Abstract
Background
Recent studies indicate that short subclinical episodes of herpes simplex virus type 2 (HSV-2) are the predominant form of skin and mucosal viral shedding. We evaluated whether standard or high-dose antiviral therapy reduced the frequency of such shedding.
Methods
To determine whether short episodes of genital HSV shedding are suppressed on standard dose (SD) and high-dose (HD) antiviral therapy, HSV-2 seropositive, HIV seronegative persons in Seattle, WA were enrolled into three separate but complementary randomized, open-label, cross-over studies comparing 1) no medication to aciclovir 400 mg twice daily (SD-ACV), 2) valaciclovir 500 mg daily (SD-VAL) to aciclovir 800 mg three times daily (TID) (HD-ACV), and 3) SD-VAL to HD-VAL (1 gm TID). Study arms lasted 4–7 weeks, separated by one week wash-out. Participants obtained genital swabs four times daily for quantitative HSV DNA PCR. The primary endpoint was within-person comparison of shedding rate on each study arm.
Results
Of 113 participants randomized, 90 were eligible for analysis of the primary endpoint. Participants collected 23,605 swabs; of these 1272 (5·4%) had HSV detected. HSV shedding was significantly higher during the no medication arm (18·1% of swabs) compared with SD-ACV (1.2% of swabs, IRR=0·05, 95% CI=0·03–0·08). Breakthrough reactivations occurred on all doses (SD-ACV 1·2%, SD-VAL 5·2%, HD-ACV 4·2%, and HD-VAL 3·3% of swabs). HD-VAL was associated with less shedding compared with SD-VAL (IRR=0·54, 95% CI=0·44–0·66), likely due to more rapid clearance of mucosal HSV (4·7 logs/6 hours on HD-VAL vs. 4·4 logs/6 hours on SD-VAL, (p=0·02)). However, the annualized breakthrough episodes was similar on SD-VAL (22·6) and HD-ACV (20·2, p=0·54) and SD-VAL (14.9) and HD-VAL (16·5, p=0·34). Regardless of dose, breakthrough episodes were short (median 7–10 hours) and 80% were subclinical. Studies were not designed to make inter-trial comparisons between antiviral doses. Except for increased incidence of headaches on HD-VAL, all regimens were well-tolerated.
Conclusions
Short bursts of subclinical genital HSV reactivation are frequent, even during high-dose antiherpes therapy, and likely account for continued transmission of HSV-2 during suppressive antiviral therapy. More potent antiviral therapy is needed to abolish HSV-2 transmission
Introduction
Herpes simplex virus type 2 (HSV-2) infection is a global epidemic,1 and significantly increases the risk of HIV-1 acquisition.2 Despite the increasing use of suppressive antiviral therapy over the past 2 decades, little alteration in the prevalence of, or complications from, HSV-2 infection has occurred.3, 4 Numerous studies have shown the clinical impact of daily antiviral therapy in reducing genital lesions5, 6 and suppressing detection of HSV-2 on genital mucosal surfaces (“shedding”).7, 8 However, treatment with valaciclovir 500 mg daily results in only 48% reduction in the risk of sexual transmission.9 Moreover, suppressive aciclovir is not effective in reducing the risk of HIV transmission or acquisition in HSV-2 seropositive persons.10–12 The discrepancy between potent suppression of clinical symptoms and failure of antivirals to fully prevent HSV-2 transmission is not well understood.
Recent studies using intensive genital secretion collection have shown that HSV-2 episodes are 3 times more frequent than previously appreciated.13 Nearly 50% of reactivations last <12 hours, with presumed rapid clearance by the mucosal immune system.13, 14 Mathematical models suggest that these short bursts of shedding are even more frequent than current sampling methods detect, comprising 70–85% of all reactivations.15 Local host immune responses appear to control the severity, duration, and amount of virus detected during an outbreak.16 Trials using once daily genital sampling indicate that antivirals are effective in reducing high-copy number, long episodes of HSV reactivation.7 However, whether antivirals suppress short reactivation events is unknown. This is clinically important, as failure to do so could account for the reduced effectiveness of antiviral drugs in clinical studies of HSV-2 transmission.
We conducted a series of complementary clinical studies to evaluate if either standard or high doses of aciclovir and valaciclovir affect the frequency of short subclinical genital bursts of HSV reactivation among HSV-2 seropositive persons.
