Abstract
Historically, déjà vu has been linked to seizure activity in temporal lobe epilepsy, and clinical reports suggest that many patients experience the phenomenon as a manifestation of simple partial seizures. We review studies on déjà vu in epilepsy with reference to recent advances in the understanding of déjà vu from a cognitive and neuropsychological standpoint. We propose a decoupled familiarity hypothesis, whereby déjà vu is produced by an erroneous feeling of familiarity which is not in keeping with current cognitive processing. Our hypothesis converges on a parahippocampal dysfunction as the locus of déjà vu experiences. However, several other temporal lobe structures feature in reports of déjà vu in epilepsy. We suggest that some of the inconsistency in the literature derives from a poor classification of the various types of déjà experiences. We propose déjà vu/déjà vécu as one way of understanding déjà experiences more fully. This distinction is based on current models of memory function, where déjà vu is caused by erroneous familiarity and déjà vécu by erroneous recollection. Priorities for future research and clinical issues are discussed.
1. Introduction
Déjà vu is a transitory mental state whereby a novel experience, such as a first time visit to a new city, feels as if it is familiar. Although the scientific literature on déjà vu is limited, much of it comes from the study of epilepsy, and it is this literature that we review here. Following a recent upsurge of interest in déjà vu following an influential review [1], experimental paradigms have been developed which produce an analogue of déjà vu in the laboratory (e.g., [2–4]). In this paper we take the view that déjà vu is a memory-based illusion, originating from the erroneous activation of the epistemic feeling of familiarity. As such, we are interested in how déjà vu is experienced in epilepsy, how it relates to memory in epilepsy more generally, and neural accounts of recognition memory in epilepsy. In turn, we elucidate what the study of epilepsy can contribute to our understanding of the déjà vu phenomenon.
The study of déjà experiences in temporal lobe epilepsy (TLE) has a long history, dating back at least to Hughlings-Jackson's 19th century description of the “dreamy state [5]”:
“[W]hat is occupying the attention is what has occupied it before, and indeed has been familiar, but has been for a time forgotten, and now is recovered with a slight sense of satisfaction as if it had been sought for.
Hughlings-Jackson (1888, page 202).”
Hughlings-Jackson [6] coined the phrase “dreamy states” to describe the nebulous mental states that occur as part of simple partial seizures (SPSs), referring to them as “double consciousness” and suggesting that “These are all voluminous mental states and yet of different kinds; no doubt they ought to be classified ….” Hughlings-Jackson did not use the term déjà vu to describe these peculiar experiences, but it is clear that one aspect of this dreamy state is the feeling of déjà vu. Hughlings-Jackson emphasises familiarity, preempting one of the first scientific attempts to classify the experience, typically defined as “… any subjectively inappropriate impression of familiarity of a present experience with an undefined past” (Neppe, 1983, page 3 [7]). Contemporary descriptions of a dreamy state converge on the same notion of a disturbance to consciousness, uneasiness, and dreamy qualities (see [8]).
We review déjà vu in TLE following four different strands within the literature: (1) surveys of the incidence and subjective qualities of the phenomenon and comparison with healthy individuals (questionnaires and surveys); (2) links between pathological brain abnormalities and déjà vu (structural and metabolic neuroimaging studies); (3) investigations of the neuroanatomical bases of déjà experiences (intracortical brain stimulation studies); (4) unusual ictal manifestations of a mnestic quality (single case reports).
We begin, however, by summarising research into the déjà vu phenomenon to produce a concrete account of this nebulous experience. For a full review of déjà experiences in neurology, psychiatry, and healthy participants, see O'Connor and Moulin [9]. The current view of déjà vu in healthy participants is that it is a memory error which exposes the nature of the memory system. According to models of memory and consciousness, our memory system is guided by feedback from experiential states and cognitive cues (experience based metacognition, e.g., [10]). For example, memory retrieval is guided by feelings of fluency and familiarity. A quick assessment of familiarity for part of an encoded item, for instance, is a likely signal that some more information about that item can be sought for [11]. The experience of novelty, on the other hand, signals the need to encode information in a scene or location [12].
In short, déjà vu occurs when the feeling of familiarity is decoupled either from the actual experience of a stimulus which is unfamiliar (see also Spatt [13]) or when familiarity is felt for material for which the prior experience is forgotten or unknown. The resulting mismatch is an erroneous feeling of familiarity for a stimulus which is simultaneously known by the experient to be unfamiliar. This second part, of knowing that the feeling is wrong is critical for the experience—feelings of false familiarity which are unopposed by the knowledge that they are false would be termed false positives within a recognition memory paradigm. Such errors are relatively common and are easy to produce in the laboratory, especially when the information is semantically related to a target episode, where the gist of an original trace is incorrectly recognized as old, such as endorsing the word “sofa” as old when it was the word “settee” that was studied [14]. Likewise, brief subliminal presentations of words in recognition tests can increase the likelihood of the item being endorsed as having occurred on a previous study list on the basis of familiarity [15]. To reiterate, this does not produce déjà vu, since the participant is not aware of the “false” nature of the familiarity for the previously unpresented items in the recognition memory test.
Other decouplings of epistemic feelings and memory processing can occur. For instance, jamais vu (often described as the inverse of déjà vu—a sense of unfamiliarity for a familiar stimulus), by our view would arise because someone has the experience of novelty for a stimulus which they nonetheless know to be familiar. One other similar clash of evaluations between consciousness and memory is the tip-of-the-tongue (ToT) phenomenon; people can feel with some certainty that they know a word but are temporarily unable to reproduce it (e.g., [16]). The critical issue with the ToT is that it seems to signal that material is accessible without actually accessing that material itself, suggesting a reflective “feeling” which is part of the retrieval process from memory, and yet which does not yield the sought-for item itself, and is somewhat disconnected from it.
The nearest laboratory analogue for our account of déjà vu in healthy participants is “recognition without identification” (RWI)—a line of reasoning began by Brown and Marsh [2]. In their study, participants shallowly processed pictures of visual scenes and were one week (Experiment 1) and three weeks (Experiment 2) later asked to report the likelihood they had visited the places. They were able to instill a bias in retrieval which the participants were not aware of, and some compared this to déjà vu. Picking up on this work, Cleary and Reyes [17] have produced a déjà vu-like experience in healthy participants by arranging for them to process materials during an encoding phase in such a way that they are able to “recognise” materials but not know from where they encountered the information. For instance, participants may perceive a set of scenes in a study phase and see very similar scenes in a test phase. There is sufficient overlap that the participant feels that they have encountered the scene but cannot pinpoint the source of this feeling. Interestingly, there are similar accounts of déjà vu historically. Knight [18] experienced déjà vu whilst in Tibet and, subsequently, suggested that this was due to the vivid descriptions of Laputa in Swift's Gulliver's Travels, a similarity which was not apparent or retrieved at the time of the déjà vu. Cleary proposes a similarity hypothesis of déjà vu arising due to familiarity with a scene caused by an overlap with a previous experience which is undetected by the person experiencing it.
Familiarity is an entity which is relatively well understood in the brain (for review see [19]). For the memory researcher, familiarity is often encountered in reference to recognition memory, where there is a well established dichotomy of recollection and familiarity, the dual process view. According to this view, humans can endorse items as “old” on the basis of two separate processes, recollection and familiarity. Recollection is associated with a feeling of “mental time travel” which arises alongside the retrieval of specifics from the prior encoding experience. This may include contextual details such as the source of a piece of information but also fragmentary images, feelings, and thoughts. Familiarity, on the other hand, is a mere sensation of prior experience, or just “knowing” that something has been encountered before, devoid of any such evocative contextual information. A predominant view is that familiarity is experienced along a continuum, such that people can evaluate the strength of familiarity, but that recollection is an all-or-nothing state [20]. These issues are critical for the neuroscience of memory since they pertain to different brain areas—the hippocampus and a network of associated brain regions mainly within the medial temporal lobe (MTL) is responsible for recollection, and a more isolated perirhinal region is responsible for familiarity [21]. Figure 1 displays a simplified graphical outline of these brain areas, which we will refer to throughout this paper. Much research is done in this area by using subjective report; participants are reliably able to distinguish between familiarity and recollection in their recognition test performance, and this produces reliable differences in neural activation as indexed by fMRI [22].
Figure 1.
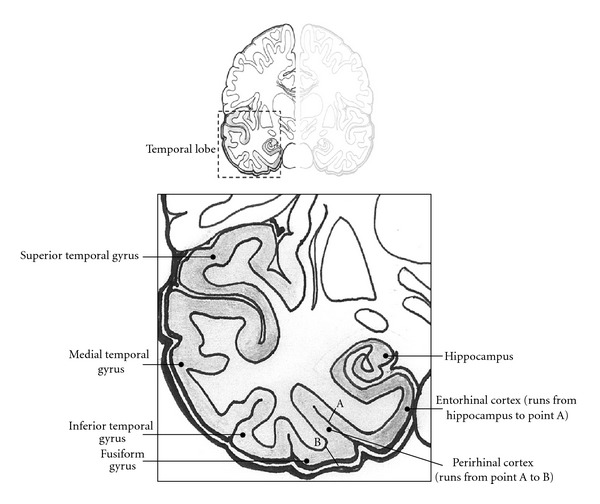
Coronal section of the temporal lobe showing key structures.
It is not surprising therefore that researchers have considered the separate contribution of recollection and familiarity in TLE [23–28]. The consensus is that whereas there is impaired recollection in TLE, familiarity is largely spared. This means that people with TLE can make effective memory judgments on the basis of an assessment of familiarity, but lack recollection. This is manifested not only in a lack of contextual details in their memories of personal experiences [29] but also on recognition memory tests [27].
