Abstract
Lmna −/− mice display multiple tissue defects and die by 6–8 weeks of age reportedly from dilated cardiomyopathy with associated conduction defects. We sought to determine whether restoration of lamin A in cardiomyocytes improves cardiac function and extends the survival of Lmna −/− mice. We observed increased total desmin protein levels and disorganization of the cytoplasmic desmin network in ∼20% of Lmna −/− ventricular myocytes, rescued in a cell-autonomous manner in Lmna −/− mice expressing a cardiac-specific lamin A transgene (Lmna −/−; Tg). Lmna −/−; Tg mice displayed significantly increased contractility and preservation of myocardial performance compared to Lmna −/− mice. Lmna −/−; Tg mice attenuated ERK1/2 phosphorylation relative to Lmna −/− mice, potentially underlying the improved localization of connexin43 to the intercalated disc. Electrocardiographic recordings from Lmna −/− mice revealed arrhythmic events and increased frequency of PR interval prolongation, which is partially rescued in Lmna −/−; Tg mice. These findings support our observation that Lmna −/−; Tg mice have a 12% median extension in lifespan compared to Lmna −/− mice. While significant, Lmna −/−; Tg mice only have modest improvement in cardiac function and survival likely stemming from the observation that only 40% of Lmna −/−; Tg cardiomyocytes have detectable lamin A expression. Cardiomyocyte-specific restoration of lamin A in Lmna −/− mice improves heart-specific pathology and extends lifespan, demonstrating that the cardiac pathology of Lmna −/− mice limits survival. The expression of lamin A is sufficient to rescue certain cellular defects associated with loss of A-type lamins in cardiomyocytes in a cell-autonomous fashion.
Introduction
Nuclear lamins are type V intermediate filament proteins that are implicated in a variety of cellular processes, including DNA replication, gene transcription and chromatin organization [1], [2], [3], [4]. Mutations within the A-type lamin gene, LMNA, are associated with over 13 different tissue-specific diseases, collectively termed laminopathies. These include autosomal Emery-Dreifuss muscular dystrophy (EDMD2/3), limb-girdle muscular dystrophy type 1B (LGMD1B), and dilated cardiomyopathy with conduction defects 1A (CMD1A). Adipose, bone, and neural tissues can also be affected in laminopathies, which may resemble aspects of accelerated or premature aging (for review see Worman, et al. 2010) [5], [6].
Mice lacking the Lmna gene appear normal at birth but progressively display multiple tissue defects, including muscular dystrophy and dilated cardiomyopathy, with a noticeable reduction in growth rate beginning as early as 2 weeks of age followed by premature death at 6–8 weeks [7]. Further analysis of hearts from Lmna −/− mice has revealed a rapid development of left ventricular dilatation coupled with decreased systolic function beginning after 2 weeks of age [8]. Dosage of A-type lamins can also influence cardiac function as Lmna+/− mice display cardiac conduction defects with a late onset of dilated cardiomyopathy [9]. However, expression of either major isoform alone, lamin A only [10] or lamin C only [11], is sufficient to prevent phenotypes observed in Lmna −/− mice, indicating that either isoform can largely compensate for the other. Interestingly, only homozygous—but not heterozygous—knock-in mouse models for either muscular dystrophy-associated or cardiac-specific LMNA mutations [12], [13] display dilated cardiomyopathy with conduction defects and premature death. In contrast, humans heterozygous for the corresponding missense mutations [14], [15] develop cardiac and skeletal muscle pathology, indicating that there are subtle differences in disease manifestation between rodents and humans.
In this study, we tested whether cardiomyocyte-specific expression of lamin A [16] can improve cardiac function and increase lifespan of Lmna −/− mice. We show better preservation of myocardial performance and reduced occurrence of conduction abnormalities for Lmna −/− mice expressing the cardiac transgene. These observations are consistent with a partial restoration of localization and protein levels of desmin, connexin43 and ERK1/2 phosphorylation. The heterogenic expression of the cardiac lamin A transgene in Lmna −/− hearts underlies this partial restoration and limits lifespan extension, however, with this model we are able to investigate cell-autonomous and non-cell autonomous roles which lends insight into the biology of A-type lamins in the cardiac system.
Results
Cardiac-specific expression of FLAG-tagged human lamin A in Lmna −/− mice
To determine whether cardiac-specific expression of lamin A can improve heart function in Lmna −/− mice, we crossed transgenic mice expressing FLAG-tagged human lamin A under the α-myosin heavy chain promoter [16] with Lmna +/− mice to ultimately produce litters containing both Lmna −/− mice and Lmna −/− mice expressing the cardiac-specific LMNA transgene (Lmna −/−; Tg). FLAG-lamin A is highly expressed in Lmna −/−; Tg cardiac tissue as measured by Western analysis using an A-type lamin antibody ( Figure 1A ), and expression is specific to cardiac tissue in both Lmna +/+; Tg and Lmna −/−; Tg mice ( Figure 1B ). Indirect immunofluorescence microscopy using antibodies against myosin heavy chain (MF-20) and FLAG ( Figure 1C ) indicate that ∼35% of ventricular myocytes express the FLAG-lamin A transgene in both Lmna +/+; Tg and Lmna −/−; Tg hearts ( Figure 1D ). Finally, expression of the cardiac-specific transgene in Lmna −/−; Tg mice does not improve the growth defect of Lmna −/− mice as measured by their body weights compared to Lmna +/+ mice ( Figure 1E ). Collectively, these results show that the FLAG-lamin A transgene is highly – though mosaically – expressed and specific to cardiomyocytes.
Figure 1. FLAG-lamin A is highly expressed, tissue-specific, and mosaic.
(A) Western blot analysis of Lmna +/+, Lmna −/−, and Lmna −/−; Tg heart lysates that were probed with lamin A/C antibody. (B) Western blot analysis of heart, gastrocnemius, quadriceps, thymus, spleen, kidney, liver, and lung from Lmna +/+; Tg (upper panel) and Lmna −/−; Tg (lower panel) mice probing with lamin A/C antibody. The asterisk indicates a cross-reactive band present in all lanes. (C) Indirect immunofluorescence micrograph of Lmna −/−; Tg heart cross-section stained for MF-20 (cardiomyocytes; green), FLAG (transgene; red), and DAPI (nuclei, blue). Note that the mosaic expression of the transgene is limited to only cardiomyocytes. Scale bar denotes 100 µm. Inset is zoomed in view from the same image. (D) Quantitation of the percent of cardiomyocytes expressing FLAG-lamin A (N = 3 each; ∼500 cardiomyocytes were scored for each group) (E) Body weights of Lmna +/+, Lmna −/−, and Lmna −/−; Tg mice (N = 10, 11, and 8 at 4–6 weeks respectively and N = 10, 7, and 5 at 6–8 weeks respectively).
