Abstract
Regulation of cytosolic Ca2+ concentration ([Ca2+]cyt) in airway smooth muscle (ASM) is a key aspect of airway contractility and can be modulated by inflammation. Mitochondria have tremendous potential for buffering [Ca2+]cyt, helping prevent Ca2+ overload, and modulating other intracellular events. Here, compartmentalization of mitochondria to different cellular regions may subserve different roles. In the present study, we examined the role of Ca2+ buffering by mitochondria and mitochondrial Ca2+ transport mechanisms in the regulation of [Ca2+]cyt in enzymatically dissociated human ASM cells upon exposure to the proinflammatory cytokines TNF-α and IL-13. Cells were loaded simultaneously with fluo-3 AM and rhod-2 AM, and [Ca2+]cyt and mitochondrial Ca2+ concentration ([Ca2+]mito) were measured, respectively, using real-time two-color fluorescence microscopy in both the perinuclear and distal, perimembranous regions of cells. Histamine induced a rapid increase in both [Ca2+]cyt and [Ca2+]mito, with a significant delay in the mitochondrial response. Inhibition of the mitochondrial Na+/Ca2+ exchanger (1 μM CGP-37157) increased [Ca2+]mito responses in perinuclear mitochondria but not distal mitochondria. Inhibition of the mitochondrial uniporter (1 μM Ru360) decreased [Ca2+]mito responses in perinuclear and distal mitochondria. CGP-37157 and Ru360 significantly enhanced histamine-induced [Ca2+]cyt. TNF-α and IL-13 both increased [Ca2+]cyt, which was associated with decreased [Ca2+]mito in the case of TNF-α but not IL-13. The effects of TNF-α on both [Ca2+]cyt and [Ca2+]mito were affected by CGP-37157 but not by Ru360. Overall, these data demonstrate that in human ASM cells, mitochondria buffer [Ca2+]cyt after agonist stimulation and its enhancement by inflammation. The differential regulation of [Ca2+]mito in different parts of ASM cells may serve to locally regulate Ca2+ fluxes from intracellular sources versus the plasma membrane as well as respond to differential energy demands at these sites. We propose that such differential mitochondrial regulation, and its disruption, may play a role in airway hyperreactivity in diseases such as asthma, where [Ca2+]cyt is increased.
Keywords: lung, bronchial smooth muscle, calcium regulation, sarcoplasmic reticulum, inflammation, mitochondrial sodium-calcium exchange, mitochondrial calcium uniporter
mitochondria have been known for several decades to have a role in Ca2+ sequestration (76, 82). Metabolism or ATP production could be enhanced by mitochondrial Ca2+ uptake, and perinuclear mitochondria might be involved in shaping nuclear Ca2+ signals (9, 10, 23, 64). Whether mitochondria contribute to regulate cytosolic Ca2+ concentration ([Ca2+]cyt) is subject to debate (10, 48, 64).
The importance of defining the role of mitochondrial Ca2+ buffering in human airway smooth muscle (ASM) lies in understanding the mechanisms by which [Ca2+]cyt is enhanced in airway diseases such as asthma, which contribute to airway hyperresponsiveness. Mitochondrial dysfunction and some ultrastructural changes in mitochondria (swelling and loss of cristae) have been observed with airway inflammation (1, 39). In human ASM, mitochondria are present in large numbers (79), and, interestingly, mitochondrial biogenesis is enhanced in asthma (28). Whether such changes can contribute to altered Ca2+ homeostasis or other features of asthma, such as airway remodeling, is not known.
Mitochondrial Ca2+ uptake during agonist stimulation has been observed in different cell types (13, 25, 33, 38, 48, 55, 56, 65) and is thought to decrease local Ca2+ gradients for sarcoplasmic reticulum (SR)/endoplasmic reticulum Ca2+ refilling while maintaining or enhancing store-operated Ca2+ entry (SOCE) (24, 37, 41, 45, 50, 81). Some theoretical models of Ca2+ signaling in smooth muscle that include mitochondria also suggest a role for modulating [Ca2+]cyt regulatory mechanisms (66, 67). In this regard, mitochondria are often located near the SR and plasma membrane (PM), and this physical proximity may be important (25, 33, 63, 69), especially to buffer locally high levels of Ca2+ during stimulation (27, 53, 56, 57). These studies raise the question of the relation between the spatial localization of mitochondria and their ability to buffer or release Ca2+. Recent work has established that mitochondria contain sophisticated Ca2+ uptake and release mechanisms that may alter [Ca2+]cyt either globally or in the vicinity of the SR and PM (10, 13, 16, 22, 25, 33, 38, 43, 48, 52, 60, 62, 63, 65, 70). There is currently little information on these roles for mitochondria in ASM cells.
Several studies have suggested that airway inflammation increases airway contractility by modulating Ca2+ regulatory mechanisms (2–4, 61, 71, 72, 74, 77, 84). Proinflammatory mediators, including cytokines such as TNF-α and IL-13, increase agonist-induced [Ca2+]cyt responses in ASM cells (74, 75, 84). The role of mitochondria in shaping these signals has not been examined. Furthermore, little is known about the effects of inflammation on mitochondrial buffering of [Ca2+]cyt in human ASM. In other smooth muscle types, inflammatory cytokines disrupt mitochondria-SR interactions and Ca2+ buffering (64, 65). Studies have also shown that short-term exposure to TNF-α temporary increases basal mitochondrial Ca2+ in the lung endothelium, leading to an increase of mitochondrial ROS generation (19, 51, 68). A recent study (13) has suggested that H2O2 may modulate both [Ca2+]mito and [Ca2+]cyt in rat ASM cells. Accordingly, we hypothesized that mitochondrial Ca2+ transport modulates the [Ca2+]cyt increase induced by agonist stimulation in human ASM cells and that inflammation affects mitochondrial Ca2+ transport. As compartmentalization may play an important role in the Ca2+ buffering capacity of mitochondria, mitochondrial Ca2+ responses were investigated in perinuclear area and distal (perimembranous) regions of cells. The role of mitochondria transport was examined using inhibitors of the mitochondrial uniporter (MCU) (7, 21, 30–32) and mitochondrial Na+/Ca2+ exchanger (NCX) (22, 47, 56, 83) and by simultaneously monitoring [Ca2+]cyt and mitochondrial Ca2+ concentration ([Ca2+]mito) in the presence of TNF-α or IL-13.
MATERIALS AND METHODS
Isolation of human ASM cells.
These techniques have been previously described (58, 78). Briefly, ASM cells were enzymatically dissociated from third- to sixth-generation bronchi of lung samples incidental to patient surgery at Mayo Clinic (approved and considered exempt by the Institutional Review Board of Mayo Clinic). Normal lung areas were identified, and dissected tissues were placed in ice-cold HBSS (Invitrogen, Carlsbad, CA) supplemented with 10 mM HEPES and 2 mM Ca2+. Under light microscopy, bronchioles were freed of cartilage, the epithelium, and surrounding tissues, and ASM tissue was dissected out. Samples were minced, and cells were dissociated using papain and collagenase with ovomucoid/albumin separation as per the manufacturer's instructions (Worthington Biochemical, Lakewood, NJ). Cell pellets were resuspended and plated in sterile culture flasks or eight-well glass-bottomed Lab-Tek chambers (Nalge Nunc, Rochester, NY). Cells were maintained at 37°C (5% CO2-95% air) using phenol red-free DMEM-F-12 medium (Invitrogen) supplemented with 10% FBS until ∼80% confluent. Before experiments, cells were washed in PBC (Invitrogen), and medium was changed to DMEM-F-12 lacking serum for 48 h. All experiments were performed in cells from passages 1–3 of subculture. Periodic assessment of the ASM phenotype (smooth muscle actin and myosin and agonist receptors as well as lack of fibroblast markers) was performed. Cell viability was tested by the exclusion of trypan blue.
Real-time fluorescence imaging.
ASM cells plated on eight-well Lab-Tek chambers were incubated in 2.5 μM fluo-3 AM (Invitrogen, [Ca2+]cyt indicator, excitation: 488 nm and emission: 510 nm) with 2.5 μM rhod-2 AM (Invitrogen, [Ca2+]mito indicator, excitation: 568 nm and emission: 590 nm) for 40 min at room temperature followed by an extensive wash in dye-free solution. In control experiments, ASM cells were loaded with either fluo-3 AM or rhod-2 AM. In other experiments, cells were loaded with 2.5 μM rhod-2 AM for 40 min and then with 500 nM MitoTracker green (Invitrogen) for 5 min at room temperature to verify that rhod-2 was present specifically in mitochondria. To simultaneously visualize the SR and mitochondria, ASM cells were loaded with 1 μM BODIPY-FL-thapsigargin (endoplasmic reticulum Ca2+ pumps, Invitrogen) for 30 min and then with 500 nM MitoTracker red CMXRos (Invitrogen) for 5 min at room temperature.
