Abstract
Central nervous system catheter infections are a serious complication in the treatment of hydrocephalus. These infections are commonly caused by Staphylococcus epidermidis and Staphylococcus aureus, both known to form biofilms on the catheter surface. Our objective was to generate a novel murine model of central nervous system catheter-associated biofilm infection using a clinical S. aureus isolate and characterize the nature of the inflammatory response during biofilm growth. Silicone catheters were precoated with S. aureus to facilitate bacterial attachment, whereupon infected or sterile catheters were stereotactically inserted into the lateral ventricle of the brain in C57BL/6 mice and evaluated at regular intervals through day 21 postinsertion. Animals tolerated the procedure well, with no clinical signs of illness or bacterial growth seen in the control group. Bacterial titers associated with central nervous system catheters were significantly elevated compared to those from the surrounding parenchyma, consistent with biofilm formation and minimal planktonic spread of infection. Catheter-associated bacterial burdens progressively increased, with maximal colonization achieved at day 7 postinfection. Analysis of inflammatory infiltrates by fluorescence-activated cell sorting (FACS) revealed significant macrophage and neutrophil influx, which peaked at days 3 and 5 to 7, respectively. In contrast, there were no detectable immune infiltrates associated with tissues surrounding sterile catheters. Biofilm infection led to significant increases in chemokine (CXCL1 and CCL2) and proinflammatory cytokine (interleukin 17 [IL-17]) expression in tissues surrounding infected central nervous system catheters. Based on these results, we propose this approach is a valid animal model for further investigations of catheter-associated central nervous system shunt infections.
INTRODUCTION
Cerebrospinal fluid (CSF) shunt placement for the treatment of hydrocephalus is one of the most common procedures performed by pediatric neurosurgeons in the United States, with tens of thousands of shunts implanted annually (29). Unfortunately, 30 to 40% of all CSF shunts in pediatric patients fail within the first year, resulting in a shunt revision to primary placement ratio of 3:1 in many health care centers (14, 29). One of the most common causes of shunt failure is infection, reported in 5 to 30% of cases (14). In addition to causing shunt failure, these catheter infections are associated with a higher risk of seizures, decreased intellectual performance, and a 2-fold increase in long-term mortality (14). Therefore, studies designed to advance our understanding as to how bacteria colonize these catheters and evade antimicrobial killing in the central nervous system could have a dramatic impact on the development of treatment modalities for these serious infections.
The most common organisms responsible for central nervous system catheter infections, Staphylococcus epidermidis and Staphylococcus aureus, are both known to form biofilms (14, 25). Biofilms are organized communities of bacteria that attach and aggregate on the catheter surface. Within biofilms, bacteria can survive in a protected environment, evading the host immune response and antimicrobial agents, presumably by a bacterially derived extracellular matrix and other immunomodulatory factors (25). The biofilm's ability to evade the host immune response and antimicrobial agents makes it difficult to manage central nervous system catheter infections nonsurgically, such that catheter removal is currently recommended for effective treatment (39).
Although an intracranial infection model, using New Zealand White rabbits, has been used to study rifampin-impregnated CSF shunt catheters, no other animal models for central nervous system catheter infection have been reported to date (15). Development of a mouse model represents a valuable new approach to identify important factors in the host immune response to central nervous system biofilms through the broad array of immunologic tools designed for use in rodents, as well as many genetically engineered strains of mice that will allow exploration of specific mechanisms of the immune response to staphylococcal infection.
Establishing a catheter model within the central nervous system was necessary, because the immune response in this compartment often differs from that seen in the periphery due to the inherent anti-inflammatory environment of the normal central nervous system (15). However, in response to injury or infection, the central nervous system is capable of mounting a robust immune response, albeit deviant in some respects, from similar insults in the periphery (15, 23, 27). Resident glial cells in the central nervous system parenchyma are activated in addition to peripheral immune cells, such as neutrophils and macrophages, which invade the central nervous system in response to infection (15, 27). A better understanding of the interactions between the immune system and the bacteria present in the biofilm is essential in order to make progress in the management of central nervous system catheter infections.
While bacterial growth characteristics and other adaptations required for biofilm formation are being extensively investigated by microbiologists, very little is known about host-biofilm interactions, particularly with regard to the immune response to catheter biofilm infections within the central nervous system. Therefore, to explore this issue, we have developed a novel model of central nervous system catheter infection in the mouse. This technique achieved consistent catheter-associated infections with S. aureus, mimicking what is seen in humans with ventricular shunt infections (14). Mice implanted with infected catheters demonstrated more pronounced innate immune cell infiltrates and elevated proinflammatory cytokine and chemokine levels compared to those implanted with sterile catheters. As confirmed by scanning electron microscopy, the majority of bacteria were associated with the catheter surface compared to surrounding tissues, confirming the establishment of bona fide biofilm infection. Based on these findings, we propose that this model serves as a powerful tool to identify important factors in the host immune response to central nervous system biofilms. Understanding the interactions between the neuroimmune system and the biofilms that form on infected catheters will allow us to explore novel management strategies for these central nervous system infections that are classically recalcitrant to conventional antibiotic therapy.
MATERIALS AND METHODS
Mouse strain.
Male C57BL/6 mice (The Jackson Laboratory, Bar Harbor, ME) were used at 8 to 9 weeks of age in all of the experiments described herein. Each experiment was performed independently at least 3 times with 4 to 5 mice per group (sterile or infected catheter). The animal use protocol was approved by the University of Nebraska Medical Center Institutional Animal Care and Use Committee and is in accord with the National Institutes of Health (NIH) guidelines for the use of rodents.
Catheter preparation and implantation.
All experiments were performed using a strain of methicillin-susceptible Staphylococcus aureus (MSSA) obtained from a pediatric patient with a central nervous system catheter infection at Arkansas Children's Hospital, specifically an infection of a ventriculo-peritoneal shunt (ACH1719). This strain is an hlb lysogen that is hemolytic on sheep blood agar. Among genes associated with virulence and known to be part of the accessory genome, sarT, sdrC, sdrE, scn, sak, and chp are present in this strain, while cna, pvl, sdrD, and sea are not. Hollow bore silicone catheters (2 mm in length, 1 mm in diameter; Cook Medical Inc., Bloomington, IN) were incubated overnight in mouse serum to facilitate bacterial adhesion to the catheter. Subsequently, catheters were incubated for 4 h with 2 × 104 CFU/ml S. aureus, which reproducibly resulted in catheter colonization of 5 to 10 × 103 CFU/catheter. Catheters incubated in serum alone were used as controls to evaluate the foreign body response in the uninfected mice.