Characterization of molecular phenotypes associated with cardiac structure and remodeling in Lmna −/− and Lmna −/−; Tg hearts
Western analysis was performed on lysates derived from hearts of 5–7 week old Lmna +/+ Lmna −/−, and Lmna −/−; Tg mice to determine whether molecular phenotypes are restored in the presence of cardiac lamin A expression. Desmin, previously determined to be mislocalized in Lmna −/− cardiomyocytes [8], exhibits an approximately 3-fold increase in total protein levels from Lmna −/− hearts ( Figure 2A ). This finding in cardiomyocytes contrasts with skeletal muscle myoblasts from Lmna −/− mice, where desmin levels are reduced [17]. Expression of FLAG-lamin A in the heart results in attenuation of total desmin that is only approximately 2-fold increased compared to Lmna +/+ hearts. Desmin is mislocalized in Lmna −/− ventricular myocytes, resulting in diminished staining in the majority of intercalated discs ( Figures 2B vs. 2C) as well as increased cytoplasmic desmin staining in ∼21% of ventricular myocytes (P<0.001) ( Figure 2E ). Less than 1% of transgene-expressing ventricular myocytes from Lmna −/−; Tg mice display increased cytoplasmic desmin (P<0.001) ( Figures 2D and 2E ), suggesting cell-autonomous rescue of desmin localization. Due to the mosaic nature of our transgene expression, we continued to observe increased cytoplasmic desmin in the transgene-non-expressing ventricular myocytes, but also noted a 38% reduction in the fraction of these cells, possibly due to neighboring transgene-expressing ventricular myocytes (P<0.01; see Discussion). These findings are consistent with the increase of desmin in hearts of Lmna −/− mice and the partial attenuation of desmin levels in Lmna −/−; Tg mice.
Figure 2. Transgenic expression of lamin A in Lmna −/− cardiomyocytes results in partial restoration of desmin phenotype but not cardiac remodeling markers.
(A) Western blot analysis of Lmna +/+, Lmna −/−, and Lmna −/−; Tg whole heart lysates. Desmin is increased in Lmna −/− heart lysates compared to Lmna +/+ and is partially attenuated in Lmna −/−; Tg mice (middle blot). α-tubulin is shown as a loading control (bottom blot). (B–D) Indirect immunofluorescence micrographs showing desmin (red), FLAG (green), and DAPI (blue) in Lmna +/+ (B), Lmna −/− (C), and Lmna −/−; Tg (D) heart ventricle sections. Asterisks mark intercalated discs, which co-localize with a pan-cadherin antibody (not shown) and arrows indicate ventricular myocytes with accumulated desmin in the cytoplasm. Scale bar denotes 50 µm. (D) Note the lack of ventricular myocytes positive for both desmin cytoplasmic accumulation and transgene expression. (E) Graph summarizing the desmin cytoplasmic accumulation phenotype observed in Lmna −/− ventricular myocytes across different mouse backgrounds aged 5–7 weeks (N = 3 for Lmna +/+, Lmna −/−, and Lmna −/−; Tg; ** P<0.01; *** P<0.001).
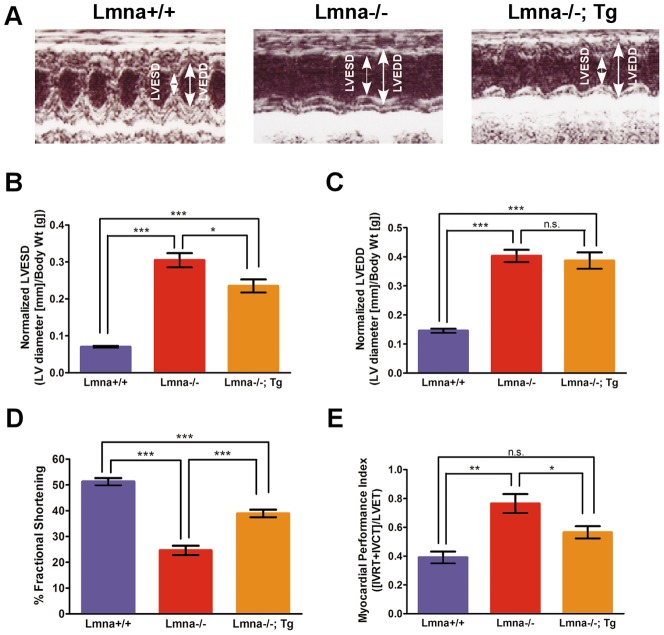
Improved cardiac contractile function in transgenic Lmna −/− mice
To determine whether transgenic expression of FLAG-lamin A improves cardiac function in Lmna −/− mice, transthoracic echocardiograms were performed at 4–8 weeks of age. Echocardiographic measurements comparing Lmna +/+ and Lmna +/+; Tg mice showed no significant difference (Figure S1 A–C). Lmna −/− mice display impaired contractility and left ventricular dilatation ( Figure 3A ). They have significantly increased (P<0.001) left ventricular end-systolic diameters (LVESD) and left ventricular end-diastolic diameters (LVEDD) normalized for body weight between Lmna −/− and Lmna +/+ littermates ( Figures 3B & 3C ). Additionally, Lmna −/− hearts are enlarged as shown by the ∼55% increase in the left ventricular mass index (LVMI) (Figure S2). Fractional shortening decreased by 50% in Lmna −/− mice compared to Lmna +/+ littermates (P<0.001) ( Figure 3D ) and consistent with previous observations [8]. As a second measure of cardiac function, the myocardial performance index (MPI) was calculated (See methods for further details; increased MPI scores have been shown previously to reflect LV systolic and/or diastolic dysfunction [18]). Lmna −/− mice display 100% increase in MPI compared to Lmna +/+ littermates (P<0.001) ( Figure 3E ), consistent with decreased cardiac contractility in Lmna −/− mice.
