Abstract
OBJECTIVE
The fetus is thought to play a central role in the onset of labor. Pulmonary surfactant protein (SP)-A, secreted by the maturing fetal lung, has been implicated in the mechanisms initiating parturition in mice. The present study was conducted to determine whether amniotic fluid concentrations of SP-A and SP-B change during human parturition.
STUDY DESIGN
Amniotic fluid SP-A and SP-B concentrations were measured with sensitive and specific ELISA in the following groups of pregnant women: 1) mid-trimester of pregnancy between 15th and 18th weeks of gestation (n=29); 2) term pregnancy not in labor (n=28); and 3) term pregnancy in spontaneous labor (n=26). Non-parametric statistics were used for analysis.
RESULTS
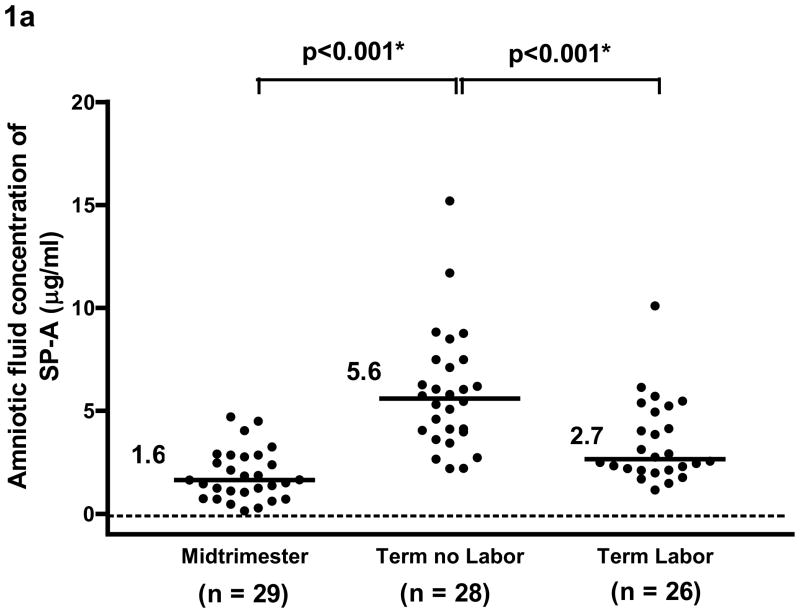
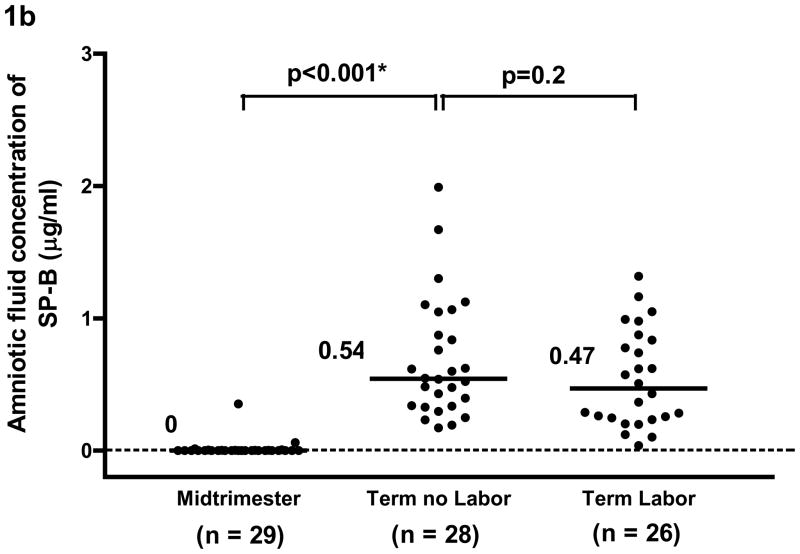
SP-A was detected in all amniotic fluid samples. SP-B was detected in 24.1% (7/29) of mid-trimester samples and in all samples at term. The median amniotic fluid concentration of SP-A and SP-B were significantly higher in women at term than in women in the mid-trimester (SP-A term no labor: median 5.6 μg/ml, range 2.2–15.2 μg/ml vs. mid-trimester: median 1.64 μg/ml, range 0.1–4.7 μg/ml; and SP-B term no labor: median 0.54 μg/ml, range 0.17–1.99 μg/ml vs. mid-trimester: median 0 μg/ml, range 0–0.35 μg/ml; both p<0.001). The median amniotic fluid SP-A concentration in women at term in labor was significantly lower than that in women at term not in labor (term in labor: median 2.7 μg/ml, range 1.2–10.1 μg/ml vs. term no labor: median 5.6 μg/ml, range 2.2–15.2 μg/ml; p<0.001). There was no significant difference in the median amniotic fluid SP-B concentrations between women in labor and those not in labor (term in labor: median 0.47 μg/ml range 0.04–1.32 μg/ml vs. term no labor: median 0.54 μg/ml range 0.17–1.99 μg/ml; p=0.2).
CONCLUSION
The amniotic fluid concentration of surfactant protein-A decreases in spontaneous human parturition at term.
Keywords: Surfactant protein, SP-A, SP-B, amniotic fluid, term, labor, parturition
INTRODUCTION
Pulmonary surfactant, a complex molecule matrix containing lipids (90%) and specific proteins (10%), reduces surface tension at the air-liquid interface of the lung [1–3]. Lipids in surfactant are critical for pulmonary function and attention has shifted to the role of the surfactant proteins (SPs). Currently, four types of SP have been characterized: SP-A, B, C and D. SP-A and SP-D are relatively hydrophilic, C-type collagenous lectins, involved in host defense and the regulation of surfactant structure or homeostasis [4–9]. In contrast, SP-B and C are relatively hydrophobic and play important roles in surfactant homeostasis and function after birth [10–12]. SP-A is the most abundant of SP in human lung.