Methods
Study Design
Participants were randomized to study medication for a specified period, followed by a one week washout period, and then crossed over to the other study medication for the remaining study period (Figure 1); all trials were open label. Trial 1 was designed to determine whether antiviral therapy suppressed short subclinical episodes of HSV shedding, compared no medication to standard dose aciclovir (SD-ACV, ACV 400 mg BID). Participants were followed for 4 weeks on each study arm. Trials 2 and 3 evaluated whether increasing doses of antivirals or alterations in dosing schedule could provide more potent suppression of short shedding episodes. Participants on the second trial (SD-VAL (VAL 500 mg daily) vs. high dose (HD) ACV (HD-ACV) (ACV 800 mg TID)) were followed for 7 weeks on each arm, and participants on the third trial (SD-VAL (VAL 500 mg daily) vs. HD-VAL (VAL 1 mg TID) were followed for 5 weeks on each arm. The “high dose” dosages were selected to provide a 2–3 fold (HD-ACV)6, 17 and an 8-fold (HD-VAL)17 increase in the mean daily plasma ACV area under the curve (AUC) concentration, relative to SD-VAL. The HD-VAL dose was selected because there are extensive clinical data about the safety of this dose, which is also used for treatment of herpes zoster 17 The longer duration of study arms in Trials 2 and 3 was based on the anticipated lower shedding rates during antiviral therapy, and the need to have sufficient power to detect smaller differences in shedding rates between the two arms.18
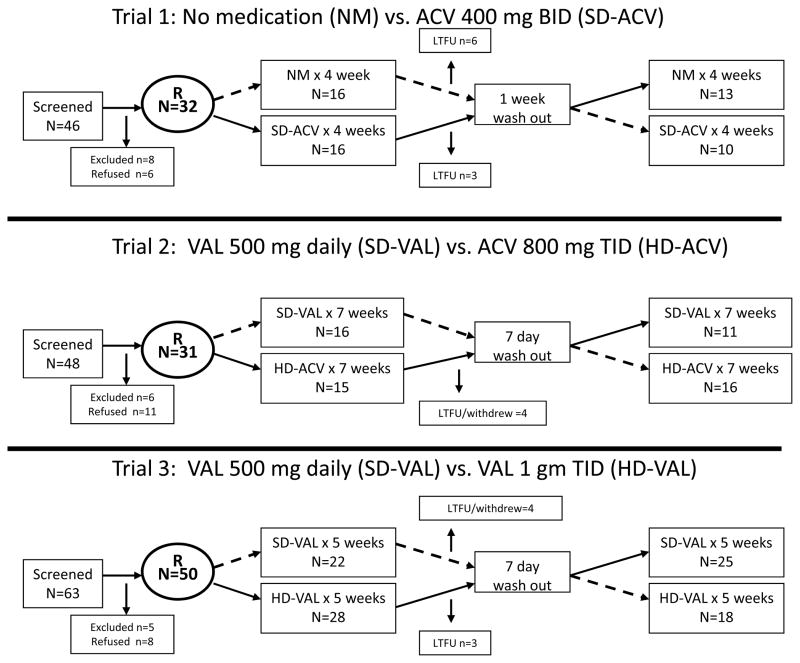
Figure 1.
Enrollment, randomization and follow-up of participants. R=Randomized, NM=no medication, SD-ACV=aciclovir 400 mg twice daily, SD-VAL=valaciclovir 500 mg daily, HD-ACV=aciclovir 800 mg three times daily, HD-VAL=valaciclovir 1 gm three times daily, LTFU=Lost to follow up
Participants self-collected a single genital swab four times daily, approximately every 4–6 hours while awake, during each study arm19. Women swabbed the vaginal vault, the entire labia majora and labia minora, the perineum and perianal area. Men swabbed the penile shaft, scrotum, perineum and perianal area. Participants recorded genital symptoms and lesions daily.20 Participants were seen biweekly for swab collection, medication administration, assessment of side effects, and genital examination, if symptomatic. Randomization was performed using a computerized random number generator and was stratified by gender. The randomization was performed at a ratio of 1:1 for all drug arms.
Study Population
HSV-2 seropositive, HIV seronegative healthy adults, ≥18 years of age were recruited at the University of Washington (UW) Virology Research Clinic between November 2006 and July 2010. In Trial 1, eligible participants had serologic evidence of HSV-2; both symptomatic and asymptomatic persons were recruited. In Trials 2 and 3, participants were required to have at least 4 clinical HSV-2 recurrences in the past year or laboratory documented primary genital HSV-2 within the past 6 months. Participants with prior adverse reaction to aciclovir, pregnant women, and those unable to comply with study procedures were excluded. Recruitment was through newspaper ads, fliers, and word of mouth. These studies were approved by the UW Human Subjects Division and all participants provided written informed consent.
Laboratory Methods
HSV serostatus was determined by Western Blot.21 Genital swabs were placed in 1mL of 1X PCR buffer and stored at 4°C. DNA was extracted from genital swabs, and HSV was quantitated by real-time polymerase chain reaction (PCR), using HSV specific primers to glycoprotein B.22 Samples containing ≥150 copies of HSV DNA per ml of PCR buffer were considered positive.23 Laboratory personnel were blinded to clinical data. Viral thymidine kinase (TK) sequencing was performed on swabs from one participant with extraordinarily high HSV shedding rates.24
Study Endpoints
The primary outcome was frequency of HSV detection, measured by the number of swabs with HSV detected divided by the number of swabs collected during each treatment arm per participant, for each study. Secondary outcomes by treatment arm included number of HSV shedding episodes, duration of HSV shedding episodes, and maximum quantity of HSV DNA detected per shedding episode. Episodes were defined as any number of swabs with HSV detected immediately preceded by two negative swabs and followed by two negative swabs. Episode duration was extrapolated by estimating start and stop times at the midpoint between the last HSV-positive and first HSV-negative swabs. Participants who did not collect at least one swab during each study arm were excluded from the analyses.
Safety
During all trials, adverse events (AEs) were assessed at each study visit. Biweekly white blood count, renal and liver function tests were monitored among persons enrolled in Trial 3. AEs were graded using NCI common terminology criteria (version 3.0).
