Abstract
Glioblastoma (GBM) is a highly aggressive primary brain tumor with a poor prognosis. Despite aggressive therapy with surgery, radiotherapy, and chemotherapy, nearly all patients succumb to disease within 2 years. Several studies have supported the presence of stem-like cells in brain tumor cultures that are CD133-positive, are capable of self-renewal, and give rise to all cell types found within the tumor, potentially perpetuating growth. CD133 is a widely accepted marker for glioma-derived cancer stem cells; however, its reliability has been questioned, creating a need for other identifiers of this biologically important subpopulation. We used a panel of 20 lectins to identify differences in glycan expression found in the glycocalyx of undifferentiated glioma-derived stem cells and differentiated cells that arise from them. Fluorescently labeled lectins that specifically recognize α-N-acetylgalactosamine (GalNAc) and α-N-acetylglucosamine (GlcNAc) differentially bound to the cell surface based on the state of cellular differentiation. GalNAc and GlcNAc were highly expressed on the surface of undifferentiated cells and showed markedly reduced expression over a 12-day duration of differentiation. Additionally, the GalNAc-recognizing lectin Dolichos biflorus agglutinin was capable of specifically selecting and sorting glioma-derived stem cell populations from an unsorted tumor stock and this subpopulation had proliferative properties similar to CD133+ cells in vitro and also had tumor-forming capability in vivo. Our preliminary results on a single cerebellar GBM suggest that GalNAc and GlcNAc are novel biomarkers for identifying glioma-derived stem cells and can be used to isolate cancer stem cells from unsorted cell populations, thereby creating new cell lines for research or clinical testing.
Introduction
Glioblastoma (GBM) is the most common and most aggressive primary brain tumor of adults, accounting for 52% of all cases [1–3]. In the United States, there are 2–3 cases of GBM diagnosed per 100,000 each year. Standard of care for these tumors includes surgery, chemotherapy, and radiotherapy. Despite this, patients typically live <2 years after diagnosis [1–3]. Several studies have supported the presence of stem-like cells in brain tumor cultures [4–6], which are highly tumorigenenic and have the ability to self-renew and to give rise to all of cell types with distinct lineages found within the tumor. GBM stem cells or cancer stem cells (CSCs) are most often identified through expression of CD133, a marker that is also present in nonmalignant neural progenitor cells [5,6]. The mRNA expression of CD133 stem cell antigen correlates with the survival of GBM patients, lending support to the current brain tumor stem cell hypothesis [7]. However, using CD133 exclusively as a marker for GBM tumor-derived CSCs (GBM-CSCs) is problematic because it is not consistently expressed in all GBMs and CD133-negative cells have been shown to give rise to tumors in transplant assays [8,9]. Recent reports question the use of CD133 for fluorescence activated cell sorting (FACS) because its expression is dependent on environmental, genetic, and chemical factors [4,9,10], making it possible to miss a population of CD133-positive cells during sorting. When present, CD133 expression can be a useful marker for enriching for GBM stem cells, yet its low expression by some tumors suggests that additional markers need to be explored. Conventionally, it is believed that therapeutic treatments are selectively toxic to differentiated or differentiating cells, which form the bulk of the tumor [5,6,9], whereas CSCs persist as a distinct subpopulation that are resistant to treatment and lead to recurrence [5,6,9]. Therefore, the identification of new biomarkers and the development of specific therapies targeted toward CSCs hold promise for patient survival and improved quality of life.
Lectins are a family of carbohydrate-binding proteins that recognize and distinguish specific sugar structures and have been extensively used to identify, characterize, and isolate novel cell subpopulations on the basis of their defining carbohydrate groups on the cell surface. For example, the lectin Dolichos biflorus agglutinin (DBA), which recognizes α-N-acetylgalactosamine (GalNAc), was used to characterize mouse embryonic stem cell (mESC) cultures by nondestructive measures [11]. Presentation of GalNAc on the mESC surface is rapidly diminished during differentiation, preceding the downregulation of other better-known protein markers of pluripotency [11]. Similarly, lectins Lycopersicon (tomato) esculetum lectin (LEL), Ricinus communis agglutinin-I (RCA-I), and Concanavalin A (ConA) have been used to identify pluripotent human ESCs [12]. Lectins have also been used to investigate metastatic processes in many cancer types [13–16], as well as to document the repertoire of glycoepitopes on the surface of embryonic carcinoma cells [17,18]. These results indicate that glycans can be used as markers to define specific stages of stemness in multiple cell types.
In this study we attempted to identify glycans that are unique to GBM-CSC undifferentiated state through nondestructive techniques (flow cytometry). We used neurosphere cultures derived from a cerebellar GBM and a panel of 20 lectins to determine the cell surface glycan expression patterns of CD133+ GBM-CSCs. Five lectins that recognize GalNAc and 2 lectins that recognize α-N-acetylglucosamine (GlcNAc) were highly reactive toward undifferentiated CD133+ GBM tumor-derived cells. Presentation of GalNAc and GlcNAc on the cell surface is rapidly diminished during differentiation, in conjunction with the GBM-CSC marker CD133, and pluripotent stem cell markers OCT4 and SOX2. Additionally, we used DBA lectin to target GalNAc and successfully sorted CD133+ GBM-CSCs (CTB-1). These findings establish the use of lectins and cell surface glycans (GalNAc and GlcNAc) to characterize CSCs derived from GBMs, independently of the widely accepted marker CD133.
Materials and Methods
Tissue culture
Cell preparations were derived from a GBM that involved the brainstem and cerebellum, which was partially resected from a consenting patient at Emory University Hospitals. The specimen was examined by a neuropathologist to verify that the tumor was diagnostic of GBM prior to establishing a culture. Specimens were cut into <1 mm3 fragments, washed with Hank's balanced salt solution (Mediatech), and digested with 1 mg/mL collagenase/dispase (Roche). Digested fragments were then filtered using a 70-μm cell mesh and washed with Hank's balanced salt solution or phosphate buffered saline (PBS) 2×. Supernatant was overlayed on histopaque ficoll (to remove red blood cells) and centrifuged at 2,000 rpm for 35–40 min. After removing the top layer, the lower layer was collected and centrifuged and resuspended in neurobasal media. Neurosphere cultures were maintained in neurobasal media supplemented with the following growth factors: hEGF (20 ng/mL), hFGF-b (10 ng/mL), and heparin (2 μg/mL) (StemCell Technologies). Neurospheres were mechanically dissociated and harvested or passaged for continual growth. Neurospheres that were differentiated were placed on 12-well plates coated with polyornithine (Sigma; 20 μg) and laminin (Sigma; 10 μg). After 24 h, media were replaced with growth-factor-free media and allowed to differentiate up to 12 days. Samples were harvested at days 1 and 12.