By this view, we might expect an increase in déjà vu in patients with TLE which is in some way related to their memory deficit according to the cognitive account above. This could occur in a chronic form due to the fact that familiarity is intact in TLE, but the retrieval of contextual information is impaired. One possibility is that déjà vu experiences in TLE are a clinical manifestation of RWI. This may mean that déjà vu in TLE is an earnest error driven by forgetting—scenes may be genuinely familiar to the subject of experience, but they cannot retrieve how they encountered the information previously. Data-driven accounts such as this [30–32] view déjà vu as a bottom-up process, whereby familiarity is actually a relevant assessment of a scene, but the person cannot access why they feel that way (similar things have been produced using hypnosis, e.g., [33]). An alternative hypothesis presented by Spatt [13] suggests that déjà vu is a result of false activation of an isolated parahippocampal based familiarity system; in TLE this occurs when familiarity is generated erroneously as part of seizure activity, and is not appropriate to the current scene. The main way to differentiate these two accounts would be knowing what triggers déjà vu. If it is due to an underlying chronic memory problem we would expect to see it outside of seizure activity, whereas if it is a top-down brain-based memory error, we would expect that it was related to seizure activity. The first part of the review considers these issues by looking at the incidence and phenomenology of déjà vu.
2. Incidence and Phenomenology of Déjà Experiences
Anecdotal reports of déjà vu consistently suggest that it is experienced as part of SPSs in some TLE patients. Given the frequency of these reports, one might expect then that déjà vu is experienced more commonly in TLE than in the normal population. Several researchers have examined the incidence and nature of déjà experiences in TLE compared to healthy participants. The key issue is whether there is any diagnostic value to déjà experiences.
A recent large-scale questionnaire study by Adachi et al. [34] addressed this question. They administered the Inventory of Déjà vu Experiences Assessment (IDEA, [35]) scale, which obtains measures of frequency of occurrence and the subjectively perceived psychobehavioural consequences of having déjà vu. In their sample of 312 patients, 143 had TLE; these were matched with a control group of 402 healthy adults. Intriguingly, they found that significantly fewer patients had some form of déjà vu (63.1%), compared with controls (76.1%). This does not tally with the idea stated above that due to temporal lobe dysfunction, and resulting deficits in recollection, people with TLE are disposed to a memory-based déjà vu experience.
This might suggest that there is nothing unique about the déjà vu experience in epilepsy. Adachi and colleagues were also motivated to investigate whether the subjective experience of déjà vu could be predictive of seizure activity. They asked patients to differentiate between instances where they were aware the déjà vu was part of seizure activity (seizure recognition form—SR) and those when it occurred as a nonepileptic subjective event (nonseizure recognition—NSR). The SR form was experienced by 24% of patients and most commonly in TLE, whereas 55.6% had the NSR form, and 16.3% encountered both. In those patients experiencing both types, it was found that the SR form tended to occur more frequently at night, was reported as occurring more recently, and was associated with negative affect and more dissociative features; hence there were several phenomenological differences between the two. However, Adachi et al. [34] found no association between the SR form and mesial temporal sclerosis (MTS) but patients with MTS experienced the NSR form less frequently than others, that is, they actually, again, had lower levels of the “healthy” form of déjà vu.
The finding of a lower incidence of any déjà vu in patients therefore provides evidence that déjà vu is a “healthy” phenomenon, actually occurring less frequently in people with damage to the temporal lobe. This suggests that memory dysfunction per se does not lead to déjà vu experience, as was shown in a recent group study examining the relationship between verbal memory and types of SPSs in 42 TLE patients [36]. This also seems reasonable given that other forms of memory impairment which stem from temporal lobe atrophy also do not lead to higher levels of déjà vu, for example, Alzheimer's disease or even healthy aging [37]. One way of examining this issue aside of imaging reports of hippocampal sclerosis would be to examine correlations between memory performance and incidence of déjà vu.
It seems likely that TLE patients in particular, however, are more likely to report déjà vu experiences due to the different and perhaps more striking phenomenological characteristics when it is experienced as part of a seizure. Future research should explore this phenomenology further. One possibility might be that it is false feelings of recollection and not familiarity which are elevated in epilepsy—we return to this idea below.
Another questionnaire study compared the qualitative characteristics of déjà vu and jamais vu in 15 TLE patients with 115 university students [38]. Similar to Adachi et al.'s [34] healthy controls, they found that 75% of students experienced déjà vu, and approximately 25% had experienced jamais vu. Déjà vu was experienced more for places than people in both controls and patients, but there were qualitative differences in the circumstances under which it occurred. For example, ictal déjà vu was more likely to occur in familiar locations; for people, it was unrelated to the degree of familiarity of the person; it generally did not evoke feelings of surprise, and it was more likely in a state of mental relaxation. In controls, however, déjà vu was more likely to occur for unfamiliar locations and people, cause surprise, and happen more often in a state of mental fatigue. Common characteristics between the two types included the average duration of the phenomenon (in the order of seconds) and a feeling of nostalgia. In contrast to Adachi et al. [34], the authors did not find déjà experiences to be associated with negative emotion, although jamais vu tended to be associated with negative emotions and fear.
Other recent studies have sought to investigate the phenomenological characteristics of déjà vu in TLE through comprehensively assessing patient's subjective reports of their experiences. In their survey, Johanson et al. [39] conducted a content analysis of 262 verbal reports of subjective experiences arising from partial seizures in a sample of 40 patients, of whom 19 had TLE. Immediately following an SPS, participants called a telephone number and attempted to describe what was felt during the ictal and postictal period. The authors found sensory and bodily sensations occurred most frequently, but “hallucinatory” experiences were also common. In total, 29% of recorded seizures consisted of clear visual hallucinations and 18% were classified as “misperceived memories,” including déjà vu, a feeling of “prescience,” and the concomitant retrieval of actual episodic memories. In a different study, Sadler and Rahey [40] describe how only 3 out of the 218 patients surveyed experienced prescience, or a profound sense of “knowing” what was about to happen next. It seems evident then that there are identifiable memory based experiences distinct from déjà vu, which warrant attention in themselves.
The “misperceived memories” in Johanson et al.'s study [39] were common in patients with temporal lobe foci, as were feelings of depersonalization and bizarreness. As with Adachi et al. [34], they found fear and negative affect to be the most common emotions. Sengoku et al. [41] attempted to specifically classify these varying phenomena alongside the emotions experienced in order to assess any potential relationship with a tendency to develop psychotic symptoms. They classified the recurrent “dreamy states” of 13 patients as déjà vu, jamais vu, or reminiscences and assessed the prevailing emotions accompanying each. They found that patients with jamais vu experienced more negative affect and fear and were much more likely to develop psychotic symptoms compared to those with déjà vu and reminiscence, which were primarily associated with positive or neutral affect. We describe case reports of distressing déjà experiences below. In summary, evidence from questionnaire studies is conclusive of a preferential involvement of the temporal lobes in the formation of déjà vu, due to its occurrence as part of temporal lobe seizures, and its association with emotion, suggesting activity in the amygdala. However, the bottom-up “recognition without identification” interpretation of the phenomenon in TLE seems not to hold because people with TLE do not have déjà vu more than the healthy population, and can differentiate between healthy déjà vu and seizure-related déjà vu. O'Connor and Moulin [42] examined such “data-driven” accounts of déjà vu in their discussion of a single case of TLE following encephalitis. They report a man who experienced prolonged periods of déjà vu before complex partial seizures during which he was initially frightened and disturbed. The case described how he tried to reduce the sensation: “[I] went through a period of looking away from what I was recognizing, hoping that this would get rid of the déjà vu. I now know that looking away, or at other things does not help, because the déjà vu follows my line of vision and hearing” (page 145). This converges on the idea that, rather than déjà vu being driven by the mismatch of current experience with similar previous perceptual experiences, it is an illusory epistemic feeling arising from the alteration of higher order processing as a result of seizure activity.
The qualitative differences found between déjà vu originating from seizures and in the healthy population reviewed above seem to converge on underlying medial temporal brain pathology, as implied by Adachi et al. [34]. Despite the finding that déjà vu is less common in epilepsy than healthy people, it is nevertheless more predictable and consistent in patients experiencing the phenomenon frequently as part of their SPS. It seems from this that a chronic form of familiarity-based memory error is not behind déjà experience in TLE. The next issue is whether a familiarity-based account holds more generally, and to examine this issue we look in more detail at the temporal lobes.
3. Neuroimaging Studies
Hypometabolism during fluorine-18 fluorodeoxyglucose positron emission tomography (18FDG PET) has previously been shown to be a good indicator of the brain regions involved in the genesis and discharge of ictal events in TLE [43]. The first study to utilize this method in assessing the functional anatomy of déjà vu used a semiquantitative analysis to compare the metabolic activity in a range of neuroanatomical areas in patients with and without seizure-related déjà vu [44]. Patients were selected from a series of surgical candidates for intractable epilepsy; the sample included 14 patients with nonlesional unilateral TLE experiencing déjà vu habitually alongside SPS, and 17 patients not experiencing déjà vu matched for a number of epilepsy-related variables. Upon initial qualitative analysis, the groups' FDG PET images were indistinguishable as a similar number of patients in each showed either normal uptake, or diffuse hypometabolism of temporal areas in the left and right hemispheres. However, using principal component analysis, they derived a factor named the “visual network,” primarily including mesial temporal structures, the parietal cortex, and other visual association areas where significant hypometabolism was found predominantly in the left hemisphere of déjà vu patients, regardless of side of seizure onset. Interpreting their findings, the authors suggest that déjà vu may be the result of initial activation in mesial temporal structures, followed by spreading discharge into visual pathways; the activation of such a network helps explain the mnestic quality associated with the distorted visual cognition during déjà vu experiences.
