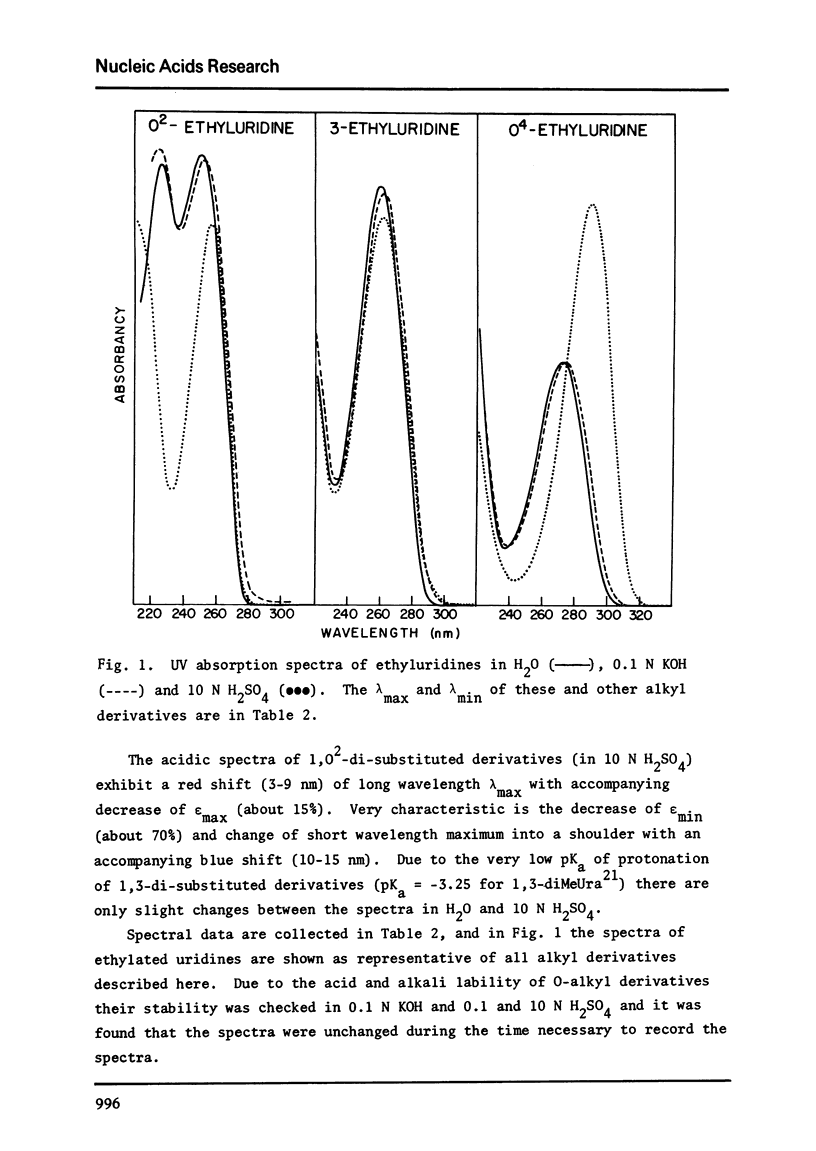
Abstract
In non-aqueous solution, diazomethane and diazoethane react with the O2, O4 and N-3 sites of uridine, thymidine, 1-methyluracil and 1-methylthymine. Diazoethane has a higher affinity for alkylating oxygens than does diazomethane. The relative ratio of O2:O4:N-3 methyl products is 1:2:16 and of ethyl products the ratio is 1:1:2. When the diazoethane reaction is performed in neutral buffered solution, the same proportion of O2:O4:N-3 ethyl products is found, but the extent of reaction is very low. O2-alkylation greatly labilizes the glycosidic bond of thymidine and uridine toward acid hydrolysis. All O2 and O4 alkyl 1-substituted 2,4-dioxopyrimidines are dealkylated in weak acid but the O2 alkyl group is the more stable.