Antibodies and lectins
The following primary antibodies and lectins were used in this study: mouse monoclonal against human OCT4, Sox2, Actin, and CD133 (Santa Cruz); rabbit monoclonal against human glial fibrillary acidic protein (GFAP), Ki67, and cleaved Caspase 3 (Cell Signaling); mouse monoclonal against human CD133-glycosylation (CD133-G; Miltenyi Biotec); and fluorescently labeled secondary antibody (Molecular Probes). The following lectins were used in this study: (full-length names for each lectin can be found in Supplementary Table S1; Supplementary Data are available online at www.liebertonline.com/scd) Concanavalin A (CON A), DBA, Peanut agglutinin (PNA), RCA 120, Soybean agglutinin (SBA), Ulex europaeus agglutinin I (UEA I), Wheat germ agglutinin (WGA), Griffonia simplificola lectin I (GSL I), Lens culinaris agglutinin (LCA), Phaseolus vulgaris Erythroagglutinin (PHA-E), Phaseolus vulgaris Leucoagglutinin (PHA-L), Pisum sativum agglutinin (PSA), Succinylated WGA, Griffonia simplificola lectin II (GSL II), Datura stramonium lectin (DSL), Erythrina cristagalli lectin (ECL), Jacalin, LEL, Solanum tuberosum lectin (STL), and Vicia villosa agglutinin (VVA) (Vector Labs).
FACS sorting
Neurospheres were grown as described previously and dissociated at desired point of maturity using Accutase (Chemicon). CTB-1 CSCs in single-cell suspension were labeled with either CD-133-gycosylation antibody or fluorescein isothiocyanate (FITC)-labeled DBA for 15 min and centrifuged, supernatant was discarded, and CTB-1 CSCs were re-suspended in neurobasal cell media. Cells were sorted based on their antibody or lectin reactivity. Collected fractions were grown as neurospheres in low-binding T25 flasks using neurobasal media with growth factors as mentioned previously.
Magnetic bead sorting
CTB-1 CSCs in single-cell suspension were labeled with FITC-labeled DBA for 15 min and centrifuged, supernatant was discarded, and CTB-1 CSCs were re-suspended in neurobasal cell media. Anti-FITC magnetic beads (Miltenyi Biotec) were added for 15 min and centrifuged, supernatant was discarded, and CTB-1 CSCs were re-suspended in neurobasal cell media. Collected fractions were grown as neurospheres in low-binding T25 flasks using neurobasal media with growth factors as mentioned previously.
Flow cytometry analysis
Neurospheres were differentiated on polyornithine- (20 μg) and laminin- (10 μg) coated 12-well plates and were harvested at days 1 and 12. CSCs in single-cell suspension were centrifuged and washed 2×in PBS. Fixed (1% para formaldehyde [PFA] for 5 min) and unfixed CSCs were labeled with lectin (1:100 per 2×105 cells) and antibodies. Fluorescently labeled secondary antibodies were conjugated to the primary antibody. Oct4, Sox2, 4′,6-diamidino-2-phenylindole (DAPI), Cleaved Caspase 3, and Ki67 labeling required cell to be made permeable (0.5% Triton X-100 for 3–5 min, Wash 2×). Cells were blocked in 1% bovine serum albumin for 30 min.
Immunofluorescent labeling
Neurospheres were grown on polyornithine- (20 μg) and laminin- (10 μg) coated chamber slides (Lab-Tek) and allowed to differentiate in neurobasal media in the absence of growth factors. Cells were fixed (1% PFA for 5 min) at days 1 and 12, and then labeled with GFAP antibody—to show astrocytic differentiation—and DAPI—to show the location of the nucleus. Isotype-matching fluorescently labeled secondary was used to identify the GFAP primary antibody.
Implantation of neurospheres
Intracranial tumor growth
About 1×105 CD133+-sorted GBM-derived neurosphere cells were intracranially injected into the right frontal hemisphere of NOD/SCID mice. Animals were immobilized on a Narishige stereotactic frame with a custom-designed mouse adaptor using aseptic techniques and body temperature was maintained on a heating pad. About 5 μL of cell suspension was injected with a sterile, glass Hamilton syringe. The needle is used to pierce through the skull of 6-week-old mice and is introduced 2.5 mm left of the bregma at 3.5-mm depth in the striatum. Animals were given Meloxicam post-op and repeated every 8 h if signs of pain were observed. Mice were observed daily by veterinary and research lab staff for neurological signs of distress.
Injection of cells in the flank and immunohistochemistry
Mice were subcutaneously injected with 1×104 cells in the flank region. Once tumor was palpable or the mouse showed signs of distress, the tumor was harvested following sacrifice by cervical dislocation and fixed in 10% neutral-buffered formalin. Following processing and embedding, paraffin tissue blocks were sectioned at 5-micron thickness, mounted on glass microscope slides, and stained for hematoxylin and eosin (H+E). H&E- and Ki67-stained (Biocare Medical) sections were reviewed and photographed.
Cell density (proliferation) assay
The sulforhodamine B (SRB) assay system was used for determination of cell proliferation, based on the measurement of cellular protein content using the dye SRB (Acid Red 52). At specific endpoints, cells were washed, fixed with 50% trichloroacetic acid (TCA), and then air dried and stained (20–30 min) with SBA. Cells were washed with 1% acetic acid and solubilized with 10 mM Tris and samples were read on a spectrophotometer at 565 nm.
Statistical analysis
P value of neurosphere diameter for proliferation was determined by a 1-way analysis of variance. Student's t-test was used for analysis of fluorescence intensities of differentiated and undifferentiated cells.
Results
Cell sorting of CD133+ GBM-derived CSCs using magnetic bead separation
Enzymatically digested tumor cells in single-cell suspension were labeled with biotinylated CD133 antibody. Metallic beads coupled with streptavidin were added to the mixture and cells positive for CD133 were successfully separated from the total population. It has been reported that CD133+ GBM-derived CSCs make up ∼0.1%–10% of the tumor, with the average level reported being <1% [19–22]. Similar to reports on CSC subpopulations, our CD133+ population represented <1% of the total tumor parent population [19–22]. After replicate sorts, our CD133+-positive population averaged 40%–50% of the total sorted population collected. CD133+ cells formed neurospheres and were grown until they reached 150 μm in diameter, at which time they were passaged or frozen and stored. Afterward, neurospheres were plated onto chamber slides where they differentiated into cells that were positive for the astrocytic marker GFAP (Fig. 1). All flow cytometry analyses were done using a cell count of 2×104. Thus, we are able to isolate CD133+ GBM-derived CSCs from GBM and demonstrate that they are capable of differentiating into astrocytic tumor cells.