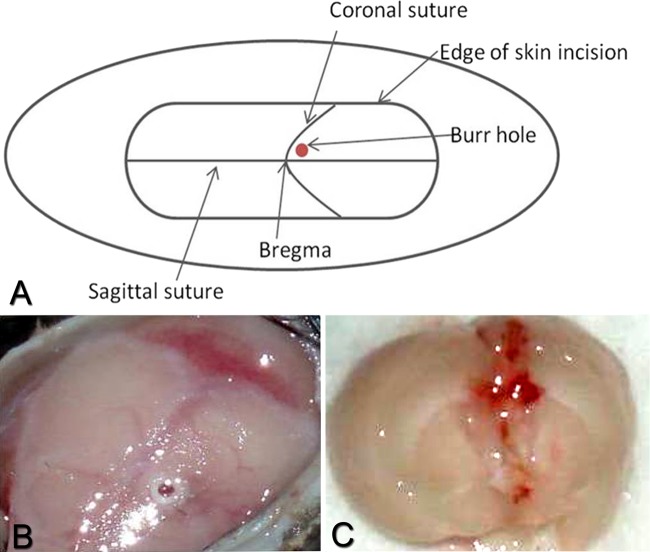
Catheters were placed into the lateral brain ventricle under general ketamine and xylazine anesthesia (100 to 200 mg/kg and 5 to 13 mg/kg intraperitoneally [i.p.], respectively). A 1-cm longitudinal incision was made in the scalp to expose the underlying skull sutures. A rodent stereotaxic apparatus (Stoelting, Wood Dale, IL) was used for accurate catheter placement into the left ventricle using the following coordinates obtained from reference 31 and include (relative to bregma) +0.02 mm rostral, +1.0 mm lateral, and −2.0 mm deep from the surface of the brain. A small burr hole was made at these coordinates and the catheter gently inserted vertically. The leading edge of the catheter was slightly beveled to ease insertion. Following catheter placement, the burr hole was sealed with bone wax and the skin incision closed using VetBond surgical glue (3M, St. Paul, MN).
Scanning electron microscopy.
Infected catheters were carefully removed from mice at day 10 postinfection. The catheter segments were longitudinally bisected and fixed in 10% neutral buffered formalin with postfixation in 2% paraformaldehyde with 2.5% glutaraldehyde and secondary fixation in 1% osmium tetroxide. They were then washed in phosphate buffer, dehydrated in a graded ethanol series, and dried. Fixed specimens were mounted on aluminum stubs with carbon tabs and colloidal silver paste and sputter coated with gold-palladium using a Hummer VI sputter coater (Anatech, Battle Creek, MI). Samples were viewed using a Quanta 200 scanning electron microscope (FEI, Hillsboro, OR) operated at 25 kV.
Quantitation of viable bacteria from central nervous system parenchyma and catheters.
Catheters and surrounding brain tissues were collected at days 1, 3, 5, 7, 10, 14, and 21 following catheter implantation from a total of 4 to 5 mice per group. The homogenization of brain tissues for bacterial titer determination and inflammatory mediator measurement is well established and provides the opportunity to procure samples for quantitative culture and protein and RNA analysis from a single animal (22). To prepare homogenates for bacterial culture, as well as downstream inflammatory mediator analysis, catheters and the surrounding lesion sites were sectioned with 1 to 2 mm on all sides. The tissue was then homogenized in 500 μl sterile phosphate-buffered saline (PBS) supplemented with a complete protease inhibitor cocktail tablet (Roche) and 160 units/ml RNase inhibitor (Promega, Madison, WI) using a Polytron homogenizer (Brinkmann Instruments, Westbury, NY). A 20-μl aliquot was removed for culture via serial 10-fold dilutions on tryptic soy agar (TSA) plates supplemented with 5% sheep blood. The remaining slurry was centrifuged, with the resulting supernatant collected for future cytokine analysis. Catheters were sonicated in sterile PBS to remove adherent bacteria, the titer was determined, and bacteria were inoculated onto blood agar plates. This method has been successfully utilized to characterize bacterial burdens in a subcutaneous catheter biofilm model and has been readily adapted to our central nervous system model (4).
ELISAs.
Inflammatory mediators were measured in supernatants of homogenized catheter-associated brain tissues as previously described (2, 20, 21). The brain tissue surrounding the catheter was collected and homogenized as outlined above for quantitation of bacterial growth. After homogenization, samples were centrifuged and inflammatory mediators measured in the resultant supernatant. Commercially available enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems, Minneapolis, MN) were used to evaluate samples for CXCL1, CXCL2, and interleukin 17 (IL-17). Mediator levels were adjusted based on the amount of total protein extracted from brain tissues for normalization as previously described (2, 20, 21).
Immunohistochemical staining and confocal microscopy.
To evaluate the nature of inflammatory infiltrates during central nervous system biofilm infection, brain tissues were collected for immunohistochemical staining and confocal microscopy. Neutrophil and macrophage/microglia accumulations in the brain tissues surrounding the catheters were evaluated using Ly-6G (US Biological, Swampscott, MA) and Iba-1 (BioCare Medical, Concord, CA) immunofluorescence staining, respectively. Brain tissues from mice in each experimental group (infected versus sterile catheter) were removed at days 7, 10, 14, and 21 following implantation. Prior to removal of the brain tissue, the mice underwent transcardiac perfusion with PBS to remove leukocytes from the vasculature and then 4% paraformaldahyde for tissue fixation. The brains were then immersed in serial concentrations of sucrose-PBS (5%, 10%, 30%) and stored at 4°C for 18 to 24 h before freezing in optimal cutting tissue (OCT) medium on dry ice. Serially interrupted (50 μm) 10-μm cryostat sections of brain tissues were prepared, mounted onto SuperFrost glass slides (Fisher Scientific, Houston, TX), and air dried for 30 min at room temperature prior to storage at −80°C. To initiate staining, tissues were equilibrated to room temperature for 10 min and fixed in ice-cold methanol. Catheter-associated tissues were incubated with the appropriate primary antibodies overnight at 4°C in a humidified chamber. Following numerous rinses in PBS, tissues were incubated with the appropriate secondary antibodies (Abs), when indicated, for 1 h at room temperature. Upon completion of the staining protocol, slides were coverslipped using the Prolong antifade reagent (Invitrogen, Carlsbad, CA) and imaged using a Zeiss laser scanning confocal microscope. The specificity of antibody staining was confirmed by the absence of fluorescent signal following incubation of tissues with secondary antibodies alone.