More recently, Guedj et al. [45] conducted a similar FDG PET study, but used voxel-based analysis to identify specific structures within the medial temporal lobes responsible for déjà vu, rather than the regional analysis described earlier [44]. This study was theoretically motivated by the proposed specialization of medial temporal structures in familiarity-based recognition memory [46]. The sample included two matched groups of 8 TLE patients, differing only with respect to the presence or absence of déjà vu as a subjective manifestation during seizures. Further, a matched control group of 20 neurologically intact healthy subjects was used. In selecting participants, Guedj et al. [45] excluded patients displaying morphological abnormalities evident from brain MRI scans due to past research suggesting these can lead to decreased metabolism [47]. This was in contrast to Adachi et al. [44], in which a number of patients were found to have atrophy or abnormal signal intensity on MRI images. In comparison to healthy subjects, the déjà vu group displayed significant ipsilateral hypometabolism in parahippocampal regions (entorhinal and perirhinal cortices), the superior temporal sulcus, and superior temporal gyrus. No significant hypometabolism was found in the hippocampus in the déjà vu group; in comparison, TLE patients without déjà vu only displayed a significant hypometabolism within the hippocampus compared to controls. Analyses at the individual level further suggested a higher incidence of hypometabolism of the superior temporal gyrus and parahippocampus in patients with déjà vu, and no difference between groups in the number showing hypometabolism of the hippocampus and amygdala. The results therefore support the finding of significant hypometabolism in the medial temporal lobe (MTL) found in Adachi et al.'s [44] PET study, but do not corroborate their suggestion of the involvement of the parietal cortex. Instead, the superior temporal gyrus was implicated. The authors note that this area receives inputs from visual and auditory association areas; similar to the explanation provided by Adachi et al. [44] regarding the parietal cortex. The metabolic findings are interpreted by this group as lending support to the thesis that the parahippocampal regions are specifically and independently involved in familiarity-based recognition, and, hence, déjà vu experiences.
As the questionnaire surveys demonstrate, déjà vu and other SPSs appear to manifest alongside emotions of fear and occasionally pleasant feelings of nostalgia. As the amygdala is established as a structure pivotal in emotional processing, Van Paesschen et al. [48] examined whether abnormal amygdalae in patients with intractable TLE has any localizing value for SPSs. They recorded descriptions of SPSs from interviews with 50 patients before combining brain MRI and fluid attenuated inversion recovery (FLAIR) to obtain amygdala relaxation time mapping (AT2). SPS manifestations were classified into 15 types. As well as emotions such as fear, these also included déjà vu, which was defined as “a stereotypical feeling of familiarity, recognition, or reminiscence that the patient recognized as abnormal” (page 858). Importantly, they excluded patients with evidence of multifocal seizure onset and those with MRI evidence of foreign tissue lesions. They assessed the AT2 ipsilateral to the seizure focus, and found an equal number of patients with normal and abnormal amygdalae. Patients with an abnormal AT2 reported a higher median number of SPS to those with normal AT2 (6 compared to 3) and stepwise logistic regression analyses displayed that déjà vu, cephalic and warm sensations, gustatory hallucinations, and indescribable strange sensations were the best predictors in discriminating between those with abnormal AT2 and those without. Of these, déjà vu emerged as the best predictor of abnormal AT2. Déjà vu was more common amongst patients with right than left-sided AT2 abnormalities, although overall SPS frequency did not lateralize.
The authors conclude that in patients with an abnormal AT2 ipsilateral to the seizure focus, a larger number of SPS manifestations are reported. As they note, in order to confirm their conclusion, depth recordings would be necessary to rule out the possibility that the ictal onset was not within the hippocampus, where sclerotic tissue was found in 80% of patients. However, they found that the small number of patients with an abnormal AT2 in the absence of hippocampal sclerosis (HS) presented with a high number of SPS types, comparable to that of the patients with both abnormal AT2 and HS. Furthermore, the patients with both normal amygdala and hippocampi had a low number of SPS types, comparable to that of the patient group with normal AT2 and unilateral HS.
The neuroimaging studies reviewed above point to a number of key structures. Of the three papers, one points to hypometabolism in parahippocampal regions in the absence of any structural abnormality in TLE patients experiencing déjà vu [45]; a second metabolic study identified a larger network involving areas of the visual cortex and the possibility of hemispheric differences, but these authors did not control for structural damage [44]; the third study using an MRI methodology showed a relationship between abnormal amygdala activation and a range of experiential phenomenon [48]. On the whole, these studies fail to identify a common region responsible for déjà experiences. As such, only Guedj et al. [45] lend support to the idea of a disturbance in parahippocampal familiarity mechanisms alone. However, they do pinpoint dysfunctional activation in networks critical for memory function more generally. In the next section, the activation of more specific regions through direct stimulation of the brain is explored with the aim of elucidating the involvement of hippocampal and parahippocampal recognition memory structures to déjà vu experiences.
4. Intracortical Brain Stimulation Studies
Hughlings-Jackson's seminal paper described the involvement of the anterior MTL (including entorhinal and perirhinal cortices) as the genesis of “dreamy states,” including reminiscences (déjà vu) and vivid visual hallucinations, and characterized these as separate to a number of other SPS manifestations [49]. Since then, a number of studies emerged throughout the latter half of the twentieth century that attempted to assess the specific brain regions involved in déjà vu-type experiences by using intracortical brain stimulation as part of surgical evaluation for resective surgery in TLE. Later, Penfield rejected both Jackson's method of classifying the dreamy state and his conclusions about its localization in the MTL [50, 51]. Instead, he viewed other sensory illusions as akin to déjà vu (called interpretive illusions), and classed vivid recollections of past memories separately as experiential hallucinations. Both of these types of experience, he concluded, were dependent on activation of the lateral temporal neocortex, in particular the superior temporal gyrus.
Following Penfield's rebuttal of Jackson's earlier evidence, a number of other studies emerged, all finding that stimulation of the MTL was able to provoke déjà vu in patients either directly [52], or from after-discharge spreading from lateral areas to the amygdala [53, 54]. However, the lateral site of administration in Gloor et al.'s study [53] was the middle temporal gyrus, which did not support Penfield's earlier finding.
Some years later, Bancaud et al. [55] attempted to resolve this issue with a further assessment of 16 TLE patients involving the recording of subjective seizure manifestations during intracortical stimulation, chemical activation and spontaneous ictal events. Following attainment of relevant clinical data (surface EEG, semiology, imaging), depth electrode stereotaxic EEG (SEEG) was performed routinely to localize patients' seizures; this included a variety of stimulation sites at lateral and medial temporal regions. Their sample was diverse, including patients with a variety of aetiologies, and differing abnormalities on brain MRI; however, the authors document the observations of each individual patient. This provides a rich account of their findings, including direct quotations from patients and the anatomical sites being stimulated, of which many evoked phenomenological experiences. Despite the variability in subjective reports between and within seizures, the authors attempt to categorise the 57 experiences into five different types of dreamy state: memories of complete scenes; vague reminiscences; déjà experiences; strangeness-familiarity; nonclassifiable. Of these, memories, reminiscences, and déjà experiences were recorded the most.
Déjà experiences featured the feeling of “reliving” the current experience prompting the use of the term “déjà vécu” (already lived) for the first time in the scientific literature. We have subsequently developed a theoretical differentiation between déjà vu and déjà vécu, but it is one which overlaps with Bancaud et al.'s view [55] that déjà vécu is a longer and more intense form of déjà vu—the difference is neatly captured in the translations of déjà vu (already seen) and déjà vécu (already lived). We return to our technical definition below.
Bancaud et al. [55] classified strangeness-familiarity together due to reports that the erroneous familiarity associated with the present environment was actually experienced as a bizarre feeling. In terms of “visual hallucinations”, reminiscences and memory flashbacks were distinguished based on the vividness of detail reported by the patient. Interestingly, despite the visual quality of most dreamy states, rarely did patients report visual disturbance with the current perceptual environment. Instead, the visual element of these subjective experiences was essentially a memory, and Bancaud et al. [55], like Jackson [49], argue that elaborate visual hallucinations are distinct from the memory experiences in déjà vu and dreamy states.
There were a number of subjective phenomenon that rarely cooccurred with dreamy states, including oro-alimentary automatisms, loss of contact and other sensory hallucinations. Neuroanatomically, dreamy states were provoked primarily by stimulation of the anterior hippocampus, followed by the temporal neocortex, and to a lesser extent the amygdala. Dreamy states arising from spontaneous seizures always involved the amygdala, anterior hippocampus, and neocortex. Notably, activity always spread to the amygdala and neocortex following stimulation of the anterior portion of the hippocampus. Stimulations to the amygdala and hippocampus involving dreamy states almost always spread to the neocortex (17/20 times) but stimulation of the temporal neocortex only spread to these mesial temporal structures 8/15 times; this difference was found to be nonsignificant, however. Taking into account the frequency with which each of these structures evoked dreamy states (due to isolated stimulation, or site of afterdis charge), the temporal cortex produced the most, followed by the anterior hippocampus, and lastly the amygdala. However, the authors note that when considering the small number of SEEG contacts in the MTL as compared to the temporal cortex, it was easier to generate dreamy states from stimulation to this area. In fact, the findings suggested it was about ten times more likely to evoke dreamy states from the medial temporal structures (anterior hippocampus/amygdala) than temporal neocortical sites. Like others, Bancaud et al. [55] also found the superior temporal gyrus an effective site in producing dreamy states, supporting Penfield's previous observations [50, 51]. Laterality effects were not found for MT structures or the temporal neocortex.
