Abstract
We developed a chondroitin sulfate - polyethylene glycol (CS-PEG) adhesive hydrogel with numerous potential biomedical applications. The carboxyl groups on chondroitin sulfate (CS) chains were functionalized with N-hydroxysuccinimide (NHS) to yield chondroitin sulfate succinimidyl succinate (CS-NHS). Following purification, the CS-NHS molecule can react with primary amines to form amide bonds. Hence, using six arm polyethylene glycol amine PEG-(NH2)6 as a crosslinker we formed a hydrogel which was covalently bound to proteins in tissue via amide bonds. By varying the initial pH of the precursor solutions, the hydrogel stiffness, swelling properties, and kinetics of gelation could be controlled. The sealing/adhesive strength could also be modified by varying the damping and storage modulus properties of the material. The adhesive strength of the material with cartilage tissue was shown to be ten times higher than that of fibrin glue. Cells encapsulated or in direct contact with the material remained viable and metabolically active. Furthermore, CS-PEG material produced minimal inflammatory response when implanted subcutaneously in a rat model and enzymatic degradation was demonstrated in vitro. This work establishes an adhesive hydrogel derived from biological and synthetic components with potential application in wound healing and regenerative medicine.
1. Introduction
In many areas of medicine including tissue engineering, surgery, regenerative medicine, and drug delivery, there exists a need for materials which are both adhesive and in situ forming. These materials can be composed of synthetic, biological or a combination of such materials. Synthetic biomaterials allow for distinct control of the scaffold’s mechanical properties, but are usually not bioactive. Biological molecules can provide cues to stimulate cells to proliferate, migrate, differentiate and produce extracellular matrix (ECM). However, controlling batch-to-batch variability is a difficult challenge when working with biological molecules. Therefore, developing a biomaterial with both biological and synthetic components with tissue adhesive properties is of significant interest.
The glycosaminoglycan (GAG) chondroitin sulfate is a biological polymer that is abundant in the extracellular matrix (ECM) of tissues throughout the body. It serves both important structural and biological functions. Different classes of chondroitin sulfate molecules can elicit different and sometimes even opposite cell responses. For example, chondroitin sulfate-A (CS-A) inhibits axonal growth while chondroitin sulfate-E (CS-E) has the opposite effect [1,2]. Other such biological functions regulated in part by CS include inflammation [3,4], coagulation [5,6], stem cell niche creation [7], enzymatic activity [3,8], complement activity [9], and apoptosis [4]. Strategically incorporating the appropriate chondroitin sulfate molecule into an adhesive for specific applications could therefore improve the tissue regeneration properties of that biomaterial.
Polyethyelene glycol (PEG) is a synthetic polymer used extensively in scaffolds for tissue engineering applications such as articular cartilage [10-14], neural [15-17], and bladder [18] tissue regeneration. PEG is essentially non immunogenic [19], antifouling [20-22], and non toxic. Thus, it can serve as an inert synthetic molecule to control scaffold mechanical properties.
In this publication, we characterize the mechanical, swelling, adhesive, biodegradation and anti-inflamatory properties of a CS-PEG hydrogel material which covalently adheres to tissue through amide bonds.
2. Materials and Methods
2.1. Synthesis of Chondroitin Sulfate Succinimidyl Succinate (CS-NHS)
The synthesis of this material has been described previously [23]. Briefly, using PBS as a buffer, 10% CS (w/v) (25 kDa, New Zealand Pharmaceuticals Ltd, Palmerston North, New Zealand), 67% EDC (w/v) and 25% NHS (w/v) were combined in a (7:1.5:1.5) (v/v) ratio CS:EDC:NHS and allowed to react for 10 minutes at 37 °C. The product was frozen and then precipitated using -20 °C EtOH. CS-NHS was washed with -20 °C EtOH nine times and then dried under high vacuum overnight. The humidity in the room was controlled using the building’s environmental control system and was measured using a hydrometer (00592W Wireless Thermo-Hydrometer, Chaney Instruments Co., Lake Geneva, Wisconsin).
2.2. NHS Quantification
The hydroxamate assay was used to quantify the total NHS groups present in the CS-NHS compound [24-26]. Each sample (1 mL, 0.1-0.3 mg/mL CS-NHS) was mixed with 0.2 mL 2N NaOH. Following incubation at 60°C for 10 minutes, the solutions were placed on ice for 1 minute and then mixed with 1.5 mL of 0.85 N HCl. Then 1mL of each sample was combined with 250 μL 5% FeCl3 in 0.1 N HCl and the absorbance was measured at 500 nm (solutions of 0-0.5 mM NHS were used as standards). Unbound NHS was quantified by washing the CS-NHS powder with EtOH and that EtOH solution was diluted in ddH2O and used as a sample.
2.3. Preparation of CS-PEG Hydrogels
Hydrogels with 1:1 ratio of CS-NHS to PEG-(NH2)6 were prepared by mixing 10% CS-NHS in PBS with 10% PEG-(NH2)6 (15 kDa, Sunbio, Orinda, CA), which had varying pH, and allowing components to react for 10 minutes at 37 °C yielding 10% 1:1 CS-PEG gels. Unless otherwise stated, the initial pH of the PEG-(NH2)6 solution is assumed to be 7.3. For the 1:2 10% CS-PEG gels, 6.7% CS-NHS in PBS and 13.3% PEG-(NH2)6 with pH of 7.3 were used. For the 2:1 10% CS-PEG gels, 13.3% CS-NHS in PBS and 6.7% PEG-(NH2)6 with pH of 7.3 were used. The cellular gels used in the cytotoxicity studies, were prepared as described above, with the only exception being that the PEG-(NH2)6 contained 40 × 106 cells/ml. After mixing the two components, the gels contained 20 × 106 cells/ml.
2.4. Measurement of Uniaxial Compressive Moduli
The gel moduli were measured using an Electroforce 3200 testing instrument (Bose, Eden Prairie, MN). Data for calculating the compression modulus was collected by compressing cylindrical gels from 0 to 10% strain at 0.44% per second. The modulus was calculated by fitting a best fit curve in the linear region of the stress vs strain plot (4 to 9% strain). Data for the storage modulus were acquired by first compressing the gels to 6% strain and then recording the stress as strain was oscillated between 5 and 7% at 1Hz. The storage modulus was calculated as described previously [27].
2.5. Swelling Ratio Analysis
The gels were incubated in PBS or water and their wet weight was measured at different times. After swelling equilibrium was reached, the gels were lyophilized, and the dry weight was measured.
