Abstract
In a previous publication1 we reported that the tyrosine selective reagent, tetraitromethane, causes complete inactivation of E. coli 30S ribosomes for poly U directed non-enzymatic phe-tRNA binding. This inactivation was demonstrated to be due to the chemical modification of the protein moiety of the ribosome. We have no identified the proteins of the 30S particle inactivated by this modification. Using a method of ribosome reconstruction we have found that unmodified proteins S1, S11, and S21 are essential for the restoration of the phe-tRNA binding activity of tetranitromethane inactivated ribosomes. We propose that these three proteins are intimately involved in the 30S ribosome binding site for tRNA.
Full text
PDF
















Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cantrell M., Craven G. R. Chemical inactivation of Escherichia coli 30 S ribosomes with maleic anhydride: identification of the proteins involved in polyuridylic acid binding. J Mol Biol. 1977 Sep 25;115(3):389–402. doi: 10.1016/0022-2836(77)90161-9. [DOI] [PubMed] [Google Scholar]
- Craven G. R., Gavin R., Fanning T. The transfer RNA binding site of the 30 S ribosome and the site of tetracycline inhibition. Cold Spring Harb Symp Quant Biol. 1969;34:129–137. doi: 10.1101/sqb.1969.034.01.019. [DOI] [PubMed] [Google Scholar]
- Craven G. R., Gupta V. Three-dimensional organization of the 30S ribosomal proteins from Escherichia coli. I. Preliminary classification of the proteins. Proc Natl Acad Sci U S A. 1970 Nov;67(3):1329–1336. doi: 10.1073/pnas.67.3.1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doyle R. J., Bello J., Roholt O. A. Probable protein crosslinking with tetranitromethane. Biochim Biophys Acta. 1968 Jun 26;160(2):274–276. doi: 10.1016/0005-2795(68)90103-7. [DOI] [PubMed] [Google Scholar]
- Gabbay E. J., Sanford K., Baxter C. S., Kapicak L. Specific interaction of peptides with nucleic acids. Evidence for a "selective bookmark" recognition hypothesis. Biochemistry. 1973 Oct 9;12(21):4021–4029. doi: 10.1021/bi00745a001. [DOI] [PubMed] [Google Scholar]
- Girshovich A. S., Bochkareva E. S., Kramarov V. M., Ovchinnikov YuA E. coli 30 S and 50 S ribosomal subparticle components in the localization region of the tRNA acceptor terminus. FEBS Lett. 1974 Sep 1;45(1):213–217. doi: 10.1016/0014-5793(74)80847-1. [DOI] [PubMed] [Google Scholar]
- Gorini L. Ribosomal discrimination of tRNAs. Nat New Biol. 1971 Dec 29;234(52):261–264. doi: 10.1038/newbio234261a0. [DOI] [PubMed] [Google Scholar]
- Hardy S. J., Kurland C. G., Voynow P., Mora G. The ribosomal proteins of Escherichia coli. I. Purification of the 30S ribosomal proteins. Biochemistry. 1969 Jul;8(7):2897–2905. doi: 10.1021/bi00835a031. [DOI] [PubMed] [Google Scholar]