Summarizing the findings, the authors conclude that for dreamy states to occur, the MTL and lateral temporal neocortex must all be involved, at least in the spread of activity in the initial 10 s following stimulation. Additionally, ipsilateral MTL stimulation was harder to generate dreamy states than on the nonepileptic contralateral side; a result also demonstrated by Halgren et al. [52] thus lending support to the conclusions drawn from questionnaire studies that déjà vu is actually part of “healthy” brain activity. In normal event processing, the hippocampus is involved in memory consolidation and retrieval, the lateral temporal neocortex is involved in object perception and identification and has connections to auditory and visual pathways, and the amygdala processes emotion; therefore the simultaneous activity of this network of regions during seizure propagation causes the common manifestations of the dreamy state. These authors were the first to specifically link the dreamy state to declarative memory systems.
Stemming from Bancaud et al.'s [55] findings, Bartolomei et al. [56] aimed to assess the role of subhippocampal structures (perirhinal and entorhinal cortices) in déjà experiences. They analysed the experiences generated from 240 stimulations in the rhinal cortices, amygdala and hippocampus in 24 patients undergoing presurgical evaluation. Overall, the subjective manifestations of stimulations in the rhinal cortices did not differ from those found in mesial temporal areas in this study, and no convincing effect of laterality was found. However, the frequency of different subjective phenomenon did differ between entorhinal and perirhinal cortices, and between rhinal cortices and other limbic structures; in particular, déjà vu and other memory phenomena. Stimulation of the entorhinal cortex was found to provoke déjà vu most often, whereas the perirhinal cortex was associated more with memory reminiscences; very few stimulations in the hippocampus and amygdala produced either of these responses. Overall then, Bartolomei et al. [56] displayed that illusions of familiarity were more likely to arise from stimulation of subhippocampal structures; they suggest that the previously reported involvement of other MTL structures may have arisen due to the localized discharge to the rhinal cortices. In support of their findings, they discuss animal and human studies of human recognition memory that implicate the rhinal cortices in familiarity discrimination.
These researchers later presented a more in-depth analysis of one particular patient who provided well-described experiential phenomenon [57]. Having established the roles of subhippocampal structures in dreamy states, they were motivated to address the burgeoning issue of how the mnestic content of experiential phenomenon may map onto episodic and semantic memory systems. During SEEG investigation, this patient only reported experiencing experiential phenomenon following stimulation of contacts located in what they termed “the perirhinal region.” Functional coupling analysis was used to determine the network of regions involved, and a strong relationship in the theta range was observed between the anterior hippocampus, perirhinal region and visual areas. The subjective manifestation of the first stimulation was associated with the production of an image of a lake and some bushes, which the patient recalled as being a place he frequents. A second higher intensity stimulation in the same region resulted in the same pattern of after discharge, and this time the patient experienced a visual image which he later confirmed was his brother's friend. These visual images related to decontextualised memories of people and places he had seen very often; essentially semantic memories. The authors suggest that activation of the visual areas, and the perirhinal regions involvement in context free memory explain this.
Vignal et al. also attempted to delineate dreamy states with reference to key memory related structures [58]. They investigated 16 patients, looking to compare any differences between provoked and spontaneous experiences. They found that 45% of dreamy states were evoked by amygdala stimulation, 37.5% by the hippocampus, and 17.5% by the parahippocampal gyrus. There was a preference for dreamy states to arise from right sided MTL stimulation, and in no cases was déjà vécu reported from stimulation of the left MTL. Further, they displayed that both déjà vécu and visual hallucinations only involved an after discharge to other medial temporal structures, and not the middle or superior temporal gyri as others have previously found [50, 51, 55]. This was found for provoked and spontaneous seizures, and interestingly, after discharge to temporal neocortex was associated with a lack of, or termination of dreamy states. Furthermore, in spontaneous seizures not involving the dreamy state, after discharge always spread to the whole temporal neocortex.
Based on their findings, Vignal et al. [58] attempt to resolve some of the past inconsistencies in the classification of these subjective phenomenon. They note the distinction made by Penfield and Perot [51] between déjà vécu; involving the altered perception of the present environment, and “experiential hallucinations”; essentially involving a past memory. In contrast, Vignal et al. [58] suggest there is a semiological continuity between the two experiences, as they were often reported to coexist during dreamy states. Citing the above findings of Bartolomei et al. [56], they propose that epileptic discharge restricted to the entorhinal cortex may result in a “pure” form of déjà vécu, but as activity is propagated to other hippocampal structures and the amygdala, widespread functional activation of the fronto-temporal autobiographical memory network may result in a more intense visual mnestic experience.
One theme suggested by the stimulation studies of Chauvel and colleagues [55–58] is that there is a continuum between feelings of déjà vu, déjà vécu, and recollection of actual scenes. This notion is borne out by Milton et al. [59] who describe a patient with a recently identified syndrome of TLE, transient epileptic amnesia (TEA, [60]). Milton et al.'s [59] patient was a 66-year-old male who presented with an extensive loss of autobiographical memories from his past that remained following AED treatment. He had never previously experienced déjà vu, but following his treatment period reported approximately 6-7 episodes that had the characteristics of déjà vécu. Strikingly, after these episodes he then reported the spontaneous retrieval of a number of remote memories that were previously inaccessible during the time he experienced the TEA attacks. These memories were verified by his wife. Formal testing revealed that one of the recovered memories was retrieved with the same level of contextual richness, or episodicity, as matched controls. For future research, it would be interesting to look at the occurrence of intrusive memories in TLE, since it may be that there is more overlap between the characteristics of déjà experiences and intrusive memories than there is from forgetfulness. Indeed, our top down hypothesis, whereby déjà vu in epilepsy derives from seizure related activity posits that déjà vu is essentially an uncontrolled and random mental event not unlike “mind pops” or intrusive memories in healthy populations.
The evidence outlined above appears to fit partially with current dual process theories of memory, and the neuroanatomical sites that have been proposed to be responsible for their action; familiarity-based recognition is proposed to be more reliant on anterior subhippocampal areas, where as recollection is reliant on the hippocampus and a distributed network of MTL regions [46]. Disruption to the familiarity system within parahippocampal structures has been suggested by Spatt [13] as a mechanism of déjà vu, and this is supported by the studies that found entorhinal cortex involvement in the phenomenon. However, as Bartolomei et al. [61] point out, isolated disruption to one localized area seems unlikely to be the only cause of such a complex high-level phenomenon, especially in light of recent evidence displaying memory processing is reliant on synchronization of multiple MTL structures, specifically in the theta band [62]. They therefore continued with the functional connectivity approach advocated in their earlier study [56] and assessed neural coupling and signal correlations between MT structures during SEEG recordings in TLE patients. They found that stimulations inducing déjà vu were associated with neural coupling between the rhinal cortices, hippocampus and amygdala, in both theta and beta bands; therefore arguing against Spatt's isolated familiarity system hypothesis [13]. Instead, their results favour the suggestion that déjà experiences may lie along a continuum [58]. The déjà vu found to be localized to the rhinal cortices may represent a lower level disruption of familiarity circuits, but as additional MT structures (hippocampus, amygdala) are recruited, a more intense déjà vecu phenomena occurs, sometimes with vivid visual distortion and associations with past memories leading to a feeling of having already lived the current experience.
5. Déjà Vu and Déjà Vécu
Our contribution to the déjà vu literature has been to attempt to classify déjà experiences into two forms according to the dual process view of recognition memory (see Table 1). It seems plausible to theorize that just as erroneous sensations of familiarity occur to give rise to déjà vu, erroneous sensations of remembering or recollecting may occur. This distinction between false sensations of familiarity and recollection might help further classify the déjà states, and more broadly speaking the dreamy state in epilepsy.
Table 1.
Overview of recollection and familiarity.
| Brain region | Subjective state | Contextual or source information | |
|---|---|---|---|
| Recollection | Hippocampus | “Remembering” | Some |
| Familiarity | Perihippocampal Regions | Finding familiar or “just knowing” | None |
We will not reproduce our whole argument here (see [63, 64]), but essentially we suggest that the déjà phenomenon can be separated into familiarity (déjà vu) and recollection (déjà vécu) errors. (For descriptions of déjà vu- and déjà vécu-like experiences induced in healthy participants see [33, 65].) To the best of our knowledge, the term déjà vécu was first used scientifically by Bancaud and colleagues [55], as noted above, although no clear justification of the term is provided (and ultimately Bancaud et al. [55] grouped together déjà vu and déjà vécu experiences). Funkhouser [65] attempted a fractionation of déjà experiences into three kinds, one of which was déjà vécu. Notably, to illustrate this experience he uses an oft-cited passage (cf., Hughlings Jackson, above [5]) from Dickens' David Copperfield (our emphasis):
“We have all some experience of a feeling, that comes over us occasionally, of what we are saying and doing having been said and done before, in a remote time—of our having been surrounded, dim ages ago, by the same faces, objects, and circumstances—of our knowing perfectly what will be said next, as if we suddenly remember it!
This fictional account contains contextual details, associated feelings and thoughts. As such, it parallels recollection, where in remembering an event we are able to draw up contextual information, and the specifics of a prior experience [67]. Thus far, our empirical support for the existence of a recollective process at play in déjà vécu comes from cases of recollective confabulation—dementia patients described as having “persistent déjà vu” who confabulate contextual details and false statements referring to previous study episodes [63, 68]. These are striking errors made more frequently for novel than mundane events and always accompanied by feelings of mental time travel, and reproducing the present as if it is contextual information from the past. With such extreme déjà experiences, it is possible to isolate the memory deficit using experimental tasks [68]. A few other cases have been reported with the same classification in their descriptions (e.g., [69]).