2.6. Measuring Gelation Times of CS-PEG Hydrogels
Using a 2-200 μL pipetman, the CS-NHS and PEG-(NH2)6 components were mixed and pipetted up and down until the solutions could no longer be pipetted. The time at which this happened was designated as the gelation time.
2.7. Chondrocyte Isolation from Articular Cartilage
Bovine articular cartilage was collected from the femoral condyles and the patellofermoral groove of a calf knee (Research 87, Inc. Boylston, MA) and stored in high glucose DMEM substituted with 100 U/mL penicillin and 10 μg/mL streptomycin (pen/strep). The chondrocytes were isolated as described previously [28]. The cartilage was minced using a scalpel and forceps, and the minced tissue was incubated in collagenase solution for 16 hours at 37 °C with 5% CO2. The collagenase solution was composed of high glucose DMEM substituted with 6% (v/v) FBS, 350 U/ml collagenase type II (Worthington Biochemical Corporation, Lakewood, NJ) and pen/strep. The cells were isolated from any undigested ECM using a 70 μm cell strainer.
2.8. Cell Culture and Viability Analysis
Following cell encapsulation, the hydrogel constructs were cultured for varying time points in medium containing high glucose DMEM, 10% fetal bovine serum FBS, 10 mM HEPES, 0.4 mM L-proline, 50 μg/mL ascorbic acid, 0.1 mM non-essential amino acids, 100 U/mL penicillin and 10 μg/mL streptomycin. Viability analysis of the encapsulated cells was done using the manufacturer’s guidelines for the LIVE/DEAD Viability/Cytotoxicity Kit *for mammalian cells (Invitrogen, Carlsbad, CA). Briefly, thin (< 300 μm) sections were taken from each gel and were incubated in live/dead medium for 30 minutes. Live/dead medium contained DMEM, 4 μM calcein AM, and 4 μM ethidium homodimer-1. The gel slices were washed with PBS 3 times and then analyzed using fluorescence microscopy with a 485 ± 10 nm optical filter for calcein AM (live cells) and a 530 ± 12.5 nm optical filter for ethidium homodimer-1 (dead cells). The live and dead cell images were merged using Adobe Photoshop software.
WST-1 reagent (Roche Applied Science, Indianapolis, IN) was also used to assess viability of chondrocytes as described by the manufacturer. Briefly, chondrocytes were grown to confluence after which hydrogels were placed on top to cover the cells. Following 24 hours of incubation at 37°C and 5% CO2, the gels were removed and the wells were washed 6 times with PBS and 3 times with DMEM. To each well 100 μL of 10% WST-1 reagent in media was added. Following incubation at 37°C and 5% CO2 for 3 hours the absorbance at 440nm was measured in each well. Cells in control wells were cultured without hydrogels.
2.9. Burst Pressure and Adhesion Experiments
A modification of ASTM standard F2392-04 was used to characterize the sealing properties of the adhesive. Briefly, incisions (4 mm) were made in collagen membranes (Vista International Packaging, Kenosha, WI) and sealed using 200 μL of 10% 1:1 CS-PEG hydrogel. Hyaluronan (HA) (Lifecore Biomedical, Chaska, MN) of different concentration and molecular weight was doped in the material as well. After incubating for 15 minutes at 37°C, the sealed membranes were placed in PBS solution overnight. The burst pressures were measured by clamping down the membranes and applying force with PBS at 1 mL per minute on one side of the membrane. The pressure at which the incision burst and the mode of failure (adhesive vs. cohesive) were recorded.
Adhesion was quantified using a modification of ASTM standard F2255-05. Bovine articular cartilage tissue was extracted as described above and was cut into thin sections such that each section included superficial, mid and deep zone cartilage. Sections were glued together using either 20 μL 10% 1:1 CS-PEG or fibrin glue and following 15 minutes of gelation, the sections were transferred to PBS solution. Prior to measuring the adhesive strength, the glued tissue was mounted on aluminum fixtures using a cyanoacrylate glue. The tissue was pulled apart using an Electroforce 3200 testing instrument (Bose, Eden Prairie, MN) and stress and strain were recorded.
2.10. Enzymatic Degradation of CS-PEG Hydrogels
The hydrogels (100 μL 10% 1:1 CS-PEG) were prepared by mixing 50 μL 10% CS-NHS in PBS with 50 μL 10% PEG-(NH2)6 and allowing the two solutions to react for 10 minutes at 37 °C. Each gel was incubated 3 times for 2 hours in fresh enzyme buffer without enzyme. The enzyme buffer was prepared based on the manufacturer’s specifications; 50 mM Tris, 60 mM sodium acetate, 0.02% BSA with a pH of 8.0. Following incubation, the gels were split into two groups. One group was incubated in enzyme buffer without chondroitinase ABC (Sigma, St. Louis, MO) while the other contained the enzyme (0.1 units per gel). At different time points, gels from each group (n=3) were incubated in 1 mM Zn2+, a chondroitinase ABC inhibitor, followed by 3 washes with ddH2O for 30 minutes each. After lyophilization, the dry weights were measured.
2.11. In Vivo Studies and Histology
All procedures were performed with prior approval from the Johns Hopkins Animal Care and Use Committee. Eight-week-old male Sprague-Dawley rats were subcutaneously injected dorsally with 200 μL of either 10% 1:1 CS-PEG or 10% (w/v) PEG-DA (Sunbio, Orinda, CA) solutions using a 20 G and 25 G needle, respectively. The PEG-DA solution was then polymerized trans-cutaneously using an initiator system described previously [29] and a 530 nm LED (USA Photonics Inc., Nyack, NY) with a 120 second light exposure at the maximum power setting. Each animal received 3 injections of each polymer and one animal was sacrificed per time point. Following euthanasia, the implants were extracted and fixed in 10% formalin overnight. A series of EtOH solutions and xylene were used to dehydrate and clear the samples, respectively. After embedding overnight in paraffin, sections were cut to 5 μm thickness, mounted on a microscope slide and allowed to dry for an hour on a 40°C plate. Following rehydration, the sections were stained using hematoxylin and eosin (H&E).
2.12 Statistical Analysis
One-way ANOVA was used to detect significant effects among groups. Tukey’s multiple comparison tests were used to detect significant differences between groups, and a p-value ≤ 0.5 was considered significant.
3. Results
3.1. Reaction Mechanism of Gelation and Tissue Adhesion
We synthesized a CS-PEG tissue adhesive which forms a three dimensional hydrogel network. For example, when CS-NHS is mixed with PEG-(NH2)6, the two molecules will react to form a hydrogel (Fig. 1A-D). CS-NHS will also react with tissue, serving as an adhesive between the hydrogel and surrounding tissue (Fig. 1E). The reaction occurs between the NHS activated carboxyl group of CS-NHS (Fig. 1F) and the primary amines found both in PEG-(NH2)6 (Fig. 1G,H) and in proteins of tissue.
