Abstract
BACKGROUND AND PURPOSE
Phagocyte function is critical for host defense against infections. Defects in phagocytic function lead to several primary immunodeficiencies characterized by early onset of recurrent and severe infections. In this work, we further investigated the effects of BAY 41-2272, a soluble guanylate cyclase (sGC) agonist, on the activation of human peripheral blood monocytes (PBM) and THP-1 cells.
EXPERIMENTAL APPROACH
THP-1 cells and PBM viability was evaluated by methylthiazoletetrazolium assay; reactive oxygen species production by lucigenin chemiluminescence; gene and protein expression of NAPDH oxidase components by qRT-PCR and Western blot analysis, respectively; phagocytosis and microbicidal activity by co-incubation, respectively, with zymosan and Escherichia coli; and cytokine release by elisa.
KEY RESULTS
BAY 41-2272, compared with the untreated group, increased spreading of monocytes by at least 35%, superoxide production by at least 50%, and gp91PHOX and p67PHOX gene expression 20 to 40 times, in both PBM and THP-1 cells. BAY 41-2272 also augmented phagocytosis of zymosan particles threefold compared with control, doubled microbicidal activity against E. coli and enhanced the release of TNF-α and IL-12p70 by both PBM and THP-1 cells. Finally, by inhibiting sGC with ODQ, we showed that BAY 41-2272-induced superoxide production and phagocytosis is not dependent exclusively on sGC activation.
CONCLUSIONS AND IMPLICATIONS
In addition to its ability to induce vasorelaxation and its potential application for therapy of vascular diseases, BAY 41-2272 was shown to activate human mononuclear phagocytes. Hence, it is a novel pro-inflammatory drug that may be useful for controlling infections in the immunocompromised host.
Keywords: BAY 41-2272, cGMP, NADPH oxidase, gp91PHOX, p67PHOX, microbicidal activity
Introduction
Phagocytes are responsible for the initiation of the immune response and their primary functions are the identification, ingestion (phagocytosis) and elimination of microorganisms, cellular debris and circulating immune complexes (Flannagan et al., 2009). Phagocytes are very important not only as effector cells but also as regulators of inflammatory responses, by releasing cytokines and several other mediators (Aggelidou et al., 2002). Primary defects in phagocytes result in the early onset of recurrent infections that lead to death if not properly managed (Samarghitean et al., 2009).
Chronic granulomatous disease (CGD) is an example of defective phagocytes, where these cells are unable to produce reactive oxygen species (ROS), resulting in flawed microbial killing (Geha et al., 2007). ROS are involved in phagocyte microbicidal mechanisms and are produced by the NADPH oxidase system, which consists of four main components, namely p47PHOX and p67PHOX, found in the cytosol; and p22PHOX and gp91PHOX, the so-called membrane cytochrome b558. These components are encoded, respectively, by NCF1, NCF2, CYBA and CYBB genes (El-Benna et al., 2005). IFN-γ therapy reduces the relative risk of severe infections in 70% in patients with CGD (Ezekowitz et al., 1988; Kobayashi et al., 2008); however, the mechanism involved in this reduction, initially attributed to positive regulation of iNOS (inducible NOS), has not been confirmed (Condino-Neto et al., 1996). Therefore, other mechanisms have been investigated to explain the beneficial effects of IFN-γ in CGD (Condino-Neto and Newburger, 2000).
One of the most biologically relevant actions of NO is its binding to the haeme moiety in the soluble guanylyl cyclase (sGC), leading to intracellular accumulation of the second messenger molecule, cGMP, which, in turn, regulates numerous pathophysiological processes (Pacher et al., 2007). There is extensive literature showing the relevance of NO in the microbicidal mechanisms against several intracellular pathogens in experimental models (Bogdan, 2001). In addition, cGMP has been shown to be a key messenger in many cellular immune processes, such as differentiation, chemotaxis, proliferation and release of soluble mediators (Kobialka and Gorczyca, 2001; Ciuman et al., 2006).
NO-independent sGC activators have emerged as valuable tools for investigating the physiopathology of the NO–sGC–cGMP signalling pathway (Stasch et al., 2001). One of most studied NO-independent sGC stimulator is BAY 41-2272, which is reported to act in synergism with NO to produce anti-aggregating, anti-proliferative and vasodilator effects (Evgenov et al., 2007). Our group has recently demonstrated that BAY 41-2272 increases the production of cGMP, release of superoxide and CYBB gene expression in human THP-1 mononuclear cells (Oliveira-Junior et al., 2007). These findings raise a new perspective for the development of new therapies for phagocyte defects and to treat infections by directly activating phagocytes and other cells of the immune system.
The aim of this work was to further evaluate the effects of BAY 41-2272 on the activation of human peripheral blood monocytes (PBM) and THP-1 cells. We specifically investigated the production of superoxide, expression of genes encoding components of the NADPH oxidase system, spreading, phagocytosis, microbicidal activity and cytokine release by these cells, all important indicators of phagocyte function.
Methods
Isolation and culture of human PBM and THP-1 cells
Peripheral blood monocyte cells (PBMC) were obtained from healthy volunteers and were isolated by density-gradient centrifugation over Ficoll-Paque® (Sigma Chemical Co., St. Louis, MO, USA), followed by anti-CD16 antibody-labelled magnetic beads columns for PBM isolation (MACS Separator, Mylteni Biotec, Bergish Gladbach, Germany). The cells were washed three times with PBS and re-suspended in RPMI 1640 culture medium supplemented with 10% heat-inactivated fetal calf serum (Gibco, Grand Island, NY, USA). THP-1 cells obtained from ATCC (Manassas, VA, USA) (human acute monocytic leukaemia cell line) were maintained as previously described (Tsuchiya et al., 1980).
Cell viability
Cell viability was assessed using a method based on the mitochondrial reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT; 5 mg·mL−1) to formazan (Thomazzi et al., 2005). Cells (1 × 106 cells·mL−1, 100 µL) were treated with either BAY 41-2272 (1.0, 3.0 and 10.0 µM), phorbol myristate acetate (PMA; 30 and 300 nM) or dimethyl sulfoxide (DMSO, drug vehicle, 0.7%) for either 30 or 120 min at 37°C. Preliminary screening experiments were carried out to select effective drug concentrations not toxic to cells. Previous studies have demonstrated that concentrations of BAY 41-2272 (up to 10.0 µM) are effective in inducing the production of cGMP and cAMP in mononuclear cells (Oliveira-Junior et al., 2007).
Measurement of superoxide production
Superoxide anion production by PBM and THP-1 cells was measured as described previously (Hatanaka et al., 2006). After a 48 h incubation with or without BAY 41-2272 (1.0 and 3.0 µM) or IFN-γ (100 U·mL−1), a lucigenin (1 mM)-preloaded cell suspension (1 × 106 cells·mL−1, 100 µL) was stimulated by adding PMA (30 nM) or IFN-γ (100 U·mL−1). The chemiluminescence was monitored for 2 h using a microplate luminometer reader (EG&G Berthold LB96V, Bad Wildbad, Germany), and the results are expressed as relative light units (RLU). Peak levels of RLU were recorded to calculate the mean of the maximum superoxide production during the observation period.
