Background: Most amyotrophic lateral sclerosis (ALS) cases are characterized with TDP-43(+), ubiquitin(+) inclusions in their diseased spinal cord motor neurons.
Results: Mice with targeted depletion of TDP-43 expression in the spinal cord motor neurons developed a range of ALS-like phenotypes.
Conclusion: TDP-43 is essential for the survival and functioning of mammalian spinal cord motor neurons.
Significance: Loss of TDP-43 function could be one major cause for neurodegeneration in ALS with TDP-43 proteinopathies.
Keywords: Amyotropic Lateral Sclerosis (Lou Gehrig's Disease), Cell Death, Mouse Genetics, Neurodegenerative Diseases, RNA-binding Proteins, Motor Dysfunction, Motor Neuron Loss, TDP-43, Targeted Gene Knock-out In Mice
Abstract
ALS, or amyotrophic lateral sclerosis, is a progressive and fatal motor neuron disease with no effective medicine. Importantly, the majority of the ALS cases are with TDP-43 proteinopathies characterized with TDP-43-positive, ubiquitin-positive inclusions (UBIs) in the cytosol. However, the role of the mismetabolism of TDP-43 in the pathogenesis of ALS with TDP-43 proteinopathies is unclear. Using the conditional mouse gene targeting approach, we show that mice with inactivation of the Tardbp gene in the spinal cord motor neurons (HB9:Cre-Tardbplx/−) exhibit progressive and male-dominant development of ALS-related phenotypes including kyphosis, motor dysfunctions, muscle weakness/atrophy, motor neuron loss, and astrocytosis in the spinal cord. Significantly, ubiquitinated proteins accumulate in the TDP-43-depleted motor neurons of the spinal cords of HB9:Cre–Tardbplx/− mice with the ALS phenotypes. This study not only establishes an important role of TDP-43 in the long term survival and functioning of the mammalian spinal cord motor neurons, but also establishes that loss of TDP-43 function could be one major cause for neurodegeneration in ALS with TDP-43 proteinopathies.
Introduction
Amyotrophic lateral sclerosis (ALS)2 (also known as Lou Gehrig's disease and motor neuron disease) is a progressive, fatal, adult-onset degenerative disorder of motor neurons in the primary motor cortex, corticospinal tracts, brainstem, and spinal cord, leading to paralysis of voluntary muscles (1–3). Currently, the incidence of ALS is 1–2 per 100,000 each year and its prevalence is 4–6 per 100,000 of the total population, with a lifetime ALS risk of 1 in 400 to 1 in 1,000 (4). Most incidences of ALS are sporadic but ∼10% of patients have a familial history (known as familial ALS; fALS). In both sALS and fALS, there are progressive manifestations of dysfunction of the lower motor neurons and cortical motor neurons but without sensory symptoms (3). Age and gender are documented sALS risk factors (5) with a male-to-female ratio of 3:2 among patients. Among the mutations associated with ALS, those in the copper-zinc superoxide dismutase (SOD1) gene have long been thought to cause ALS through a toxic gain of function rather than impairment of the antioxidant function of the SOD1 enzyme (6). Other genes with mutations associated with the fALS include alsin (ALS2), senataxin (ALS4), vesicle-associated membrane protein (VAPB, ALS8), Angiogenin, and the p150 subunit of dynactin (DCTN1) (3). More than 30 mutations in the TDP-43-coding region of Tardbp have also been identified in ALS patients with or without apparent family history, corresponding to ∼4% of fALS and less than 1% of sALS (7). Most patients with the TDP-43 mutation(s) develop a classical ALS phenotype without cognitive deficit suggesting an important role of TDP-43 in the development of ALS (7–9).