Recently, SP-A was implicated in a mechanism initiating parturition in mice [13]. The experimental basis for this is that: 1) the amniotic fluid concentration of SP-A increased with advancing gestational age in mice; 2) the administration of SP-A into the amniotic cavity on E15 of gestation caused preterm delivery; and 3) intra-amniotic injection of a neutralizing antibody to SP-A was associated with prolonged gestation [13]. The proposed hypothesis is that SP-A produced by the maturing fetal lung is secreted into the amniotic cavity where it induces local macrophage activation and infiltration of these cells into the uterine wall, leading to a pro-inflammatory response, suspension of progesterone action and parturition [13].
It is unclear whether SP-A plays a role in the initiation of parturition in humans. The present study was conducted to determine whether human spontaneous parturition at term is associated with changes in amniotic fluid concentrations of SP-A and SP-B.
MATERIALS AND METHODS
Study design
A cross-sectional study was conducted to measure SP-A, SP-B and total protein concentration in amniotic fluid samples obtained from women during the second trimester (15–18 weeks) of pregnancy (n=29), at term (≥37 weeks of gestation) not in labor (n=28), and at term gestation in spontaneous labor (n=26). Amniotic fluid was obtained by trans-abdominal amniocentesis. The fluid was centrifuged and the supernatant stored. Patients with meconium stained amniotic fluid samples were excluded. Second trimester amniotic fluid samples were obtained from patients who had consented to have an amniocentesis for prenatal diagnosis of genetic disorders. All patients from this group delivered a normal neonate at term. Women at term, but not in labor, underwent amniocentesis for the assessment of fetal lung maturity prior to cesarean section, whereas those in labor underwent amniocentesis because of either uncertain gestational age or for the diagnosis of intra-amniotic infection. All women at term had no evidence of medical or obstetrical complications, had negative amniotic fluid cultures for microorganisms and delivered a normal infant with neonatal birthweight of more than 2500 grams. Amniotic fluid not required for clinical assessment was centrifuged for 10 minutes at 4°C and the supernatant was aliquoted and stored at −70°C until analysis.
All women provided informed consent prior to the collection of amniotic fluid. The collection and utilization of the samples was approved by the Human Investigation Committee of the Sotero del Rio Hospital, Santiago, Chile (a major affiliate of the Catholic University of Santiago), the Pennsylvania Hospital and the IRB of the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Many of these samples were used in previous studies of cytokines, matrix metalloproteinases and chemokines in human parturition [14–17].
ELISA for determining the SP-A and SP-B concentrations in amniotic fluid
SP-A and SP-B concentrations in amniotic fluid were determined by a sandwich ELISA system which had been validated for amniotic fluid as previously described in detail [18,19]. The inter-assay coefficient of variation (CV) for SP-A and SP-B was 33.5% and 34.2% and the intra-assay CV was 7.7% and 18.7%, respectively. The sensitivity of SP-A and SP-B was 10 ng/ml and 2 ng/ml, respectively.
Statistical analysis
Mann-Whitney U test was utilized to determine differences in the median between groups. Contingency tables and Chi-square tests were employed for comparisons of proportions. The statistical package used was SPSS 12.0 (SPSS Inc, Chicago, Illinois).
RESULTS
SP-A was detected in all amniotic fluid samples. SP-B was detected in 24.1% (7/29) of mid-trimester samples (as early as 15th week of gestation) and in all samples at term. Table I summarizes demographic and clinical characteristics of the three study groups. As expected, the median maternal age of women in the mid-trimester group was significantly higher than women at term not in labor. Among women at term, there was no significant difference in median maternal age, gestational age at amniocentesis and neonatal birthweight.
Table I.
Demographic and clinical characteristics
| Characteristics | Mid-trimester (n=29) | p* | Term no labor (n=28) | p** | Term in labor (n=26) |
|---|---|---|---|---|---|
| Maternal age, years | 36 (26–40) | <0.001* | 24 (14–42) | 0.2 | 22 (15–36) |
| Gestational age at amniocentesis, weeks | 16 (15–18) | <0.001* | 39.3 (38–42) | 0.3 | 40 (38–42) |
| Gestational age at delivery, weeks | 39 (37–42) | 0.6 | 39.3 (38–42) | 0.3 | 40 (38–42) |
| Birthweight, grams | 3460 (2866–4043) | 0.3 | 3330 (2810–4470) | 0.2 | 3325 (2540–3760) |
| Male gender, n (%) | 17 (58.6%) | 0.1 | 11 (39.3%) | 0.03* | 18 (69.2%) |
Values are presented as median (range) or numbers (percent); *p<0.05
Comparison between mid-trimester and Term no labor.
Comparison between Term no labor and Term in labor
The median amniotic fluid concentration of SP-A and of SP-B was significantly higher in women at term not in labor than those in the mid-trimester [SP-A; term no labor: median 5.6 μg/ml, range 2.2–15.2 μg/ml vs. mid-trimester: median 1.64 μg/ml, range 0.1–4.7 μg/ml, p<0.001 (see Figure 1a) and SP-B, term no labor: median 0.54 μg/ml, range 0.17–1.99 μg/ml vs. mid-trimester: median 0 μg/ml, range 0–0.35 μg/ml, p<0.001, (see Figure 1b)].
Figure 1.
Figure 1a. and 1b. The median concentration of SP-A and of SP-B in amniotic fluid was significantly higher at term than in the mid-trimester of pregnancy (SP-A term no labor: median 5.6 μg/ml, range 2.2–15.2 μg/ml vs. mid-trimester: median 1.64 μg/ml, range 0.1–4.7 μg/ml; p<0.001 and SP-B term no labor: median 0.54 μg/ml, range 0.17–1.99 μg/ml vs. mid-trimester: median, 0 μg/ml, range 0–0.35 μg/ml; p<0.001). The median amniotic fluid concentration of SPA in women at term in spontaneous labor was significantly lower than in those not in labor (term in labor: median 2.7 μg/ml, range 1.2–10.1 μg/ml vs. term no labor: median 5.6 μg/ml, range 2.2–15.2 μg/ml; p<0.001). There was no significant difference in the median amniotic fluid concentration of SP-B between women with and without labor (term in labor: median 0.47 μg/ml, range 0.04–1.32 μg/ml vs. term no labor: median 0.54 μg/ml, range 0.17–1.99μg/ml; p=0.2). *p<0.05.