Quantitation of cellular infiltrates by FACS.
To quantitate immune cell infiltrates during central nervous system biofilm infection, cells associated with sterile and infected catheters were recovered from the brain parenchyma and quantitated by fluorescence-activated cell sorting (FACS) analysis as previously described (23, 30). Briefly, mice were perfused to remove leukocytes from the vasculature, and the entire ipsilateral hemisphere was removed for analysis. Tissues were minced and filtered through a 70-μm nylon mesh cell strainer. The resulting slurry was then subjected to collagenase/DNase digestion to facilitate immune cell retrieval and centrifuged on a discontinuous Percoll gradient as previously described to remove cell debris and red blood cells (RBCs) (23, 30). Upon isolation, cells underwent extensive washing and incubation in FcBlock (BD Biosciences, San Diego, CA) to minimize nonspecific binding to Fc receptors. Cells were then stained with an antibody panel to discriminate neutrophils (Ly-6G+, CD11b+, CD45high), macrophages (Ly-6G−, CD11b+, CD45high) and microglia (Ly-6G−, CD11b+, CD45low-intermediate) (30, 34) and analyzed using a BD FACSAria with compensation set based on the staining of single fluorochromes alone and correction for autofluorescence using unstained cells. Controls included cells stained with directly conjugated isotype control antibodies to assess the degree of nonspecific staining.
Generation of S. aureus static biofilms in vitro.
The ability of MSSA ACH1719 to form biofilms in vitro was assessed using a microtiter plate assay as previously described (3). Briefly, overnight cultures of MSSA ACH1719, UAMS-1, and USA300 LAC were incubated with tryptic soy broth (TSB) in a 96-well polystyrene microtiter plate coated with mouse serum. The latter two strains were used for comparisons with MSSA ACH1719, since they are well-established biofilm-producing clinical isolates (3, 38). The plates were incubated for 24 h at 37°C and then gently washed to remove any nonadherent bacteria. The remaining biofilm was then fixed with 100% EtOH and stained with crystal violet for visualization. For confocal microscopy analysis, static biofilms were generated on sterile glass chamber slides (Fisher Scientific, Houston, TX) treated with 20% human plasma (generous gift of Steve Carson, UNMC) in sterile carbonate-bicarbonate buffer overnight (9). The plasma-coating buffer was then removed, and each chamber was inoculated with 2 ml ACH1719, diluted to an optical density at 600 nm (OD600) value of approximately 0.050. The slides were incubated at 37°C under static aerobic conditions for 2 days, with fresh medium replaced every 24 h. In preparation for confocal microscopy, medium was removed and replaced with 1 ml of a 1:100 dilution of Syto9 in PBS (Invitrogen, Carlsbad, CA). After incubating for 30 min, the stain was removed and the biofilm was visualized using a Zeiss laser scanning confocal microscope.
Statistics.
Significant differences between experimental groups were determined with Sigma Stat, using the unpaired Student t test at the 95% confidence interval. As repeated measurements are not made on the same animal given the necessary sacrifice of the mouse for tissue collected, this was determined to be more appropriate than analysis of variance (ANOVA).
RESULTS
Establishment of a murine model of central nervous system catheter infection.
This murine model of central nervous system catheter infection was developed to study the complex interactions between central nervous system immunity and biofilm formation in vivo, taking advantage of the multiple immunologic tools available for use in the mouse. As described earlier, all experiments were performed using a methicillin-susceptible S. aureus isolate obtained from a pediatric patient with a central nervous system catheter infection (ACH1719). This isolate was selected because it caused a catheter-associated biofilm within the central nervous system, increasing the clinical relevance of this model. Importantly, when evaluated using microtiter plate assays with crystal violet staining, this isolate is capable of forming in vitro biofilms similar to that seen with the UAMS-1 and USA300 LAC strains of S. aureus, both well-characterized biofilm-producing clinical strains of S. aureus (Fig. 1A) (4, 37). Static biofilm growth in glass chamber slides also demonstrated a thick lawn of biofilm formation when stained with the vital stain Syto9 (Fig. 1B and C).
Fig 1.
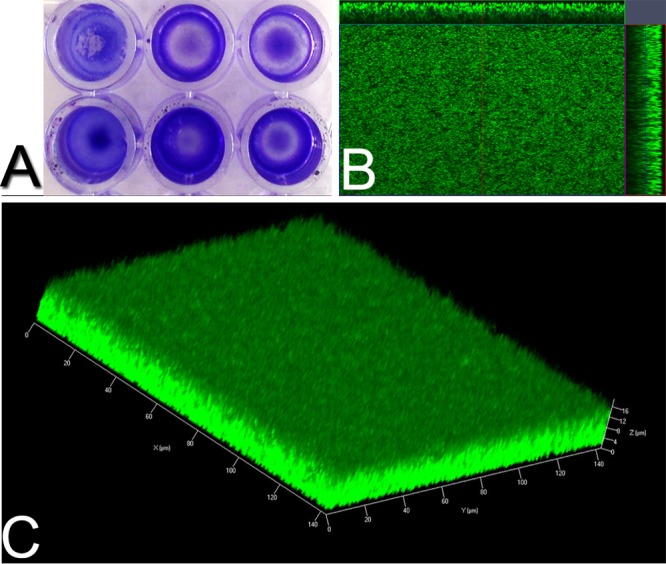
S. aureus strain ACH1719 is capable of forming biofilm in vitro. (A) In microtiter plate cultures (performed in triplicate), ACH1719 forms biofilm grossly equivalent to that observed with USA300 LAC (not shown) and UAMS-1. Static glass chamber biofilm culture stained with Syto9 (green) and scanned at 1-μm intervals demonstrates a thick lawn of biofilm growth with ACH1719. (B) Orthogonal view; (C) three-dimensional Z-stack reconstruction.