The techniques for [Ca2+]cyt imaging of human ASM cells have been previously described (58, 73, 78). Cells were visualized using an epifluorescence imaging system (MetaFluor, Universal Imaging, Downingtown, PA) on a Nikon Diaphot inverted microscope (Fryer Instruments, Edina, MN) with a ×40/1.3 numerical aperture oil-immersion lens (or a ×60/1.3 numerical aperture oil-immersion lens) and a 12-bit Photometric Cascade digital camera system (Roper Scientific, Tucson, AZ). Images were collected at 1 Hz. Cells were initially perfused with 2 mM Ca2+ HBSS, and baseline fluorescence was established. Changes in fluorescence were analyzed by selecting individual software-defined regions of interest (ROIs; 3 × 3 pixels) delineating mitochondrial versus cytoplasmic areas.
Calibrations.
We performed empirical calibrations of fluorescence values for fluo-3 AM and rhod-2 AM to be able to compare [Ca2+]cyt and [Ca2+]mito. Although these are not ratiometric dyes, Ca2+ can be estimated by essentially mapping fluorescence intensity to known Ca2+ levels. Calibrations were performed with ASM cells loaded with either fluo-3 AM or rhod-2 AM (2.5 μM) perfused successively with 11 K2EGTA/CaEGTA buffers (free Ca2+ ranging from 0 to 39 μM, Ca2+ calibration buffer kit no. 1, Invitrogen). Gray-level values of fluorescence intensities at various settings used with the epifluorescence imaging system and for each Ca2+ concentration buffer were converted to a family of calibration curves as previously described (54, 59, 80).
Statistical analysis.
Four bronchial samples were used to obtain ASM cells. [Ca2+]cyt responses of ∼6–8 cells/visual field and [Ca2+]mito responses of at least 40 mitochondria/cell were measured, and all experiments were repeated at least 3 times for each sample obtained; n denotes the number of cells that were analyzed. In experiments where Ca2+ responses were compared in the presence or absence of specific drugs, a paired t-test was used, whereas population studies were compared using an unpaired t-test or one-way ANOVA with repeated measures as appropriate. A Bonferroni correction was applied for multiple comparisons. Statistical significance was established at P < 0.05. All values are expressed as means ± SE.
RESULTS
Dye loading of ASM cells.
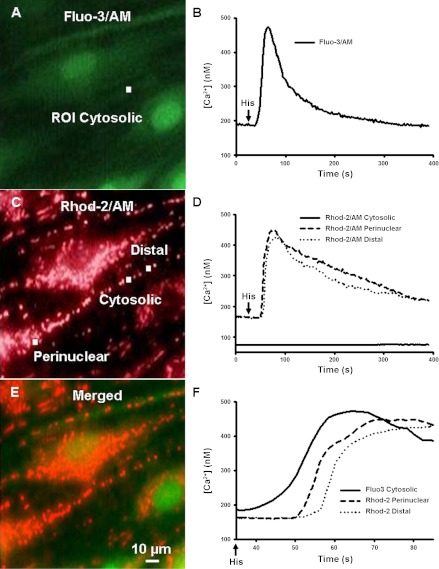
To simultaneously measure [Ca2+]cyt and [Ca2+]mito, human ASM cells were loaded with fluo-3 AM and rhod-2 AM (Fig. 1). The distribution of fluo-3 AM was diffuse, whereas rhod-2 AM was localized to punctate areas typical of mitochondria. Ca2+ responses were measured in cells that exhibited a fusiform shape with a centrally located nucleus typical of healthy human ASM cells and showing homogenous loading with fluo-3 AM and punctate loading of rhod-2 AM (Fig. 1, A–C). In some experiments, cells were loaded with rhod-2 and MitoTracker green to confirm that the pattern of rhod-2 staining was restricted to mitochondria (Fig. 1, D–F). In a second set of experiments, ASM cells were loaded with BODIPY-FL-thapsigargin and then MitoTracker red CMXRos (Fig. 1, G–I). The pattern of MitoTracker red CMXRos staining was similar to that of rhod-2 and MitoTracker green (not shown). We observed that mitochondria are present in large numbers within human ASM cells, primarily around the nucleus, where they form a dense interconnected network. The SR Ca2+ pumps visualized with BODIPY-FL-thapsigargin appeared to be highly localized around the nucleus (Fig. 1G), where a dense network of mitochondria was also observed (Fig. 1, H and I). BODIPY-FL-thapsigargin fluorescence was far less intense elsewhere within the cells. Image analysis of 10 ASM cells revealed that the network of mitochondria juxtaposed with the SR Ca2+ pumps was within 40 μm of the centroid of the nucleus. These mitochondria were defined as perinuclear for subsequent analyses. Mitochondria located >50 μm from the centroid of the nucleus and within 10 μm of the cell membrane were defined as distal for subsequent analyses.
Fig. 1.
Dye loading of human airway smooth muscle (ASM) cells. A and B: fluorescence images of human ASM cells loaded with 2.5 μM fluo-3 AM (A) and 2.5 μM rhod-2 AM (B) to estimate cytosolic ([Ca2+]cyt) and mitochondrial Ca2+ concentration ([Ca2+]mito), respectively. C: relative distribution of fluo-3 AM and rhod-2 AM fluorescence with the merged image of the two fluorescence images. D and E: in a separate set of experiments, human ASM cells were loaded simultaneously with rhod-2 AM (D) and then with 500 nM MitoTracker green (E) for 5 min to visualize mitochondria and showed consistent and matching patterns of mitochondria in the perinuclear areas and more distal (potentially perimembranous) areas of cells. F: an overlay of the two fluorescence images confirmed that the pattern of rhod-2 staining was mainly within mitochondria. G and H: in additional experiments, ASM cells were loaded with 1 μM BODIPY-FL-thapsigargin (G; endoplasmic reticulum Ca2+ pumps) and then with 500 nM MitoTracker red CMXRos (H). I: overlays of the two fluorescence images along with image analysis of the mitochondrial network were used to define the perinuclear regions. N, nucleus. Bars = 10 μm.
As shown in Fig. 1, B and E, some weaker rhod-2 AM fluorescence could be observed in the cytosol. Therefore, it was important to verify that the rhod-2 readings within the ROIs did not reflect cytosolic Ca2+. ASM cells were loaded with MitoTracker green and rhod-2 AM (Fig. 2, A–C), and ROIs (3 × 3 pixels) used to measure change in rhod-2 AM fluorescence were placed within the mitochondria (identified with MitoTracker green), in the cytosol or near mitochondria where some weaker rhod-2 AM background fluorescence was observed (Fig. 2C). Transient increases in rhod-2 fluorescence induced by 10 μM histamine completely overlapped with regions of MitoTracker green fluorescence (region 1 in Fig. 2C). In ROIs outside of the mitochondria (Mitotracker green fluorescence), histamine did not induce a rhod-2 fluorescence response (e.g., regions 2 and 3 in Fig. 2C). These data confirmed that the responses obtained with rhod-2 AM were specific to change in [Ca2+]mito and not [Ca2+]cyt (Fig. 2D).
Fig. 2.
Transient increases in rhod-2 fluorescence are localized within mitochondria. A and B: fluorescence images of human ASM cells loaded with MitoTracker green (A) and rhod-2 AM (B). C: overlay of the two fluorescence images with examples of regions of interest (ROIs) chosen within the mitochondria (region 1), in the cytosol (region 2), and near mitochondria (region 3), where some background fluorescence could be detected. Bar = 10 μm. D: measurement of rhod-2 fluorescence expressed as [Ca2+] (in nM) in the ROIs shown in C. A transient increase in [Ca2+] in response to histamine (His; 10 μM) was observed for the ROI chosen within the mitochondria (region 1, solid line) and not for the ROIs placed in either the cytosol (region 2, dotted line) or near the mitochondria (region 3, dashed line). E and F: MitoTracker green-loaded cells excited at 488 nm showed significant specific fluorescence at 501 nm, corresponding to MitoTracker green (E), whereas excitation of the cells at 568 nm (rhod-2 AM excitation wavelength) did not show significant (above background) fluorescence at 590 nm (F). G and H: similarly, excitation of ASM cells loaded with 2.5 μM rhod-2 AM at 488 nm (G; Mitotracker green excitation wavelength) did not result in any nonspecific fluorescence at 501 nm (above background), whereas excitation at 568 nm resulted in specific fluorescence at 590 nm, corresponding to rhod-2 AM (H). Ex, excitation; Em, emission.
To confirm the specificity of our fluorescent imaging, ASM cells were loaded with only MitoTracker green or rhod-2 AM. MitoTracker green-loaded cells excited at 488 nm showed specific fluorescence at 501 nm, corresponding to MitoTracker green, whereas excitation at 568 nm (the rhod-2 AM excitation wavelength) did not show significant (above background) specific fluorescence at 590 nm (Fig. 2, E and F). Similarly, the excitation of rhod-2 AM-loaded cells with 488 nm (the Mitotracker green excitation wavelength) did not result in any nonspecific fluorescence at 501 nm (above background; Fig. 2G), whereas excitation at 568 nm resulted in specific fluorescence at 590 nm, corresponding to rhod-2 AM.
