Abstract
CD8 T cells stimulated with a suboptimal dose of anti-CD3 antibodies (100 pg/ml) in the presence of IL-15 retain a naïve phenotype with expression of CD45RA, CD28, CD27 and CCR7 but acquire new functions and differentiate into immunosuppressive T cells. CD8+CCR7+ Tregs express FOXP3 and prevent CD4 T cells from responding to T-cell receptor stimulation and entering the cell cycle. Naïve CD4 T cells are more susceptible to inhibition than memory cells. The suppressive activity of CD8+CCR7+ Tregs is not mediated by IL-10, TGF-β, CTLA-4, CCL4 or adenosine and relies on interference with very early steps of the TCR signaling cascade. Specifically, CD8+CCR7+ Tregs prevent TCR-induced phosphorylation of ZAP70 and dampen the rise of intracellular calcium in CD4 T cells. The inducibility of CD8+CCR7+ Tregs is correlated to the age of the individual with peripheral blood lymphocytes of donors older than 60 years yielding low numbers of FOXP3low CD8 Treg cells. Loss of CD8+CCR7+ Tregs in the elderly host may be of relevance in the aging immune system as immunosenescence is associated with a state of chronic smoldering inflammation.
Introduction
Regulatory T cells (Tregs) are now recognized as a critical T-cell subset that participates in normal and dysfunctional immune responses. Tregs guard tolerance to self and play a pivotal role in downregulating autoreactive and pathogenic immunity (1). Also, Tregs control immune responses to pathogens and allergens and have been implicated in defining the balance between the host and commensal microbial flora (2).
A defining molecular feature of Tregs is the expression of the FOXP3 transcription factor which has emerged as a common denominator of cells specializing in regulatory function (3). The critical role of FOXP3 in determining the functionality of Tregs is emphasized by the severe multi-organ autoimmune disease that occurs in FOXP3-deficient mice (scurfy mice). Similarly, humans defective for FOXP3 develop a multitude of immune-mediated pathologies (4). While the expression of FOXP3 is not unique for Tregs, particularly not for human T cells most of which transiently express FOXP3 after T-cell receptor-mediated activation (5), this transcription factor is useful in identifying and monitoring Treg populations.
Much of what is now known about Tregs has been learned from CD4+FOXP3+ Tregs but it is likely that each Treg population has unique conditions of induction and mechanisms through which it functions. CD4+FOXP3+ Tregs develop in the thymus as a distinct lineage that is separated from conventional CD4 T cells during the time of positive selection (6, 7). Thymic FOXP3+ cells are characterized by a selected repertoire of T cell receptors that distinguishes them from conventional T cells. Alternatively, FOXP3+ Tregs can be induced by converting mature peripheral T cells into FOXP3 positivity (8–10).
To distinguish such inducible Tregs from the natural, thymus-derived population, they have been named adaptive Tregs. A multitude of experimental conditions has been described which allow the generation of inducible/adaptive Tregs, including chronic suboptimal stimulation of the antigen receptor, introduction of antigen via the oral route and antigen exposure during the homeostatic expansion of lymphocytes in lymphopenic hosts (11–13). Also, it has been suggested that in vitro activation of naïve T cells in the presence of IL-2 and TGF-β is sufficient to achieve outgrowth of Tregs with immunosuppressive properties (14). Even subtle differences in induction conditions may be relevant for the functional profile, the stability and tissue trafficking behavior of the resulting Treg populations (15, 16) and all of these parameters will have a major impact on the potential use of Treg in vivo. In an attempt to understand the heterogeneity of the different FOXP3+ Treg populations in mice, a recent study has compared broad gene-expression profiles of a multitude of converted FOXP3+ cell subsets with those isolated from unmanipulated tissues. The transcriptional signatures of the different FOXP3 populations revealed marked and unexpected heterogeneity arguing for significant diversity within the pool of Tregs (17).
In line with a heterogeneous composition of the Treg pool, a multitude of molecular mechanisms has been identified that underpin the suppressive activity of regulatory lymphocytes (18, 19). While immunosuppressive pathways may be influenced by the tissue environment in which they occur and the cells that are targeted, some common denominators have emerged (20). Tregs can communicate with responder T cells via the release of mediators or through contact-dependent receptor-ligand interactions. Naïve T cells can be stopped from entering the activation cycle by disrupting activation signals, e.g. by absorbing the critical growth factor IL-2. As many Treg populations express the CD25 IL-2 receptor on their surface, mopping away IL-2 may be one mode of interfering with immune responses. Tregs may also acquire the ability to kill effector T cells or destroy antigen-presenting cells, thus disrupting immune activation. IL-10 secretion has been closely linked with the ability to dampen immune responses. Similarly, adenosine has been implicated in mediating suppressive functions of Tregs (21). Activation of the adenosine receptors expressed on many of the immune and inflammatory cells increases intracellular cAMP concentrations, a signal that inhibits cytokine production and cellular expansion. Adenosine-producing Tregs may thus participate in a negative feedback loop that controls host immunity and inflammation. Alternative immunoregulatory molecules secreted by Tregs include CCL4, which allows CD8 Tregs to specifically target chemokine receptor-expressing effector cells (22). Finally, CD4 Tregs constitutively express CTLA-4 and this molecule has been implicated in their suppressive function (23).
While considerable effort has gone into understanding and characterizing CD4+ Tregs, much less is known about CD8+ Tregs but CD8 Tregs are considered essential participants in normal and pathogenic immune responses (24). About 25 years ago early studies identified human CD8+ suppressor cells in patients with disseminated or lepromatous leprosy (25, 26). A role for CD8+ Tregs has also been suspected in autoimmune disease, allotransplantation and infectious disease (27). Limited information is available on distinct subsets or lineage markers among CD8 Tregs, but CD28 expression or the lack thereof appears to be a distinctive feature of different CD8+ Treg subsets in humans. In this context, it is important that the loss of CD28 is a feature of end-differentiated effector T cells and that CD8+CD28− T cells are now recognized as a marker population in the aged immune system. Thus, CD8+CD28− T cells with suppressive properties are part of the memory T cell compartment but would not reside among naïve T cells. The thymus of healthy humans contains CD8+ T cells that express classical Treg markers (CD25, FOXP3, GITR, CTLA-4) and inhibit through a contact-dependent mechanism (28). The circulating T-cell pool of humans includes CD8+ Tregs that are characterized by the expression of CD103 (29); immunosuppressive CD8+ T cells in heart transplant recipients have been described to carry CD27, CD38, and HLA-DR (30). Regulatory CD8+CD25+ cells expressing CTLA-4 and FOXP3 have been assumed to be involved in the efficacy of experimental treatment with a modified anti-CD3 mAb in autoimmune type 1 diabetes mellitus patients (31). HLA-E–restricted CD8+ Tregs may play a role in controlling self-reactive CD4+ T cells in this disease (32). CD8+CD25+FOXP3+ T cells exert influence over self-reactive CD4+ T cells during the course of multiple sclerosis (33). CD8+CD25+FOXP3+ suppressive T cells in colorectal cancer tissue may contribute to immune escape and disease progression (34). Expansion of CD8+FOXP3+ T cells with suppressive potential in the colorectal mucosa following infection with simian immunodeficiency virus correlates with diminished antiviral T cell response and viral control in primates (35). Unlike CD4+FOXP3+ Tregs generated in the thymus, the majority of the reported CD8+ Tregs require antigen stimulation (36). However, conditions for CD8+ Treg induction, their phenotypes and their mechanisms of action vary among reports and still remain poorly understood.
Here, we report on a novel CD8+ T-cell subset that can be distinguished from previously described Treg subphenotypes. Induction requirements include low-dose stimulation of the T-cell receptor in combination with the T-cell growth factor IL-15. The cells retain a phenotype that is classically encountered on naïve CD8+ T cells, as they are positive for CD45RA, CD28, CD27 and CCR7. The mechanisms through which they exert their suppressive function is independent from secreted cytokines and target preferentially naïve CD4+ T cells. CD8+CCR7+ Tregs regulate CD4 T cells by inhibiting very early events in the CD4+ T cell activation cascade. Specifically, CD4+ T cells suppressed by CD8+CCR7+ Tregs fail to phosphorylate the membrane-proximal adaptor molecule ZAP70 and to mobilize intracellular Ca(2+), localizing the inhibitory signal to pinnacle steps in the T-cell receptor–dependent signaling process.
Materials and Methods
Cell Isolation and Culture
Healthy individuals aged 18–85 years of age were recruited into the study. The protocol was approved by the Institutional Review Board and appropriate informed consent was obtained. PBMCs were isolated by gradient centrifugation using Lymphocyte Separation Medium (Lonza, Basel, Switzerland). For the initial dose finding studies, PBMCs (2×105/well) were stimulated with indicated concentrations of soluble anti-CD3 mAb (Orthoclone OKT3, BioLegend, San Diego, CA) and IL-15 (Invitrogen, Carlsbad, CA) in 96-well plates.
For the induction of CD8 Tregs, PBMCs were cultured in 24-well plates at a concentration of 1×106 cells/ml in RPMI medium supplemented with Pen/Strep/Glutamine, 10% FCS, 5 ng/ml recombinant IL-15 and 0.1 ng/ml of anti-CD3/OKT3. After six days, CD8+ T cells were purified by negative selection with a CD8+ T cell isolation kit II (Miltenyi Biotec), followed by separation of CD8+CCR7+ and CD8+CCR7− T cells through PE-CCR7 antibody (BD Biosciences) and anti-PE microbeads (Miltenyi Biotec). Alternatively, CD8+CCR7+ T cells from the cultures were isolated using a naïve CD8+ T cell isolation kit (Miltenyi Biotec, Bergisch Gladbach, Germany) according to the manufacturer’s guidelines. CD4+ T cells were purified from unstimulated PBMC using a naïve CD4+ T cell isolation kit (Miltenyi Biotec) and memory CD4+ T cells were obtained using a CD4+ T cell isolation kit followed by depletion of naïve CD4+ T cells using CD45RA magnetic beads (both Miltenyi Biotec). Purity of cell populations was monitored by flow cytometry and was consistently >90%.
CFSE-labeling of CD4+ T cells
Purified naïve and/or memory CD4+ T cells were labeled with 5 µM of CFSE (Invitrogen, Carlsbad, CA) for 5 min at 37°C and were washed for 30 min with warm RPMI 10% FCS at 37°C prior to use.
Suppression Assays
Suppression assays were performed by culturing CFSE-labeled naïve or memory CD4+ T cells (responder cells) and CD8+CCR7+ T cells (Tregs) at a 1:1 ratio, unless otherwise indicated. Parallel cultures contained a mixture of CD4 responder cells and CD8+CCR7− T cells at a 1:1 ratio. Cells were stimulated with anti-CD3/CD28-coupled beads (Invitrogen, Carlsbad, CA) at a 1:1 bead/cell ratio. After four days, cells were stained with CD4-PerCP and CD8-PE to discriminate each T-cell population and proliferation of CD4+ T cells was detected as the dilution of CFSE on flow cytometry. Alternatively, CD4+ T cells were stimulated with allogeneic monocytes that were derived from the donor of the CD8+CCR7+ T cells. In some experiments, anti-TGF-β Abs (R&D Systems, Minneapolis, MN), anti-IL-10 Abs (R&D Systems), anti-CCL4 Abs (R&D Systems), anti-HLA-ABC Abs (Serotec, Oxford, UK), and adenosine 2A antagonist, ZM241385 (Tocris, Bristol, UK) were added to the culture. Control cultures received isotype control antibodies. To explore the role of secreted products, CD8+CCR7+ T cells were isolated at day 6 of the induction cultures and were fixed with 4% paraformaldehyde (PFA) solution (30 min at 4°C) before coculturing them with CD4+ T cells. To study the effect of IL-2 in overcoming suppression, different concentrations of recombinant human IL-2 (GenScript, Piscataway, NJ) were added at the beginning of the culture period.