Full text
PDF

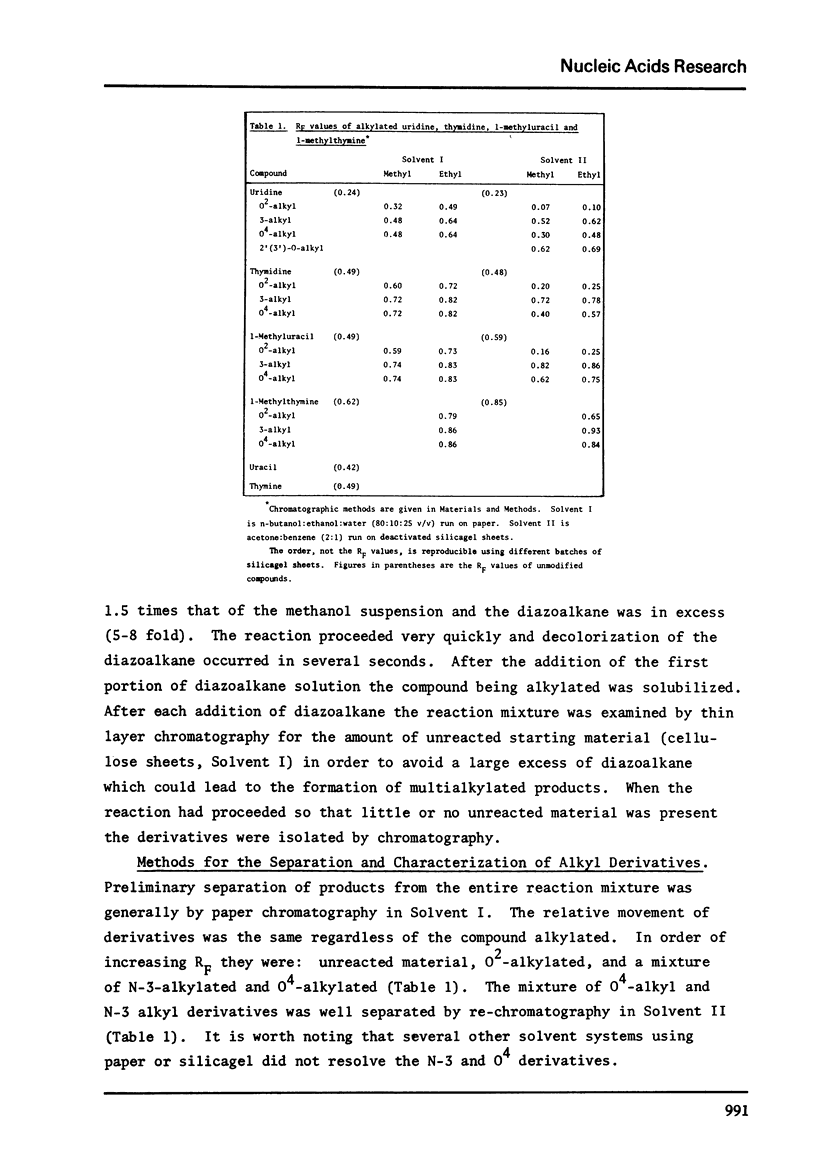
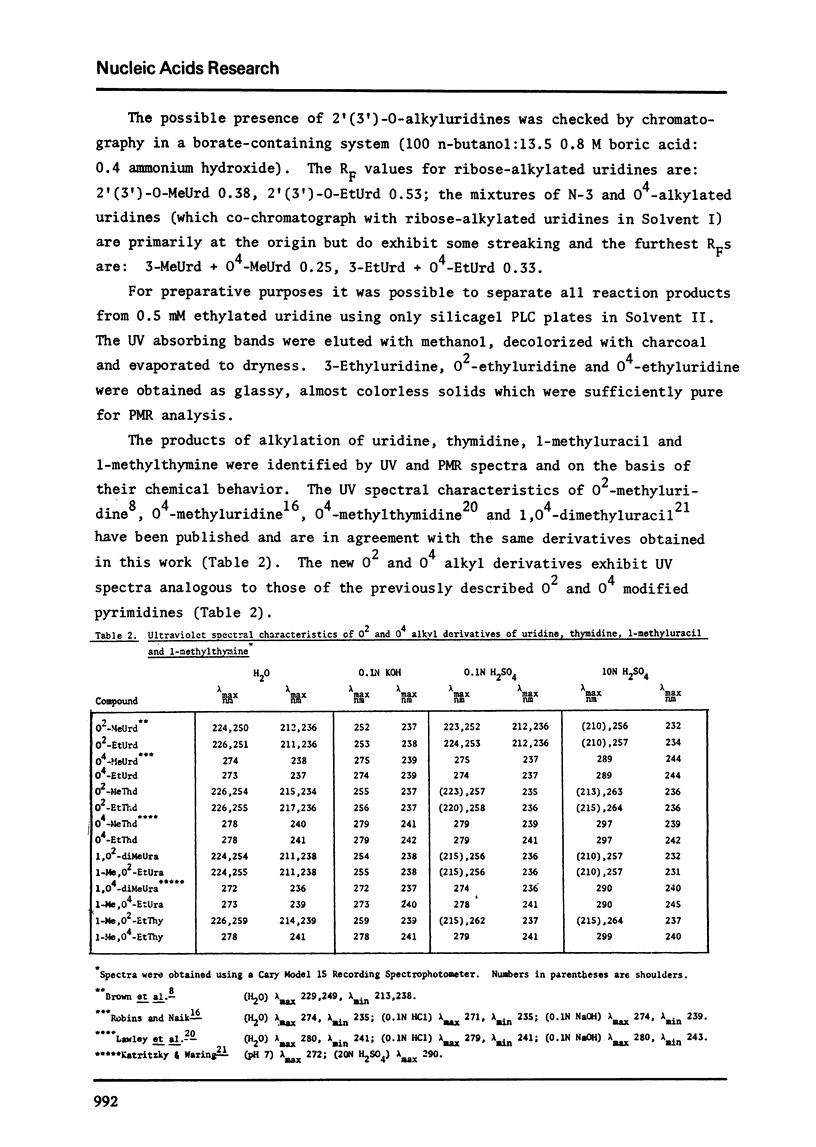
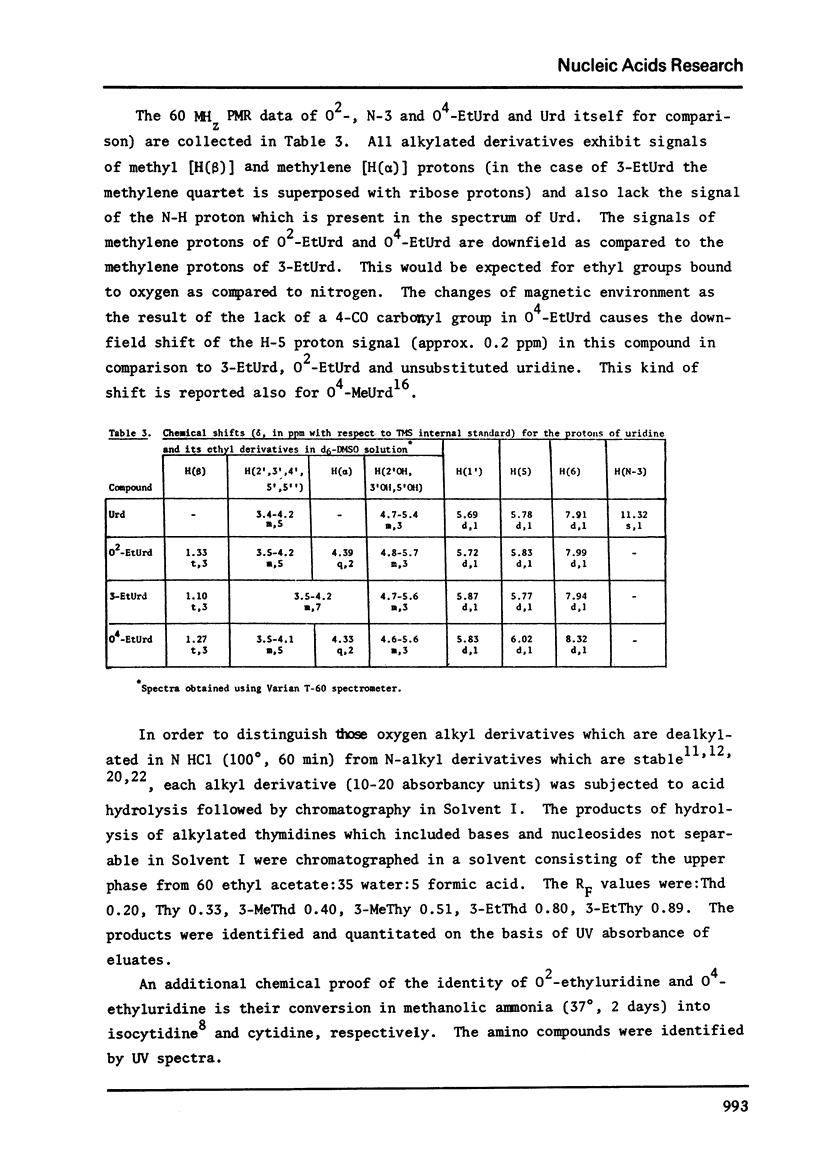
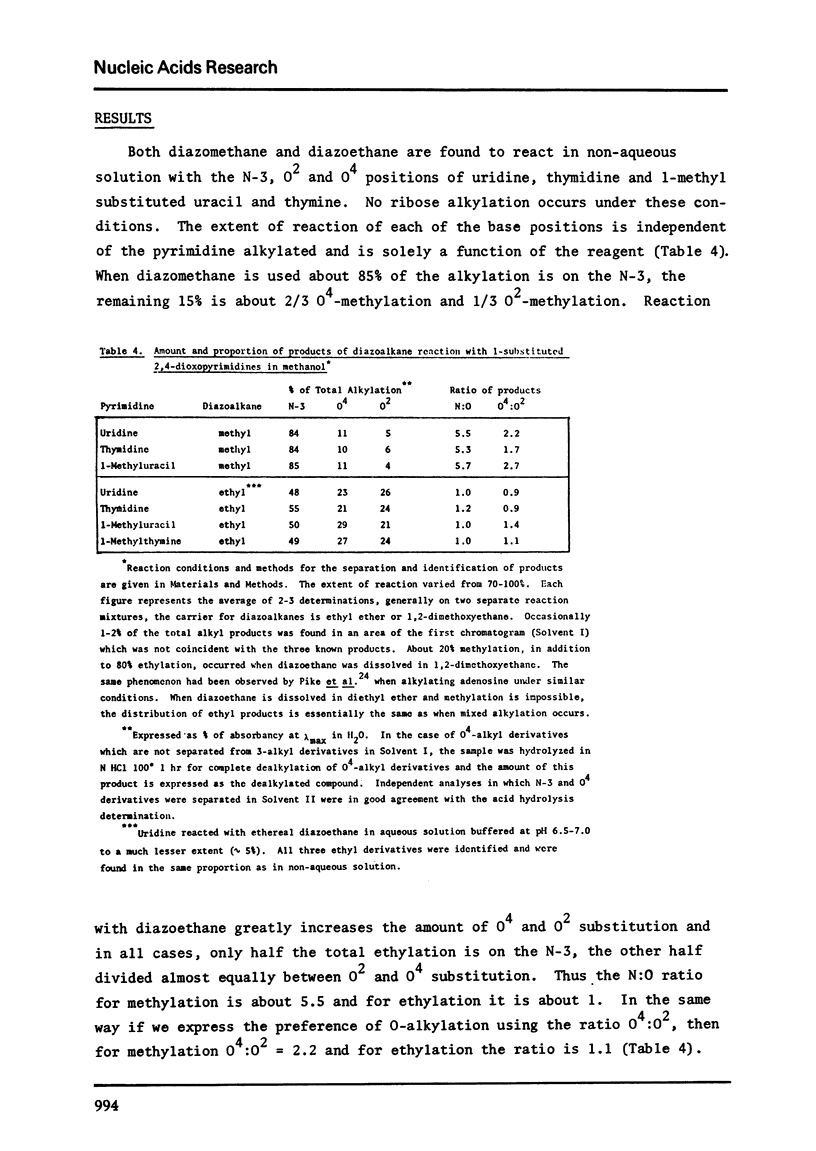





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- FOX J. J., SHUGAR D. Spectrophotometric studies of nucleic acid derivatives and related compounds as a function of pH. II. Natural and synthetic pyrimidine nucleosides. Biochim Biophys Acta. 1952 Oct;9(4):369–384. doi: 10.1016/0006-3002(52)90181-9. [DOI] [PubMed] [Google Scholar]
- Farmer P. B., Foster A. B., Jarman M., Tisdale M. J. The alkylation of 2'-deoxyguanosine and of thymidine with diazoalkanes. Some observations on o-alkylation. Biochem J. 1973 Sep;135(1):203–213. doi: 10.1042/bj1350203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman O. M., Mahapatra G. N., Dash B., Stevenson R. Studies on the action of diazomethane on deoxyribonucleic acid. The action of diazomethane on deoxyribonucleosides. Biochim Biophys Acta. 1965 Jun 8;103(2):286–297. doi: 10.1016/0005-2787(65)90168-1. [DOI] [PubMed] [Google Scholar]
- GOMPPER R. THE REACTIONS OF DIAZOMETHANE WITH HETEROCYCLIC COMPOUNDS. Adv Heterocycl Chem. 1963;10:245–286. doi: 10.1016/s0065-2725(08)60751-5. [DOI] [PubMed] [Google Scholar]