Although not guided by contemporary memory theory, Funkhouser [65] makes a compelling classification of déjà vu and déjà vécu. He describes déjà vu as a more nebulous experience, and differentiates it from déjà vécu thus as being “primarily or even exclusively a mental happening”. He suggests that déjà vu has no accompanying feeling of prescience, resonating with the idea of mental time travel in déjà vécu.
To some extent it is difficult given the lack of consensus with regards to definitions of various déjà experiences, and the reliance on a retrospective classification of subjective reports to know whether distinctions between déjà vu and déjà vécu will be clinically useful or even be observed in clinical cases. It remains a priority to take this classification, a priori, into cases of people with déjà experiences either spontaneously or under stimulation, to examine its utility. Certainly, we suggest that there is some evidence in the epilepsy literature that déjà vu and déjà vécu are separable experiential phenomenon; a distinction identifiable qualitatively in the context of spontaneous and provoked epileptic activity. Moreover, the differences in their phenomenology are attributable to the functionally independent neural substrates of memory to which their genesis relies upon. If nothing else, we call for a tightening up of the terminology around déjà vu and the recognition memory system. For instance, Spatt's [13] account of a parahippocampal system being at fault in déjà vu (which receives support from this review) is summed up thus: “This memory system is responsible for judgments of familiarity. The result is that a momentary perceived scene is given the characteristics of familiarity that normally accompany a conscious recollection.” [13, page 6]. This quote clearly confounds the two processes of familiarity and recollection. A better phrasing might be that the scene is given characteristics of familiarity without complete retrieval.
In terms of brain regions, the work here suggests that parahippocampal areas are involved in the familiarity felt in déjà vu, but, also that the more extensive the network implicated, the more complex and complete the déjà experience is [58]. By this view, we could see that mere familiarity results from activation of a more restricted network but with extra recruitment or after discharge to connected areas, the more recollection-like the experience becomes, and ultimately, partial retrieval may result in uncontrolled visual imagery and emotional content. But this is a view of the neurological picture which does not receive uniform support. Takeda et al. [70] describe a TLE patient with persistent and debilitating déjà experiences, “…Whatever happens to me in daily life, I feel that I have experienced it before. I feel like I'm living again the same life that I have lived before.” (page 196). Subsequent imaging revealed hyperperfusion of the left medial temporal area during blood perfusion single-photon-emission computed tomography (SPECT) and integration of MRI data identified the hyperperfused area to be in the left entorhinal cortex. The patient's persistent déjà vu was assumed to be a result of epileptic activity, and he was subsequently administered 15 mg/day diazepam for three days, which almost completely abolished it. SPECT was carried out again during this period, this time showing no sign of hyperperfusion in the medial temporal area. Takeda et al.'s [70] patient's description of his experience would appear to be consistent with our characterization of déjà vécu. Critically, however, the imaging evidence suggested localized dysfunction in the entorhinal cortex was responsible, which is not in line with our view of the larger hippocampal network involved in more intense and elaborate versions of the experience (recollection-based déjà vécu).
Two other recent case studies found contrasting results. In one, frequent episodes of déjà vu were the result of medial neurocysticercosis localized in the amygdala; following right-sided amygdalectomy sparing the hippocampus, the patient's episodes of déjà experiences ceased [71]. In another, a patient with TEA experienced what he described as “Groundhog Day,” or the reliving of events, following a particularly severe amnestic attack; MRI investigation revealed high signal intensity in the left hippocampus and a PET scan displayed dramatic hyperperfusion in the left MTL [72]. It does seem unlikely that such a complex phenomenological experience can be explained by a single structure, and, certainly, the most informative study would come from a comparison of all available investigation methods (SEEG, imaging) on groups of patients experiencing different degrees of déjà experiences. Unfortunately, however, as we have seen, such patients are uncommon and invasive brain recording methods cannot be used ad hoc for research purposes. Again, this calls for a need for a better characterization and description of déjà experiences (which we have attempted in Table 2).
Table 2.
A preliminary classification of déjà vu, déjà vécu, and involuntary memories.
| Déjà vu | Déjà vécu | Involuntary memories | |
|---|---|---|---|
| Feelings | |||
| Familiarity | • | • | • |
| Prescience | • | ||
| Mental time travel | • | • | |
| Characterisation | “Prior occurrence” | “Prior experience” | “Prior episode” |
| Content | |||
| Emotion | • | • | |
| Visual images | • | ||
| Source or context | • | • | |
| Narrative-like coherence | • | ||
| A defined past | • | ||
| Recall | None | Partial | Complete |
| Time course | Shorter | Longer | — |
Note: we would see that all these experiences should share these same characteristics; they would all be experienced involuntarily and they will all be relatively brief mental phenomena.
Table 2 presents déjà vu as a relatively simple brief experience. It is an illusion merely of prior occurrence. Our definition suggests that it is not a rich evocative experience based on the activation of a network of regions, but merely a glitch in the sensation of familiarity which is decoupled from current processing, thoughts, and goals. Déjà vécu, on the other hand, is a longer experience where the epistemic feeling is one of mental time travel and of partial retrieval of context. In such forms of déjà experience the person may have time to reflect on the phenomena and search for the source of the prior experience, and as they do so, they may have the sensation that the experience is unraveling, or that they actually experienced the present moment before as a dream or imagining. Prescience, the feeling that they know what is about to happen, will commonly be felt. This feeling of what is about to happen actually resonates with recent views of the hippocampal system as part of a remember-imagining system which generates dynamic mental representations from disperse cortical sites for both past and future events [73]. Most critically, a déjà vécu experience, in comparison to a déjà vu experience, will feature emotions (usually negative) and on the whole will feel more intense than a déjà vu experience.
We have also tentatively proposed a third category of experience, stemming from the evidence in this review that déjà experiences appear along a continuum with memory retrieval. In essence, the chief difference between déjà vécu and involuntary memory will be that the subject is certain of a defined prior episode, that is, their experience maps onto a definite event in their past. But there will also be no striking clash of mental evaluations—involuntary memories, whilst possibly being intrusive or distressing, do not produce a decoupling of epistemic feelings and content. It will feel like a memory—and it will contain information which allows it to be experienced as such. These involuntary memories may be either true or false recollections, but they will be bound by a sort of coherence in keeping with the reconstructive nature of personal episodic memories [57, 74].
The classifications in Table 2 have been generated by consideration of the data presented in this review, but also by expectations of the phenomenology and purpose of recollection and familiarity. The definition of déjà vu seems rather basic and unremarkable, but we suggest that this is the experience which is closest to healthy déjà vu—a brief mental state which does not involve the activation of a large neural network. It does resonate with the process of familiarity too—which is seen as a mere assessment of prior occurrence, which if sufficiently strong triggers other processes and a conscious memory search. It is likely, that just as familiarity can be thought of as a trace strength process, so can déjà vu, with stronger and weaker forms. We return to this comparison with the trace strength and dual process debate below. Of course, the value of any such classification system is the extent to which it is used and helps us understand data, which is a priority for future research.
6. Clinical Remarks
As a rule, family doctors, clinical neuropsychologists, and neurologists alike would see epilepsy as the most likely cause of an elevated level of déjà experiences (for an overview of déjà vu in neurology, see [75]). Other causes of the experience to a pathological degree not discussed here include substance abuse [76], depersonalization or derealisation [77, 78], or migraine [79]. We are aware of cases of debilitating déjà vu which seem to derive from clinical levels of anxiety in cognitively intact people, and this is an area of research worthy of further investigation.
For the most part, the déjà experiences in TLE are unremarkable. However, a handful of recent case reports document the occurrence of more debilitating forms of déjà experiences. For example, Cohen et al. [80] report a man with a long history of complex TLE, who after experiencing a concussion following a seizure, began to encounter vivid intrusions of traumatic events. However, investigation of his history and questioning his family revealed that although aspects of these events did occur, the memories he was recalling were not veridical. Moreover, the traumatic and distressing characteristics of these false intrusions were sufficient for a diagnosis of post traumatic stress disorder (PTSD). According to his wife the intrusion of these memories was precipitated by seizures, suggesting that abnormal medial temporal ictal activity created an integration of different sources of information. We return to false memories in TLE below.
Murai and Fukao [81] also report a case where a close relationship was observed between TLE, unusual experiential phenomenon, and psychiatric symptoms. They describe a 23-year-old male TLE patient who experienced déjà vécu like interictal psychotic episodes. Initially, he only reported experiencing déjà vu auras, but this had developed into a reduplicative paramnesia in which he persistently felt like he was reliving an isolated four year period of his life. This constant cycle of interpreting his current world as a replay of past events became so debilitating that the patient had attempted suicide on a number of occasions. The authors suggested that his initial déjà vu experiences were likely to have arisen from mild abnormalities in the mesial temporal familiarity circuit, but chronic epileptiform activity may have resulted in neuroplastic regenerative changes involving the hippocampus and a distributed memory network responsible for recollection. In an attempt to differentiate déjà vécu from hallucinatory memories, we have suggested that involuntary memories contain visual imagery—and it is this that possibly allows the experient to feel that they have retrieved a memory, rather than having a déjà vécu. Déjà vécu may have a sense of pastness and of some prior context and an expectation of what will happen next, but without the generation of material which can corroborate the experience, in this sense recall of information is partial and inaccessible, such as with the ToT.
In sum, within epilepsy, if it were possible to better classify déjà experiences, we think that it would give some indication of whether familiarity or recollective networks were involved in the epileptic activity. Certainly, déjà vécu, prescience and emotional states are, we think, indicative of a larger network being affected by epileptic changes. The clinical examination of consciousness is a contentious one (see [82]), but possibly the forms of consciousness represented in familiarity and recollection are somewhat better specified and more extensively researched than others, and offer an opportunity to begin to classify the dreamy state further. This surely would have clinical repercussions in understanding epileptic loci and the impact certain forms of epilepsy have on subjective experience.