Figure 3. Lmna −/−; Tg mice display improved contractile function compared to Lmna −/− littermates.
(A) Representative echocardiograms of Lmna −/−, Lmna −/−; Tg, and control littermates. (B) Normalized left ventricular end-systolic diameter (LVESD) measurements are increased in Lmna −/− hearts compared to control hearts and are improved in Lmna −/−; Tg hearts. (C) Normalized left ventricular end-diastolic diameter (LVEDD) measurements are increased in Lmna −/− hearts and are not improved in Lmna −/−; Tg hearts. (D) Fractional shortening is decreased in Lmna −/− hearts compared to control hearts and is improved in Lmna −/−; Tg hearts. (E) Myocardial performance index (MPI) is increased (worse) in Lmna −/− mice, but is improved in Lmna −/−; Tg mice. For all of the above experiments: Control, N = 13; Lmna −/−, N = 15; Lmna −/−; Tg, N = 12 for 4–8 weeks of age. One-way ANOVA analyses were performed (B–E) and significant genotype differences are listed above for each panel. Post-tests were performed between genotypes and significance is listed as follows: * P<0.05; ** P<0.01; *** P<0.001; n.s. not significant.
The expression of FLAG-lamin A partially restores cardiac contractility in Lmna −/− mice ( Figures 3A, 3B, 3D ). Compared to Lmna −/− mice, Lmna −/−; Tg mice significantly attenuate dilation of LVESD by ∼25% (P<0.01) ( Figure 3B ) and display ∼60% improvement in fractional shortening (P<0.001) although fractional shortening is still 20% lower in comparison to control littermates (P<0.001) ( Figure 3D ). Similarly, the MPI is partially improved by ∼25% in Lmna −/−; Tg mice compared to Lmna −/− mice (P<0.01) ( Figure 3E ). However, hearts of Lmna −/−; Tg mice still exhibit cardiac enlargement, as LVEDD ( Figure 3C ) and LVMI (Figure S2) are not significantly changed compared to hearts of Lmna −/− mice. Furthermore, mRNA expression of atrial natriuretic factor and brain natriuretic peptide, hallmarks of cardiac remodeling and physical stress, are not significantly changed in hearts of Lmna −/−; Tg mice compared to those from Lmna −/− mice (Figure S3). Collectively, these data indicate that the mosaic expression of FLAG-lamin A in Lmna −/− cardiomyocytes results in partial but significant restoration of cardiac contractility compared to Lmna −/−, yet fails to ameliorate cardiac dilation and remodeling.
Characterization of molecular phenotypes associated with cardiac conduction in both Lmna −/− and Lmna −/−; Tg mice
Increased ERK signaling as reflected by levels of phosphorylated ERK1/2 (pERK1/2) has been observed in hearts of LmnaH222P/H222P mice [19] and in other cell systems with knockdown of A-type lamin or emerin expression [20]. Consistently, we detect a 2.5-fold increase of pERK1/2 in Lmna −/− hearts relative to total ERK1/2 levels (P<0.01) ( Figure 4A and 4B ). Although we were unable to achieve significance, Lmna −/−; Tg (n = 11) hearts trend towards a reduced pERK1/2 level compared to hearts from Lmna −/− mice (n = 16), suggesting that there may be a partially decreased cellular stress response. Increases in ERK1/2 activity have been shown to negatively affect gap junction communication through phosphorylation of connexin43 (Cx43) [21], [22]. We find no significant change in levels of total Cx43 as detected by the NT1 antibody. However, in Lmna −/− hearts we detect a significant increase in CT1 signal, an antibody which predominantly recognizes Cx43 found in the cytoplasm and is increased in ischemic hearts [23] ( Figure 4A ). The increase in CT1 signal is not significantly attenuated in the hearts from Lmna −/−; Tg mice. In addition to changes detectable by immunoblot, we also observe a decreased amount of gap junctional Cx43 in hearts from 5–7 week old Lmna −/− mice compared to similarly aged Lmna +/+ mice via immunofluorescence ( Figures 4C and 4D ). Hearts from Lmna −/−; Tg mice appear to have more Cx43 present at the gap junction than hearts from Lmna −/− mice ( Figure 4E ). To quantitatively assay the loss of Cx43 in the intercalated discs of ventricular myocytes from 5–7 week old Lmna −/− mice, we formulated a co-association index for scoring Cx43 at intercalated discs (see methods). Intercalated discs from Lmna +/+ ventricular myocytes display a mean Cx43 index of 0.70, which is reduced to 0.39 in Lmna −/− ventricular myocytes ( Figure 4F ). Intercalated discs from Lmna −/−; Tg ventricular myocytes display a mean Cx43 index of 0.53. When normalized against the mean index for Lmna+/+ ventricular myocytes for each experiment, ventricular myocytes from Lmna −/− mice display 45% decrease in Cx43 localization at the intercalated disc (P<0.01) ( Figure 4G ). In contrast, ventricular myocytes from Lmna −/−; Tg mice display 25% decrease in Cx43 localization at the intercalated disc compared to Lmna+/+ ventricular myocytes (P = 0.11), suggesting a partial restoration of Cx43 localization at the intercalated disc.
Figure 4. Transgenic expression of lamin A in Lmna −/− cardiomyocytes partially restores ERK1/2 activation and gap junction protein localization.