CYBB, CYBA, NCF1 and NCF2 mRNA expression
mRNA was obtained after lysis of PBM and THP-1 cells (1 × 107 cells·mL−1) previously treated for 48 h with IFN-γ (100 U·mL−1) or BAY 41-2272 (1.0 and 3.0 µM) using Trizol reagent (Invitrogen, Carlsbad, CA, USA) followed by reverse transcription with Super Script III RT and random hexamers (Invitrogen) to cDNA. For quantitative real-time PCR analysis, SYBR green MasterMix (Applied Biosystems, Foster City, CA, USA) was used in an ABI 5700 thermocycler. All results were normalized to human β-actin. Table 1 shows primers sequence used in the reactions.
Table 1.
Real-time PCR primers sequences of genes encoding components of NADPH system
| Protein/gene | Forward primer | Reverse primer |
|---|---|---|
| p22phox (CYBA) | 5′-atg tgg gcc aac gaa cag-3′ | 5′-gta ctc cag cag gca cac aa-3′ |
| gp91phox (CYBB) | 5′-ttg tgg aaa ccc tcc tat ga-3′ | 5′-aaa acc gca cca acc tct c.-3′ |
| p47phox (NCF1) | 5′-cct ctt tcc agt gca ttt aag g-3′ | 5′-gat gtg acg gat gaa ggt gtc-3′ |
| p67phox (NCF2) | 5′-cgg aca aga agg act gga ag-3′ | 5′-gga agt aag cca ctg cca ag-3′ |
| β-actin | 5′- tca ccg agc gcg gct-3′ | 5′-taa tgt cac gca cga ttt ccc-3′ |
p22PHOX, p47PHOX and p67PHOX protein expression by Western blot
PBM were first treated with IFN-γ (100 U·mL−1) or BAY 41-2272 (1.0 and 3.0 µM) for 48 h. After removal of the medium, cells (1 × 107 cells·mL−1) were washed in PBS and lysed in ice-cold lysis buffer [2% Triton X-100, 10 mM Tris–HCl (pH 8), 150 mM NaCl, 2 mM NaN3, 2 mM EDTA] containing protease inhibitors (Roche Molecular Biochemicals, Indianapolis, IN, USA) for 45 min at 4°C. The soluble proteins (20 µg) were separated by 7.5% SDS-PAGE, and the gel was electroblotted on nitrocellulose membrane (Hybond-ECL; Amersham, Buckinghamshire, UK) and then probed with monoclonal antibodies for p22PHOX, p47PHOX, p67PHOX or β-actin, according to the method described by Chen et al. (2004). The immunoblots were labelled with horseradish-labelled anti-mouse IgG and detected via enhancement with chemiluminescence reagent. The total pixel intensity analyses of the blots were conducted by using ImageJ 1.44 (developed by Wayne Rasband, National Institutes of Health, Bethesda, MD, USA; available at http://rsb.info.nih.gov/ij/index.html).
gp91PHOX protein expression by flow cytometry
Gp91PHOX expression in PBM (1 × 106 cells·mL−1) treated with PMA (30 nM), IFN-γ (100 U·mL−1) or BAY 41-2272 (1.0 and 3.0 µM) for 48 h was assessed by using mAbs directed against an external epitope of gp91phox (7D5; Yamauchi et al., 2001). Fluorescence was measured on a FACSCanto II flow cytometry (BD Biosciences, San Diego, CA, USA), and results are expressed as percentage of cells expressing gp91PHOX and mean fluorescence intensity (MFI) values in arbitrary units (au).
Spreading assay
After a 1 or 48 h incubation with BAY 41-2272 (1.0 and 3.0 µM), IFN-γ (100 U·mL−1) or PMA (30 nM), PBM and THP-1 cells were labelled with a fluorescent dye, calcein-AM (Blease et al., 1999). For spreading assays, 100 µL of 1 × 106 cells·mL−1 labelled cells were placed over microscope glass slides for 1 h. Analysis was performed using a phase-contrast microscope (Nikon, Tokyo, Japan) at 400× magnification. The results are expressed as the percentage of adherent cells from a total of 200 cells.
Phagocytosis assay
After a 48 h treatment with or without BAY 41-2272 (1.0 and 3.0 µM) or IFN-γ (100 U·mL−1), PBM and THP-1 cells (1 × 106 cells·mL−1) were incubated with zymosan (Sigma; 1 × 107 particles·mL−1) for 2 h and stimulated by adding PMA (30 nM) or IFN-γ (100 U·mL−1). Phagocyting cells were defined on phase-contrast microscope (Nikon) as cells that internalized two or more particles. Results are expressed as a percentage of phagocyting cells from a total of 200 cells.
Microbicidal activity
An enteropathogenic Escherichia coli (EPEC) strain was used for microbicidal assays. EPEC were cultured overnight in brain heart infusion broth (BHI – Fluka, Bucks, Switzerland) under shaking, at 37°C, and its concentrations were estimated photometrically (an optical density of 1.0 at 600 nm was equivalent to 1 × 108 bacteria·mL−1). The CFU assay was performed as described previously (Bellinati-Pires et al., 1995). A proportion of 1:10 of cells and EPEC, opsonized or not with fresh human serum (FHS), were mixed and incubated at 37°C under shaking for 30, 60, 90 and 120 min, after which cells were lysed with 1.5% Triton X-100. Recovered bacteria were re-suspended in BHI broth and plated on BHI agar for an overnight incubation at 37°C. The colonies were counted and the CFU number was converted to % bacteria survived (Bellinati-Pires et al., 1995). Microbicidal activity was calculated as follows: 100 –[(CFU at time t/CFU at time 0) × 100] and expressed as % of maximum microbicidal activity (minimum % bacteria survived).
Cytokine production
PBM and THP-1 cells were cultured at 1 × 106 cells·mL−1 into 12-well plates treated with BAY 41-2272 (1.0 and 3.0 µM) or IFN-γ (100 U·mL−1) for 48 h. Supernatants were collected and assayed by elisa for IL-12p70 or TNF-α release (BD Biosciences) as described previously (Pitzurra et al., 2000).
BAY 41-2272 actions through sGC pathway
We performed a number of experiments using ODQ [100 µM 1H-(1,2,4) oxadiazole (4,3-α) quinoxaline-1-one], a specific sGC inhibitor, to investigate the effects of BAY 41-2272 on superoxide production and phagocytosis mediated through the sGC pathway. We established two groups of treatments: (i) cells treated for 2 h together or not with PMA (30 nM), IFN-γ (100 U·mL−1) or BAY 41-2272 (1.0 and 3.0 µM); (ii) cells pretreated with ODQ together or not with BAY 41-2272 for 48 h before PMA or IFN-γ stimulation. In both treatments, ODQ was given 1 h before treatments to guarantee its binding and inactivation of sGC. After this, we performed the same protocols described above, and analysed superoxide release and phagocytosis.
Data analysis and statistical procedures
Results are expressed as mean ± SEM of n experiments. Statistical analysis was carried out using a two-way ANOVA followed by the Bonferroni post-test for multiple comparisons (Erceg-Hurn and Mirosevich, 2008). Graphpad Prism version 5.0 (GraphPad, San Diego, CA, USA) was used to perform statistical analysis; results were deemed significant if P < 0.05.
Drugs, chemicals reagents and other materials
Human IFN-γ was purchased from R&D Systems (Minneapolis, MN), and PMA from Sigma (St. Louis, MO, USA). BAY 41-2272 was provided by Pharma Research Center, Bayer AG (Wuppertal, Germany).