Figure 1.
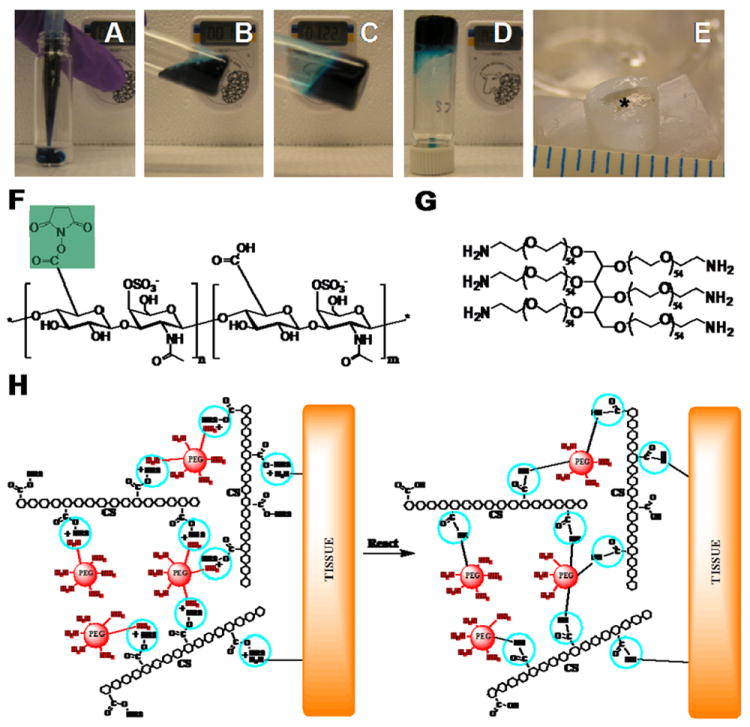
CS-NHS reacts with primary amines of both PEG-(NH2)6 and proteins of tissue to form a covalently bound hydrogel to tissue. (A-D) Solutions of PEG-(NH2)6 (colored with blue dye) and CS-NHS (clear solution) are mixed to form a hydrogel. (E) The same hydrogel without dye (star) in a cartilage defect after 11 days of swelling in PBS. The NHS activated carboxyl groups of CS-NHS (F) react with the primary amines of PEG-(NH2)6 (G) and the primary amines of proteins in tissue (H, reactive groups circled). The new bonds that form are amide bonds. (H) The material in contact with tissue before (left) and after (right) it has reacted with primary amines. NHS = N-Hydroxysuccinimide; PEG = Poly(ethylene glycol); CS = Chondroitin Sulfate.
As determined by the hydroxamate method [24-26], 23.8 ± 0.5 % of the carboxyl groups of chondroitin sulfate were activated with NHS using EDC/NHS chemistry as outlined in Scheme 1. NHS activated carboxyl groups will react with primary amines to form amide bonds (Fig. 1H). Since each molecule of chondroitin sulfate has approximately 11 activated carboxyl groups, it can react with several primary amines at a time such that if a diamine or higher order amine is used, a network can be formed that is also covalently bound to tissue.
Scheme 1.
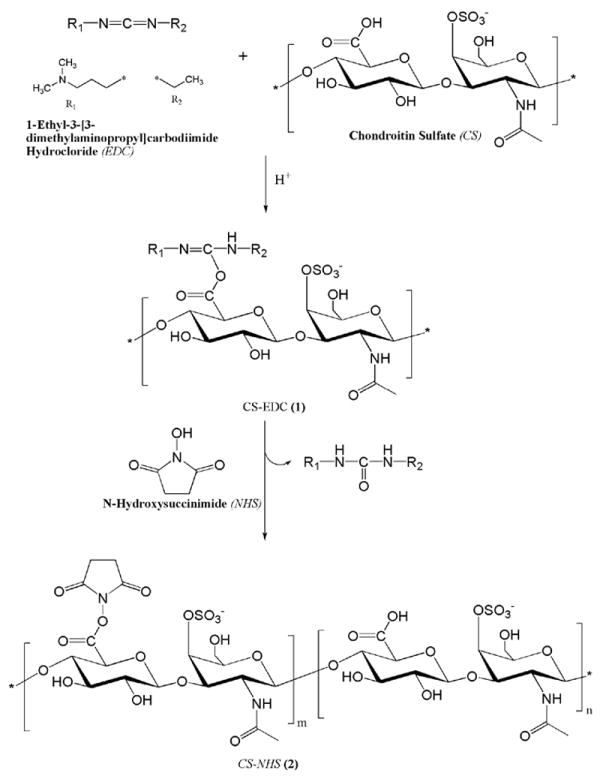
Synthesis of CS-NHS.
3.2. Effect of Humidity During Synthesis on Gel Properties
NHS activated carboxyl groups are very sensitive to hydrolysis in the presence of moisture which effects the stability of the adhesive components and ultimately their performance. In addition, ethanol (EtOH) is very hydroscopic. Decreased moisture lowers hydrolysis of the NHS activated carboxyl groups. This leads to more activated carboxyl groups per CS molecule and thus higher crosslinking densities and tangent moduli. This becomes particularly relevant during the -20°C EtOH washing steps during synthesis. Our data has shown that if humidity is decreased from 60 to 20% during this step, the tangent moduli of the gels is significantly increased (Fig. 2A).
Figure 2.
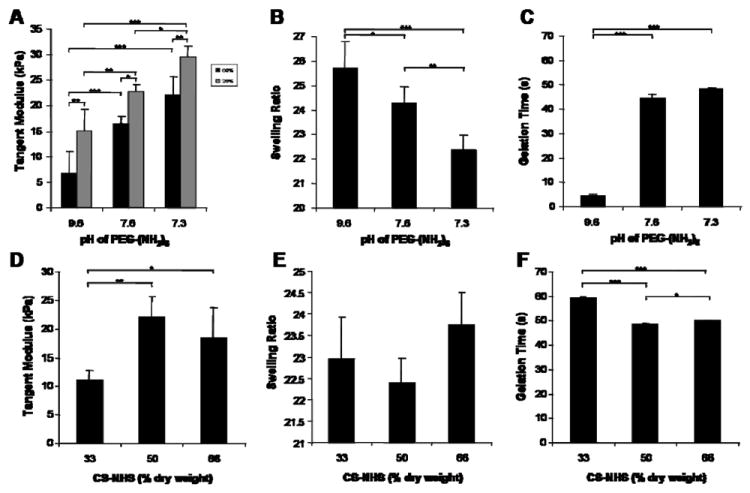
Effects of PEG-(NH2)6 solution pH, polymer ratios and synthesis conditions on mechanical, swelling and gelation properties of 10% (w/v) CS-PEG hydrogels. For panels A-C, 50% (% dry weight) CS-NHS and 50% (% dry weight) PEG-(NH2)6 were used. (A) The tangent modulus increases as pH decreases. Also, if humidity is decreased during the EtOH washing steps of the synthesis, the modulus consistently increases. (B) The swelling ratios as a function of pH show the opposite trend than that of the moduli. (C) As pH decreases, the time it takes the components to form a hydrogel increases. (D-F) when the concentration of CS to PEG is varied, there is a significant decrease in modulus at lower concentrations of CS-NHS, but it has little effect on swelling ratio and gelation time. Values are reported in mean ± SD with a sample size of 5 per treatment for tangent moduli and swelling ratio experiments, and a sample size of 3 for gelation time experiments. *p<0.05, **p<0.01, ***p<0.001.