TDP-43, or TAR DNA-binding protein-43 (10), is an ubiquitously expressed nuclear protein encoded by one of the mRNA isoforms from the highly conserved Tardbp gene (11). It is a RNA-binding protein involved in transcriptional repression, pre-mRNA splicing, and translation (12, 13). TDP-43 has also been identified as the major pathological signature protein of the intracellular inclusions typical for disease cells of a range of neurodegenerative diseases, including the frontotemporal lobar degeneration with ubiquitin-positive, Tau- and α-synuclein-negative inclusions (FTLD-U) and ALS (13–17). TDP-43 molecules in the diseased cells of the patient brains or spinal cords are characterized by abnormal ubiquitination, hyperphosphorylation, and partial cleavage to generate ∼25-kDa and 35-kDa C-terminal fragment(s). Furthermore, TDP-43 is partially or completely cleared from the nuclei of either neuronal or glial cells containing the TDP-43(+) and ubiquitin(+) aggregates/inclusions, or UBIs, in the cytoplasm (18).
Several mouse models have been established for ALS disease including the strains of rodents that are transgenic with SOD1, ALS2 knock-out mice, and mice with genetically engineered genes coding for the neurofilament subunits (reviewed in Refs. 19 and 20). Among these, the mutant human SOD1 (hSOD1) transgenic mouse model is currently the most widely used because it shares several clinical phenotypes with the ALS patients. The first symptom of the hSOD1 mice is a fine “jittering/tremor” in one or more of the limbs, which appears at ∼90 to 100 days of age (21, 22). At later stages, the cytopathological features of the hSOD1 transgenic mice include motor neuron loss with astrocytosis, the presence of SOD1-positive inclusions including Lewy body-like hyaline inclusions/astrocyte hyline inclusions, and vacuole formation (23).
Overexpression of TDP-43 in transgenic rodents could also lead to development of motor neuron disease-like symptoms. These transgenic rodents develop one or more of several symptoms, which include motor neuron dysfunction, muscle defect-related pathology, and neuronal loss (24–27). The lifespans of some of these transgenic mouse lines are short, likely due to the cytotoxicity of the overexpressed TDP-43 as well as the relatively lower motor neuron specificity of the promoters, e.g. Thy1, prion, etc., used to express the transgenes (24, 26–28). Finally, the appearance of cells with cytoplasmic TDP-43(+) UBIs and TDP-43-depleted nuclei at later stages of pathogenesis of the TDP-43 transgenic mice (25, 26) suggest that the disease phenotypes in the TDP-43 transgenic mice may result in part from loss of function of TDP-43. However, the pathological phenotypes of the mice could also be caused entirely by gain of toxicity from overexpression of the exogenous TDP-43.
Thus, the relative contributions of loss of function and gain of cytotoxicity to the neurodegeneration in FTLD-U and ALS with TDP-43(+) UBIs are not clear (reviewed in Refs. 16 and 29–31). Also, regardless of its currently known biochemical and structural properties, the physiological functions of TDP-43 in different mammalian tissues are also unknown. Previously, we have shown, by gene targeting approaches in mice, that TDP-43 is important for mouse early embryonic development (32). As described in the following, we have taken advantage of the Tardbplx mouse line from that study and generated mice with spinal cord motor neuron-specific knock-out of TDP-43 expression. Furthermore, these mice exhibit pathological phenotypes in striking similarity to ALS.
EXPERIMENTAL PROCEDURES
Generation of HB9 Promotor-driven Conditional Tardbp Gene Knock-out Mice
The Tardbp allele of the C57BL/6j mice was knocked out specifically in the postmitotic motor neurons in the spinal cord by crossing mice carrying the Tardbp conditional allele (Tardbplx) with mice carrying a Cre-recombinase transgene driven by the HB9-promoter (HB9:Cre)(see “Results” for more details). The viabilities and weights of the mice were monitored regularly. Genotyping of the mice was performed by PCR of genomic DNAs from the tail biopsies in comparison to the nail samples.
Tests of Motor Neuron Functions
For the limb-clasping test, the mice were suspended by pulling their tails. For the rotarod test, the mice were trained before testing to exclude differences in motivation and motor learning. Mice were first trained for four consecutive days. In the testing phase, they were put on the rod rotating constantly at 2.5 rpm, and the speed was gradually increased to 25 rpm over a 3-min period. The timer was stopped when the mice fell from the rod or when they gripped the rod and started to rotate with it.