Statistical Methods
Estimates of shedding frequency were based upon natural history studies using four times/day sampling13, 14, and within-person correlation was estimated to be 0.27. Using a sample size calculation for repeated-measures binary data25 and assuming a genital shedding rate of 10% on no medication, Trial 1 had 80% power to detect a 35% reduction in genital shedding rates on SD-ACV. Assuming a genital shedding rate of 3% on SD-VAL,7 Trials 2 and 3 had 80% power to detect a 50% reduction in HSV genital shedding rates on HD-ACV or HD-VAL as compared with SD-VAL; a 20% loss to follow up was assumed for all trials. The first day in each treatment period was excluded from the analysis. Using Poisson regression, HSV genital shedding rates and episodes rates were compared by treatment arm, including a scale parameter for overdispersion and the log number of swabs as the offset. Sequence and period effects were examined within the model. Generalized estimating equations were used to compare shedding rates on no medication and drug arms in Trials 2 and 3, excluding 3 participants who participated in both Trial 1 and Trial 3. Linear mixed models (LMM) were used to compare episode rate, episode duration, maximum HSV log10 copy number during episodes, and HSV log10 copy number among positive swabs by treatment arm, and to evaluate the association between episode duration and maximum HSV log10 copy number. Episode kinetics were calculated by computing the slope of a linear regression line from the beginning to peak (expansion) or peak to end (clearance) of an episode.26 The Wilcoxon signed rank test for matched pairs was used to compare drug adherence rates by treatment as measured by pill counts. Correlation between adherence and genital shedding rate was measured using the Spearman correlation coefficient. The annualized episode rate was calculated by enumerating the number of episodes and dividing by person-years of follow-up per treatment arm. Two-sided p-values<0.05 were considered statistically significant. Data were analyzed using Stata 10·1.
Role of the funding source: Valaciclovir was provided at no cost for Trial 3 by GlaxoSmithKline. The study design, conduct and analyses were performed solely by the academic investigators associated with the trial. The funding source had no role in analyzing or interpreting the data or writing the manuscript.
Results
Demographics
Of 157 participants screened, 90 were enrolled and collected samples on both study arms; (Figure 1); 3 participants enrolled in both Trial 1 and Trial 3. The median age was 43 years (IQR 30–51). Forty-nine (54%) participants were women and 76 (84%) were Caucasian. Fifty (56%) participants were HSV-2 seropositive only; 39 (43%) were HSV-1 and HSV-2 seropositive. The median duration of HSV-2 infection was 7·6 years (IQR 1·8–19·5). The demographics of the study population were similar in each of the trials (supplemental Table 1).
Genital HSV shedding rates during no medication, standard dose, and high dose therapy
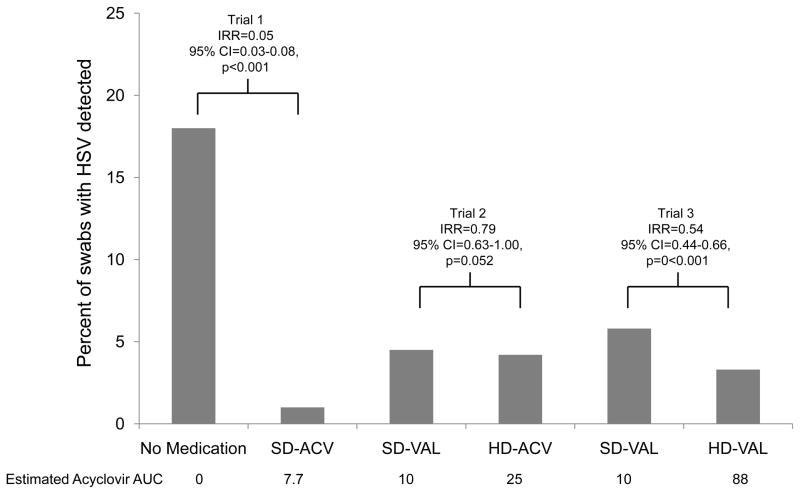
Ninety participants collected 23,605 (86%) of a possible 27,319 swabs. Swabs were collected a median of 5·7 hours apart (IQR=4·5–7·2 hours, range 2·0–13·5 hours). Twenty-three participants collected 2123 swabs while receiving no medication, and HSV was found in 384 (18·1%) of these swabs (Table 1A, Figure 2). While all doses of aciclovir reduced the frequency of detection of HSV as compared with no medication (all p-values ≤0·003), breakthrough reactivation occurred at all drug doses (1·2–5·8% of swabs) (Figure 2). Twenty-five (1·2%) of 2129 swabs had detectable HSV during SD-ACV (IRR=0·05, 95% CI=0·03–0·08, compared with no medication). There was a trend toward a decreased proportion of swabs with HSV detected collected from 27 participants during HD-ACV as compared with SD-VAL (198 (4·2%) of 4709 swabs vs. 209 (4·5%) of 4663 swabs, IRR=0·79, 95% CI=0·63–1·00). A more substantial dose response in rate of HSV shedding was noted during HD-VAL; 164 (3·3%) of 4973 swabs collected from 43 participants had HSV detected as compared with SD-VAL (292 (5·8%) of 5008 swabs) (IRR=0·54, 95% CI=0·44–0·66). However, HSV was still detected from 3·3% of swabs on HD-VAL. Sequence effects were not found for any trial (data not shown). As period effects were present for all three trials, each model was adjusted for the period effect.
Table 1A.
