Abstract
Langerhans cells (LCs) were first described by Paul Langerhans, in 1868, as dendritically shaped cells, which were located in the squamous epithelia of epidermis. Later on, these cells were identified in all stratified squamous epithelium of mammals. Dendritic cells (DCs) play an important role in local defense mechanisms in the epithelium. LCs are situated usually in the suprabasal layer of stratified squamous epithelia of oral mucosa and epidermis of skin. They constitute 3% of the cell population in epidermis. LCs are thought to act as antigen presenting cells (APCs) during initiation of immune responses. With the help of APCs, the lymphocytes are able to recognize and respond to specific microbes. In this paper we have reviewed the origin, distribution, demonstration and mechanism of action of LCs and their role in different pathological conditions.
Keywords: Antigen presenting cells, clear cells, dendritic cells, Langerhans cells
ORIGIN OF LANGERHANS CELLS
The Langerhans cells (LCs) originate from the bone marrow and then migrate into the epithelium to perform the function of antigen recognition and presentation. Studies have shown that the dendritic cells (DCs) are developmentally similar. They share a common origin and then differentiate into different types in response to different environmental stimuli (Zuniga et al. 2004) [Figure 1]. Earlier studies of DCs in the rat (Klinkert, 1984; Bowers and Berkovitz, 1986), the mouse (Steinman and Cohn, 1973), and humans (Caux et al., 1992)[1] suggested the presence of cell types of progenitor cells, which were developmentally distinct, the myeloid and the lymphoid series. These cells can then differentiate into distinct DC subtypes and perform distinct functions.[1] Lymphoid progenitors give rise to precursors that differentiate into thymic DCs, interdigitating DCs and plasmacytoid DCs. Myeloid progenitors give rise to precursors of monocyte/macrophage series, LCs, interstitial DCs, germinal center DCs, and peripheral blood DCs [Figure 2]. This indicates that common precursors can give rise to different DC subtypes, under specific environmental conditions.
Figure 1.
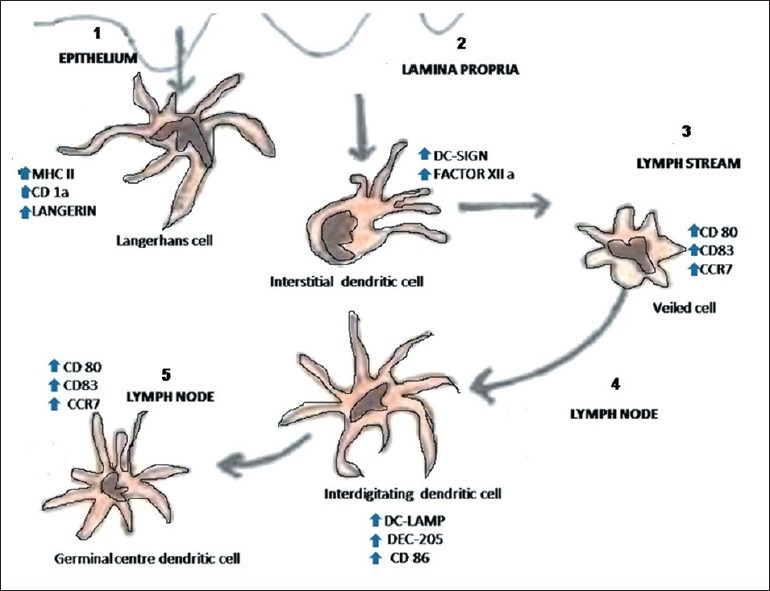
Types of dendritic cells. (Abbreviations; MHC - Major histocompatibility antigen, DC-SIGN - Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin, DC-LAMP - Dendritic cell-lysosomal associated membrane protein, DEC - Dendritic cell receptor for endocytosis.)
Figure 2.