7. Conclusions
We started this paper with the aim of examining the familiarity processes in TLE, and how they might be behind the déjà experiences in this group. We took as our starting point the contemporary definition of déjà vu which hinges upon familiarity, and drew upon theoretical mechanisms of déjà vu in healthy groups. An examination of the literature suggests that chronic RWI is not behind déjà experiences in TLE. There are several strands to this argument. First we have observed that déjà vu in TLE does not appear to be data driven, but persists whatever stimulus is being attended to [42]. Second, seizure and nonseizure-related forms of déjà experiences can be differentiated by TLE patients [34]. This suggests that there is seizure-like cause of the phenomenon, and a more “normal” form. Third, we might expect that if a recognition failure is behind déjà vu, then those people who had déjà vu should have poorer memories, which is not generally observed [36]. Finally, our own clinical experience is that people with TLE have déjà vu in objectively novel locations and experiences. The RWI account requires only for a similarity between a previous event and a subsequent experience which goes undetected. In very novel situations with little or no overlap with any prior experience, we would not expect déjà vu to occur, but our own experience is that this is just when déjà vu is most likely to be experienced. That said, RWI experiments are relatively straight forward, and it is a research priority to examine the neural substrates of such effects in comparison to déjà vu, and to test groups of TLE people who do and do not experience déjà vu on RWI tasks.
Our alternative account is the decoupled familiarity hypothesis, where familiarity is experienced in a way that is at odds with the outputs of current processing. By our account, in a déjà vu experience, familiarity is free-floating and unrelated to the sensed environment, and not triggered by the environment—it is a random top-down brain event. This is an idea which receives some support from parahippocampal hypometabolism findings, structural damage and cortical stimulation in TLE. Spatt [13] produced a similar parahippocampal account of déjà vu, but his account emphasised the role of the familiarity system in pattern matching. According to the pattern-matching account, déjà vu arises because a present experience is erroneously matched to a prior experience. The limitation of this erroneous pattern matching account is that it fails to explain the clash in evaluations experienced in déjà vu. One aspect of the pattern matching account is that it should also generate false memories, on the occasions when the false match is very strong. However, our impression is that false memory syndromes are rare in TLE, and, although this issue remains ripe for further research, it seems that déjà vu is not related to the generation of false memories and confabulation (but see the single case reported in [80]).
We also started this paper with the aim of elucidating the neural mechanisms underlying déjà vu in TLE. We find that there is generally a large number of brain regions responsible for the generation of déjà vu in epilepsy. Many studies converge on parahippocampal regions, but as the field advances, larger networks of regions are being identified [61]. Our view is that there is imprecision in the neural regions described as a direct consequence of a lack of precision in the definitions of déjà experiences. Along with other authors, we suggest that déjà vu-déjà vécu, and retrieval of fragmentary images and emotions probably lie along a continuum. A research priority would be to begin to use common definitions and examples across laboratories and participants. Because so much research outside the déjà vu literature differentiates experiences of recollection and familiarity, we suggest that this is a good place to start. We also propose that the field begins to consider déjà vu-like fragmentary images retrieved unintentionally in TLE as involuntary memories [83, 84]. Table 2 gives an overview of the differences between these states, and could form the beginnings of definitions for use by others. In sum, in finding some support for our decoupled familiarity hypothesis, it makes sense from a memory theory point of view that decoupled recollection should occur too—hence our theory of déjà vécu.
Our final concluding remark considers whether déjà vu experiences in TLE might illuminate the processes at work in “healthy” déjà vu. We have previously criticised studies using RWI (e.g., [3]) on the grounds that laboratory memory experiments may be contaminated by social desirability in the levels of déjà vu reporting, and that the sensations produced are insipid versions of what occurs in daily life [9]. On the other hand, using TLE to understand déjà vu has been criticised on the grounds that healthy déjà vu is unlikely to derive from seizure-like activity (even though there seem to be definite physiological triggers for déjà vu, such as stress or fatigue [1]). In support of the difference between healthy and epileptic déjà vu are the reports which show people with TLE can differentiate between seizure related and “healthy” déjà vu. Like forms of the déjà experience itself, there may be a continuum of experience between healthy and TLE experiences, and again, the true nature of these experiences may never be elucidated without hypotheses based on neuroscientific models and a consensus amongst researchers regarding definitions. Certainly, the classification of déjà vu we have attempted here will be of use to déjà experiences of any aetiology.
Déjà experiences in epilepsy do reveal something about the working of the brain. Within epilepsy, restricted activation of memory networks used for memory retrieval on a physiological level results in peculiar experiential phenomena-restricted (or artificially isolated) activation of the familiarity network leads to déjà vu, and activation of a wider network, again, restricted insofar as it does not lead to complete retrieval, leads to déjà vécu. Thus, déjà experiences expose the nature of the circuitry and the epistemic feelings associated with retrieval processes. For future work, it would be interesting to consider neuromodulation in déjà experiences, since it appears there can be pharmacological causes which lead to déjà vu [69, 76]. Similarly, a number of authors have pinpointed specific neuro-oscillations in the theta range as being critical in the activation of déjà experiences [61, 63].
On a more theoretical level, the nature of the networks involved in déjà vu and déjà vécu echo those supposedly for the separate memory experiences (and processes) of familiarity and recollection. In fact, there is debate as to whether familiarity and recollection lie along a continuum (e.g., [85, 86]) or represent two distinct processes (e.g., [87, 88]). The most recent work by Bartolomei et al. [61] suggests a network account of déjà vu, which is similar to the idea that familiarity and recollection lie on a unidimensional scale, with their differences being captured in the notion of trace strength. According to single-process accounts, high-strength memory traces can be retrieved with the sensation of recollection (or at least “high confidence”), whereas a low-strength memory traces elicit mere familiarity with the cue. Chauvel et al.'s studies seem to address this issue from a direct electrophysiological viewpoint. The “trace strength” in these experiments could be thought of literally, as the level of current applied to the cortex. Their results suggest that the larger the initial stimulation the more extensive the discharge to surrounding regions (e.g., [57, 58]). This leads to a more nuanced view of whether human recognition memory relies on dual or single processes—trace strength at one specific point predicts the extent of activation to more dispersed and specialized circuits in concert with the original stimulated point. Thus, the coactivation and synchronization of brain areas seen in after discharges leading to déjà vécu and involuntary memories may represent an account of how a putatively single trace can recruit multiple brain regions and mental representations to produce recollection, which by our view, would be anatomically and procedurally separate from the sensation of familiarity. Artificially induced déjà experiences in epileptic patients seem an ideal test of single-versus dual-process accounts of recognition memory in humans.
References
- 1.Brown AS. A review of the Déjà Vu experience. Psychological Bulletin. 2003;129(3):394–413. doi: 10.1037/0033-2909.129.3.394. [DOI] [PubMed] [Google Scholar]
- 2.Brown AS, Marsh EJ. Evoking false beliefs about autobiographical experience. Psychonomic Bulletin and Review. 2008;15(1):186–190. doi: 10.3758/pbr.15.1.186. [DOI] [PubMed] [Google Scholar]