3.3. Effect of pH on Gel Properties
The stiffness, swelling, and gelation properties of the CS-PEG hydrogel can be controlled by varying the initial pH of the PEG-(NH2)6 solution. After mixing the two components, the primary amines in the PEG-(NH2)6 react to form amide bonds, thus lowering the concentration of primary amines in solution and dropping the pH back down to a physiologically relevant pH of ~7.4 within the time it takes for the gel to form (5-49 seconds at 37°C). As initial pH increases the unconfined compression modulus and gelation time decrease while swelling ratio increases (Fig. 2A-C).
3.4. Effect of Polymer Ratios on Gel Properties
The composition of the biological and synthetic component can be manipulated without significantly affecting material properties. A ratio of 1:1 yielded the most crosslinked gels as evidenced by the higher modulus, and the gel with lower CS-NHS content had a significantly lower modulus (Fig. 2D). Despite the drop in modulus for the 1:2 CS-PEG gels, the equilibrium swelling ratio did not significantly change and the reaction kinetics were not greatly affected as the ratio between the two polymers was varied (Fig. 2E-F).
3.5. Viability Studies
To assess how reaction conditions or various concentrations of HEPES buffer may affect cell viability, chondrocytes were encapsulated in CS-PEG hydrogels and stained using the Live/Dead Viability/Cytotoxicity following one day of encapsulation. The majority of cells fluoresced green indicating viability as demonstrated in Fig. 3A signifying that the gel is not toxic to chondrocytes.
Figure 3.
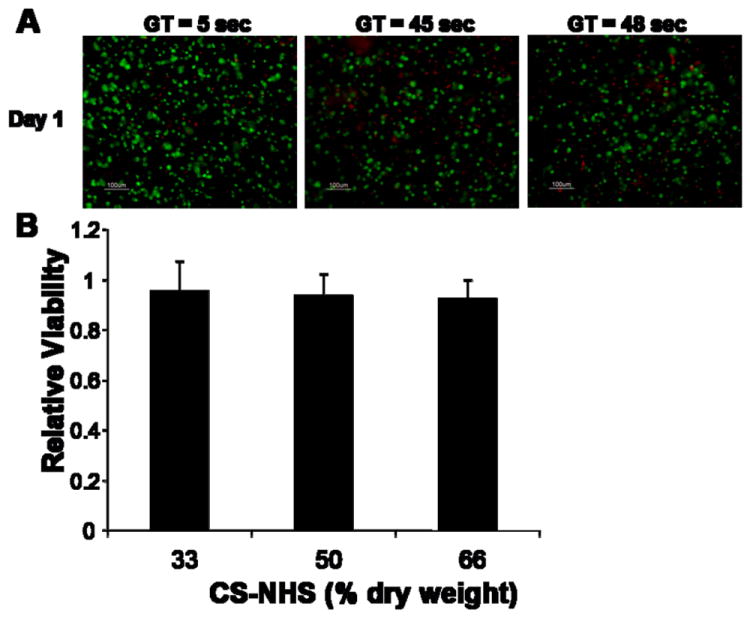
Effect of CS-PEG hydrogels with varying gelation times (GT) and polymer ratios on cytotoxicity to chondrocytes. (A) Live/Dead analysis of chondrocytes following one day of encapsulation in hydrogels with varying gelation times. For all gelation times, most of the cells are alive (green) with very few cells dead (red). (B) Chondrocytes were exposed to CS-PEG hydrogels with varying CS-NHS content and viability was quantified using the WST-1 reagent. Values are normalized to controls (monolayer of cells without exposure to hydrogel) and are reported in mean ± SD for a sample size of 4 per treatment. No significant differences was observed between the treatments and the control.
To further assess cytotoxicity, the CS-PEG scaffold was incubated with a monolayer of chondrocytes for 24 hours and WST-1 reagent was used to quantify viability (Fig. 3B). WST-1 reagent is cleaved by mitochondrial enzymes to form formazan which absorbs strongly at 440 nm. Therefore, the higher the absorbance at 440 nm, the more metabolically active, and therefore, viable the cells are. As the CS-NHS component of the scaffold is varied from 33 to 66% of dry weight, there is no effect on cell viability, and no statistically significant difference from control, where cells are not exposed to gel. Similar results for encapsulation and incubation with the material were found in using mesenchymal stem cells (MSCs), intervertebral disc cells including nucleus pulposus and annulus fibrosus cells, corneal cells including endothelial, stromal and epithelial cells [23] and foreskin fibroblasts (data not shown).
3.6. Effects of Temperature on Gelation Time
To better characterize setting times of the gel, temperature was investigated as a potential means of controlling gelation time. We found that the two components form a hydrogel more quickly as temperature increases (Fig. 4A).
Figure 4.