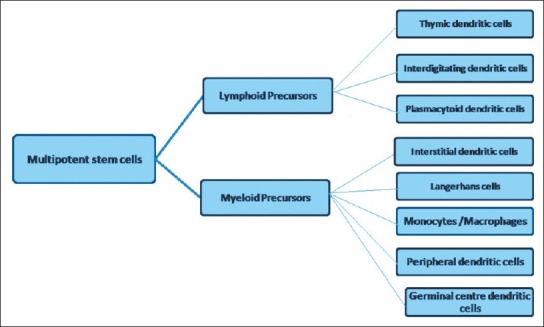
Myeloid and Lymphoid series further differentiating into dendritic cell subsets. (Abbreviation; DCs - dendritic cells)
Frlinger, Katz et al., in 1979, suggested that LCs are bone marrow derived cells that begin to appear in the epidermis by about seven weeks of gestation.[1] They could be recognized by their positive staining for ATPase as demonstrated by Wolff et al. At this stage, they are fewer, smaller and less dendritic than at a later fetal stage. LCs begin to express CD1a reactivity at gestational stage of sixty days.[1]
DISTRIBUTION OF LANGERHANS CELLS
A study has shown that the epithelium of the oral mucosa contains about 37 times as many T-lymphocytes as the epidermis of normal skin (van Loon et al., 1989).[2] The authors also concluded that CD4+ and CD8+ T-cell subsets are equally present in the epithelium of the oral mucosa, and the TH-lymphocytes (T helper cells) play a more important role.[2]
LCs are seen in histological sections stained with hematoxylin and eosin as ‘clear cells’ in suprabasal layers of mucous membranes.[3] The LCs can be visualized as cells with their bodies generally in the middle portion of stratum spinosum. Delicate dendritic processes can often be observed extending to the level of stratum corneum. Their cytoplasmic processes are found in stratum corneum and usually directed towards epithelial surface [Figure 3]. Many studies have shown variation in LCs density in relation to age and location in different body regions.[4] According to morphology, LCs have been classified into two types i.e. Type1 LCs, which are pyramidal in shape, located in suprabasal layer, and Type2 LCs, which are spherical in shape, located in the basal layer. Type 1 contains numerous Birbeck granules, electrolucent cytoplasm and long-branched dendritic processes whereas Type 2 cells, with fewer Birbeck granules, electron dense cytoplasm and shorter dendritic processes [Figure 4].[5]
Figure 3.
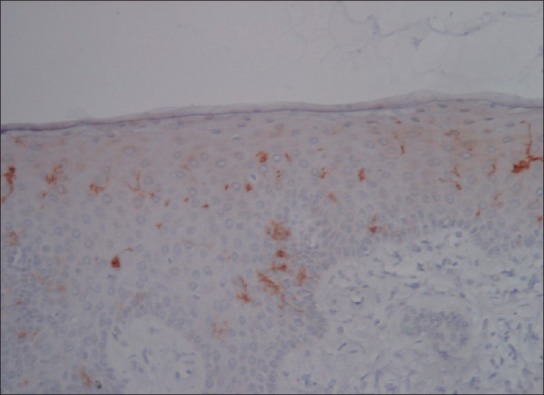
Photomicrograph of gingival epithelium showing CD1a positive Langerhans cells within the stratum spinosum
Figure 4.
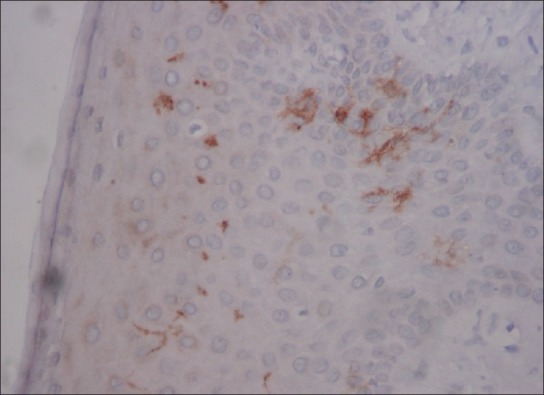
Photomicrograph of gingival epithelium showing CD1a positive Langerhans cells with long dendritic processes and some with spherical morphology
STAINING OF LANGERHANS CELLS
Hematoxylin and eosin staining
LCs in sections stained with routine hematoxylin and eosin stains cannot be well appreciated. They appear as ‘clear cells’ having a clear halo around their nuclei. LCs being present in the suprabasal layers are also referred to as ‘high level clear cells’, melanocytes being the ‘low level clear cells’ as present in basal cell layer of stratified squamous epithelium. All these cells, except Merkel cells, lack desmosomal junctions to adjacent cells. During histologic processing the cytoplasm shrinks around the nucleus to produce the clear halo.[3]
Special staining
One of the first techniques employed to demonstrate LCs was the metal impregnation staining technique using heavy metals such as gold chloride, osmium iodide or zinc osmium iodide and uranyl acetate–lead staining. Zinc osmium iodide, although gives good staining, also stains keratinocytes and melanocytes.[5]
Enzyme histochemistry
The actual sites of cellular enzymatic activity can be demonstrated by enzyme histochemical techniques like ATPase, which is an excellent method for the identification of human LCs.[5] Other enzymes used are adenosinediphosphatase (ADPase), 5 nucleotidases and amino-peptidases.
