Abstract
OBJECTIVE
The purpose of this multicenter study was to confirm the validity and reliability of the Pediatric Cardiac Quality of Life Inventory (PCQLI).
METHODS
Seven centers recruited pediatric patients (8–18 years of age) with heart disease (HD) and their parents to complete the PCQLI and generic health-related quality of life (Pediatric Quality of Life Inventory [PedsQL]) and non–quality of life (Self-Perception Profile for Children [SPPC]/Self-Perception Profile for Adolescents [SPPA] and Youth Self-Report [YSR]/Child Behavior Checklist [CBCL]) tools. PCQLI construct validity was assessed through correlations of PCQLI scores between patients and parents and with severity of congenital HD, medical care utilization, and PedsQL, SPPC/SPPA, and YSR/CBCL scores. PCQLI test-retest reliability was evaluated.
RESULTS
The study enrolled 1605 patient-parent pairs. Construct validity was substantiated by the association of lower PCQLI scores with Fontan palliation and increased numbers of cardiac operations, hospital admissions, and physician visits (P < .001); moderate to good correlations between patient and parent PCQLI scores (r = 0.41–0.61; P <.001); and fair to good correlations between PCQLI total scores and PedsQL total (r = 0.70–0.76), SPPC/SPPA global self-worth (r = 0.43–0.46), YSR/CBCL total competency (r = 0.28–0.37), and syndrome and Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition-oriented scale (r = −0.58 to −0.30; P < .001) scores. Test-retest reliability correlations were excellent (r = 0.78–0.90; P < .001).
CONCLUSIONS
PCQLI scores are valid and reliable for children and adolescents with congenital and acquired HD and may be useful for future research and clinical management. Pediatrics 2010;126:498–508
Keywords: congenital heart disease, acquired heart disease, health-related quality of life, reliability, validity
In recent decades, new surgical techniques and advances in cardiopulmonary bypass, intensive care, heart transplantation, and interventional catheterization have significantly reduced mortality rates for children with heart disease (HD).1 This growing population of survivors has important neurodevelopmental, psychosocial, and physical morbidities resulting from their disease and/or its management that may affect quality of life (QoL).2,3 Health-related QoL (HRQoL) is defined as the influence of a specific illness, medical therapy, or health services policy on the ability of the patient to both function in and derive personal satisfaction from various physical, psychological, and social life contexts.4 Evaluation of HRQoL is important because it allows for better communication among patients, parents, and health care providers; prioritization of problems in part on the basis of patient and/or parent preferences; monitoring of changes over time or in response to specific therapies; and screening for other significant medical and psychosocial problems.5 Despite the known advantages, validated assessment of HRQoL in the pediatric cardiac population is lacking.6 A more-complete understanding of the impact of HD on HRQoL in the pediatric population may improve patient-centered medical treatment and outcomes.7
Several characteristics inherent to the pediatric cardiac population have limited HRQoL assessment, including the broad age range, changing developmental capabilities, and the variety of defects, therapies (medical, surgical, and interventional), and outcomes. The existing, disease-specific, HRQoL instruments for the pediatric cardiac population each have salient limitations, namely, narrow age ranges,8,9 limited data to support broad applicability to other geographic regions or demographic groups within the United States,8–10 and/or an inability to discriminate among various types of HD.8–10 The Pediatric Cardiac Quality of Life Inventory (PCQLI) was developed to address these limitations.11 It is a self-administered, disease-specific, HRQoL tool for children and adolescents with HD that features a wide age range, parent-proxy reporting, and the ability to differentiate among congenital HD subpopulations.11
Limited validity and reliability analyses exist for disease-specific, pediatric, cardiac HRQoL measures, including the PCQLI.8–12 However, valid, reliable instruments are required for quantitative HRQoL assessment, which may be used to improve patient outcomes systematically. The PCQLI subscales have been shown to have excellent internal consistency and to correlate well with each other and with the PCQLI total.11 The purpose of this large, multicenter study was to confirm the validity and reliability of the PCQLI by evaluating the impact of important clinical and patient factors (disease severity, medical care utilization, patient-parent consensus, and patient self-perception, competency, and behavior) on HRQoL in the pediatric cardiac population. We hypothesized that PCQLI scores would be reliable (similar between baseline testing and retesting) and valid (inversely correlated with disease severity and medical utilization, similar between patient and parent respondents, and negatively associated with poorer self-perception, decreased competency, and increased behavioral and emotional problems).
METHODS
Study Design
This study was a prospective, cross-sectional survey of pediatric patients with congenital or acquired HD and their parents or guardians. It was performed at 7 large, pediatric cardiovascular care centers in the United States between November 2004 and December 2008. The institutional review boards of all participating institutions approved the study.
Subject Selection
Patients were eligible for participation if they had congenital or acquired HD, were 8 to 18 years of age, were fluent in English, and were attending an outpatient cardiology visit. Patients were excluded if they had a significant co-morbid medical condition or major developmental delay or came to the clinic because of an acute change in their clinical status. Parents (an inclusive term for parents or guardians of eligible patients) were excluded if they were not fluent in English or had a major developmental delay. All eligible, consenting/assenting patients and parents were enrolled in the study.
Inventory Administration
Patient-parent pairs completed a battery of questionnaires that included a disease-specific, pediatric, cardiac HRQoL instrument (PCQLI),11 a generic HRQoL instrument (Pediatric Quality of Life Inventory [PedsQL] Generic Core Scales 4.0),13 and non–QoL instruments (Self-Perception Profile for Children [SPPC]/Self-Perception Profile for Adolescents [SPPA], Child Behavior Checklist [CBCL], and Youth Self-Report [YSR]).14–17 All generic HRQoL and non–QoL inventories used in the study are internally consistent and valid.13–17
Reliability testing included patient-parent pairs available to complete inventories on 2 occasions (baseline testing and retesting). Baseline testing (all inventories including the PCQLI) was completed ≥10 days before an outpatient cardiology visit. Retesting (PCQLI and brief interim history) was performed on the day of the appointment, before patients saw their cardiology provider. Patients and/or parents were excluded from test-retest reliability analysis if any significant life event or change in patient clinical status occurred between study visits. Patient-parent pairs who were unavailable for testing on 2 occasions were recruited for construct validity testing and completed the baseline inventories on the day of the appointment, before seeing their cardiology provider. Inventory completion was supervised, to minimize data contamination resulting from patient-parent discussion.