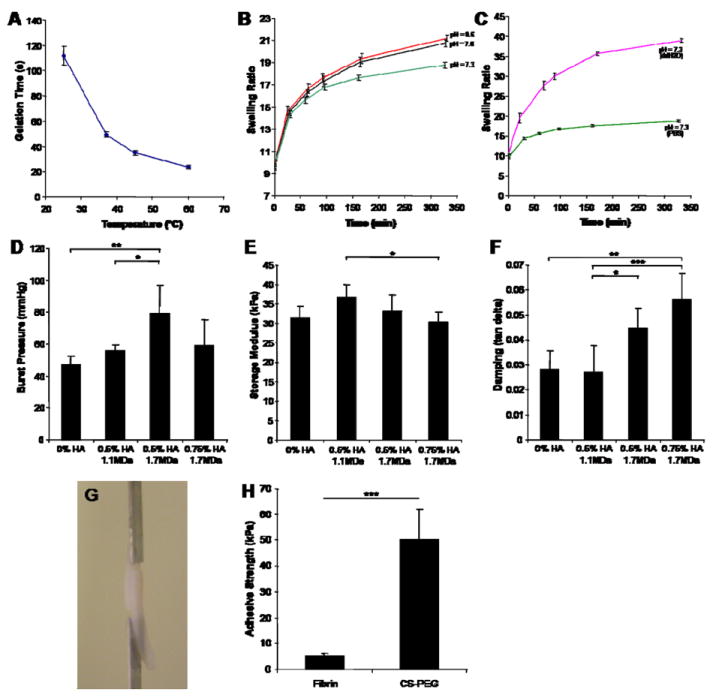
Swelling, kinetics of gelation, sealing and adhesive properties of the CS-PEG hydrogel adhesive. (A) There is an inverse relationship between gelation time and temperature. (B,C) Varying the crosslinking density and the osmolarity of the swelling medium has little effect on when swelling equilibrium is reached. In all cases, swelling equilibrium is achieved within five and a half hours. (C) When ddH2O is used instead of PBS, swelling equilibrium is again reached within five and a half hours even though the gels swell twice as much. (D-F) Effects of HA thickener on the burst pressure, storage modulus and damping properties of the hydrogels. (G) The glued cartilage pieces were pulled apart using a Bose ELF 3200 Mechanical Tester. (H) Adhesive strengths of CS-PEG and fibrin glue were significantly different and were an order of magnitude apart. Values are reported in mean ± SD with a sample size of 5 per treatment except for gelation time experiments which had a sample size of 3. *p<0.05, **p<0.01, ***p<0.001.
3.7. Swelling Properties
Gels with the same CS to PEG ratio reached swelling equilibrium at the same time independent of crosslinking density or the osmolarity of the swelling medium. For example, the hydrogels with varying initial pH of the PEG-(NH2)6 solution all reached swelling equilibrium within five and a half hours (Fig 4B). Swelling equilibrium was defined as the swelling ratio at which the wet weight of the hydrogel was no longer significantly changing. In addition, when the swelling medium was changed from PBS to ddH2O, the gels swelled almost twice as much at equilibrium (Fig. 4C). However, the time it took to reach swelling equilibrium was again five and a half hours. The in vivo swelling is expected to resemble the swelling ratio in PBS, as this more closely resembles the osmolarity of physiologic conditions. Additionally, when filling a defect, the gel will swell under confined conditions in which case swelling is controlled by the stiffness of the surrounding tissue. For example, in an articular cartilage defect, very little swelling was observed (Fig. 1E).
3.8. Adhesive and Sealing properties of CS-PEG
In an earlier publication [23], we evaluate the sealing properties of one formulation of the material using corneal tissue. Here we further characterize the material’s sealing properties by varying stiffness and damping using hyaluronan. The molecular weight and concentration of the HA molecule being doped in the hydrogel were chosen in order to change the material’s stiffness and damping. This led to a change in the hydrogel’s sealing properties. Also, HA with molecular weight in the MDa range was used because it is physiologically relevant [30], and it is retained within the hydrogel for an extended period of time. Doping in 0.5% (w/v) 1.1 MDa HA to the CS-PEG adhesive increased the burst pressure and the storage modulus while maintaining a constant damping (measured by tan δ) (Fig. 4D-F). The increase in burst pressure could therefore be attributed to the increase in storage modulus. When the molecular weight of HA was increased to 1.7 MDa while maintaining concentration at 0.5% (w/v) HA, there was a significant increase in burst pressure and damping while there was a decrease in storage modulus when compared to the 0.5% (w/v) 1.1 MDa HA. Therefore, the increase in burst pressure could be attributed to the increase in damping of the material. The effect of damping can again be seen when we compare CS-PEG gels to CS-PEG gels doped with 0.75% (w/v) 1.7 MDa HA. The burst pressure increases for the gels with HA even though the storage moduli are the same, due to the increase in damping. Therefore, storage modulus and damping of the material directly affect burst pressure outcomes.
The adhesive strength of CS-PEG was compared to fibrin glue in lap-shear tension using a modified protocol of ASTM standard F2255-05 (Fig. 4G). Bovine cartilage tissue was used as a substrate. CS-PEG showed an order of magnitude increase in adhesive strength when compared to fibrin glue (Fig. 4H) thus demonstrating the superior adhesive properties of the material.
3.9. Biological Activity
CS can influence biological responses of cells, which can be useful for guiding tissue repair and regenerative medicine applications. However, chemically modifying CS could compromise its biological activity. For example, following acrylate functionalization of 14% of the hydroxyl groups found on HA, the gels formed would not degrade with hyaluronidase (HAse) treatment [31]. To test whether significant change to the chemical structure of CS occurred, CS-PEG hydrogels were exposed to chondroitinase ABC and the dry weight was recorded at several time points (Fig. 5A-C). The enzyme (0.1 U enzyme per 100 μL 10% (w/v) hydrogel construct) was able to degrade approximately 70% of the hydrogel within 30 hours. From this we can conclude that the modification that takes place has not rendered CS biologically unrecognizable.
Figure 5.
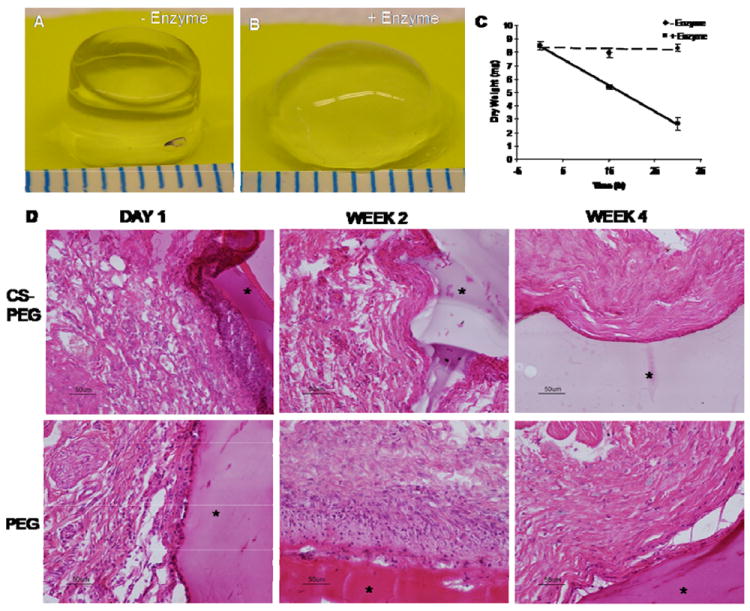
Biological activity of CS-PEG gels. Fifteen hours after incubation CS-PEG gels in buffer (A) without and (B) with chondroitinase ABC. (C) Within 30 hours, most of the gel had been degraded by the enzyme. (D) H&E staining of CS-PEG and PEG gels after subcutaneous injection in a rat model. In each section, the gels are marked with a star. CS-PEG = chondroitin sulfate-poly(ethylene glycol) gel. Values are reported in mean ± SD with a sample size of 3 per treatment.