Immunohistochemistry
A large number surface and cytoplasmic antigens have been localized on LCs by means of antibodies used in different techniques like, immunofluorescence or immunoperoxidase techniques. One of the most prominent features of LCs is the high level of expression of MHC class II antigens. In addition to this, MHC class I antigens are also expressed by LCs. Leukocyte common antigen (CD45), a marker of cells of hematopoietic origin, is also expressed by LCs and shows stresses their bone marrow origin.[5] Many other antigens like Fc-IgG, CD11, C3b/C4b, CD25, CD29, CD54, CD1D and CD83 are found only in subpopulations of LCs or are present in such trace amounts that their detection is only possible by using extremely sensitive methods. Among all the markers used, CD1a immunolabeling is considered to be the most reliable method to identify the human LCs. Studies have shown that immature DCs express immunohistochemical markers CD1a, b, c and d.[5] Recent studies have shown that Langerin seems to be the more specific marker for LCs.
It has been proposed that Langerin induces the formation of Birbeck granules. These LC-specific organelles are distributed within the cytoplasm and are thought to be involved in antigen processing.[4]
Electron microscopy
Electron microscopy studies have shown that LCs exhibit a cleaved or folded nucleus and no tonofilaments or desmosomes. The Birbeck granules are characteristically present in their cytoplasm, which have a three-dimensional profile of a disc. Their size varies from 100 nm to 1 μm. These granules seem to be most specific marker for LCs. Birbeck granules show a vesicle at one end and sometimes at both ends. In cross sections, the granule with vesicle at one end shows a characteristic appearance, which is termed as ‘tennis racket’ appearance [Figure 5].
Figure 5.
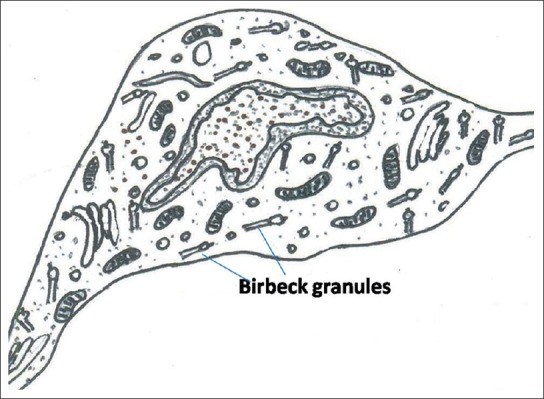
Langerhans cell with cleaved nucleus and numerous Birbeck granules within the cytoplasm
Presence of transverse striations has also been observed within the Birbeck granule. It is believed that they are formed due to invagination of the cell membrane.[6] Grune and Stratton, 1983, have used six electron microscopic criteria of specificity for LCs: indented or lobulated nucleus, Birbeck granules, absence of tonofilaments, well-developed Golgi apparatus with a clear cytoplasm, absence of desmosomes and absence of melanosomes and pre-melanosomes.[5]
MECHANISM OF FUNCTION OF LANGERHANS CELLS
The DCs and other APCs must quickly respond to the invading bacterial structures (e.g. lipopolysaccharides, proteoglycans) called pathogen-associated molecular patterns. This function is accomplished through toll-like receptors (TLRs), which are a major class of signaling receptors. Receptors involved in antigen recognition are toll-like receptors (TLRs) and C-type lectin receptors. TLRs are pattern recognition receptors associated along with pathogen-associated molecular patterns (PAMPs). Depending on their localization within the tissue and the state of differentiation, DCs, LCs and certain tissue macrophages express C-type lectin receptors.[7] C-type lectin receptors include membrane bound C-type lectins - CD207 i.e. Langerin, CD206 i.e. Mannose receptors, CD205 and CD209. CD1a, which is a transmembrane antigen, is normally found on cortical thymocytes as well as LCs.
