Abstract
Domestic coal combustion has had profound adverse effects on the health of millions of people worldwide. In China alone several hundred million people commonly burn raw coal in unvented stoves that permeate their homes with high levels of toxic metals and organic compounds. At least 3,000 people in Guizhou Province in southwest China are suffering from severe arsenic poisoning. The primary source of the arsenic appears to be consumption of chili peppers dried over fires fueled with high-arsenic coal. Coal samples in the region were found to contain up to 35,000 ppm arsenic. Chili peppers dried over high-arsenic coal fires adsorb 500 ppm arsenic on average. More than 10 million people in Guizhou Province and surrounding areas suffer from dental and skeletal fluorosis. The excess fluorine is caused by eating corn dried over burning briquettes made from high-fluorine coals and high-fluorine clay binders. Polycyclic aromatic hydrocarbons formed during coal combustion are believed to cause or contribute to the high incidence of esophageal and lung cancers in parts of China. Domestic coal combustion also has caused selenium poisoning and possibly mercury poisoning. Better knowledge of coal quality parameters may help to reduce some of these health problems. For example, information on concentrations and distributions of potentially toxic elements in coal may help delineate areas of a coal deposit to be avoided. Information on the modes of occurrence of these elements and the textural relations of the minerals and macerals in coal may help predict the behavior of the potentially toxic components during coal combustion.
The U.S. Environmental Protection Agency (EPA) recently issued a report to Congress on the health impacts of 189 potentially hazardous air pollutants (HAPs) emitted from coal-burning electric utility generators (1). In this report the EPA concludes that, with the exception of mercury, there is no compelling evidence to indicate that trace element emissions cause human health problems. The absence of detectable health problems is, in part, caused by the fact that the coals burned in the U.S. generally contain low to modest concentrations of HAP elements and that many coal-burning utilities use sophisticated pollution control systems that efficiently reduce the emissions of HAPs (2).
Such is not the case in many developing countries, especially in homes where coal is used for heating and cooking. Domestic use of coal can present serious human health problems because the coals generally are mined locally with little regard to their composition, and the coals are commonly burned in poorly vented or unvented stoves, directly exposing residents to the emissions.
This paper briefly describes health problems believed to be caused by, or exacerbated by, trace elements or organic compounds emitted during domestic combustion of coal. Although the examples used to illustrate these problems are taken from China, people in many other developing and undeveloped countries use coal in a similar way and may suffer from similar health problems.
China is the world’s largest coal producer and coal consumer. Coal production has increased steadily during the past 25 years to nearly 1.4 billion metric tons in 1996 (5). In contrast to most developed countries, such as the U.S., where domestic coal use constitutes a small fraction of 1% of coal consumption, a substantial portion of China’s coal is used for domestic energy needs. Smith and Liu (3) estimate that worldwide 330 million people rely on coal for domestic energy needs and as many as 2.5–3 billion people are using even poorer-quality biomass fuels. However, Florig (4) estimates that more than 75% of China’s primary energy needs are supplied by domestic coal. Coal stoves and small coal boilers provide more than 50% of the energy for urban households and 22% of rural households rely on coal (4). About 70 percent of the population in China resides in rural areas, thus Florig’s data would indicate that about 400 million people in China rely on coal for their domestic energy needs.