FIG. 1.
Culturing, sorting, and differentiation of CD133-positive GBM-derived CSCs. (A) Schematic representation of CD133-positive cell sorting. (B) Sorted, dispersed, and expanded CD133-positive CSCs from patient with a high-grade glioma. Neurospheres were cultured in DMEM serum-free media with growth factors mentioned in the experimental procedures (scale bar=200 μm). (C) Differentiation of neurospheres in the absences of growth factors. Immunostaining shows that the protein GFAP [red (DAPI:blue)] is present after differentiation, indicating astrocytic differentiation. (D) Schematic drawing of CD133 protein, including the 8 putative glycosylation sites. Glycosylation is believed to shield epitopes on CD133 protein. CSCs, cancer stem cells; DBA, Dolichos biflorus agglutinin; DAPI, 4′,6-diamidino-2-phenylindole; DMEM, Dulbecco's modified Eagle medium; GBM, glioblastoma; GFAP, glial fibrillary acidic protein.
Lectins identify GalNAc and GlcNAc on the surface of CD133+ GBM-CSCs
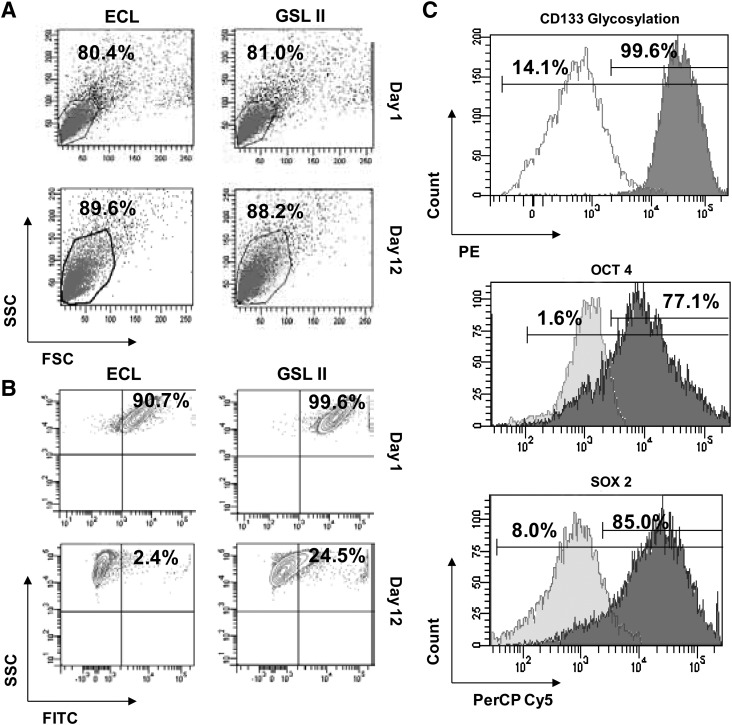
Lectins provide a useful tool for recognizing specific carbohydrates on the surface of cells and identify changes in glycosylation patterns. We used a panel of 20 lectins to study the patterning of CD133+ GBM-CSC surface glycosylation during differentiation (Supplementary Table S1). Neurospheres derived from CD133+ GBM-CSCs were grown as described previously and plated onto polyornithine- and laminin-coated tissue-culture-grade plastics. Within 10 h, neurospheres adhered and extensions became visible. Differentiation continued for 12 days, and harvested cells (day 1 and day 12) (Fig. 2A) were labeled with the panel of lectins. A quantitative evaluation (flow cytometry) of the cell populations showed lectins DBA, PNA, SBA, GSL I, and VVA, all of which recognize the glycan moiety GalNAc, highly labeled undifferentiated GBM-CSCs at day 1 (Fig. 2B, C). Reactivity dramatically decreased after 12 days of differentiation. Similarly, lectins ECL and GSL II, which recognize the glycan moiety GlcNAc, were highly reactive against undifferentiated GBM-CSCs. This reactivity noticeably decreased after 12 days of differentiation (Fig. 3A, B). Further, the reactivity of lectins for GalNAc and GlcNAc was highly correlated with the expression of GBM-CSC marker CD133-G and embryonic stem cell markers OCT4 and SOX2 (Fig. 3C). OCT4 and SOX2 have been well studied in CSCs, in conjunction with CD133 [23–26]. This result shows that specific lectins can identify the glycans GalNAc and GlcNAc expressed on the surface of undifferentiated GBM-CSCs, but that this reactivity is dramatically attenuated when cells are differentiated.
FIG. 2.
Differentiation, α-N-acetylgalactosamine-recognizing lectin labeling, and flow cytometry of CD133-positive cells. (A) Sorted CD133-positive cells were grown as neurospheres and differentiated in the absence of growth factors on laminin-coated plates over a 12-day time course. At day 1, some cells with the rounded, undifferentiated stem cell phenotype were apparent. After 12 days of differentiation, in the absence of growth factors, cells with the rounded, undifferentiated phenotype were still present. (B) Primary GBM tumor cells were sorted for CD133 positivity, expanded as neurospheres (∼2 weeks), and differentiated on laminin-coated plates (12 days). Cells were gated base and analyzed base on SSC versus FSC. (C) Cells from (B) were analyzed for reactivity to fluorescently labeled DBA, PNA, SBA, GSL I, and VVA lectins by flow cytometry. Arrows indicate morphology commonly seen in cells with stemness. After 12 days of differentiation there are stem-like cells still present. PNA, Peanut agglutinin; SBA, Soybean agglutinin; GSL I, Griffonia simplificola lectin I; VVA, Vicia villosa agglutinin; SSC, side scatter; FSC, forward scatter; FITC, fluorescein isothiocyanate.
FIG. 3.
Differentiation, α-N-acetylglucosamine-recognizing lectin labeling, and flow cytometry of CD133-positive cells. (A) Primary GBM tumor cells were sorted for CD133 positivity, expanded as neurospheres (∼2 weeks), and differentiated on laminin-coated plates (12 days). Cells were gated base and analyzed base on SSC versus FSC. (B) Cells from (A) were analyzed for positive reactivity to fluorescently labeled GSL II, and ECL by flow cytometry. (C) Cells from (B) were analyzed for CD133 glycosylation, OCT4, and SOX2 expression. ECL, Erythrina cristagalli lectin; GSL II, Griffonia simplicifolia lectin II; PE, phycoerythrin.