Results
BAY 41-2272 does not affect cell viability
Since a high rate of toxicity would not be an appropriate feature for a potential treatment, we evaluated the effects of BAY 41-2272 on cell viability. The MTT reduction assay showed that neither 30 nor 120 min exposure of human PBM or THP-1 cells to BAY 41-2272 (1.0–10.0 µM) caused any toxic effect (two-way anova, n= 6, P > 0.05; Table 2). The vehicle used for BAY 41-2272 (0.7% DMSO) also does not affect cell viability.
Table 2.
Effect of BAY 41-2272 on the viability of PBM or THP-1 cells
| MTT reduction (% control) | |||
|---|---|---|---|
| Cells | Treatment | 30 min | 120 min |
| PBM | 1.0 µM BAY 41-2272 | 95.32 ± 5.41 | 104.30 ± 2.10 |
| 3.0 µM BAY 41-2272 | 98.62 ± 2.35 | 102.80 ± 0.44 | |
| 10.0 µM BAY 41-2272 | 98.01 ± 3.34 | 99.12 ± 3.00 | |
| 3.0 µM BAY 41-2272 + 100 U·mL−1 IFN-γ | 72 ± 1.45 | 44 ± 2.65* | |
| 0.7% DMSO | 98.75 ± 2.87 | 96.07 ± 3.05 | |
| 30 nM PMA | 94.03 ± 3.32 | 92.13 ± 2.72 | |
| 300 nM PMA | 92.10 ± 0.38 | 68.45 ± 2.62* | |
| THP-1 | 1.0 µM BAY 41-2272 | 102.00 ± 5.20 | 101.40 ± 1.96 |
| 3.0 µM BAY 41-2272 | 98.05 ± 0.65 | 106.85 ± 2.62 | |
| 10.0 µM BAY 41-2272 | 97.98 ± 3.06 | 105.78 ± 1.70 | |
| 3.0 µM BAY 41-2272 + 100 U·mL−1 IFN-γ | 78 ± 2.31 | 56 ± 0.20* | |
| 0.7% DMSO | 106.44 ± 2.07 | 102.05 ± 4.17 | |
| 30 nM PMA | 98.45 ± 2.65 | 94.76 ± 3.10 | |
| 300 nM PMA | 94.65 ± 2.54 | 84.33 ± 2.23* | |
Human PBM or THP-1 cell suspensions (2 × 106 cells·mL−1) were incubated at 37°C for 30 min or 120 min with BAY 41-2272 (1.0–10.0 µM) or PMA (30 or 300 nM). Each experiment was carried out in quintuplicate (n= 5).
The results are expressed as MTT reduction (% control, mean ± SEM).
anova, P < 0.05.
BAY 41-2272 increases IFN-γ- and PMA-induced superoxide production by human PBM and THP-1 cells
As ROS is known to have an important function in cell-mediated microbial death, we assessed whether BAY 41-2272 directly stimulated ROS production, as assayed by lucigenin-chemiluminescence. We used IFN-γ and PMA to stimulate ROS production and enable comparison between these treatments and BAY 41-2272 stimulation. BAY 41-2272 (1.0 and 3.0 µM) increased the amount of superoxide released by PBM and THP-1 cells after direct stimulation for 2 h (two-way anova, n= 6, P < 0.05) and also after 48 h with the 3.0 µM dose. BAY 41-2272 primed the PBM and THP-1 cells so that both IFN-γ and PMA were more potent at releasing superoxide after stimulation for 48 h (two-way anova, n= 6, P < 0.05; Figure 1A,B).
Figure 1.

BAY 41-2272 primed THP-1 cells and PBM for a potent release of superoxide after IFN-γ or PMA stimulation. BAY 41-2272 treatment (1.0 or 3.0 µM) for 2 and 48 h increases superoxide release in THP-1 (A) and PBM (B), as assessed by lucigenin-chemiluminescence assay, and was even more effective with subsequent stimulation with PMA (30 nM) or IFN-γ (100 U·mL−1) for 2 h. Results are expressed in RLU as a mean ± SEM of maximum production (n= 6 healthy volunteers, in triplicate for PBM, or n= 6 independent experiments, in triplicate, for THP-1). CONT: no stimulus. *P < 0.05, compared with basal production; **P < 0.05, compared with basal and BAY 41-2272 2 h treatment, by two-way anova followed by Bonferroni test.
BAY 41-2272 augments the expression of CYBB and NCF2 genes
The activation of the NADPH oxidase system involves the phosphorylation of p47PHOX and assembly of its components (El-Benna, 2005). In addition, regulation of the activity of the NADPH oxidase system also involves gene expression (Condino-Neto et al., 1998). Therefore, we assessed whether BAY 41-2272 had an effect on CYBB, CYBA, NCF1 and NCF2 gene expression. Treatment of PBM and THP-1 cells with BAY 41-2272 for 48 h caused a significant increase in CYBB and NCF2 expression, but not in CYBA and NCF1, both at 1.0 and 3.0 µM (two-way anova, n= 6, P < 0.05; Figure 2) compared with basal gene expression.
Figure 2.
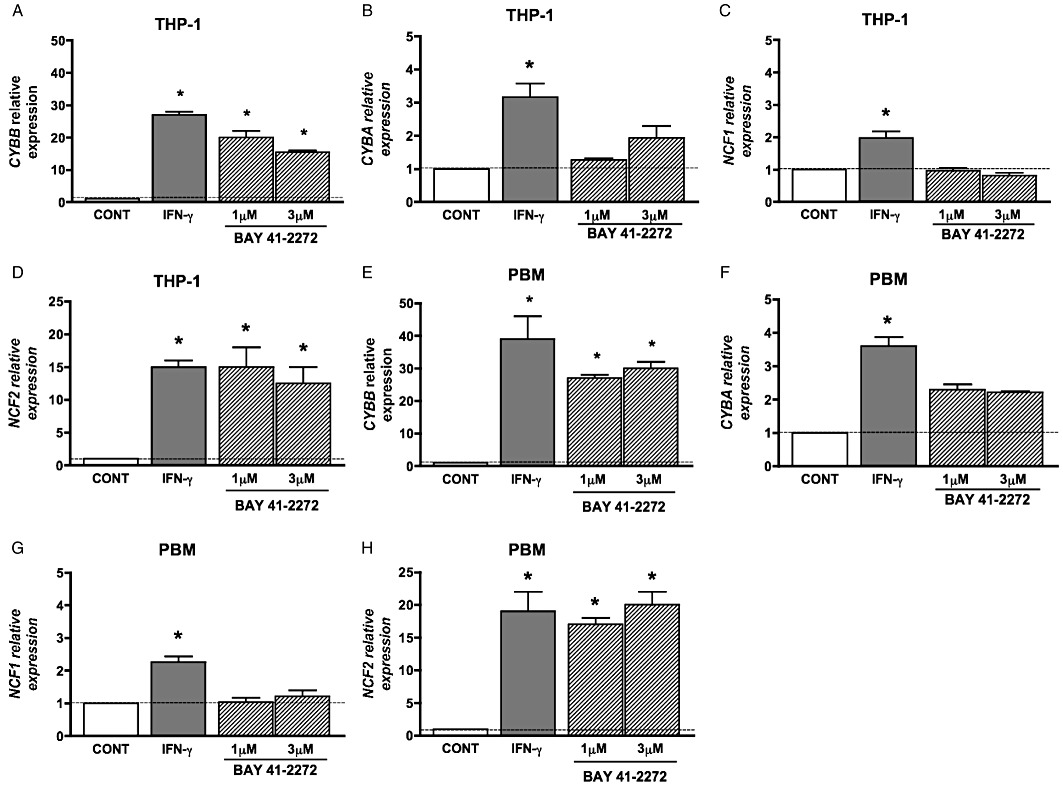
BAY 41-2272 increases CYBB (gp91PHOX) and NCF2 (p67PHOX), but not CYBA (p22PHOX) or NCF1 (p47PHOX), expression in THP-1 cells (A–B–C–D) and PBM (E–F–G–H). THP-1 (A–B–C–D) and PBM (E–F–G–H) treated with BAY 41-2272 (1.0 or 3.0 µM) increases CYBB and NCF2 expression levels after 48 h, quantified by qRT-PCR normalized to β-actin. BAY 41-2272 was less effective than IFN-γ (100 U·mL−1) treatment for the same time. Dashed line represents expression of genes in untreated cells. Results are shown as mean ± SEM of expression relative to untreated group (n= 6 healthy volunteers, in triplicate for PBM, or n= 6 independent experiments, in triplicate for THP-1). *P < 0.05, compared with basal expression, by two-way anova followed by Bonferroni test.