Flow cytometry
The phenotype of both CD8+ and CD4+ T cells was assessed by flow cytometry using the following anti-human mAbs: anti-CD4-FITC/APC, anti-CD8-FITC/PE/PerCP, anti-CD25-APC, anti-CD27-PE, anti-CD28-FITC, anti-CD28-PE, anti-CD40L-PE, anti-CD45RA-APC, anti-CD45RO-FITC, anti-CD56-FITC, anti-CD69-FITC, anti-CD71-PE, anti-CD103-PE, anti-CCR7-PE/PerCPCy5.5/PE-Cy7, anti-pZAP70-PE, anti-TNFR2-PE (all from BD Biosciences), anti-FOXP3-PE (Miltenyi Biotec), anti-PD-1-PE (Biolegend, San Diego, CA), anti-CTLA-4-PE, anti-GITR-APC, anti-CD122-eFluor710 (all from eBioscience, San Diego, CA). Corresponding isotype controls (all from BD Biosciences) or fluorescence-minus-one (FMO) served as controls. All flow cytometric analyses were performed on an LSR II flow cytometer (BD Biosciences) and the data were analyzed using FlowJo software.
Confocal Microscopy
CD4+CD45RA+ CD4 T cells were loaded with Fluo-4/AM (Molecular Probes, NY) as previously described (37) and mixed with anti-CD3/CD28 beads at a 1:1 ratio. CD8+CCR7+ T cells were added and cells were immediately subjected to confocal microscopy. Fluorescence images were collected using 488 nm excitation light and emission at 520–550 nm. All images were captured by a Zeiss confocal LSM 510 META (Carl Zeiss, Thornwood, NY) using a Plan-Apochromat 63x/1.4 Oil DIC objective. LSM software (Carl Zeiss), Imaris (Bitplane, St. Paul, MN) and ImageJ software (NIH, Bethesda, MD) were applied to quantify fluorescence signals.
Statistical analysis
Statistical analysis was performed with GraphPad Prism 4.03 software, using Wilcoxon matched pair tests or paired t-tests as appropriate. Differences with a p-value <0.05 were considered significant.
Results
Expansion of CD8+CCR7+ T cells with low-dose anti-CD3 and IL-15
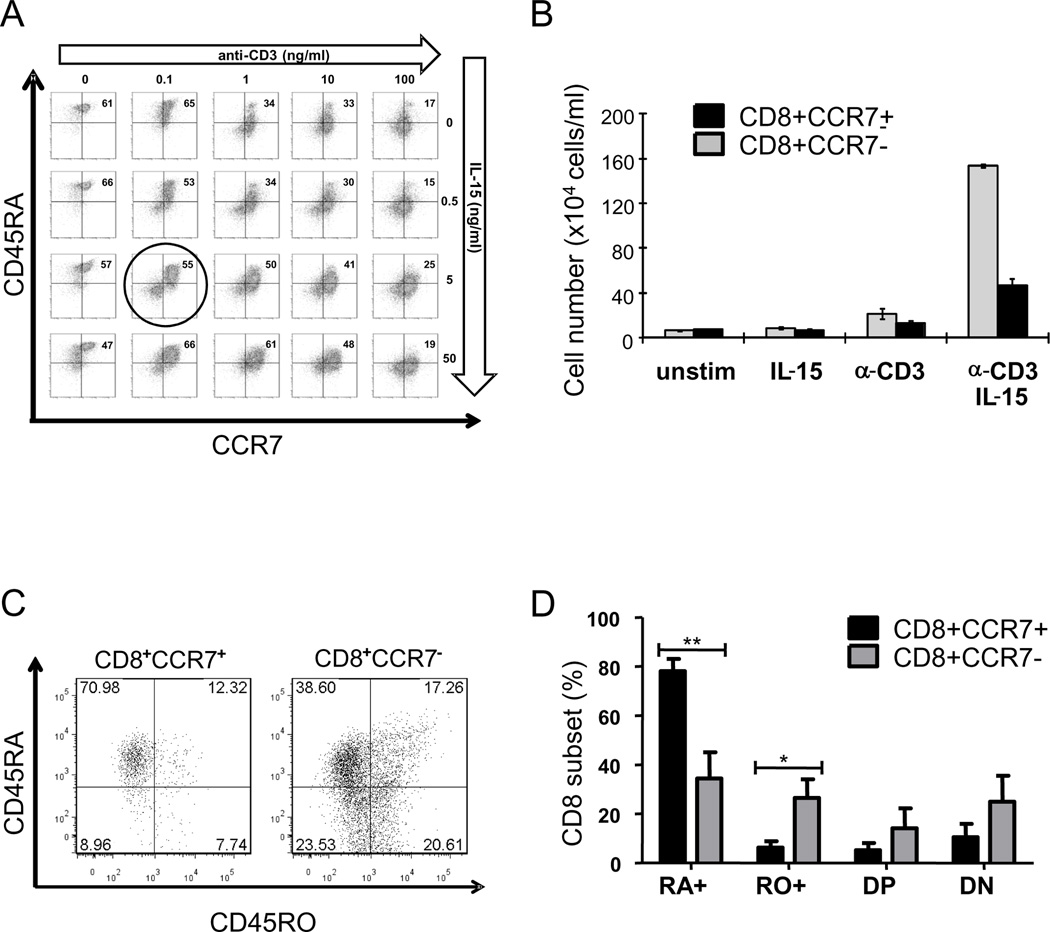
In search for CD8 subpopulations with regulatory functions, we stimulated fresh PBMCs with increasing doses of soluble anti-CD3 Abs and tested to which degree the growth factor IL-15 was able to expand CD8 subphenotypes. As shown in Fig. 1, concentrations of anti-CD3 between 1 and 100 ng/ml induced rapid conversion of naïve CD8+CD45RA+CCR7+ T cells into memory T cells. Within six days of activation, the majority of CD8 T cells lost CD45RA expression and, with the highest concentrations of anti-CD3 antibodies (100 ng/ml), also downregulated CCR7. In cultures driven with 100 ng/ml anti-CD3 only a small population of less than 20% of the CD8 T cells retained the naïve phenotype of CD8+CD45RA+CCR7+ (Fig. 1A). Interestingly, with a low-dose activation signal, naïve CD8 T cells were able to avoid conversion into the memory phenotype and continued to be positive for both CD45RA as well as CCR7. Stimulated with anti-CD3 concentrations of just 0.1 ng/ml, 55–65% of all CD8 cells were phenotypically indistinguishable from the naïve subset.
Figure 1. Induction and expansion of CD8+CCR7+ T cells.
(A). PBMCs were collected from healthy individuals, plated at 2×106 per well and stimulated with anti-CD3 Abs at increasing doses. Recombinant human IL-15 at concentrations of 0–50 ng/ml was added at the beginning of the culture period. On day 6, cells were harvested and stained with Abs specific for CD8, CD45RA and CCR7 and analyzed by flow cytometry. Conditions used in subsequent experiments are indicated by a circle. (B) Cells were cultured with (0.1 ng/ml) or without anti-CD3, with (5 ng/ml) or without IL-15 or the combination of anti-CD3 and IL-15. On day 6, cell numbers of CD8+CCR7− and CD8+CCR7+ cells were quantified by flow cytometry. Results are expressed as mean cell recovery ± SEM. (C) Cells were induced with anti-CD3 (0.1 ng/ml) and IL-15 (5 ng/ml) over a six-day culture, stained with anti-CD8, anti-CCR7, anti-CD45RA and anti CD45RO antibodies and analyzed by flow cytometry. Expression of CD45RA and CD45RO was determined on cells gated for CD8+CCR7+ (left) and CD8+CCR7− (right). A representative scatterblot is shown. (D) The percentage of CD45RA single positive (RA+), CD45RO single positive (RO+), CD45RA and CD45RO double positive (DP) and double negative (DN) cells is shown for day 6 CD8+CCR7+ (black bars) and CD8+CCR7− (gray bars) cells as the mean ± SEM of four independent experiments. ** p<0.005; * p<0.05.
Indication that the low-dose TCR-mediated signal functionally altered CD8 T cells came from experiments that demonstrated increased responsiveness to the T-cell growth factor IL-15. Nonactivated CD8 T cells were barely able to grow in response to increasing concentrations of IL-15 (Fig. 1A). Even low-dose anti-CD3 stimulation rendered the cells IL-15 responsive with a more than tenfold increase in cell numbers by day 6. Within the outgrowing cells, 75% had a CD8+CCR7− phenotype and about 25% retained the CD8+CCR7+ naïve phenotype (Fig. 1B). The expansion of CD8+CCR7+ T cells was specific for the T-cell growth factor IL-15 (Supplemental Fig. 1). Exogenous IL-2 added at doses of 50–300 U/ml failed to facilitate the induction of CD8+CCR7+ T cells. The expansion of CD8+CCR7+ T cells required signals delivered from other cells present in the mixed population of PBMC. Neither purified CD8+CCR7+ nor purified CD8+CCR7− T cells were able to multiply in cultures driven by low-dose anti-CD3 and IL-15 (Supplemental Fig. 2).
To determine whether CD8 cells stimulated with suboptimal doses of anti-CD3 (0.1 ng/ml) and IL-15 expressed both the naïve marker CD45RA as well as the memory marker CD45RO, we isolated CD8+CCR7+ and CD8+CCR7− T cells after six days of induction culture and assessed the surface phenotype by flow cytometry. The vast majority of CD8+CCR7+ cells had a naïve phenotype; only a small proportion of about 10% of the cells co-expressed CD45RO. In contrast, more than half of the CD8+CCR7− T cells had converted and acquired CD45RO expression (Fig. 1C, 1D).
Since we were particularly interested in the functional properties of CD8 T cells that maintained a naïve phenotype despite TCR-mediated and cytokine-supported stimulation, we selected the activation condition of 0.1 ng/ml soluble anti-CD3 Abs combined with 5 ng/ml IL-15 for all further experiments.