Tissue Preparation and Immunostaining Analysis
The mice were sacrificed under deep anesthesia, and perfused transcardially with 4% paraformaldehyde in PBS (pH 7.4). The spinal cords were dissected and the lumber segments (L3–L6) were identified using the ribs and vertebrae as the guide. The segments were processed for cryoprotection, and 100 serial cross-sections were made of the lumbar spinal cords at a thickness of 10 μm. Every 12th section (12 sections total from each animal) was stained with 1% cresyl violet (Sigma). Each section was visualized with an Axioimage-Z1 light microscope at ×20 magnification, and the cells were counted manually by tracing the perimeter of each motor neuron. The cell counts were made within an area demarcated by a horizontal line drawn through the central canal and encompassing the ventral horn of the gray matter to include layers 7–9. Initially, we identified the motor neurons using the following criteria: 1) the presence of a large nucleolus located within the nucleus surrounded by light blue-staining cytoplasm; and 2) a cell soma area over 100 μm2. The γ motor neurons range in their soma areas from 100 to 250 μm2, whereas the soma areas of the larger α motor neurons range from 250 to 1100 μm2.
In parallel sets of the sections, the motor neurons were also analyzed by immunofluorescence staining with single or combined use of several different antibodies: a goat antibody directed against the choline acetyltransferase (ChAT; Chemicon), a rabbit anti-TDP-43 (Proteintech), a rabbit anti-GFAP (Milopore), a rabbit anti-Iba1 (Wako), a mouse anti-MAP2 (Sigma), a mouse anti-ubiquitin (Zymed Laboratories Inc.), a mouse anti-neuronal nuclei (NeuN), and a mouse anti-human ERRγ (PPMX). The sections were preincubated in PBS solution containing 10% (v/v) normal donkey serum and 0.2% Triton X-100 for 1 h, and then incubated overnight with one or more of the antibodies. Antibody against the rabbit/mouse IgG (Jackson ImmunoResearch) was used for secondary detection. The images were taken from the ZEISS-LSM710 and LSM780 confocal microscope.
Immunoblotting Analysis
Total extracts of the spinal cords were prepared by homogenization of the tissue in the RIPA buffer (0.1% SDS, 1% Nonidet P-40, 0.5% sodium deoxycholate, 5 mm EDTA, 150 mm NaCl, 50 mm Tris-HCl, pH 8.0) containing protease and phosphatase inhibitors. The RIPA-soluble and urea-soluble fractions of the proteins from the spinal cords were obtained by centrifugation of the tissue homogenate at 15,000 × g at 4 °C for 30 min. The supernatant was collected as the “RIPA-soluble” fraction. The pellet was washed 3 times with RIPA buffer and then solublized in the urea buffer (7 m urea, 2 m thiourea, 4% CHAPS, 30 mm Tris-HCl, pH 8.5) to give the “urea-soluble” fraction. 4 μg of RIPA extract or ×4 equivalent volumes of urea extract per lane were separated on a 10% Tris glycine SDS-PAGE gel. Immunoblotting analysis of the RIPA-soluble and urea-soluble fractions of the spinal cord extracts followed standard procedures with use of appropriate antibodies. For comparing the protein levels of WT, Tardbplx/− mice, and HB9:Cre-Tardbplx/− mice, quantification of band intensity was performed using Image J software (NIH). For TDP-43/ChAT expression quantification, protein levels relative to wild type (WT) mice was determined using TDP-43/ChAT immunoblots within the linear range of band intensity after normalization to tubulin expression.
RESULTS
Generation of Mice with Conditional Knock-out of Tardbp Expression in the Spinal Cord Motor Neurons
The Tardbp gene was inactivated in the motor neurons with use of the loxP/cre recombination system. We first generated a conditional allele of the Tardbp locus by flanking exons 2 and 3 with loxP sites (Tardbplx allele), which was suitable for Cre-mediated Tardbp-floxed gene inactivation (Fig. 1A; Ref. 32). We then crossed the mice with motor neuron-specific Mnx1 (HB9):Cre mice to generate the HB9:Cre-Tardbplx/− mice (Fig. 1).
FIGURE 1.