Health Problems Caused by Trace Element Emissions
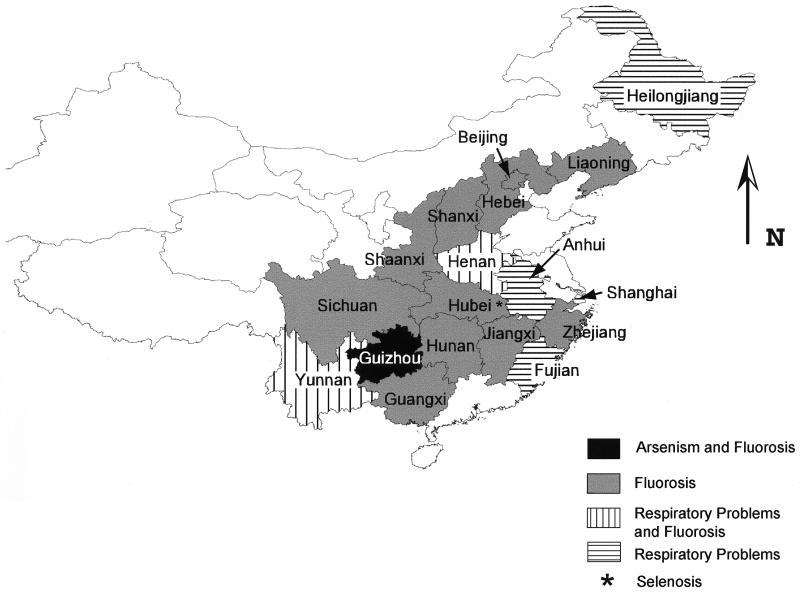
Wood had long been the primary source of energy in southwest China, but by the early part of this century the forests were largely denuded and the residents were forced to seek other sources of fuel. In southwest Guizhou Province (Fig. 1), surface exposures of coal are plentiful, and coal quickly became the primary fuel for domestic use. Unfortunately, some of these coals have undergone mineralization, causing their enrichment in potentially toxic trace elements such as arsenic, fluorine, mercury, antimony, and thallium.
Figure 1.
Map of China indicating provinces in which there are reported health problems caused by domestic coal combustion.
Burning the mineralized coals in unvented stoves volatilizes the toxic elements and exposes the local population to the toxic elements in the emissions. The situation is exacerbated by the practice of drying crops directly over the coal fires. In the autumn it is commonly cool and damp in the higher elevations of Guizhou Province. It is common practice for the residents of this region to dry their corn and chili peppers directly over the burning coals.
Arsenic.
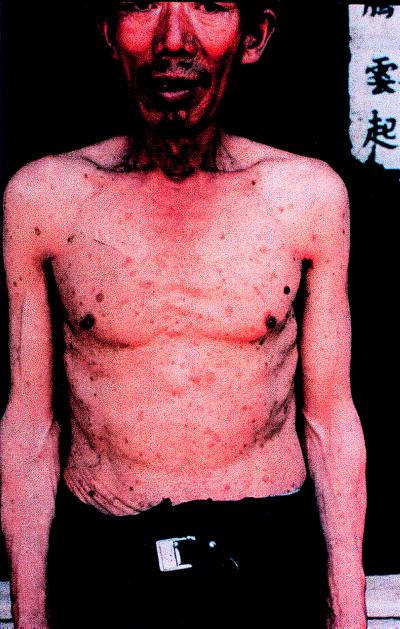
Chronic arsenic poisoning, which affects at least 3,000 people in Guizhou Province, has been described by Zheng and others (6). Those affected exhibit typical symptoms of arsenic poisoning, including hyperpigmentation (flushed appearance, freckles), hyperkeratosis (scaly lesions on the skin, generally concentrated on the hands and feet), Bowen’s disease (dark, horny, precancerous lesions of the skin: Fig. 2), and squamous cell carcinoma.
Figure 2.
Extensive scaly lesions (hyperkeratosis) are evident on the chest of a resident of this region. The dark spot over the left breast was diagnosed as Bowen’s disease.
Zheng and others (6) have shown that chili peppers dried over open coal-burning stoves may be a principal vector for the arsenic poisoning. Fresh chili peppers have less than 1 ppm arsenic. In contrast, chili peppers dried over high-arsenic coal fires can have more than 500 ppm arsenic. Significant amounts of arsenic also may come from other tainted foods, ingestion of dust (samples of kitchen dust contained as much as 3,000 ppm arsenic), and from inhalation of indoor air polluted by arsenic derived from coal combustion. The arsenic content of drinking water samples was below the Environmental Protection Agency’s drinking water standard (7) of 50 ppb and does not appear to be an important factor.
Detailed chemical and mineralogical characterization of the arsenic-bearing coal samples from this region recently was conducted by Belkin and coworkers (8–10). They analyzed about 25 coal samples that they had collected from several locations within Guizhou Province. Instrumental neutron activation analyses of the coal indicate arsenic concentrations as high as 35,000 ppm. The magnitude of this concentration can be seen by comparison with U.S. coals. The mean concentration for arsenic in nearly 10,000 U.S coal samples is approximately 22 ppm, with a maximum value of about 2,000 ppm (11).