Genital HSV shedding rates by treatment arm
| Trial 1 | Trial 2 | Trial 3 | ||||
|---|---|---|---|---|---|---|
| No med N=23 |
SD-ACV N=23 |
SD-VAL N=27 |
HD-ACV N=27 |
SD-VAL N=43 |
HD-VAL N=43 |
|
| Overall | ||||||
| Swabs with HSV detected | 384/2123 (18.1) | 25/2129 (1.2) | 209/4663 (4.5) | 198/4709 (4.2) | 292/5008 (5.8) | 164/4973 (3.3) |
| Days with HSV detected | 143/599 (23.9) | 21/604 (3.5) | 113/1251 (9.0) | 110/1274 (8.6) | 120/1378 (8.8) | 94/1395 (6.7) |
| Per-Participant* | Median (IQR) | |||||
| Swabs collected | 96 (88–107) | 96 (85–106) | 180 (155–189) | 178 (168 –193) | 121 (110–130) | 117 (105–131) |
| Days on study | 27 (26–28) | 28 (26–29) | 48 (48–49) | 49 (48 – 50) | 34 (34–35) | 34 (34–35) |
| Percent of swabs with HSV detected | 11 (0–32) | 0 (0–1) | 2 (1–4) | 1 (0–3) | 3 (0–9) | 1 (0 – 4) |
| Percent of days with HSV detected | 21 (0–38) | 0 (0–4) | 4 (2–13) | 2 (0–6) | 5 I0–15) | 3 (0–10) |
The number of participants is as noted in Figure 1, excluding participants who were not included in the analysis due to loss to follow-up. Numbers are n(%) unless otherwise indicated
Figure 2.
Rate of HSV detection in genital swabs by antiviral dose. IRR=Incidence risk ratio, CI=confidence interval, BID=twice daily, QD=once daily, TID=three times daily AUC=area under the curve (μg.h/ml).
Characterization of HSV Shedding Episodes
Three hundred forty-four genital HSV shedding episodes occurred during the course of the 3 trials. Of 49 HSV shedding episodes during the no medication arm, 32 (65%) were not associated with genital lesions or symptoms (“subclinical”) (Table 1B). Most shedding episodes were subclinical during SD-ACV (16 (94%) of 17), SD-VAL (pooled 111 (80%) of 139 episodes), HD-ACV (68 (92%) of 74 episodes) and HD-VAL, (48 (74%) of 65 episodes). The episode rate was significantly lower on SD-ACV as compared with no medication (10·0 vs. 28·7 episodes/year, p=0·001). There was no difference in the episode rate between SD-VAL and HD-ACV (22·6 vs. 20·2 episodes/year, p=0·54) or SD-VAL and HD-VAL (14·9 vs. 16·5 episodes/year, p=0·34).
Table 1B.
Characteristics of genital HSV shedding episodes by treatment arm
| Trial 1 | Trial 2 | Trial 3 | ||||
|---|---|---|---|---|---|---|
| No med | SD-ACV | SD-VAL | HD-ACV | SD-VAL | HD-VAL | |
| Number of episodes | 49 | 17 | 81 | 74 | 58 | 65 |
| Median episodes/person, IQR | 1 (0–4) | 0 (0–1) | 2 (1–5) | 1 (0–3) | 1 (0–2) | 1 (0–2) |
| Person years of follow-up | 1.71 | 1.70 | 3.59 | 3.66 | 3.89 | 3.93 |
| Number of subclinical episodes n(%) | 32 (65) | 16 (94) | 74 (91) | 68 (92) | 37 (64)* | 48 (74)++ |
| Annualized episode rate(episodes/person-years) | 28.7 | 10.0 | 22.6 | 20.2 | 14.9 | 16.5 |
| Number of episodes of known duration | 36 | 15 | 69 | 57 | 42 | 41 |
| Median episode duration, IQR(hours) | 13 (7–73) | 7 (6–13) | 8 (6–18) | 8 (6– 19) | 10 (7–42) | 7 (6–9) |
| Maximum log10 copies/mL, IQR | 3.3 (2.5–5.7) | 2.9 (2.4–3.4) | 2.7 (2.4–3.1) | 2.8 (2.5 –3.2) | 3.0 (2.4–4.5) | 2.5 (2.3–2.7) |
IQR=Interquartile range
missing for 3 episodes
missing for 11 episodes
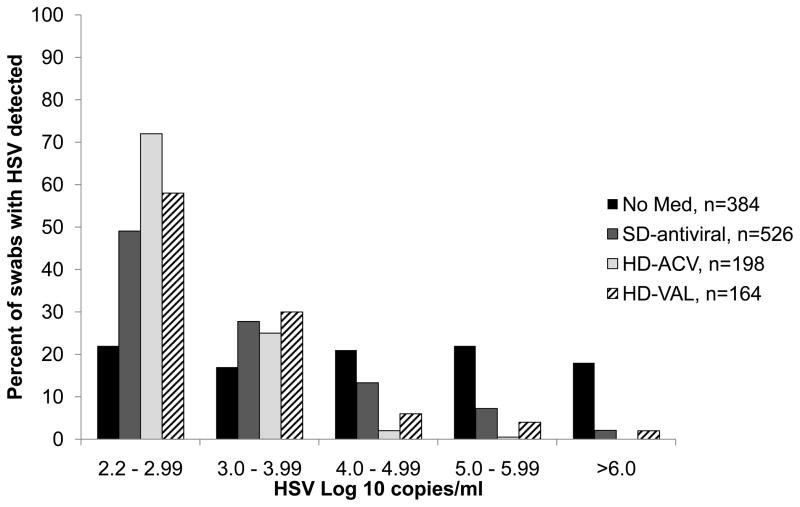
The median maximum quantity of HSV detected per episode was significantly higher during no medication (3·3 log10 copy number/ml) compared with SD-ACV (2·9 log10 copy number/ml), (p=0·02). The quantity of HSV detected during episodes on SD-VAL and HD-ACV was similar (2·7 vs. 2·8 log10 copies/ml, p=0·66), and was significantly lower on HD-VAL compared with SD-VAL (2·5 vs. 3·0 log10 copies/ml, p=0·001). The distribution of quantity of HSV detected on each antiviral dose is shown in Figure 3.
Figure 3.