Fluorescently labeled DBA and cell surface GalNAc can sort GBM-CSCs to establish new cell lines with different phenotypes
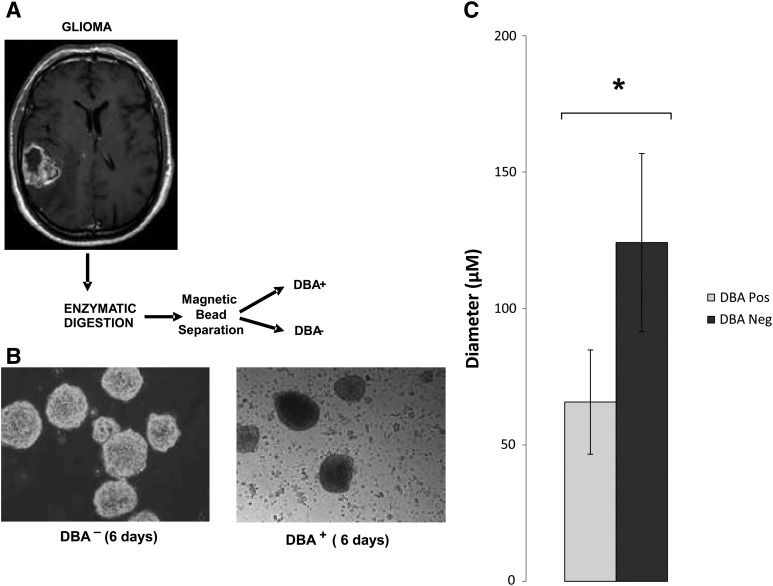
GBM cells, in single-cell suspension, were treated with fluorescently labeled DBA lectin and 2 technologies were employed to separate positively labeled cells from the parent population. BD FACS Aria cell sorter successfully separated and quantitatively analyzed positively labeled cells for DBA lectin from nonlabeled (negative control population) and dual-labeled cells (fluorescently labeled DBA lectin and fluorescently labeled Actin antibody) (Supplementary Fig. S1A, B). DBA has specificity for GalNAc and can be competed off cells using 200 mM free (unbound) GalNAc (Supplementary Fig. S1C), suggesting that a nonspecific interaction between DBA lectin and proteins or other glycans is unlikely. A second method was developed using the autoMACS Pro Separator (Miltenyi Biotec), a benchtop-automated magnetic cell sorter used within a sterile tissue culture cabinet. Using autoMACS magnetic beads that recognized fluorescent labeling we were able to separate fluorescent DBA lectin positively labeled cells from nonlabeled cells (Fig. 4A). DBA-lectin-positive cells (CTB-1) made up <1% of the total parent population and proliferated at a slower rate than the DBA-lectin-negative population (Fig. 4B). The measurement of neurosphere diameters demonstrated that DBA-lectin-bound cells produced smaller neurospheres, suggesting slower proliferation and/or increased cell death, even after repeat passaging (Fig. 4C). Neurospheres derived from DBA-lectin-negative cells were limited in their passage number because they would readily plate down. Ki67, a marker of proliferation, was higher in day 1 DBA-lectin-negative populations (23.8%) than in DBA-lectin-positive populations (16%) (Fig. 5). This trend continued throughout differentiation and, by day 12, 24.5% of DBA-lectin-negative sorted cells were Ki67 positive, while DBA-lectin-positive sorted cells showed a lower Ki67 reactivity (4.9%). Cleaved caspase 3, an indicator of apoptosis, was elevated in the DBA-lectin-negative population at day 1 (25.5%) versus the DBA-lectin-positive population (14.6%); however, by day 12, apoptosis increased in the DBA-lectin-positive population to almost half of the cells (46%) while the DBA-lectin-negative population shows a decrease in cell death (19.6%) (Fig. 5). Increased proliferation in DBA-lectin-negative populations can also be visualized by microscopy at day 1 and day 12 (Supplementary Fig. S2). At day 12 the DBA-lectin-negative population is denser than the DBA-lectin-positive population (Supplementary Fig. S2); however, both populations are able to produce GFAP-positive cells (astrocytes), similar to CD133-positive sorted cells (Supplementary Fig. S2). Positively sorted DBA lectin cells also show high reactivity for lectins PNA, SBA, VVA, and GSL-I, which recognize GalNAc, and were diminished by day 12 (Fig. 6A). GSL-II and ECL, which recognize GlcNAc, are also highly reactive for DBA-lectin-positive sorted cell populations and show reduced reactivity by day 12 (Fig. 6A). DBA-negative sorted cells, on the other hand, showed low reactivity toward GalNAc- and GlcNAc-recognizing lectins and this continued to decrease by day 12 (Fig. 6B) in conjunction with differentiation. ConA, which interacts with high mannose N-glycans, remains high in both undifferentiated and differentiated populations and serves as a positive control (Fig. 6A, B). However, GBM-CSC marker CD133-G and embryonic stem cell markers OCT4 and SOX2 are high in the DBA-lectin-positive sorted cells, and similar to DBA reactivity, they decrease substantially by day 12 of differentiation (Fig. 6A). In the DBA-lectin-negative population, these stem cell markers are uniformly low at day 1 and are almost nonexistent by day 12 (Fig. 6A). Using cell sorting technology with DBA lectin, we were able to separate CSCs that have high expression of stemness markers (CD133+, OCT4+, and SOX2+) from cells with lower expression of stemness markers (CD133−, OCT4−, and SOX2− cells). Cells with lower expression of stemness markers may be already committed to differentiation [11,23,25]. The data show that lectins that recognize GalNAc can be used to sort undifferentiated GBM-CSCs from the original unsorted parent population, or from cells that show a phenotype of commitment to early differentiation, thereby generating new CSC lines.
FIG. 4.
Sorting, culturing, and measurement of neurosphere diameter. (A) Schematic representation of DBA-lectin-positive cell sorting. (B) Sorted, dispersed, and expanded DBA-lectin-positive and DBA-lectin-negative CSCs from a patient with a high-grade glioma. Neurospheres were cultured in DMEM serum-free media with growth factors mentioned in the experimental procedures. DBA-lectin-positive neurospheres (n=27) proliferate at a slower rate and therefore have smaller diameters when compared with the DBA-lectin-negative neurospheres (n=24). (C) Bar graph representation of data collected from (B); *P value ≤0.05.
FIG. 5.
GBM-CSCs at day 1 and day 12 of differentiation labeled for Ki67 and cleaved caspase-3. Bar graph representation of DBA-positive and DBA-negative sorted cells that were grown as neurospheres and differentiated on laminin-coated plates for 12 days. Cells were labeled at day 1 and day 12 of differentiation for (A) Ki67 (proliferation) and (B) cleaved caspase-3 (apoptosis) and analyzed using flow cytometry; *P value ≤0.05.