- Kuśmierek J. T., Giziewicz J., Shugar D. Preparation of O'-alkyl derivatives of cytosine and uracil nucleosides. Biochemistry. 1973 Jan 16;12(2):194–200. doi: 10.1021/bi00726a003. [DOI] [PubMed] [Google Scholar]
- Kuśmierek J. T., Shugar D. Alkylation of cytidine-5'-phosphate: mechanisms of alkylation, influence of O'-alkylation on susceptibility of pyrimidine nucleotides to some nucleolytic enzymes, and synthesis of 2'-O-alkyl polynucleotides. Acta Biochim Pol. 1973 Aug;20(4):365–381. [PubMed] [Google Scholar]
- Lawley P. D., Orr D. J., Shah S. A., Farmer P. B., Jarman M. Reaction products from N-methyl-N-nitrosourea and deoxyribonucleic acid containing thymidine residues. Synthesis and identification of a new methylation product, O4-methylthymidine. Biochem J. 1973 Sep;135(1):193–201. doi: 10.1042/bj1350193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MILES H. T. Infrared spectra and tautomeric structure in D20 solution of some pyrimidine nucleosides and nucleotides. Biochim Biophys Acta. 1956 Nov;22(2):247–253. doi: 10.1016/0006-3002(56)90147-0. [DOI] [PubMed] [Google Scholar]
- Pike L. M., Khan M. K., Rottman F. Mixed alkylation (methylation and ethylation) of adenosine by diazoethane in aqueous 1,2-dimethoxyethane. J Org Chem. 1974 Dec 13;39(25):3674–3676. doi: 10.1021/jo00939a013. [DOI] [PubMed] [Google Scholar]