To investigate in vivo biocompatibility, CS-PEG and PEG based scaffolds were injected subcutaneously into a rat animal model. The degree of inflammation elicited by the material was then evaluated histologically. Both scaffolds elicited a minor inflammatory response at 24 hours after injection (Fig. 5D). However, within two weeks and four weeks, the inflammatory response had resolved with the CS-PEG and PEG scaffold, respectively (Fig. 5D). The reduced inflammatory response seen with CS-PEG is consistent with the well-documented anti-inflammatory properties of CS. This is also further evidence that the chemical modification of CS did not significantly alter its biological functions.
4. Discussion
It has been demonstrated that the stiffness of biomaterials plays a key role in cell behavior such as adhesion, proliferation and gene expression [32,33]. Therefore having control over the mechanical properties of a biomaterial would be advantageous. In this work, we demonstrate that by varying the pH of the PEG-(NH2)6 solution prior to gelation, we can control the stiffness of the CS-PEG hydrogels. This is a consequence of two competing reactions with the NHS activated carboxyl groups of CS: hydrolysis and reaction with primary amines. Hydrolysis forms carboxyl groups and reaction with primary amines forms amide bonds. Normally, both reactions occur more often when the pH is higher, with amide formation increasing to a greater degree [34]. However, in this case, both polymers increase in molecular weight as the reaction progresses. This leads to slower diffusion rates of the polymers containing primary amines and NHS activated carboxyl groups and thus an increase in the time required for the two functional groups to react. This decrease in diffusion rate also increases the probability that hydrolysis will occur before the two reactive groups have a chance to meet. In effect, as the pH is decreased, the reaction kinetics are slowed down allowing more time for primary amines to diffuse to NHS activated carboxyl groups and react to form amide bonds. As a result, more amide bonds are formed (data not shown) leading to a more crosslinked, and therefore, stiffer, hydrogel as evidenced by the higher compression modulus (Fig. 2A). Thus, pH can be used to control the crosslinking density in this hydrogel.
For hydrogels of the same polymer composition, when the crosslinking density and modulus increase, the equilibrium swelling ratio is expected to decrease. As expected, the observed swelling ratio decreased as pH decreased (Fig. 2B), and is therefore consistent with the compression modulus data. However, equilibrium swelling did not correlate with the moduli of the gels when the CS-PEG ratio was varied. This can be explained by the variable amount of CS. Since CS is highly charged, it swells more than PEG, and therefore, the concentration of CS will affect the swelling of the gels. As a result, both CS concentration and crosslinking density influence the swelling ratio of the gels.
Time for gelation is an important parameter in a surgical setting. If the gelation time is too long, the material will become diluted with body fluids and gel properties will be compromised or the material will not remain in the desired implantation site. Alternatively, if the hydrogel forms too quickly, implantation becomes challenging and tissue adhesion is compromised. At a normal body core temperature of 37°C, the adhesive would set within 49 ± 2 sec, while at 25°C, the gelation time increased to 112 ± 8 sec (Fig. 4A). Following administration of anesthesia, body core temperature can decrease by 4°C below normal [35], which could increase time of gelation to a value between these two averages. Nevertheless, gelation time occurs in a time frame suitable for surgical applications.
In some instances, following surgery, it would be beneficial if the material is degraded and replaced by normal tissue. Biodegradation of CS-PEG can occur via hydrolysis or enzymatically. There are at least three enzymes identified in the human genome which are able to degrade CS include hyaluronidase-1 (Hyal-1), hyaluronidase-4 (Hyal-4) and PH-20. Hyal-1 and PH-20 act primarily on hyaluronan, but can also degrade CS, while Hyal-4 is specific for CS degradation [36-38]. This material is therefore subject to enzymatic degradation in vivo as modeled by the in vitro degradation.
In addition to biodegradation, it is important that biomaterials stimulate or at least not impede cell function. One solution is to utilize biological polymers as components in the scaffold. This usually involves chemical modifications to the polymer which could alter its structure significantly such that it loses its bioactivity. Therefore, we injected the CS-PEG scaffold in rats and observed that it decreased the inflammatory response when compared to a control PEG scaffold. CS has been shown to alleviate the inflammatory response by mediating cellular responses, cytokine secretion, and intracellular signaling [39]. Thus, the presence of CS could have contributed to the observed decrease in the duration of inflammation. This supports the idea that the chemical modification of the CS molecule does not alter its biological functions.
We showed that the sealing properties of the hydrogel can be controlled by doping in HA to vary the hydrogel’s modulus and damping. In agreement with other studies the burst pressures of the hydrogel improved as the bulk modulus increased [40,41]. However, this is the first study which demonstrates that the sealing properties of a hydrogel material are improved with an increase in damping. Since damping becomes important under dynamic conditions, we believe that the rate of filling is having an impact on the burst pressure outcomes. Therefore, optimizing the damping characteristics of the hydrogel would be important when sealing a dynamic tissue such as a blood vessel, lung or intestine.
Improved material adhesion to the underlying tissue has been shown to improve tissue integration in vitro and in vivo [42]. Here we show that relative to fibrin glue, CS-PEG gels have 10 times stronger adhesion to cartilage tissue (Fig. 4H). In addition to forming physical crosslinks to tissue like fibrin glue, CS-PEG is also able to form covalent crosslinks by reacting with primary amines found on collagen in the ECM to form amide bonds. Therefore, the added covalent crosslinks account for the better adhesion properties of the material.
5. Conclusion
In this paper we have demonstrated the successful synthesis of a CS-NHS macromolecule that can react with polyamines to form a hydrogel and with proteins of tissue to form an adhesive. In particular we have characterized a scaffold/adhesive composed of CS-NHS and PEG-(NH2)6. We have shown that at higher initial pH, the gels formed are softer, swell more and gel more quickly, and that altering the initial pH can tune these parameters. Varying the CS to PEG ratio does not affect swelling or gelation times, but at low CS concentrations, softer gels are formed. Swelling equilibrium is reached within five and a half hours independent of crosslinking density or osmolarity of the swelling medium. The material is not toxic to chondrocytes and is able to seal incisions made in collagen membranes. We found that damping and storage moduli are both directly related to the burst pressure of the material. Additionally, the cartilage adhesive strength of CS-PEG is ten times that of fibrin glue. Finally, we showed that the material is biodegradable and the biological activity of CS is maintained following its modification with NHS.