Induced CD8+CCR7+ T cells have immunosuppressive properties
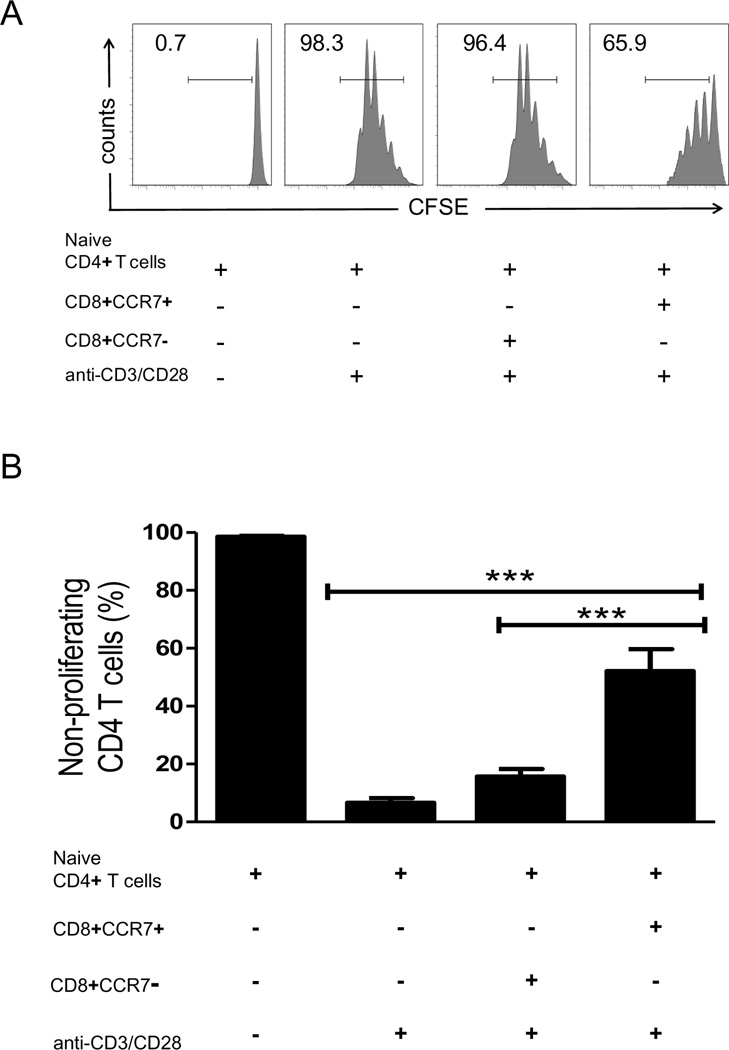
To establish a functional profile for CD8 T cells that have been induced by a combined signal of suboptimal T-cell receptor stimulation and IL-15–mediated STAT5 signaling, we compared CD8+CCR7+ and CD8+CCR7− T cells from the six-day cultures in in vitro suppression assays (Fig. 2). CFSE-labeled naïve CD4 T cells were activated with anti-CD3/CD28-coupled beads and conditions were set to obtain cell cycle entry and progression of essentially the entire population. Coculture of naïve CD4 T cells with equal numbers of autologous CD8+CCR7− T cells did not affect the growth behavior of the CD4 cells (Fig. 2A). In contrast, CD8+CCR7+ T cells were capable of suppressing clonal expansion of conventional CD4 T cells. The regulatory function of CD8+CCR7+ T cells involved the retention of a fraction of the CD4 T cells in a nondividing state. In a series of experiments, CD8+CCR7+ Tregs prevented the cell cycle entry of about 50% of CD4 T cells (Fig. 2B). Inducible CD8+CCR7+ T cells were not only highly efficient in inhibiting autologous CD4 T cells but could also downregulate the proliferative response of allogeneic naïve CD4 T cells. Allogeneic CD4 T cells stimulated by either anti-CD3-couple beads or by monocytes were susceptible to the suppressive effects of CD8+CCR7+ Tregs. In allogeneic combinations in which the stimulating antigen-presenting cells derived from the donor of the CD8 T cells, the regulatory action of CD8+CCR7+ Tregs was fully maintained (data not shown).
Figure 2. CD8+CCR7+ T cells suppress the proliferation of CD4 T cells.
CD8+CCR7− and CD8+CCR7+ T cells were generated as described in Fig. 1A. On day 6, both cell populations were purified and cocultured at a 1:1 ratio with CFSE-labeled CD4+CD45RA+ cells that were isolated from the same donor. Cocultures were stimulated with anti-CD3/CD28-coupled beads, added at a concentration of one bead per CD4 T cell. On day 4 of the coculture, proliferation of the CD4 T cells was analyzed by quantifying CFSE dilution using flow cytometry. (A) Histograms from a representative experiment are shown. Numbers represent the fraction of proliferating CD4 T cells. (B) The mean percentage ± SEM of nonproliferating CD4 T cells is shown for nine independent coculture experiments. *** p<0.0001.
Inducible CD8+CCR7+ Tregs disrupt early steps in the activation of CD4 T cells
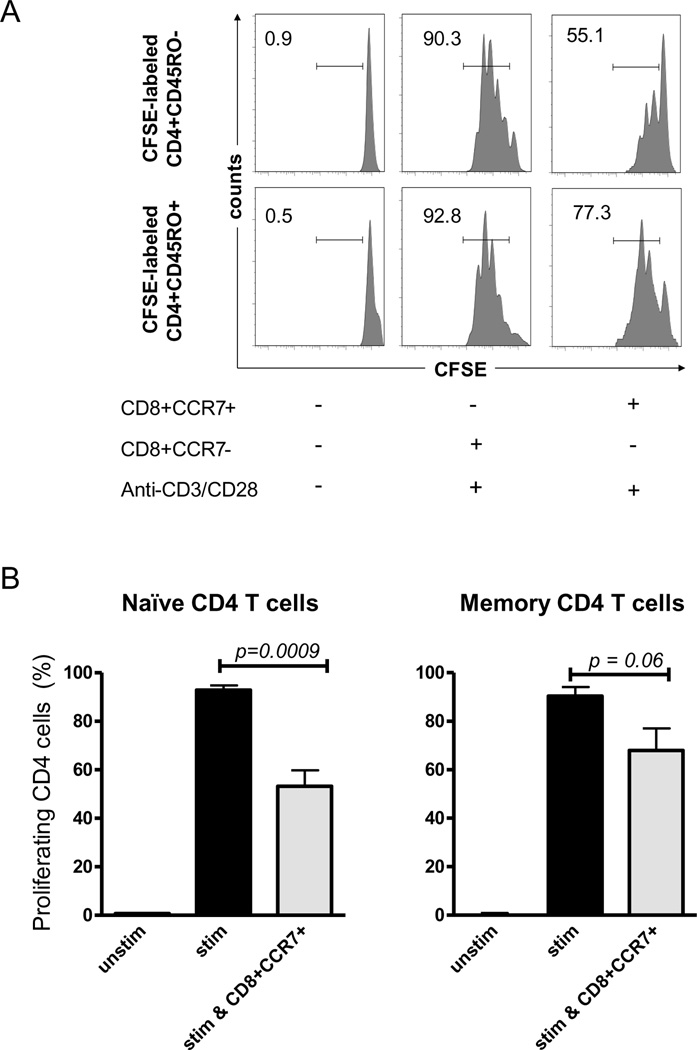
To identify the mechanisms through which CD8+CCR7+ Tregs suppress the proliferative response of CD4 T cells, we sought to delineate which CD4 T cells are susceptible to the inhibitory effects and at which stage of T cell activation CD8+CCR7+ Tregs interfere with the activation program. CD4 T cells were separated into CD4+CD45RO− naïve and CD4+CD45RO+ memory T cell populations, labeled with CFSE and cocultured with CD8+CCR7+ Tregs. Naïve CD4 T cells were consistently more susceptible to the suppressive action of the CD8 Tregs than memory CD4 T cells (Fig. 3). Whereas more than 90% of the CD4 T cells proliferated when stimulated with anti-CD3/CD28-coupled beads alone, only 55–65% of the cells entered the cell cycle in the presence of the CD8 Tregs. Memory CD4 T cells were less affected by the inhibitory action but remained partially susceptible. In the cocultures containing CD8 Tregs, 75–85% of memory CD4 T cells were able to progress through the cell cycle.
Figure 3. Naïve CD4+ T cells are more susceptible to the suppressive effect of CD8+CCR7+ Treg cells.
CD8+ CCR7− and CD8+CCR7+ T cells were purified after six days of activation with anti-CD3 (0.1 ng/ml) and IL-15 (5 ng/ml). CD4+CD45RA+ and CD4+CD45RO+ T cells derived from the same donor were labeled with CFSE and stimulated with anti-CD3/CD28 beads in the absence and presence of CD8+CCR7+ or CD8+CCR7− T cells. (A) CFSE dilution histograms are shown from a representative experiment. The frequency of proliferating CD4 T cells is indicated above each histogram. (B) Results of four independent experiments are shown as the mean ± SEM.
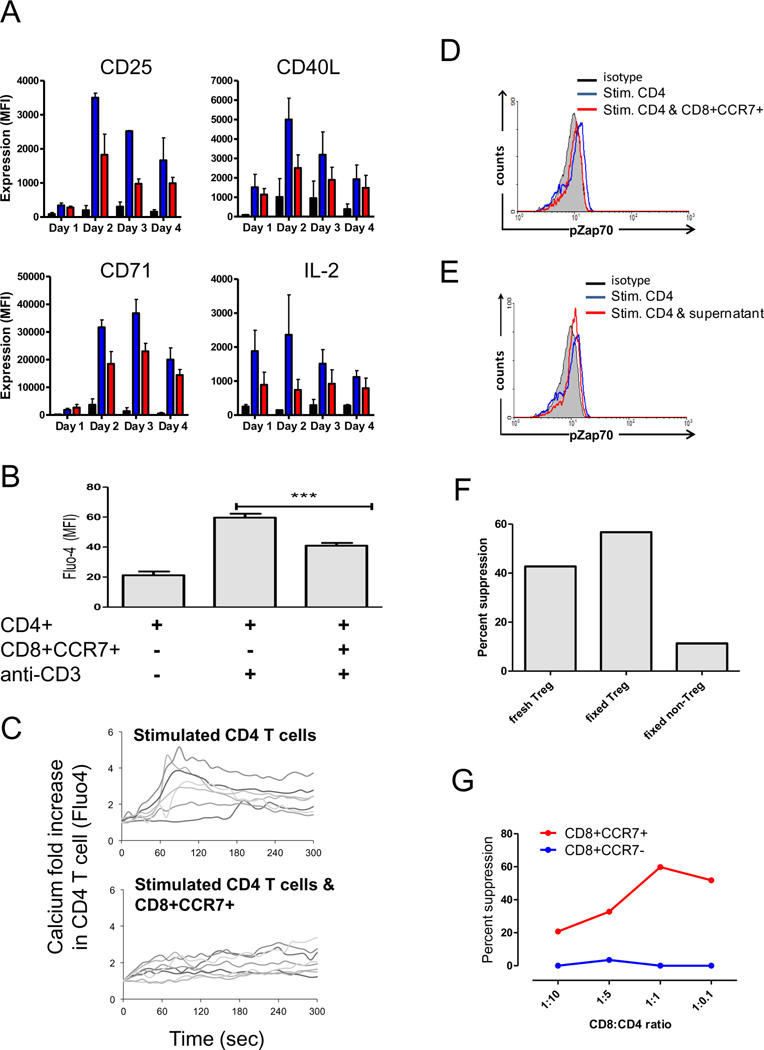
The CD8 Treg-mediated blockade of CD4 T cells entering the cell cycle suggested that the CD8 Tregs act very early in the activation cascade. Accordingly, we tested whether early steps in T cell activation are altered in the presence of CD8 Tregs. The appearance of T cell activation markers on CD4 T cells cultured with CD8+CCR7+ and CD8+CCR7− T cells were monitored by multicolor flow cytometry (Fig. 4). Stimulation of CD4 T cells with anti-CD3/CD28-coated beads induced the expected pattern of CD40L, CD25 and CD71 expression. Twenty-four hours following stimulation, the CD4 T cells began to upregulate CD40L, with peak expression occurring after 48 h. CD25 appeared on stimulated T cells after 48 h and CD71 had the slowest kinetic with maximal expression on day 3. In cultures containing CD8+CCR7+ T cells, all three activation markers were repressed. The impact of CD8+CCR7+ T cells was visible as early as 24 h when CD4 cells exposed to the CD8 Tregs had reduced expression of the early activation marker CD40L. Interference with early activation steps was confirmed by monitoring intracellular IL-2 in CD4 cells. Levels of intracellular IL-2 were reduced by 50% in CD4 T cells cocultured with CD8 Tregs (Fig. 4A).
Figure 4. CD8+CCR7+ Tregs interfere with early steps in the CD4 T cell activation cascade.