Inventory Selection
PCQLI
The PCQLI measures disease-specific, pediatric HRQoL and generates 3 scores, namely, total, disease impact subscale, and psychosocial impact subscale.11 Each subscale score has a maximum of 50 points, and their sum yields the total score. Higher scores represent better perceived HRQoL. The PCQLI has 4 forms, that is, child (ages 8–12), parent of child (parent-proxy), adolescent (ages 13–18), and parent of adolescent (parent-proxy) forms.
PedsQL
The PedsQL is a generic HRQoL instrument that assesses HRQoL in healthy, acutely ill, and chronically ill pediatric populations.13 It generates 3 scores, namely, total, physical health summary, and psychosocial health summary. Higher scores represent better perceived HRQoL. The PedsQL has 4 forms, that is, child (ages 8–12) and teen (ages 13–18) forms and 2 associated parent-proxy forms.
SPPC/SPPA
The SPPC (child form, for ages 8–12) and SPPA (adolescent form, for ages 13–18) are non–QoL measures that assess self-perception in children and adolescents, respectively. The principal construct is global self-worth.14,15 Higher scores are associated with better self-perception.
YSR/CBCL
The YSR (for patients 11–18 years of age) and CBCL (parent-proxy for patients 6–18 years of age) are non–QoL instruments that assess competencies (activities, social, school, and total), emotional state (syndrome scales), and behavioral problems (Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition [DSM-IV] scales) of children and adolescents.16,17 Total competency, the syndrome scales constituting the internalizing problems summary scale (social withdrawal, somatic complaints, and anxiety/depression), and select DSM-IV scales (affective, anxiety, somatic, and attention-deficit/hyperactivity disorders) were analyzed for this study.16,17
Demographic and Clinical Data Collection
Patient and parent demographic information and patient clinical information (diagnostic, surgical, and medical history) were collected through parent report and chart review. Patient clinical information is presented as primary diagnosis, original diagnostic category (primary diagnoses grouped into general categories), and current cardiac status (a synthesis of the patient’s original diagnostic category and interventions performed).
Statistical Analyses
Approach
Descriptive statistics were determined for the entire population. Continuous variables were expressed as means (with SDs or SEs) or medians (with ranges), depending on their distribution. Categorical variables were tabulated and expressed as proportions. All statistical comparisons used an a priori significance level of P < .05. All study inventory scoring and missing value adjustments were performed according to published guidelines.13–17 Even if missing information precluded inclusion in a specific analysis, patient and/or parent data were still included in all analyses for which there were complete data. Baseline inventories from the reliability testing were included in the validity testing analyses.
Construct Validity Assessment
To substantiate the validity of PCQLI scores, construct validity was assessed by using (1) comparison of PCQLI scores between specific congenital HD subgroups (known-groups method), (2) variation of PCQLI scores with medical care utilization (analysis of variance), (3) comparison of patient and parent PCQLI scores (convergent validity), and (4) comparison of PCQLI scores and generic HRQoL and non–QoL scores (convergent validity). For validity analyses, correlations were interpreted as follows: ≤0.20, poor agreement; 0.21 to 0.40, fair agreement; 0.41 to 0.60, moderate agreement; 0.61 to 0.80, good agreement; ≥0.81, excellent agreement.18
Association of PCQLI Scores With Severity of Congenital HD
To determine whether PCQLI scores decreased with increasing disease severity, patients with congenital HD were grouped into 3 representative subgroups, that is, (1) mild HD (no previous surgical or catheter-based intervention), (2) repaired biventricular HD, and (3) palliated single-ventricle HD. The Kruskal-Wallis 1-way analysis of variance test was used to compare PCQLI total and sub-scale scores among the 3 subgroups. In addition, a sequential Bonferroni correction (Holm’s method) was used for 2-way comparisons.19
Variation of PCQLI Scores With Medical Care Utilization
The number of occurrences for each medical care utilization variable was stratified before analysis, as follows: cardiac operations, 0, 1, 2, 3, or ≥4; HD-related hospital admissions, 0, 1, 2 to 5, 6 to 10, or ≥11; physician visits within the previous 12 months, 0, 1, 2, 3 to 5, 6 to 11, or ≥12. The Kruskal-Wallis 1-way analysis of variance test was used to evaluate the variance of PCQLI total scores with increasing medical care utilization in each respondent group.
Comparison of Patient and Parent PCQLI Scores
To assess the agreement between patient and parent scores, disattenuated20 interrater Spearman correlations were calculated for PCQLI total and subscale scores. Comparisons were made between patients and parents within each age category (child and adolescent).
Association of PCQLI Scores With Generic HRQoL and Non–QoL Scores
The relationships between the disease-specific HRQoL (PCQLI) and generic HRQoL (PedsQL) and non–QoL (SPPC/SPPA and YSR/CBCL) measures were evaluated by using the Spearman test for correlation. The PCQLI total and subscale scores were compared with the PedsQL total and summary scores, respectively. In addition, PCQLI total scores were correlated with SPPC/ SPPA global self-worth scores and with YSR/CBCL total competency, internalizing problems, and select DSM-IV scale scores.
Test-Retest Reliability Assessment
To assess the reliability of PCQLI scores, test-retest Spearman intrarater correlation coefficients were calculated. Spearman coefficients instead of Pearson coefficients were used to assess test-retest reliability because the PCQLI scores are not normally distributed, which violates the Pearson R assumption. Consistent with other biometric research, all comparisons used a predetermined significance threshold of 0.70.21 Values of ≥0.70 were considered excellent.
RESULTS
Subject Demographic Characteristics
A total of 1605 eligible patient-parent pairs enrolled in the PCQLI Testing Study (consentrate: 85%). Site-specific enrollment, patient and parent demographic, and patient clinical information is presented in Tables 1 and 2. The majority (53%–57%) of patients were male. Parents were primarily female (82%–87%). Patients and parents were predominantly white (81%–86%); black participants represented the most common minority (8%–9%). In both age groups, ~25% of patients received special educational or learning support at school or home, and 10% to 14% were in gifted programs. Approximately 75% of parents had completed some postsecondary education, with >50% having earned a bachelor’s degree or higher. The average Hollingshead socioeconomic status score of parents (mean score: 46.5–47.0) was indicative of upper-middle class.22 Patients commonly resided in a home with both parents (71%–78%). Primary diagnoses for the patient study population are shown in Table 2. Common biventricular HD primary diagnoses included septal defects, tetralogy of Fallot, aortic valve stenosis, and transposition of the great arteries. The most-frequent single-ventricle lesion was hypoplastic left heart syndrome. Common nonstructural heart defects were complete atrioventricular block and various forms of supraventricular tachycardia.