The silicone catheters utilized in this model were made of a material similar to that used in pediatric ventricular shunts and were precoated with mouse serum prior to incubation with bacteria, as previous work has demonstrated improved bacterial adherence and biofilm formation upon the addition of plasma proteins with S. aureus (3). To standardize the growth conditions for achieving reproducible catheter bacterial burdens, catheters were incubated with a range of S. aureus inoculums (102 to 108 CFU/ml) collected during exponential growth phase under aerobic conditions. Ultimately, we found that a 4-h incubation with 2 × 104 CFU/ml S. aureus produced the most consistent results, with a final titer of 6.2 × 103 CFU/catheter (±1.4 × 103 standard error of the mean [SEM]) cultured from infected catheters after rinsing and sonication (data not shown).
After precoating, catheters were inserted into the lateral ventricle using a rodent stereotaxic apparatus for consistent and accurate positioning. This location was selected because it allowed entry into the largest part of the lateral ventricle, mimicking placement seen in children (Fig. 2). Following catheter implantation, mice were monitored closely for changes in behavior, appearance, and weight. Mice implanted with sterile catheters recovered rapidly from the surgical procedure, with only decreased activity and mild weight loss observed on the first postoperative day but a return to baseline activity and weight gain on subsequent days. No abnormal movements, seizures, or other atypical behavior were noted in the sterile catheter group. In contrast, mice implanted with catheters precoated with S. aureus exhibited symptoms of illness, including decreased movement and ruffled fur, for the first week following implantation. Animals also revealed weight loss of up to 15% of their original body weight during this time period. By days 7 and 8, these mice began to improve and return to baseline appearance and activity levels. In addition, they returned to their original body weight. The mortality rate observed in the mice implanted with infected catheters was approximately 10% and was seen primarily in the first week postimplantation, as opposed to very rare mortality observed in mice implanted with sterile catheters.
Fig 2.
Overview of implantation procedure. Silicone catheters are precoated with mouse serum and incubated with 2 × 104 CFU/ml S. aureus for 4 h to facilitate bacterial attachment. Catheter fragments coated with S. aureus or serum alone were stereotactically inserted into the lateral ventricle of C57BL/6 mice. This location (A and B; from bregma: +0.02 mm rostral, +1 mm lateral, −2 mm deep) was chosen because the lateral ventricles are fairly large in diameter and more easily accessible at this location (C).
Surprisingly, 10 to 15% of mice implanted with S. aureus-coated catheters exhibited several unusual behaviors occurring between days 10 to 12 postinfection. Specifically, affected mice displayed mild seizure-like behaviors, with erratic and repetitive forelimb movement, as well as hyperactivity in some cases. This behavior resolved without intervention, and the mice behaved normally with no residual neurologic defect grossly observed for the remainder of their infection course (14 to 21 days).
Catheter-associated bacterial growth predominates in comparison to surrounding brain parenchyma.
An important issue to establish in the development of this model was confirming that bacteria were preferentially associated with the catheter, consistent with a biofilm-mediated foreign body infection. Electron microscopy of explanted catheters verified bona fide biofilm formation on the catheter surface at day 10 postinfection (Fig. 3A, B). While there was some unavoidable disruption of the biofilm mass resulting from catheter removal and processing, a large portion of the catheter surface was covered with bacteria (Fig. 3A). Higher magnification shows adherent bacteria as well as leukocytes near the catheter surface (Fig. 3B). Collectively, these findings demonstrate in vivo S. aureus biofilm formation in this model, allowing further evaluation of the innate immune response to biofilm infection within the central nervous system.
Fig 3.
Visualization of S. aureus biofilm growth on explanted catheters. Catheters were removed 10 days postinfection and visualized with scanning election microscopy. While some biofilm disruption occurred following catheter removal and processing, a large portion of the catheter surface is covered with bacteria (A; 200× magnification). Higher magnification of this area shows bacteria adhering to the catheter surface (white arrows) as well as white blood cells (*) (B; 3,000× magnification).
Catheters were removed from the brain tissue at various time points after insertion, rinsed in sterile PBS, and sonicated to disrupt the bacteria from the inner and outer catheter surfaces. In addition, the brain tissue surrounding catheters was recovered and homogenized to quantitate the degree of bacterial detachment and/or dissemination. Importantly, hematoxylin and eosin (H&E) staining confirmed that abscess formation rarely occurred in this infection model (data not shown). The cultures from both catheters and the parenchyma of mice implanted with sterile catheters were negative at all time points examined (days 1, 3, 5, 7, 10, 14, 21; data not shown). As shown in Fig. 4, mice receiving S. aureus-infected catheters demonstrated significantly higher bacterial titers on the catheter itself than on surrounding tissues, demonstrating that the infection is primarily catheter associated with limited secondary parenchymal spread, which agrees with the scanning electron microscopy results. Bacterial burdens in the parenchyma peaked at day 5 postinfection and began to resolve thereafter, correlating with the illness behavior and weight loss observed in the mice during the first week of infection.
Fig 4.
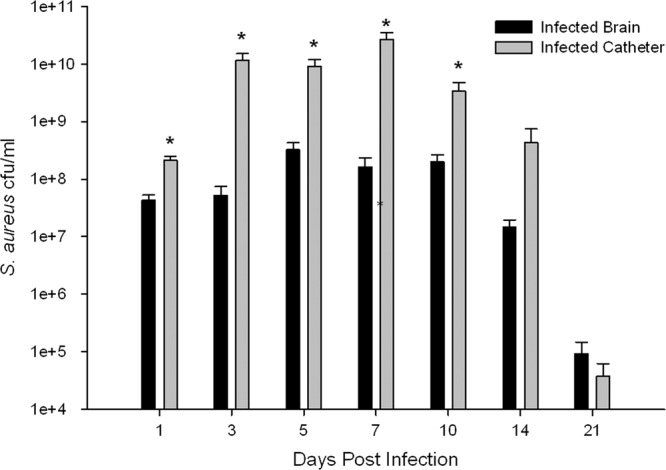
Catheter-associated bacterial growth is significantly elevated in comparison to infection in surrounding brain parenchyma. Catheters were removed from the brain tissue, rinsed in PBS, and then sonicated in 500 μl of PBS. The titer of this solution was then determined, and the solution was cultured on blood agar plates. Supernatants from homogenates of catheter-associated tissue were also cultured on blood agar plates. Results represent three replicates of independent experiments with 4 to 5 mice per group (sterile, infected) in each replicate (n = 12 to 15 total animals per group). The cultures from the catheters and parenchyma of the mice implanted with sterile catheters were negative and are not represented. *, P < 0.05.