BAY 41-2272 increases p67PHOX and gp91PHOX protein expression
As we had demonstrated that BAY 41-2272 increased the levels of expression of CYBB and NCF2, we evaluated the protein expression of NADPH oxidase components. Our Western blot results confirmed that BAY 41-2272 increased the expression of p67PHOX but no changes in protein expression of p22PHOX and p47PHOX were observed (two-way anova, n= 4, P < 0.05; Figure 3A). Similarly, the flow cytometry of gp91PHOX showed an increased expression of this protein in groups treated with BAY 41-2272 (two-way anova, n= 4, P < 0.05; Figure 3B). These results show that gene and protein expression of these components was increased (Figure 2).
Figure 3.

BAY 41-2272 increases p67PHOX and gp91PHOX protein expression, but not p22PHOX and p47PHOX. PBM (1 × 107 cells·mL−1 for Western blot and 1 × 106 cells·mL−1 for flow cytometry) treated with BAY 41-2272 (1.0 or 3.0 µM) increased p67PHOX (A) and gp91PHOX (B) expression after 48 h, as quantified by Western blot (normalized to β-actin) and flow cytometry, respectively. Cells were also treated with IFN-γ (100 U·mL−1) and PMA (30 nM for flow cytometry). Dashed line represents basal expression normalized to β-actin expression. For Western blot, results are shown as a representative blot figure and as total pixel intensity of the blots. For flow cytometry, results are shown as mean ± SEM of percentage of cells expressing gp91PHOX or mean ± SEM of MFI (n= 4 healthy volunteers, in triplicate). *P < 0.05, compared to basal protein expression, by two-way anova followed by Bonferroni test.
BAY 41-2272 increases spreading and phagocytosis
The early defence response of mononuclear phagocytes involves the ingestion of microorganisms and the generation of activation signals by these cells. BAY 41-2272 (1.0 and 3.0 µM), treatment for 1 h or 48 h, increased the phagocytosis of zymosan induced by PBM and THP-1 cells (two-way anova, n= 6, P < 0.05; Figure 4B,D). The same pattern of response was observed for spreading (Figure 4A,C).
Figure 4.

BAY 41-2272 increases spreading and phagocytosis of THP-1 cells and PBM. THP-1 (A, B) and PBM (C, D) (1 × 106 cells·mL−1) pretreated with BAY 41-2272 for 48 h increased the spreading and phagocytic activity against zymosan. BAY 41-2272 (1.0 or 3.0 µM) treatment was even more effective when PMA (30 nM) and IFN-γ (100 U·mL−1) were added for 2 h after pretreatment. Number of spreading or phagocytosing cells is expressed as a percentage ± SEM of 200 total cells counted (n= 6 healthy volunteers, in triplicate for PBM, n= 6 independent experiments for THP-1).*P < 0.05, compared with basal (spreading or phagocytosis); **P < 0.05, compared to cells not pretreated, by two-way anova followed by Bonferroni test.
BAY 41-2272 increases the bactericidal activity against E. coli
Phagocytosis and ROS generation by mononuclear phagocytes have been kinetically correlated with increased bactericidal activity (DeLeo et al., 1999). Treatment with BAY 41-2272 (1.0 or 3.0 µM) produced a significant increase in the bactericidal activity of PBM and THP-1 cells against EPEC (two-way anova, n= 6, P < 0.05; Figure 5A,B). These results showed even higher significance for opsonized bacteria.
Figure 5.

BAY 41-2272 increases microbicidal activity of THP-1 cells and PBM. Pretreatment of THP-1 (A) and PBM (B) (1 × 106 cells·mL−1) with BAY 41-2272 (1.0 or 3.0 µM) for 48 h increased their microbicidal activity against EPEC, assessed by CFU counting from recovering internalized bacteria after co-culture. As positive control, cells were treated with IFN-γ (100 U·mL−1) for 48 h. Microbicidal activity is expressed as percentage ± SEM of maximum killing (minimum bacterial survival) (n= 6 healthy volunteers, in triplicate for PBM, n= 6 independent experiments, in triplicate for THP-1). *P < 0.05, compared with basal bacterial activity; **P < 0.05, compared with cells in same treatment condition, cultured with bacteria opsonized or not, by two-way anova followed by Bonferroni test.
BAY 41-2272 increases the release of TNF-α and IL-12p70
Cytokine production is a major aspect of the immune response and they are involved in both effector and regulatory mechanisms, including mononuclear phagocytes (Geissmann et al., 2008). Culturing PBM and THP-1 cells with BAY 41-2272 (1.0 and 3.0 µM) for 48 h caused an increase in TNF-α and IL-12p70 production (two-way anova, n= 6, P < 0.05; Figure 6).
Figure 6.