TABLE 1.
Subject Demographic, Diagnostic, and Medical Information
| Children (N = 803) | Parents/Guardians of Children (N = 803) | Adolescents (N = 802) | Parents/Guardians of Adolescents (N = 802) | |
|---|---|---|---|---|
| Site-specific enrollment, n (%) | ||||
| Children’s Hospital of Philadelphia | 251 (31.2) | 251 (31.2) | 325 (40.5) | 325 (40.5) |
| Cincinnati Children’s Hospital Medical Center | 164 (20.4) | 164 (20.4) | 130 (16.2) | 130 (16.2) |
| Children’s Hospital Boston | 102 (12.7) | 102 (12.7) | 105 (13.1) | 105 (13.1) |
| University of Texas Southwestern Medical Center at Dallas | 100 (12.5) | 100 (12.5) | 95 (11.8) | 95 (11.8) |
| Children’s Hospital of Wisconsin | 117 (14.6) | 117 (14.6) | 65 (8.1) | 65 (8.1) |
| University of California, San Francisco, Children’s Hospital | 49 (6.1) | 49 (6.1) | 53 (6.6) | 53 (6.6) |
| Phoenix Children’s Hospital | 20 (2.5) | 20 (2.5) | 29 (3.6) | 29 (3.6) |
| Age, mean ± SD, y | 10.3 ± 1.5 | 41.2 ± 6.4 | 15.7 ± 1.6 | 45.7 ± 6.0 |
| Gender, n (%) | ||||
| Male | 433 (53.9) | 109 (13.6) | 455 (56.7) | 138 (17.2) |
| Female | 370 (46.1) | 694 (86.4) | 347 (43.3) | 664 (82.8) |
| Ethnicity, n (%) | ||||
| Non-Hispanic | 748 (93.2) | 764 (95.1) | 750 (93.5) | 763 (95.1) |
| Hispanic | 52 (6.5) | 38 (4.7) | 49 (6.1) | 37 (4.6) |
| Unknown/not reported | 3 (0.4) | 1 (0.1) | 3 (0.4) | 2 (0.2) |
| Race, n (%) | ||||
| White | 656 (81.7) | 672 (83.7) | 673 (83.9) | 689 (85.9) |
| Black | 66 (8.2) | 65 (8.1) | 70 (8.7) | 65 (8.1) |
| Asian | 21 (2.6) | 20 (2.5) | 9 (1.1) | 6 (0.7) |
| American Indian/Native Alaskan | 2 (0.2) | 2 (0.2) | 5 (0.6) | 4 (0.5) |
| Native Hawaiian/other Pacific Islander | 1 (0.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| >1 race/other | 56 (7.0) | 42 (5.2) | 44 (5.5) | 36 (4.5) |
| Unknown/not reported | 1 (0.1) | 2 (0.2) | 1 (0.1) | 2 (0.2) |
| Level of education for patient, n (%) | ||||
| Kindergarten to grade 5 | 639 (79.6) | NA | 4 (0.5) | NA |
| Grades 6–8 | 162 (20.2) | NA | 284 (35.4) | NA |
| Grades 9–12 | 2 (0.2) | NA | 463 (57.7) | NA |
| GED/high school or technical/trade school graduate | 0 (0.0) | NA | 37 (4.6) | NA |
| Some college or training after high school | 0 (0.0) | NA | 9 (1.1) | NA |
| Other | 0 (0.0) | NA | 5 (0.6) | NA |
| Special educational programs, n (%) | ||||
| Individualized education plan | 100 (12.5) | NA | 98 (12.2) | NA |
| Learning supports (tutor or learning disability services) | 105 (13.1) | NA | 81 (10.1) | NA |
| Special education and mainstream classes (both part-time) | 65 (8.1) | NA | 63 (7.9) | NA |
| Special education classes (part-time) without mainstream classes | 4 (0.5) | NA | 9 (1.1) | NA |
| Self-contained special education classroom (full-time) | 14 (1.7) | NA | 13 (1.6) | NA |
| Gifted program | 84 (10.5) | NA | 115 (14.3) | NA |
| None | 507 (63.1) | NA | 497 (62.0) | NA |
| Primary residence, n (%) | ||||
| Both parents | 621 (77.3) | NA | 573 (71.4) | NA |
| Mother | 97 (12.1) | NA | 117 (14.6) | NA |
| Father | 17 (2.1) | NA | 18 (2.2) | NA |
| Mother plus other | 44 (5.5) | NA | 61 (7.6) | NA |
| Father plus other | 4 (0.5) | NA | 8 (1.0) | NA |
| Shared care (~50/50) | 13 (1.6) | NA | 15 (1.9) | NA |
| Neither parent (lives with guardian) | 7 (0.9) | NA | 9 (1.1) | NA |
| Relationship of parent/guardian to patient, n (%) | ||||
| Mother | NA | 685 (85.3) | NA | 659 (82.2) |
| Father | NA | 105 (13.1) | NA | 132 (16.5) |
| Grandparent | NA | 10 (1.2) | NA | 7 (0.9) |
| Legal guardian, related or nonrelated | NA | 3 (0.4) | NA | 3 (0.4) |
| Other | NA | 0 (0.0) | NA | 1 (0.1) |
| Level of education for parent/guardian, n (%) | ||||
| Kindergarten to grade 6 | NA | 1 (0.1) | NA | 1 (0.1) |
| Grades 7–9 | NA | 10 (1.2) | NA | 7 (0.9) |
| Grades 10–11 | NA | 23 (2.9) | NA | 20 (2.5) |
| High school graduate/GED | NA | 137 (17.1) | NA | 177 (22.1) |
| Some college or trade school | NA | 215 (26.8) | NA | 207 (25.8) |
| College graduate | NA | 294 (36.6) | NA | 276 (34.4) |
| Postgraduate degree | NA | 123 (15.3) | NA | 114 (14.2) |
| Hollingshead socioeconomic status score, mean | NA | 47.0 | NA | 46.5 |
| Current medications | ||||
| Taking ≥ 1 medication on regular basis, n (%) | 445 (55.4) | NA | 470 (58.6) | NA |