Acknowledgments
The authors thank Dr. Trollsas from Abbott Laboratories, IL for the helpful discussion.
Footnotes
Funding for this work was provided by NIH (Grant # R01EB05517), and Coulter Foundation (Grant # E222008)
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Wang H, Katagiri Y, McCann TE, Unsworth E, Goldsmith P, Yu ZX, et al. Chondroitin-4-sulfation negatively regulates axonal guidance and growth. J Cell Sci. 2008;121:3083–3091. doi: 10.1242/jcs.032649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ueoka C, Kaneda N, Okazaki I, Nadanaka S, Muramatsu T, Sugahara K. Neuronal cell adhesion, mediated by the heparin-binding neuroregulatory factor midkine, is specifically inhibited by chondroitin sulfate E. Structural and functional implications of the over-sulfated chondroitin sulfate. J Biol Chem. 2000;275:37407–37413. doi: 10.1074/jbc.M002538200. [DOI] [PubMed] [Google Scholar]
- 3.Ronca F, Palmieri L, Panicucci P, Ronca G. Anti-inflammatory activity of chondroitin sulfate. Osteoarthr Cartil. 1998;6(Suppl A):14–21. doi: 10.1016/s1063-4584(98)80006-x. [DOI] [PubMed] [Google Scholar]
- 4.Campo GM, Avenoso A, Campo S, D’Ascola A, Traina P, Sama D, et al. Glycosaminoglycans modulate inflammation and apoptosis in LPS-treated chondrocytes. J Cell Biochem. 2009;106:83–92. doi: 10.1002/jcb.21981. [DOI] [PubMed] [Google Scholar]
- 5.McGee M, Wagner WD. Chondroitin sulfate anticoagulant activity is linked to water transfer: relevance to proteoglycan structure in atherosclerosis. Arterioscler Thromb Vasc Biol. 2003;23:1921–1927. doi: 10.1161/01.ATV.0000090673.96120.67. [DOI] [PubMed] [Google Scholar]
- 6.Akiyama F, Seno N, Yoshida K. Anticoagulant activity of dermatan polysulfates. Tohoku J Exp Med. 1982;136:359–365. doi: 10.1620/tjem.136.359. [DOI] [PubMed] [Google Scholar]
- 7.Hayes AJ, Tudor D, Nowell MA, Caterson B, Hughes CE. Chondroitin sulfate sulfation motifs as putative biomarkers for isolation of articular cartilage progenitor cells. J Histochem Cytochem. 2008;56:125–138. doi: 10.1369/jhc.7A7320.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Volpi N. Inhibition of human leukocyte elastase activity by chondroitin sulfates. Chem Biol Interact. 1997;105:157–167. doi: 10.1016/s0009-2797(97)00045-8. [DOI] [PubMed] [Google Scholar]
- 9.Raepple E, Hill HU, Loos M. Mode of interaction of different polyanions with the first (C1, C1), the second (C2) and the fourth (C4) component of complement--I. Effect on fluid phase C1 and on C1 bound to EA or to EAC4. Immunochemistry. 1976;13:251–255. doi: 10.1016/0019-2791(76)90223-8. [DOI] [PubMed] [Google Scholar]
- 10.Raeber GP, Lutolf MP, Hubbell JA. Molecularly engineered PEG hydrogels: a novel model system for proteolytically mediated cell migration. 2005;89:1374–1388. doi: 10.1529/biophysj.104.050682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Appelman TP, Mizrahi J, Elisseeff JH, Seliktar D. The differential effect of scaffold composition and architecture on chondrocyte response to mechanical stimulation. Biomaterials. 2009;30:518–525. doi: 10.1016/j.biomaterials.2008.09.063. [DOI] [PubMed] [Google Scholar]
- 12.Villanueva I, Klement BJ, von Deutsch D, Bryant SJ. Cross-linking density alters early metabolic activities in chondrocytes encapsulated in poly(ethylene glycol) hydrogels and cultured in the rotating wall vessel. Biotechnol Bioeng. 2009;102:1242–1250. doi: 10.1002/bit.22134. [DOI] [PubMed] [Google Scholar]
- 13.Varghese S, Hwang NS, Canver AC, Theprungsirikul P, Lin DW, Elisseeff J. Chondroitin sulfate based niches for chondrogenic differentiation of mesenchymal stem cells. Matrix Biol. 2008;27:12–21. doi: 10.1016/j.matbio.2007.07.002. [DOI] [PubMed] [Google Scholar]
- 14.Williams CG, Kim TK, Taboas A, Malik A, Manson P, Elisseeff J. In vitro chondrogenesis of bone marrow-derived mesenchymal stem cells in a photopolymerizing hydrogel. Tissue Eng. 2003;9:679–688. doi: 10.1089/107632703768247377. [DOI] [PubMed] [Google Scholar]
- 15.Mahoney MJ, Anseth KS. Three-dimensional growth and function of neural tissue in degradable polyethylene glycol hydrogels. Biomaterials. 2006;27:2265–2274. doi: 10.1016/j.biomaterials.2005.11.007. [DOI] [PubMed] [Google Scholar]
- 16.Namba RM, Cole AA, Bjugstad KB, Mahoney MJ. Development of porous PEG hydrogels that enable efficient, uniform cell-seeding and permit early neural process extension. Acta Biomater. 2009;5:1884–1897. doi: 10.1016/j.actbio.2009.01.036. [DOI] [PubMed] [Google Scholar]
- 17.Comolli N, Neuhuber B, Fischer I, Lowman A. In vitro analysis of PNIPAAm-PEG, a novel, injectable scaffold for spinal cord repair. Acta Biomater. 2008;5:1046–1055. doi: 10.1016/j.actbio.2008.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Adelow C, Segura T, Hubbell JA, Frey P. The effect of enzymatically degradable poly(ethylene glycol) hydrogels on smooth muscle cell phenotype. Biomaterials. 2008;29:314–326. doi: 10.1016/j.biomaterials.2007.09.036. [DOI] [PubMed] [Google Scholar]
- 19.Abuchowski A, van Es T, Palczuk NC, Davis FF. Alteration of immunological properties of bovine serum albumin by covalent attachment of polyethylene glycol. J Biol Chem. 1977;252:3578–3581. [PubMed] [Google Scholar]