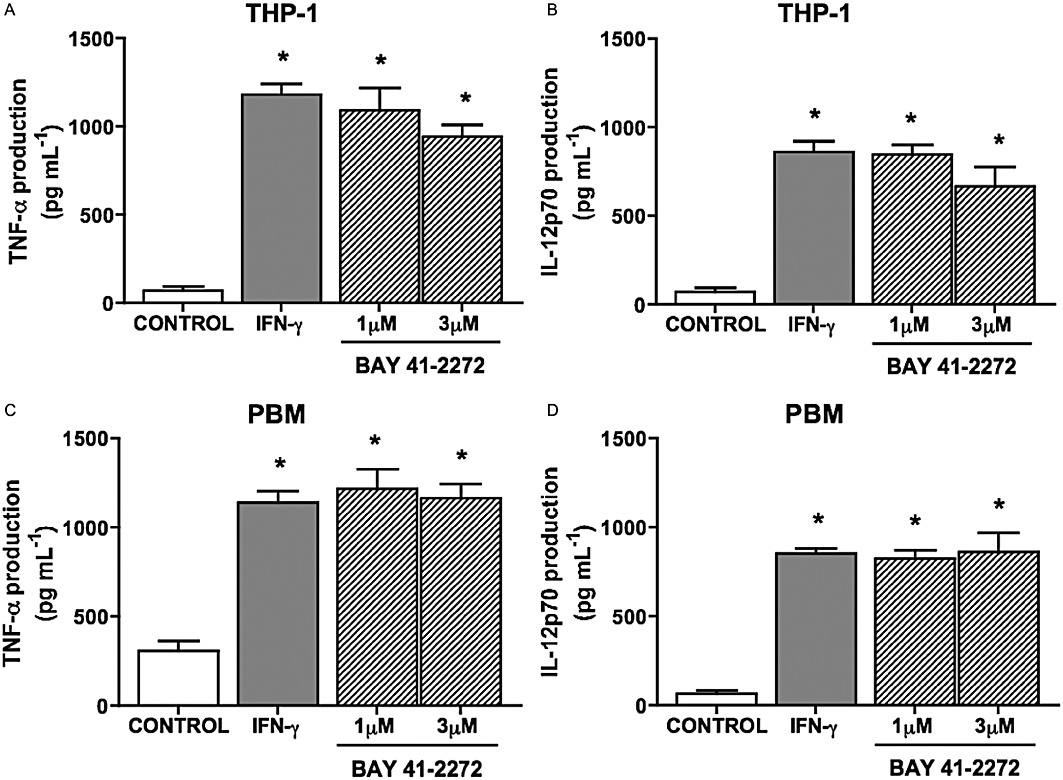
BAY 41-2272 increases the production of IL-12p70 and TNF-α by THP-1 cells and PBM. THP-1 (A,B) and PBM (C,D) cells (1 × 106 cells·mL−1) pretreated with BAY 41-2272 (1.0 or 3.0 µM) for 48 h increased TNF-α and IL-12p70 production. As positive control, cells were treated with IFN-γ (100 U·mL−1) for 48 h. Results are expressed as mean ± SEM (n= 6 healthy volunteers, in triplicate for PBM, n= 6 independent experiments, in triplicate for THP-1). *P < 0.05, compared with basal cytokine production, by one-way anova followed by Bonferroni post-test.
BAY 41-2272 produces effects on PBM and THP-1 cells through sGC pathway, specifically inhibited by ODQ
To clarify the pathways involved in the effects of BAY 41-2272, we inhibited sGC with ODQ to evaluate the specific interaction of the drug with this enzyme. We observed that treatment with ODQ did not result in a significant reduction of BAY 41-2272 effects on the parameters investigated, even when cells were treated with the higher dose of the compound (3 mM). With regard to the production of ROS (two-way anova, n= 6, P < 0.05; Figure 7), the groups treated with BAY 41-2272 plus ODQ did not show a significant reduction in ROS production in any of the concentrations used. Even when we did the pretreatment for 48 h, in the absence or presence of ODQ, BAY 41-2272 still showed considerable effects on ROS production (two-way anova, n= 6, P < 0.05; Figure 7C,D). Similar results were found for phagocytosis (two-way anova, n= 6, P < 0.05; Figure 8), using the same incubation protocol.
Figure 7.

BAY 41-2272 affects superoxide production in a manner not exclusively dependent on the sGC pathway. BAY 41-2272 treatment (1.0 or 3.0 µM) for 2 h (A,B) and 48 h (C,D) increased superoxide release in PBM and THP-1 cells (1 × 106 cells·mL−1), as assessed by lucigenin-chemiluminescence assay, even when sGC was specifically inhibited with ODQ (100 µM). We also treated cells with PMA (30 nM) or IFN-γ (100 U·mL−1) for 2 h, as controls. Dashed line represents expression of genes in untreated cells. Results are expressed in RLU as a mean ± SEM of maximum production (n= 4 healthy volunteers, in triplicate for PBM, or n= 4 independent experiments, in triplicate, for THP-1). *P < 0.05, compared with basal production by two-way anova followed by Bonferroni test.
Figure 8.

BAY 41-2272 affects phagocytosis in a manner not exclusively dependent on the sGC pathway. BAY 41-2272 treatment (1.0 or 3.0 µM) for 2 h (A,B) and 48 h (C,D) increases phagocytic activity of PBM and THP-1 cells (1 × 106 cells·mL−1) against zymosan, even when sGC was specifically inhibited with ODQ (100 µM). We also treated cells with PMA (30 nM) or IFN-γ (100 U·mL−1) for 2 h, as controls. Dashed line represents expression of genes in untreated cells. Number of phagocytosing cells is expressed as a percentage ± SEM of 200 total cells counted (n= 4 healthy volunteers, in triplicate for PBM, n= 4 independent experiments for THP-1). *P < 0.05, compared with basal spreading or phagocytosis; **P < 0.05, compared to cells not pretreated cells, by two-way anova followed by Bonferroni test.
Discussion and conclusions
The present study clearly demonstrates that 2 or 48 h treatment with BAY 41-2272 (1.0 or 3.0 µM) caused a marked increase in the activation of PBM and THP-1 cells, as demonstrated by induction of oxidative burst, increased phagocytosis and microbicidal activity, enhancement of IL-12p70 and TNF-α release, and also increased expression of CYBB and NCF2 genes in these cells. These observations support the hypothesis that BAY 41-2272 activates mononuclear phagocytes and has the potential to be used in the control of infections in the immunocompromised host.
ROS have an important function in cell-mediated microbial death and are an important indicator of monocytic cell activation (Huang et al., 2008). We used IFN-γ (a receptor-mediated activator) and PMA (a direct PKC activator) to stimulate ROS production as positive controls and, after treatment with BAY 41-2272, to test its potential for priming cells. We observed that BAY 41-2272 induced a marked increase in superoxide production, which may be related to PKC stimulation of the sGC–cGMP signalling pathway (Khare et al., 1994; Aggelidou et al., 2002). PKC promotes ‘phagocyte oxidase’ phosphorylation and activation of the NADPH oxidase system (Ueyama et al., 2007). Moreover, activation of PKC may explain the high response after just 2 h of treatment, since an increase in gene expression after this short time is not likely to occur for most genes, including the NADPH oxidase-related genes and proteins (Newburger et al., 1988).
As extensively described, activation of the NADPH oxidase system involves phosphorylation and clustering of proteins in membranes, but the maintenance of the response depends on the expression of new NADPH oxidase components (Quinn et al., 2006). Our results show that BAY 41-2272 markedly increases in the expression of CYBB and NCF2. Moreover, the protein expression of the NADPH oxidase components (p67PHOX and gp91PHOX) was also increased, confirming the data obtained by qRT-PCR. These results are similar to those found by Newburger et al. (1991), who showed an increase in the expression of these genes by up to 12 times after 24 h incubation of PBM with IFN-γ and also, similarly, Condino-Neto et al. (1998) found that IFN-γ increased the expression of CYBB in THP-1 cells. These results suggest that BAY 41-2272 stimulates not only signalling pathways but also important transcriptional factors related to the expression of CYBB and NCF2 genes, such as STAB1, CDP (Hawkins et al., 2001) and Pbx1, constitutively expressed in THP-1 (Skalnik and Neufeld, 1992; Eklund et al., 2000); HAF-1 (Newburger et al., 1994; Suzuki et al., 1998); and PU.1/IRF1/ICSBP/CBP (Eklund and Kakar, 1999). Recently, Luengo-Blanco et al. (2008) reported that binding of NF-κB transcription factor to CYBB is important for NAPDH oxidase function. Since there was a marked increased production of TNF-α, which is dependent on NF-κB activation (Ci et al., 2008), we suggest that BAY 41-2272 may activate the signalling pathways involved in NF-κB activation.