| No. of medications per patient, median (range) | 1 (0–18) | NA | 1 (0–18) | NA |
| Original diagnostic category, n (%) | ||||
| Biventricular CHD without AAO | 420 (52.3) | NA | 374 (46.6) | NA |
| Biventricular CHD with AAO | 83 (10.3) | NA | 72 (9.0) | NA |
| Single-ventricle CHD without AAO | 69 (8.6) | NA | 49 (6.1) | NA |
| Single-ventricle CHD with AAO | 67 (8.3) | NA | 35 (4.4) | NA |
| AHD with structurally normal heart | 164 (20.4) | NA | 272 (33.9) | NA |
| Current cardiac status, n (%) | ||||
| Unrepaired CHD | 108 (13.4) | NA | 97 (12.1) | NA |
| Repaired biventricular CHD | 390 (48.6) | NA | 343 (42.8) | NA |
| Palliated single-ventricle CHD | 130 (16.2) | NA | 74 (9.2) | NA |
| Heart transplant | 24 (3.0) | NA | 29 (3.6) | NA |
| AHD without intervention | 89 (11.1) | NA | 150 (18.7) | NA |
| AHD with intervention | 62 (7.7) | NA | 109 (13.6) | NA |
| Significant medical history, n (%) | ||||
| History of cerebrovascular event | 10 (1.2) | NA | 78 (9.7) | NA |
| History of seizures | 11 (1.4) | NA | 85 (10.6) | NA |
| Diagnosed learning disability | 152 (18.9) | NA | 235 (29.3) | NA |
| Diagnosed mental health disorder | 22 (2.7) | NA | 26 (3.2) | NA |
| Premature birth (<37 wk of gestation) | 238 (29.6) | NA | 126 (15.7) | NA |
| Multiple gestation | 82 (10.2) | NA | 16 (2.0) | NA |
| Age at cardiac diagnosis of < 30 d | 529 (65.9) | NA | 396 (49.4) | NA |
| Age at cardiac diagnosis of > 1 y | 178 (22.2) | NA | 278 (34.7) | NA |
AAO indicates aortic arch obstruction; AHD, acquired HD; CHD, congenital HD; GED, general equivalency diploma; NA, not applicable.
TABLE 2.
Primary Diagnoses for Patients
|
n (%)
|
|||
|---|---|---|---|
| Children (N = 803) | Adolescents (N = 802) | Total (N = 1605) | |
| Septal defects | 127 (15.8) | 71 (8.9) | 198 (12.3) |
| Atrial septal defect (patent foramen ovale, secundum, primum, or sinus venosus) | 46 | 29 | 75 |
| Ventricular septal defect (single or multiple) | 66 | 39 | 105 |
| Atrioventricular septal defect (complete or transitional) | 12 | 3 | 15 |
| Aortopulmonary window | 3 | 0 | 3 |
| Pulmonary venous anomalies | 6 (0.8) | 9 (1.1) | 15 (0.9) |
| Partial anomalous pulmonary venous connection | 3 | 3 | 6 |
| Total anomalous pulmonary venous connection | 3 | 6 | 9 |
| Right heart lesions | 138 (17.2) | 115 (14.3) | 253 (15.8) |
| Tetralogy of Fallot | 52 | 46 | 98 |
| Tetralogy of Fallot with pulmonary atresia | 16 | 9 | 25 |
| Tetralogy of Fallot with absent pulmonary valve | 6 | 3 | 9 |
| Pulmonary atresia with intact ventricular septum | 15 | 14 | 29 |
| Ebstein anomaly | 15 | 9 | 24 |
| Tricuspid stenosis | 1 | 0 | 1 |
| Pulmonary stenosis, valvar | 29 | 28 | 57 |
| Pulmonary artery stenosis, branch | 1 | 4 | 5 |
| Double-chambered right ventricle | 3 | 2 | 5 |
| Left heart lesions | 81 (10.1) | 105 (13.1) | 186 (11.6) |
| Aortic stenosis | 47 | 42 | 89 |
| Aortic insufficiency | 4 | 3 | 7 |
| Aortic insufficiency and aortic stenosis | 4 | 8 | 12 |
| Mitral stenosis | 3 | 5 | 8 |
| Mitral regurgitation | 6 | 12 | 18 |
| Mitral valve prolapse (without moderate or greater mitral regurgitation) | 2 | 8 | 10 |
| Bicuspid aortic valve (nonobstructive) | 13 | 27 | 40 |
| Other outflow-tract abnormalities in left ventricle | 2 | 0 | 2 |
| Single ventricle | 119 (14.8) | 81 (10.1) | 200 (12.5) |
| Hypoplastic left heart syndrome | 39 | 23 | 62 |
| Single-ventricle, double-inlet left ventricle | 20 | 11 | 31 |
| Single-ventricle, double-inlet right ventricle | 0 | 3 | 3 |
| Single-ventricle, mitral atresia (not hypoplastic left heart syndrome or double-outlet right ventricle/mitral atresia) | 10 | 10 | 20 |
| Single-ventricle, tricuspid atresia | 22 | 18 | 40 |
| Single-ventricle, unbalanced atrioventricular canal | 12 | 9 | 21 |
| Single-ventricle, right ventricle-aorta with pulmonary atresia | 13 | 6 | 19 |
| Single-ventricle, superoinferior ventricle (with or without straddling tricuspid valve) | 3 | 1 | 4 |
| Conotruncal anomalies | 75 (9.3) | 64 (8.0) | 139 (8.7) |
| Transposition of great arteries | 54 | 46 | 100 |
| Double-outlet right ventricle | 11 | 6 | 17 |
| Truncus arteriosus | 10 | 12 | 22 |
| Thoracic arteries and veins | 94 (11.7) | 90 (11.2) | 184 (11.5) |
| Coronary artery anomaly | 8 | 6 | 14 |
| Coarctation of aorta | 65 | 62 | 127 |