(A) Western blot analysis of Lmna +/+, Lmna −/−, and Lmna −/−; Tg mouse heart lysates. Phosphorylated-ERK1/2 (pERK1/2) levels are increased in Lmna −/− compared to Lmna +/+ and are decreased in Lmna −/−; Tg hearts while total ERK1/2 levels are unchanged. Total Cx43 protein levels detected by NT1 are unchanged, while cytoplasmic levels of Cx43 as detected by CT1 are increased. α-tubulin is shown as a loading control. (B) Quantification of pERK1/2 levels normalized to total ERK1/2 levels in 5–7 week old Lmna +/+ (N = 13), Lmna −/− (N = 16), and Lmna −/−; Tg (N = 11) mouse heart lysates. One-way ANOVA analyses were performed for significance with post-test significance values as follows: ** P<0.01; n.s. not significant. (C–E) Indirect immunofluorescence micrographs of pan-cadherin (red) and Cx43 (green) in Lmna +/+ (C), Lmna −/− (D), and (E) Lmna −/−; Tg heart ventricle sections taken at equivalent exposure times from 6-week old mice. Asterisks mark intercalated discs with qualitative decreases in Cx43 relative to pan-cadherin staining. White scale bar denotes 50 µm. (F) Distribution of individual intercalated disks summarizing relative frequency of Cx43 co-association index in 5–7-week old Lmna +/+ (N = 3; 350 intercalated discs analyzed total), Lmna −/− (N = 3; 349 intercalated discs analyzed total), and Lmna −/−; Tg (N = 3; 367 intercalated discs analyzed total) mice. A non-linear regression for Gaussian distribution was fitted for each group tested (Lmna +/+ R2 = 0.91; Lmna −/− R2 = 0.96; Lmna −/−; Tg R2 = 0.80). (G) Combined Cx43 co-association indices from multiple experiments normalized against their respective Lmna +/+ control (N = 3). One-way ANOVA analyses were performed for significance with post-test significance values as follows: ** P<0.01; n.s. not significant.
Transgenic Lmna −/− mice have less severe conduction defects
We hypothesized that if Cx43 localization was partially restored in Lmna −/−; Tg mice, there may be improvements in cardiac conduction. To test this hypothesis, we took electrocardiography (ECG) recordings of 5–7 week old mice and measured a number of parameters associated with cardiac conduction, including PR and QRS intervals. Despite the previous study reporting a significant increase in the average PR and QRS intervals for Lmna −/− mice [8], we were unable to observe a significant increase in the average PR or QRS intervals of similarly aged Lmna −/− mice after analyzing 300 beats per mouse (N = 5) using this method (Table S1). However, intermittent prolongation of the PR interval was noted in Lmna −/− mice. Since there is no consensus on the normal reference values for PR intervals, we derived the reference values by analyzing 1,500 beats from 5 Lmna+/+ littermates, which display a Gaussian distribution. PR intervals from single mice were then compared against the derived reference value and values greater than 95% of our normal reference were considered abnormally prolonged ( Figure 5A ). Using the cut-off value of >30% abnormally prolonged PR intervals, 5 out of 6 Lmna −/− mice displayed PR prolongation (Fisher's exact test, P = 0.0476), suggesting the presence of first degree atrioventricular block ( Figure 5B ). In contrast, 2 out of 5 Lmna −/−; Tg mice exhibited PR prolongation (Fisher's exact test, P = 0.2222), suggesting partial improvement in atrioventricular conduction due to cardiac-specific FLAG-lamin A expression. One Lmna +/+; Tg mouse exhibited >30% PR interval prolongation and a second mouse displayed a dropped beat (Figure S4), which might be a manifestation of sinus pause, sinoatrial or atrioventricular block, suggesting that increased expression of lamin A in a subset of normal myocytes can also result in conduction abnormalities. We also observe several arrhythmic events in Lmna −/− mice. Compared to a normal ECG tracing from a representative Lmna +/+ mouse ( Figure 5C ), the ECG of Lmna −/− mice display intermittent atrial premature beat ( Figure 5D ) and a multifocal atrial rhythm ( Figure 5E ). We were unable to locate any similar arrhythmic occurrences in Lmna −/−; Tg mice, potentially due to improved cardiac function.
Figure 5. Lmna−/−; Tg mice show improved conduction parameters compared to Lmna−/− mice by ECG analysis.
(A) Box-and-whiskers plot of pooled Lmna +/+ PR intervals (N = 5) compared with single animal PR intervals for each of the following genetic backgrounds at 5-7 weeks of age: Lmna +/+; Tg, Lmna −/− and Lmna −/−; Tg. The whiskers represent at 5–95% confidence interval and individual outliers are represented by their respective dots. (B) Fraction of mice which display >30% abnormally prolonged PR interval. Lmna +/+ (N = 5), Lmna +/+; Tg (N = 5), Lmna −/− (N = 6), and Lmna −/−; Tg (N = 5). (C–E) ECG traces from a Lmna +/+ mouse (C), a Lmna −/− mouse exhibiting an atrial premature beat (D) and a Lmna −/− mouse displaying multifocal atrial rhythm (E). Note the changes in P-wave morphology and cycle length. Arrows denote ectopic P-waves. RR intervals are shown above.
Transgene expression of FLAG-lamin A in Lmna −/− cardiomyocytes extends lifespan
Concurrent with our studies of cardiac function and molecular restoration in cardiomyocytes from Lmna −/−; Tg mice, we sought to determine whether this improvement would translate into an increased lifespan. Kaplan-Meier curves were generated from a cohort of 24 and 28 mice each for transgenic and non-transgenic Lmna −/− mice, respectively. Despite the mosaic expression of FLAG-lamin A in Lmna −/−; Tg cardiomyocytes, we observe a 12% mean increase and a 15% maximal increase in lifespan (P<0.011) of Lmna −/−; Tg mice compared to non-transgenic Lmna −/− littermates ( Figure 6 ).
Figure 6. Extension of median and maximal lifespan in Lmna −/− mice expressing cardiac lamin A.

Lmna −/−; Tg mice display a 12% and 15% increase in median and maximal lifespans, respectively, compared to Lmna −/− littermates (Lmna +/+; Tg N = 10; Lmna −/− N = 24; Lmna −/−; Tg N = 28). The Log-Rank test, which measures significance evenly across all time points, reports a significance probability of P<0.0114
Discussion
In this study, we tested the hypothesis that cardiomyocyte-specific expression of lamin A in Lmna −/− mice can restore cardiac function and increase lifespan. Through the generation of Lmna −/− mice with cardiomyocyte-specific expression of FLAG-lamin A, we observed significantly improved fractional shortening and myocardial performance index by echocardiogram, restored localization of both desmin and Cx43, and attenuated protein levels of both pERK1/2 and desmin, resulting in partial restoration of cardiac function compared to Lmna −/− mice. Despite increased cardiac contractility, cardiac remodeling in transgenic Lmna −/− mice was still evident with no amelioration of chamber dilation. We observed less Cx43 localized to the intercalated disk in ventricular myocytes of Lmna −/− mice which was partially restored in ventricular myocytes of Lmna −/−; Tg mice. This modest improvement in Cx43 localization was also consistent with our finding that the stochastic PR interval prolongation observed in Lmna −/− mice is less frequent in Lmna −/−; Tg mice. The improvements to cardiac function due to the expression of lamin A resulted in a significant—though modest—extension in lifespan compared to Lmna −/− littermates. Collectively, these data suggest that cardiomyocyte-specific expression of lamin A in Lmna −/− mice can partially rescue cardiac function and that the cardiac pathology present in Lmna −/− mice is lifespan limiting.