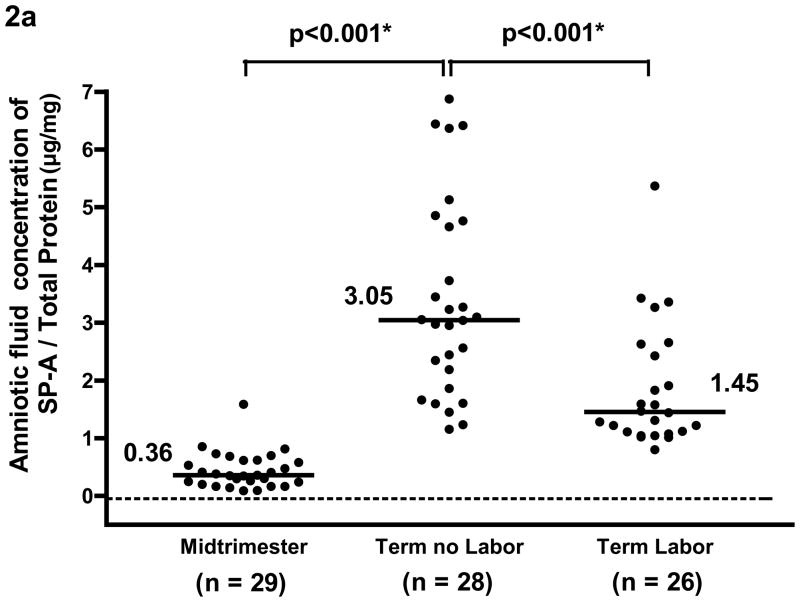
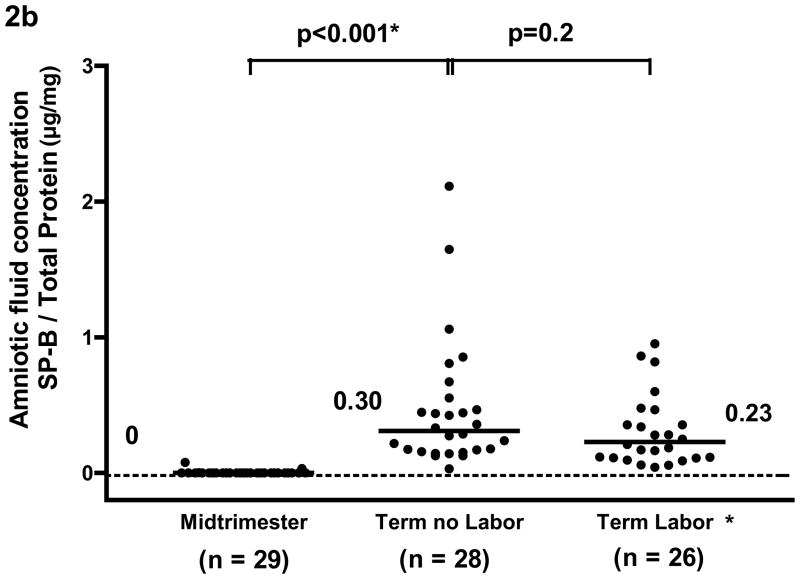
The median amniotic fluid concentration of SP-A in women at term in spontaneous labor was approximately 50% of that found in women at term not in labor (term in labor: median 2.7 μg/ml, range 1.2–10.1 μg/ml vs. term no labor: median 5.6 μg/ml, range 2.2–15.2 μg/ml, p<0.001; see Figure 1a). There was no significant difference in the median amniotic fluid SP-B concentration between the two groups of women at term (term in labor: median 0.47 μg/ml, range 0.04–1.32 μg/ml vs. term no labor: median 0.54 μg/ml, range 0.17–1.99μg/ml, p=0.2; see Figure 1b). The results remained similar after adjusting for total amniotic fluid protein content (p=0.001 and p=0.2, respectively; see Figures 2a and 2b).
Figure 2.
Figure 2a and 2b. After adjusting for total protein concentration, the median amniotic fluid concentration of SP-A and of SP-B was significantly higher at term than during the mid-trimester of pregnancy (SP-A/total protein; term no labor: median 3.05 μg/mg, range 1.15–6.87 μg/mg vs. mid-trimester: median 0.36 μg/mg, range 0.09–1.59 μg/mg; p<0.001 and SP-B/total protein, term no labor: median 0.30 μg/mg, range 0.03–2.11 μg/mg vs. mid-trimester: median 0 μg/mg, range 0–0.08 μg/mg; p<0.001). Amniotic fluid SP-A concentration after adjusting for total protein concentration in women at term in labor was lower than those not in labor (term labor: median 1.45 μg/mg, range 0.8–25.18 μg/mg vs. term not in labor: median 3.05 μg/mg, range 1.15–6.87 μg/mg; p<0.001). There was no significant difference in the median amniotic fluid concentration of SP-B/total protein in women with and without labor (term labor: median 0.23 μg/mg, range 0.04–5.58 μg/mg vs. term not in labor: median 0.30 μg/mg, range 0.03–2.11 μg/mg; p=0.2) *One data point in the term labor group was an outlier (SP-B/total protein: 5.58 μg/mg) and is not shown in the figure. *p<0.05.