Belkin and coworkers (8–10) examined polished blocks of the coal by using a scanning electron microscope (SEM) equipped with energy-dispersive x-ray analyzer (EDS) and an electron microprobe (EMP). They observed a wide variety of As-bearing mineral phases in the coal samples. Pyrite is the most common sulfide, occurring as framboids, euhedral crystals, and irregular shapes. The range of As in pyrite determined by EMP analyses is from the detection limit (≈100 ppm) in unaltered framboids to about 4.5 weight percent in grains adjacent to arsenopyrite crystals. Arsenopyrite occurs in a variety of habits, including large 150- to 250-μm crystals, narrow, 1- to 5-μm veins, and small crystals (Fig. 3A). Selenium contents in arsenopyrite are fairly uniform (0.15 to 0.2 weight percent). A third As-bearing sulfide, composed of As, Pb, and S, is present rarely.
Figure 3.
(A) SEM back-scattered electron image of polished coal showing a pyrite grain (P) and adjacent arsenopyrite crystals (A). (B) SEM back-scattered electron image of polished coal showing the deposition of a banded complex of iron oxide and As-bearing iron phosphates. (C) SEM back-scattered electron image of polished block of arsenic-rich coal. Dark areas are coal, bright areas are mainly pyrite, milky area is coal containing organically bound arsenate. Fluids moving through the fracture in the coal appears to have removed arsenic from the organic matrix. (D) X-ray map depicting the distribution of arsenic in the coal. Red areas are high concentrations, and blue areas are low concentrations. Compare distribution of arsenic to the outline of the milky area in C. A–C originally appeared in ref. 9 and are republished with the permission of the Pittsburgh Coal Conference.
Another group of As-bearing minerals contains arsenic in the 5+ valence state as arsenate commonly substituting for the phosphate group. An unidentified As-bearing iron phosphate, usually associated with banded iron oxide (Fig. 3B), as veins or masses has a P/As ratio on the order of 4. Jarosite [K2Fe63+(SO4)4(OH)12] was present as an alteration product of sulfides or as mixtures with iron oxide and commonly contained a few weight percent As. An additional As-rich phase was observed only as scattered micron-sized grains that contained only Fe and As (±O), as identified by SEM-EDS. The atomic ratio of Fe/As in this mineral is about 1 and the mineral may be scorodite, FeAsO4⋅2H2O.
Some coals display evidence of movement of epigenetic fluids and show primary phases and their alteration products. One sample examined by Belkin and coworkers (8–10) showed a complete range of diagenetic, low-As framboids that progresses through various stages to framboid pseudomorphs composed totally of As-bearing iron oxide (SEM-EDS and reflected light indicates hematite). Veins of jarosite, arsenopyrite, and iron oxide are common in some samples.
Three samples from the same location had As concentrations in excess of 3 weight percent and were mineralogically unusual. Although they contain small grains and veins of arsenopyrite and As-bearing pyrite, the concentration of these phases is completely inadequate to account for the As abundance on a whole coal basis. However, in SEM back-scattered electron images, a distinct banding characterized by differing image brightness is easily observed (Fig. 3C). Some of this banding forms box-like arrangements, but in all cases the bands appear to have sharp edges. The bands range from a few μm to tens and a few hundreds of μm in thickness. SEM-EDS results show that these bright bands are highly enriched in As (Fig. 3D). They also contain S, Fe, and traces of Al and Si (μm-sized clay particles). In fact, there is a relationship between the EDS count intensity for As and apparent brightness of the SEM image. Semiquantitative analysis by SEM-EDS demonstrate that the bright bands contain As at levels ≈3 weight percent. Fe concentrations in the bands are low but always present at levels from 0.2 to 0.4 weight percent, S is the only other major element found. By using an SEM, no discrete As-bearing phases could be resolved in these bands at 50,000 times magnification. Thin fragments of one sample were examined by an advanced field-emission transmission electron microscope. No discrete As-bearing phase could be observed by using this instrument at magnifications of 1 million times. Thus, finely dispersed arsenopyrite, As-bearing pyrite, or any other As-phase can be ruled out as the source of the As.