(A) Cocultures of CD8+CCR7+ Tregs and CD4+CD45RA+ T cells were set up as outlined in Fig. 2. After 24, 48, 72 and 96 h, CD4+ T cells were analyzed for the expression of early (CD25, CD40L) and late (CD71) activation markers by flow cytometry. The frequency of IL-2–producing CD4+ T cells was measured by intracellular staining. Mean MFI ± SEM is shown for three independent experiments. (B) CD4 T cells were loaded with the [Ca2+]i indicator dye Fluo-4/AM and mixed with CD8+CCR7+ Tregs (induced as described in Fig. 1) at a 1:1 ratio. After adding anti-CD3/CD28-coupled beads, cells were immediately analyzed by confocal microscopy. Mean fluorescence intensity of Fluo-4 was measured after 2 min in CD4 T cells that were unconnected to beads (left bar), CD4 T cells connected to beads (middle bar) and CD4 T cells in contact with beads as well as CD8 cells (right bar). Results from a minimum of 20 CD4 T cells for each condition are shown as mean ± SEM from three independent experiments. (C) [Ca2+]i traces recorded from individual CD4 T cells that were in contact with beads (top panel) or from individual CD4 T cells that were contacting CD8+CCR7+ T cells as well as anti-CD3-coupled beads. (D) CD8+CCR7+ Tregs were purified after six days of activation with anti-CD3 and IL-15. CD4+CD45RA+ T cells were isolated and stimulated with anti-CD3/CD28-coupled beads in the absence or presence of CD8+CCR7+ Tregs (ratio 1:1). After a 5 min incubation period, cells were fixed and stained for CD4 and phosphorylated ZAP70 and analyzed by flow cytometry. One representative histogram of pZAP70 expression in CD4+ T cells out of three independent experiments is shown. (E) Phosphorylated ZAP70 was measured in CD4 T cells, which were stimulated with anti-CD3/CD28 coated beads in the absence (blue line) or presence (red line) of supernatants harvested from CD8+CCR7+ Treg cultures. One representative example is shown. (F) PBMC were cultured for six days with anti-CD3 and IL-15 as described in Fig. 1. CD8+CCR7+ were isolated and either left untreated (fresh Tregs) or treated with PFA (fixed Treg). PFA-fixed CD8+CCR7− T cells served as control. The suppressive activity of all three Tregs populations was analyzed by mixing them at a 1:1 ratio with CD4 T cells activated by anti-CD3-coupled beads. CD4 T cell activation was quantified by measuring pZAP70 expression after 5 min of stimulation as described in Fig. 4C. Percent suppression was calculated based on pZAP70 MFI in cocultures versus pZAP70 MFI in CD4 T cells stimulated alone. One representative out of two experiments is shown. (G) CD8+CCR7+ Tregs were purified after six days of induction with anti-CD3 and IL-15. CD4+CD45RA+ T cells were isolated, stimulated with anti-CD3/CD28-coupled beads and mixed at increasing ratios with either CD8+CCR7+ Tregs (red line) or CD8+CCR7− T cells (blue line). After a 5 min incubation period, cells were fixed and stained for CD4 and phosphorylated ZAP70 and analyzed by flow cytometry. Percent suppression was calculated as in Fig. 4D and is shown for one representative out of two independent experiments for CD8 to CD4 T cell ratios of 1:10, 1:5, 1:1 and 1:0.1.
To apply a different method for the quantification of early steps in TCR-driven cellular activation, purified CD4+CD45RA+ T cells were loaded with a calcium-sensitive dye and intracellular calcium concentrations were quantified by confocal microscopy (Fig. 4B). CD4 T cells, CD8+CCR7+ Tregs and CD3/CD28-coupled beads were mixed and immediately processed for microscopic analysis. Calcium tracings were recorded over 5 min from CD4 T cells that were unconnected to beads, CD4 T cells that had established contact to a bead and CD4 T cells that were in contact to both, a bead as well as a CD8+CCR7+ Treg. Intracellular calcium concentrations were measured in a minimum of 20 CD4 T cells that were nonstimulated, were undergoing stimulation or were contacting CD8 T cells. Data from three independent experiments are presented in Fig. 4B and show that ligation of the TCR through bead-coupled antibodies increased intracellular calcium levels about threefold. In the presence of an interacting CD8 T cell, calcium fluxes in the CD4 T cells were blunted. To inhibit the increase in intracellular calcium, CD8 Treg had to establish membrane-to-membrane contact with the CD4 T cell. The configuration resulting in suppression of Ca(2+) signaling involved a “tri-cellular” arrangement with the CD4 T cell attached to a bead and to a CD8 T cell. Tracings from individual CD4 T cells that were either connected to beads only or to both, a bead as well as a CD8+CCR7+ cells are presented in Fig. 4C and show that the CD8 Tregs cells efficiently curbed the early calcium influx.
The earliest events in T cell activation occur within minutes following ligation of the T cell receptor. TCR engagement triggers a cascade of biochemical reactions with phosphorylation of immunoreceptor tyrosine-based activation motifs in the CD3 zeta chains which permits the docking and phosphorylation of the Syk-family kinase ZAP70 by the Src family protein kinase Lck. Through flow cytometric analysis of antibodies binding to phosphorylated ZAP70, membrane-proximal signals in T cell activation can be monitored. Five minutes after ligating the TCR of CD4 T cells with anti-CD3/CD28-coupled beads increased levels of pZAP70 are detectable (Fig. 4D). This accumulation process is prevented if CD8+CCR7+ T cells are added to the CD4 T cells. Only CD8+CCR7+ T cells were able to interfere with early phosphorylation events whereas CD8+CCR7− T control cells could not alter pZAP70 accumulation triggered by TCR stimulation (Fig. 4). Thus, inducible CD8+CCR7+ Tregs act on early events in CD4 T cell activation, counteracting the initial steps in TCR-dependent signaling.
As the measurement of phosphorylated ZAP70 allowed for the swift quantification of the suppressive effect, we address the question whether CD8+CCR7+ Tregs secreted soluble factors that modulated the responsiveness of CD4 T cells to TCR engagement. Supernatants harvested from activated CD8+CCR7+ Tregs had no suppressive effect on the ability of CD4 T cells to phosphorylate ZAP70 (Fig. 4E).
To investigate whether CD8 Tregs had to be stimulated to exhibit their suppressive action we examined whether fixed CD8+CCR7+ T cells displayed inhibitory activities (Fig. 4F). Postfixation CD8+CCR7+ Tregs were at least as effective in suppressing accumulation of phosphorylated ZAP70 in CD4 T cells undergoing stimulation as their unfixed counterparts. To control for nonspecific effects of cell fixation, we included fixed CD8+CCR7− T cells in the suppression assays. In the presence of such cells pZAP70 accumulation was essentially unchanged compared to the positive control (Fig. 4F).
To gain insights into the potency of CD8+CCR7+ Tregs, their suppressive activity was measured at varying CD8:CD4 ratios (Fig. 4G; Supplemental Fig. 3). CD8+CCR7+ T cells were induced over a culture period of six days as described in Fig. 1, purified and mixed with increasing numbers of naïve CD4 T cells. Fifteen minutes following the addition of anti-CD3 coupled beads, intracellular levels of pZAP70 were quantified by flow cytometry. CD8+CCR7− T cells left the activation cascade in CD4 T cells unaffected, independent from the cellular ratio (Fig. 4G). If CD4 T cells outnumbered CD8+CCR7+ Tregs by fivefold to tenfold, their response to TCR stimulation was only minimally suppressed. At a 1:1 ratio, CD8+CCR7+ Tregs became highly effective in inhibiting T cell activation by 50–60%. A further increased in the number of CD8 Treg enhanced suppression only marginally, suggesting that a 1:1 ratio of suppressor to effector cell is sufficient to facilitate almost optimal effects.
Phenotyping of CD8+CCR7+ Tregs
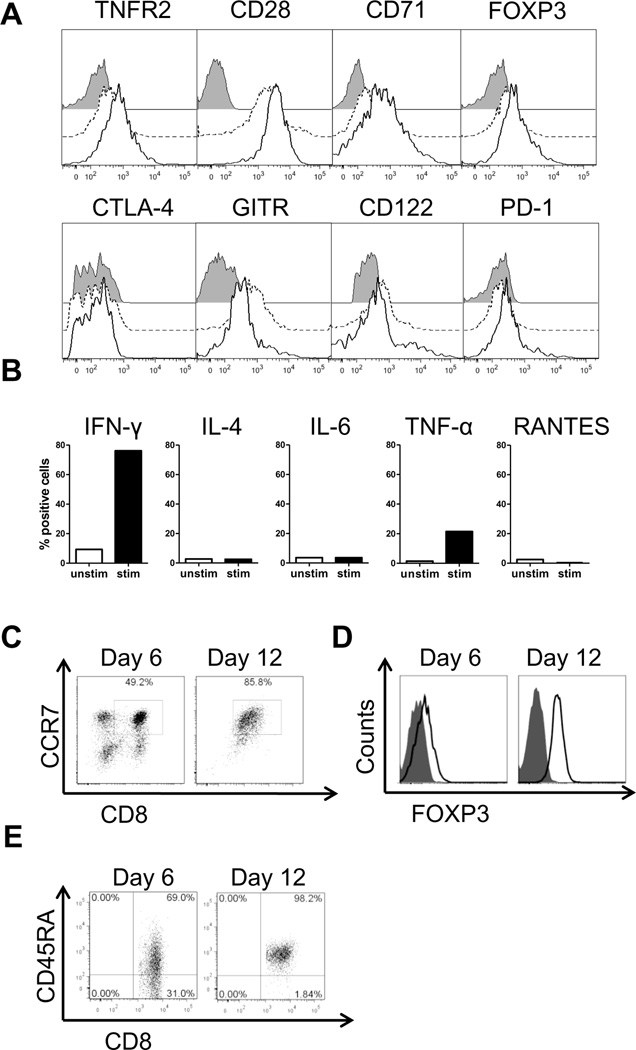
To further characterize the population of suboptimally stimulated CD8 T cells that preserve a naïve phenotype but display immunosuppressive properties, we examined whether these cells express classical regulatory cell markers. As presented in Fig. 5 and Supplemental Fig. 4, an activation period of six days with low-dose anti-CD3 and IL-15 was sufficient to induce the regulatory markers FOXP3, CD25 and CD103. The cells appeared activated and were positive for CD69 and CD71. In contrast, CTLA-4 was not expressed and no intracellular stores of CTLA-4 could be detected (data not shown). GITR and CD122 were expressed at levels similar to those on nonregulatory CD8+CCR7− cells (Fig. 5). Essentially all CD8+CCR7+ T cells remained positive for CD28 and CD27.
Figure 5. Phenotype and cytokine production profile of CD8+CCR7+ Treg cells.
(A) PBMCs were collected from healthy donors. CD8+CCR7+ T cells were induced and expanded with anti-CD3 (0.1 ng/ml) and IL-15 (5 ng/ml) as described in Fig. 1 and harvested on day 6. Cells were phenotyped by flow cytometry using antibodies to CD8, CCR7, TNFR2, CD28, CD71, FOXP3, CTLA-4, GITR, CD122 and PD-1. One histogram representative of five experiments is shown for CD8+CCR7+ Tregs (solid line) and CD8+CCR7− T cells (dashed line). Fluorescence minus one (FMO) controls are presented as filled histograms. (B) Expression of IFN-γ, IL-4, IL-6, TNF-α and RANTES was measured in nonstimulated (open bars) and PMA/ionomycin stimulated (black bars) CD8+CCR7+ Tregs using intracellular staining and flow cytometry. Cells were stimulated with PMA/ionomycin for 4 h in the presence of brefeldin A. The percentage of cytokine positive CD8+CCR7+ cells is shown for one representative out of two experiments. (C–E) PBMC were cultured with anti-CD3 (0.1 ng/ml) and IL-15 (5 ng/ml) for six days. At day 6, cells were washed and kept for an additional six days with only IL-15 (5 ng/ml). Expression of CD8 and CCR7 was analyzed at days 6 and 12 by flow cytometry and a representative scatterblot is shown in (C) FOXP3 expression in CD8+CCR7+ cells at days 6 and 12 is shown as a solid line and the FMO as a filled histogram in D. CD45RA expression on CD8+ T cells at days 6 and 12 is presented in E. For C–E, one representative example out of three independent experiments is shown.