Targeted disruption of the mouse Tardbp gene in spinal cord motor neurons. Exons 2 (E2) and 3 (E3) of TDP-43 were replaced with a ”floxed“ fragment containing exons 2 and 3 followed by a frt-flanking neo cassette. The neo cassette was then removed by germline expression of flp recombinase and the two exons were removed by HB9 promoter-driven Cre recombinase from the HB9:Cre mice. The genotypes of mice carrying the different alleles were validated by PCR of their genomic DNAs using primers (a, b, and c) shown in the figure.
HB9 is a homeodomain transcription factor that is expressed selectively in motor neurons in the developing spinal cord (E9.5) and is essential for differentiation of the postmitotic motor neurons (33). Also, HB9:Cre mice express the cre gene specifically in the spinal cord motor neurons and have been used previously to manipulate gene expression in the motor neurons (33). The motor neuron specificity of the HB9-driven Cre expression have also been verified by crossing the HB9:Cre mouse line to a ROSA26:LacZ reporter mice in several studies (33–38). Examination of the GFP signals in HB9:GFP transgenic mice have indicated that expression of HB9 is restricted in the motor neurons, but not in the sensory neurons (39).
On the other hand, it should be emphasized here that HB9 expression in cells other than the spinal cord motor neurons could not be completely ruled out. For instance, expression of HB9:Cre has also been observed in cells other than the ventral motor neurons, which could be due to HB9 promoter activity in the visceral motor neurons or in motor neuron progenitors (34). Other studies have shown that Cre is expressed in motor neurons as well as in some ventral interneurons at the embryonic stage (35, 36). In any case, as shown and described below, the use of HB9:Cre mice eliminated TDP-43 expression mainly in ChAT(+) motor neurons in the spinal cords of our conditional knock-out mice.
Development of ALS-like Morphological and Behavioral Phenotypes in HB9:Cre-Tardbplx/− Mice
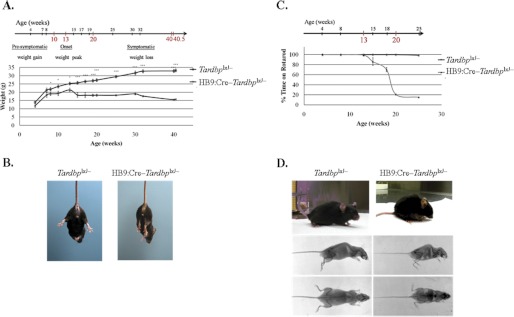
Unlike the EIIa:Cre-Tardbplx/lx mice we generated previously (32), which were lethal at the peri-implantation stage, HB9:Cre–Tardbplx/− mice were viable and phenotypically normal at birth, suggesting that TDP-43 was not essential for normal development of spinal cord motor neurons. However, although the average weight of HB9:Cre–Tardbplx/− mice was only slightly lower than the controls at early births, the difference became more prominent afterward (Fig. 2A). The body weight was shown previously to be a simple and reliable measure for the disease “onset” and progression in the ALS mouse model with transgenic expression of the mutant hSOD1 (G93A) (40), with the inflection point of the weight curve providing a simple definition of the disease onset. Interestingly, similar to the mutant hSOD1 transgenic mice, HB9:Cre–Tardbplx/− mice also showed a peak of weight gain during 90 to 100 days (∼13 weeks, Fig. 2A). Soon after that, the mice started to show significant weight loss (Fig. 2A), abnormal hind limb clasping (Fig. 2B), and deficiency in rotarod test (Fig. 2C). It should be noted here that throughout our study, Tardbplx/− mice were used as the control in comparison to the HB9:Cre-Tardbplx/− mice. Like in the study of EIIa:Cre-Tardbplx/+ mice (32), the levels of TDP-43 expression in the spinal cords of Tardbplx/− and the wild-type mice, either 10- or 20-week-old, were similar (supplemental Fig. S1A).
FIGURE 2.