- Heimark R. L., Kahan L., Johnston K., Hershey J. W., Traut R. R. Cross-linking of initiation factor IF3 to proteins of the Escherichia coli 30 S ribosomal subunit. J Mol Biol. 1976 Aug 5;105(2):219–230. doi: 10.1016/0022-2836(76)90108-x. [DOI] [PubMed] [Google Scholar]
- Kurland C. G. Structure and function of the bacterial ribosome. Annu Rev Biochem. 1977;46:173–200. doi: 10.1146/annurev.bi.46.070177.001133. [DOI] [PubMed] [Google Scholar]
- Kurland C. G. The requirements for specific sRNA binding by ribosomes. J Mol Biol. 1966 Jun;18(1):90–108. doi: 10.1016/s0022-2836(66)80079-7. [DOI] [PubMed] [Google Scholar]
- LEBOY P. S., COX E. C., FLAKS J. G. THE CHROMOSOMAL SITE SPECIFYING A RIBOSOMAL PROTEIN IN ESCHERICHIA COLI. Proc Natl Acad Sci U S A. 1964 Dec;52:1367–1374. doi: 10.1073/pnas.52.6.1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lake J. A. Aminoacyl-tRNA binding at the recognition site is the first step of the elongation cycle of protein synthesis. Proc Natl Acad Sci U S A. 1977 May;74(5):1903–1907. doi: 10.1073/pnas.74.5.1903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lake J. A., Kahan L. Ribosomal proteins S5, S11, S13 and S19 localized by electron microscopy of antibody-labeled subunits. J Mol Biol. 1975 Dec 25;99(4):631–644. doi: 10.1016/s0022-2836(75)80177-x. [DOI] [PubMed] [Google Scholar]
- Mizushima S., Nomura M. Assembly mapping of 30S ribosomal proteins from E. coli. Nature. 1970 Jun 27;226(5252):1214–1214. doi: 10.1038/2261214a0. [DOI] [PubMed] [Google Scholar]
- NIRENBERG M., LEDER P. RNA CODEWORDS AND PROTEIN SYNTHESIS. THE EFFECT OF TRINUCLEOTIDES UPON THE BINDING OF SRNA TO RIBOSOMES. Science. 1964 Sep 25;145(3639):1399–1407. doi: 10.1126/science.145.3639.1399. [DOI] [PubMed] [Google Scholar]
- Nomura M., Mizushima S., Ozaki M., Traub P., Lowry C. V. Structure and function of ribosomes and their molecular components. Cold Spring Harb Symp Quant Biol. 1969;34:49–61. doi: 10.1101/sqb.1969.034.01.009. [DOI] [PubMed] [Google Scholar]
- Riordan J. F., Christen P. Reaction of tetranitromethane with protein sulfhydryl groups. Inactivation of aldolase. Biochemistry. 1968 Apr;7(4):1525–1530. doi: 10.1021/bi00844a040. [DOI] [PubMed] [Google Scholar]
- Shih C. Y., Craven G. R. Identification of neighbor relationships among proteins in the 30 S ribosome: intermolecular cross-linkage of three proteins induced by tetranitromethane. J Mol Biol. 1973 Aug 25;78(4):651–663. doi: 10.1016/0022-2836(73)90286-6. [DOI] [PubMed] [Google Scholar]