It has been shown in a study that the TLR ligands induce the maturation of bone marrow dendritic cells. It was also observed that these mature DCs were capable of antigen recognition and antigen uptake.[8]
Toll-like receptor recognition is specific for several important pathogen-associated molecular patterns like: peptidoglycans, bacterial lipoproteins, and zymosan by Toll-like receptor 2, double-stranded RNA by Toll-like receptor 3, most lipopolysaccharide and heat-shock proteins by Toll-like receptor 4, flagellin by Toll-like receptor 5 (Akira and Hemmi, 2003).[9] The main function of C-type lectin receptors is to interact with conserved carbohydrates of microorganisms (PAMPs), and internalize them for processing and antigen presentation. Accordingly, C-type lectin receptors contain carbohydrate recognition domains for binding to sugars. Toll-like receptors rapidly transmit information about the pathogen through interleukin domains and promote the release of NFk-β. This is followed by transcription of co-stimulatory molecules and proinflammatory cytokines (e.g., IL-1, IL-12, IFN-γ) [Figure 6].
Figure 6.
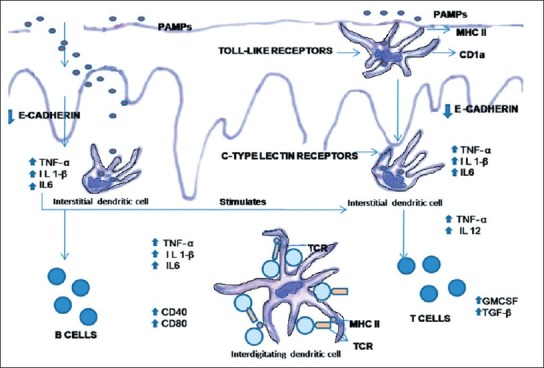
Antigen recognition by Langerhans cells present in the epithelium and initiation of T-cell response. (Abbreviations; PAMPs – Pathogen-associated molecular patterns, MHC – Major histocompatibility antigen, IL – Interleukin, TNF – Tumor necrosis factor, TGF – Transforming growth factor, GM-CSF – Granulocyte monocyte colony stimulating factor, TCR – T cell receptor)
The LCs migrate via the afferent lymphatics as veiled cells, into the par cortex of the draining lymph nodes. Within the para-cortex, the cells interdigitate with many T cells.[9] This migration carries the antigen from the skin or mucosa to the TH cells of the lymph nodes. The LCs express Class II MHC molecules for communicating with CD4+ T cells. Antigens are then degraded by proteolytic enzymes in specialized intercellular organelles. The linear fragments of antigenic peptides associate with class II MHC molecules as they are moved to the cell surface.[7]
Cytoplasmic antigens are then processed into peptides by cytoplasmic proteases. The antigen is then transported into the rough endoplasmic reticulum (RER) of the cell. Within the RER, class I and class II antigens are synthesized and assembled. Once within the rough endoplasmic reticulum, the peptides are thought to associate efficiently with newly synthesized class I molecules. Cytoplasmic antigens are thought to be processed by an organelle known as proteasome, which are multicatalytic proteinase complexes. Following synthesis in the RER, both class I and class II antigens are transported to the Golgi compartment. Within the Golgi compartment, the class I and II antigens segregate; class I molecules are transported directly to the cell surface and class II molecules arrive in the lysosomal-like compartment.[9] The migratory activity of LCs is regulated by the expression of chemokine receptors, selectins, chemo-attractants or inflammatory cytokines and integrins. NFk-β, in turn regulates the activity of these receptors.