In selected experiments, ASM cells were loaded with either fluo-3 AM or rhod-2 AM, and the responses to an agonist obtained for each Ca2+ indicator were compared with those obtained with simultaneous loading of the cells. No differences were found in terms of delays, amplitudes, etc., indicating a lack of interference between the two dyes. The ROIs (3 × 3 pixels) were placed into the cytosol to measure changes in [Ca2+]cyt (Fig. 3, A and B) or were placed at the center of well-delineated mitochondrial groups to measure changes in [Ca2+]mito (Fig. 3, C and D).
Fig. 3.
Measurement of [Ca2+]cyt and [Ca2+]mito in human ASM cells. A: fluorescence image of human ASM cells loaded with fluo-3 AM with an example of the ROI used to measure [Ca2+]cyt. B: 10 μM His first induced a rise in [Ca2+]cyt. C: the same ROI was used to measure the change in [Ca2+]mito with the same cells simultaneously loaded with rhod-2 AM. Examples of the ROIs used to measure [Ca2+]mito in the perinuclear and distal regions are also shown. D: traces obtained from the ROIs in C showed that the changes in fluorescence only occur when the ROIs are placed within the mitochondria in either the distal (dotted line) or perinuclear (dashed line) region of the cells. E and F: an overlay of the two fluorescence images (E) along with a time-extended view of [Ca2+] responses (F) from the graphs in B and D showed a significant delay between [Ca2+]cyt and [Ca2+]mito responses. Histamine (His) was added at 30 s.
[Ca2+]cyt and [Ca2+]mito responses to histamine.
In unstimulated cells, basal [Ca2+]cyt (181 ± 39 nM) and [Ca2+]mito measured either from mitochondria localized in the distal or perinuclear regions of the cells (174 ± 36 and 166 ± 30 nM, respectively, n = 31) were not significantly different (P > 0.05; Figs. 3, B and D, and 4A). In the presence of 2 mM extracellular Ca2+, exposure to 10 μM histamine induced a characteristic large increase in [Ca2+]cyt, which was followed by a rapid increase in [Ca2+]mito (Fig. 3, B and D). When the ROIs to measure changes in rhod-2 fluorescence were placed within the cytosol, we did not observe any response to histamine stimulation (Fig. 3D).
Fig. 4.
Comparison of [Ca2+]cyt and [Ca2+]mito in human ASM. A: basal [Ca2+]cyt and [Ca2+]mito from mitochondria in the distal versus perinuclear regions of cells and the His effect on the amplitude of [Ca2+] responses (peak amplitude, n = 31). Values are means ± SE. B: effect of inhibited mitochondrial transport on the delay (in s) between [Ca2+]cyt and [Ca2+]mito. When measured at the initiation of either response, the delay between [Ca2+]cyt and [Ca2+]mito was estimated at 11 ± 3 and 9 ± 3 s for the mitochondria localized in the perinuclear or distal regions of the cells, respectively. Inhibition of the mitochondrial uniporter (MCU) with Ru360 increased this delay for both mitochondria in the perinuclear regions (15 ± 2 s) and distal regions of the cells (16 ± 3 s). CGP-37157 (CGP) had no effect on the delays between [Ca2+]cyt and [Ca2+]mito. *Significant effect (P < 0.05).
The [Ca2+]mito responses were delayed by 10 ± 3 s from [Ca2+]cyt responses when measured at their initiations (P < 0.05, n = 31; Figs. 3, E and F, and 4B). This observed delay was another strong indication that we measured [Ca2+]mito and not [Ca2+]cyt. After a peak, [Ca2+]cyt rapidly decreased toward baseline, whereas the decrease in [Ca2+]mito was much slower (Fig. 3, B and D). A significant delay between [Ca2+]cyt and [Ca2+]mito peaks was estimated at 12 ± 5 s (P < 0.05, n = 31; Fig. 3F). No significant delay was found between the peaks of [Ca2+]mito responses from distal or perinuclear regions (Fig. 3D). The amplitude of [Ca2+]cyt and [Ca2+]mito responses induced by histamine were not significantly different (P > 0.05; Fig. 4A). The amplitude of [Ca2+]mito in the distal region was slightly lower than for the perinuclear region; however, there was substantially greater variability in the [Ca2+]mito responses in the distal regions (Fig. 4A). The fall of both [Ca2+] responses (starting at the peak until the return to the basal Ca2+ level) was measured by calculating the areas under the curves using SigmaPlot (Systat Software, Chicago, IL) and were significantly higher for [Ca2+]mito responses than for [Ca2+]cyt responses (69,041 ± 1,025 and 51,076 ± 1,236 nM3, respectively, P < 0.05, n = 31).
Effect of inhibited mitochondrial Ca2+ transport on [Ca2+]cyt.
To test the hypothesis that mitochondrial Ca2+ transport regulates [Ca2+]cyt, ASM cells loaded with fluo-3 AM and rhod-2 AM were perfused with 1 μM of the mitochondrial NCX inhibitor 7-chloro-5-(2-chlorophenyl)-1,5-dihydro-4,1-benzothiazepine-2(3H)-one (CGP-37157; Calbiochem, San Diego, CA) (17, 18). CGP-37157 did not significantly influence baseline [Ca2+]cyt or [Ca2+]mito (Fig. 5). However, compared with control cells, the amplitude of the [Ca2+]cyt response to 10 μM histamine was almost doubled in ASM cells that were exposed to CGP-37157 (∼195% of control, P < 0.05, n = 48; Figs. 5A and 6A), and [Ca2+]mito from perinuclear regions was also increased (∼152% of control, P < 0.05, n = 48; Figs. 5B and 6B). CGP-37157 did not increase the delay between [Ca2+]cyt and [Ca2+]mito responses (Fig. 4B). In contrast, CGP-37157 did not significantly influence the amplitude of [Ca2+]mito responses to histamine in distal regions of ASM cells (Figs. 5C and 6B). In addition, CGP-37157 significantly increased the rate of fall of the cytosolic Ca2+ response to histamine (∼60% of control), whereas the rate of fall of [Ca2+]mito responses from perinuclear or distal regions was decreased (∼20%, P < 0.05, n = 48; Figs. 5 and 6C).
Fig. 5.
Effect of inhibited mitochondrial transport on [Ca2+]cyt and [Ca2+]mito. ASM cells loaded with fluo-3 AM and rhod-2 AM were exposed to 10 μM His. A: compared with control cells (solid line), inhibition of MCU with Ru360 (dotted line) increased [Ca2+]cyt induced by His (10 μM). A similar result was obtained when the mitochondrial Na+/Ca2+ exchanger (NCX) was inhibited with CGP (dashed line). B and C: compared with control cells (solid line), Ru360 (dotted line) decreased [Ca2+]mito induced by His (10 μM) in the perinuclear regions (B) or distal regions (C) of the cells. CGP (dashed line) increased [Ca2+]mito in the perinuclear regions (B) but not in the distal regions (C) of the cells.
Fig. 6.
A–C: summary of the effects of inhibited mitochondrial transport on the amplitude of the [Ca2+]cyt response (expressed as a percentage of control; A), the amplitude of the [Ca2+]mito amplitude (B), and the rate of fall (C; expressed as a percentage of control and corresponding to the areas under the curves, starting at the peak until the return to the basal Ca2+ level). Ru360 did not affect the decay of the [Ca2+]cyt responses but decreased the associated rate of fall of [Ca2+]mito for both the perinuclear and distal regions of the cells. CGP increased the rate of fall of [Ca2+]cyt responses, which was accompanied by a decrease in the decay of [Ca2+]mito responses. Values are means ± SE. *Significant effect (P < 0.05).
In a second set of experiments, MCU was inhibited with 1 μM Ru360 (Sigma-Aldrich, St. Louis, MO) (18, 43), which also did not significantly influence baseline [Ca2+]cyt or [Ca2+]mito (Fig. 5). Compared with control ASM cells, Ru360 significantly increased the amplitude of [Ca2+]cyt responses induced by histamine (∼169% of control; Figs. 5A and 6A). The corresponding amplitude of [Ca2+]mito responses from distal, and to a lesser extent perinuclear, regions were decreased (∼40–20% of control, n = 48, P < 0.05; Figs. 5, B and C, and 6B). We also observed that Ru360 increased the delay between [Ca2+]cyt and [Ca2+]mito responses (measured at the initiation of the responses) by ∼5 ± 2 s, for both the perinuclear regions and distal regions (P < 0.05; Fig. 4B). Ru360 did not change the fall of the cytosolic Ca2+ response to histamine, but the fall of [Ca2+]mito response from perinuclear or distal regions was decreased (∼20%, P < 0.05, n = 48; Fig. 6C).