Mosaic expression of lamin A in Lmna −/− cardiomyocytes was very likely a limiting factor in many of our incompletely rescued phenotypes, but also allowed us to observe a juxtaposition of cardiomyocytes either expressing or not expressing the lamin A transgene. Other studies had previously used this approach to address cell autonomy roles in the cardiac system [24], [25]. In our study, Lmna −/−; Tg mice displayed ∼30–40% heterogeneity of lamin A transgene expression in ventricular cardiomyocytes, and we observed both cell-autonomous and non-cell-autonomous phenotypes. In general, our findings suggest the contractile defect of Lmna −/− mice can be rescued in a cell-autonomous fashion, as indicated by restoration of the desmin cytoskeletal network in lamin A-expressing Lmna −/− cardiomyocytes. We also observed a significantly decreased fraction of non-transgene expressing Lmna −/−; Tg cardiomyocytes with disorganized desmin, which we postulate as being propagated from neighboring cardiomyocytes expressing the lamin A transgene. Since mechanotransduction relies on communication between the nucleus and extracellular interactions from cell-cell contact, extracellular matrix composition, and secreted factors [26], [27], we propose that A-type lamins may play an additional role by modulating these extracellular properties to coordinate mechanosensing and transduction in a non-cell-autonomous manner.
Altered A-type lamin function also results in increased activity of ERK1/2. Although this phenomenon is universal to many cell types with abnormal A-type lamin composition [20], [28], [29], [30], the administration of the MEK inhibitor, PD98059, improves the dilated cardiomyopathy of LmnaH222P/H222P mice [31], [32], which strongly supports the notion that altered ERK1/2 activity is a critical component associated with pathogenesis. Indeed, increased ERK1/2 activity is associated with cardiac hypertrophy in other heart disease models [33]. Cx43 is the most widely distributed member of the connexin family of proteins, which forms gap junctions, facilitates cell-to-cell communication, and is found in a variety of different tissues and cell types [34]. Phosphorylation of Cx43 by ERK1/2 inhibits gap-junctional communication [21], [22], and decreased Cx43 activity at the intercalated disc in Lmna −/− mice may play a critical part in the conduction defects and premature death observed. Electrical conduction of the heart cannot be completely rescued in a cell-autonomous fashion as gap junctions must form stable connexons with neighboring cells in order to maintain a functioning channel, making it an attractive example of non-cell-autonomous function. Mice carrying a heterozygous deletion for Cx43 [35], as well as cardiac-restricted inactivation of Cx43 in adult mice [36], [37], show slowing of ventricular conduction and eventual death by ventricular tachycardia. In addition, heterogeneous or mosaic expression of Cx43 resulted in similar spontaneous ventricular arrhythmia and altered conduction velocity [25], [38]. We observed heterogeneity in the rescue of gap-junctional Cx43 levels, which we speculate contributes to the continued premature death phenotype of Lmna −/−; Tg mice through terminal arrhythmic events. Interestingly, a similar phenotype describing loss of Cx43 localization to the intercalated disc coupled with desmin aggregation has been described in D7-des mice, which encode a deletion that causes human dilated cardiomyopathy [39]. Additionally, we observed arrhythmia in two Lmna +/+; Tg mice suggesting that increased expression of lamin A in a fraction of cardiomyocytes can alter conduction—and possibly connexin activity—in an otherwise normal heart. These examples demonstrate the importance of assessing non-cell-autonomous outcomes in any transgenic and gene therapy models.
ERK1/2 activity can also result in the activation of different transcriptional pathways depending on the context of the tissue type and origin of ERK1/2 activation [40]. A-type lamins can bind and sequester c-Fos, the immediate downstream effector of pERK1/2, thereby inhibiting the activity of AP-1 [41]. Further studies have shown that active ERK1/2 can also co-localize with lamin A and c-Fos at the nuclear envelope, and the loss of A-type lamins results in increased AP-1 activity and reduced c-Fos phosphorylation [29]. Finally, ERK1/2 may additionally act upon Cx43 in a cell survival pathway, as ERK1/2 activity enhances translocation of Cx43 to the mitochondria, where Cx43 has been shown to play a protective role against cell death [42], [43], [44]. Although it is currently not known whether ERK1/2 signaling can modulate desmin filament formation in cardiomyocytes, it is becoming increasingly clear that loss of or mutation of A-type lamins may impinge on multiple pathways that lead to cardiac dysfunction.
Our data highlight the role of A-type lamins in cardiomyocyte function to both maintain efficient contraction and preserve a functional conductive network. These findings also re-emphasize the need for uniform expression in gene therapy models correcting conduction diseases.
Methods
Animals
Lmna +/− mice were obtained from Dr. Colin Stewart [7] and were backcrossed on C57BL/6J for 9 additional generations. The resulting Lmna +/− mice, B6.129S1(Cg)-Lmnatm1Stw/BkknJ, were crossed with transgenic mice (founder line 903; B6/CBA F1 hybrids) expressing FLAG-lamin A under the control of the α-myosin heavy chain promoter, which have been described elsewhere [16]. The resulting transgenic Lmna +/− mice were crossed with Lmna +/− mice to generate Lmna −/− mice expressing the cardiac-specific transgene (Lmna −/−; Tg). Details for genotyping can be found in Methods S1. Mice were bred and maintained under specific pathogen-free conditions. All experiments were performed in compliance with the University of Washington Institutional Animal Care and Use Committee (Protocol Number: 2174–23).
Tissue preparation and indirect immunofluorescence
Hearts of transgenic and non-transgenic Lmna −/− and control mice were rapidly excised and rinsed in phosphate-buffered saline (PBS) prior to mounting in Tissue-Tek O.C.T. compound and subsequent freezing in liquid nitrogen-cooled isopentane. Heart ventricle sections 8 µm in thickness were collected and mounted on Superfrost plus glass slides (Fisher). Antibodies used and detailed methods for staining and scoring desmin and connexin can be found in Methods S1.