FIG. 6.
Quantitation of lectin binding on DBA-positive (A) and DBA-negative (B) GBM-CSC (day 1 and day 12) surfaces with 8 different lectins and 3 stem cells markers. The percent of cells with specific carbohydrate expression was determined by flow cytometry using 8 different lectins. The data are means±SD of 3 independent assays of CTB-1 GBM-CSCs. CTB-1 cell line was stained with CD133, OCT4, and SOX2 antibodies in addition to lectin staining. Statistical significance (P≤0.05) is indicated by asterisk. CON A, Concanavalin A; SD, standard deviation.
GalNAc on the surface of GBM-CSCs is recognized independently from CD133-G and CD133
To determine whether the DBA-lectin-recognized glycan GalNAc is an independent structure or part of the already recognized markers CD133-G or CD133, we triple-labeled DBA-lectin-positive sorted cells at day 1 and at day 12 of differentiation with fluorescently labeled DBA lectin, anti-CD133-G, and anti-CD133. At day 1 of differentiation, we observed doubly positive–labeled cell populations for DBA lectin/CD133-G and DBA lectin/CD133, as well as singly labeled cells for DBA lectin, CD133-G, and CD133 (Supplementary Fig. S3A). However, at day 12, labeled cell populations that solely or partially included CD133-G or CD133 decreased in number, while exclusive DBA-lectin-positive populations increased by greater than 2-fold (Supplementary Fig. S3B). Combined, this data shows GalNAc, which is recognized by DBA lectin, is presented on the surface of GBM-CSCs as a structure that is independent of CD133-G and CD133.
DBA-lectin-sorted GBM-CSCs produce high-grade gliomas in NOD/SCID mice
CD133+ (CTB/CD133+) sorted GBM-CSCs were intracranially implanted (1×105) in NOD/SCID mice to demonstrate tumorigenicity. At 6 months, an infiltrative high-grade glioma was noted confirming the orthotopic tumorigenicity of these cells (Supplementary Fig. S4). To determine the tumorigenicity of DBA+-sorted cells (CTB-1/DBA+), 1×104 cells were implanted into the flank of NOD/SCID mice. These CTB-1/DBA+ cells produced a high-grade neoplasm with features of a GBM, including high cell density, an infiltrative pattern, and angiogenesis [Fig. 7A, B (H+E stain)]. DBA+-neurosphere-derived tumors also display high immune reactivity with the Ki67 antibody, confirming their high proliferation rate [Fig. 7A, B (Ki67 stain)] and correlating with our findings of Ki67-positive neurospheres (Fig. 5).
FIG. 7.
Photomicrograph of an H&E- and Ki67-stained high-grade glioma derived from CTB-1/DBA+ GBM neurospheres implanted in flank of NOD/SCID mice. (A) Solid growth pattern of high-grade glioma with high cell density and perivascular growth pattern. Ki67 labeling index was very high (greater than 40%). (B) High-grade malignancy showed features of a GBM including nuclear anaplasia, invasiveness, and proliferation of blood vessels. H&E, hematoxylin and eosin.
We also performed an SRB assay to measure neurosphere growth over 48 h to assess proliferative potential. Our data show increased growth of CD133+ and DBA+ neurospheres over a 48-h time period. At 24 h, CD133+-sorted neurosphere showed slightly more growth than CD133− neurospheres (15% greater growth). DBA+-sorted neurospheres showed less growth than CD133− neurospheres (16%) and significantly less growth than the CD133+ (29% less growth) also at 24 h. However at 48 h, CD133+ and CD133− neurospheres showed a decrease in growth while DBA+ neurospheres continued to show increased growth. Indeed after 48 h, DBA+ neurospheres showed 2% greater growth than CD133+ neurospheres and 29% more growth than CD133− neurospheres. This data suggests that DBA+ neurospheres form tumors in vivo similar to CD133+-sorted neurospheres. Further, DBA+-sorted neurospheres have proliferative properties similar to CD133+-sorted neurospheres that are considered in vitro hallmarks of tumorigenicity.
Discussion
The presence of GalNAc and GlcNAc, independent of other stem cell markers, can be used to identify GBM-CSCs. Here we report the use of lectins as a reliable indicator of stemness in GBM-CSCs derived from a cerebellar GBM. We highlight the ability of lectins to recognize GalNAc and GlcNAc as discriminating markers between undifferentiated GBM-CSCs and cells that differentiated from them. Interestingly, GalNAc and GlcNAc appear to be sugars with the most dramatic changes on the surface of human GBM-CSCs during differentiation.
Our data indicate that there are temporal differences in the expression of GalNAc and GlcNAc epitopes on the surface of GBM-CSCs as they differentiate and these differences correspond to better-known CSC markers. Current GBM-CSC markers (CD133-G, CD133, OCT4, and SOX2) can be categorized into 2 classes: (i) cell surface markers CD133-G and CD133, which are elevated in GBM-CSCs but are rapidly downregulated during lineage commitment, and (ii) OCT4 and SOX2, which are nuclear markers that are generally lost in most stem cells even further down the pathway of lineage commitment. Our observations that GalNAc and GlcNAc are downregulated in conjunction with other GBM-CSC markers now allow us to define cell populations with greater accuracy and temporal resolution. In combination with or in place of existing GBM-CSC markers, lectins that recognize GalNAc and GlcNAc can be used to identify GBM-CSCs in living cell populations without their destruction (ie, RNA extraction). The use of antibodies to identify surface markers has the potential of inducing intracellular changes upon binding to epitopes and may be difficult to compete off. This property of antibodies makes the use of lectins, which generally do not induce intracellular signaling and can be readily competed off, ideal for cell sorting and creating new lines of GBM-CSCs. For example, we were successful in using DBA lectin to sort GBM-CSCs (CTB-1/DBA+) and demonstrate their tumorigenicity in NOD/SCID mice (Fig. 7), similar to CD133+-sorted GBM-CSCs (CTB/CD133+) (Supplementary Fig. S4). Likewise, in vitro assays confirmed that DBA+-sorted cells formed proliferating neurospheres (Fig. 8). The DBA+-sorted cells were not as proliferative as CD133+ or CD133− populations at 24 h; however, growth continued to increase exclusively in the DBA+ neurospheres after 48 h, suggesting slower but continued proliferation that is a characteristic of CSCs.
FIG. 8.