Distribution of HSV quantity (log10 copies/ml) in swabs with HSV DNA detected by PCR, during no medication (black), aciclovir 400 mg twice daily and valaciclovir 500 mg daily (SD-antiviral, gray), aciclovir 800 mg three times daily (HD-ACV) (light gray) and valaciclovir 1 gm three times daily (HD-VAL) (cross-hatched). N=number of swabs
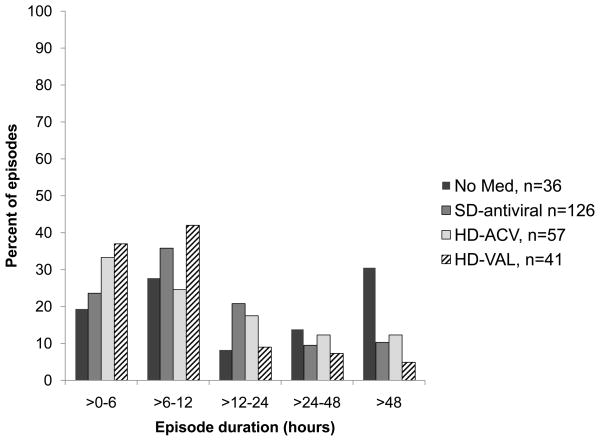
Of the 344 HSV shedding episodes, 84 episodes were of unknown duration; 44 (12.8%) were due to observing the episode at the beginning or end of the treatment arm, and 40 (11.6%) were due to missing swabs. Among the remaining 260 HSV shedding episodes, the episode duration was calculated. The median episode duration was longer on no medication as compared with SD-ACV (13 hours vs. 7 hours, p=0·01). The median episode duration was 8 hours on both SD-VAL and HD-ACV (p=0·23), and was significantly shorter on HD-VAL compared with SD-VAL (7 hours vs. 10 hours, p=0·03) (Table 1B). The median viral expansion rate was similar on SD-VAL (4·5–4·6 logs per 6 hours, range 0·2–11·5) HD-ACV (4·8 logs per 6 hours, range 0–7·9, p=0·56) and HD-VAL (4·7 log per 6 hours, range 0·1–9·3, p=0·82). However, the rate of viral clearance was faster on HD-VAL (4·7 log/6 hours, range 0·6–9·3) than on SD-VAL (4·4 log, range 0·0–8·9, p=0·016). Most shedding episodes on antiviral therapy were short regardless of dose: 75 (60%) of 126 episodes on SD-antiviral therapy, 33 (58%) of 57 episodes on HD-ACV, and 32 (78%) of 41 episodes on HD-VAL were less than 12 hours duration (Figure 4). An increased episode duration was correlated with an increased maximum log10 copy number during the episode (0·15 log higher for each 6 hours increasing duration, p<0·001).
Figure 4.
Distribution of HSV episode duration in the 260 episodes of known duration, during no medication (black), aciclovir 400 mg twice daily and valaciclovir 500 mg daily (SD-antiviral, gray), aciclovir 800 mg three times daily (HD-ACV) (light gray) and valaciclovir 1 gm three times daily (HD-VAL) (cross-hatched). N=number of episodes. Supplemental Figure 1. Quantity of HSV detected per swab in participant in Trial 2 which HSV was detected on 84 (49%) of 172 swabs during arm 1 (HD-ACV) and 70 (37%) of 189 swabs during arm 2 (SD-VAL).
Study, Drug and Swab Adherence
Demographic characteristics and shedding rates of participants who were lost to follow up and completed the study did not differ (data not shown). Between 78% and 96% of participants in all studies reported >85% adherence to study medication, with a median adherence rate of 98–100% for each study arm. No correlation between drug adherence and shedding was observed (data not shown). There were a total of 3,748 missing swabs for participants included in the final analysis, comprising 1,630 intervals when at least one swab was not collected.. Of these intervals, 1,126 (69·3%) had a swab missing at a single time point, 410 (25·2%) were missing a swab at 2–5 consecutive time points, and 91 (5·6%) were missing swabs from >5 consecutive time points. Each study arm and trial had a similar proportion of missing swabs (11·4%, 14·3% and 14·8% from Trial 1, 2, and 3 respectively).
Adverse Events and Antiviral Resistance
During HD-VAL therapy, 13 (30%) of 43 participants complained of headache on at least one day (compared with none on SD-VAL). Nausea (7%), and myalgias (5%) were also reported during HD-VAL. Two (5%) participants developed grade II neutropenia on HD-VAL and SD-VAL. No additional abnormalities in complete blood count, liver function or renal function, or AEs related to study medication were noted. For one participant with high rates of shedding on both study arms in Trial 2 (supp. Figure 1), we evaluated whether ACV resistance could be a factor in breakthrough recurrences. We sequenced the HSV TK gene from a representative swab sample, and did not detect any mutation known to confer aciclovir resistance27 (Genbank accession number HM446467).
Discussion
In a series of studies using incrementally higher doses of antivirals for HSV, we observed persistent, frequent, short HSV shedding episodes on the genital mucosa, providing a likely explanation for the inability of daily suppressive therapy to completely abrogate HSV-2 transmission. Although daily antiviral therapy nearly eliminated clinical recurrences and reduced the frequency of high copy number shedding episodes, we found that short episodes of genital HSV shedding occur frequently on antiviral therapy, even with regimens that increase the aciclovir AUC 2–3 fold (HD-ACV) or 8-fold (HD-VAL) over standard suppressive doses of aciclovir or valaciclovir. These breakthrough episodes are typically subclinical, last a median of 7–10 hours, and occur at a similar rate regardless of antiviral dose.