CO-STIMULATION
After the antigen recognition, signal transduction is mediated via the interleukins and MyD88. Thus resulting in the release of NFk-β, which is an important factor in DC maturation. Therefore interaction of the PAMPs with the C-type lectin receptors and toll-like receptors and resultant stimulation of LCs brings about antigen presentation and elicitation of cell-mediated immune response. It is observed that the variations in co-signal ligand expression and cytokine production greatly influence the antigen-presenting properties of migrating DCs in regional lymph nodes. There was delayed migration and limited induction of co-signal ligands in other DC subsets. These authors stated that oral mucosal LCs play a different role than other DC subsets. Thus the functional specialization of each subset in T cell responses against orally administrated antigens can be interpreted.[10]
LANGERHANS CELLS IN DIFFERENT PATHOLOGICAL CONDITIONS
Gingivitis and periodontitis
An increased number of LCs in gingival epithelium associated with the development of gingivitis in germ-free mice has been demonstrated.[11] The LCs increased in number with plaque accumulation during gingivitis. In this study, LCs were found five times more in inflamed gingiva in comparison to healthy gingiva from the same patients.[12] It has been suggested that, in gingivitis the LCs initiate the development of oral lymphoid follicles. These foci develop in response to pathogen-associated molecular patterns.[13] In chronic adult periodontitis, mature CD83+ DCs form immune conjugates with CD4+ T cells in the oral lymphoid foci.[1,14]
Human immunodeficiency virus infection
It has been shown that Acquired ImmunoDeficiency Syndrome (AIDS) and AIDS-related complex patients have decreased number of LCs . The LCs expressed low amounts of CD4 antigens,[15,16] which were immunologically detectable. It was assumed that the reduction in the number of LCs in HIV-1 infection was a consequence of injury to the LCs due to HIV infection. Another electron microscopic investigation carried out on the skin and mucosal biopsies of infected patients demonstrated HIV-1 virions in the extracellular space around LCs. In this study, viral particles budding from LC surface membranes were also observed.[17] HIV-1 virions are localized along with Langerin at the cell surface and in intracellular vesicles. These virions were found to be in close vicinity to invaginations which were Langerin-positive. Thus, Langerin captures HIV-1 and internalizes it into Birbeck granules of LCs.[4] It has been suggested that Langerhans could act as a reservoir for the virus as they are the target cells during HIV infection.[18] It was also stated that the LCs and other DCs are among the first cells to encounter antigens and migrate to lymph nodes to elicit T-cell responses.[19]
Oral lichen planus
Oral lichen planus (OLP) is considered to be a mucocutaneous disease caused due to a cell-mediated immune reaction. Light and electron microscopic studies suggested that an intraepithelial accumulation of LCs takes place during the early stages. In OLP, changes in the local immune regulation may result in changes in the LCs. These changes are responsible for the pathogenesis of OLP.[5] Oral lichen planus is characterized by the presence of LCs, macrophages and lymphocytes, as shown immunohistochemically, by the presence of specific antigens.[20,21] In a study it was suggested that the antigen presenting capability of LCs was increased in lichen planus.[22] Other studies have suggested that LCs and T lymphocytes are in direct contact with each other in OLP.[23]
Lichenoid drug eruptions
In lichenoid drug eruptions associated with systemic drug administration, the route of entry of the antigen is at a distant site. Thus the antigen processing and presentation results in reduction of activation of LCs. The same is applicable in cases where lichenoid reactions of the oral mucous membrane are found in sites in contact with dental metallic restorations.[24] In another study, it was shown that the number of HLA-DR+ LCs in lichenoid reactions was significantly lower than the number in OLP.[25]
Recurrent aphthous stomatitis
The significance of LCs has also been discussed related to both recurrent aphthous stomatitis and Behcet's syndrome. In these conditions, mononuclear cells are involved which interact with the epithelia. In an experimental study, there was an increase in the number of LCs in the areas of skin of Behcet's disease patients subjected to pricking with a hypodermic needle. This indicated an exaggerated immune response.[26] Langerhans were observed to possess intense enzymatic activity along with some degenerating cells. A possible reason for this kind of reaction could be disturbance in the LCs population.[5]
Contact hypersensitivity
LCs are considered necessary to induce contact hypersensitivity as the phenomenon is taking place in the epidermis. Hapten-modified LCs can induce sensitization when particular immunizing conditions are applied. The phenomenon of contact hypersensitivity has two phases: sensitization and elicitation of response. Sensitization of LCs takes place about 10 - 14 days and their number increased in the epidermis at 24 - 48 hrs. It was also found that LCs increased significantly in the connective tissue at the site of nickel application.[27]
Role of LCs in oral cancer
In oral cancer the increase in the number of LCs, macrophages and CD8+ lymphocytes in the epithelium have been demonstrated.[28,29] In a study, the functional role of LCs in squamous cell carcinomas arising from the tongue was studied. It showed a close relationship of LCs with lymphocytes in terms of their number and distribution along the epithelium.[30] An increase in the number of CD8 positive lymphocytes and LCs had been observed in the stroma, adjacent to tumor cells as well as in the connective tissue which was far away from the tumor. A suggested possibility is that in cancer, the LCs present tumor antigens to the lymphocytes, thus playing a similar role as in case of allergic reactions.