Western analysis
Tissues were homogenized using the TH homogenizer (Omni International) in a lysis buffer (10 µL/mg tissue) containing the following: 2% SDS, 250 mM sucrose, 75 mM urea, 1 mM DTT, 50 mM Tris (pH 7.5), 25 µg/mL aprotinin, and 10 µg/mL leupeptin [13], [19]. Antibodies used and further details can be found in Methods S1.
Echocardiography
Transgenic and non-transgenic Lmna −/− mice and control littermates received echocardiography examination (Siemens Acuson CV70). A mixture of 0.5% isoflurane with O2 was administered through a nose cone to provide adequate sedation but minimal cardiac suppression during echocardiography. Heart rate was monitored and kept above 400 bpm to minimize any effect on any parameters [45]. Pathologists performing the echocardiography examination were blinded to the transgene genotype for Lmna −/− mice. Further details can be found in Methods S1.
ECG recordings
Electrocardiography was recorded using TA11ETA-F10 transmitters (Data Science International, St.Paul, MN). ECG data were recorded using Dataquest A.R.T™software (Data Sciences International) and the first 300 beats were analyzed using LabChart7 Pro (ADInstruments). Further details can be found in Methods S1.
Statistical analysis
Data are expressed as mean ± S.E.M. and one-way ANOVA analyses were performed where relevant with Bonferroni post tests to compare genotypes of specific age groups. A value of P<0.05 was considered statistically significant. For lifespan analysis, a Kaplan-Meier curve was generated and Log Rank Tests were performed with a value of P<0.05 that was considered statistically significant. All statistical analyses and graphs were performed using GraphPad Prism 5.02.
Supporting Information
Lmna +/+ and Lmna +/+; Tg hearts show no significant difference in cardiac function. (A & B) No significant difference is noted in (A) fractional shortening, (B) myocardial performance index (MPI), or (C) left ventricular mass index (LVMI) of Lmna +/+ and Lmna +/+; Tg hearts at 4–8 weeks of age as measured by echocardiography. Two-tailed unpaired t-tests were used to determine P-values which are listed for each panel. (Lmna +/+, N = 6; Lmna +/+; Tg, N = 7).
(TIF)
Lmna−/− hearts are enlarged relative to control littermates and Lmna−/− ; Tg hearts are not significantly improved. Left ventricular mass of 4–8 week old mice was measured and normalized to body weight to resolve the LVMI. One-way ANOVA was performed and significant genotype differences are listed for each panel. Bonferonni post-tests were performed between genotypes and significance is listed as follows: * P<0.05; n.s. not significant. (Control, N = 13; Lmna−/−, N = 15; Lmna−/−; Tg, N = 12).
(TIF)
mRNA levels of global remodeling markers, ANF and BNP, are enriched in Lmna −/− and Lmna −/−; Tg mice. qPCR of cardiac remodeling mRNA's for Lmna −/− and Lmna −/−; Tg hearts. Data are presented as fold-increase over Lmna +/+ hearts. Global cardiac remodeling mRNA's, ANF and BNP, are all increased in Lmna −/− hearts and are not significantly changed in Lmna −/−; Tg hearts. (Lmna+/+, N = 4; Lmna −/−, N = 7; Lmna −/−; Tg, N = 5).
(TIF)
Dropped beat in Lmna +/+; Tg heart. An isolated case of a dropped heartbeat was noted in a single Lmna +/+; Tg heart during ECG recording which could reflect either a sinus pause or sino-atrial block.
(TIF)
Average ECG parameters from 5–7 week old mice. ECG parameters of individual mice from Lmna+/+ and Lmna −/− mice either expressing or not expressing FLAG-lamin A in cardiomyocytes. Similar mouse genotypes are grouped together with mouse ID displayed and parameters are averaged. Each parameter from an individual mouse represents an averaged value from the first 300 beats recorded. Parameters include RR interval, Heart Rate, PR interval, P duration, and QRS interval.
(TIF)
Additional information and specifics on methods with supporting references. Further details include genotyping primers, qPCR primers, specific antibodies used for both immunofluorescence and Western blotting, details on image analysis, and animal techniques.
(DOC)
Acknowledgments
The authors would like to thank Sara Mamman, Rubysue Mangalindan, Ashot Safarli and other members of the Ladiges lab in the Department of Comparative Medicine for handling and maintenance of mouse colonies. The MF20 myosin antibody (Fischman) was obtained from the Developmental Studies Hybridoma Bank under the auspices of the NICHD, maintained by University of Iowa (Biological Sciences, Iowa City, IA 52242).