- 20.Dalsin JL, Hu BH, Lee BP, Messersmith PB. Mussel adhesive protein mimetic polymers for the preparation of nonfouling surfaces. J Am Chem Soc. 2003;125:4253–4258. doi: 10.1021/ja0284963. [DOI] [PubMed] [Google Scholar]
- 21.Dalsin JL, Lin L, Tosatti S, Voros J, Textor M, Messersmith PB. Protein resistance of titanium oxide surfaces modified by biologically inspired mPEG-DOPA. Langmuir. 2005;21:640–646. doi: 10.1021/la048626g. [DOI] [PubMed] [Google Scholar]
- 22.Zhang F, Kang ET, Neoh KG, Wang P, Tan KL. Reactive coupling of poly(ethylene glycol) on electroactive polyaniline films for reduction in protein adsorption and platelet adhesion. Biomaterials. 2002;23:787–795. doi: 10.1016/s0142-9612(01)00184-3. [DOI] [PubMed] [Google Scholar]
- 23.Strehin I, Ambrose WM, Schein O, Salahuddin A, Elisseeff J. Synthesis and characterization of a chondroitin sulfate-polyethylene glycol corneal adhesive. J Cataract Refract Surg. 2009;35:567–576. doi: 10.1016/j.jcrs.2008.11.035. [DOI] [PubMed] [Google Scholar]
- 24.Lipmann F, Tuttle LC. A specific micromethod for the determination of acyl phosphates. J Biol Chem. 1945;159:21–28. [Google Scholar]
- 25.Schnaar RL, Lee YC. Polyacrylamide gels copolymerized with active esters. A new medium for affinity systems. Biochemistry. 1975;14:1535–1541. doi: 10.1021/bi00678a030. [DOI] [PubMed] [Google Scholar]
- 26.Iwata H, Matsuda S, Mitsuhashi K, Itoh E, Ikada Y. A novel surgical glue composed of gelatin and N-hydroxysuccinimide activated poly(L-glutamic acid): Part 1. Synthesis of activated poly(L-glutamic acid) and its gelation with gelatin. Biomaterials. 1998;19:1869–1876. doi: 10.1016/s0142-9612(98)00095-7. [DOI] [PubMed] [Google Scholar]
- 27.Menard KP. Dynamic mechanical analysis: A practical introduction. Boca Raton, Fla.: CRC Press; 1999. [Google Scholar]
- 28.Kim TK, Sharma B, Williams CG, Ruffner MA, Malik A, McFarland EG, et al. Experimental model for cartilage tissue engineering to regenerate the zonal organization of articular cartilage. Osteoarthritis Cartilage. 2003;11:653–664. doi: 10.1016/s1063-4584(03)00120-1. [DOI] [PubMed] [Google Scholar]
- 29.Cruise GM, Hegre OD, Scharp DS, Hubbell JA. A sensitivity study of the key parameters in the interfacial photopolymerization of poly(ethylene glycol) diacrylate upon porcine islets. Biotechnol Bioeng. 1998;57:655–665. doi: 10.1002/(sici)1097-0290(19980320)57:6<655::aid-bit3>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 30.Balazs EA, Watson D, Duff IF, Roseman S. Hyaluronic acid in synovial fluid. I. Molecular parameters of hyaluronic acid in normal and arthritic human fluids. Arthritis Rheum. 1967;10:357–376. doi: 10.1002/art.1780100407. [DOI] [PubMed] [Google Scholar]
- 31.Miki D, Dastgheib K, Kim T, Pfister-Serres A, Smeds KA, Inoue M, et al. A photopolymerized sealant for corneal lacerations. Cornea. 2002;21:393–399. doi: 10.1097/00003226-200205000-00012. [DOI] [PubMed] [Google Scholar]
- 32.Peyton SR, Raub CB, Keschrumrus VP, Putnam AJ. The use of poly(ethylene glycol) hydrogels to investigate the impact of ECM chemistry and mechanics on smooth muscle cells. Biomaterials. 2006;27:4881–4893. doi: 10.1016/j.biomaterials.2006.05.012. [DOI] [PubMed] [Google Scholar]
- 33.Peyton SR, Kim PD, Ghajar CM, Seliktar D, Putnam AJ. The effects of matrix stiffness and RhoA on the phenotypic plasticity of smooth muscle cells in a 3-D biosynthetic hydrogel system. Biomaterials. 2008;29:2597–2607. doi: 10.1016/j.biomaterials.2008.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hermanson GT. Bioconjugate Techniques. San Diego: Academic Press; 1996. NHS esters; pp. 139–140. [Google Scholar]
- 35.Kurz A. Thermal care in the perioperative period. Best Pract Res Clin Anaesthesiol. 2008;22:39–62. doi: 10.1016/j.bpa.2007.10.004. [DOI] [PubMed] [Google Scholar]
- 36.Csoka AB, Frost GI, Stern R. The six hyaluronidase-like genes in the human and mouse genomes. Matrix Biol. 2001;20:499–508. doi: 10.1016/s0945-053x(01)00172-x. [DOI] [PubMed] [Google Scholar]
- 37.Stern R. Devising a pathway for hyaluronan catabolism: are we there yet? Glycobiology. 2003;13:105R–115R. doi: 10.1093/glycob/cwg112. [DOI] [PubMed] [Google Scholar]
- 38.Jedrzejas MJ, Stern R. Structures of vertebrate hyaluronidases and their unique enzymatic mechanism of hydrolysis. Proteins. 2005;61:227–238. doi: 10.1002/prot.20592. [DOI] [PubMed] [Google Scholar]
- 39.Iovu M, Dumais G, du Souich P. Anti-inflammatory activity of chondroitin sulfate. Osteoarthritis Cartilage. 2008;16(Suppl 3):S14–8. doi: 10.1016/j.joca.2008.06.008. [DOI] [PubMed] [Google Scholar]
- 40.Berglund JD, Mohseni MM, Nerem RM, Sambanis A. A biological hybrid model for collagen-based tissue engineered vascular constructs. Biomaterials. 2003;24:1241–1254. doi: 10.1016/s0142-9612(02)00506-9. [DOI] [PubMed] [Google Scholar]
- 41.Isayeva IS, Gent AN, Kennedy JP. Synthesis and burst strength of water-swollen immunoisolatory tubules. J Polym Sci A Polym Chem. 2002;40:2075–2084. [Google Scholar]
- 42.Wang DA, Varghese S, Sharma B, Strehin I, Fermanian S, Gorham J, et al. Multifunctional chondroitin sulphate for cartilage tissue-biomaterial integration. Nat Mater. 2007;6:385–392. doi: 10.1038/nmat1890. [DOI] [PMC free article] [PubMed] [Google Scholar]