- 3.Cleary AM. Recognition memory, familiarity, and déjà vu experiences. Current Directions in Psychological Science. 2008;17(5):353–357. [Google Scholar]
- 4.Cleary AM, Ryals AJ, Nomi JS. Can déjà vu result from similarity to a prior experience? support for the similarity hypothesis of déjà vu. Psychonomic Bulletin and Review. 2009;16(6):1082–1088. doi: 10.3758/PBR.16.6.1082. [DOI] [PubMed] [Google Scholar]
- 5.Hughlings-Jackson J. On a particular variety of epilepsy (“intellectual aura”), one case with symptoms of organic brain disease. Brain. 1888;11(2):179–207. [Google Scholar]
- 6.Hughlings-Jackson J. On right or left-sided spasm at the onset of epileptic paroxysms, and on crude sensation warnings, and elaborate mental states. Brain. 1880;3(2):192–206. [Google Scholar]
- 7.Neppe VM. The incidence of déjà vu. Parapsychological Journal of South Africa. 1983;4:94–106. [Google Scholar]
- 8.Gloor P. Experiential phenomena of temporal lobe epilepsy. Facts and hypotheses. Brain. 1990;113(6):1673–1694. doi: 10.1093/brain/113.6.1673. [DOI] [PubMed] [Google Scholar]
- 9.O’Connor AR, Moulin CJA. Recognition without identification, erroneous familiarity, and déjà vu. Current Psychiatry Reports. 2010;12(3):165–173. doi: 10.1007/s11920-010-0119-5. [DOI] [PubMed] [Google Scholar]
- 10.Arango-Muñoz S. Two levels of metacognition. Philosophia. 2011;39(1):71–82. [Google Scholar]
- 11.Metcalfe J, Schwartz BL, Joaquim SG. The cue-familiarity heuristic in metacognition. Journal of Experimental Psychology. 1993;19(4):851–861. doi: 10.1037//0278-7393.19.4.851. [DOI] [PubMed] [Google Scholar]
- 12.Tulving E, Markowitsch HJ, Craik FIM, Habib R, Houle. S. Novelty and familiarity activations in PET studies of memory encoding and retrieval. Cerebral Cortex. 1996;6:71–79. doi: 10.1093/cercor/6.1.71. [DOI] [PubMed] [Google Scholar]
- 13.Spatt J. Déjà vu: possible parahippocampal mechanisms. Journal of Neuropsychiatry and Clinical Neurosciences. 2002;14(1):6–10. doi: 10.1176/jnp.14.1.6. [DOI] [PubMed] [Google Scholar]
- 14.Schacter DL, Dodson CS. Misattribution, false recognition and the sins of memory. Philosophical Transactions of the Royal Society B. 2001;356(1413):1385–1393. doi: 10.1098/rstb.2001.0938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jacoby LL, Whitehouse K. An Illusion of memory: false recognition influenced by unconscious perception. Journal of Experimental Psychology. 1989;118(2):126–135. [Google Scholar]
- 16.Schwartz BL. Sparkling at the end of the tongue: the etiology of tip-of-the-tongue phenomenology. Psychonomic Bulletin and Review. 1999;6(3):379–393. doi: 10.3758/bf03210827. [DOI] [PubMed] [Google Scholar]
- 17.Cleary AM, Reyes NL. Scene recognition without identification. Acta Psychologica. 2009;131(1):53–62. doi: 10.1016/j.actpsy.2009.02.006. [DOI] [PubMed] [Google Scholar]
- 18.Knight EF. Where Three Empires Meet: A Narrative of Recent Travel in Kashmir, Western Tibet, Gilgit and the Adjoining Countries. London, UK: Longmans, Green & Co.; 1895. [Google Scholar]
- 19.Mandler G. Familiarity breeds attempts. Perspectives on Psychological Sciences. 2008;3:390–399. doi: 10.1111/j.1745-6924.2008.00087.x. [DOI] [PubMed] [Google Scholar]
- 20.Yonelinas AP. The nature of recollection and familiarity: a review of 30 years of research. Journal of Memory and Language. 2002;46(3):441–517. [Google Scholar]
- 21.Eichenbaum H, Yonelinas AP, Ranganath C. The medial temporal lobe and recognition memory. Annual Review of Neuroscience. 2007;30:123–152. doi: 10.1146/annurev.neuro.30.051606.094328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Henson RNA, Rugg MD, Shallice T, Josephs O, Dolan RJ. Recollection and familiarity in recognition memory: an event-related functional magnetic resonance imaging study. Journal of Neuroscience. 1999;19(10):3962–3972. doi: 10.1523/JNEUROSCI.19-10-03962.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bird CM, Shallice T, Cipolotti L. Fractionation of memory in medial temporal lobe amnesia. Neuropsychologia. 2007;45(6):1160–1171. doi: 10.1016/j.neuropsychologia.2006.10.011. [DOI] [PubMed] [Google Scholar]
- 24.Blaxton TA, Theodore WH. The role of the temporal lobes in recognizing visuospatial materials: remembering versus knowing. Brain and Cognition. 1997;35(1):5–25. doi: 10.1006/brcg.1997.0902. [DOI] [PubMed] [Google Scholar]
- 25.Cohn M, McAndrews MP, Moscovitch M. Associative reinstatement: a novel approach to assessing associative memory in patients with unilateral temporal lobe excisions. Neuropsychologia. 2009;47(13):2989–2994. doi: 10.1016/j.neuropsychologia.2009.06.029. [DOI] [PubMed] [Google Scholar]
- 26.Moran M, Seidenberg M, Sabsevitz D, Swanson S, Hermann B. The acquisition of face and person identity information following anterior temporal lobectomy. Journal of the International Neuropsychological Society. 2005;11(3):237–248. doi: 10.1017/S1355617705050290. [DOI] [PubMed] [Google Scholar]
- 27.Moscovitch DA, McAndrews MP. Material-specific deficits in “remembering” in patients with unilateral temporal lobe epilepsy and excisions. Neuropsychologia. 2002;40(8):1335–1342. doi: 10.1016/s0028-3932(01)00213-5. [DOI] [PubMed] [Google Scholar]
- 28.Thaiss L, Petrides M. Source versus content memory in patients with a unilateral frontal cortex or a temporal lobe excision. Brain. 2003;126(5):1112–1126. doi: 10.1093/brain/awg112. [DOI] [PubMed] [Google Scholar]
- 29.Illman NA, Rathbone CJ, Kemp S, Moulin CJA. Autobiographical memory and the self in a case of transient epileptic amnesia. Epilepsy and Behavior. 2011;21(1):36–41. doi: 10.1016/j.yebeh.2011.02.022. [DOI] [PubMed] [Google Scholar]
- 30.Dashiell JF. Fundamentals of Objective Psychology. Boston, Mass, USA: Houghton Mifflin Company; 1937. [Google Scholar]
- 31.Leeds M. One form of paramnesia: the illusion of déjà vu. Journal of the American Society for Psychical Research. 1944;38:24–42. [Google Scholar]
- 32.Osborn HF. Illusions of memory. North American Review. 1884;138:476–486. [Google Scholar]
- 33.O’Connor AR, Barnier AJ, Cox RE. Déjà vu in the laboratory: a behavioral and experiential comparison of posthypnotic amnesia and posthypnotic familiarity. International Journal of Clinical and Experimental Hypnosis. 2008;56(4):425–450. doi: 10.1080/00207140802255450. [DOI] [PubMed] [Google Scholar]
- 34.Adachi N, Akanuma N, Ito M, et al. Two forms of déjà vu experiences in patients with epilepsy. Epilepsy and Behavior. 2010;18(3):218–222. doi: 10.1016/j.yebeh.2010.02.016. [DOI] [PubMed] [Google Scholar]
- 35.Adachi N, Adachi T, Kimura M, Akanuma N, Kato M. Development of the Japanese version of the Inventory of Déjà vu Experiences Assessment (IDEA) Seishin Igaku. 2001;43:1223–1231. [Google Scholar]
- 36.Vederman AC, Holtzer R, Zimmerman ME, Devinsky O, Barr WB. Ictal mnemestic aura and verbal memory function. Epilepsy and Behavior. 2010;17(4):474–477. doi: 10.1016/j.yebeh.2010.01.018. [DOI] [PubMed] [Google Scholar]
- 37.Moulin CJA, Buchanan SM, Bradley R, Karadoller DZ. Déjà vu in healthy aging. In press. [Google Scholar]
- 38.Fukao K, Murai T, Yamada M, Sengoku A, Kusumi T. Déjà Vu and Jamais Vu as Ictal Symptoms: qualitative comparison with those occurring in normal subjects using a questionnaire. Epilepsia. 2005;46:28–29. [Google Scholar]
- 39.Johanson M, Valli K, Revonsuo A, Wedlund JE. Content analysis of subjective experiences in partial epileptic seizures. Epilepsy and Behavior. 2008;12(1):170–182. doi: 10.1016/j.yebeh.2007.10.002. [DOI] [PubMed] [Google Scholar]
- 40.Sadler RM, Rahey S. Prescience as an aura of temporal lobe epilepsy. Epilepsia. 2004;45(8):982–984. doi: 10.1111/j.0013-9580.2004.68603.x. [DOI] [PubMed] [Google Scholar]
- 41.Sengoku A, Toichi M, Murai T. Dreamy states and psychoses in temporal lobe epilepsy: mediating role of affect. Psychiatry and Clinical Neurosciences. 1997;51(1):23–26. doi: 10.1111/j.1440-1819.1997.tb02361.x. [DOI] [PubMed] [Google Scholar]
- 42.O’Connor AR, Moulin CJA. The persistence of erroneous familiarity in an epileptic male: challenging perceptual theories of déjà vu activation. Brain and Cognition. 2008;68(2):144–147. doi: 10.1016/j.bandc.2008.03.007. [DOI] [PubMed] [Google Scholar]
- 43.Lee EM, Im KC, Kim JH, et al. Relationship between hypometabolic patterns and ictal scalp EEG patterns in patients with unilateral hippocampal sclerosis: an FDG-PET study. Epilepsy Research. 2009;84(2-3):187–193. doi: 10.1016/j.eplepsyres.2009.02.005. [DOI] [PubMed] [Google Scholar]
- 44.Adachi N, Koutroumanidis M, Elwes RDC, et al. Interictal 18FDG PET findings in temporal lobe epilepsy with deja vu. Journal of Neuropsychiatry and Clinical Neurosciences. 1999;11(3):380–386. doi: 10.1176/jnp.11.3.380. [DOI] [PubMed] [Google Scholar]
- 45.Guedj E, Aubert S, McGonigal A, Mundler O, Bartolomei F. Déjà-vu in temporal lobe epilepsy: metabolic pattern of cortical involvement in patients with normal brain MRI. Neuropsychologia. 2010;48(7):2174–2181. doi: 10.1016/j.neuropsychologia.2010.04.009. [DOI] [PubMed] [Google Scholar]