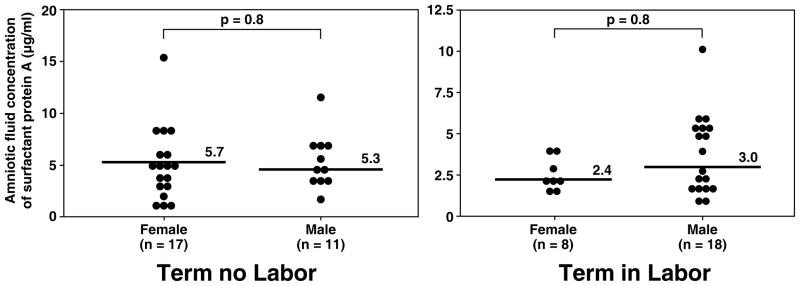
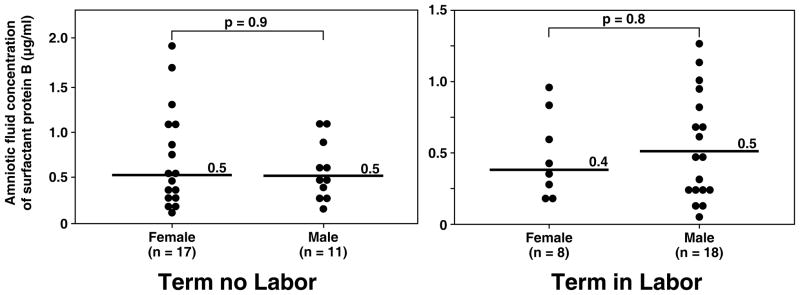
Although more male fetuses were included in the group of women at term in labor than in those not in labor [(in labor: 69.2% (18/26) vs. not in labor: 39.3% (11/28); p=0.03); Table I], there was no significant difference in the median amniotic fluid concentration of SP-A and SP-B stratified by fetal gender in both the term in labor and term not in labor groups (all p>0.05; see Figure 3 and 4).
Figure 3.
Amniotic fluid concentrations of SP-A in women at term with and without labor stratified by gender of the fetus. There was no significant difference in the median amniotic fluid concentration of SP-A stratified by fetal gender in both the term in labor and term not in labor groups (term no labor SP-A female: median 5.7 μg/ml, range 2.2–15.2 μg/ml vs. male: median 5.3 μg/ml, range 2.6–11.7 μg/ml; p=0.8 and term in labor SP-A female: median 2.4 μg/ml, range 1.7–4.1 μg/ml vs. male: median 3.0 μg/ml, range 1.2–10.1 μg/ml; p=0.2).
Figure 4.
Amniotic fluid concentrations of SP-B in women at term with and without labor stratified by gender of the fetus. There was no significant difference in the median amniotic fluid concentration of SP-B stratified by fetal gender in both the term in labor and term not in labor groups (term no labor SP-B, female: median 0.5 μg/ml, range 0.2–1.9 μg/ml vs. male: median 0.5 μg/ml, range 0.2–1.1 μg/ml; p=0.9 and term in labor SP-B, female: median 0.4 μg/ml, range 0.2–0.9 μg/ml vs. male: median 0.5 μg/ml, range 0.04–1.3 μg/ml; p=0.8).
DISCUSSION
Principal findings of this study
1) The median amniotic fluid SP-A concentration, but not SP-B, is lower in women with spontaneous labor at term than in women not in labor; 2) SP-A and SP-B amniotic fluid concentrations are higher in term gestation than in the mid-trimester of pregnancy.
Physiology of SP-A and SP-B
SP-A is a large, oligomeric protein from the collectin family of C-type lectins, encoded by two genes (SP-A1 and SP-A2) located on chromosome 10. SP-A plays important roles in host defense against infection and is required for the formation of tubular myelin, the major form of alveolar surfactant. SP-A binds bacteria, viruses, allergens, apoptotic cells and functions as an opsonin enhancing uptake by alveolar macrophages, monocytes, neutrophils and dendritic cells [4,7,11,12]. SP-A may also induce or inhibit the production of cytokines and inflammatory mediators [eg: tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β], reactive oxygen species and nitric oxide metabolites depending on the types of pathogens or stimuli, activation state of cells, and cellular receptors. The CD91-calreticulin complex, signal-inhibitory regulatory protein-α (SIRP-α) or Toll-like receptors (TLRs) 2 and 4 have been proposed as mediators of cellular responses to SP-A [7,12,20]. In the context of the host response of the lung, the immunomodulatory properties of SP-A might benefit the host [7,12]. For example, in the resting state, clearance of apoptotic cells or pathogens without invoking a robust inflammatory response may serve as a protective role in the lung. However, in the context of infection, the recruitment of immune cells or inflammatory mediators might be warranted [7,12]. While SP-A deficient mice are susceptible to infection and lack tubular myelin, surfactant function does not significantly alter by the lack of SP-A [21,22]. SP-A has been detected in amniotic fluid, maternal and umbilical cord blood [23,24].
SP-B is a hydrophobic protein encoded by a single gene located on chromosome 2.[25] SP-B plays a critical role in surfactant homeostasis by promoting adsorption of lipid surfactant into the expanding surface film and maintaining its stability during respiratory cycles to lower the surface tension within the alveolar spaces [1,26]. Infants with inherited deficiency of SP-B develop severe respiratory distress at birth [27]. SP-B has been detected in amniotic fluid and in adult serum [28].
Alveolar type II cells synthesize all four surfactant proteins and surfactant lipids. Surfactant proteins are also synthesized by airway cells including clara cells and submucosal cells. SP-A, but not SP-B, has been localized to non-pulmonary sites [12] including brain, trachea, testis, salivary glands, lacrimal glands, heart, prostate, kidney, pancreas, vagina [29] and human chorio-amniotic membranes [30], supporting the role of SP-A in host defense rather than reducing surface tension.