In addition to ROS release, phagocytosis is an essential function that should be analysed in phagocytes. Signal pathways activated by BAY 41-2272 are involved in cGMP generation and are linked to morphological changes required for migration, phagocytosis and spreading (Borán and García, 2007). We showed that BAY 41-2272 increased phagocytosis and this can be explained by PKG activation, as this was recently shown by Bóran et al. (2008) to promote a rapid release and expansion of filaments in microglia cells and increase in the phagocytosis function after treatment with ANP (another sGC stimulator) and dibutyryl-cGMP (permeable analogue of cGMP).
The phagocytosis activity and oxidative burst by mononuclear phagocytes have been kinetically correlated with increased bactericidal activity. It is recognized that ROS are highly toxic to bacteria such as E. coli (Nauseef, 2007), and this increase is probably related to the amount of oxidative burst and phagocytosis. However, other important parameters such as acidification of phagosome pH and release of lysosomal enzymes and granules (Ismail et al., 2002) have not yet been evaluated. Therefore, we cannot exclude a possible involvement of other factors such as the expression of surface molecules like Fc and complement receptors, making the phagocytosis more efficient and generating other signals that increase the phagocyte killing (Jongstra-Bilen et al., 2003; Luo et al., 2006; Rhein et al., 2008).
All these effects may be related to cytokine production involved with cellular activation and enhancement of the immune response. According to our expectations, treatment with BAY 41-2272 generated an increase in IL-12p70 release, since activated monocytes and macrophages produced high levels of these cytokines (Murray et al., 2006). BAY 41-2272 also promoted an increased production of TNF-α, strongly suggesting that this drug can activate MAPKs or NF-κB signalling pathways (Igbal and Zaidi, 2008). In addition, TNF-α production is thought to act like an autocrine, signalling cell activation by acting on the TNF-α receptor in monocyte cells (Deering and Orange, 2006).
Since we observed a wide spectrum of functions activated in treated phagocytes, we wondered whether all these effects of BAY 41-2272 resulted from an effect involving the sGC cyclase pathway. We observed that treatment with ODQ, a sGC specific inhibitor, did not result in a significant reduction of BAY 41-2272-induced superoxide production and phagocytosis. These results show that the effects of the compound are not related only to the sGC cyclase pathway, presenting activity on other signalling pathways.
Oliveira-Junior et al. (2007) showed that BAY 41-2272 is able to generate an increased production of cAMP, another important second messenger. Based on this, we suggest that other important pathways are activated by this compound. This increased production of cAMP could be due to a direct effect of the drug on the enzyme adenylate cyclase or an indirect effect through the production of cGMP. In addition to this hypothesis, Derbyshire et al. (2009) showed that the catalytic domains of GCs contain a catalytic and allosteric site capable of binding to both GTP and ATP nucleotides. This same group showed that the action of YC-1, a BAY 41-2272 analogue, leads to the production of cGMP and cAMP. This another alternative action of the drug, beyond the pathway involving sGC–cGMP–PKG.
Based on our results using ODQ and the results found by Koglin et al. (2002), showing BAY 41-2272 binding sites at two isoforms of sGC, we started to perform an in silico analysis of proteins with potential binding sites for BAY 41-2272. In our initial analysis, we found four proteins with potential binding sites for this compound and functions related to differentiation/activation of myelomonocytic cells (data not shown). These results open a new way of thinking for BAY 41-2272 analysis and new pathways involved in the activation of human myelomonocytic cells.
In summary, our present study consistently demonstrated that BAY 41-2272 activates human mononuclear phagocytes and has a great potential to control infections. However, the cardiovascular effects of BAY 41-2272 limit its clinical application, and this should be addressed in future in in vivo experimental and pre-clinical studies. Moreover, additional studies to clarify the signalling pathways involved in phagocyte activation will help to elucidate and modulate the effects of BAY 41-2272 both in the cardiovascular and immune systems. In addition, the effects of BAY 41-2272 in other immune cells, such as lymphocytes, are also needed to elucidate how this drug works and its potential applications for the immunocompromised host.
Acknowledgments
This work was supported by grants from Fundação de Amparo à Pesquisa do Estado de São Paulo (06/51004-2 and 06/52483-1) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (470978/2008-2).
Glossary
- BAY 41-2272
5-cyclopropyl-2-[1-(2-fluoro-benzyl)-1H-pyrazolo[3,4-b]pyridin-3-yl]-pyrimidin-4-ylamine
- BHI
brain heart infusion broth
- CFU
colony-forming unit
- CGD
chronic granulomatous disease
- DMSO
dimethyl sulfoxide
- EPEC
enteropathogenic Escherichia coli
- FHS
fresh human serum
- IL-12p70
IL-12 subunit p70
- MTT
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- ODQ
1H-(1,2,4) oxadiazole (4,3-alpha) quinoxaline-1-one
- PBM
peripheral blood monocytes
- PBMC
peripheral blood monocyte cell
- PMA
phorbol myristate acetate
- ROS
reactive oxygen species
- sGC
soluble guanylyl cyclase
Conflicts of interest
None.