Langerhans cell histiocytosis
Langerhans cell histiocytosis (LCH) mainly affects young children and is characterized by accumulation of CD1a+ LCs within the epidermis, dermis, bones and occasionally lymphoid organs, lungs and digestive tract. The altered expression of intercellular adhesion molecules may play an important role in the pathogenesis of the disease as it is important for the migration and distribution of LCs.[31–33] It has also been observed that during the chronic form of the disease, the LCs are functionally immature. Studies have also shown that some extrinsic signals play a role in maintaining them in an immature state.[34]
It is well documented that LCs play an important role in the immune responses, particularly, in antigen presentation to the T lymphocytes. Thus, they should be phenotypically and functionally studied in correlation with the other cells of the immune system.
Footnotes
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- 1.Cutler CW, Jotwani R. Dendritic cells at the oral mucosal interface. J Dent Res. 2006;85(8):678–89. doi: 10.1177/154405910608500801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lu FX, Jacobson RS. Oral mucosal immunity and HIV/SIV Infection. J Dent Res Mar. 2007;86:216–26. doi: 10.1177/154405910708600305. [DOI] [PubMed] [Google Scholar]
- 3.5th edition. Missouri: Mosby Year Book Inc; 1996. Ten Kate AR: Oral Histology. Development, structure and Function. [Google Scholar]
- 4.de Witte L, Nabatov A, Pion M, Fluitsma D, de Jong MA, de Gruijl T, et al. Langerin is a natural barrier to HIV-1 transmission by Langerhans cells. Nature Med. 2007;13(3):367–71. doi: 10.1038/nm1541. [DOI] [PubMed] [Google Scholar]
- 5.Lombardi T, Hauser C, Budtz-Jörgensen E. Langerhans cells: structure, function and role in oral pathological conditions. J Oral Pathol Med. 1993;22:193–202. doi: 10.1111/j.1600-0714.1993.tb01056.x. [DOI] [PubMed] [Google Scholar]
- 6.Bodineau A, Coulomb B, Folliguet M, Igondjo-Tchen S, Godeau G, Brousse N, et al. Do Langerhans cells behave similarly in elderly and younger patients with chronic periodontitis. Arch Oral Biol. 2007;52:189–94. doi: 10.1016/j.archoralbio.2006.09.007. [DOI] [PubMed] [Google Scholar]
- 7.Elder D, Elviysor R, Jawosky C, Johnson B. 8th ed. Philadelphia, PA: Lippincott Williams and Wilkins; 1997. Lever's Histopathology of the skin. [Google Scholar]
- 8.Datta SK, Redecke V, Prilliman KR, Takabayashi K, Corr M, Tallant T, et al. A subset of toll-like receptor ligands induces cross-presentation by bone marrow-derived dendritic cells. J Immunol. 2003;170:4102–10. doi: 10.4049/jimmunol.170.8.4102. [DOI] [PubMed] [Google Scholar]
- 9.Cutler CW, Jotwani R. Antigen presentation and the role of dendritic cells in periodontitis. Periodontol 2000. 2004;35:135–57. doi: 10.1111/j.0906-6713.2004.003560.x. [DOI] [PubMed] [Google Scholar]
- 10.Aramaki O, Chalermsarp N, Otsuki M, Tagami J, Azuma M. Differential expression of co-signal molecules and migratory properties in four distinct subsets of migratory dendritic cells from the oral mucosa. Biochem Biophys Res Commun. 2011;413:407–13. doi: 10.1016/j.bbrc.2011.08.099. [DOI] [PubMed] [Google Scholar]