Effect of TNF-α and inhibited mitochondrial Ca2+ transport on [Ca2+]cyt.
ASM cells were incubated for 48 h with TNF-α (20 ng/ml) (72) and then loaded with fluo-3 AM and rhod-2 AM. Inhibitors such as CGP-37157 (mitochondrial NCX inhibitor) or Ru360 (MCU inhibitor) were subsequently added. Basal [Ca2+]cyt was moderately increased after the exposure to TNF-α (∼50 nM, P < 0.05, n = 40; Fig. 7A). In contrast, basal [Ca2+]mito was unaffected by TNF-α (Fig. 7, B and C). Consistent with previous observations (74, 75, 84), TNF-α significantly increased the amplitude of the [Ca2+]cyt response to histamine (10 μM) compared with the vehicle control (∼330% of control, P < 0.05, n = 40; Figs. 7A and 8A). In contrast, the amplitude of the [Ca2+]mito responses was substantially smaller in TNF-α-exposed cells. However, the effect of TNF-α was only observed in mitochondria localized to the perinuclear region of the cells (∼60% of control, P < 0.05, n = 32; Figs. 7, B and C, and 8B). CGP-37157 slightly reduced the effect of TNF-α on the [Ca2+]cyt response to histamine (∼277% of the amplitude of control, P < 0.05, n = 35; Figs. 7A and 8A), and this was correlated with a corresponding increase in [Ca2+]mito within the perinuclear region (∼155% of the amplitude of control, P < 0.05, n = 37; Figs. 7, B and C, and 8B). As in control cells, CGP-37157 did not influence the amplitude of [Ca2+]mito from the distal regions of ASM cells treated with TNF-α. In contrast to CGP-37157, Ru360 had no effect on the increase of [Ca2+]cyt (Figs. 7A and 8A) or the decrease of the [Ca2+]mito response to histamine (10 μM) induced by TNF-α (Figs. 7, B and C, and 8B). The delay between [Ca2+]cyt and [Ca2+]mito responses was not significantly changed by TNF-α and the subsequent inhibition of MCU or NCX (P > 0.05, n = 37). The fall of [Ca2+]cyt was increased by TNF-α (∼40–50% of control, P < 0.05, n = 37; Fig. 8C). Ru360 or CGP-37157 did not significantly further increase the fall of [Ca2+]cyt, which was found to be more variable than for control cells. TNF-α decreased the fall of [Ca2+]mito from perinuclear regions (∼20% of control, P < 0.05, n = 37; Fig. 8C), which was, in contrast, significantly increased by CGP-37157 (∼40% of control, P < 0.05, n = 37; Fig. 8C). TNF-α with or without CGP-37157 did not change the fall of [Ca2+]mito from distal regions. Ru360 did not affect the fall of [Ca2+]mito (P > 0.05; Fig. 8C).
Fig. 7.
Effect of the proinflammatory cytokine TNF-α and inhibited mitochondrial transport on [Ca2+]cyt and [Ca2+]mito. ASM cells incubated for 48 h with TNF-α and then loaded with fluo-3 AM and rhod-2 AM were exposed to 10 μM His. A: compared with the vehicle control, TNF-α increased [Ca2+]cyt responses induced by His (10 μM). A similar result was obtained when MCU was inhibited with Ru360 (dotted line). Inhibition of mitochondrial NCX with CGP (dashed line) decreased [Ca2+]cyt in TNF-α-exposed cells. B and C: TNF-α decreased [Ca2+]mito responses to His measured from mitochondria in the perinuclear regions (B) but not from mitochondria in the distal regions of cells (C). Ru360 did not further decrease [Ca2+]mito in TNF-α-exposed cells. In contrast, CGP significantly increased [Ca2+]mito measured from mitochondria in the perinuclear regions of cells but not from mitochondria in the distal regions.
Fig. 8.
A and B: summary of the effect of TNF-α and inhibited mitochondrial transport on the amplitude of [Ca2+]cyt (A) and [Ca2+]mito (B) responses. C: the rate of fall expressed as a percentage of the vehicle control and corresponding to the areas under the curves (starting at the peak until the return to the basal Ca2+ level). TNF-α increased the decay of [Ca2+]cyt and decreased the decay of [Ca2+]mito responses from the distal regions of the cells. Ru360 increased the decay of [Ca2+]cyt responses but not [Ca2+]mito responses. Compared with the vehicle control, CGP increased the decay of [Ca2+]cyt and [Ca2+]mito responses from the perinuclear regions of the cells. Values are means ± SE. *Significant effect (P < 0.05).
Effect of IL-13 and inhibited mitochondrial Ca2+ transport on [Ca2+]cyt.
ASM cells were incubated for 48 h with IL-13 (20 ng/ml) (72), and experimental protocols similar to those with TNF-α were conducted. IL-13 also induced an increase of the amplitude of the [Ca2+]cyt response to histamine (10 μM) compared with the vehicle control, albeit not as substantial as with TNF-α (∼220% of control, P < 0.05, n = 20; Figs. 9A and 10A). Basal [Ca2+]cyt and [Ca2+]mito were not affected by IL-13 (Fig. 9, A–C). No significant change of the amplitude of the [Ca2+]mito response to histamine was observed in the distal or perinuclear regions of the cells exposed to IL-13 (P > 0.05, n = 20; Fig. 9, B and C, and 10B). The delay between [Ca2+]cyt and [Ca2+]mito responses was also not significantly changed by IL-13 (P > 0.05, n = 20). However, the delays observed after the exposure to IL-13 or TNF-α were much variable than in control cells. Inhibition of MCU with Ru360 increased the [Ca2+]cyt response to histamine, whereas inhibition of mitochondrial NCX with CGP-37157 reduced the effect of IL-13 (∼272% and ∼177% of control, respectively, P < 0.05, n = 20; Figs. 9A and 10A). In ASM cells treated with IL-13, Ru360 decrease the amplitude of the [Ca2+]mito response to histamine in the distal regions (∼46% of control, P < 0.05, n = 23) and perinuclear regions of the cells (∼38% of control, P < 0.05, n = 23; Figs. 9, B and C, and 10B). CGP-37157 increased the amplitude of the [Ca2+]mito response to histamine in the perinuclear regions of the cells treated with IL-13 (∼165% of control, P < 0.05, n = 19; Figs. 9B and 10B). In the distal regions, the effect of CGP-37157 was found to be more variable and overall not significant (Fig. 10B). The fall of [Ca2+]cyt was increased by IL-13 (∼50% of control, P < 0.05, n = 23), whereas the fall of [Ca2+]mito was decreased (∼40% of control, P < 0.05, n = 23; Fig. 10C). Ru360 did not further increase the fall of [Ca2+]cyt or [Ca2+]mito compared with IL-13-treated cells. In contrast, CGP-37157 significantly increased the fall of [Ca2+]mito (∼35% of control, P < 0.05, n = 23) but not the fall of [Ca2+]cyt compared with IL-13 alone (Fig. 10C).
Fig. 9.
Effect of IL-13 and inhibited mitochondrial transport on [Ca2+]cyt and [Ca2+]mito. A: compared with the vehicle control, IL-13 increased [Ca2+]cyt responses to His. Inhibition of MCU with Ru360 (dotted line) further increased [Ca2+]cyt responses in IL-13-exposed cells, whereas inhibition of mitochondrial NCX with CGP (dashed line) decreased [Ca2+]cyt responses. B and C: effects of IL-13 and inhibited mitochondrial transport on [Ca2+]mito responses. Compared with the vehicle control, IL-13 had no effect on [Ca2+]mito responses to His measured from both mitochondria in the perinuclear (B) and distal (C) regions of cells. Ru360 significantly decreased [Ca2+]mito in IL-13-exposed cells, whereas CGP increased [Ca2+]mito responses measured from mitochondria in the perinuclear regions but not from mitochondria in the distal regions of cells.
Fig. 10.
A and B: summary of the effects of IL-13 and inhibited mitochondrial transport on the amplitude of [Ca2+]cyt (A) and [Ca2+]mito (B) responses (expressed as a percentage of the vehicle control). Values are means ± SE. *Significant effect (P < 0.05). C: the rate of fall expressed as a percentage of the vehicle control and corresponding to the areas under the curves (starting at the peak until the return to the basal Ca2+ level). IL-13 increased the decay of [Ca2+]cyt and [Ca2+]mito responses from the distal regions of the cells, whereas it decreased the decay of [Ca2+]mito responses from the perinuclear regions. Ru360 did not further increased the decay of [Ca2+]cyt responses but decreased the decay of [Ca2+]mito responses from both the perinuclear and distal regions of the cells. Compared with the vehicle control, CGP increased the decay of [Ca2+]cyt and [Ca2+]mito responses from the perinuclear regions of the cells. Values are means ± SE. *Significant effect (P < 0.05).