At day 14 postimplantation, 40% of the infected mice appeared to have spontaneously cleared their infection from the catheter and surrounding parenchyma, and by day 21 postimplantation, 50% of the infected mice had undetectable bacterial burdens. The mice with persistent infection did not have greater weight loss or clinical signs of illness during their postimplantation course. As bacterial quantification occurs at the time of sacrifice, it is impossible to determine if the initial bacterial growth was lower in the mice with spontaneous resolution of infection. Alternatively, low numbers of viable bacteria may remain but are below the threshold of detection using the methodology in this study and/or cannot be cultured because of a persister state (40).
Central nervous system catheter infection induces robust neutrophil and macrophage recruitment.
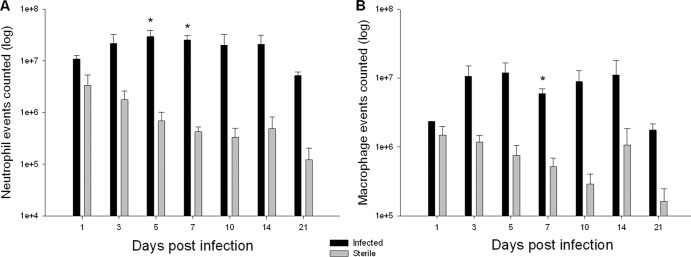
To evaluate whether central nervous system catheter infection induces innate immune cell influx into surrounding tissues, catheter-associated cells were recovered from the brain tissues of mice receiving infected or sterile catheters and analyzed by FACS. Cells were identified based on the following markers: neutrophils (Ly-6G+, CD11b+, CD45high), macrophages (Ly-6G−, CD11b+, CD45high), and microglia (Ly-6G−, CD11b+, CD45low-intermediate). No differences were observed in the numbers of microglia present in tissues from animals implanted with either infected or sterile catheters over the 21-day course examined (data not shown).
Neutrophil influx into tissues surrounding infected catheters was increased at day 3 postinfection and remained elevated through day 14, after which it began to decrease (Fig. 5). In contrast, a small increase in neutrophil influx was observed with sterile catheters on day 1, likely resulting from acute tissue damage associated with catheter insertion. Importantly, total neutrophil counts in the hemisphere containing the infected catheter were significantly higher than sterile catheters throughout the time course evaluated. A similar pattern was observed with regard to macrophage influx, where macrophage infiltrates were significantly higher in tissues surrounding infected catheters than in tissue surrounding sterile catheters (Fig. 5). Again, sterile catheters demonstrated a small increase in macrophage influx on day 1 following infection, with a decrease thereafter. Macrophage infiltrates were increased at day 3 postimplantation and remained elevated through day 14, after which they began to decline (Fig. 5), coinciding with infection resolution in a subset of animals.
Fig 5.
Central nervous system catheter infection induces robust neutrophil and macrophage recruitment. Catheter-associated cells were recovered from infected and sterile mice at multiple time points following catheter implantation using a Percoll gradient method and analyzed by FACS. Results are presented as the absolute number of positive cells for each population, averaged from independent experiments. Results represent three replicates of independent experiments with 4 to 5 mice per group (sterile, infected) in each replicate (n = 12 to 15 total animals per group). *, P < 0.05.
Immunofluorescence staining provides positional information about cellular infiltrates relative to the infection site, which is not possible with flow cytometry. Immunofluorescence staining corroborated the results observed with flow cytometry, with an increase in neutrophil (Ly-6G+) and macrophages/microglia (Iba-1+) in tissues surrounding infected catheters in comparison with sterile catheters. In the first week following infection, neutrophils and macrophages/microglia were diffusely distributed throughout the brain parenchyma. At later dates postinfection (days 14 and 21), neutrophils were primarily localized to areas adjacent to infected catheters. No significant differences were observed in the distribution of the macrophages/microglia in the weeks following infection, but this may be due to the inability to distinguish between macrophages and microglia in tissue sections.
Inflammatory mediator expression is elevated following central nervous system catheter infection.
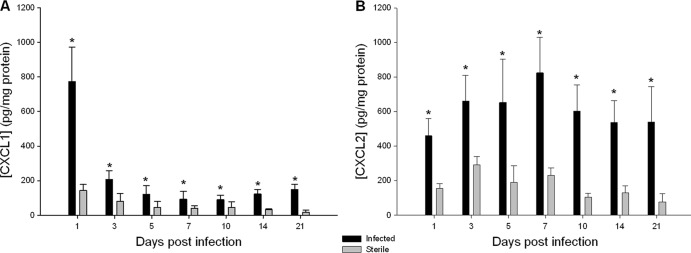
To evaluate whether central nervous system catheter infection actively induced changes in cytokine and chemokine expression throughout the course of infection, homogenates of catheter-associated tissues were analyzed for CXCL1, CXCL2, and IL-17 by ELISA. CXCL1, a chemokine produced by macrophages, neutrophils, and microglia, is an important neutrophil chemoattractant for initiating and organizing the response to infection (18, 37). In this model, CXCL1 expression peaked at day 1 following infection (Fig. 6), likely providing an initial signal to recruit neutrophils to address the developing biofilm infection. Interestingly, CXCL1 levels rapidly diminished and were not significantly elevated during later stages of infection. A small increase in CXCL1 expression was observed in the tissues surrounding the sterile catheters; however, this was a comparatively minor increase compared to infected tissues and likely a response to the tissue trauma resulting from catheter insertion into the ventricle.
Fig 6.
Elevated levels of proinflammatory chemokines CXCL1 (A) and CXCL2 (B). Supernatants from homogenates of catheter-associated tissue were analyzed for CXCL1 and CXCL2 by ELISA. Results were normalized to the amount of total protein recovered to account for differences in tissue sampling size. Results represent three replicates of independent experiments with 4 to 5 mice per group (sterile, infected) in each replicate (n = 12 to 15 total animals per group). *, P < 0.05.