Studies measuring clinical effectiveness of aciclovir and valaciclovir in immunocompetent persons have shown relatively similar efficacy, except in persons with very frequent recurrences.6 Initial clinical trials of aciclovir and valaciclovir showed high efficacy in preventing genital herpes recurrences.5, 6 Trials that evaluated suppression of subclinical shedding also demonstrated a pronounced effect, although efficacy was greater when measured by viral culture (~95%)8 than PCR (70–80%).7 Because of the high success in reducing clinical and subclinical shedding, valaciclovir 500 mg daily was selected as the dose to test for reduction in sexual transmission of HSV-2. Interestingly, this dose only reduced the sexual transmission of HSV-2 by 48% among HSV-2 discordant couples, despite a 73% reduction in subclinical shedding in the same population.9 In the trials presented here, participants experienced approximately 1–2 shedding episodes per month on both standard and high dose antiviral therapy. Despite the short duration of these episodes, a substantial proportion, over 20% on standard dose antiviral therapy, had >104 HSV copies/ml. These high copy number shedding episodes were not eliminated with HD-VAL and HSV was still detected from 3% of swabs. Our data indicate that anti-HSV therapy, while clinically effective, does not substantially alter the underlying pathobiology of frequent, subclinical HSV-2 reactivation. That we could not eliminate or even alter the frequency of shedding episodes using HD-VAL suggests that the maximal benefit of shedding reduction has likely been reached for currently available antivirals.
Recent modeling suggests observed shedding patterns can be replicated through simulated release of frequent, small amounts of HSV from neurons. 15 The short episodes found in this trial may reflect the earliest HSV replication events during or after neuronal HSV reactivation. Possible explanations for this finding may include inadequate aciclovir concentrations in neurons due to the blood brain barrier 28 or less efficient conversion to aciclovir tri-phosphate in neurons as compared to epithelial cells. 29 Our finding that the kinetics of viral clearance, but not expansion, are faster on HD-VAL support the hypothesis that these antivirals do not suppress the release of virions into the genital tract. Our trials had several strengths, including a rigorous cross-over design which allowed for intra-person comparisons, and motivated participants who complied with intensive swabbing schedules. The high medication adherence rate and the inclusion of a three times daily drug regimen, as well as the similarity in episode frequency and duration across trials suggest that our findings are not influenced by the absorption kinetics of these antivirals, but by their underlying mechanism of action. In addition, we utilized a validated highly sensitive and specific PCR assay,23 minimizing the likelihood that detection of HSV was due to false positives. While the threshold copy number necessary or sufficient for transmission has not been established, HSV transmission from culture negative, PCR positive secretions has been documented.30 In addition, the linear relationship between log copy numbers and probability of a viral replication in tissue media suggests that a threshold for infectivity is unlikely to be defined.31
An ideal study design for these trials would have been multiple dose cross-overs in each participant.7 However, the intensity of collecting the swabs on a frequent basis made this impractical, as such a design would have necessitated intensive swabbing for nearly 150 days for each participant. Our studies were not powered to compare shedding rates on different antiviral doses between trials, as the cross-over design relied on comparison of the shedding rates within each person. The similarity in shedding rates on all doses of medication (1·2%–5·2% of swabs) is remarkable in that it demonstrates the reproducibility of results. However, given the differences in study design and population, we caution against direct comparisons between the antiviral doses of the three trials. We recruited people with clinically recognized disease who would be expected to have higher shedding rates to the two trials using HD-ACV and HD-VAL, to better detect a difference in shedding rates between the study arms. Despite this attempt to demonstrate such a difference, even very high doses of valaciclovir were associated with breakthrough genital HSV shedding. We conducted there trials at a single site, and enrolled mostly white, healthy, sexually active adults. The generalizability of these data to other geographic regions or racial/ethnic groups is unknown. In addition, we cannot extrapolate these findings to immunosuppressed patients. Overall, our 3 studies enrolled the patient populations most relevant to current treatment guidelines.32
Our results demonstrate that short episodes of genital HSV reactivation occur frequently among HSV-2 seropositive persons in the presence of standard dose valaciclovir and cannot be eradicated with high dose antivirals. While currently available anti-HSV therapy provides benefit for individual patients by preventing clinical HSV recurrences, more potent suppressive therapies, including antiviral drugs or immunotherapy in the form of therapeutic vaccines, are needed to provide substantial public health benefits, such as prevention of HSV-2 transmission and HIV-1 acquisition and transmission.
Supplementary Material
Panel: Research in Context.
Systematic Review
A meta-analysis of 14 randomized clinical trials of antiviral therapy for HSV-2 showed a 47% global RR reduction of recurrence in patients on suppressive therapy compared to placebo 33. Aciclovir and valaciclovir also suppress genital herpes shedding by 70–80% compared to placebo.7 Suppression of genital herpes with aciclovir and valaciclovir have been studied for public health benefit; both to prevent HSV-2 transmission9 and HIV transmission12 and acquisition10, 11. Despite 70% reduction in genital HSV-2 shedding, HSV-2 transmission was suppressed by only 48%9. Furthermore, HSV-2 suppression was not effective in preventing HIV transmission12 or acquisition10, 11. These results have led to questions about why there is a discrepancy in shedding rates and clinical outcomes, and whether higher doses of antiviral would be more effective.
Interpretation
The current study demonstrates that short, subclinical shedding persists on both standard and high dose aciclovir and valaciclovir. While the HSV shedding rate was reduced by 50% on the highest doses of valaciclovir (1 gm three times daily), the rate of breakthrough shedding episodes remained the same, at ~16–20 episodes/year. These data suggest that novel therapies are needed to completely prevent HSV reactivation.