DISCUSSION
During agonist stimulation, mitochondrial Ca2+ uptake of cytosolic Ca2+ has been described in many cell types (13, 25, 33, 38, 48, 55, 56, 65). In this regard, spatial localization of mitochondria may play an important role. Mitochondria are often located near the SR or PM, and a common hypothesis is that mitochondria alter SR Ca2+ release and reuptake as well as PM Ca2+ influx/efflux (10, 13, 16, 25, 33, 38, 43, 48, 52, 60, 62, 63, 65, 70). Inflammation may further modulate some mitochondrial functions (19, 51, 68) and the relationship between mitochondria and the SR (64, 65). In the present study, we examined the relation between [Ca2+]mito and [Ca2+]cyt in human ASM cells during agonist stimulation. More importantly, we showed that inflammatory mediators, such as TNF-α, increase [Ca2+]cyt by disrupting mitochondrial Ca2+ transport. Here, both mitochondrial NCX (mediating Ca2+ release in excitable cells) and MCU (mediating Ca2+ uptake) appear to be involved. However, an important distinction is that the contribution of these mechanisms may vary depending on the perinuclear versus more distal localization of mitochondria.
Under baseline [Ca2+]cyt conditions, mitochondria have relatively “low affinity” for Ca2+ (32, 48, 64, 69). In accordance, we found that inhibition of mitochondrial Ca2+ regulatory mechanisms, such as mitochondrial NCX with CGP-37157 or MCU with Ru360, did not influence [Ca2+]cyt or [Ca2+]mito in unstimulated ASM cells. After agonist stimulation, rapid increases in [Ca2+]cyt appeared to be buffered by mitochondria via the activation of both mitochondrial NCX and MCU. While inhibition of mitochondrial Ca2+ transport was expected to modulate [Ca2+]mito, the changes in [Ca2+]cyt support the idea that mitochondria contribute to [Ca2+]cyt regulation in human ASM cells. Here, we observed a significant delay between the initiation and peak of increase in [Ca2+]cyt versus [Ca2+]mito. Inhibition of mitochondrial Ca2+ uptake with Ru360 but not mitochondrial Ca2+ release modulate these delays, suggesting that mitochondrial Ca2+ buffering is involved in shaping [Ca2+]cyt responses, as predicted by some theoretical models of Ca2+ signaling in smooth muscle cells (66, 67).
Cytosolic Ca2+ is enhanced in airway diseases such as asthma, and it has been proposed that inflammation is a key factor. We and others (2–4, 61, 71, 74, 75, 77, 84) have shown that airway inflammation increases airway contractility, and this is correlated with an increase in agonist-induced [Ca2+]cyt responses. Several [Ca2+]cyt regulatory mechanisms have been reported to be modulated by airway inflammation, but the role of mitochondrial Ca2+ buffering has not been previously examined. In this study, we showed that TNF-α, which increases [Ca2+]cyt, concurrently decreases [Ca2+]mito by inhibiting MCU. In contrast, IL-13, while also enhancing the [Ca2+]cyt response to histamine, does not alter [Ca2+]mito substantially. These novel data suggest an important mechanism by which cytokines may increase [Ca2+]cyt and thus enhance contractility: impaired mitochondrial Ca2+ buffering. There is currently very little information in this regard. A previous study (68) in the lung endothelium found that a 10-min infusion of TNF-α increases basal [Ca2+]mito for up to 1 h. This was correlated with an increase of mitochondrial H2O2. However, the effects on [Ca2+]cyt or changes in [Ca2+]mito after a long exposure to TNF-α were not examined. In our study, after 48 h of exposure to TNF-α, we observed a moderate increase of basal [Ca2+]cyt, but basal [Ca2+]mito was unaffected. IL-13 did not change basal [Ca2+]cyt or [Ca2+]mito. However, the responses to an agonist were clearly affected, indicating that altered mitochondrial Ca2+ buffering may modulate cytokine effects in the short term (agonist responses) as well as in the long term (basal Ca2+ with potential downstream genomic effects). Here, a number of [Ca2+]cyt regulatory mechanisms may be involved in their interactions with mitochondria. PM NCX, sarco(endo)plasmic reticulum Ca2+-ATPase (SERCA), and SOCE have been found to be affected by both cytokines (26, 72, 74, 84), which, in turn, may affect [Ca2+]mito, but their contributions remain to be determined. Recent studies (13, 68) have indicated that inflammation could increase H2O2 production by mitochondria, which, in turn, could affect both [Ca2+]cyt and [Ca2+]mito. The role of inflammation on mitochondrial Ca2+ uptake and Ca2+ release is not clear. In our study, we found that Ru360 did not further decrease [Ca2+]mito in ASM cells treated with TNF-α, suggesting that MCU was inhibited. An increase of mitochondrial Ca2+ uptake leads to ROS generation and ATP production, and high mitochondrial [Ca2+] is detrimental to mitochondrial functions. In our study, it was not clear if after 48 h of exposure to TNF-α the MCU is inhibited to prevent a high level of mitochondrial H2O2 or whether TNF-α is involved in regulating the energetic demand of the cells. Potential downstream genomic effects also remain to be explored, especially in ASM.
Mitochondrial Ca2+ transport is activated by large [Ca2+]cyt changes in the micromolar range (32, 48, 64, 69). Given that global [Ca2+]cyt elevations in most tissues are only in the low micromolar range, local [Ca2+]cyt levels in the vicinity of the SR or PM must reach high enough levels to activate mitochondrial Ca2+ buffering (27, 53, 56, 57). A recent study (27) estimated that [Ca2+] is 5–10 times higher in these microdomains than elsewhere in the cytosol. Depending on the [Ca2+]mito level, subsequent Ca2+ release back into the cytosol may be fast or slow (62). In non-ASM tissues, mitochondria respond to local [Ca2+]cyt in the vicinity of open Ca2+ channels (20, 37, 62). In vascular and gastrointestinal smooth muscles, Ca2+ release via inositol 1,4,5-triphosphate (IP3) receptor (IP3R) channels (83) and ryanodine receptor (RyR) channels (83) results in large [Ca2+]mito elevations, as it appears to do in ASM cells based on our results. Accordingly, the physical proximity of mitochondria to the SR and PM may be important in terms of buffering SR Ca2+ release or modulating influx and might explain some of the differences that we observed between [Ca2+]mito from distal or perinuclear regions.
Consistent with an expected loss of mitochondrial Ca2+ extrusion and mitochondrial Ca2+ uptake (8, 10, 62), inhibition of mitochondrial NCX with CGP-37157 increased [Ca2+]mito, whereas inhibition of MCU with Ru360 decreased [Ca2+]mito. However, an interesting and novel observation in our study was the differential roles of mitochondrial NCX versus MCU in the perinuclear versus distal regions of ASM cells. In the distal regions, CGP-37157 had no significant effect, whereas Ru360 consistently decreased [Ca2+]mito. Here, based on the typical fusiform shape of ASM cells, we propose that the distal mitochondria may be close to the PM and thus affected by and, in turn, influence Ca2+ influx. Accordingly, a role for the uniporter in buffering Ca2+ influx is likely to take precedence in these cellular areas. Mitochondrial Ca2+ uptake is also associated with increased ATP production. As the distal area of ASM cells is primarily associated with the contractile apparatus, the energetic demand might be higher than in the perinuclear area and favor mitochondrial Ca2+ uptake. In TNF-α-exposed cells, Ru360 did not further decrease [Ca2+]mito, suggesting an inhibitory effect of TNF-α on MCU. These differences between the two regions remain to be further explored in the context of cytokine effects on energy availability for the contractile apparatus in the distal areas of ASM cells versus other effects, such as in the SR or nuclear Ca2+ reuptake in the perinuclear area.
Impaired mitochondrial Ca2+ buffering has been reported to enhance [Ca2+]cyt release in different cell types (8, 10, 14, 62). The regulation of [Ca2+]cyt by mitochondria may reflect the Ca2+ buffering capacity (either global or localized) of the mitochondria or/and by indirect effects of some mitochondrial function. The physical proximity of mitochondria to the SR had led several recent studies (11, 34, 36, 66) to examine possible functional interactions between the SR and mitochondria. A common hypothesis is that the mitochondria alter SR Ca2+ refilling by competing with SERCA pumps for the removal of cytosolic Ca2+ (49). In smooth muscle (as in other muscles), SERCA is the mechanism for filling the SR, with two Ca2+ pumped back for every ATP consumed against a gradient of ∼1 mM in the SR lumen (42). SERCA activity is directly proportional to local [Ca2+]cyt. By affecting local [Ca2+]cyt as well as ATP, mitochondria may affect SERCA activity. It has been proposed that mitochondria could generate microdomains of low Ca2+ next to SERCA pumps, and this ability was found to be dependent on mitochondrial Ca2+ uptake and not on mitochondrial Ca2+ release (29, 40). Our findings of inhibitory effects of Ru360 on [Ca2+]cyt in the perinuclear areas is consistent with the above scenario. Whether SR luminal Ca2+ is actually affected in human ASM cells remains to be examined.