SP-A and SP-B in mid-trimester amniotic fluid
The finding that amniotic fluid concentration of SP-A and B in women at term is higher than in the mid-trimester of pregnancy is consistent with that of previous studies [18,30–33]. Pryhuber and colleagues reported that amniotic fluid concentration of SP-A, SP-B and the lecithin-sphingomyelin (L/S) ratio increased with advancing gestational age [18]. The mean amniotic fluid concentration of SP-A increased gradually after 19–23 weeks of gestation, followed by a quadratic increase between 30–40 weeks of gestation. In the current study, SP-A was detectable in the amniotic fluid at 15th–18th week of gestation. SPA expression has been detected by in situ hybridization in the human fetal respiratory tract by 16 weeks [18,34]. At that time, SP-A protein and mRNA expression were observed only in the tracheal epithelium. However, expression by bronchiolar cells and pre-type II lining terminal airways was not observed until the 19th–20th week of gestation [18,34]. It is possible that amniotic fluid SP-A in the early mid-trimester derives from extra-pulmonary origin, most likely chorioamniotic membranes. The presence of SP-A in amniotic fluid cavity at 17 weeks of patient with fetal tracheal atresia (without tracheo-esophageal fistula) supported this view [35].
The observation that amniotic fluid SP-B was detectable in 24% of the mid-trimester samples and as early as the 15th week of gestation was unexpected. However, all of the samples except one had low concentrations of SP-B. Contrary to the present findings, SP-B has been previously reported to be first detectable in amniotic fluid at 31 weeks of gestation [18]. SP-B mRNA and pro-protein have been detected in lung tissue as early as the 13th–15th weeks of gestation [36–38]. The mature SP-B protein has been detected after 19 weeks [37,38] and not consistently detectable until the 24th week when the epithelial cell lining begins to differentiate into type II cells containing lamellar bodies.
Amniotic fluid concentration of SP-A and SP-B in women at term with and without labor
Animals studies suggests that SP-A is incorporated into myelinic and vesicular forms of extracellular surfactant [39] and that labor can stimulate the release of surfactant into the pulmonary airways [40,41]. In humans, the concentration of SP-A in umbilical cord blood is higher in newborns delivered after spontaneous labor than in those delivered by elective cesarean section [24]. Cho et al suggested that this could be due to an increased transfer of pulmonary SP-A to the fetal circulation during labor [24]. Our finding that the median amniotic fluid SP-A concentration was lower in women at term in labor than in those not in labor was unexpected and not consistent with the view that SP-A secreted by the lung may play a role in the onset of parturition. Condon et al proposed that SP-A could initiate parturition by directly stimulating amniotic fluid fetal macrophages to migrate into the uterine wall where they would produce pro-inflammatory cytokines and activate nuclear factor kappa B leading to suspension of the progesterone action and uterine quiescence [13,42]. However, there is no information available about the amniotic fluid concentrations of SP-A in mice who were in labor compared to those without labor. A recent study in placental bed biopsy and postpartum hysterectomy specimens from pregnant patients with male fetuses reported that macrophages in human myometrium after labor are all maternal origin [43]. This study suggested that the trafficking of fetal macrophages in labor seemed to be different between human and mice. Although in vitro experiments provided evidence that surfactant lipids and SP-A could stimulate prostaglandin production in human amnion [44,45], there was no significant difference in SP-A mRNA expression in the chorioamniotic membranes of women at term not in labor and those in labor [35]. Finally, the observation that mice with a genetic deletion [46] or an over-expression [47] of the SP-A gene reproduce normally contradicts the role of SP-A as an essential factor of labor.
The current study does not exclude a role for SP-A in the mechanism of parturition. Human labor is associated with an increased production of prostaglandin and pro-inflammatory cytokines by intrauterine tissue, amniotic fluid and invading macrophages [48–51]. The decreased amniotic fluid concentration of SP-A may be due to the uptake by macrophages and other cells including amnion. However, there is no experimental evidence at this point that this is the case. Alternatively, SP-A could potentially suppress an inflammatory response in intra-uterine tissues during normal gestation [7]. A decrease in amniotic fluid SP-A could be associated with an acute intrauterine inflammatory response and parturition [7]. A recent study indicated that human fetal membranes from women in spontaneous labor, but without histologic chorioamnionitis, had a gene expression pattern consistent with inflammation [52].
Another possible explanation for the observed lower amniotic fluid concentration of SP-A in women at term in labor than in those without labor is that SP-A may be incorporated into tubular myelin [39] in preparation for neonatal exposure to the noxious elements present in room air after birth. Incorporation of SP-A into the complex surfactant molecule matrix present in the lung leads to a decrease in amniotic fluid SP-A concentration. In an animal experiment, a time-dependent movement of exogenous SP-A from extracellular components into pneumocyte type II cells was observed after intra-tracheal instillation of SP-A [39] suggesting that alveolar clearance and recycling of SP-A into Type II cells or tubular myelin could occur in humans. Finally, since there is an increased concentration of neutrophil elastase in the amniotic fluid of women at term in labor [53] and the incubation of bronchoalveolar lavage fluid from patients with cystic fibrosis (that contained high concentration of protease) with SP-A caused a time dependent degradation of SP-A [54]. It is also possible that the decreased concentrations of SPA in the amniotic fluid are due to the action of proteases during term labor [53,54].
Strength and limitation of the study
This is the first study examining the question whether the concentrations of SP-A and SP-B in amniotic fluid change in human parturition. Since this is a retrospective cross sectional study, the chronological events or causal relationships would be difficult to specify. Moreover, the ELISA assay system validated for amniotic fluid [18,19] had slightly high inter- and intra-assay coefficients of variation. So far, there was no commercially available assay for surfactant proteins (except SP-D).
Conclusion
In summary, our study demonstrates that the amniotic fluid concentrations of SPA, but not of SP-B, are lower in normal pregnancies at term in spontaneous labor than in those not in labor.
Acknowledgments
This research was supported in part by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, NIH, DHHS.