| Interrupted aortic arch | 5 | 6 | 11 |
| Patent ductus arteriosus | 7 | 2 | 9 |
| Vascular rings and slings (with or without tracheal stenosis) | 1 | 1 | 2 |
| Aortic root/ascending aorta dilation | 8 | 13 | 21 |
| Electrophysiologic conditions | 121 (15.1) | 193 (24.1) | 314 (19.6) |
| Sinus node dysfunction | 2 | 0 | 2 |
| First-degree atrioventricular block | 0 | 2 | 2 |
| Second-degree atrioventricular block | 2 | 3 | 5 |
| Complete atrioventricular block | 26 | 29 | 55 |
| Supraventricular tachycardia | 41 | 67 | 108 |
| Junctional ectopic tachycardia | 0 | 2 | 2 |
| Ventricular tachycardia | 7 | 16 | 23 |
| Long QT syndrome | 37 | 54 | 91 |
| Premature ventricular contractions | 4 | 5 | 9 |
| Vasodepressor syncope | 2 | 15 | 17 |
| Cardiomyopathy | 29 (3.6) | 57 (7.1) | 86 (5.4) |
| Cardiomyopathy, dilated | 17 | 30 | 47 |
| Cardiomyopathy, hypertrophic | 7 | 24 | 31 |
| Cardiomyopathy, restrictive | 5 | 3 | 8 |
| Other | 13 (1.6) | 17 (2.1) | 30 (1.9) |
| Pericarditis | 1 | 1 | 2 |
| Endocarditis | 2 | 3 | 5 |
| Kawasaki disease | 5 | 6 | 11 |
| Aneurysm, ventricular | 1 | 1 | 2 |
| Cardiac tumor | 1 | 3 | 4 |
| Pulmonary hypertension, primary | 2 | 3 | 5 |
| Myocardial infraction | 1 | 0 | 1 |
The original diagnostic category and current cardiac status exhibited similar trends between age groups. The most-prevalent original diagnostic category was biventricular congenital HD without aortic arch obstruction (eg, septal defects, transposition of the great arteries, or tetralogy of Fallot [46%–52%]), followed by acquired HD with a structurally normal heart (eg, electrophysiologic diagnoses or cardiomyopathy [20%–34%]). Consistent with the original diagnostic category, repaired biventricular congenital HD was the most-common current cardiac status (43%–49%). A substantial minority (16%) of children had palliated single-ventricle congenital HD culminating in a Fontan operation; however, this current cardiac status was less common among adolescents (9%). Heart transplant recipients constituted only 3% to 4% of the study population.
Validity Analyses
Association of PCQLI Scores With Severity of Congenital HD
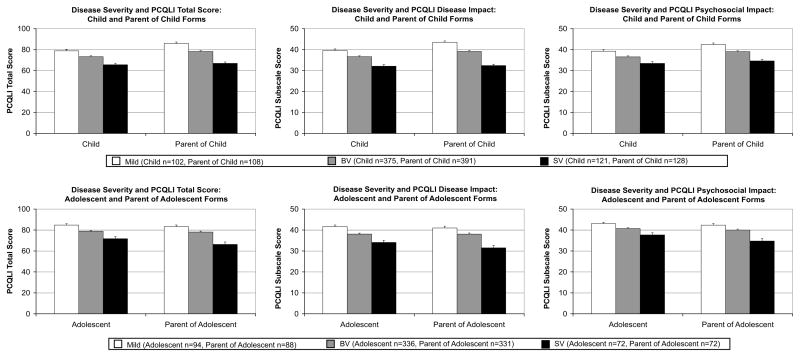
PCQLI scores differed significantly among disease severity subgroups (P < .001) (Fig 1). Furthermore, patients in the repaired biventricular HD subgroup had significantly lower PCQLI total and subscale scores than did patients in the mild HD subgroup, and patients in the palliated single-ventricle subgroup had significantly lower PCQLI total and subscales scores than did patients in the repaired biventricular HD and mild HD subgroups (P ≤ .036). These results were consistent across all age categories and respondent types and confirmed quantitatively the widespread, qualitative, clinical observations regarding the association of lower HRQoL with increased disease severity.
FIGURE 1.
Association of severity of congenital HD with PCQLI scores. Values are expressed as means, with error bars representing SEMs. All 3-way (P < .001) and 2-way (P ≤.036) comparisons were statistically significant. BV indicates repaired biventricular HD subgroup; SV, palliated single-ventricle subgroup; mild, mild HD subgroup.
Variation of PCQLI Scores With Medical Care Utilization
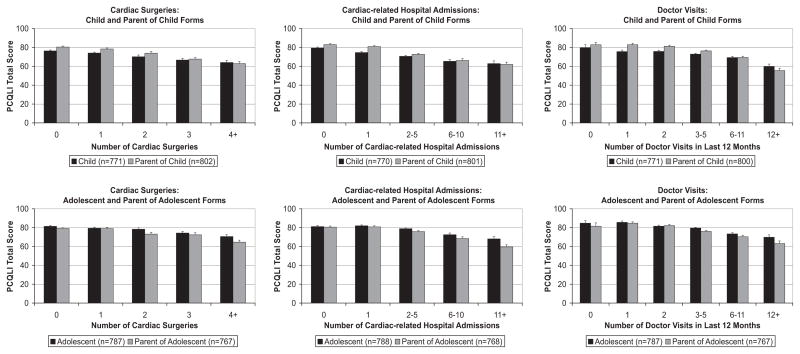
Increased numbers of cardiac operations, HD-related hospital admissions, and physician visits in the previous 12 months were associated with lower PCQLI total scores (P < .001) (Fig 2). These results were consistent across all 4 forms.
FIGURE 2.
Variation of PCQLI total scores with medical care utilization factors. Values are expressed as means, with error bars representing SEMs. All comparisons were significant (P < .001).
Comparison of Patient and Parent PCQLI Scores
Comparison of child and parent-of-child PCQLI scores revealed moderate correlations, as follows: disease impact subscale scores, 0.55; psychosocial impact subscale scores, 0.41; total scores, 0.50 (N = 759; P < .001). Comparison of adolescent and parent-of-adolescent PCQLI scores revealed moderate to good correlations, as follows: disease impact subscale scores, 0.61; psychosocial impact subscale scores, 0.45; total scores, 0.56 (N = 748; P < .001).