Mitochondrial Ca2+ fluxes can also regulate IP3R and RyR channel activities and thus SR Ca2+ release (6, 11, 12, 14, 15, 34, 36). Here, the effects appear to be cell type specific, with both inhibition and promotion of SR Ca2+ release being reported (6, 11, 12, 15). Cheranov et al. (14) demonstrated that CGP-37157 increases the driving force for [Ca2+]mito release and elevates local [Ca2+]cyt near IP3R channels or RyR channels, thereby affecting SR Ca2+ release. Such a scenario may explain the observed increase in [Ca2+]cyt in the presence of CGP-37157, especially in the perinuclear areas, where the SR is likely to be located. Additionally, H2O2 production, which is dependent on mitochondrial Ca2+ uptake, may be induced by histamine and affect both the IP3 signaling cascade and SOCE (13) as well as ATP production. ATP is also an important effector of IP3Rs and is dependent on [Ca2+]mito and ROS production. The potential effect of H2O2, notably on the fall of the [Ca2+]cyt response, might explain the slow decay of [Ca2+]cyt observed with CGP-37157. We cannot exclude a nonspecific effect of CGP-37157, but this is generally observed with a concentration of >10 μM (for a review, see Ref. 18), which was not used here. Smaller doses of CGP-37157 had no effect on both [Ca2+]cyt or [Ca2+]mito in our primary ASM cells (data not shown).
Recently, the molecular identities of MCU [MCU/mitochondrial Ca2+ uptake 1 (MICU1)] and NCX (also called NCLX) (7, 21, 22, 47) have been uncovered, and the development of molecular tools may help us to better understand the relation between MCU and NCX with other established Ca2+ regulatory mechanisms. In this regard, studies examining MICU1 and NCX have relied on the specificity of Ru360 and CGP-37157 (18). Accordingly, these pharmacological inhibitors form an appropriate first step toward examining [Ca2+]mito in primary cells such as human ASM, which tend to be recalcitrant to RNA interference approaches targeting MICU1 and NCX (pilot studies).
In addition to SR Ca2+ release and reuptake, PM Ca2+ influx/efflux is a major component of [Ca2+]cyt regulation in ASM. We (5, 75, 84) have previously demonstrated the importance of SOCE in ASM, triggered after SR Ca2+ depletion. It is important to recognize that the extent of Ca2+ influx via SOCE is not simply dependent on local PM Ca2+ gradients but is regulated by the state of SR repletion, [Ca2+]cyt-dependent fast and slow inactivation, and even by other intracellular signaling cascades (e.g., diacylglycerol) (50). There is now evidence in non-ASM tissues that mitochondria can also influence SOCE (35, 44, 50). However, this effect may be indirect. By taking up Ca2+ released from the SR, mitochondrial Ca2+ buffering can enhance store depletion and hence maintain SOCE (48, 50). The available information suggests that the physical proximity of mitochondria to the PM may be important (50). A recent study (46) has shown that the inhibition of mitochondrial NCX is fundamental to SOCE regulation in endothelial cells. Whether these scenarios hold true in ASM cells has not been determined. Our finding that Ru360 reduces [Ca2+]mito while elevating [Ca2+]cyt is suggestive. However, a more interesting observation is that in the presence of TNF-α, Ru360 did not affect either [Ca2+]cyt or [Ca2+]mito. We (84) have previously shown that TNF-α increases SOCE. Therefore, we would have expected [Ca2+]mito to be enhanced in the presence of TNF-α, allowing SOCE to be maintained. The reasons underlying this discrepancy are not clear but may involve cytokine-induced dysregulation of MCU itself.
In the present study, we found that the effects of TNF-α versus IL-13 on [Ca2+]mito were substantially different, especially depending on the location of the mitochondria. The functional relevance of these differences remains to be determined. However, it is possible that although both cytokines, which are important in asthma, can robustly enhance [Ca2+]cyt, the mechanisms by which this is achieved may differ, including mitochondrial Ca2+ regulation.
Overall, the present study demonstrates that mitochondria buffer [Ca2+]cyt in human ASM cells and contribute to the modulation of [Ca2+]cyt. Considering the importance of [Ca2+]cyt regulatory mechanisms in enhancing [Ca2+]cyt under conditions such as inflammation (relevant to diseases such as asthma), our study underlines the emerging importance of mitochondria, especially MCU, in inflammatory airway disease.
GRANTS
This work was supported by National Heart, Lung, and Blood Institute Grants HL-74309 (to G. C. Sieck), HL-090595 (to C. M. Pabelick), HL-088029 (to Y. S. Prakash), and HL-056470 (to Y. S. Prakash and G. C. Sieck).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
AUTHOR CONTRIBUTIONS
Author contributions: P.F.D., B.Y., and M.A.T. performed experiments; P.F.D., B.Y., and M.A.T. analyzed data; P.F.D., C.M.P., Y.S.P., and G.C.S. interpreted results of experiments; P.F.D., Y.S.P., and G.C.S. prepared figures; P.F.D., C.M.P., Y.S.P., and G.C.S. drafted manuscript; P.F.D., Y.S.P., and G.C.S. edited and revised manuscript; C.M.P., Y.S.P., and G.C.S. conception and design of research; Y.S.P. and G.C.S. approved final version of manuscript.
REFERENCES
- 1. Aguilera-Aguirre L, Bacsi A, Saavedra-Molina A, Kurosky A, Sur S, Boldogh I. Mitochondrial dysfunction increases allergic airway inflammation. J Immunol 183: 5379–5387, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Amrani Y. TNF-alpha and calcium signaling in airway smooth muscle cells: a never-ending story with promising therapeutic relevance. Am J Respir Cell Mol Biol 36: 387–388, 2007 [DOI] [PubMed] [Google Scholar]
- 3. Amrani Y, Chen H, Panettieri RA., Jr Activation of tumor necrosis factor receptor 1 in airway smooth muscle: a potential pathway that modulates bronchial hyper-responsiveness in asthma? Respir Res 1: 49–53, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Amrani Y, Panettieri RA., Jr Modulation of calcium homeostasis as a mechanism for altering smooth muscle responsiveness in asthma. Curr Opin Allergy Clin Immunol 2: 39–45, 2002 [DOI] [PubMed] [Google Scholar]
- 5. Ay B, Prakash YS, Pabelick CM, Sieck GC. Store-operated Ca2+ entry in porcine airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 286: L909–L917, 2004 [DOI] [PubMed] [Google Scholar]
- 6. Balemba OB, Bartoo AC, Nelson MT, Mawe GM. Role of mitochondria in spontaneous rhythmic activity and intracellular calcium waves in the guinea pig gallbladder smooth muscle. Am J Physiol Gastrointest Liver Physiol 294: G467–G476, 2008 [DOI] [PubMed] [Google Scholar]
- 7. Baughman JM, Perocchi F, Girgis HS, Plovanich M, Belcher-Timme CA, Sancak Y, Bao XR, Strittmatter L, Goldberger O, Bogorad RL, Koteliansky V, Mootha VK. Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature 476: 341–345, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Bernardi P. Mitochondrial transport of cations: channels, exchangers, and permeability transition. Physiol Rev 79: 1127–1155, 1999 [DOI] [PubMed] [Google Scholar]
- 9. Bruce JI, Giovannucci DR, Blinder G, Shuttleworth TJ, Yule DI. Modulation of [Ca2+]i signaling dynamics and metabolism by perinuclear mitochondria in mouse parotid acinar cells. J Biol Chem 279: 12909–12917, 2004 [DOI] [PubMed] [Google Scholar]
- 10. Carafoli E. The fateful encounter of mitochondria with calcium: how did it happen? Biochim Biophys Acta 1797: 595–606, 2010 [DOI] [PubMed] [Google Scholar]
- 11. Chalmers S, McCarron JG. Inhibition of mitochondrial calcium uptake rather than efflux impedes calcium release by inositol-1,4,5-trisphosphate-sensitive receptors. Cell Calcium 46: 107–113, 2009 [DOI] [PubMed] [Google Scholar]
- 12. Chalmers S, McCarron JG. The mitochondrial membrane potential and Ca2+ oscillations in smooth muscle. J Cell Sci 121: 75–85, 2008 [DOI] [PubMed] [Google Scholar]