CXCL2 has chemotactic properties similar to those of CXCL1, recruiting neutrophils to sites of infection or injury (5, 20, 35). Interestingly, in contrast to CXCL1, CXCL2 expression progressively increased out to day 7 postinfection and steadily declined thereafter (Fig. 6). It is possible that sustained CXCL2 levels allow for continued recruitment of new neutrophils in an attempt to resolve the infection, although other chemokines or alternative neutrophil chemoattractants such as complement split products and/or bacterium-derived N-formyl-methionyl peptides (f-Met-Leu-Phe) not examined in the current study may also participate in this process.
IL-17 is an important antimicrobial cytokine, known to play an important role in the response to brain abscesses and cutaneous infections caused by S. aureus as well as other models of infection (1, 12, 24, 30, 33). In the setting of a catheter-based infection, IL-17 expression is significantly higher in infected than in sterile tissues; however, the physiological significance of the low levels observed in this setting is unclear (Fig. 7). Tumor necrosis factor alpha (TNF-α) and monocyte chemoattractant protein 1 (MCP-1) were also analyzed; however, there were no significant differences observed in their expressions (data not shown).
Fig 7.
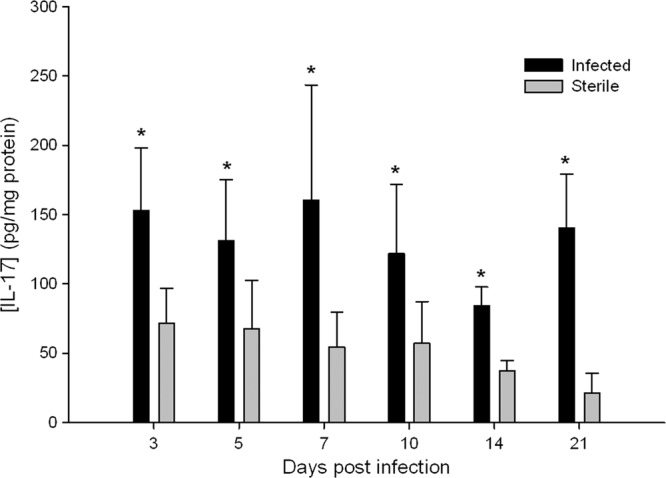
Antimicrobial cytokine IL-17 levels are elevated in infected central nervous system catheters. Supernatants from homogenates of catheter-associated tissue were analyzed for IL-17 by ELISA. Results were normalized to the amount of total protein recovered to account for differences in tissue sampling size. Results represent three replicates of independent experiments with 4 to 5 mice per group (sterile, infected) in each replicate (n = 12 to 15 total animals per group). *, P < 0.05.
DISCUSSION
Cerebrospinal fluid (CSF) shunt infections are a frequent and serious complication in the treatment of hydrocephalus in the pediatric population, with a reported incidence of 5 to 30% (29). In addition to shunt failure, these catheter infections are associated with a higher risk of seizures, decreased intellectual performance, and a 2-fold increase in long-term mortality (14). Therefore, these infections present a challenge that is costly in terms of both patient morbidity and health care expense. As such, studies designed to advance our understanding as to how bacteria colonize these catheters and evade antimicrobial killing in the central nervous system could have a dramatic impact on the development of treatment modalities for these serious infections.
Our animal model results in a reproducible catheter-associated infection in the mouse, similar to the findings seen in humans with ventricular shunt infections. Mice implanted with infected catheters demonstrate more pronounced innate immune cell infiltrates and elevated proinflammatory cytokine and chemokine levels compared to those of mice implanted with sterile catheters. The biofilms observed with scanning electron microscopy analysis are similar to the few images available of similar catheters removed from humans with central nervous system catheter infections (14). In addition, the mild seizure-like activity observed in a subset of infected mice parallels the clinical features of human central nervous system catheter infections. Based on these findings, we propose that this serves as a useful animal model for evaluating CSF shunt infections.
To maximize the clinical relevance of this model, we selected the implantation site, catheter material, and bacterial isolate to reflect human disease as closely as possible. The implantation site utilized in this model enters the lateral ventral, as seen in the placement of ventricular catheters in human patients. The bacterial strain selected for these experiments was a methicillin-susceptible Staphylococcus aureus sample from a child with a ventriculo-peritoneal shunt infection, selected because it is capable of forming a catheter-associated biofilm within the central nervous system, which was verified by our in vitro studies as well as scanning electron microscopy analysis of infected catheters taken from the mouse brain. Several studies have demonstrated significant differences in biofilm formation, toxin production, and regulatory functions when laboratory strains are compared with staphylococcal clinical isolates (3, 6, 8, 10, 17, 28). The use of clinical strains provides valuable information that is much more readily translatable to human disease processes and treatment than is possible with traditional laboratory strains of bacteria.
S. aureus and S. epidermidis are the organisms most commonly associated with central nervous system catheter infections. These organisms are known to form biofilms on catheter surfaces, making diagnosis and treatment of central nervous system catheter infections difficult (14). The ability of biofilms to evade host immune clearance and antimicrobial agents makes it difficult to resolve central nervous system catheter infections nonsurgically, such that catheter removal is currently recommended to effectively treat these infections (39). Studies in peripheral biofilm infection models and in vitro models have been limited to date. In the periphery, most data suggest that immunity is attenuated in response to a biofilm infection since organisms are not efficiently cleared (10, 13, 36, 38). In one recent study, proinflammatory responses were attenuated in a mouse model of subcutaneous catheter-associated biofilm infection (38). Specifically, biofilm infection in the periphery was typified by reductions in several proinflammatory mediators, including IL-1β, CXCL2, and CCL2, which contrasts with the prominent proinflammatory response observed in this central nervous system infection model (i.e., IL-17, CXCL1, and CXCL2). This difference may reflect the impact of bacterial strain variations in the immune response observed during biofilm infections, as a methicillin-resistant S. aureus (MRSA) strain was utilized in the peripheral model versus the methicillin-susceptible S. aureus strain utilized in the current study. It may also reflect the impact of the infection methodology, as the bacteria are given the opportunity to colonize the catheter in vitro in our model, without pressure from the immune system at the time of colonization. This may allow a greater degree of bacterial replication and greater bacterial burden than seen in models in which the device is implanted sterilely and then infected in situ. Another potential reason for this disparity could be the impact of the site of infection on the immune response elicited following biofilm-mediated infection. In particular, the immune milieu in the central nervous system is inherently different than in the periphery, with unique innate immune cell types. Another recent study utilizing a steel pin tibial infection model illustrates the differences that can be observed with biofilm infections depending on the site of infection, as a MRSA infection model noted increases in proinflammatory cytokines IL-17, IL-1β, IL-2, IL-6, and IL-12p70, similar to the increase in inflammatory markers seen in our infection model (32). A model of joint arthroplasty in mice demonstrated an important role for IL-1β in the response to staphylococcal biofilm infections in mice, with increased bacterial burdens and decreased neutrophil recruitment in mice deficient for IL-1β but not in those deficient for TLR2, further demonstrating the complexity inherent in evaluating in vivo systems of biofilm infection.