Reference List
- 1.Whitsett JA, Weaver TE. Hydrophobic surfactant proteins in lung function and disease. N Engl J Med. 2002;347:2141–2148. doi: 10.1056/NEJMra022387. [DOI] [PubMed] [Google Scholar]
- 2.Weaver TE, Whitsett JA. Function and regulation of expression of pulmonary surfactant-associated proteins. Biochem J. 1991;273(Pt 2):249–264. doi: 10.1042/bj2730249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jobe AH, Ikegami M. Surfactant and acute lung injury. Proc Assoc Am Physicians. 1998;110:489–495. [PubMed] [Google Scholar]
- 4.McCormack FX, Whitsett JA. The pulmonary collectins, SP-A and SP-D, orchestrate innate immunity in the lung. J Clin Invest. 2002;109:707–712. doi: 10.1172/JCI15293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cockshutt AM, Weitz J, Possmayer F. Pulmonary surfactant-associated protein A enhances the surface activity of lipid extract surfactant and reverses inhibition by blood proteins in vitro. Biochemistry. 1990;29:8424–8429. doi: 10.1021/bi00488a032. [DOI] [PubMed] [Google Scholar]
- 6.Dobbs LG, Wright JR, Hawgood S, Gonzalez R, Venstrom K, Nellenbogen J. Pulmonary surfactant and its components inhibit secretion of phosphatidylcholine from cultured rat alveolar type II cells. Proc Natl Acad Sci USA. 1987;84:1010–1014. doi: 10.1073/pnas.84.4.1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kishore U, Greenhough TJ, Waters P, Shrive AK, Ghai R, Kamran MF, Bernal AL, Reid KB, Madan T, Chakraborty T. Surfactant proteins SP-A and SP-D: structure, function and receptors. Mol Immunol. 2006;43:1293–1315. doi: 10.1016/j.molimm.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 8.Rice WR, Ross GF, Singleton FM, Dingle S, Whitsett JA. Surfactant-associated protein inhibits phospholipid secretion from type II cells. J Appl Physiol. 1987;63:692–698. doi: 10.1152/jappl.1987.63.2.692. [DOI] [PubMed] [Google Scholar]
- 9.Suzuki Y, Fujita Y, Kogishi K. Reconstitution of tubular myelin from synthetic lipids and proteins associated with pig pulmonary surfactant. Am Rev Respir Dis. 1989;140:75–81. doi: 10.1164/ajrccm/140.1.75. [DOI] [PubMed] [Google Scholar]
- 10.Whitsett JA, Nogee LM, Weaver TE, Horowitz AD. Human surfactant protein B: structure, function, regulation, and genetic disease. Physiol Rev. 1995;75:749–757. doi: 10.1152/physrev.1995.75.4.749. [DOI] [PubMed] [Google Scholar]
- 11.Whitsett JA. Surfactant proteins in innate host defense of the lung. Biol Neonate. 2005;88:175–180. doi: 10.1159/000087580. [DOI] [PubMed] [Google Scholar]
- 12.Wright JR. Immunoregulatory functions of surfactant proteins. Nat Rev Immunol. 2005;5:58–68. doi: 10.1038/nri1528. [DOI] [PubMed] [Google Scholar]
- 13.Condon JC, Jeyasuria P, Faust JM, Mendelson CR. Surfactant protein secreted by the maturing mouse fetal lung acts as a hormone that signals the initiation of parturition. Proc Natl Acad Sci USA. 2004;101:4978–4983. doi: 10.1073/pnas.0401124101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Romero R, Gomez R, Galasso M, Munoz H, Acosta L, Yoon BH, Svinarich D, Cotton DB. Macrophage inflammatory protein-1 alpha in term and preterm parturition: effect of microbial invasion of the amniotic cavity. Am J Reprod Immunol. 1994;32:108–113. doi: 10.1111/j.1600-0897.1994.tb01101.x. [DOI] [PubMed] [Google Scholar]
- 15.Espinoza J, Chaiworapongsa T, Romero R, Gomez R, Kim JC, Yoshimatsu J, Edwin S, Rathnasabapathy C, Yoon BH. Evidence of participation of soluble CD14 in the host response to microbial invasion of the amniotic cavity and intra-amniotic inflammation in term and preterm gestations. J Matern Fetal Neonatal Med. 2002;12:304–312. doi: 10.1080/jmf.12.5.304.312. [DOI] [PubMed] [Google Scholar]
- 16.Park KH, Chaiworapongsa T, Kim YM, Espinoza J, Yoshimatsu J, Edwin S, Gomez R, Yoon BH, Romero R. Matrix metalloproteinase 3 in parturition, premature rupture of the membranes, and microbial invasion of the amniotic cavity. J Perinat Med. 2003;31:12–22. doi: 10.1515/JPM.2003.002. [DOI] [PubMed] [Google Scholar]
- 17.Esplin MS, Romero R, Chaiworapongsa T, Kim YM, Edwin S, Gomez R, Gonzalez R, Adashi EY. Amniotic fluid levels of immunoreactive monocyte chemotactic protein-1 increase during term parturition. J Matern Fetal Neonatal Med. 2003;14:51–56. doi: 10.1080/jmf.14.1.51.56. [DOI] [PubMed] [Google Scholar]
- 18.Pryhuber GS, Hull WM, Fink I, McMahan MJ, Whitsett JA. Ontogeny of surfactant proteins A and B in human amniotic fluid as indices of fetal lung maturity. Pediatr Res. 1991;30:597–605. doi: 10.1203/00006450-199112000-00023. [DOI] [PubMed] [Google Scholar]
- 19.McMahan MJ, Mimouni F, Miodovnik M, Hull WM, Whitsett JA. Surfactant associated protein (SAP-35) in amniotic fluid from diabetic and nondiabetic pregnancies. Obstet Gynecol. 1987;70:94–98. [PubMed] [Google Scholar]