Sulforhodamine B assay for proliferation. (A) CTB/CD133+, CTB-1/DBA+, and CTB/CD133− cell populations begin to form neurospheres within 24 h. (B) Quantitative representation of sulforhodamine B assay (n=9). *P value ≤0.05 versus CTB/CD133− time equivalent. **P value ≤0.05 versus positive population (CTB/CD133+ or CTB-1/DBA+) time equivalent.
When cells are labeled for all 3 surface markers (DBA, CD133-G, and CD133), we note a much smaller population than when we label with a single surface marker. This supports the notion that, although GalNAc is clearly identified as an independent structure (Supplementary Fig. 3A, B), there may be some GalNAc residues found within the CD133-G and CD133 structures. Differences in the affinity of lectins versus antibodies for their targets can contribute to these labeling differences. For example, the association constant of DBA for GalNAc ligands is in the range of 1×103 mol−1, whereas the association constant for an antibody usually ranges between 105 and 1012 mol−1. Further characterization of the DBA epitopes in GBM-CSCs should help resolve this question. Nonetheless, independent DBA labeling remains present on GBM-CSCs and follows an expression trend that is similar to other stem cell markers, even in the near absence of CD133-G and CD133. GalNAc may be a more reliable marker for GBM-CSCs and CSCs in general given the limitations of CD133 that have been described in several reports. GalNAc and GlcNAc residues have been given limited attention with GBM-CSCs, and may also be present on CSCs in other cancer types.
An increase in apoptosis, as indicated by an elevated amount of cleaved caspase 3, may be a contributing factor to the small percentage of CSCs that exist within a tumor. At day 1, in the DBA-lectin-positive sorted CSCs, cleaved caspase 3 was low while OCT4 was high, which has been reported in other CSCs [23]. However upon differentiation, our data showed that by day 12, OCT4 is hardly present (Fig. 5) and cleaved caspase 3 increased by 3-fold (Supplementary Fig. S2), indicating an increase in apoptosis with cell differentiation. In addition, by day 12 in the same population, proliferation rates decreased to 1/3 that noted at day 1, perturbing the production of replacement cells. Most interesting, DBA recognition of GalNAc, OCT4, and SOX2 levels was almost the same at day 12, in both DBA-lectin-positive and DBA-lectin-negative sorted populations. We expected apoptosis and proliferation to be similar. However, there were statistically significant differences between the 2 populations. This causes us to question whether there are multiple subpopulations of GBM-CSCs that coexist and uniquely contribute to the formation of the tumor; this has been reported in other CSC lines [27]. However, additional research is needed to confirm this theory of multiple subsets of GBM-CSCs that coexist in the same tumor.
The use of lectins to identify cell surface markers is highlighted by their ability to discriminate between GBM-derived CSCs and differentiated tumor cells. Because differentiated tumor cells have lost their ability to self-renew, lectins are likely to have utility as an indicator of differentiation commitment. Interestingly, in our study, the only glycans to change substantially on the cell surface during differentiation were GalNAc and GlcNAc (Table 1). It will be interesting to determine how GalNAc and GlcNAc epitopes are maintained on the surface of GBM-derived CSCs and to identify any proteins or lipids that may carry these glycans. The current marker for GBM-derived CSCs, CD133, has known limits in its usage. CD133 is not restricted to CSCs, but its expression is more widespread and has been found on differentiated tumor cells [28,29]. Almost all CD133-related experiments to date have made use of 2 antibodies, AC133 and AC141, which are widely reported to bind to glycosylation on CD133. Little is known about the molecular nature of the epitopes recognized by the antibodies [29,30]. Since the nature of the glycosylated epitopes has not been defined, it is possible that the antibodies AC133 and AC141 nonspecifically recognize glycosylation on molecules other than CD133 [31]. Reports show that CD133 glycosylation can be downregulated independently of CD133, which is believed to be differentially folded as a result of glycosylation, therefore shielding specific proteomic epitopes [28]. Our results show that CD133 expression diminishes during differentiation, but this may actually be a masking effect of CD133 glycosylation. The CD133 antibody that we used recognizes the N-terminal epitope, which has been found to be masked by ganglioside binding during differentiation [32]. However, the AC133 antibody (CD133-G) that we used recognizes glycosylation in the second extracellular loop and therefore is not related to the N-terminus. An additional complicating factor is the presence of alternatively spliced isoforms of CD133. Several alternatively spliced forms of CD133 have been reported in human, and although to date they have not been identified, researchers believe that CD133 isoforms lacking the AC133 and AC141 epitopes could exist [33–35]. In total, there is justifiable concern with the exclusive use of CD133 or its glycosylation as a marker for GBM-CSCs and GalNAc- and GlcNAc-recognizing lectins may be a suitable alternative. The embryonic stem cell markers OCT4 and SOX2 are reliable indicators of stemness, but their use is time consuming and will require the destruction of the cell.
Table 1.
Summary of Data Obtained Using a Panel of 20 Fluorescein-Isothiocyanate-Labeled Lectins on Unfixed hGBM Cancer Stem Cells (CTB-1), Grown on Laminin in CTB-1 Neuronal Media at Day 1 and Day 12 in the Absence of Growth Factors
| Lectin | Day 1 | Day 12 |
|---|---|---|
| CON A | + | + |
| DBA | + | − |
| PNA | + | − |
| RCA 120 | + | + |
| SBA | + | − |
| UEA I | − | − |
| WGA | + | + |
| GSL I | + | − |
| LCA | + | + |
| PHA-E | + | + |
| PHA-L | + | + |
| PSA | + | + |
| Succinylated WGA | + | + |
| GSL II | + | − |
| DSL | + | + |
| ECL | + | - |
| Jacalin | + | + |
| LEL | + | + |
| STL | + | + |
| VVA | + | − |
The experiments were repeated on fixed with the same result. Lectins that showed changes in reactivity between day 1 and day 12 are in bold.
CON A, Concanavalin A; DBA, Dolichos biflorus agglutinin; PNA, Peanut agglutinin; LEL, Lycopersicon esculetum lectin; RCA, Ricinus communis agglutinin; SBA, Soybean agglutinin; UEA I, Ulex europaeus agglutinin I; WGA, Wheat germ agglutinin; GSL I, Griffonia simplificola lectin I; LCA, Lens culinaris agglutinin; PHA-E, Phaseolus vulgaris Erythroagglutinin; PHA-L, Phaseolus vulgaris Leucoagglutinin; PSA, Pisum sativum agglutinin; SWGA, Succinylated Wheat Germ agglutinin; DSL, Datura stramonium lectin; ECL, Erythrina cristagalli lectin; LEL, Lycopersicon esculentum lectin; STL, Solanum tuberosum lectin; VVA, Vicia villosa agglutinin; hGBM, human glioblastoma multiforme.
