis. 1993;147:301–306. doi: 10.1164/ajrccm/147.2.301. [DOI] [PubMed] [Google Scholar]
- 6.Rabe KF, Hurd S, Anzueto A, Barnes PJ, Buist SA, Calverley P, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2007;176:532–555. doi: 10.1164/rccm.200703-456SO. [DOI] [PubMed] [Google Scholar]
- 7.Saha S, Brightling CE. Eosinophilic airway inflammation in COPD. Int J Chron Obstruct Pulmon Dis. 2006;1:39–47. doi: 10.2147/copd.2006.1.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bafadhel M, Saha S, Siva R, McCormick M, Monteiro W, Rugman P, et al. Sputum IL-5 concentration is associated with a sputum eosinophilia and attenuated by corticosteroid therapy in COPD. Respiration. 2009;78:256–262. doi: 10.1159/000221902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Douwes J, Gibson P, Pekkanen J, Pearce N. Non-eosinophilic asthma: importance and possible mechanisms. Thorax. 2002;57:643–648. doi: 10.1136/thorax.57.7.643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cowan DC, Cowan JO, Palmay R, Williamson A, Taylor DR. Effects of steroid therapy on inflammatory cell subtypes in asthma. Thorax. 2010;65:384–390. doi: 10.1136/thx.2009.126722. [DOI] [PubMed] [Google Scholar]
- 11.Simpson JL, Scott R, Boyle MJ, Gibson PG. Inflammatory subtypes in asthma: assessment and identification using induced sputum. Respirology. 2006;11:54–61. doi: 10.1111/j.1440-1843.2006.00784.x. [DOI] [PubMed] [Google Scholar]
- 12.Pavord ID, Sterk PJ, Hargreave FE, Kips JC, et al. Clinical applications of assessment of airway inflammation using induced sputum. Eur Respir J. 2002;37:40s–43s. doi: 10.1183/09031936.02.00004002. [DOI] [PubMed] [Google Scholar]
- 13.Pizzichini E, Pizzichini MM, Efthimiadis A, Evans S, Morris MM, Squillace D, et al. Indices of airway inflammation in induced sputum: reproducibility and validity of cell and fluid-phase measurements. Am J Respir Crit Care Med. 1996;154:308–317. doi: 10.1164/ajrccm.154.2.8756799. [DOI] [PubMed] [Google Scholar]
- 14.Haldar P, Pavord ID, Shaw DE, Berry MA, Thomas M, Brightling CE, et al. Cluster analysis and clinical asthma phenotypes. Am J Respir Crit Care Med. 2008;178:218–224. doi: 10.1164/rccm.200711-1754OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bateman ED, Hurd SS, Barnes PJ, Bousquet J, Drazen JM, FitzGerald M, et al. Global strategy for asthma management and prevention: GINA executive summary. Eur Respir J. 2008;31:143–178. doi: 10.1183/09031936.00138707. [DOI] [PubMed] [Google Scholar]
- 16.Brusasco V, Crapo R, Viegi G. Coming together: the ATS/ERS consensus on clinical pulmonary function testing. Eur Respir J. 2005;26:1–2. doi: 10.1183/09031936.05.00034205. [DOI] [PubMed] [Google Scholar]
- 17.Kelly MM, Keatings V, Leigh R, Peterson C, Shute J, Venge P, et al. Analysis of fluid-phase mediators. Eur Respir J Suppl. 2002 Sep;37:24s–39s. [PubMed] [Google Scholar]
- 18.Belda J, Leigh R, Parameswaran K, O’Byrne PM, Sears MR, Hargreave FE. Induced sputum cell counts in healthy adults. Am J Respir Crit Care Med. 2000;161:475–478. doi: 10.1164/ajrccm.161.2.9903097. [DOI] [PubMed] [Google Scholar]
- 19.Keatings VM, Collins PD, Scott DM, Barnes PJ. Differences in interleukin-8 and tumor necrosis factor-alpha in induced sputum from patients with chronic obstructive pulmonary disease or asthma. Am J Respir Crit Care Med. 1996;153:530–534. doi: 10.1164/ajrccm.153.2.8564092. [DOI] [PubMed] [Google Scholar]
- 20.Jeffery PK, Laitinen A, Venge P. Biopsy markers of airway inflammation and remodelling. Respir Med. 2000;94((suppl F)):S9–S15. doi: 10.1016/s0954-6111(00)90127-6. [DOI] [PubMed] [Google Scholar]
- 21.Jeffery PK. Differences and similarities between chronic obstructive pulmonary disease and asthma. Clin Exp Allergy. 1999;29((suppl 2)):14–26. [PubMed] [Google Scholar]
- 22.Robinson DS. Th-2 cytokines in allergic disease. Br Med Bull. 2000;56:956–968. doi: 10.1258/0007142001903625. [DOI] [PubMed] [Google Scholar]
- 23.Soler N, Ewig S, Torres A, Filella X, Gonzalez J, Zaubet A. Airway inflammation and bronchial microbial patterns in patients with stable chronic obstructive pulmonary disease. Eur Respir J. 1999;14:1015–1022. doi: 10.1183/09031936.99.14510159. [DOI] [PubMed] [Google Scholar]