In this model, a portion of the infected mice had undetectable bacterial burdens at days 14 and 21 postinfection. This may be due to limitations in the sensitivity of culturing viable organisms, as there may be very small numbers of bacteria present at this time point. Alternatively, residual organisms may have entered a persister state as small colony variants, which can be difficult to detect using traditional culture methods (40). In future studies, this possibility will be evaluated with alternate culture methods as well as imaging techniques, including electron microscopy. Mice are also known to be relatively resistant to infection with S. aureus and may therefore be capable of reducing their bacterial burden without antibiotic treatment, unlike human hosts. This has been demonstrated in multiple animal models, in which large numbers of bacteria are required to induce infection in pneumonia, skin and renal abscess models, and intravenous sepsis (7, 11, 19, 26). In addition, both skin and brain abscess models of S. aureus infection in mice have shown that mice are capable of reducing their bacterial burden without definitive antimicrobial therapy (2, 7). Despite these limitations, development of a mouse catheter-associated biofilm infection model represents a powerful new approach to identify important factors in the host immune response to central nervous system biofilms through the broad array of immunologic tools available for use in rodents. The interactions between host immune cells and the biofilm represent a relatively unexplored niche, particularly in the central nervous system, and this mouse model will be a valuable tool for investigating these interactions. Using in vivo studies is important when investigating the complex interactions between the biofilm and the host response in the central nervous system, as these relationships are difficult to accurately replicate in vitro.
There are many possibilities for future studies utilizing this animal model, given the scope of the clinical problem it is designed to explore. Studies under way in our laboratory include a direct comparison of this catheter-based infection model to a parenchymal brain abscess infection, to better delineate the alterations in the immune response that occur based on infection modality alone, with the same location and bacterial strain. In addition, the roles of the adaptive immune system in central nervous system catheter infections have yet to be explored. Studies in peripheral biofilm models suggest that CD4 T cells may play a role in the immune response to biofilm infection in the periphery, but this has yet to be explored in the central nervous system (32). CD4 T cells are known to play a significant role in parenchymal brain abscesses (16); therefore, investigation of their role, if any, in catheter-based infections should also be pursued. Alterations in immune markers, bacterial kinetics, and pathology in response to novel antibiotic therapies have not been previously described but can also be explored using this model.
The development of this murine model of central nervous system catheter infection is an important advancement, as it now allows an investigation of the pathology and host response to these infections. Defining the nature of this immune response could provide valuable tools for screening patients at higher risk for these infections and for providing adjunctive therapy to current antibiotic treatment regimens. New treatment and diagnostic modalities are imperative given the high costs, in health care expense and patient morbidity, associated with CSF shunt infections.
ACKNOWLEDGMENTS
We thank Amy Aldrich, Teresa Fritz, Amanda Angle, and Tyler Scherr for their assistance with FACS, in vitro biofilm assays, and immunohistochemical staining. We thank Nada Harik, at the University of Arkansas for Medical Sciences, Little Rock, AR, for providing the staphylococcal strain used in these experiments. We also thank Charles Kuszynski, Megan Michalak, and Victoria Smith in the UNMC Cell Analysis Facility for assistance with FACS analysis. We thank Geoffrey Talmon and Melissa Holzapfel with the UNMC Electron Microscopy facility for assistance with processing and obtaining EM images.
This work was supported by grants from the National Institute of Neurological Disorders and Stroke to J.N.S. (1K08NS069812-01) and National Institute of Allergy and Infectious Disease (NIAID) P01 AI083211 Project 4 to T.K.
Footnotes
Published ahead of print 2 July 2012
REFERENCES
- 1. Almansa R, et al. 2011. Imbalanced pro- and anti-Th17 responses (IL-17/granulocyte colony-stimulating factor) predict fatal outcome in 2009 pandemic influenza. Crit. Care 15:448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Baldwin AC, Kielian T. 2004. Persistent immune activation associated with a mouse model of Staphylococcus aureus-induced experimental brain abscess. J. Neuroimmunol. 151:24–32 [DOI] [PubMed] [Google Scholar]
- 3. Beenken KE, Blevins JS, Smeltzer MS. 2003. Mutation of sarA in Staphylococcus aureus limits biofilm formation. Infect. Immun. 71:4206–4211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Beenken KE, et al. 2004. Global gene expression in Staphylococcus aureus biofilms. J. Bacteriol. 186:4665–4684 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Bell MD, et al. 1996. Recombinant human adenovirus with rat MIP-2 gene insertion causes prolonged PMN recruitment to the murine brain. Eur. J. Neurosci. 8:1803–1811 [DOI] [PubMed] [Google Scholar]
- 6. Blevins JS, Beenken KE, Elasri MO, Hurlburt BK, Smeltzer MS. 2002. Strain-dependent differences in the regulatory roles of sarA and agr in Staphylococcus aureus. Infect. Immun. 70:470–480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Bunce C, Wheeler L, Reed G, Musser J, Barg N. 1992. Murine model of cutaneous infection with Gram-positive cocci. Infect. Immun. 60:2636–2640 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Cassat J, et al. 2006. Transcriptional profiling of a Staphylococcus aureus clinical isolate and its isogenic agr and sarA mutants reveals global differences in comparison to the laboratory strain RN6390. Microbiology 152:3075–3090 [DOI] [PubMed] [Google Scholar]