Funding Statement
Contract grant sponsor: National Institute on Aging at the National Institutes of Health; Contract grant number: R01 AG024287. Contract grant sponsor: National Institutes of Health; Contract grant number: T32 HL007312. Contract grant sponsor: National Institute of Arthritis and Musculoskeletal and Skin Diseases at the National Institutes of Health; Contract grant number: R01 AR048997. Contract grant sponsor: National Institutes of Health; Contract grant number: HL085686. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Lee DC, Welton KL, Smith ED, Kennedy BK (2009) A-type nuclear lamins act as transcriptional repressors when targeted to promoters. Exp Cell Res 315: 996–1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Shumaker DK, Solimando L, Sengupta K, Shimi T, Adam SA, et al. (2008) The highly conserved nuclear lamin Ig-fold binds to PCNA: its role in DNA replication. J Cell Biol 181: 269–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Spann TP, Goldman AE, Wang C, Huang S, Goldman RD (2002) Alteration of nuclear lamin organization inhibits RNA polymerase II-dependent transcription. J Cell Biol 156: 603–608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Spann TP, Moir RD, Goldman AE, Stick R, Goldman RD (1997) Disruption of nuclear lamin organization alters the distribution of replication factors and inhibits DNA synthesis. J Cell Biol 136: 1201–1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Worman HJ, Ostlund C, Wang Y (2010) Diseases of the nuclear envelope. Cold Spring Harb Perspect Biol 2: a000760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Burtner CR, Kennedy BK (2010) Progeria syndromes and ageing: what is the connection? Nat Rev Mol Cell Biol 11: 567–578. [DOI] [PubMed] [Google Scholar]
- 7. Sullivan T, Escalante-Alcalde D, Bhatt H, Anver M, Bhat N, et al. (1999) Loss of A-type lamin expression compromises nuclear envelope integrity leading to muscular dystrophy. J Cell Biol 147: 913–920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Nikolova V, Leimena C, McMahon AC, Tan JC, Chandar S, et al. (2004) Defects in nuclear structure and function promote dilated cardiomyopathy in lamin A/C-deficient mice. J Clin Invest 113: 357–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Wolf CM, Wang L, Alcalai R, Pizard A, Burgon PG, et al. (2008) Lamin A/C haploinsufficiency causes dilated cardiomyopathy and apoptosis-triggered cardiac conduction system disease. J Mol Cell Cardiol 44: 293–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Davies BS, Barnes RH, 2nd, Tu Y, Ren S, Andres DA, et al (2010) An accumulation of non-farnesylated prelamin A causes cardiomyopathy but not progeria. Hum Mol Genet 19: 2682–2694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Fong LG, Ng JK, Lammerding J, Vickers TA, Meta M, et al. (2006) Prelamin A and lamin A appear to be dispensable in the nuclear lamina. J Clin Invest 116: 743–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Mounkes LC, Kozlov SV, Rottman JN, Stewart CL (2005) Expression of an LMNA-N195K variant of A-type lamins results in cardiac conduction defects and death in mice. Hum Mol Genet 14: 2167–2180. [DOI] [PubMed] [Google Scholar]
- 13. Arimura T, Helbling-Leclerc A, Massart C, Varnous S, Niel F, et al. (2005) Mouse model carrying H222P-Lmna mutation develops muscular dystrophy and dilated cardiomyopathy similar to human striated muscle laminopathies. Hum Mol Genet 14: 155–169. [DOI] [PubMed] [Google Scholar]
- 14. Bonne G, Mercuri E, Muchir A, Urtizberea A, Becane HM, et al. (2000) Clinical and molecular genetic spectrum of autosomal dominant Emery-Dreifuss muscular dystrophy due to mutations of the lamin A/C gene. Ann Neurol 48: 170–180. [PubMed] [Google Scholar]
- 15. Fatkin D, MacRae C, Sasaki T, Wolff MR, Porcu M, et al. (1999) Missense mutations in the rod domain of the lamin A/C gene as causes of dilated cardiomyopathy and conduction-system disease. N Engl J Med 341: 1715–1724. [DOI] [PubMed] [Google Scholar]
- 16. Wang Y, Herron AJ, Worman HJ (2006) Pathology and nuclear abnormalities in hearts of transgenic mice expressing M371K lamin A encoded by an LMNA mutation causing Emery-Dreifuss muscular dystrophy. Hum Mol Genet 15: 2479–2489. [DOI] [PubMed] [Google Scholar]
- 17. Frock RL, Kudlow BA, Evans AM, Jameson SA, Hauschka SD, et al. (2006) Lamin A/C and emerin are critical for skeletal muscle satellite cell differentiation. Genes Dev 20: 486–500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Dai DF, Santana LF, Vermulst M, Tomazela DM, Emond MJ, et al. (2009) Overexpression of catalase targeted to mitochondria attenuates murine cardiac aging. Circulation 119: 2789–2797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Muchir A, Pavlidis P, Decostre V, Herron AJ, Arimura T, et al. (2007) Activation of MAPK pathways links LMNA mutations to cardiomyopathy in Emery-Dreifuss muscular dystrophy. J Clin Invest 117: 1282–1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Muchir A, Wu W, Worman HJ (2009) Reduced expression of A-type lamins and emerin activates extracellular signal-regulated kinase in cultured cells. Biochim Biophys Acta 1792: 75–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Warn-Cramer BJ, Cottrell GT, Burt JM, Lau AF (1998) Regulation of connexin-43 gap junctional intercellular communication by mitogen-activated protein kinase. J Biol Chem 273: 9188–9196. [DOI] [PubMed] [Google Scholar]
- 22. Ruch RJ, Trosko JE, Madhukar BV (2001) Inhibition of connexin43 gap junctional intercellular communication by TPA requires ERK activation. J Cell Biochem 83: 163–169. [DOI] [PubMed] [Google Scholar]
- 23. Sosinsky GE, Solan JL, Gaietta GM, Ngan L, Lee GJ, et al. (2007) The C-terminus of connexin43 adopts different conformations in the Golgi and gap junction as detected with structure-specific antibodies. Biochem J 408: 375–385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Gary-Bobo G, Parlakian A, Escoubet B, Franco CA, Clement S, et al. (2008) Mosaic inactivation of the serum response factor gene in the myocardium induces focal lesions and heart failure. Eur J Heart Fail 10: 635–645. [DOI] [PubMed] [Google Scholar]
- 25. Gutstein DE, Morley GE, Fishman GI (2001) Conditional gene targeting of connexin43: exploring the consequences of gap junction remodeling in the heart. Cell Commun Adhes 8: 345–348. [DOI] [PubMed] [Google Scholar]
- 26. Gonzalez S, Aguilera S, Urzua U, Quest AF, Molina C, et al. (2011) Mechanotransduction and epigenetic control in autoimmune diseases. Autoimmun Rev 10: 175–179. [DOI] [PubMed] [Google Scholar]