References
- Aggelidou E, Hillhouse EW, Grammatopoulos DK. Up-regulation of nitric oxide synthase and modulation of the guanylate cyclase activity by corticotropin-releasing hormone but not urocortin II or urocortin III in cultured human pregnant myometrial cells. Proc Natl Acad Sci U S A. 2002;99:3300–3305. doi: 10.1073/pnas.052296399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellinati-Pires R, Salgado M, Hypolito IP, Grumach AS, Carneiro-Sampaio M. Application of a fluorochrome-lysostaphin assay to the detection of phagocytic and bactericidal disturbances in human neutrophils and monocytes. J Investig Allergol Clin Immunol. 1995;5:337–342. [PubMed] [Google Scholar]
- Blease K, Chen Y, Hellewell PG, Burke-Gaffney A. Lipoteichoic acid inhibits lipopolysaccharide-induced adhesion molecule expression and IL-8 release in human lung microvascular endothelial cells. J Immunol. 1999;163:6139–6147. [PubMed] [Google Scholar]
- Bogdan C. Nitric oxide and immune response. Nature. 2001;2:907–916. doi: 10.1038/ni1001-907. [DOI] [PubMed] [Google Scholar]
- Borán MS, García A. The cyclic GMP-protein kinase G pathway regulates cytoskeleton dynamics and motility in astrocytes. J Neurochem. 2007;102:216–230. doi: 10.1111/j.1471-4159.2007.04464.x. [DOI] [PubMed] [Google Scholar]
- Bóran MS, Baltrons MA, Garcia A. The ANP-cGMP-protein kinase G pathway induces a phagocytic phenotype but decreases inflammatory gene expression in microglial cells. Glia. 2008;56:394–411. doi: 10.1002/glia.20618. [DOI] [PubMed] [Google Scholar]
- Chen KC, Zhou Y, Xing K, Krysan K, Lou MF. Platelet derived growth factor (PDGF)-induced reactive oxygen species in the lens epithelial cells: the redox signaling. Exp Eye Res. 2004;78:1057–1067. doi: 10.1016/j.exer.2004.02.004. [DOI] [PubMed] [Google Scholar]
- Ci X, Song Y, Zeng F, Zhang X, Li H, Wang X, et al. Ceftiofur impairs pro-inflammatory cytokine secretion through the inhibition of the activation of NF-kappaB and MAPK. Biochem Biophys Res Commun. 2008;372:73–77. doi: 10.1016/j.bbrc.2008.04.170. [DOI] [PubMed] [Google Scholar]
- Ciuman M, Siednienko J, Czyzyk R, Witwicka H, Kołosionek E, Kobiałka M, et al. Cyclic GMP-dependent protein kinase and soluble guanylyl cyclase disappear in elicited rat neutrophils. Biochim Biophys Acta. 2006;1760:1618–1623. doi: 10.1016/j.bbagen.2006.09.002. [DOI] [PubMed] [Google Scholar]
- Condino-Neto A, Newburger P. Interferon-gamma improves splicing efficiency of CYBB gene transcripts in an interferon-responsive variant of chronic granulomatous disease due to a Splice site consensus region mutation. Blood. 2000;95:3548–3554. [PubMed] [Google Scholar]
- Condino-Neto A, Muscara MN, Bellinati-Pires R, Carneiro-Sampaio MM, Brandão AC, Grumach AS, et al. Effect of therapy with recombinant human interferon-gamma on the release of nitric oxide by neutrophils and mononuclear cells from patients with chronic granulomatous disease. J Interferon Cytokine Res. 1996;16:357–364. doi: 10.1089/jir.1996.16.357. [DOI] [PubMed] [Google Scholar]
- Condino-Neto A, Whitney C, Newburger PE. Dexamethasone but not indomethacin inhibits human phagocyte nicotinamide adenine dinucleotide phosphate oxidase activity by down-regulating expression of genes encoding oxidase components. J Immunol. 1998;161:4960–4967. [PubMed] [Google Scholar]
- Deering RP, Orange J. Development a clinical assay to evaluate toll-like receptor function. Clin Vaccine Immunol. 2006;13:68–76. doi: 10.1128/CVI.13.1.68-76.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeLeo FR, Allen LA, Apicella M, Nauseef WM. NADPH oxidase activation and assembly during phagocytosis. J Immunol. 1999;163:6732–6740. [PubMed] [Google Scholar]
- Derbyshire ER, Fernhoff NB, Deng S, Marletta MA. Nucleotide regulation of soluble guanylate cyclase substrate specificity. Biochemistry. 2009;48:7519–7524. doi: 10.1021/bi900696x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eklund EA, Kakar R. Recruitment of CREB-binding protein by PU.1, IFN-regulatory factor-1, and the IFN consensus sequence-binding protein is necessary for IFN-gamma-induced p67phox and gp91phox expression. J Immunol. 1999;163:6095–6105. [PubMed] [Google Scholar]
- Eklund EA, Jalava A, Kakar R. Tyrosine phosphorylation of HoxA10 decreases DNA binding and transcriptional repression during interferon gamma -induced differentiation of myeloid leukemia cell lines. J Biol Chem. 2000;275:20117–20126. doi: 10.1074/jbc.M907915199. [DOI] [PubMed] [Google Scholar]
- El-Benna J, Dang PM, Gougerot-Pocidalo MA, Elbim C. Phagocyte NADPH oxidase: a multicomponent enzyme essential for host defenses. Arch Immunol Ther Exp (Warsz) 2005;53:199–206. [PubMed] [Google Scholar]
- Erceg-Hurn DM, Mirosevich VM. Modern robust statistical methods: an easy way to maximize the accuracy and power of your research. Am Psychol. 2008;63:591–601. doi: 10.1037/0003-066X.63.7.591. [DOI] [PubMed] [Google Scholar]
- Evgenov OV, Kohane DS, Bloch KD, Stasch JP, Volpato GP, Bellas E, et al. Inhaled agonists of soluble guanylate cyclase induce selective pulmonary vasodilatation. Am J Respir Crit Care Med. 2007;176:1138–1145. doi: 10.1164/rccm.200707-1121OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezekowitz RA, Dinauer MC, Jaffe HS, Orkin SH, Newburger PE. Partial correction of the phagocyte defect in patients with X-linked chronic granulomatous disease by subcutaneous interferon gamma. N Engl J Med. 1988;319:146–151. doi: 10.1056/NEJM198807213190305. [DOI] [PubMed] [Google Scholar]
- Flannagan RS, Cosío G, Grinstein S. Antimicrobial mechanisms of phagocytes and bacterial evasion strategies. Nat Rev Microbiol. 2009;7:355–366. doi: 10.1038/nrmicro2128. [DOI] [PubMed] [Google Scholar]
- Geha RS, Notarangelo LD, Casanova JL, Chapel H, Conley ME, Fischer A, et al. Primary immunodeficiency diseases: An update from the International Union of Immunological Societies Primary Immunodeficiency Diseases Classification Committee. J Allergy Clin Immunol. 2007;120:776–794. doi: 10.1016/j.jaci.2007.08.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geissmann F, Auffray C, Palframan R, Wirrig C, Ciocca A, Campisi L, et al. Blood monocytes: distinct subsets, how they relate to dendritic cells, and their possible roles in the regulation of T-cell responses. Immunol Cell Biol. 2008;86:398–408. doi: 10.1038/icb.2008.19. [DOI] [PubMed] [Google Scholar]
- Hatanaka E, Levada-Pires AC, Pithon-Curi TC, Curi R. Systematic study on ROS production induced by oleic, linoleic, and g-linolenic acids in human and rat neutrophils. Free Radic Biol Med. 2006;41:1124–1132. doi: 10.1016/j.freeradbiomed.2006.06.014. [DOI] [PubMed] [Google Scholar]
- Hawkins SM, Kohwi-Shigematsu T, Skalnik DG. The matrix attachment region-binding protein SATB1 interacts with multiple elements within the gp91phox promoter and is down-regulated during myeloid differentiation. J Biol Chem. 2001;276:44472–44480. doi: 10.1074/jbc.M104193200. [DOI] [PubMed] [Google Scholar]
- Huang YJ, Chen IS, Tseng CP, Day YJ, Lin YC, Liao CH. 2R,3R)-2-(3′,4′-dihydroxybenzyl)-3-(3″,4″-dimethoxybenzyl)butyrolactone suppresses fMLP-induced superoxide production by inhibiting fMLP-receptor binding in human neutrophils. Biochem Pharmacol. 2008;75:688–697. doi: 10.1016/j.bcp.2007.10.002. [DOI] [PubMed] [Google Scholar]