Association of PCQLI Scores With Generic HRQoL and Non–QoL Scores
The associations between PCQLI scores and PedsQL, SPPC/SPPA, and YSR/CBCL scores were statistically significant (P < .0001) (Table 3). Good correlations existed between PCQLI and PedsQL scores; correlation coefficients ranged from 0.61 (child PCQLI disease impact subscale scores versus PedsQL physical health summary scores) to 0.76 (adolescent PCQLI total scores versus PedsQL total scores). PCQLI total scores were moderately correlated with SPPC/SPPA global self-worth scores for both age groups. Fair correlations between PCQLI total scores and YSR/CBCL total competency scores were noted, and fair to moderate inverse correlations existed between PCQLI total scores and both YSR/CBCL internalizing problems summary scale scores and DSM-IV scale scores for all groups.
TABLE 3.
Association of PCQLI Scores With Generic HRQoL and Non–QoL Scores
| Instrument Score | PCQLI Score | Spearman Correlation Coefficients
|
|||
|---|---|---|---|---|---|
| Child | Parent of Child | Adolescent | Parent of Adolescent | ||
| PedsQL | N = 760 | N = 765 | N = 778 | N = 765 | |
| Total | Total | 0.70 | 0.70 | 0.76 | 0.72 |
| Physical health summary | Disease impact subscale | 0.61 | 0.67 | 0.67 | 0.68 |
| Psychosocial health summary | Psychosocial impact subscale | 0.64 | 0.62 | 0.69 | 0.64 |
| SPPC/SPPA | N = 758 | N = 743 | |||
| Global self-worth | Total | 0.46 | 0.43 | ||
| YSR/CBCL | N = 277a | N = 754 | N = 759 | N = 733 | |
| Total competency | Total | 0.33 | 0.33 | 0.28 | 0.37 |
| Internalizing problems | Total | −0.51 | −0.52 | −0.58 | −0.54 |
| DSM-IV | |||||
| Affective disorder | Total | −0.54 | −0.47 | −0.56 | −0.52 |
| Anxiety disorder | Total | −0.41 | −0.45 | −0.41 | −0.41 |
| Somatic disorder | Total | −0.33 | −0.34 | −0.39 | −0.38 |
| Attention-deficit/hyperactivity disorder | Total | −0.34 | −0.30 | −0.30 | −0.37 |
All Spearman correlation coefficients were statistically significant (P < .001).
The child sample for the YSR was limited to children 11 to 12 years of age, because the YSR is designed for patients 11 to 18 years of age.
Reliability Analyses
The median interim time between baseline testing and retesting was 18 days (range: 10–42 days). Test-retest reliability correlations were excellent (correlations: ≥ 0.70), and all Spearman intrarater correlation coefficients were statistically significant (P < .001) (Table 4).
TABLE 4.
PCQLI Test-Retest Reliability
| PCQLI Form | N | Spearman Correlation Coefficients
|
||
|---|---|---|---|---|
| PCQLI Disease Impact Subscale Score | PCQLI Psychosocial Impact Subscale Score | PCQLI Total Score | ||
| Child | 291 | 0.82 | 0.78 | 0.82 |
| Parent of child | 290 | 0.87 | 0.82 | 0.86 |
| Adolescent | 194 | 0.87 | 0.84 | 0.88 |
| Parent of adolescent | 190 | 0.90 | 0.83 | 0.89 |
All Spearman correlation coefficients were statistically significant (P < .001).
DISCUSSION
This large, multicenter study showed that lower HRQoL was associated with greater disease severity and medical care utilization, poorer patient self-perception and competency, and increased behavioral and emotional problems in the pediatric cardiac population. These data substantiate the validity and reliability of PCQLI scores in a geographically diverse patient population and support the use of the PCQLI for HRQoL assessment among children and adolescents (8–18 years of age) with congenital or acquired HD in clinical and research settings.11
PCQLI assessment may identify opportunities to improve the HRQoL of pediatric patients with HD. PCQLI scores were inversely related to increasing medical care utilization (cardiac operations, HD-related hospitalizations, and physician visits in the previous 12 months). This relationship parallels findings by Josie et al22 for the pediatric asthma population. Because the medical care utilization required to maximize overall outcomes varies according to patient, disease type, and treatment group, the PCQLI may identify opportunities to alter the timing, manner, and intensity of care to best balance required care and HRQoL. In addition, the PCQLI may identify patient predictors of lower HRQoL that may be more amenable to modification than clinical factors. In particular, psychosocial factors may allow for intervention, because lower PCQLI scores were associated with poorer self-perception, lower competency, and increased behavioral and emotional problems. Identification of modifiable psychosocial, neurodevelopmental, and physical morbidity factors with the PCQLI may permit interventions that improve HRQoL in the pediatric HD population.
PCQLI scores may be used to improve patient care and HRQoL. In addition to identifying patients who have poorer HRQoL scores, the PCQLI may detect areas for improvement in communication between patients, parents, and care providers that may affect overall management.23 Moons et al6 determined that, of 70 QoL studies concerning the pediatric cardiac population published between 1980 and 2003, only 1 measured patient-perceived HRQoL. This lack of patient self-assessment is problematic, because the perceptions of patients and parents regarding patient HRQoL are similar but not the same. PCQLI assessment of the perceptions of both patients and parents might be valuable to practitioners because it would identify instances of disparate perspectives.23 Understanding the perspectives of the patient, parent, and practitioner is crucial to the development of a patient care plan and may improve treatment adherence.24–26
In conjunction with its clinical uses, the PCQLI may improve patient-centered research. The variation between PCQLI and PedsQL scores suggests that different information is obtained by using the disease-specific PCQLI measure. From a research perspective, this implies that use of both generic and disease-specific HRQoL assessment tools would be advisable. A generic measure would allow for comparison of HRQoL between a disease-specific group and healthy children and/or children with other chronic diseases. In addition, a disease-specific measure such as the PCQLI may detect small but important changes in HRQoL over time or differences among clinically relevant subgroups and/or treatment regimens.