- 46.Brown MW, Aggleton JP. Recognition memory: what are the roles of the perirhinal cortex and hippocampus? Nature Reviews Neuroscience. 2001;2(1):51–61. doi: 10.1038/35049064. [DOI] [PubMed] [Google Scholar]
- 47.Knowlton RC, Laxer KD, Klein G, et al. In vivo hippocampal glucose metabolism in mesial temporal lobe epilepsy. Neurology. 2001;57(7):1184–1190. doi: 10.1212/wnl.57.7.1184. [DOI] [PubMed] [Google Scholar]
- 48.Van Paesschen W, King MD, Duncan JS, Connelly A. The amygdala and temporal lobe simple partial seizures: a prospective and quantitative MRI study. Epilepsia. 2001;42(7):857–862. doi: 10.1046/j.1528-1157.2001.042007857.x. [DOI] [PubMed] [Google Scholar]
- 49.Hughlings-Jackson J. On temporary paralysis after epileptiform and epileptic seizures; a contribution to the study of dissolution of the nervous system. Brain. 1881;3(4):433–451. [Google Scholar]
- 50.Mullan S, Penfield W. Illusions of comparative interpretation and emotion: production by epileptic discharge and by electrical stimulation in the temporal cortex. AMA Archives of Neurology and Psychiatry. 1959;81:269–284. [PubMed] [Google Scholar]
- 51.Penfield W, Perot P. The brain’s record of auditory and visual experience: a final summary and discussion. Brain. 1963;86(4):595–696. doi: 10.1093/brain/86.4.595. [DOI] [PubMed] [Google Scholar]
- 52.Halgren E, Walter RD, Cherlow DG, Crandall PH. Mental phenomena evoked by electrical stimulation of the human hippocampal formation and amygdala. Brain. 1978;101(1):83–117. doi: 10.1093/brain/101.1.83. [DOI] [PubMed] [Google Scholar]
- 53.Gloor P, Olivier A, Quesney LF. The role of the limbic system in experimental phenomena of temporal lobe epilepsy. Annals of Neurology. 1982;12(2):129–144. doi: 10.1002/ana.410120203. [DOI] [PubMed] [Google Scholar]
- 54.Gloor P. Experiential phenomena of temporal lobe epilepsy. Facts and hypotheses. Brain. 1990;113(6):1673–1694. doi: 10.1093/brain/113.6.1673. [DOI] [PubMed] [Google Scholar]
- 55.Bancaud J, Brunet-Bourgin F, Chauvel P, Halgren E. Anatomical origin of deja vu and vivid “memories” in human temporal lobe epilepsy. Brain. 1994;117(1):71–90. doi: 10.1093/brain/117.1.71. [DOI] [PubMed] [Google Scholar]
- 56.Bartolomei F, Barbeau E, Gavaret M, et al. Cortical stimulation study of the role of rhinal cortex in déjà vu and reminiscence of memories. Neurology. 2004;63(5):858–864. doi: 10.1212/01.wnl.0000137037.56916.3f. [DOI] [PubMed] [Google Scholar]
- 57.Barbeau E, Wendling F, Régis J, et al. Recollection of vivid memories after perirhinal region stimulations: synchronization in the theta range of spatially distributed brain areas. Neuropsychologia. 2005;43(9):1329–1337. doi: 10.1016/j.neuropsychologia.2004.11.025. [DOI] [PubMed] [Google Scholar]
- 58.Vignal JP, Maillard L, McGonigal A, Chauvel P. The dreamy state: hallucinations of autobiographic memory evoked by temporal lobe stimulations and seizures. Brain. 2007;130(1):88–99. doi: 10.1093/brain/awl329. [DOI] [PubMed] [Google Scholar]
- 59.Milton F, Butler CR, Zeman AZJ. Transient epileptic amnesia: déjà vu heralding recovery of lost memories. Journal of Neurology, Neurosurgery and Psychiatry. 2011;82(10):1178–1179. doi: 10.1136/jnnp.2009.200147. [DOI] [PubMed] [Google Scholar]
- 60.Butler CR, Graham KS, Hodges JR, Kapur N, Wardlaw JM, Zeman AZJ. The syndrome of transient epileptic amnesia. Annals of Neurology. 2007;61(6):587–598. doi: 10.1002/ana.21111. [DOI] [PubMed] [Google Scholar]
- 61.Bartolomei F, Barbeau EJ, Nguyen T, et al. Rhinal-hippocampal interactions during déjà vu. Clinical Neurophysiology. 2012;123(3):489–495. doi: 10.1016/j.clinph.2011.08.012. [DOI] [PubMed] [Google Scholar]
- 62.Mormann F, Osterhage H, Andrzejak RG, et al. Independent delta/theta rhythms in the human hippocampus, entorhinalcortex. Frontiers in Human Neuroscience. 2008 ;2(3) doi: 10.3389/neuro.09.003.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.O’Connor AR, Lever C, Moulin CJA. Novel insights into false recollection: a model of déjà vécu. Cognitive Neuropsychiatry. 2010;15(1–3):118–144. doi: 10.1080/13546800903113071. [DOI] [PubMed] [Google Scholar]
- 64.Illman NA, Moulin CJA, O’Connor AR, Chauvel P. Déjà experiences in Epilepsy: contributions from memory research. In: Zeman A, et al., editors. Memory and Epilepsy—The State of the Art. Oxford University Press; [Google Scholar]
- 65.Funkhouser A. Three Types of Déjà vu. Mental Science Network. 1995;57:20–22. [Google Scholar]
- 66.Dickens C. Three types of Déjà vu. Mental Science Network. 1850;57:20–22. [Google Scholar]
- 67.Conway MA. Memory and the self. Journal of Memory and Language. 2005;53(4):594–628. [Google Scholar]
- 68.Moulin CJA, Conway MA, Thompson RG, James N, Jones RW. Disordered memory awareness: recollective confabulation in two cases of persistent déjà vecu. Neuropsychologia. 2005;43(9):1362–1378. doi: 10.1016/j.neuropsychologia.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 69.Kalra S, Chancellor A, Zeman A. Recurring déjà vu associated with 5-hydroxytryptophan. Acta Neuropsychiatrica. 2007;19(5):311–313. doi: 10.1111/j.1601-5215.2007.00245.x. [DOI] [PubMed] [Google Scholar]
- 70.Takeda Y, Kurita T, Sakurai K, Shiga T, Tamaki N, Koyama T. Persistent déjà vu associated with hyperperfusion in the entorhinal cortex. Epilepsy and Behavior. 2011;21(2):196–199. doi: 10.1016/j.yebeh.2011.03.031. [DOI] [PubMed] [Google Scholar]
- 71.Lee DJ, Owen CM, Khanifar E, Kim RC, Binder DK. Isolated amygdala neurocysticercosis in a patient presenting with déjà vu and olfactory auras: case report. Journal of Neurosurgery. 2009;3(6):538–541. doi: 10.3171/2009.2.PEDS08140. [DOI] [PubMed] [Google Scholar]
- 72.Butler CR, Zeman A. A case of transient epileptic amnesia with radiological localization. Nature Clinical Practice Neurology. 2008;4(9):516–521. doi: 10.1038/ncpneuro0857. [DOI] [PubMed] [Google Scholar]
- 73.Addis DR, Wong AT, Schacter DL. Remembering the past and imagining the future: common and distinct neural substrates during event construction and elaboration. Neuropsychologia. 2007;45(7):1363–1377. doi: 10.1016/j.neuropsychologia.2006.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Conway MA, Singer JA, Tagini A. The self and autobiographical memory: correspondence and coherence. Social Cognition. 2004;22(5):491–529. [Google Scholar]
- 75. Wild E. Deja Vu in Neurology. Journal of Neurology. 2005;252:1–7. doi: 10.1007/s00415-005-0677-3. [DOI] [PubMed] [Google Scholar]
- 76.Singh S. Adolescent salvia substance abuse. Addiction. 2007;102(5):823–824. doi: 10.1111/j.1360-0443.2007.01810.x. [DOI] [PubMed] [Google Scholar]
- 77.Harper MA. Déjà vu and depersonalisation in normal subjects. Australian and New Zealand Journal of Psychiatry. 1969;3:67–74. [Google Scholar]
- 78.Sierra M, Berrios GE. The Cambridge Depersonalisation Scale: a new instrument for the measurement of depersonalisation. Psychiatry Research. 2000;93(2):153–164. doi: 10.1016/s0165-1781(00)00100-1. [DOI] [PubMed] [Google Scholar]
- 79.Sacks O. Migraine. London, UK: Faber and Faber; 1970 . [Google Scholar]
- 80.Cohen ML, Rozensky RH, Zlatar ZZ, Averbuch RN, Cibula JE. Posttraumatic stress disorder caused by the misattribution of seizure-related experiential responses. Epilepsy and Behavior. 2010;19(4):652–655. doi: 10.1016/j.yebeh.2010.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Murai T, Fukao K. Paramnesic multiplication of autobiographical memory as a manifestation of interictal psychosis. Psychopathology. 2003;36(1):49–51. doi: 10.1159/000069655. [DOI] [PubMed] [Google Scholar]
- 82.Gloor P. Consciousness as a neurological concept in epileptology: a critical review. Epilepsia. 1986;27(2):S14–S26. doi: 10.1111/j.1528-1157.1986.tb05737.x. [DOI] [PubMed] [Google Scholar]
- 83.Rasmussen AS, Berntsen D. The unpredictable past: spontaneous autobiographical memories outnumber autobiographical memories retrieved strategically. Consciousness and Cognition. 2011;20(4):1842–1846. doi: 10.1016/j.concog.2011.07.010. [DOI] [PubMed] [Google Scholar]
- 84.Berntsen D. Involuntary autobiographical memories. Applied Cognitive Psychology. 1996;10:435–454. [Google Scholar]
- 85.Rotello CM, Macmillan NA, Reeder JA, Wong M. The remember response: subject to bias, graded, and not a process-pure indicator of recollection. Psychonomic Bulletin and Review. 2005;12(5):865–873. doi: 10.3758/bf03196778. [DOI] [PubMed] [Google Scholar]
- 86.Dunn JC. Remember-Know: a matter of confidence. Psychological Review. 2004;111(2):524–542. doi: 10.1037/0033-295X.111.2.524. [DOI] [PubMed] [Google Scholar]
- 87.Diana RA, Reder LM, Arndt J, Park H. Models of recognition: a review of arguments in favor of a dual-process account. Psychonomic Bulletin and Review. 2006;13(1):1–21. doi: 10.3758/bf03193807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Yonelinas AP. Receiver-operating characteristics in recognition memory: evidence for a dual-process model. Journal of Experimental Psychology. 1994;20(6):1341–1354. doi: 10.1037//0278-7393.20.6.1341. [DOI] [PubMed] [Google Scholar]