To define the nature of bonding in the arsenic-bearing phases, a reconnaissance study of two high-arsenic samples was conducted by using high-energy x-rays from a synchrotron source (12). Collection of diffraction spectrum intensity across the XANES (x-ray absorption near-edge structure) and EXAFS (extended x-ray absorption fine structure) regions of an absorption spectrum can provide three-dimensional information on the electronic state and chemical coordination for each crystallographic site of the chosen element. Results from this work demonstrate that ≈100% of the As in one sample is AsO43− and that about 75% of the As in the other sample is AsO43− with the balance (25%) as sulfide-bound As. Thus, for the two coals examined, the preponderance of the As is in the 5+ valence state.
An interesting and potentially important relationship exists between arsenic and gold. Southwest Guizhou Province contains numerous gold mines and deposits that have been identified as Carlin-type gold deposits (13). Carlin-type deposits are characterized by very fine-grained gold (generally less than 1 μm) and are associated with arsenic, antimony, mercury, and thallium. In Carlin-type deposits of the Great Basin region of Nevada, the sedimentary host rock is usually rich in organic matter. The Guizhou Province deposits also contain thallium and mercury (14–16). Ashley and others (13) concluded that the Chinese and Great Basin deposits formed from low-salinity fluids at relatively low temperatures. Li and Peters (17), using fluid inclusion data, indicate that the Guizhou fluids ranged in temperature from 150°C to 240°C, comparable to the U.S. occurrences. It is geologically reasonable to assume that the introduction of arsenic into the coal strata is related to gold mineralization, although the exact mechanism is uncertain.
Mineralogical characterization of the coals from Guizhou Province may help elucidate the geologic process that created the high-arsenic coals and the relationship of the high-arsenic coals to the gold. Knowledge of these processes and relationships may help determine the regional distribution of these environmentally dangerous coals. Information on the arsenic mineralogy also may help us to anticipate the behavior of arsenic during coal combustion. Preliminary characterization of residual ash in coal-burning stoves indicates high retention of arsenic. Mineralogical characterization in conjunction with combustion tests may determine whether one or more of the arsenic-bearing phases is primarily responsible for adsorption of arsenic on the chili peppers.
Fluorine.
The health problems caused by fluorine volatilized during domestic coal use are far more extensive than those caused by arsenic (Fig. 1). More than 10 million people in Guizhou Province and surrounding areas suffer from various forms of fluorosis (18, 19), and it also has been reported from 13 other provinces, autonomous regions, and municipalities in China (20).
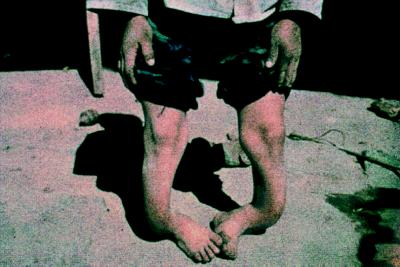
Typical symptoms of fluorosis include mottling of tooth enamel (dental flurosis) and various forms of skeletal fluorosis, including osteosclerosis, limited movement of the joints, and outward manifestations such as knock knees, bow legs, and spinal curvature. Fluorosis combined with nutritional deficiencies in children can result in severe bone deformation (Fig. 4).
Figure 4.
Bone deformation caused by nutritional deficiency combined with exposure to high levels of fluorine from domestic coal combustion.
The etiology of fluorosis is similar to that of arsenism in that the disease is derived from foods dried over coal-burning stoves. Zheng and Huang (18) have demonstrated that adsorption of fluorine by corn dried over unvented ovens burning high (>200 ppm) fluorine coal is the probable cause of the extensive dental and skeletal fluorosis in southwest China. The mode of occurrence of fluorine in the coal is unknown. The problem is compounded by the use of clay as a binder for making briquettes. The clay used is a high-fluorine (mean value of 903 ppm) residue formed by intense leaching of a limestone substrate. Ando and others (20) determined fluorine contents of coals from two mines (559 and 802 ppm) and in the associated soils (592 and 669 ppm). They estimated that 97% of the fluoride exposure came from food consumption and 2% from direct inhalation. Zhang and Cao (19) report mean fluorine levels in coals from 11 regions in China to range from 203 to 1,513 ppm with a maximum value of 3,762 ppm.