The ability of a disease-specific HRQoL instrument to distinguish among clinically meaningful subgroups within a particular disease population is critically important. Disease-specific HRQoL measures used in adult cardiac27,28 and chronic pediatric noncardiac22,29,30 disease populations can discriminate between discrete disease-specific subgroups across a wide age range, irrespective of respondent type (patient or parent). The pediatric cardiac disease-specific HRQoL tools currently available (ConQoL Index and PedsQL 3.0 cardiac module) can distinguish among disease severity subgroups but only within select subsets of a study population (eg, age or respondent type).9,10 In contrast, PCQLI total and subscale scores can differentiate between congenital HD severity subgroups irrespective of age category, score examined, or respondent type. From a research perspective, this is an important development that should facilitate cross-sectional and prospective studies of HRQoL in clinically important subgroups in the pediatric HD population.
This study has several inherent limitations. The study population was predominantly white, with few Hispanic, Asian, or Native American participants, and was restricted to English-speaking patients and parents. The majority of patients lived in 2-parent households. In addition, parent respondents were predominantly mothers. Furthermore, the parent respondent population had above-average socioeconomic status and educational backgrounds. Because any of these factors may potentially restrict the external validity of the PCQLI, additional research is required to substantiate the PCQLI for general application.
CONCLUSIONS
PCQLI scores were inversely correlated with increased disease severity and medical care utilization, poorer self-perception and competency, and increased behavioral and emotional problems. PCQLI scores were valid and reliable for children and adolescents (8–18 years of age) with HD. The ability of the PCQLI to distinguish among population subgroups irrespective of score examined, age category, or respondent type represents an advance over currently available pediatric, cardiac disease-specific, HRQoL instruments. The external validity and responsiveness of PCQLI scores are being evaluated currently in multicenter studies. The PCQLI should be useful in clinical screening, surveillance, and evaluation for clinical management and future research studies for children and adolescents with congenital or acquired HD.
WHAT’S KNOWN ON THIS SUBJECT
Little is known regarding the impact of important clinical and patient factors (disease severity, medical care utilization, patient-parent consensus, and patient self-perception, competency, and behavior) on HRQoL in the pediatric cardiac population.
WHAT THIS STUDY ADDS
This large, multicenter study showed that lower HRQoL was associated with greater disease severity and medical care utilization, poorer patient self-perception and competency, and increased behavioral and emotional problems in the pediatric cardiac population. The PCQLI is valid and reliable.
Acknowledgments
Research and project support was provided by the National Institute of Child Health and Human Development (grant 5-K23-HD048637), the American Heart Association, Pennsylvania/Delaware Affiliate (now Great Rivers Affiliate) (Beginning Grant-in-Aid 0465467U), the Cincinnati Children’s Hospital Research Foundation, and the Children’s Hospital of Philadelphia Institutional Development Fund.
The following persons participated in patient enrollment, data collection, study coordination, or manuscript preparation: Cincinnati Children’s Hospital Medical Center: Richard Ittenbach, PhD, Shawna Hottinger, MS, Katelyn Mellion, BS, Kaleigh Coughlin, BA, Brett Morgan, BS, Loran Carroll, BS, Melanie Riedel, BS, Michael Whalen, Aimee Baker, MA, Baiyang Wang, MS; Children’s Hospital of Philadelphia: Linda Hurd, MSN, CRNP, Janice Prodell, RN, Lynda Ahearn, RN, Lydia Kruge, BA, Anita Pudusseri, BS, Darryl Powell, Andrew Schissler, BSE, Josie Welkom, BA, Stanley O. Dunn, BA; Children’s Hospital Boston: Annette Baker, MSN, CRNP, Jill Cotter, BA, Danielle Martin, BA, Erica Denhoff, BA, Ellen McGrath, BSN; University of California, San Francisco, Children’s Hospital: David Teitel, MD, Laura Robertson, MD; Phoenix Children’s Hospital: Melissa Hill, PA-C; Children’s Hospital of Wisconsin: Stuart Berger, MD, Nancy Ghanayem, MD, Lisa Young-Borkowski, MSN, Mary Krolikowski, MSN, Angie Klemm, Mara Koffarnus; University of Texas Southwestern Medical Center at Dallas: Gloria Williams, BA.
Funded by the National Institutes of Health (NIH).
ABBREVIATIONS
- CBCL
Child Behavior Checklist
- HD
heart disease
- HRQoL
health-related quality of life
- PCQLI
Pediatric Cardiac Quality of Life Inventory
- PedsQL
Pediatric Quality of Life Inventory
- QoL
quality of life
- SPPA
Self-Perception Profile for Adolescents
- SPPC
Self-Perception Profile for Children
- YSR
Youth Self-Report
- DSM-IV
Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition
Footnotes
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose.