- 11.Van Kooyk Y, Engering A, Lekkerkerker AN, Ludwig IS, Geijtenbeek TB. Pathogens use carbohydrates to escape immunity induced by dendritic cells. Curr Opin Immunol. 2004;16:488–93. doi: 10.1016/j.coi.2004.05.010. [DOI] [PubMed] [Google Scholar]
- 12.Bos IR, Burkhardt A. Interepithelial cells of the oral mucosa.Light and electron microscopic observations in germfree, specific pathogen-free and conventionalized mice. J Oral Pathol. 1980;9:65–81. doi: 10.1111/j.1600-0714.1980.tb01389.x. [DOI] [PubMed] [Google Scholar]
- 13.DiFranco CF, Toto PD, Rowden G, Gargiulo AW, Keene JJ, Jr, Connelly E. Identification of Langerhans cells in human gingival epithelium. J Periodontol. 1985;56:48–54. doi: 10.1902/jop.1985.56.1.48. [DOI] [PubMed] [Google Scholar]
- 14.Jotwani R, Palucka AK, Al-Quotub M, Nouri-Shirazi M, Kim J, Bell D, et al. Mature dendritic cells infiltrate the T cell-rich region of oral mucosa in chronic periodontitis: in situ, in vivo, and in vitro studies. J Immunol. 2001;167:4693–700. doi: 10.4049/jimmunol.167.8.4693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wood GS, Warner NL, Warnke RA. Anti-Leu-3/T4 antibodies react with cells of monocyte/macrophage and Langerhans lineage. J Immunol. 1983;131:212–6. [PubMed] [Google Scholar]
- 16.Groh V, Tani M, Harrer A, Wolff K, Stingl G. Leu-3/T4 expression on epidermal Langerhans cells in normal and diseased skin. J Invest Dermatol. 1986;86:115–20. doi: 10.1111/1523-1747.ep12284090. [DOI] [PubMed] [Google Scholar]
- 17.Rappersberger K, Gartner S, Schenk P, Stingl G, Groh V, Tschachler E, et al. Langerhans’ cells are an actual site of HIV- 1 replication. Intervirology. 1988;29:185–94. doi: 10.1159/000150045. [DOI] [PubMed] [Google Scholar]
- 18.Neideckhen H, Lutz G, Bauer R, Kreysel HW. Langerhans cells as primary target and vehicle for transmission of HIV. Lancet. 1987;2:519–20. doi: 10.1016/s0140-6736(87)91843-5. [DOI] [PubMed] [Google Scholar]
- 19.Cameron PU, Handley AJ, Baylis DC, Solomon AE, Bernard N, Purcell DF, et al. Preferential infection of dendritic cells during HIV type 1 infection of blood leukocytes. J Virol. 2007;81:2297–306. doi: 10.1128/JVI.01795-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Regezi JA, Stewart JC, Lloyd RV, Headington JT. Immunohistochemical staining of Langerhans cells and macrophages in oral lichen planus. Oral Surg Oral Med Oral Pathol. 1985;60:396–402. doi: 10.1016/0030-4220(85)90262-2. [DOI] [PubMed] [Google Scholar]
- 21.Ishh T. Immunohistochemical demonstration of T cell subsets and accessory cells in oral lichen planus. J Oral Pathol. 1987;16:356–61. doi: 10.1111/j.1600-0714.1987.tb00708.x. [DOI] [PubMed] [Google Scholar]
- 22.Farthing PM, Matear P, Cruchley AT. The activation of Langerhans cells in oral lichen planus. J Oral Pathol Med. 1990;19:81–5. doi: 10.1111/j.1600-0714.1990.tb00801.x. [DOI] [PubMed] [Google Scholar]