- 13. Chen T, Zhu L, Wang T, Ye H, Huang K, Hu Q. Mitochondria depletion abolishes agonist-induced Ca2+ plateau in airway smooth muscle cells: potential role of H2O2. Am J Physiol Lung Cell Mol Physiol 298: L178–L188, 2010 [DOI] [PubMed] [Google Scholar]
- 14. Cheranov SY, Jaggar JH. Mitochondrial modulation of Ca2+ sparks and transient KCa currents in smooth muscle cells of rat cerebral arteries. J Physiol 556: 755–771, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Collins TJ, Lipp P, Berridge MJ, Li W, Bootman MD. Inositol 1,4,5-trisphosphate-induced Ca2+ release is inhibited by mitochondrial depolarization. Biochem J 347: 593–600, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cox DA, Matlib MA. Modulation of intramitochondrial free Ca2+ concentration by antagonists of Na+-Ca2+ exchange. Trends Pharmacol Sci 14: 408–413, 1993 [DOI] [PubMed] [Google Scholar]
- 17. Cox DA, Matlib MA. A role for the mitochondrial Na+-Ca2+ exchanger in the regulation of oxidative phosphorylation in isolated heart mitochondria. J Biol Chem 268: 938–947, 1993 [PubMed] [Google Scholar]
- 18. Csordas G, Varnai P, Golenar T, Sheu SS, Hajnoczky G. Calcium transport across the inner mitochondrial membrane: molecular mechanisms and pharmacology. Mol Cell Endocrinol 353: 109–113, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Dada LA, Sznajder JI. Mitochondrial Ca2+ and ROS take center stage to orchestrate TNF-alpha-mediated inflammatory responses. J Clin Invest 121: 1683–1685, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Dai J, Kuo KH, Leo JM, van Breemen C, Lee CH. Rearrangement of the close contact between the mitochondria and the sarcoplasmic reticulum in airway smooth muscle. Cell Calcium 37: 333–340, 2005 [DOI] [PubMed] [Google Scholar]
- 21. De Stefani D, Raffaello A, Teardo E, Szabo I, Rizzuto R. A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature 476: 336–340, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Drago I, Pizzo P, Pozzan T. After half a century mitochondrial calcium in- and efflux machineries reveal themselves. EMBO J 30: 4119–4125, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Duchen MR. Mitochondria and calcium: from cell signalling to cell death. J Physiol 529: 57–68, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Duszynski J, Koziel R, Brutkowski W, Szczepanowska J, Zablocki K. The regulatory role of mitochondria in capacitative calcium entry. Biochim Biophys Acta 1757: 380–387, 2006 [DOI] [PubMed] [Google Scholar]
- 25. Franzini-Armstrong C. ER-mitochondria communication. How privileged? Physiology (Bethesda) 22: 261–268, 2007 [DOI] [PubMed] [Google Scholar]
- 26. Gao YD, Zou JJ, Zheng JW, Shang M, Chen X, Geng S, Yang J. Promoting effects of IL-13 on Ca2+ release and store-operated Ca2+ entry in airway smooth muscle cells. Pulm Pharmacol Ther 23: 182–189, 2010 [DOI] [PubMed] [Google Scholar]
- 27. Giacomello M, Drago I, Bortolozzi M, Scorzeto M, Gianelle A, Pizzo P, Pozzan T. Ca2+ hot spots on the mitochondrial surface are generated by Ca2+ mobilization from stores, but not by activation of store-operated Ca2+ channels. Mol Cell 38: 280–290, 2010 [DOI] [PubMed] [Google Scholar]
- 28. Girodet PO, Ozier A, Bara I, Tunon de Lara JM, Marthan R, Berger P. Airway remodeling in asthma: new mechanisms and potential for pharmacological intervention. Pharmacol Ther 130: 325–337, 2011 [DOI] [PubMed] [Google Scholar]
- 29. Glitsch MD, Bakowski D, Parekh AB. Store-operated Ca2+ entry depends on mitochondrial Ca2+ uptake. EMBO J 21: 6744–6754, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Gunter TE, Buntinas L, Sparagna G, Eliseev R, Gunter K. Mitochondrial calcium transport: mechanisms and functions. Cell Calcium 28: 285–296, 2000 [DOI] [PubMed] [Google Scholar]
- 31. Gunter TE, Gunter KK. Uptake of calcium by mitochondria: transport and possible function. IUBMB Life 52: 197–204, 2001 [DOI] [PubMed] [Google Scholar]
- 32. Gunter TE, Yule DI, Gunter KK, Eliseev RA, Salter JD. Calcium and mitochondria. FEBS Lett 567: 96–102, 2004 [DOI] [PubMed] [Google Scholar]
- 33. Hajnoczky G, Saotome M, Csordas G, Weaver D, Yi M. Calcium signalling and mitochondrial motility. Novartis Found Symp 287: 105–117; discussion 117–121, 2007 [DOI] [PubMed] [Google Scholar]
- 34. Hernandez-SanMiguel E, Vay L, Santo-Domingo J, Lobaton CD, Moreno A, Montero M, Alvarez J. The mitochondrial Na+/Ca2+ exchanger plays a key role in the control of cytosolic Ca2+ oscillations. Cell Calcium 40: 53–61, 2006 [DOI] [PubMed] [Google Scholar]
- 35. Hoth M, Button DC, Lewis RS. Mitochondrial control of calcium-channel gating: a mechanism for sustained signaling and transcriptional activation in T lymphocytes. Proc Natl Acad Sci USA 97: 10607–10612, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Ishii K, Hirose K, Iino M. Ca2+ shuttling between endoplasmic reticulum and mitochondria underlying Ca2+ oscillations. EMBO Rep 7: 390–396, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Kuba M, Higure Y, Susaki H, Hayato R, Kuba K. Bidirectional Ca2+ coupling of mitochondria with the endoplasmic reticulum and regulation of multimodal Ca2+ entries in rat brown adipocytes. Am J Physiol Cell Physiol 292: C896–C908, 2007 [DOI] [PubMed] [Google Scholar]
- 38. Maack C, O'Rourke B. Excitation-contraction coupling and mitochondrial energetics. Basic Res Cardiol 102: 369–392, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Mabalirajan U, Dinda AK, Kumar S, Roshan R, Gupta P, Sharma SK, Ghosh B. Mitochondrial structural changes and dysfunction are associated with experimental allergic asthma. J Immunol 181: 3540–3548, 2008 [DOI] [PubMed] [Google Scholar]
- 40. Malli R, Frieden M, Osibow K, Zoratti C, Mayer M, Demaurex N, Graier WF. Sustained Ca2+ transfer across mitochondria is Essential for mitochondrial Ca2+ buffering, store-operated Ca2+ entry, and Ca2+ store refilling. J Biol Chem 278: 44769–44779, 2003 [DOI] [PubMed] [Google Scholar]
- 41. Malli R, Frieden M, Trenker M, Graier WF. The role of mitochondria for Ca2+ refilling of the endoplasmic reticulum. J Biol Chem 280: 12114–12122, 2005 [DOI] [PubMed] [Google Scholar]
- 42. Marin J, Encabo A, Briones A, Garcia-Cohen EC, Alonso MJ. Mechanisms involved in the cellular calcium homeostasis in vascular smooth muscle: calcium pumps. Life Sci 64: 279–303, 1999 [DOI] [PubMed] [Google Scholar]
- 43. Matlib MA, Zhou Z, Knight S, Ahmed S, Choi KM, Krause-Bauer J, Phillips R, Altschuld R, Katsube Y, Sperelakis N, Bers DM. Oxygen-bridged dinuclear ruthenium amine complex specifically inhibits Ca2+ uptake into mitochondria in vitro and in situ in single cardiac myocytes. J Biol Chem 273: 10223–10231, 1998 [DOI] [PubMed] [Google Scholar]
- 44. Montero M, Alonso MT, Carnicero E, Cuchillo-Ibanez I, Albillos A, Garcia AG, Garcia-Sancho J, Alvarez J. Chromaffin-cell stimulation triggers fast millimolar mitochondrial Ca2+ transients that modulate secretion. Nat Cell Biol 2: 57–61, 2000 [DOI] [PubMed] [Google Scholar]
- 45. Munoz E, Valero RA, Quintana A, Hoth M, Nunez L, Villalobos C. Nonsteroidal anti-inflammatory drugs inhibit vascular smooth muscle cell proliferation by enabling the Ca2+-dependent inactivation of calcium release-activated calcium/orai channels normally prevented by mitochondria. J Biol Chem 286: 16186–16196, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Naghdi S, Waldeck-Weiermair M, Fertschai I, Poteser M, Graier WF, Malli R. Mitochondrial Ca2+ uptake and not mitochondrial motility is required for STIM1-Orai1-dependent store-operated Ca2+ entry. J Cell Sci 123: 2553–2564, 2010 [DOI] [PubMed] [Google Scholar]
- 47. Palty R, Silverman WF, Hershfinkel M, Caporale T, Sensi SL, Parnis J, Nolte C, Fishman D, Shoshan-Barmatz V, Herrmann S, Khananshvili D, Sekler I. NCLX is an essential component of mitochondrial Na+/Ca2+ exchange. Proc Natl Acad Sci USA 107: 436–441, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Parekh AB. Mitochondrial regulation of intracellular Ca2+ signaling: more than just simple Ca2+ buffers. News Physiol Sci 18: 252–256, 2003 [DOI] [PubMed] [Google Scholar]