- Shimizu M., Craven G. R. Chemical inactivation of Escherichia coli 30-S ribosomes by iodination. Identification of proteins involved in tRNA binding. Eur J Biochem. 1976 Jan 2;61(1):307–315. doi: 10.1111/j.1432-1033.1976.tb10023.x. [DOI] [PubMed] [Google Scholar]
- Shimizu M., Craven G. R. Further evidence for the participation of proteins S 3, S 14 and S 19 in tRNA binding to E. coli 30 S subunits. Mol Biol Rep. 1976 Nov;3(2):105–111. doi: 10.1007/BF00423223. [DOI] [PubMed] [Google Scholar]
- Smolarsky M., Tal M. Novel method for measuring polyuridylic acid binding to ribosomes. Biochim Biophys Acta. 1970 Feb 18;199(2):447–452. doi: 10.1016/0005-2787(70)90087-0. [DOI] [PubMed] [Google Scholar]
- Sokolovsky M., Riordan J. F., Vallee B. L. Tetranitromethane. A reagent for the nitration of tyrosyl residues in proteins. Biochemistry. 1966 Nov;5(11):3582–3589. doi: 10.1021/bi00875a029. [DOI] [PubMed] [Google Scholar]
- Sommer A., Traut R. R. Identification by diagonal gel electrophoresis of nine neighboring protein pairs in the Escherichia coli 30 S ribosome crosslinked with methyl-4-mercaptobutyrimidate. J Mol Biol. 1975 Oct 5;97(4):471–481. doi: 10.1016/s0022-2836(75)80054-4. [DOI] [PubMed] [Google Scholar]
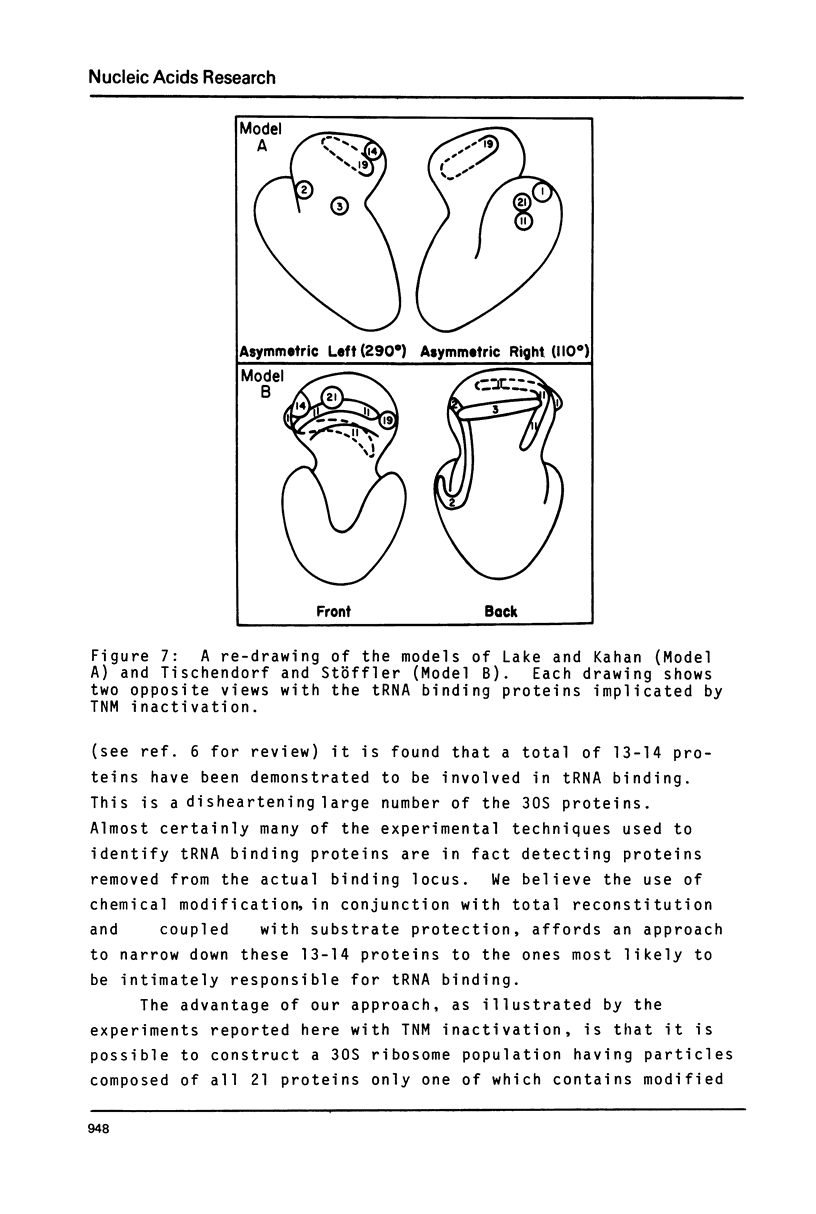
- Tischendorf G. W., Zeichhardt H., Stöffler G. Architecture of the Escherichia coli ribosome as determined by immune electron microscopy. Proc Natl Acad Sci U S A. 1975 Dec;72(12):4820–4824. doi: 10.1073/pnas.72.12.4820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toulmé J. J., Hélène C. Specific recognition of single-stranded nucleic acids. Interaction of tryptophan-containing peptides with native, denatured, and ultraviolet-irradiated DNA. J Biol Chem. 1977 Jan 10;252(1):244–249. [PubMed] [Google Scholar]
- Traub P., Nomura M. Structure and function of Escherichia coli ribosomes. VI. Mechanism of assembly of 30 s ribosomes studied in vitro. J Mol Biol. 1969 Mar 28;40(3):391–413. doi: 10.1016/0022-2836(69)90161-2. [DOI] [PubMed] [Google Scholar]
- Voynow P., Kurland C. G. Stoichiometry of the 30S ribosomal proteins of Escherichia coli. Biochemistry. 1971 Feb 2;10(3):517–524. doi: 10.1021/bi00779a026. [DOI] [PubMed] [Google Scholar]
- Wittmann H. G., Stöfflet G., Hindennach I., Kurland C. G., Birge E. A., Randall-Hazelbauer L., Nomura M., Kaltschmidt E., Mizushima S., Traut R. R. Correlation of 30S ribosomal proteins of Escherichia coli isolated in different laboratories. Mol Gen Genet. 1971;111(4):327–333. doi: 10.1007/BF00569784. [DOI] [PubMed] [Google Scholar]
- van Duin J., Kurland C. G., Dondon J., Grunberg-Manago M. Near neighbors of IF3 bound to 30S ribosomal subunits. FEBS Lett. 1975 Nov 15;59(2):287–290. doi: 10.1016/0014-5793(75)80394-2. [DOI] [PubMed] [Google Scholar]