- 23.el-Labban NG. An ultrastructural study of Langerhans cells crossing the basal complex in oral lichen planus. Arch Oral Biol. 1977;22:629–31. doi: 10.1016/0003-9969(77)90077-2. [DOI] [PubMed] [Google Scholar]
- 24.Bolewska J, Reibl J. T lymphocytes, Langerhans cells and HLA-DR expression on keratinocytes in oral lesions associated with amalgam restorations. J Oral Pathol Med. 1989;18:525–8. doi: 10.1111/j.1600-0714.1989.tb01356.x. [DOI] [PubMed] [Google Scholar]
- 25.McCartan BE, Lamey PJ. Expression of CD1 and HLA-DR by Langerhans cells (LC) in oral lichenoid drug eruptions (LDE) and idiopathic oral lichen planus (LP) J Oral Pathol Med. 1997;26:176–80. doi: 10.1111/j.1600-0714.1997.tb00454.x. [DOI] [PubMed] [Google Scholar]
- 26.Salto T, Honma T. Epidermal Langerhans cells, macrophages and lymphocytes after the prick test in patients with behcet's syndrome and recurrent oral ulcerations. Biomed Res. 1981;2:2658–9. [Google Scholar]
- 27.van Loon LA, van Elsas PW, Bos JD, ten Harkel-Hagenaar HC, Krieg SR, Davidson CL. T-lymphocyte and Langerhans cell distribution in normal and allergically induced oral mucosa in contact with nickel-containing dental alloys. J Oral Pathol. 1998;17:129–37. doi: 10.1111/j.1600-0714.1988.tb01899.x. [DOI] [PubMed] [Google Scholar]
- 28.Gatter KC, Morris HB, Roach B, Mortimer P, Fleming KA, Mason DY. Langerhans cells and T cells in human skin tumors: an immunohistochemical study. Histopathology. 1984;8:229–44. doi: 10.1111/j.1365-2559.1984.tb02338.x. [DOI] [PubMed] [Google Scholar]
- 29.Löning T, Broemel H, Becker WM, Otto HF. Identification of inflammatory cell phenotypes in human oral carcinomas by means of monoclonal antibodies. Arch Oral Biol. 1983;28:1093–100. doi: 10.1016/0003-9969(83)90164-4. [DOI] [PubMed] [Google Scholar]
- 30.Kurihara K, Hashimoto N. The pathological significance of Langerhans cells in oral cancer. J Oral Pathol. 1985;14:289–98. doi: 10.1111/j.1600-0714.1985.tb00496.x. [DOI] [PubMed] [Google Scholar]
- 31.de Graaf JH, Tamminga RY, Dam-Meiring A, Kamps WA, Timens W. The presence of cytokines in Langerhans’ cell histiocytosis. J Pathol. 1996;180:400–6. doi: 10.1002/(SICI)1096-9896(199612)180:4<400::AID-PATH701>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 32.Tazi A, Moreau J, Bergeron A, Dominique S, Hance AJ, Soler P. Evidence that Langerhans cells in adult pulmonary Langerhans cell histiocytosis are mature dendritic cells: importance of the cytokine microenvironment. J Immunol. 1999;163:3511–5. [PubMed] [Google Scholar]
- 33.Egeler RM, Favara BE, van Meurs M, Laman JD, Claassen E. Differential In situ cytokine profiles of Langerhans-like cells and T cells in Langerhans cell histiocytosis: abundant expression of cytokines relevant to disease and treatment. Blood. 1999;94:4195–201. [PubMed] [Google Scholar]
- 34.Geissmann F, Lepelletier Y, Fraitag S, Valladeau J, Bodemer C, Debré M, et al. Differentiation of Langerhans cells in Langerhans cell histiocytosis. Blood. 2001;97:1241–8. doi: 10.1182/blood.v97.5.1241. [DOI] [PubMed] [Google Scholar]