Acknowledgments
The paper is in memory of Laura P. Olin, ARNP, a dedicated clinician who participated in the conduct of these studies and passed away in 2009.
Funding: NIH STI-TM-CRC-DAP(CJ), K23 AI079394(CJ), K23 AI087206(JS), K24 AI071113(AW), PO1 AI30731(AM,DMK,LC,AW). GlaxoSmithKline provided valaciclovir for Trial 3.
Footnotes
Presented in part at the 18th International Society of STD Research Conference, London, United Kingdom, June 28-July 1, 2009, and the 19th International Society of STD Research Conference, Quebec City, Canada July 10-13, 2011.
Trial Registration: ClinicalTrials.gov NCT00362297, NCT00723229, NCT01346475).
Conflicts of Interest:
CJ is a research investigator for AiCuris, GmbH. AW and LC are consultants for AiCuris GmbH, which is developing treatments for HSV. LC is the head of the scientific advisory board for and holds stock (<1% of company) in Immune Design Corp. LC and DMK are listed as coinventors on several patents describing antigens and epitopes to which T-cell responses to HSV-2 are directed.
AM and DMK are consultants for Immune Design Corp.
DMK is a consultant to Sanofi-Pasteur and Agenus Inc, for HSV-2 vaccines, and is contracted by Coridon Pty Ltd, Vical, Inc, and PATH for preclinical evaluation of a candidate HSV-2 vaccine. MS, SK, SS, MLH, JTS no conflicts.
Contributions of authors: CJ, AW, and LC designed the trials. SK and SS collected the data and CJ supervised this process. MLH and DMK performed laboratory assays. MS, AM, JTS, and CJ analyzed the data. CJ drafted the manuscript. All authors contributed to data interpretation and manuscript editing. All authors approved the final version of the manuscript.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Looker KJ, Garnett GP, Schmid GP. An estimate of the global prevalence and incidence of herpes simplex virus type 2 infection. Bulletin of the World Health Organization. 2008;86:805–12A. doi: 10.2471/BLT.07.046128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Freeman EE, Weiss HA, Glynn JR, Cross PL, Whitworth JA, Hayes RJ. Herpes simplex virus 2 infection increases HIV acquisition in men and women: systematic review and meta-analysis of longitudinal studies. AIDS. 2006;20(1):73– 83. doi: 10.1097/01.aids.0000198081.09337.a7. [DOI] [PubMed] [Google Scholar]
- 3.Xu F, Sternberg MR, Kottiri BJ, et al. Trends in herpes simplex virus type 1 and type 2 seroprevalence in the United States. Jama. 2006;296(8):964–73. doi: 10.1001/jama.296.8.964. [DOI] [PubMed] [Google Scholar]
- 4.Morris SR, Bauer HM, Samuel MC, Gallagher D, Bolan G. Neonatal Herpes Morbidity and Mortality in California, 1995–2003. Sexually Transmitted Diseases. 2008;35(1):14–8. 0.1097/OLQ.0b013e3180f62bc7. [PubMed] [Google Scholar]
- 5.Mertz GJ, Jones CC, Mills J, et al. Long-term acyclovir suppression of frequently recurring genital herpes simplex virus infection. A multicenter double-blind trial. Jama. 1988;260(2):201–6. [PubMed] [Google Scholar]
- 6.Reitano M, Tyring S, Lang W, et al. Valaciclovir for the suppression of recurrent genital herpes simplex virus infection: a large-scale dose range-finding study. International Valaciclovir HSV Study Group. J Infect Dis. 1998;178(3):603–10. doi: 10.1086/515385. [DOI] [PubMed] [Google Scholar]
- 7.Gupta R, Wald A, Krantz E, et al. Valacyclovir and acyclovir for suppression of shedding of herpes simplex virus in the genital tract. J Infect Dis. 2004;190(8):1374–81. doi: 10.1086/424519. [DOI] [PubMed] [Google Scholar]
- 8.Wald A, Zeh J, Barnum G, Davis LG, Corey L. Suppression of subclinical shedding of herpes simplex virus type 2 with acyclovir. Ann Intern Med. 1996;124(1 Pt 1):8–15. doi: 10.7326/0003-4819-124-1_part_1-199601010-00002. [DOI] [PubMed] [Google Scholar]
- 9.Corey L, Wald A, Patel R, et al. Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N Engl J Med. 2004;350(1):11–20. doi: 10.1056/NEJMoa035144. [DOI] [PubMed] [Google Scholar]
- 10.Celum C, Wald A, Hughes J, et al. Effect of aciclovir on HIV-1 acquisition in herpes simplex virus 2 seropositive women and men who have sex with men: a randomised, double-blind, placebo-controlled trial. Lancet. 2008;371(9630):2109–19. doi: 10.1016/S0140-6736(08)60920-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Watson-Jones D, Weiss HA, Rusizoka M, et al. Effect of herpes simplex suppression on incidence of HIV among women in Tanzania. N Engl J Med. 2008;358(15):1560–71. doi: 10.1056/NEJMoa0800260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Celum C, Wald A, Lingappa JR, et al. Acyclovir and transmission of HIV-1 from persons infected with HIV-1 and HSV-2. N Engl J Med. 2010;362(5):427–39. doi: 10.1056/NEJMoa0904849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mark K, Wald A, Magaret A, et al. Rapidly Cleared Episodes of Herpes Simplex Virus Reactivation in Immunocompetent Adults. J Infect Dis. 2008;198(8):1141–9. doi: 10.1086/591913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mark KE, Wald A, Magaret AS, et al. Rapidly cleared episodes of oral and anogenital herpes simplex virus shedding in HIV-infected adults. J Acquir Immune Defic Syndr. 