- Robins M. J., Naik S. R., Lee A. S. Nucleic acid related compounds. 12. The facile and high-yield stannous chloride catalyzed monomethylation of the cis-glycol system of nucleosides by diazomethane. J Org Chem. 1974 Jun 28;39(13):1891–1899. doi: 10.1021/jo00927a022. [DOI] [PubMed] [Google Scholar]
- Robins M. J., Naik S. R. Nucleic acid related compounds. 1. Methylation and transformation of 4-methoxy-2-pyrimidinone 1- -D-ribofuranoside into 2'-O-methyl nucleoside components of ribonucleic acid, their analogs, and derivatives. Biochemistry. 1971 Sep 14;10(19):3591–3597. doi: 10.1021/bi00795a017. [DOI] [PubMed] [Google Scholar]
- SCANNELL J. P., CRESTFIELD A. M., ALLEN F. W. Methylation studies on various uracil derivatives and on an isomer of uridine isolated from ribonucleic acids. Biochim Biophys Acta. 1959 Apr;32:406–412. doi: 10.1016/0006-3002(59)90613-4. [DOI] [PubMed] [Google Scholar]
- Singer B. Methylation and ethylation of uridylic acid and thymidylic acid. Reactivity of the ring and phosphate as a function of pH and alkyl group. Biochemistry. 1975 Oct 7;14(20):4353–4357. doi: 10.1021/bi00691a001. [DOI] [PubMed] [Google Scholar]