- Igbal J, Zaidi M. TNF-induced MAP kinase activation oscillates in time. Biochem Biophys Res Commun. 2008;371:906–911. doi: 10.1016/j.bbrc.2008.03.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ismail N, Olano JP, Feng HM, Walker DH. Current status of immune mechanisms of killing of intracellular microorganisms. FEMS Microbiol Lett. 2002;207:111–120. doi: 10.1111/j.1574-6968.2002.tb11038.x. [DOI] [PubMed] [Google Scholar]
- Jongstra-Bilen J, Harrison R, Grinstein S. Fcgamma-receptors induce Mac-1 (CD11b/CD18) mobilization and accumulation in the phagocytic cup for optimal phagocytosis. J Biol Chem. 2003;278:45720–45729. doi: 10.1074/jbc.M303704200. [DOI] [PubMed] [Google Scholar]
- Khare S, Tien XY, Wilson D, Wali RK, Bissonnette BM, Scaglione-Sewell B, et al. The role of protein kinase-C alpha in the activation of particulate guanylate cyclase by 1 alpha,25-dihydroxyvitamin D3 in CaCo-2 cells. Endocrinology. 1994;135:277–283. doi: 10.1210/endo.135.1.7912183. [DOI] [PubMed] [Google Scholar]
- Kobayashi S, Murayama S, Takanashi S, Takahashi K, Miyatsuka S, Fujita T, et al. Clinical features and prognoses of 23 patients with chronic granulomatous disease followed for 21 years by a single hospital in Japan. Eur J Pediatr. 2008;167:1389–1394. doi: 10.1007/s00431-008-0680-7. [DOI] [PubMed] [Google Scholar]
- Kobialka M, Gorczyca WA. The effect of cGMP on the function of immune system cells. Postepy Hig Med Dosw. 2001;55:195–210. [PubMed] [Google Scholar]
- Koglin M, Stasch JP, Behrends S. BAY 41-2272 activates two isoforms of nitric oxide- sensitive guanylyl cyclase. Biochem Biophys Res Commun. 2002;292:1057–1062. doi: 10.1006/bbrc.2002.6764. [DOI] [PubMed] [Google Scholar]
- Luengo-Blanco M, Prando C, Bustamante J, Aragão-Filho WC, Pereira PV, Rehder J, et al. Essential role of nuclear factor-kappaB for NADPH oxidase activity in normal and anhidrotic ectodermal dysplasia leukocytes. Blood. 2008;112:1453–1460. doi: 10.1182/blood-2007-07-099267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo Y, Cook E, Fries BC, Casadevall A. Phagocytic efficacy of macrophage-like cells as a function of cell cycle and Fcgamma receptors (FcgammaR) and complement receptor (CR)3 expression. Clin Exp Immunol. 2006;145:380–387. doi: 10.1111/j.1365-2249.2006.03132.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray HW, Tsai CW, Liu J, Ma X. Responses to Leishmania donovani in mice deficient in interleukin-12 (IL-12), IL-12/IL-23, or IL-18. Infect Immun. 2006;74:4370–4374. doi: 10.1128/IAI.00422-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nauseef WM. How human neutrophils kill and degrade microbes: an integrated view. Immunol Rev. 2007;219:88–102. doi: 10.1111/j.1600-065X.2007.00550.x. [DOI] [PubMed] [Google Scholar]
- Newburger PE, Ezekowitz RA, Whitney C, Wright J, Orkin SH. Induction of phagocyte cytochrome b heavy chain gene expression by interferon γ. Proc Natl Acad Sci U S A. 1988;85:5215–5219. doi: 10.1073/pnas.85.14.5215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newburger PE, Dai Q, Whitney C. In vitro regulation of human phagocyte cytochrome b heavy and light chain gene expression by bacterial lipopolysaccharide and recombinant human cytokines. J Biol Chem. 1991;266:16171–16177. [PubMed] [Google Scholar]
- Newburger PE, Skalnik DG, Hopkins PJ, Eklund EA, Curnutte JT. Mutations in the promoter region of the gene for gp91-phox in X-linked chronic granulomatous disease with decreased expression of cytochrome b558. J Clin Invest. 1994;94:1205–1211. doi: 10.1172/JCI117437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira-Junior EB, Thomazzi SM, Rehder J, Antunes E, Codino-Neto A. Effects of BAY 41-2272, an activator of nitric oxide-independent site of soluble guanylate cyclase, on human NADPH oxidase system from THP-1 cells. Eur J Pharmacol. 2007;567:43–49. doi: 10.1016/j.ejphar.2007.04.018. [DOI] [PubMed] [Google Scholar]
- Pacher P, Beckman JS, Liaudet L. Nitric oxide and peroxynitrite in health and disease. Physiol Rev. 2007;87:315–424. doi: 10.1152/physrev.00029.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitzurra L, Cherniak R, Giammarioli M, Perito S, Bistoni F, Vecchiarelli A. Early induction of interleukin-12 by human monocytes exposed to Cryptococcus neoformans mannoproteins. Infect Immun. 2000;68:558–563. doi: 10.1128/iai.68.2.558-563.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinn MT, Ammons MCB, DeLeo FR. The expanding role of NADPH oxidase in health and disease: no longer just agents of death and destruction. Clin Sci (Lond) 2006;111:1–20. doi: 10.1042/CS20060059. [DOI] [PubMed] [Google Scholar]
- Rhein LM, Perkins M, Gerard NP, Gerard C. FcgammaRIII is protective against Pseudomonas aeruginosa pneumonia. Am J Respir Cell Mol Biol. 2008;38:401–406. doi: 10.1165/rcmb.2007-0309OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samarghitean C, Ortutay C, Vihinen M. Systematic classification of primary immunodeficiencies based on clinical, pathological, and laboratory parameters. J Immunol. 2009;183:7569–7575. doi: 10.4049/jimmunol.0901837. [DOI] [PubMed] [Google Scholar]
- Skalnik DG, Neufeld EJ. Sequence-specific binding of HMG-I(Y) to the proximal promoter of the gp91-phox gene. Biochem Biophys Res Commun. 1992;187:563–569. doi: 10.1016/0006-291x(92)91231-e. [DOI] [PubMed] [Google Scholar]
- Stasch JP, Becker EM, Alonso-Alija C, Apeler H, Dembowsky K, Feurer A, et al. NO-independent regulatory site on soluble guanylate cyclase. Nature. 2001;410:212–215. doi: 10.1038/35065611. [DOI] [PubMed] [Google Scholar]
- Suzuki S, Kumatori A, Haagen IA. PU.1 as an essential activator for the expression of gp91(phox) gene in human peripheral neutrophils, monocytes, and B lymphocytes. Proc Natl Acad Sci U S A. 1998;95:6085–6090. doi: 10.1073/pnas.95.11.6085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomazzi SM, Moreira J, De Nucci G, Antunes E. Inhibitory effects on human eosinophil chemotaxis in vitro by BAY 41-2272, an activator of nitric oxide-independent site of soluble guanylate cyclase. Biochem Pharmacol. 2005;69:875–882. doi: 10.1016/j.bcp.2004.12.007. [DOI] [PubMed] [Google Scholar]
- Tsuchiya S, Yamabe M, Yamaguchi Y, Kobayashi Y, Konno T, Tada K. Establishment and characterization of a human acute monocytic leukemia cell line (THP-1) Int J Cancer. 1980;26:171–176. doi: 10.1002/ijc.2910260208. [DOI] [PubMed] [Google Scholar]
- Ueyama T, Tatsuno T, Kawasaki T, Tsujibe S, Shirai Y, Sumimoto H, et al. A regulated adaptor function of p40phox: distinct p67phox membrane targeting by p40phox and p47phox. Mol Biol Cell. 2007;18:441–454. doi: 10.1091/mbc.E06-08-0731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamauchi A, Yu L, Potgens AJ, Kuribayashi F, Nunoi H, Kanegasaki S, et al. Location of the epitope for 7D5, a monoclonal antibody raised against human flavocytochrome b558, to the extracellular peptide portion of primate gp91phox. Microbiol Immunol. 2001;45:249–257. doi: 10.1111/j.1348-0421.2001.tb02614.x. [DOI] [PubMed] [Google Scholar]