- 20.Guillot L, Balloy V, McCormack FX, Golenbock DT, Chignard M, Si-Tahar M. Cutting edge: the immunostimulatory activity of the lung surfactant protein-A involves Toll-like receptor 4. J Immunol. 2002;168:5989–5992. doi: 10.4049/jimmunol.168.12.5989. [DOI] [PubMed] [Google Scholar]
- 21.LeVine AM, Kurak KE, Wright JR, Watford WT, Bruno MD, Ross GF, Whitsett JA, Korfhagen TR. Surfactant protein-A binds group B streptococcus enhancing phagocytosis and clearance from lungs of surfactant protein-A-deficient mice. Am J Respir Cell Mol Biol. 1999;20:279–286. doi: 10.1165/ajrcmb.20.2.3303. [DOI] [PubMed] [Google Scholar]
- 22.LeVine AM, Bruno MD, Huelsman KM, Ross GF, Whitsett JA, Korfhagen TR. Surfactant protein A-deficient mice are susceptible to group B streptococcal infection. J Immunol. 1997;158:4336–4340. [PubMed] [Google Scholar]
- 23.Doyle IR, Hermans C, Bernard A, Nicholas TE, Bersten AD. Clearance of Clara cell secretory protein 16 (CC16) and surfactant proteins A and B from blood in acute respiratory failure. Am J Respir Crit Care Med. 1998;158:1528–1535. doi: 10.1164/ajrccm.158.5.9712097. [DOI] [PubMed] [Google Scholar]
- 24.Cho K, Matsuda T, Okajima S, Matsumoto Y, Sagawa T, Fujimoto S, Kobayashi K. Factors influencing pulmonary surfactant protein A levels in cord blood, maternal blood and amniotic fluid. Biol Neonate. 1999;75:104–110. doi: 10.1159/000014085. [DOI] [PubMed] [Google Scholar]
- 25.Nogee LM. Genetics of the hydrophobic surfactant proteins. Biochim Biophys Acta. 1998;1408:323–333. doi: 10.1016/s0925-4439(98)00078-7. [DOI] [PubMed] [Google Scholar]
- 26.Hawgood S. Surfactant protein B: structure and function. Biol Neonate. 2004;85:285–289. doi: 10.1159/000078169. [DOI] [PubMed] [Google Scholar]
- 27.Nogee LM. Alterations in SP-B and SP-C expression in neonatal lung disease. Annu Rev Physiol. 2004;66:601–623. doi: 10.1146/annurev.physiol.66.032102.134711. [DOI] [PubMed] [Google Scholar]
- 28.Hermans C, Dong P, Robin M, Jadoul M, Bernard A, Bersten AD, Doyle IR. Determinants of serum levels of surfactant proteins A and B and Clara cell protein CC16. Biomarkers. 2003;8:461–471. doi: 10.1080/13547500310001647021. [DOI] [PubMed] [Google Scholar]
- 29.MacNeill C, Umstead TM, Phelps DS, Lin Z, Floros J, Shearer DA, Weisz J. Surfactant protein A, an innate immune factor, is expressed in the vaginal mucosa and is present in vaginal lavage fluid. Immunology. 2004;111:91–99. doi: 10.1111/j.1365-2567.2003.01782.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Miyamura K, Malhotra R, Hoppe HJ, Reid KB, Phizackerley PJ, Macpherson P, Lopez BA. Surfactant proteins A (SP-A) and D (SP-D): levels in human amniotic fluid and localization in the fetal membranes. Biochim Biophys Acta. 1994;1210:303–307. doi: 10.1016/0005-2760(94)90233-x. [DOI] [PubMed] [Google Scholar]
- 31.King RJ, Ruch J, Gikas EG, Platzker AC, Creasy RK. Appearance of paoproteins of pulmonary surfactant in human amniotic fluid. J Appl Physiol. 1975;39:735–741. doi: 10.1152/jappl.1975.39.5.735. [DOI] [PubMed] [Google Scholar]
- 32.Shelley SA, Balis JU, Paciga JE, Knuppel RA, Ruffolo EH, Bouis PJ., Jr Surfactant “apoproteins” in human amniotic fluid: an enzyme-linked immunosorbent assay for the prenatal assessment of lung maturity. Am J Obstet Gynecol. 1982;144:224–228. doi: 10.1016/0002-9378(82)90632-9. [DOI] [PubMed] [Google Scholar]
- 33.Dilger I, Schwedler G, Dudenhausen JW. Determination of the pulmonary surfactant-associated protein SP-B in amniotic fluid with a competition ELISA. Gynecol Obstet Invest. 1994;38:24–27. doi: 10.1159/000292439. [DOI] [PubMed] [Google Scholar]
- 34.Khoor A, Gray ME, Hull WM, Whitsett JA, Stahlman MT. Developmental expression of SPA and SP-A mRNA in the proximal and distal respiratory epithelium in the human fetus and newborn. J Histochem Cytochem. 1993;41:1311–1319. doi: 10.1177/41.9.8354874. [DOI] [PubMed] [Google Scholar]
- 35.Han YM, Romero R, Kim YM, Kim JS, Richani K, Friel LA, Kusanovic JP, Jeanty C, Vitale S, Nien JK, et al. Surfactant protein-A mRNA expression by human fetal membranes is increased in histological chorioamnionitis but not in spontaneous labour at term. J Pathol. 2007;211:489–496. doi: 10.1002/path.2131. [DOI] [PubMed] [Google Scholar]
- 36.Liley HG, White RT, Warr RG, Benson BJ, Hawgood S, Ballard PL. Regulation of messenger RNAs for the hydrophobic surfactant proteins in human lung. J Clin Invest. 1989;83:1191–1197. doi: 10.1172/JCI114000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pryhuber GS. Regulation and function of pulmonary surfactant protein B. Mol Genet Metab. 1998;64:217–228. doi: 10.1006/mgme.1998.2722. [DOI] [PubMed] [Google Scholar]