In conclusion, a novel aspect of this work is the establishment of GalNAc and GlcNAc as nondestructive markers for GBM-derived CSCs and as a reliable readout for initial differentiation events, at a level of temporal resolution that was not previously possible. Our current investigation has the limitations of a solitary GBM used for analysis and therefore these same studies should be applied to great number of glial neoplasms from a variety of locations [36,37]. However, lectins can be used as recognition tools for these glycan markers that are located on the surface of cells, allowing easy access for cell sorting, culturing, and harvesting of a specific cell population. This offers an alternative to previous methodologies that are limiting and may require destructive techniques.
Supplementary Material
Acknowledgments
A very special thank you to Dianne Alexis, MLS, QIHC(ASCP), for all histology performed in this project. The authors thank Saju Samuel (COO; Jeevan Biosciences, Inc.) for administrative support of this project. Linda J. Roback and Louise McCormick, Ph.D., for technical and laboratory support. Rachel J. Nash (Jeevan Biosciences, Inc.) for data analysis and support. This project was funded in part by the Public Health Service National Institutes of Health (NIH) awards CA149107 (D.J.B.), CA139035 (D.J.B.), and Jeevan Biosciences, INC.-IANAFS.
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Brat DJ. Scheithauer BW. Fuller GN. Tihan T. Newly codified glial neoplasms of the 2007 WHO classification of tumours of the central nervous system: angiocentric glioma, pilomyxoid astrocytoma and pituicytoma. Brain Pathol. 2007;17:319–324. doi: 10.1111/j.1750-3639.2007.00082.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fuller GN. Scheithauer BW. The 2007 revised world health organization (WHO) classification of tumours of the central nervous system: newly codified entities. Brain Pathol. 2007;17:304–307. doi: 10.1111/j.1750-3639.2007.00084.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Louis DN. Ohgaki H. Wiestler OD. Cavenee WK. Burger PC. Jouvet A. Scheithauer BW. Kleihues P. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114:97–109. doi: 10.1007/s00401-007-0243-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Griguer CE. Oliva CR. Gobin E. Marcorelles P. Benos DJ. Lancaster JR. Gillespie GY., Jr. CD133 is a marker of bioenergetic stress in human glioma. PLoS One. 2008;3:e3655. doi: 10.1371/journal.pone.0003655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hemmati HD. Nakano I. Lazareff JA. Masterman-Smith M. Geschwind DH. Bronner-Fraser M. Kornblum HI. Cancerous stem cells can arise from pediatric brain tumors. Proc Natl Acad Sci U S A. 2003;100:15178–15183. doi: 10.1073/pnas.2036535100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kondo T. Setoguchi T. Taga T. Persistence of a small subpopulation of cancer stem-like cells in the C6 glioma cell line. Proc Natl Acad Sci U S A. 2004;101:781–786. doi: 10.1073/pnas.0307618100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.P. Metellus IN-M. Delfino C. Colin C. Coulibaly B. Fina F. Chinot OL. Figarella-Branger D. Ouafik L. Prognostic impact of stem cell marker CD133 in 61 glioblastoma patients treated with concomitant chemoradiation: a prospective study. J Clin Oncol 2010 ASCO Annual Meeting Proceedings (Post-Meeting Edition); 2010. p. 2027. [Google Scholar]
- 8.Beier D. Hau P. Proescholdt M. Lohmeier A. Wischhusen J. Oefner PJ. Aigner L. Brawanski A. Bogdahn U. Beier CP. CD133(+) and CD133(−) glioblastoma-derived cancer stem cells show differential growth characteristics and molecular profiles. Cancer Res. 2007;67:4010–4015. doi: 10.1158/0008-5472.CAN-06-4180. [DOI] [PubMed] [Google Scholar]
- 9.Lathia JD. Venere M. Rao MS. Rich JN. Seeing is believing: are cancer stem cells the loch ness monster of tumor biology? Stem Cell Rev. 2011;7:227–237. doi: 10.1007/s12015-010-9194-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sun Y. Kong W. Falk A. Hu J. Zhou L. Pollard S. Smith A. CD133 (Prominin) negative human neural stem cells are clonogenic and tripotent. PLoS One. 2009;4:e5498. doi: 10.1371/journal.pone.0005498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nash R. Neves L. Faast R. Pierce M. Dalton S. The lectin Dolichos biflorus agglutinin recognizes glycan epitopes on the surface of murine embryonic stem cells: a new tool for characterizing pluripotent cells and early differentiation. Stem Cells. 2007;25:974–982. doi: 10.1634/stemcells.2006-0224. [DOI] [PubMed] [Google Scholar]
- 12.Venable A. Mitalipova M. Lyons I. Jones K. Shin S. Pierce M. Stice S. Lectin binding profiles of SSEA-4 enriched, pluripotent human embryonic stem cell surfaces. BMC Dev Biol. 2005;5:15. doi: 10.1186/1471-213X-5-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Plattner VE. Wagner M. Ratzinger G. Gabor F. Wirth M. Targeted drug delivery: binding and uptake of plant lectins using human 5637 bladder cancer cells. Eur J Pharm Biopharm. 2008;70:572–576. doi: 10.1016/j.ejpb.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 14.Miettinen M. Holthofer H. Lehto VP. Miettinen A. Virtanen I. Ulex europaeus I lectin as a marker for tumors derived from endothelial cells. Am J Clin Pathol. 1983;79:32–36. doi: 10.1093/ajcp/79.1.32. [DOI] [PubMed] [Google Scholar]
- 15.Lalwani AK. Carey TE. Goldstein IJ. Peters BP. Lectin binding characteristics of squamous cell carcinomas of the head and neck. Acta Otolaryngol. 1996;116:125–131. doi: 10.3109/00016489609137725. [DOI] [PubMed] [Google Scholar]