2010;54(5):482–8. doi: 10.1097/QAI.0b013e3181d91322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schiffer JT, Abu-Raddad L, Mark KE, et al. Frequent release of low amounts of herpes simplex virus from neurons: results of a mathematical model. Sci Transl Med. 2009;1(7):7–16. doi: 10.1126/scitranslmed.3000193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Schiffer JT, Abu-Raddad L, Mark KE, et al. Mucosal host immune response predicts the severity and duration of herpes simplex virus-2 genital tract shedding episodes. Proc Natl Acad Sci U S A. 2010;107(44):18973–8. doi: 10.1073/pnas.1006614107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Beutner KR, Friedman DJ, Forszpaniak C, Andersen PL, Wood MJ. Valaciclovir compared with acyclovir for improved therapy for herpes zoster in immunocompetent adults. Antimicrob Agents Chemother. 1995;39(7):1546–53. doi: 10.1128/aac.39.7.1546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Magaret A, Johnston C, Wald A. Use of the designation “shedder” in mucosal detection of herpes simplex virus DNA involving repeated sampling. Sex Transm Infect. 2009;85(4):270–75. doi: 10.1136/sti.2008.034751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mark K, Wald A, Magaret A, et al. Rapidly Cleared Episodes of Herpes Simplex Virus Reactivation in Immunocompetent Adults. The Journal of Infectious Diseases. 2008;198(8):1141–9. doi: 10.1086/591913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wald A, Zeh J, Selke S, et al. Reactivation of genital herpes simplex virus type 2 infection in asymptomatic seropositive persons. N Engl J Med. 2000;342(12):844–50. doi: 10.1056/NEJM200003233421203. [DOI] [PubMed] [Google Scholar]
- 21.Ashley RL, Militoni J, Lee F, Nahmias A, Corey L. Comparison of Western blot (immunoblot) and glycoprotein G-specific immunodot enzyme assay for detecting antibodies to herpes simplex virus types 1 and 2 in human sera. J Clin Microbiol. 1988;26(4):662–7. doi: 10.1128/jcm.26.4.662-667.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jerome KR, Huang ML, Wald A, Selke S, Corey L. Quantitative stability of DNA after extended storage of clinical specimens as determined by real-time PCR. J Clin Microbiol. 2002;40(7):2609–11. doi: 10.1128/JCM.40.7.2609-2611.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Magaret AS, Wald A, Huang ML, Selke S, Corey L. Optimizing PCR positivity criterion for detection of herpes simplex virus DNA on skin and mucosa. J Clin Microbiol. 2007;45(5):1618–20. doi: 10.1128/JCM.01405-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Czartoski T, Liu C, Koelle DM, Schmechel S, Kalus A, Wald A. Fulminant, acyclovir-resistant, herpes simplex virus type 2 hepatitis in an immunocompetent woman. J Clin Microbiol. 2006;44(4):1584–6. doi: 10.1128/JCM.44.4.1584-1586.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Diggle PJ, Liang KY, Zeger SL. Analysis of longitudinal data. New York: Oxford University Press; 1994. [Google Scholar]
- 26.Schiffer JT, Wald A, Selke S, Corey L, Magaret A. The Kinetics of Mucosal Herpes Simplex Virus-2 Infection in Humans: Evidence for Rapid Viral-Host Interactions. Journal of Infectious Diseases. 2011;204(4):554–61. doi: 10.1093/infdis/jir314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Morfin F, Thouvenot D. Herpes simplex virus resistance to antiviral drugs. Journal of Clinical Virology. 2003;26(1):29–37. doi: 10.1016/s1386-6532(02)00263-9. [DOI] [PubMed] [Google Scholar]
- 28.Lycke J, Malmestrom C, Stahle L. Acyclovir Levels in Serum and Cerebrospinal Fluid after Oral Administration of Valacyclovir. Antimicrob Agents Chemother. 2003;47(8):2438–41. doi: 10.1128/AAC.47.8.2438-2441.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Brandi G, Schiavano GF, Balestra E, Tavazzi B, Perno C-F, Magnani M. The potency of acyclovir can be markedly different in different cell types. Life Sciences. 2001;69(11):1285–90. doi: 10.1016/s0024-3205(01)01213-9. [DOI] [PubMed] [Google Scholar]
- 30.Brown ZA, Wald A, Morrow RA, Selke S, Zeh J, Corey L. Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant. Jama. 2003;289(2):203–9. doi: 10.1001/jama.289.2.203. [DOI] [PubMed] [Google Scholar]
- 31.Wald A, Huang M-L, Carrell D, Selke S, Corey L. Polymerase Chain Reaction for Detection of Herpes Simplex Virus (HSV) DNA on Mucosal Surfaces: Comparison with HSV Isolation in Cell Culture. Journal of Infectious Diseases. 2003;188(9):1345–51. doi: 10.1086/379043. [DOI] [PubMed] [Google Scholar]
- 32.Workowski KA, Berman S. Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm Rep. 2010;59(RR-12):1–110. [PubMed] [Google Scholar]
- 33.Lebrun-Vignes B, Bouzamondo A, Dupuy A, Guillaume J-C, Lechat P, Chosidow O. A meta-analysis to assess the efficacy of oral antiviral treatment to prevent genital herpes outbreaks. Journal of the American Academy of Dermatology. 2007;57(2):238–46. doi: 10.1016/j.jaad.2007.02.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.