- 49. Parekh AB. Store-operated Ca2+ entry: dynamic interplay between endoplasmic reticulum, mitochondria and plasma membrane. J Physiol 547: 333–348, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Parekh AB, Putney JW., Jr Store-operated calcium channels. Physiol Rev 85: 757–810, 2005 [DOI] [PubMed] [Google Scholar]
- 51. Parthasarathi K, Ichimura H, Quadri S, Issekutz A, Bhattacharya J. Mitochondrial reactive oxygen species regulate spatial profile of proinflammatory responses in lung venular capillaries. J Immunol 169: 7078–7086, 2002 [DOI] [PubMed] [Google Scholar]
- 52. Perocchi F, Gohil VM, Girgis HS, Bao XR, McCombs JE, Palmer AE, Mootha VK. MICU1 encodes a mitochondrial EF hand protein required for Ca2+ uptake. Nature 467: 291–296, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Petersen OH, Petersen CC, Kasai H. Calcium and hormone action. Annu Rev Physiol 56: 297–319, 1994 [DOI] [PubMed] [Google Scholar]
- 54. Pitter JG, Maechler P, Wollheim CB, Spat A. Mitochondria respond to Ca2+ already in the submicromolar range: correlation with redox state. Cell Calcium 31: 97–104, 2002 [DOI] [PubMed] [Google Scholar]
- 55. Poburko D, Santo-Domingo J, Demaurex N. Dynamic regulation of the mitochondrial proton gradient during cytosolic calcium elevations. J Biol Chem 286: 11672–11684, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Pozzan T, Rizzuto R. The renaissance of mitochondrial calcium transport. Eur J Biochem 267: 5269–5273, 2000 [DOI] [PubMed] [Google Scholar]
- 57. Pozzan T, Rizzuto R, Volpe P, Meldolesi J. Molecular and cellular physiology of intracellular calcium stores. Physiol Rev 74: 595–636, 1994 [DOI] [PubMed] [Google Scholar]
- 58. Prakash YS, Iyanoye A, Ay B, Mantilla CB, Pabelick CM. Neurotrophin effects on intracellular Ca2+ and force in airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 291: L447–L456, 2006 [DOI] [PubMed] [Google Scholar]
- 59. Prakash YS, Kannan MS, Sieck GC. Regulation of intracellular calcium oscillations in porcine tracheal smooth muscle cells. Am J Physiol Cell Physiol 272: C966–C975, 1997 [DOI] [PubMed] [Google Scholar]
- 60. Reddy PH. Mitochondrial dysfunction and oxidative stress in asthma: implications for mitochondria-targeted antioxidant therapeutics. Pharmaceuticals (Basel) 4: 429–456, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Risse PA, Jo T, Suarez F, Hirota N, Tolloczko B, Ferraro P, Grutter P, Martin JG. Interleukin-13 inhibits proliferation and enhances contractility of human airway smooth muscle cells without change in contractile phenotype. Am J Physiol Lung Cell Mol Physiol 300: L958–L966, 2011 [DOI] [PubMed] [Google Scholar]
- 62. Rizzuto R, Bernardi P, Pozzan T. Mitochondria as all-round players of the calcium game. J Physiol 529: 37–47, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Rizzuto R, Marchi S, Bonora M, Aguiari P, Bononi A, De Stefani D, Giorgi C, Leo S, Rimessi A, Siviero R, Zecchini E, Pinton P. Ca2+ transfer from the ER to mitochondria: when, how and why. Biochim Biophys Acta 1787: 1342–1351, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Rizzuto R, Pozzan T. Microdomains of intracellular Ca2+: molecular determinants and functional consequences. Physiol Rev 86: 369–408, 2006 [DOI] [PubMed] [Google Scholar]
- 65. Romagnoli A, Aguiari P, De Stefani D, Leo S, Marchi S, Rimessi A, Zecchini E, Pinton P, Rizzuto R. Endoplasmic reticulum/mitochondria calcium cross-talk. Novartis Found Symp 287: 122–131, 2007 [PubMed] [Google Scholar]
- 66. Roux E, Marhl M. Role of sarcoplasmic reticulum and mitochondria in Ca2+ removal in airway myocytes. Biophys J 86: 2583–2595, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Roux E, Noble PJ, Noble D, Marhl M. Modelling of calcium handling in airway myocytes. Prog Biophys Mol Biol 90: 64–87, 2006 [DOI] [PubMed] [Google Scholar]
- 68. Rowlands DJ, Islam MN, Das SR, Huertas A, Quadri SK, Horiuchi K, Inamdar N, Emin MT, Lindert J, Ten VS, Bhattacharya S, Bhattacharya J. Activation of TNFR1 ectodomain shedding by mitochondrial Ca2+ determines the severity of inflammation in mouse lung microvessels. J Clin Invest 121: 1986–1999, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Ruiz-Meana M, Fernandez-Sanz C, Garcia-Dorado D. The SR-mitochondria interaction: a new player in cardiac pathophysiology. Cardiovasc Res 88: 30–39, 2010 [DOI] [PubMed] [Google Scholar]
- 70. Ryu SY, Beutner G, Kinnally K, Dirksen RT, Sheu SS. Single channel characterization of the mitochondrial ryanodine receptor in heart mitoplasts. J Biol Chem 286: 21324–21329, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Sakai Y, Kwan CY. Calcium regulation and contractile dysfunction of smooth muscle. Biol Signals 2: 305–312, 1993 [DOI] [PubMed] [Google Scholar]
- 72. Sathish V, Delmotte PF, Thompson MA, Pabelick CM, Sieck GC, Prakash YS. Sodium-calcium exchange in intracellular calcium handling of human airway smooth muscle. PLoS One 6: e23662, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Sathish V, Leblebici F, Kip SN, Thompson MA, Pabelick CM, Prakash YS, Sieck GC. Regulation of sarcoplasmic reticulum Ca2+ reuptake in porcine airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 294: L787–L796, 2008 [DOI] [PubMed] [Google Scholar]
- 74. Sathish V, Thompson MA, Bailey JP, Pabelick CM, Prakash YS, Sieck GC. Effect of proinflammatory cytokines on regulation of sarcoplasmic reticulum Ca2+ reuptake in human airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 297: L26–L34, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Sieck GC, White TA, Thompson MA, Pabelick CM, Wylam ME, Prakash YS. Regulation of store-operated Ca2+ entry by CD38 in human airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 294: L378–L385, 2008 [DOI] [PubMed] [Google Scholar]
- 76. Slater EC, Cleland KW. The effect of calcium on the respiratory and phosphorylative activities of heart-muscle sarcosomes. Biochem J 55: 566–590, 1953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Tirumurugaan KG, Jude JA, Kang BN, Panettieri RA, Walseth TF, Kannan MS. TNF-α induced CD38 expression in human airway smooth muscle cells: role of MAP kinases and transcription factors NF-κB and AP-1. Am J Physiol Lung Cell Mol Physiol 292: L1385–L1395, 2007 [DOI] [PubMed] [Google Scholar]
- 78. Townsend EA, Thompson MA, Pabelick CM, Prakash YS. Rapid effects of estrogen on intracellular Ca2+ regulation in human airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 298: L521–L530, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Trian T, Benard G, Begueret H, Rossignol R, Girodet PO, Ghosh D, Ousova O, Vernejoux JM, Marthan R, Tunon-de-Lara JM, Berger P. Bronchial smooth muscle remodeling involves calcium-dependent enhanced mitochondrial biogenesis in asthma. J Exp Med 204: 3173–3181, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Tsien R, Pozzan T. Measurement of cytosolic free Ca2+ with quin2. Methods Enzymol 172: 230–262, 1989 [DOI] [PubMed] [Google Scholar]
- 81. Varadi A, Cirulli V, Rutter GA. Mitochondrial localization as a determinant of capacitative Ca2+ entry in HeLa cells. Cell Calcium 36: 499–508, 2004 [DOI] [PubMed] [Google Scholar]
- 82. Vasington FD, Murphy JV. Ca ion uptake by rat kidney mitochondria and its dependence on respiration and phosphorylation. J Biol Chem 237: 2670–2677, 1962 [PubMed] [Google Scholar]
- 83. Ward SM, Baker SA, de Faoite A, Sanders KM. Propagation of slow waves requires IP3 receptors and mitochondrial Ca2+ uptake in canine colonic muscles. J Physiol 549: 207–218, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. White TA, Xue A, Chini EN, Thompson M, Sieck GC, Wylam ME. Role of transient receptor potential C3 in TNF-alpha-enhanced calcium influx in human airway myocytes. Am J Respir Cell Mol Biol 35: 243–251, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]