- Singer B. Reaction of guanosine with ethylating agents. Biochemistry. 1972 Oct 10;11(21):3939–3947. doi: 10.1021/bi00771a017. [DOI] [PubMed] [Google Scholar]
- Singer B., Sun L., Fraenkel-Conrat H. Reaction of adenosine with ethylating agents. Biochemistry. 1974 Apr 23;13(9):1913–1920. doi: 10.1021/bi00706a021. [DOI] [PubMed] [Google Scholar]
- Sun L., Singer B. Reaction of cytidine with ethylating agents. Biochemistry. 1974 Apr 23;13(9):1905–1913. doi: 10.1021/bi00706a020. [DOI] [PubMed] [Google Scholar]
- Sun L., Singer B. The specificity of different classes of ethylating agents toward various sites of HeLa cell DNA in vitro and in vivo. Biochemistry. 1975 Apr 22;14(8):1795–1802. doi: 10.1021/bi00679a036. [DOI] [PubMed] [Google Scholar]
- TSinger B., Fraenkel-Conrat H. The specificity of different classes of ethylating agents toward various sites in RNA. Biochemistry. 1975 Feb 25;14(4):772–782. doi: 10.1021/bi00675a020. [DOI] [PubMed] [Google Scholar]
- Wierzchowski K. L., Litońska E., Shugar D. Infrared and ultraviolet studies on the tautomeric equilibria in aqueous medium between monoanionic species of uracil, thymine, 5-fluorouracil, and other 2,4-diketopyrimidines. J Am Chem Soc. 1965 Oct 20;87(20):4621–4629. doi: 10.1021/ja00948a039. [DOI] [PubMed] [Google Scholar]