In addition, we investigated which cytokines are being produced by the Treg population. Unstimulated CD8+CCR7+ cells produced minimal IFN-γ, IL-4, IL-6, TNF-α and RANTES (Fig. 5B). However, following stimulation with PMA and ionomycin for 4 h, CD8+CCR7+ cells contained considerable intracellular stores of IFN-γ and TNF-α. IL-4, IL-6 or RANTES were not detected, even after activation (Fig. 5B).
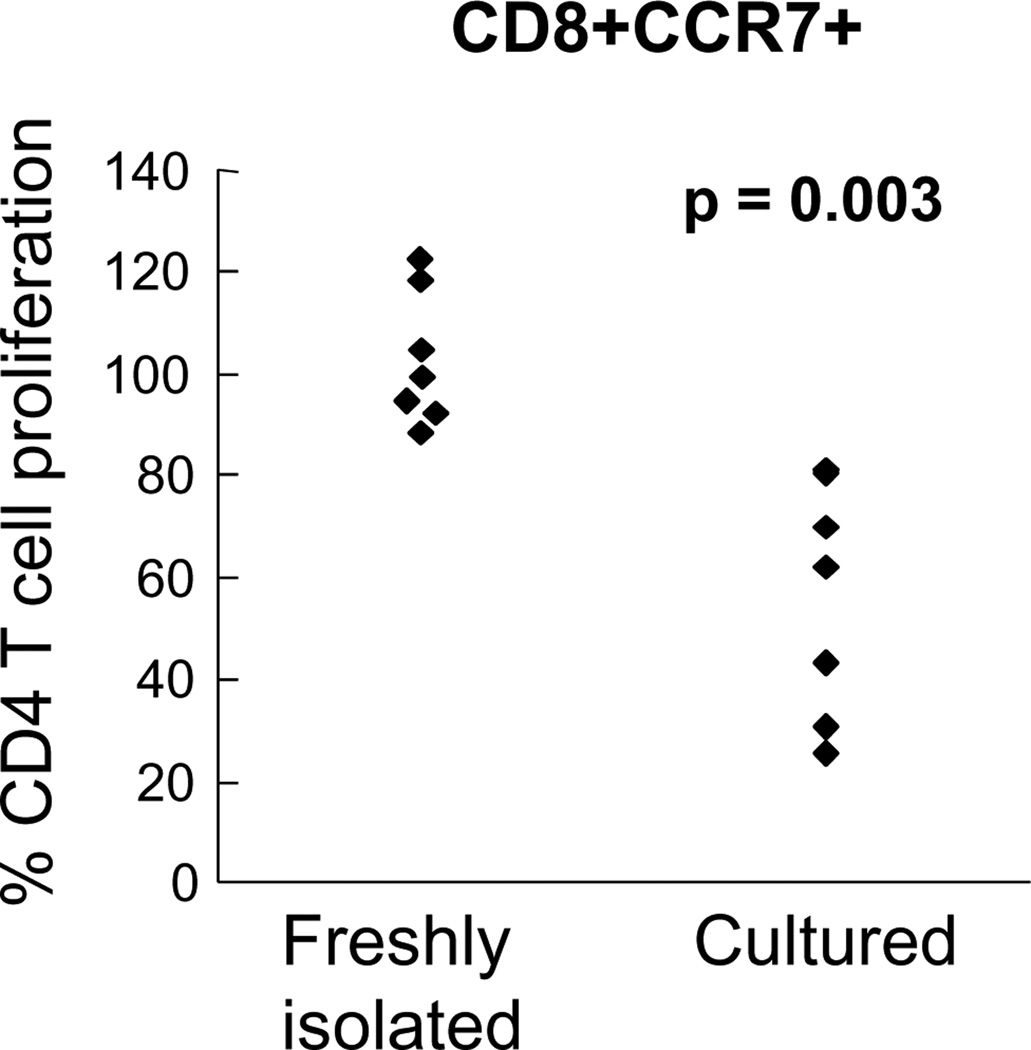
The phenotype of CD8+CCR7+ Tregs remained stable as they were continuously cultured for an additional week with only low doses of IL-15. As shown in Fig. 5C–E, continuous culture of the CD8+CCR7+ cells resulted in population that was homogenously positive for CCR7 and CD45RA. FOXP3 expression was enhanced suggesting that the culture conditions with low-dose IL-15 favored survival of T cells with regulatory function. Such continuously cultured cells displayed a similar cytokine production profile as freshly induced CD8+CCR7+ Tregs, also supporting the concept that functional capabilities were preserved beyond the induction culture. In contrast, freshly isolated CD8+CCR7+ T cells did not possess inhibitory function (Fig. 6) but rather required an induction period with TCR cross-linking and IL-15. Side-by-side comparison of fresh and cultured CD8+CCR7+ T cells revealed a clear-cut functional separation with only the stimulated CD8+CCR7+ T cells displaying regulatory abilities (Fig. 6).
Figure 6. Induction of CD8+CCR7+ Tregs.
CD8+CCR7+ T cells were either purified from fresh PBMCs or from cultures stimulated for six days with anti-CD3 (0.1 ng/ml) and IL-15 (5 ng/ml). CD4+CD45RA+ T cells were isolated from the same donor, labeled with CFSE and cocultured with the CD8 T cells and anti-CD3/CD28 beads as described above. Proliferation of CD4 T cells was assessed by CFSE dilution and relative proliferation was determined as the percentage of CD4 T cells proliferating in the absence and presence of CD8+CCR7+ Tregs.
Inducibility of CD8+CCR7+ Treg cells
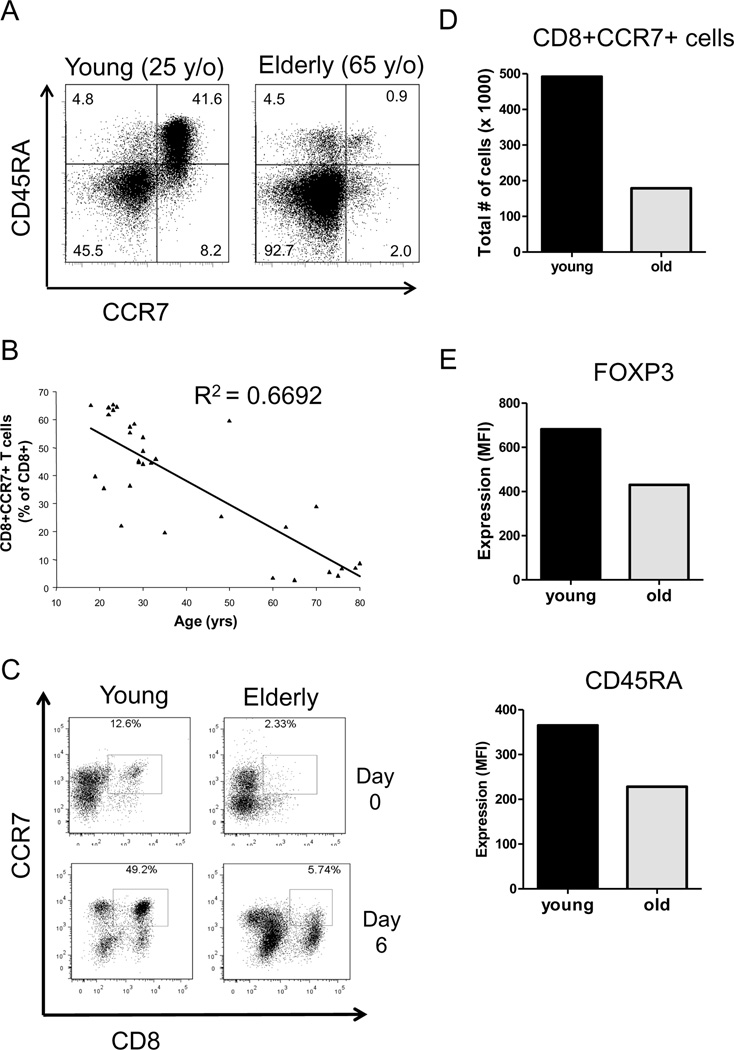
In a series of experiments, the yield of CD8+CCR7+ T cells by the end of a six-day induction period varied widely. In an effort to identify host factors that determine how many suppressive CD8+CCR7+ T cells can be generated from PBMCs, we compared donor characteristics from high- and low-yielding cultures. Donor age emerged as an important predictor in how effective CD8+CCR7+ Tregs can be induced (Fig. 7A and 7B). In the age group of 20–30 year old individuals, more than 50% of the recovered CD8 T cells had the marker profile CD8+CD45RA+CCR7+ T cells. Only a few of these young donors had less than 40% CCR7-expressing CD8 T cells by the end of the induction culture. Conversely, in most of the individuals older than 60 years of age, low-dose TCR cross-linking and culture with IL-15 yielded mostly CCR7-negative cells. Elderly donors from whom a sufficient number of CD8+CCR7+ Tregs could be generated were an exception. The frequencies of naïve CD8+CCR7+ T cells are age-dependent (38), raising the possibility that with progressive age the precursor pool for inducible CD8+CCR7+ Tregs declines. In addition, CD8+CCR7+ T cells from elderly donors expanded less vigorously than those in young donors (Fig. 7C and 7D). In young individuals, CD8+CCR7+ T cells accounted for 10–15% of cells prior to stimulation and reached up to 50% by the end of the six-day culture period. In contrast, old donors had only 2–10% of CD8+CCR7+ T cells at the beginning and upon stimulation with anti-CD3 and IL-15 such cells remained a minor subpopulation. By the end of the induction period the yield of CD8+CCR7+ T cells was about threefold higher in young versus old donors (Fig. 7D). Since FOXP3 expression has been linked to suppressive function, we compared the expression of FOXP3 in cells generated from young and old persons. As shown in Fig. 7E, FOXP3 and CD45RA expression were both lower in CD8+CCCR7+ Tregs derived from individuals older than 50 years of age.
Figure 7. Induction of CD8+CCR7+ Tregs is impaired in elderly individuals.
PBMCs were isolated from healthy individuals 18 to 81 years old and stimulated with anti-CD3 and IL-15 as described. On day 6, cells were harvested, stained with anti-CD8, anti-CD45RA and anti-CCR7 antibodies and analyzed by flow cytometry. (A) Representative results from a young (25 years; left) and an elderly donor (65 years; right) are shown. (B) The frequency of induced CD8+CCR7+ cells is plotted against the age of the donor. (C) Expression of CD8 and CCR7 is compared for a representative young and old donor on day 0 and day 6. (D) The total number of CD8+CCR7+ cells recovered at day 6 is shown for one representative experiment. (E) Expression of FOXP3 and CD45RA was analyzed by flow cytometry in day-6 CD8+CCR7+ cells derived from young (black bars) and old (grey bars) individuals. One representative out of three experiments is shown.