- 38.Stahlman MT, Gray ME, Whitsett JA. The ontogeny and distribution of surfactant protein B in human fetuses and newborns. J Histochem Cytochem. 1992;40:1471–1480. doi: 10.1177/40.10.1527371. [DOI] [PubMed] [Google Scholar]
- 39.Savov J, Wright JR, Young SL. Incorporation of biotinylated SP-A into rat lung surfactant layer, type II cells, and clara cells. Am J Physiol Lung Cell Mol Physiol. 2000;279:L118–L126. doi: 10.1152/ajplung.2000.279.1.L118. [DOI] [PubMed] [Google Scholar]
- 40.Rooney SA, Gobran LI, Wai-Lee TS. Stimulation of surfactant production by oxytocin-induced labor in the rabbit. J Clin Invest. 1977;60:754–759. doi: 10.1172/JCI108828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Marino PA, Rooney SA. The effect of labor on surfactant secretion in newborn rabbit lung slices. Biochim Biophys Acta. 1981;664:389–396. doi: 10.1016/0005-2760(81)90061-8. [DOI] [PubMed] [Google Scholar]
- 42.Mendelson CR, Condon JC. New insights into the molecular endocrinology of parturition. J Steroid Biochem Mol Biol. 2005;93:113–119. doi: 10.1016/j.jsbmb.2004.12.027. [DOI] [PubMed] [Google Scholar]
- 43.Kim CJ, Kim JS, Kim YM, Cushenberry E, Richani K, Espinoza J, Romero R. Fetal macrophages are not present in the myometrium of women with labor at term. Am J Obstet Gynecol. 2006;195:829–833. doi: 10.1016/j.ajog.2006.06.052. [DOI] [PubMed] [Google Scholar]
- 44.Lopez BA, Newman GE, Phizackerley PJ, Turnbull AC. Surfactant stimulates prostaglandin E production in human amnion. Br J Obstet Gynaecol. 1988;95:1013–1017. doi: 10.1111/j.1471-0528.1988.tb06506.x. [DOI] [PubMed] [Google Scholar]
- 45.Sun K, Brockman D, Campos B, Pitzer B, Myatt L. Induction of surfactant protein A expression by cortisol facilitates prostaglandin synthesis in human chorionic trophoblasts. J Clin Endocrinol Metab. 2006;91:4988–4994. doi: 10.1210/jc.2006-1472. [DOI] [PubMed] [Google Scholar]
- 46.Korfhagen TR, Bruno MD, Ross GF, Huelsman KM, Ikegami M, Jobe AH, Wert SE, Stripp BR, Morris RE, Glasser SW, et al. Altered surfactant function and structure in SP-A gene targeted mice. Proc Natl Acad Sci USA. 1996;93:9594–9599. doi: 10.1073/pnas.93.18.9594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Elhalwagi BM, Zhang M, Ikegami M, Iwamoto HS, Morris RE, Miller ML, Dienger K, McCormack FX. Normal surfactant pool sizes and inhibition-resistant surfactant from mice that overexpress surfactant protein A. Am J Respir Cell Mol Biol. 1999;21:380–387. doi: 10.1165/ajrcmb.21.3.3676. [DOI] [PubMed] [Google Scholar]
- 48.Mitchell MD, Chang MC, Chaiworapongsa T, Lan HY, Helliwell RJ, Romero R, Sato TA. Identification of 9alpha,11beta-prostaglandin F2 in human amniotic fluid and characterization of its production by human gestational tissues. J Clin Endocrinol Metab. 2005;90:4244–4248. doi: 10.1210/jc.2004-2496. [DOI] [PubMed] [Google Scholar]
- 49.Romero R, Munoz H, Gomez R, Parra M, Polanco M, Valverde V, Hasbun J, Garrido J, Ghezzi F, Mazor M, et al. Increase in prostaglandin bioavailability precedes the onset of human parturition. Prostaglandins Leukot Essent Fatty Acids. 1996;54:187–191. doi: 10.1016/s0952-3278(96)90015-0. [DOI] [PubMed] [Google Scholar]
- 50.Keski-Nisula LT, Aalto ML, Kirkinen PP, Kosma VM, Heinonen ST. Myometrial inflammation in human delivery and its association with labor and infection. Am J Clin Pathol. 2003;120:217–224. doi: 10.1309/KC6K-DTX9-8LFY-B3J7. [DOI] [PubMed] [Google Scholar]
- 51.Keelan JA, Marvin KW, Sato TA, Coleman M, McCowan LM, Mitchell MD. Cytokine abundance in placental tissues: evidence of inflammatory activation in gestational membranes with term and preterm parturition. Am J Obstet Gynecol. 1999;181:1530–1536. doi: 10.1016/s0002-9378(99)70400-x. [DOI] [PubMed] [Google Scholar]
- 52.Haddad R, Tromp G, Kuivaniemi H, Chaiworapongsa T, Kim YM, Mazor M, Romero R. Human spontaneous labor without histologic chorioamnionitis is characterized by an acute inflammation gene expression signature. Am J Obstet Gynecol. 2006;195:394–24. doi: 10.1016/j.ajog.2005.08.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Helmig BR, Romero R, Espinoza J, Chaiworapongsa T, Bujold E, Gomez R, Ohlsson K, Uldbjerg N. Neutrophil elastase and secretory leukocyte protease inhibitor in prelabor rupture of membranes, parturition and intra-amniotic infection. J Matern Fetal Neonatal Med. 2002;12:237–246. doi: 10.1080/jmf.12.4.237.246. [DOI] [PubMed] [Google Scholar]
- 54.Rubio F, Cooley J, Accurso FJ, Remold-O’Donnell E. Linkage of neutrophil serine proteases and decreased surfactant protein-A (SP-A) levels in inflammatory lung disease. Thorax. 2004;59:318–323. doi: 10.1136/thx.2003.014902. [DOI] [PMC free article] [PubMed] [Google Scholar]