- 16.Hohenberger P. Liewald F. Schlag P. Herfarth C. Lectins and immunohistochemistry of colorectal cancer, its recurrences and metastases. Eur J Surg Oncol. 1990;16:289–297. [PubMed] [Google Scholar]
- 17.Muramatsu H. Hamada H. Noguchi S. Kamada Y. Muramatsu T. Cell-surface changes during in vitro differentiation of pluripotent embryonal carcinoma cells. Dev Biol. 1985;110:284–296. doi: 10.1016/0012-1606(85)90088-0. [DOI] [PubMed] [Google Scholar]
- 18.Ozawa M. Muramatsu T. The glycoprotein-bound large carbohydrates from embryonal carcinoma cells carry receptors for several lectins recognizing N-acetylgalactosamine and galactose. J Biochem. 1985;97:317–324. doi: 10.1093/oxfordjournals.jbchem.a135056. [DOI] [PubMed] [Google Scholar]
- 19.Dick JE. Looking ahead in cancer stem cell research. Nat Biotechnol. 2009;27:44–46. doi: 10.1038/nbt0109-44. [DOI] [PubMed] [Google Scholar]
- 20.Lasky JL., 3rd Choe M. Nakano I. Cancer stem cells in pediatric brain tumors. Curr Stem Cell Res Ther. 2009;4:298–305. doi: 10.2174/157488809789649278. [DOI] [PubMed] [Google Scholar]
- 21.Li C. Heidt DG. Dalerba P. Burant CF. Zhang L. Adsay V. Wicha M. Clarke MF. Simeone DM. Identification of pancreatic cancer stem cells. Cancer Res. 2007;67:1030–1037. doi: 10.1158/0008-5472.CAN-06-2030. [DOI] [PubMed] [Google Scholar]
- 22.Singh SK. Clarke ID. Hide T. Dirks PB. Cancer stem cells in nervous system tumors. Oncogene. 2004;23:7267–7273. doi: 10.1038/sj.onc.1207946. [DOI] [PubMed] [Google Scholar]
- 23.Chen YC. Hsu HS. Chen YW. Tsai TH. How CK. Wang CY. Hung SC. Chang YL. Tsai ML, et al. Oct-4 expression maintained cancer stem-like properties in lung cancer-derived CD133-positive cells. PLoS One. 2008;3:e2637. doi: 10.1371/journal.pone.0002637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ong CW. Kim LG. Kong HH. Low LY. Iacopetta B. Soong R. Salto-Tellez M. CD133 expression predicts for non-response to chemotherapy in colorectal cancer. Mod Pathol. 2010;23:450–457. doi: 10.1038/modpathol.2009.181. [DOI] [PubMed] [Google Scholar]
- 25.Saigusa S. Tanaka K. Toiyama Y. Yokoe T. Okugawa Y. Ioue Y. Miki C. Kusunoki M. Correlation of CD133, OCT4, and SOX2 in rectal cancer and their association with distant recurrence after chemoradiotherapy. Ann Surg Oncol. 2009;16:3488–3498. doi: 10.1245/s10434-009-0617-z. [DOI] [PubMed] [Google Scholar]
- 26.Baal N. Reisinger K. Jahr H. Bohle RM. Liang O. Munstedt K. Rao CV. Preissner KT. Zygmunt MT. Expression of transcription factor Oct-4 and other embryonic genes in CD133 positive cells from human umbilical cord blood. Thromb Haemost. 2004;92:767–775. doi: 10.1160/TH04-02-0079. [DOI] [PubMed] [Google Scholar]
- 27.Hwang-Verslues WW. Kuo WH. Chang PH. Pan CC. Wang HH. Tsai ST. Jeng YM. Shew JY. Kung JT, et al. Multiple lineages of human breast cancer stem/progenitor cells identified by profiling with stem cell markers. PLoS One. 2009;4:e8377. doi: 10.1371/journal.pone.0008377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kemper K. Sprick MR. de Bree M. Scopelliti A. Vermeulen L. Hoek M. Zeilstra J. Pals ST. Mehmet H. Stassi G. Medema JP. The AC133 epitope, but not the CD133 protein, is lost upon cancer stem cell differentiation. Cancer Res. 2010;70:719–729. doi: 10.1158/0008-5472.CAN-09-1820. [DOI] [PubMed] [Google Scholar]
- 29.Miraglia R. Caruso S. Maruzzelli L. Luca A. Gridelli B. Role of sonography in the diagnosis and treatment of a large iatrogenic pseudoaneurysm of a hepatic artery. J Clin Ultrasound. 2010;38:450–453. doi: 10.1002/jcu.20679. [DOI] [PubMed] [Google Scholar]
- 30.Bidlingmaier S. Zhu X. Liu B. The utility and limitations of glycosylated human CD133 epitopes in defining cancer stem cells. J Mol Med. 2008;86:1025–1032. doi: 10.1007/s00109-008-0357-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Potgens AJ. Schmitz U. Kaufmann P. Frank HG. Monoclonal antibody CD133-2 (AC141) against hematopoietic stem cell antigen CD133 shows crossreactivity with cytokeratin 18. J Histochem Cytochem. 2002;50:1131–1134. doi: 10.1177/002215540205000814. [DOI] [PubMed] [Google Scholar]
- 32.Taieb N. Maresca M. Guo XJ. Garmy N. Fantini J. Yahi N. The first extracellular domain of the tumour stem cell marker CD133 contains an antigenic ganglioside-binding motif. Cancer Lett. 2009;278:164–173. doi: 10.1016/j.canlet.2009.01.013. [DOI] [PubMed] [Google Scholar]
- 33.Shmelkov SV. Jun L. St. Clair R. McGarrigle D. Derderian CA. Usenko JK. Costa C. Zhang F. Guo X. Rafii S. Alternative promoters regulate transcription of the gene that encodes stem cell surface protein AC133. Blood. 2004;103:2055–2061. doi: 10.1182/blood-2003-06-1881. [DOI] [PubMed] [Google Scholar]
- 34.Yu Y. Flint A. Dvorin EL. Bischoff J. AC133-2, a novel isoform of human AC133 stem cell antigen. J Biol Chem. 2002;277:20711–20716. doi: 10.1074/jbc.M202349200. [DOI] [PubMed] [Google Scholar]
- 35.Fargeas CA. Joester A. Missol-Kolka E. Hellwig A. Huttner WB. Corbeil D. Identification of novel Prominin-1/CD133 splice variants with alternative C-termini and their expression in epididymis and testis. J Cell Sci. 2004;117:4301–4311. doi: 10.1242/jcs.01315. [DOI] [PubMed] [Google Scholar]
- 36.Broniscer A. Gajjar A. Supratentorial high-grade astrocytoma and diffuse brainstem glioma: two challenges for the pediatric oncologist. Oncologist. 2004;9:197–206. doi: 10.1634/theoncologist.9-2-197. [DOI] [PubMed] [Google Scholar]
- 37.Utsuki S. Oka H. Miyajima Y. Kijima C. Yasui Y. Fujii K. Adult cerebellar glioblastoma cases have different characteristics from supratentorial glioblastoma. Brain Tumor Pathol. 2011 2011 Nov 11;:1–9. doi: 10.1007/s10014-011-0070-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.