CD8+CCR7+ mediated suppression is independent from IL-10, TGF-β or consumption of IL-2
Many Treg populations function by releasing immunosuppressive cytokines that target neighboring T cell populations or modulate the antigen-presenting functions of accessory cells. CD8+CCR7+ T cells exerted their regulatory properties in the absence of antigen-presenting cells (Fig. 2), emphasizing that the suppression requires direct CD8-CD4 T cell communication. Furthermore, by monitoring cell numbers in cocultures, we excluded that CD8+CCR7+ T cells represent killer cells that lyse CD4 T cells. Levels of perforin and granzyme-specific transcripts in CD8+CCR7+ T cells were low (data not shown).
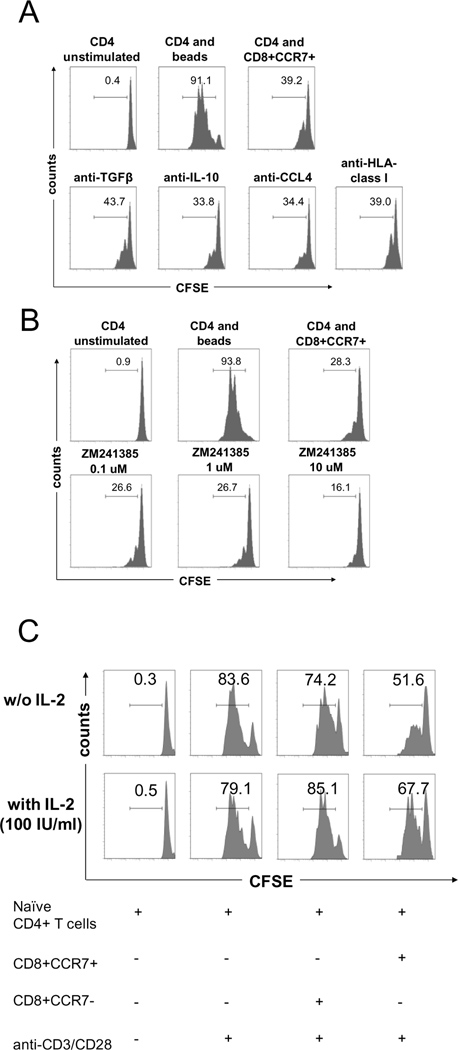
IL-10 and TGF-β are considered classical suppressive cytokines; in addition, a CD8 Treg variant has been described that depends on CCL4 to regulate T cell proliferation (5). To implicate IL-10, TGF-β or CCL4 in the mechanism of suppression by CD8+CCR7+ Tregs, optimal concentrations of antibodies were included in the CD4-CD8 T cell cocultures (Fig. 8A). Blockade of the cytokines, as well as CCL4, left the suppressive properties of the CD8+CCR7+ T cells unaffected. In parallel suppression assays, the need for HLA class I recognition was tested by blocking access to these molecules with anti-HLA class I antibodies. Similar to the cytokine blockade experiments, anti-HLA class I antibodies failed to abrogate the suppressive effects of CD8+CCR7+ Tregs. Finally, we explored whether inducible CD8+CCR7+ T cells release adenosine to trigger accumulation of intracellular cAMP as a means of immunosuppression. In these experiments, adenosine receptors were blocked with the small molecule inhibitor ZM241385. Increasing doses of ZM241385 (0.1–10 µM) had no effect on the ability of CD8 Tregs to downregulate CD4 T cell proliferation (Fig. 8B).
Figure 8. CD8+CCR7+ Treg-mediated suppression is independent of TGF-β, IL-10, CCL4, adenosine or HLA-class I recognition.
CD8+CCR7+ Tregs and CD4+CD45RA+ T cells were prepared and cocultured as outlined in Fig. 2. Proliferation of CD4 T cells was quantified by CFSE dilution after 96 h of coculture. (A) Abs reactive to TGF-β (100 μg/ml), IL-10 (100 μg/ml), CCL4 (100 μg/ml) or HLA-class I molecules (100 μg/ml) were added at the beginning of the cocultures. (B) Varying doses of the small molecule inhibitor ZM241385 which blocks the adenosine A2A receptor were added to the cocultures at day 0. (C) Cocultures were supplemented with exogenous IL-2 at the beginning of the culture period. Results for 100 U/ml of recombinant human IL-2 are shown.
As IL-2 is required to amplify and sustain T cell proliferation following TCR ligation, we examined whether supplementation of exogenous IL-2 could alleviate the suppressive properties of CD8+CCR7+ T cells. Concentrations of supplemented IL-2 at 25 U/ml had no effect at all and could not overcome CD8 Treg-mediated inhibition of CD4 T cell proliferation (data not shown). High doses of IL-2 at 100 U/ml increased cell cycle progression of proliferating cells; however, a large fraction of cells remained quiescent (Fig. 8C), suggesting that the exogenous IL-2 allowed expansion of CD4 T cells that had entered the cell cycle but could not break CD8+CCR7+ Treg-mediated suppression. These results are consistent with the observation that CD8+CCR7+ T cells interfere with early TCR signaling events.
Discussion
Here, we describe a novel subset of CD8 T cells which displays regulatory function. In distinction to other regulatory T cell populations, the CD8 Tregs characterized in this study have a naïve phenotype and maintain CCR7 expression. CD8+CCR7+ Tregs can be reliably induced by stimulation with a very low dose of TCR cross-linking anti-CD3 Abs. Functionally, they suppress CD4+ T cell activation and proliferation by interfering with very early steps in the T-cell receptor activation cascade, upstream of ZAP70 phosphorylation. Their suppressive action is independent of IL-10 and TGF-β release. CD8+CCR7+ Tregs conditioned for six days with low-dose TCR cross-linking are distinct from circulating CD8+CCR7+ T cells, as freshly isolated CD8+CCR7+ T cells have no suppressive capacity and do not express any of the regulatory markers present on the adaptive CD8+ Tregs. Adaptive CD8+ Tregs are generated from PBMCs through a bona fide induction mechanism and are not merely expanded from a pre-existing CD8+CCR7+ regulatory T cell population. A strong correlation between donor age and inducibility of these anti-inflammatory CD8 T cells suggests a potentially important immunoregulatory role for CD8+CCR7+ Tregs in the process of immunosenescence.
The CD8+CCR7+ Tregs described here are a novel population, clearly distinct from CD8 suppressor T cells characterized previously (36, 39–46). Murine CD8 Treg populations have been subdivided into Qa-1 restricted and non-Qa-1 restricted subsets. Qa-1 restricted Tregs recognize the mouse homologue of HLA-E, a molecule implicated in the presentation of self as well as foreign antigens (47). Non-Qa-1 restricted murine CD8 Tregs have been phenotyped as CD8+CD28− and function by interfering with antigen-presenting cells (48). CD8+CCR7+ Tregs identified in the current study overlap with previously described human Tregs in that they express the classical regulatory marker FOXP3 (49), but their naïve phenotype and their mechanism-of-action assigns them to a novel CD8 subset. Based on phenotypic profiling, CD8+CCR7+ Tregs are reminiscent of a recently described population of long-lived human memory T-cell population that displays enhanced self-renewal and multipotent capacity (50). These cells, which have been designated as memory stem T cells (Tscm), reside within the CD45RO−, CCR7+, CD45RA+, CD122+ T-cell compartment and thus display remarkable phenotypic similarities to the regulatory T cells described here. However, there are distinguishing features which separate Tscm and Tregs. Most significantly, Tscm represent a clonally expanded primordial memory subset which emerges after antigenic stimulation and possesses increased proliferative and reconstitutive capacities. Tscm exhibit superior anti-tumor responses in vivo making it very unlikely that they have immunosuppressive functions.
Adaptive CD8+CCR7+ Tregs are promptly induced in PBMCs of young individuals but the yield of induction is low in individuals during the second half of life. Loss of this population in donors over the age of 65 is quite dramatic with over 90% reduction compared to younger adults. While humans may carry multiple different Treg populations that can possibly compensate for each other, the almost complete loss of CD8+CCR7+ Tregs in the elderly is remarkable. Aging of the immune system is a complex process in which the reserve of naïve T cells dwindles, memory T cell populations expand and excess proinflammatory cytokines are produced by immune and nonimmune cells (51–53). Some inflammatory syndromes, such as large vessel vasculitis, occur almost exclusively in individuals older than 50 years of age (54).
Other inflammatory diseases display features of premature immune aging, especially rheumatoid arthritis and atherosclerosis. Molecular deficiencies implicated in the link between inflammation and immune aging include DNA damage repair pathways and telomeric maintenance and protection (55–57). In essence, the elderly is prone to unopposed inflammation and many of the age-related pathologies are characterized by inflammatory tissue damage. In that setting, it may be of particular importance that the elderly essentially lacks the ability to generate anti-inflammatory CD8+CCR7+ T cells. Increased inflammation in the elderly manifests with both a rise in circulating cytokines, as well as aberrant infiltration, and activation of immune cells. Progressive age is associated with a steady decline in the frequency of CD8 naïve T cells whereas end-differentiated CD8+CD28− T cells account for a considerable proportion of the CD8 compartment (38, 58). Frequencies of CD8+CD28− T cells are a potent predictor of vaccine nonresponsiveness in the elderly, emphasizing the functional importance of replacement of naïve CD8 T cells by memory/effector cells (59). The inability to induce CD8+CCR7+ Tregs is disproportionally more impaired than the decline in the naïve CD8 population, suggesting that aging individuals may selectively lose precursor cells that give rise to CD8+CCR7+ Tregs. In pilot studies, we have not been able to induce CD8+CCR7+ Tregs from purified CD8+CD45RO+ memory T cells. It is also possible that age-related defects in cellular metabolism jeopardize the survival of CD8 T cells that differentiate into Tregs.
Generation of CD8+CCR7+ Tregs was greatly enhanced in the presence of IL-15, suggesting that the STAT5 signaling pathway has a role (60). STAT5 has been implicated in regulating FOXP3 transcription (61). However, IL-2 failed to promote the induction of CD8+CCR7+ Tregs, despite its ability to initiate the STAT5 signaling pathway; possibly due to its stronger ability to co-activate PI3K. In the current experiments, induction of CD8+CCR7+ Tregs was strongly affected by combinatorial signals transmitted through the T cell receptor and the STAT5 pathway. Both of these pathways could be subject to age-related changes that impact the potential of the aging donor to generate sufficient anti-inflammatory CD8 Tregs. The IL-15 dependence of the CD8+CCR7+ Tregs raises the question whether they are a counterpart of the murine CD8+CD44+CD122+ Tregs that have recently been described to be involved in controlling autoimmunity in vivo (62). IL-15 deficient mice lack such CD44+CD122+ Tregs. In variance to the current CD8+CCR7+ Tregs, IL-15–dependent murine CD8 Tregs express a typical memory marker (CD44) and are markedly increased upon immunization with KLH/CFA, suggesting that they represent a memory T cell population.