Mercury.
There is also considerable concern about the health effects of mercury and the proportion of anthropogenic mercury in the environment (21). So far, there is no direct evidence of health problems caused by mercury released from coal but there are circumstances where poisoning from mercury released from coal combustion may be occurring. Zhou and Liu (22) reported on chronic thallium poisoning in Guizhou Province, China, where the source of the thallium poisoning appears to be from vegetables grown on a mercury/thallium-rich mining slag. Most symptoms, such as hair loss, are typical of thallium poisoning. However, loss of vision in several patients from this region was considered to be unique (R. Dart, personal communication). Mineralogical analysis of the coal being used in the homes of people having visual impairment revealed abundant mercury minerals. Chemical analysis of a coal sample being used in Guizhou Province, China, indicates a mercury concentration of 55 ppm, which is about 200 times the average mercury concentration in U.S. coals.
Selenium.
Zheng and others (23) report nearly 500 cases of human selenosis in southwest China that are attributed to the use of selenium-rich carbonaceous shales known locally as “stone coal.” The stone coals have as much as 8,390 ppm selenium. This selenosis is attributed to the practice of using combustion ash as a soil amendment. This process introduced large amounts of selenium into the soil and resulted in selenium uptake by crops. Symptoms of selenium poisoning include hair and nail loss.
Organic Compounds.
Esophageal cancer is a common fatal cancer and the fourth-leading cause of cancer death in China. Parts of Henan Province in north-central China (Fig. 1) have some of the highest rates of esophageal cancer in the world, with annual age-adjusted mortality rates of up to 169 per 100,000 and cumulative death rates of over 20% by age 75 for both sexes (24). Many studies have been carried out on the high incidence of esophageal and lung cancers in China, but the dominant causative agents of the cancer remain unclear. Polycyclic aromatic hydrocarbons (PAHs) released during unvented coal combustion in homes in China, have been cited as the primary cause for the highly elevated incidence of lung cancer (25). The PAH levels in homes burning “smoky” coal are so high that the resulting lung cancer mortality rate is five times the national average of China (26).
Conclusions
A better knowledge of coal quality parameters may help to minimize some of the health problems caused by domestic coal use. Information on the concentrations and distributions of potentially toxic elements in coal may assist people dependent on local coal sources to avoid those areas of a coal deposit having undesirably high concentrations of toxic compounds. Information on the modes of occurrence of potentially toxic elements and the textural relations of the minerals and macerals in which they occur may help us to anticipate the behavior of the potentially toxic components during coal cleaning, combustion, weathering, and leaching. Coal characterization offers geoscientists opportunities to directly contribute improved public health.