- 27. Dahl KN, Ribeiro AJ, Lammerding J (2008) Nuclear shape, mechanics, and mechanotransduction. Circ Res 102: 1307–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Favreau C, Delbarre E, Courvalin JC, Buendia B (2008) Differentiation of C2C12 myoblasts expressing lamin A mutated at a site responsible for Emery-Dreifuss muscular dystrophy is improved by inhibition of the MEK-ERK pathway and stimulation of the PI3-kinase pathway. Exp Cell Res 314: 1392–1405. [DOI] [PubMed] [Google Scholar]
- 29. Gonzalez JM, Navarro-Puche A, Casar B, Crespo P, Andres V (2008) Fast regulation of AP-1 activity through interaction of lamin A/C, ERK1/2, and c-Fos at the nuclear envelope. J Cell Biol 183: 653–666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Rodriguez J, Calvo F, Gonzalez JM, Casar B, Andres V, et al. (2010) ERK1/2 MAP kinases promote cell cycle entry by rapid, kinase-independent disruption of retinoblastoma-lamin A complexes. J Cell Biol 191: 967–979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Muchir A, Shan J, Bonne G, Lehnart SE, Worman HJ (2009) Inhibition of extracellular signal-regulated kinase signaling to prevent cardiomyopathy caused by mutation in the gene encoding A-type lamins. Hum Mol Genet 18: 241–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Wu W, Muchir A, Shan J, Bonne G, Worman HJ (2011) Mitogen-activated protein kinase inhibitors improve heart function and prevent fibrosis in cardiomyopathy caused by mutation in lamin A/C gene. Circulation 123: 53–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kodama H, Fukuda K, Pan J, Sano M, Takahashi T, et al. (2000) Significance of ERK cascade compared with JAK/STAT and PI3-K pathway in gp130-mediated cardiac hypertrophy. Am J Physiol Heart Circ Physiol 279: H1635–1644. [DOI] [PubMed] [Google Scholar]
- 34. Laird DW (2006) Life cycle of connexins in health and disease. Biochem J 394: 527–543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Guerrero PA, Schuessler RB, Davis LM, Beyer EC, Johnson CM, et al. (1997) Slow ventricular conduction in mice heterozygous for a connexin43 null mutation. J Clin Invest 99: 1991–1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Danik SB, Liu F, Zhang J, Suk HJ, Morley GE, et al. (2004) Modulation of cardiac gap junction expression and arrhythmic susceptibility. Circ Res 95: 1035–1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Eckardt D, Theis M, Degen J, Ott T, van Rijen HV, et al. (2004) Functional role of connexin43 gap junction channels in adult mouse heart assessed by inducible gene deletion. J Mol Cell Cardiol 36: 101–110. [DOI] [PubMed] [Google Scholar]
- 38. Gutstein DE, Danik SB, Lewitton S, France D, Liu F, et al. (2005) Focal gap junction uncoupling and spontaneous ventricular ectopy. Am J Physiol Heart Circ Physiol 289: H1091–1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Gard JJ, Yamada K, Green KG, Eloff BC, Rosenbaum DS, et al. (2005) Remodeling of gap junctions and slow conduction in a mouse model of desmin-related cardiomyopathy. Cardiovasc Res 67: 539–547. [DOI] [PubMed] [Google Scholar]
- 40. Aplin M, Christensen GL, Schneider M, Heydorn A, Gammeltoft S, et al. (2007) Differential extracellular signal-regulated kinases 1 and 2 activation by the angiotensin type 1 receptor supports distinct phenotypes of cardiac myocytes. Basic Clin Pharmacol Toxicol 100: 296–301. [DOI] [PubMed] [Google Scholar]
- 41. Ivorra C, Kubicek M, Gonzalez JM, Sanz-Gonzalez SM, Alvarez-Barrientos A, et al. (2006) A mechanism of AP-1 suppression through interaction of c-Fos with lamin A/C. Genes Dev 20: 307–320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Polontchouk L, Ebelt B, Jackels M, Dhein S (2002) Chronic effects of endothelin 1 and angiotensin II on gap junctions and intercellular communication in cardiac cells. Faseb J 16: 87–89. [DOI] [PubMed] [Google Scholar]
- 43. Lu G, Haider H, Porollo A, Ashraf M (2010) Mitochondria-specific transgenic overexpression of connexin-43 simulates preconditioning-induced cytoprotection of stem cells. Cardiovasc Res 88: 277–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Rottlaender D, Boengler K, Wolny M, Michels G, Endres-Becker J, et al. (2010) Connexin 43 acts as a cytoprotective mediator of signal transduction by stimulating mitochondrial KATP channels in mouse cardiomyocytes. J Clin Invest 120: 1441–1453. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 45. Roth DM, Swaney JS, Dalton ND, Gilpin EA, Ross J Jr (2002) Impact of anesthesia on cardiac function during echocardiography in mice. Am J Physiol Heart Circ Physiol 282: H2134–2140. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Lmna +/+ and Lmna +/+; Tg hearts show no significant difference in cardiac function. (A & B) No significant difference is noted in (A) fractional shortening, (B) myocardial performance index (MPI), or (C) left ventricular mass index (LVMI) of Lmna +/+ and Lmna +/+; Tg hearts at 4–8 weeks of age as measured by echocardiography. Two-tailed unpaired t-tests were used to determine P-values which are listed for each panel. (Lmna +/+, N = 6; Lmna +/+; Tg, N = 7).
(TIF)
Lmna−/− hearts are enlarged relative to control littermates and Lmna−/− ; Tg hearts are not significantly improved. Left ventricular mass of 4–8 week old mice was measured and normalized to body weight to resolve the LVMI. One-way ANOVA was performed and significant genotype differences are listed for each panel. Bonferonni post-tests were performed between genotypes and significance is listed as follows: * P<0.05; n.s. not significant. (Control, N = 13; Lmna−/−, N = 15; Lmna−/−; Tg, N = 12).
(TIF)
mRNA levels of global remodeling markers, ANF and BNP, are enriched in Lmna −/− and Lmna −/−; Tg mice. qPCR of cardiac remodeling mRNA's for Lmna −/− and Lmna −/−; Tg hearts. Data are presented as fold-increase over Lmna +/+ hearts. Global cardiac remodeling mRNA's, ANF and BNP, are all increased in Lmna −/− hearts and are not significantly changed in Lmna −/−; Tg hearts. (Lmna+/+, N = 4; Lmna −/−, N = 7; Lmna −/−; Tg, N = 5).
(TIF)
Dropped beat in Lmna +/+; Tg heart. An isolated case of a dropped heartbeat was noted in a single Lmna +/+; Tg heart during ECG recording which could reflect either a sinus pause or sino-atrial block.
(TIF)
Average ECG parameters from 5–7 week old mice. ECG parameters of individual mice from Lmna+/+ and Lmna −/− mice either expressing or not expressing FLAG-lamin A in cardiomyocytes. Similar mouse genotypes are grouped together with mouse ID displayed and parameters are averaged. Each parameter from an individual mouse represents an averaged value from the first 300 beats recorded. Parameters include RR interval, Heart Rate, PR interval, P duration, and QRS interval.
(TIF)
Additional information and specifics on methods with supporting references. Further details include genotyping primers, qPCR primers, specific antibodies used for both immunofluorescence and Western blotting, details on image analysis, and animal techniques.
(DOC)