For a number of previously described CD8+ Tregs, the mechanism of CD4+ T cell suppression has been defined to be either IL-10 (63, 64), TGF-β (65), or CCL4-dependent (5). This new subset of adaptive CD8+ Tregs we describe here differs from the previously described ones, as it does not suppress through any of the conventional routes (IL-10, TGF-β or CCL4). Additionally, transfer of supernatant from activated CD8+ Tregs had no suppressive effect on CD4+ T cells, further supporting the concept that suppression is not mediated through a secreted protein. These data suggest that the mechanism of suppression is likely to be cell-contact dependent. Furthermore, the fact that CD8+CCR7+ Tregs were able to suppress ZAP70 phosphorylation as early as 5–10 min after CD3/CD28 stimulation indicates that suppression does not require novel protein synthesis. Together, these findings strongly argue against a secreted mediator mediating suppression, corroborating our data that blockade of IL-10, TGF-β and CCL4 had no effect on CD4 T cell suppression. Instead, suppression is more likely to be mediated through a receptor-ligand pair. The interference of CD8 Tregs with proximal steps of TCR-dependent signaling was confirmed by quantifying TCR-driven rises in intracellular calcium, a function regulated by activated phospholipase C, which is immediately distal from ZAP70 phosphorylation. CD8 Tregs that could make contact to a CD4 T cell were able to blunt the typical increase in cytoplasmic Ca(2+). These confocal imaging experiments established that CD8 Tregs form a membrane interface with the CD4 T cell they regulate and confirmed that the CD8 Tregs did not kill the CD4 T cell. Suppression mediated via a direct T-T cell contact, not involving the antigen-presenting cells or killing, is a novel mechanism that requires additional studies to identify the relevant molecular events.
Both naïve and memory CD4+ T cells are sensitive to the suppression, but, notably, suppression was consistently more pronounced in naïve CD4+ T cells. As naïve and memory CD4+ T cells are quite distinct from each other in terms of phenotype and function, increased susceptibility of naïve CD4+ T cells to CD8+ T cell suppression could be due to differential expression of a receptor through which suppression is mediated. Preference to inhibit naïve over memory CD4 T cells appears to be a unique feature of CD8+CCR7+ Tregs. CD4+ Tregs are known to suppress naïve and memory CD4+ T cells equally well.
Like other Treg populations, CD8+CCR7+ Tregs express the IL-2 receptor α-chain (CD25) and the observed CD4+ T cell suppression could be due to the consumption of IL-2 by the CD8+ Tregs. Consumption of IL-2 has been proposed to be the mechanism by which CD4+ Tregs suppress responder CD4+ T cells (66). In our system, addition of high amounts of exogenous IL-2 partly alleviated CD4+ T cell suppression. The observed effect was, however, very moderate indicating that IL-2 consumption is not the main mechanism by which CD8+CCR7+ Tregs suppress. Instead, it is possible that exogenous IL-2 simply augments the expansion of the CD4+ T cells that escaped CD8+CCR7+ Treg-suppression in the first place. The CD4-CD8 cocultures consistently contained a small subset of CD4+ T cells (5–10%) which appeared able to proliferate despite the presence of CD8+ Tregs. Phenotypic differences between susceptible and resistant CD4 T cells may be an important clue towards a molecular definition of the molecules facilitating the “veto signal” to CD4 T cells.
Physiologic relevance of adaptive CD8+CCR7− T cells is suggested by the wide variation in the ability of donors to give rise to the population and the strict influence of donor age. Whether adaptive CD8+CCR7+ T cells exist in vivo, how they are induced and where they function remains to be elucidated. Conditions of low-antigen exposure may exist in chronically infected hosts and it has been proposed that Treg may critically define the host microbe relationship during chronic infection (27). The expression of CCR7 predicts that the Tregs described here would preferentially mimic the trafficking pattern of naïve cells and home to primary lymphoid tissues. The homing chemokines CCL19 and CCL21, which can recruit CCR7-expressing cells are also abundantly present in tertiary lymphoid structures, such as in the chronically inflamed joint of patients with rheumatoid arthritis (67, 68) suggesting possible involvement of CD8+CCR7+ T cells in the regulation of autoimmunity (69). Studies in lymphoid tissues and autoimmune lesions are needed to provide insight into the role that CD8+CCR7+ Tregs have in normal and pathogenic immune responses.
Supplementary Material
Acknowledgments
This work was supported by grants from the National Institutes of Health (AI 44142, AR 42527, EY 11916, HL 58000, AI 57266, and AI 90019).
Footnotes
Disclosures
The authors have no financial conflicts of interest.
REFERENCES
- 1.Sakaguchi S, Yamaguchi T, Nomura T, Ono M. Regulatory T cells and immune tolerance. Cell. 2008;133:775–787. doi: 10.1016/j.cell.2008.05.009. [DOI] [PubMed] [Google Scholar]
- 2.Belkaid Y, Tarbell K. Regulatory T cells in the control of host-microorganism interactions (*) Annu Rev Immunol. 2009;27:551–589. doi: 10.1146/annurev.immunol.021908.132723. [DOI] [PubMed] [Google Scholar]
- 3.Zheng Y, Rudensky AY. Foxp3 in control of the regulatory T cell lineage. Nat Immunol. 2007;8:457–462. doi: 10.1038/ni1455. [DOI] [PubMed] [Google Scholar]
- 4.Ziegler SF. FOXP3: of mice and men. Annu Rev Immunol. 2006;24:209–226. doi: 10.1146/annurev.immunol.24.021605.090547. [DOI] [PubMed] [Google Scholar]
- 5.Wang J, Ioan-Facsinay A, van der Voort EI, Huizinga TW, Toes RE. Transient expression of FOXP3 in human activated nonregulatory CD4+ T cells. Eur J Immunol. 2007;37:129–138. doi: 10.1002/eji.200636435. [DOI] [PubMed] [Google Scholar]
- 6.Lio CW, Hsieh CS. A two-step process for thymic regulatory T cell development. Immunity. 2008;28:100–111. doi: 10.1016/j.immuni.2007.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hsieh CS, Zheng Y, Liang Y, Fontenot JD, Rudensky AY. An intersection between the self-reactive regulatory and nonregulatory T cell receptor repertoires. Nat Immunol. 2006;7:401–410. doi: 10.1038/ni1318. [DOI] [PubMed] [Google Scholar]
- 8.Zheng SG, Wang JH, Stohl W, Kim KS, Gray JD, Horwitz DA. TGF-beta requires CTLA-4 early after T cell activation to induce FoxP3 and generate adaptive CD4+CD25+ regulatory cells. J Immunol. 2006;176:3321–3329. doi: 10.4049/jimmunol.176.6.3321. [DOI] [PubMed] [Google Scholar]
- 9.Apostolou I, von Boehmer H. In vivo instruction of suppressor commitment in naive T cells. J Exp Med. 2004;199:1401–1408. doi: 10.1084/jem.20040249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kretschmer K, Apostolou I, Hawiger D, Khazaie K, Nussenzweig MC, von Boehmer H. Inducing and expanding regulatory T cell populations by foreign antigen. Nat Immunol. 2005;6:1219–1227. doi: 10.1038/ni1265. [DOI] [PubMed] [Google Scholar]
- 11.Fantini MC, Becker C, Monteleone G, Pallone F, Galle PR, Neurath MF. Cutting edge: TGF-beta induces a regulatory phenotype in CD4+CD25− T cells through Foxp3 induction and down-regulation of Smad7. J Immunol. 2004;172:5149–5153. doi: 10.4049/jimmunol.172.9.5149. [DOI] [PubMed] [Google Scholar]
- 12.Curotto de Lafaille MA, Lino AC, Kutchukhidze N, Lafaille JJ. CD25− T cells generate CD25+Foxp3+ regulatory T cells by peripheral expansion. J Immunol. 2004;173:7259–7268. doi: 10.4049/jimmunol.173.12.7259. [DOI] [PubMed] [Google Scholar]
- 13.Mucida D, Kutchukhidze N, Erazo A, Russo M, Lafaille JJ, Curotto de Lafaille MA. Oral tolerance in the absence of naturally occurring Tregs. J Clin Invest. 2005;115:1923–1933. doi: 10.1172/JCI24487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zheng SG, Wang JH, Gray JD, Soucier H, Horwitz DA. Natural and induced CD4+CD25+ cells educate CD4+CD25− cells to develop suppressive activity: the role of IL-2, TGF-beta, and IL-10. J Immunol. 2004;172:5213–5221. doi: 10.4049/jimmunol.172.9.5213. [DOI] [PubMed] [Google Scholar]
- 15.Hori S. Developmental plasticity of Foxp3+ regulatory T cells. Curr Opin Immunol. 2010;22:575–582. doi: 10.1016/j.coi.2010.08.004. [DOI] [PubMed] [Google Scholar]
- 16.Horwitz DA, Zheng SG, Gray JD. Natural and TGF-beta-induced Foxp3(+)CD4(+) CD25(+) regulatory T cells are not mirror images of each other. Trends Immunol. 2008;29:429–435. doi: 10.1016/j.it.2008.06.005. [DOI] [PubMed] [Google Scholar]
- 17.Feuerer M, Hill JA, Kretschmer K, von Boehmer H, Mathis D, Benoist C. Genomic definition of multiple ex vivo regulatory T cell subphenotypes. Proc Natl Acad Sci U S A. 2010;107:5919–5924. doi: 10.1073/pnas.1002006107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vignali DA, Collison LW, Workman CJ. How regulatory T cells work. Nat Rev Immunol. 2008;8:523–532. doi: 10.1038/nri2343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shevach EM. Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity. 2009;30:636–645. doi: 10.1016/j.immuni.2009.04.010. [DOI] [PubMed] [Google Scholar]
- 20.Yamaguchi T, Wing JB, Sakaguchi S. Two modes of immune suppression by Foxp3(+) regulatory T cells under inflammatory or non-inflammatory conditions. Semin Immunol. 2011;23:424–430. doi: 10.1016/j.smim.2011.10.002. [DOI] [PubMed] [Google Scholar]
- 21.Ernst PB, Garrison JC, Thompson LF. Much ado about adenosine: adenosine synthesis and function in regulatory T cell biology. J Immunol. 2010;185:1993–1998. doi: 10.4049/jimmunol.1000108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Joosten SA, van Meijgaarden KE, Savage ND, de Boer T, Triebel F, van der Wal A, de Heer E, Klein MR, Geluk A, Ottenhoff TH. Identification of a human CD8+ regulatory T cell subset that mediates suppression through the chemokine CC chemokine ligand 4. Proc Natl Acad Sci U S A. 2007;104:8029–8034. doi: 10.1073/pnas.0702257104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sakaguchi S, Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T. Regulatory T cells: how do they suppress immune responses? Int Immunol. 2009;21:1105–1111. doi: 10.1093/intimm/dxp095. [DOI] [PubMed] [Google Scholar]
- 24.Kapp JA. Special regulatory T-cell review: Suppressors regulated but unsuppressed. Immunology. 2008;123:28–32. doi: 10.1111/j.1365-2567.2007.02773.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ottenhoff TH, Elferink DG, Klatser PR, de Vries RR. Cloned suppressor T cells from a lepromatous leprosy patient suppress Mycobacterium leprae reactive helper T cells. Nature. 1986;322:462–464. doi: 10.1038/322462a0. [DOI] [PubMed] [Google Scholar]