ABBREVIATIONS
- SEM
scanning electron microscope
- EDS
energy-dispersive x-ray analyzer
References
- 1.U.S. Environmental Protection Agency. Study of Hazardous Air Pollutant Emissions from Electric Utility Steam-Generating Units. Washington, DC: Government Printing Office; 1998. EPA-453/R-98-004A. [Google Scholar]
- 2.U.S. Environmental Protection Agency. Study of Hazardous Air Pollutant Emissions from Electric Utility Steam-Generating Units: Interim Final Report. Washington, DC: Government Printing Office; 1996. , EPA-453/R-96–013a. [Google Scholar]
- 3.Smith K R, Liu Y. In: Epidemiology of Lung Cancer. Samet J M, editor. New York: Dekker; 1994. pp. 151–184. [Google Scholar]
- 4.Florig H K. Environ Sci Technol News. 1997;31:274A–279A. doi: 10.1021/es972315t. [DOI] [PubMed] [Google Scholar]
- 5.Coleman L L. International Coal: 1997 Edition. Washington, DC: The National Mining Association; 1998. [Google Scholar]
- 6.Zheng B, Yu X, Zhand J, Zhou D. 30th Intl Geologic Congress Abstr. 1996;3:410. [Google Scholar]
- 7.U.S. Environmental Protection Agency. Water Quality Criteria 1972. Washington, DC: Government Printing Office; 1973. , EPA-R3–73033. [Google Scholar]
- 8.Belkin H E, Zheng B, Finkelman R B. Fourth International Symposium on Environmental Geochemistry. Reston, VA: U.S. Geological Survey; 1997. , U.S. Geological Survey Open-File Report 97–496, p. 10. [Google Scholar]
- 9.Belkin H E, Zheng B, Zhou D, Finkelman R B. Fourteenth Annual International Pittsburgh Coal Conference, CD-ROM. University of Pittsburgh; 1997. [Google Scholar]
- 10.Belkin H E, Warwick P, Zheng B, Zhou D, Finkelman R B. Fifteenth Annual International Pittsburgh Coal Conference, CD-ROM. University of Pittsburgh; 1998. [Google Scholar]
- 11.Bragg L J, Oman J K, Tewalt S J, Oman C L, Rega N H, Washington P M, Finkelman R B. U. S. Geological Survey Coal Quality (coalqual), Version 2.0. Reston, VA: U.S. Geological Survey; 1997. , U. S. Geological Survey Open-File Report 97–134 (CD-ROM). [Google Scholar]
- 12.Huffman G P, Huggins F E, Shah N, Zhao J. Fuel Processing Technol. 1994;39:47–62. [Google Scholar]
- 13.Ashley R P, Cunningham C G, Bostick N H, Dean W E, Chou I-M. Ore Geol Rev. 1991;6:131–151. [Google Scholar]
- 14.Qian H, Chen W, Hu Y. Geol J Universities. 1995;1:45–52. (in Chinese; English abstract). [Google Scholar]
- 15.Chen D, Wang H, Ren D. Acta Mineralogia Sinica. 1996;16:307–314. (in Chinese; English abstract). [Google Scholar]
- 16.He M, Wu X. Chinese J Geochem. 1997;16:75–79. [Google Scholar]
- 17.Li Z, Peters S G. Geological Society of America Program for 1996 Annual Meeting. Boulder, CO: Geological Society of America; 1996. p. A153. (abstr.). [Google Scholar]
- 18.Zheng B, Huang R. In: Developments in Geoscience, Contributions to 28th International Geologic Congress: 1989. Washington DC, editor. Beijing, China: Science Press; 1989. pp. 171–176. [Google Scholar]
- 19.Zhang Y, Cao S R. Fluoride. 1996;29:207–211. [Google Scholar]
- 20.Ando M, Tadano M, Asanuma S, Matsushima S, Wanatabe T, Kondo T, Sakuai S, Ji R, Liang C, Cao S. Environ Health Perspect. 1998;106:239–244. doi: 10.1289/ehp.98106239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.U.S. Environmental Protection Agency. Mercury Study Report to Congress: White Paper. Washington, DC: Government Printing Office; 1998. EPA-453/R-98-004B. [Google Scholar]
- 22.Zhou D, Liu D. J Environ Health. 1985;48:14–18. [Google Scholar]
- 23.Zheng B, Hong Y, Zhao W, Zhou H, Xia W, Su H, Mao D, Yan L, Thornton I. Chinese Sci Bull. 1992;37:1725–1729. [Google Scholar]
- 24.Crowley S S, Orem W H, Roth M J, Finkelman R B, Scroggs E A, Willett J. Fifteenth Annual Meeting of the Society for Organic Petrology, Abstracts and Program. Vol. 15. The Society for Organic Petrology; 1998. pp. 99–97. [Google Scholar]
- 25.Mumford J L, He X Z, Chapman R S, Cao S R, Harris D B, Li X M, Xian W Z, Jiang C W, Xu J C, Chuang J C, et al. China Sci. 1987;235:217–220. doi: 10.1126/science.3798109. [DOI] [PubMed] [Google Scholar]
- 26.Mumford J L, Li X, Hu F, Lu X B, Chaung J C. Carcinogenesis. 1995;16:3031–3036. doi: 10.1093/carcin/16.12.3031. [DOI] [PubMed] [Google Scholar]