References
- 1.Lloyd-Jones D, Adams R, Carnethon M, et al. Heart disease and stroke statistics: 2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2009;119(3):480–486. doi: 10.1161/CIRCULATIONAHA.108.191259. [DOI] [PubMed] [Google Scholar]
- 2.Bellinger DC, Wypij D, duPlessis AJ, et al. Neurodevelopmental status at eight years in children with dextro-transposition of the great arteries: the Boston Circulatory Arrest Trial. J Thorac Cardiovasc Surg. 2003;126(5):1385–1396. doi: 10.1016/s0022-5223(03)00711-6. [DOI] [PubMed] [Google Scholar]
- 3.Mahle WT, Clancy RR, Moss EM, Gerdes M, Jobes DR, Wernovsky G. Neurodevelopmental outcome and lifestyle assessment in school-aged and adolescent children with hypoplastic left heart syndrome. Pediatrics. 2000;105(5):1082–1089. doi: 10.1542/peds.105.5.1082. [DOI] [PubMed] [Google Scholar]
- 4.Drotar D. Measuring Health-Related Quality of Life in Children and Adolescents. Mahwah, NJ: Erlbaum; 1998. [Google Scholar]
- 5.Higginson IJ, Carr AJ. Measuring quality of life: using quality of life measures in the clinical setting. BMJ. 2001;322(7297):1297–1300. doi: 10.1136/bmj.322.7297.1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Moons P, Van Deyk K, Budts W, De Geest S. Caliber of quality-of-life assessments in congenital heart disease: a plea for more conceptual and methodological rigor. Arch Pediatr Adolesc Med. 2004;158(11):1062–1069. doi: 10.1001/archpedi.158.11.1062. [DOI] [PubMed] [Google Scholar]
- 7.Janse AJ, Uiterwaal CS, Gemke RJ, Kimpen JL, Sinnema G. A difference in perception of quality of life in chronically ill children was found between parents and pediatricians. J Clin Epidemiol. 2005;58(5):495–502. doi: 10.1016/j.jclinepi.2004.09.010. [DOI] [PubMed] [Google Scholar]
- 8.Kendall L, Lewin RJ, Parsons JM, Veldtman GR, Quirk J, Hardman GE. Factors associated with self-perceived state of health in adolescents with congenital cardiac disease attending paediatric cardiologic clinics. Cardiol Young. 2001;11(4):431–438. doi: 10.1017/s1047951101000555. [DOI] [PubMed] [Google Scholar]
- 9.Macran S, Birks Y, Parsons J, et al. The development of a new measure of quality of life for children with congenital cardiac disease. Cardiol Young. 2006;16(2):165–172. doi: 10.1017/S1047951106000102. [DOI] [PubMed] [Google Scholar]
- 10.Uzark K, Jones K, Burwinkle TM, Varni JW. The Pediatric Quality of Life Inventory in children with heart disease. Prog Pediatr Cardiol. 2003;18(2):141–148. [Google Scholar]
- 11.Marino BS, Shera D, Wernovsky G, et al. The development of the Pediatric Cardiac Quality of Life Inventory: a quality of life measure for children and adolescents with heart disease. Qual Life Res. 2008;17(4):613–626. doi: 10.1007/s11136-008-9323-8. [DOI] [PubMed] [Google Scholar]
- 12.Uzark K, Jones K, Slusher J, Limbers CA, Burwinkle TM, Varni JW. Quality of life in children with heart disease as perceived by children and parents. Pediatrics. 2008;121(5) doi: 10.1542/peds.2006-3778. Available at: www.pediatrics.org/cgi/content/full/121/5/e1060. [DOI] [PubMed] [Google Scholar]
- 13.Varni JW, Seid M, Rode CA. The PedsQL: measurement model for the Pediatric Quality of Life Inventory. Med Care. 1999;37(2):126–139. doi: 10.1097/00005650-199902000-00003. [DOI] [PubMed] [Google Scholar]
- 14.Harter S. Manual for the Self-Perception Profile for Children. Denver, CO: University of Denver; 1985. [Google Scholar]
- 15.Harter S. Manual for the Self-Perception Profile for Adolescents. Denver, CO: University of Denver; 1985. [Google Scholar]
- 16.Achenbach T. Child Behavior Checklist for Ages 6–18. Burlington, VT: University of Vermont; 2001. [Google Scholar]
- 17.Achenbach T. Youth Self-Report for Ages 1 1–18. Burlington, VT: University of Vermont; 2001. [Google Scholar]
- 18.Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33(1):159–174. [PubMed] [Google Scholar]
- 19.Holm S. A simple sequentially rejective multiple test procedure. Scand J Stat. 1979;6(2):65–70. [Google Scholar]
- 20.Zimmerman DW, Williams RH. Properties of the Spearman correction for attenuation for normal and realistic non-normal distributions. Appl Psychol Measure. 1997;21(3):271–278. [Google Scholar]
- 21.Nunnally JC. Psychometic Theory. 2. New York, NY: McGraw-Hill; 1978. [Google Scholar]
- 22.Josie KL, Greenley RN, Drotar D. Health-related quality-of-life measures for children with asthma: reliability and validity of the Children’s Health Survey for Asthma and the Pediatric Quality of Life Inventory 3.0 Asthma Module. Ann Allergy Asthma Immunol. 2007;98(3):218–224. doi: 10.1016/S1081-1206(10)60710-7. [DOI] [PubMed] [Google Scholar]
- 23.Marino BS, Tomlinson RS, Drotar D, et al. Quality-of-life concerns differ among patients, parents, and medical providers in children and adolescents with congenital and acquired heart disease. Pediatrics. 2009;123(4) doi: 10.1542/peds.2008-2572. Available at: www.pediatrics.org/cgi/content/full/123/4/e708. [DOI] [PubMed] [Google Scholar]
- 24.McAllister S, Buckner EB, White-Williams C. Medication adherence after heart transplantation: adolescents and their issues. Prog Transplant. 2006;16(4):317–323. doi: 10.1177/152692480601600406. [DOI] [PubMed] [Google Scholar]
- 25.Byrne A, MacDonald J, Buckley S. Reading, language and memory skills: a comparative longitudinal study of children with Down syndrome and their mainstream peers. Br J Educ Psychol. 2002;72(4):513–529. doi: 10.1348/00070990260377497. [DOI] [PubMed] [Google Scholar]
- 26.Lawrence K, Stilley CS, Olshansky E, Bender A, Webber SA. Further exploration: maturity and adherence in adolescent and young adult heart transplant recipients. Prog Transplant. 2008;18(1):50–54. doi: 10.1177/152692480801800110. [DOI] [PubMed] [Google Scholar]
- 27.Middel B, Bouma J, de Jongste M, et al. Psychometric properties of the Minnesota Living with Heart Failure Questionnaire (MLHF-Q) Clin Rehabil. 2001;15(5):489–500. doi: 10.1191/026921501680425216. [DOI] [PubMed] [Google Scholar]
- 28.Green CP, Porter CB, Bresnahan DR, Spertus JA. Development and evaluation of the Kansas City Cardiomyopathy Questionnaire: a new health status measure for heart failure. J Am Coll Cardiol. 2000;35(5):1245–1255. doi: 10.1016/s0735-1097(00)00531-3. [DOI] [PubMed] [Google Scholar]
- 29.Quittner AL, Buu A, Messer MA, Modi AC, Watrous M. Development and validation of the Cystic Fibrosis Questionnaire in the United States: a health-related quality-of-life measure for cystic fibrosis. Chest. 2005;128(4):2347–2354. doi: 10.1378/chest.128.4.2347. [DOI] [PubMed] [Google Scholar]
- 30.Wallenstein GV, Carranza-Rosenzweig J, Kosinski M, Blaisdell-Gross B, Gajria K, Jhingran P. A psychometric comparison of three patient-based measures of asthma control. Curr Med Res Opin. 2007;23(2):369–377. doi: 10.1185/030079906X167426. [DOI] [PubMed] [Google Scholar]