- 26.Modlin RL, Kato H, Mehra V, Nelson EE, Fan XD, Rea TH, Pattengale PK, Bloom BR. Genetically restricted suppressor T-cell clones derived from lepromatous leprosy lesions. Nature. 1986;322:459–461. doi: 10.1038/322459a0. [DOI] [PubMed] [Google Scholar]
- 27.Joosten SA, Ottenhoff TH. Human CD4 and CD8 regulatory T cells in infectious diseases and vaccination. Hum Immunol. 2008;69:760–770. doi: 10.1016/j.humimm.2008.07.017. [DOI] [PubMed] [Google Scholar]
- 28.Cosmi L, Liotta F, Lazzeri E, Francalanci M, Angeli R, Mazzinghi B, Santarlasci V, Manetti R, Vanini V, Romagnani P, Maggi E, Romagnani S, Annunziato F. Human CD8+CD25+ thymocytes share phenotypic and functional features with CD4+CD25+ regulatory thymocytes. Blood. 2003;102:4107–4114. doi: 10.1182/blood-2003-04-1320. [DOI] [PubMed] [Google Scholar]
- 29.Uss E, Rowshani AT, Hooibrink B, Lardy NM, van Lier RA, ten Berge IJ. CD103 is a marker for alloantigen-induced regulatory CD8+ T cells. J Immunol. 2006;177:2775–2783. doi: 10.4049/jimmunol.177.5.2775. [DOI] [PubMed] [Google Scholar]
- 30.Colovai AI, Mirza M, Vlad G, Wang S, Ho E, Cortesini R, Suciu-Foca N. Regulatory CD8+CD28− T cells in heart transplant recipients. Hum Immunol. 2003;64:31–37. doi: 10.1016/s0198-8859(02)00742-5. [DOI] [PubMed] [Google Scholar]
- 31.Bisikirska B, Colgan J, Luban J, Bluestone JA, Herold KC. TCR stimulation with modified anti-CD3 mAb expands CD8+ T cell population and induces CD8+CD25+ Tregs. J Clin Invest. 2005;115:2904–2913. doi: 10.1172/JCI23961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jiang H, Canfield SM, Gallagher MP, Jiang HH, Jiang Y, Zheng Z, Chess L. HLA-E-restricted regulatory CD8(+) T cells are involved in development and control of human autoimmune type 1 diabetes. J Clin Invest. 2010;120:3641–3650. doi: 10.1172/JCI43522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Correale J, Villa A. Role of CD8+ CD25+ Foxp3+ regulatory T cells in multiple sclerosis. Ann Neurol. 2010;67:625–638. doi: 10.1002/ana.21944. [DOI] [PubMed] [Google Scholar]
- 34.Chaput N, Louafi S, Bardier A, Charlotte F, Vaillant JC, Menegaux F, Rosenzwajg M, Lemoine F, Klatzmann D, Taieb J. Identification of CD8+CD25+Foxp3+ suppressive T cells in colorectal cancer tissue. Gut. 2009;58:520–529. doi: 10.1136/gut.2008.158824. [DOI] [PubMed] [Google Scholar]
- 35.Nigam P, Velu V, Kannanganat S, Chennareddi L, Kwa S, Siddiqui M, Amara RR. Expansion of FOXP3+ CD8 T cells with suppressive potential in colorectal mucosa following a pathogenic simian immunodeficiency virus infection correlates with diminished antiviral T cell response and viral control. J Immunol. 2010;184:1690–1701. doi: 10.4049/jimmunol.0902955. [DOI] [PubMed] [Google Scholar]
- 36.Suzuki M, Konya C, Goronzy JJ, Weyand CM. Inhibitory CD8+ T cells in autoimmune disease. Hum Immunol. 2008;69:781–789. doi: 10.1016/j.humimm.2008.08.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pryshchep S, Goronzy JJ, Parashar S, Weyand CM. Insufficient deactivation of the protein tyrosine kinase lck amplifies T-cell responsiveness in acute coronary syndrome. Circ Res. 2010;106:769–778. doi: 10.1161/CIRCRESAHA.109.206052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Czesnikiewicz-Guzik M, Lee WW, Cui D, Hiruma Y, Lamar DL, Yang ZZ, Ouslander JG, Weyand CM, Goronzy JJ. T cell subset-specific susceptibility to aging. Clin Immunol. 2008;127:107–118. doi: 10.1016/j.clim.2007.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Filaci G, Fenoglio D, Indiveri F. CD8(+) T regulatory/suppressor cells and their relationships with autoreactivity and autoimmunity. Autoimmunity. 2011;44:51–57. doi: 10.3109/08916931003782171. [DOI] [PubMed] [Google Scholar]
- 40.Wilson RW, Chenggis ML, Unger ER. Longitudinal study of human papillomavirus infection of the female urogenital tract by in situ hybridization. Arch Pathol Lab Med. 1990;114:155–159. [PubMed] [Google Scholar]
- 41.Lu L, Cantor H. Generation and regulation of CD8(+) regulatory T cells. Cell Mol Immunol. 2008;5:401–406. doi: 10.1038/cmi.2008.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Smith TR, Kumar V. Revival of CD8+ Treg-mediated suppression. Trends Immunol. 2008;29:337–342. doi: 10.1016/j.it.2008.04.002. [DOI] [PubMed] [Google Scholar]
- 43.Vlad G, Chang CC, Colovai AI, Vasilescu ER, Cortesini R, Suciu-Foca N. Membrane and soluble ILT3 are critical to the generation of T suppressor cells and induction of immunological tolerance. Int Rev Immunol. 2010;29:119–132. doi: 10.3109/08830180903281185. [DOI] [PubMed] [Google Scholar]
- 44.Konya C, Goronzy JJ, Weyand CM. Treating autoimmune disease by targeting CD8(+) T suppressor cells. Expert Opin Biol Ther. 2009;9:951–965. doi: 10.1517/14712590903020759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kapp JA, Bucy RP. CD8+ suppressor T cells resurrected. Hum Immunol. 2008;69:715–720. doi: 10.1016/j.humimm.2008.07.018. [DOI] [PubMed] [Google Scholar]
- 46.Skaggs BJ, Singh RP, Hahn BH. Induction of immune tolerance by activation of CD8+ T suppressor/regulatory cells in lupus-prone mice. Hum Immunol. 2008;69:790–796. doi: 10.1016/j.humimm.2008.08.284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sarantopoulos S, Lu L, Cantor H. Qa-1 restriction of CD8+ suppressor T cells. J Clin Invest. 2004;114:1218–1221. doi: 10.1172/JCI23152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jiang H, Chess L. An integrated view of suppressor T cell subsets in immunoregulation. J Clin Invest. 2004;114:1198–1208. doi: 10.1172/JCI23411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jarvis LB, Matyszak MK, Duggleby RC, Goodall JC, Hall FC, Gaston JS. Autoreactive human peripheral blood CD8+ T cells with a regulatory phenotype and function. Eur J Immunol. 2005;35:2896–2908. doi: 10.1002/eji.200526162. [DOI] [PubMed] [Google Scholar]
- 50.Gattinoni L, Lugli E, Ji Y, Pos Z, Paulos CM, Quigley MF, Almeida JR, Gostick E, Yu Z, Carpenito C, Wang E, Douek DC, Price DA, June CH, Marincola FM, Roederer M, Restifo NP. A human memory T cell subset with stem cell-like properties. Nat Med. 2011;17:1290–1297. doi: 10.1038/nm.2446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Aspinall R, Goronzy JJ. Immune senescence. Curr Opin Immunol. 2010;22:497–499. doi: 10.1016/j.coi.2010.07.002. [DOI] [PubMed] [Google Scholar]
- 52.Buchholz VR, Neuenhahn M, Busch DH. CD8+ T cell differentiation in the aging immune system: until the last clone standing. Curr Opin Immunol. 2011;23:549–554. doi: 10.1016/j.coi.2011.05.002. [DOI] [PubMed] [Google Scholar]
- 53.Weng NP, Akbar AN, Goronzy J. CD28(−) T cells: their role in the age-associated decline of immune function. Trends Immunol. 2009;30:306–312. doi: 10.1016/j.it.2009.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mohan SV, Liao YJ, Kim JW, Goronzy JJ, Weyand CM. Giant cell arteritis: immune and vascular aging as disease risk factors. Arthritis Res Ther. 2011;13:231. doi: 10.1186/ar3358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Weyand CM, Fujii H, Shao L, Goronzy JJ. Rejuvenating the immune system in rheumatoid arthritis. Nat Rev Rheumatol. 2009;5:583–588. doi: 10.1038/nrrheum.2009.180. [DOI] [PubMed] [Google Scholar]
- 56.Shao L, Fujii H, Colmegna I, Oishi H, Goronzy JJ, Weyand CM. Deficiency of the DNA repair enzyme ATM in rheumatoid arthritis. J Exp Med. 2009;206:1435–1449. doi: 10.1084/jem.20082251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Fujii H, Shao L, Colmegna I, Goronzy JJ, Weyand CM. Telomerase insufficiency in rheumatoid arthritis. Proc Natl Acad Sci U S A. 2009;106:4360–4365. doi: 10.1073/pnas.0811332106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Naylor K, Li G, Vallejo AN, Lee WW, Koetz K, Bryl E, Witkowski J, Fulbright J, Weyand CM, Goronzy JJ. The influence of age on T cell generation and TCR diversity. J Immunol. 2005;174:7446–7452. doi: 10.4049/jimmunol.174.11.7446. [DOI] [PubMed] [Google Scholar]
- 59.Goronzy JJ, Fulbright JW, Crowson CS, Poland GA, O'Fallon WM, Weyand CM. Value of immunological markers in predicting responsiveness to influenza vaccination in elderly individuals. J Virol. 2001;75:12182–12187. doi: 10.1128/JVI.75.24.12182-12187.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Cheng G, Yu A, Malek TR. T-cell tolerance and the multi-functional role of IL-2R signaling in T-regulatory cells. Immunol Rev. 2011;241:63–76. doi: 10.1111/j.1600-065X.2011.01004.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Vang KB, Yang J, Mahmud SA, Burchill MA, Vegoe AL, Farrar MA. IL-2,-7, and-15, but not thymic stromal lymphopoeitin, redundantly govern CD4+Foxp3+ regulatory T cell development. J Immunol. 2008;181:3285–3290. doi: 10.4049/jimmunol.181.5.3285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kim HJ, Wang X, Radfar S, Sproule TJ, Roopenian DC, Cantor H. CD8+ T regulatory cells express the Ly49 Class I MHC receptor and are defective in autoimmune prone B6-Yaa mice. Proc Natl Acad Sci U S A. 2011;108:2010–2015. doi: 10.1073/pnas.1018974108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Andersen MH, Sorensen RB, Brimnes MK, Svane IM, Becker JC, thor Straten P. Identification of heme oxygenase-1-specific regulatory CD8+ T cells in cancer patients. J Clin Invest. 2009;119:2245–2256. doi: 10.1172/JCI38739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Shi Z, Okuno Y, Rifa'i M, Endharti AT, Akane K, Isobe K, Suzuki H. Human CD8+CXCR3+ T cells have the same function as murine CD8+CD122+ Treg. Eur J Immunol. 2009;39:2106–2119. doi: 10.1002/eji.200939314. [DOI] [PubMed] [Google Scholar]
- 65.Bhowmick S, Clark RB, Brocke S, Cone RE. Antigen-specific splenic CD4+ and CD8+ regulatory T cells generated via the eye, suppress experimental autoimmune encephalomyelitis either at the priming or at the effector phase. Int Immunol. 2011;23:119–128. doi: 10.1093/intimm/dxq461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sakaguchi S. Regulatory T cells: history and perspective. Methods Mol Biol. 2011;707:3–17. doi: 10.1007/978-1-61737-979-6_1. [DOI] [PubMed] [Google Scholar]
- 67.Takemura S, Braun A, Crowson C, Kurtin PJ, Cofield RH, O'Fallon WM, Goronzy JJ, Weyand CM. Lymphoid neogenesis in rheumatoid synovitis. J Immunol. 2001;167:1072–1080. doi: 10.4049/jimmunol.167.2.1072. [DOI] [PubMed] [Google Scholar]
- 68.Seyler TM, Park YW, Takemura S, Bram RJ, Kurtin PJ, Goronzy JJ, Weyand CM. BLyS and APRIL in rheumatoid arthritis. J Clin Invest. 2005;115:3083–3092. doi: 10.1172/JCI25265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Klimiuk PA, Goronzy JJ, Weyand CM. IL-16 as an anti-inflammatory cytokine in rheumatoid synovitis. J Immunol. 1999;162:4293–4299. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.