- 24.O’Donnell R, Breen D, Wilson S, Djukanovic R. Inflammatory cells in the airways in COPD. Thorax. 2006;61:448–454. doi: 10.1136/thx.2004.024463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gibson PG, Simpson JL, Saltos N. Heterogeneity of airway inflammation in persistent asthma: evidence of neutrophilic inflammation and increased sputum interleukin-8. Chest. 2001;119:1329–1336. doi: 10.1378/chest.119.5.1329. [DOI] [PubMed] [Google Scholar]
- 26.Berry M, Morgan A, Shaw DE, Parker D, Green R, Brightling C, et al. Pathological features and inhaled corticosteroid response of eosinophilic and non-eosinophilic asthma. Thorax. 2007;62:1043–1049. doi: 10.1136/thx.2006.073429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Belvisi MG. Regulation of inflammatory cell function by corticosteroids. Proc Am Thorac Soc. 2004;1:207–214. doi: 10.1513/pats.200402-002MS. [DOI] [PubMed] [Google Scholar]
- 28.Fukakusa M, Bergeron C, Tulic MK, Fiset PO, Al DO, Laviolette M, et al. Oral corticosteroids decrease eosinophil and CC chemokine expression but increase neutrophil, IL-8, and IFN-gamma-inducible protein 10 expression in asthmatic airway mucosa. J Allergy Clin Immunol. 2005;115:280–286. doi: 10.1016/j.jaci.2004.10.036. [DOI] [PubMed] [Google Scholar]
- 29.Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Bradding P, et al. Asthma exacerbations and sputum eosinophil counts: a randomised controlled trial. Lancet. 2002;360((9347)):1715–1721. doi: 10.1016/S0140-6736(02)11679-5. [DOI] [PubMed] [Google Scholar]
- 30.Siva R, Green RH, Brightling CE, Shelley M, Hargadon B, McKenna S, et al. Eosinophilic airway inflammation and exacerbations of COPD: a randomised controlled trial. Eur Respir J. 2007;29:906–913. doi: 10.1183/09031936.00146306. [DOI] [PubMed] [Google Scholar]
- 31.Jeffery PK. Comparison of the structural and inflammatory features of COPD and asthma. Giles F. Filley Lecture. Chest. 2000;117((suppl 1)):251S–260S. doi: 10.1378/chest.117.5_suppl_1.251s. [DOI] [PubMed] [Google Scholar]
- 32.Pavord ID, Wardlaw AJ. The A to E of airway disease. Clin Exp Allergy. 2010;40:62–67. doi: 10.1111/j.1365-2222.2009.03410.x. [DOI] [PubMed] [Google Scholar]
- 33.Wardlaw AJ, Silverman M, Siva R, Pavord ID, Green R. Multi-dimensional phenotyping: towards a new taxonomy for airway disease. Clin Exp Allergy. 2005;35:1254–1262. doi: 10.1111/j.1365-2222.2005.02344.x. [DOI] [PubMed] [Google Scholar]
- 34.Haldar P, Brightling CE, Hargadon B, Gupta S, Monteiro W, Sousa A, et al. Mepolizumab and exacerbations of refractory eosinophilic asthma. N Engl J Med. 2009;360:973–984. doi: 10.1056/NEJMoa0808991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Spanevello A, Dragonieri S, Maestrelli P. Towards a better phenotyping of chronic obstructive pulmonary diseases. Respiration. 2009;78:253–255. doi: 10.1159/000229191. [DOI] [PubMed] [Google Scholar]
- 36.Han MK, Agusti A, Calverley PM, Celli BR, Criner G, Curtis JL, et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. Am J Respir Crit Care Med. 2010;182:598–604. doi: 10.1164/rccm.200912-1843CC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bon JM, Leader JK, Weissfeld JL, Coxson HO, Zheng B, Branch RA, et al. The influence of radiographic phenotype and smoking status on peripheral blood biomarker patterns in chronic obstructive pulmonary disease. PLoS One. 2009;4:e6865. doi: 10.1371/journal.pone.0006865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Miller M, Ramsdell J, Friedman PJ, Cho JY, Renvall M, Broide DH. Computed tomographic scan-diagnosed chronic obstructive pulmonary disease-emphysema: eotaxin-1 is associated with bronchodilator response and extent of emphysema. J Allergy Clin Immunol. 2007;120:1118–1125. doi: 10.1016/j.jaci.2007.08.045. [DOI] [PubMed] [Google Scholar]
- 39.D'Armiento JM, Scharf SM, Roth MD, Connett JE, Ghio A, Sternberg D, et al. Eosinophil and T cell markers predict functional decline in COPD patients. Respir Res. 2009;10:113. doi: 10.1186/1465-9921-10-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material