- 9. Cassat JE, Lee CY, Smeltzer MS. 2007. Investigation of biofilm formation in clinical isolates of Staphylococcus aureus. Methods Mol. Biol. 391:127–144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Cerca N, Brooks JL, Jefferson KK. 2008. Regulation of the intercellular adhesin locus regulator (icaR) by SarA, sigmaB, and IcaR in Staphylococcus aureus. J. Bacteriol. 190:6530–6533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Cheng AG, et al. 2009. Genetic requirements for Staphylococcus aureus abscess formation and persistence in host tissues. FASEB J. 23:3393–3404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Cho JS, et al. 2010. IL-17 is essential for host defense against cutaneous Staphylococcus aureus infection in mice. J. Clin. Invest. 120:1762–1773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Costerton JW, Stewart PS, Greenberg EP. 1999. Bacterial biofilms: a common cause of persistent infections. Science 284:1318–1322 [DOI] [PubMed] [Google Scholar]
- 14. Fux CA, et al. 2006. Biofilm-related infections of cerebrospinal fluid shunts. Clin. Microbiol. Infect. 12:331–337 [DOI] [PubMed] [Google Scholar]
- 15. Hampl JA, et al. 2003. Rifampin-impregnated silicone catheters: a potential tool for prevention and treatment of CSF shunt infections. Infection 31:109–111 [DOI] [PubMed] [Google Scholar]
- 16. Holley MM, Kielian T. 2012. Th1 and Th17 cells regulate innate immune responses and bacterial clearance during central nervous system infection. J. Immunol. 188:1360–1370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Jefferson KK, Pier DB, Goldmann DA, Pier GB. 2004. The teicoplanin-associated locus regulator (TcaR) and the intercellular adhesin locus regulator (IcaR) are transcriptional inhibitors of the ica locus in Staphylococcus aureus. J. Bacteriol. 186:2449–2456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Johnson EA, et al. 2011. Increased expression of the chemokines CXCL1 and MIP-1alpha by resident brain cells precedes neutrophil infiltration in the brain following prolonged soman-induced status epilepticus in rats. J. Neuroinflammation 8:41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jongerius I, et al. 2012. Staphylococcus aureus virulence is enhanced by secreted factors that block innate immune defenses. J. Innate Immun. 4:301–311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kielian T, Barry B, Hickey WF. 2001. CXC chemokine receptor-2 ligands are required for neutrophil-mediated host defense in experimental brain abscesses. J. Immunol. 166:4634–4643 [DOI] [PubMed] [Google Scholar]
- 21. Kielian T, Bearden ED, Baldwin AC, Esen N. 2004. IL-1 and TNF-alpha play a pivotal role in the host immune response in a mouse model of Staphylococcus aureus-induced experimental brain abscess. J. Neuropathol. Exp. Neurol. 63:381–396 [DOI] [PubMed] [Google Scholar]
- 22. Kielian T, Haney A, Mayes PM, Garg S, Esen N. 2005. Toll-like receptor 2 modulates the proinflammatory milieu in Staphylococcus aureus-induced brain abscess. Infect. Immun. 73:7428–7435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Kielian T, et al. 2007. MyD88-dependent signals are essential for the host immune response in experimental brain abscess. J. Immunol. 178:4528–4537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kimizuka Y, et al. 2012. Roles of IL-17 in an experimental Legionella pneumonia model. Infect. Immun. 80:1121–1127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kockro RA, et al. 2000. Use of scanning electron microscopy to investigate the prophylactic efficacy of rifampin-impregnated CSF shunt catheters. J. Med. Microbiol. 49:441–450 [DOI] [PubMed] [Google Scholar]
- 26. Kraft WG, Johnson PT, David BC, Morgan DR. 1986. Cutaneous infection in normal and immunocompromised mice. Infect. Immun. 52:707–713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Lucas SM, Rothwell NJ, Gibson RM. 2006. The role of inflammation in CNS injury and disease. Br. J. Pharmacol. 147(Suppl 1):S232–S240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. McCallum N, Bischoff M, Maki H, Wada A, Berger-Bachi B. 2004. TcaR, a putative MarR-like regulator of sarS expression. J. Bacteriol. 186:2966–2972 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. McGirt MJ, et al. 2003. Risk factors for pediatric ventriculoperitoneal shunt infection and predictors of infectious pathogens. Clin. Infect. Dis. 36:858–862 [DOI] [PubMed] [Google Scholar]
- 30. Nichols JR, et al. 2009. TLR2 deficiency leads to increased Th17 infiltrates in experimental brain abscesses. J. Immunol. 182:7119–7130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Paxinos G, Franklin KBJ. 2007. The mouse brain in stereotaxic coordinates, 3rd ed Academic Press, London, United Kingdom [Google Scholar]
- 32. Prabhakara R, Harro JM, Leid JG, Harris M, Shirtliff ME. 2011. Murine immune response to a chronic Staphylococcus aureus biofilm infection. Infect. Immun. 79:1789–1796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Puel A, et al. 2011. Chronic mucocutaneous candidiasis in humans with inborn errors of interleukin-17 immunity. Science 332:65–68 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Reichel CA, et al. 2006. Chemokine receptors Ccr1, Ccr2, and Ccr5 mediate neutrophil migration to postischemic tissue. J. Leukoc. Biol. 79:114–122 [DOI] [PubMed] [Google Scholar]
- 35. Shiratori M, Tozaki-Saitoh H, Yoshitake M, Tsuda M, Inoue K. 2010. P2X7 receptor activation induces CXCL2 production in microglia through NFAT and PKC/MAPK pathways. J. Neurochem. 114:810–819 [DOI] [PubMed] [Google Scholar]
- 36. Singh PK, Parsek MR, Greenberg EP, Welsh MJ. 2002. A component of innate immunity prevents bacterial biofilm development. Nature 417:552–555 [DOI] [PubMed] [Google Scholar]
- 37. Skuljec J, et al. 2011. CCL5 induces a pro-inflammatory profile in microglia in vitro. Cell Immunol. 270:164–171 [DOI] [PubMed] [Google Scholar]
- 38. Thurlow LR, et al. 2011. Staphylococcus aureus biofilms prevent macrophage phagocytosis and attenuate inflammation in vivo. J. Immunol. 186:6585–6596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Tunkel AR, et al. 2004. Practice guidelines for the management of bacterial meningitis. Clin. Infect. Dis. 39:1267–1284 [DOI] [PubMed] [Google Scholar]
- 40. von Eiff C. 2008. Staphylococcus aureus small colony variants: a challenge to microbiologists and clinicians. Int. J. Antimicrob. Agents 31:507–510 [DOI] [PubMed] [Google Scholar]