ALS-like phenotypes of HB9:Cre–Tardbplx/− mice. A, top, time scale of the progression of ALS-like phenotypes of the HB9:Cre–Tardbplx/− mice, as exemplied in A–D. Below, time course of the body weight changes (weights at different ages, mean ± S.E.). The mean body weight of HB9:Cre–Tardbplx/− mice is statistically different from that of the Tardbplx/− mice after 8 weeks (*, p < 0.05; ***, p < 0.005). The disease onset of the HB9:Cre–Tardbplx/− mice is defined as the start of the weight loss at around 13 weeks. B, hindlimb clasping test. Abnormal clasping of a 20-week-old HB9:Cre–Tardbplx/− mouse is exemplied in the right panel as compared with a control littermate in the left panel. C, rotarod test. The indices (average time on the rotarod after normalization to the control, mean ± S.E.) are plotted against the ages of the mice. D, kyphosis phenotype of the mutant mouse (right panels) in comparison to the control (left panels) at 20 weeks of age. The lower 4 panels are the contrast enhancement radiography of the skeletons. n = 6 for each group.
Although all of the above clinical signs were similar between the HB9:Cre–Tardbplx/− mice and the mutant hSOD1 transgenic mice, the disease progression after the symptom onset was somewhat slower in HB9:Cre–Tardbplx/− mice. Significantly, HB9:Cre–Tardbplx/− mice exhibited a kyphosis phenotype beginning at 20 weeks of age and it became severe at 24 weeks, as exemplified in Fig. 2D. The kyphosis likely resulted from weakening of the extensor thoracic paraspinal muscles, as observed in ALS patients (41) as well as in the mutant hSOD1 (G93A) transgenic mice (42). The apparent muscle weakness of HB9:Cre–Tardbplx/− mice was accompanied with muscle atrophy as revealed by morphological analysis of the gastrocnemius and soleus muscles of the mice (supplemental Fig. S2). Finally, similar to ALS (3), the development of ALS-like phenotypes in HB9:Cre–Tardbplx/− mice were also male dominant, with a male/female ratio of 3:1. The average lifespan of the HB9:Cre–Tardbplx/− mice showing ALS-like phenotypes was 10 months.
Loss of Motor Neurons and Enhancement of Astrocytosis in the Spinal Cord of HB9:Cre-Tardbplx/− Mice
ALS was defined as a neurodegenerative disorder characterized by progressive muscle weakness indicating the degeneration of motor neurons in the primary motor cortex, brainstem, and spinal cord. Progressive atrophy of the muscle fibers, which were denervated as their corresponding motor neurons degenerated, then led to weakening of the affected muscles. We reasoned that the restricted population of the spinal cord motor neurons in HB9:Cre-Tardbplx/− mice might be affected by the depletion of TDP-43. Indeed, immunofluorescence staining and histology analysis of the spinal cords showed a decrease of ChAT-positive motor neurons among the ventral horn cells of 20-week-old HB9:Cre–Tardbplx/− mice (Fig. 3A). Furthermore, among the remaining ChAT-positive motor neurons of HB9:Cre–Tardbplx/− mice, 62% lost the immunostaining signals of TDP-43, as exemplified in the confocal images (Fig. 3A and supplemental Fig. S3) and statistically presented in the histogram of Fig. 3A.
FIGURE 3.
Motor neuron loss in the spinal cords of 20-week-old HB9:Cre–Tardbplx/− mice. A, immunofluorescence co-staining of ChAT and TDP-43 showing that the number of ChAT-positive motor neurons (MN) in the ventral horn of the lumbar spinal cord of 20-week-old HB9:Cre–Tardbplx/− mice (lower two rows of panels) was reduced in comparison to the control (upper two rows). The 2nd and 4th rows are magnified photos of the boxed areas in the 1st and 3rd rows of photos, respectively. The scale bars are 50 μm. The quantitative comparison is shown by the bar histogram below the photographs. Note the depletion of TDP-43 in most, although not all, of the ChAT(+) cells, or motor neurons. Also, ∼62% of the ChAT(+) cells of the mutant mice are without TDP-43 expression. ***, p < 0.05. B, top, photomicrographs showing the cresyl violet (Nissl)-stained sections through the lumbar spinal cord from a HB9:Cre–Tardbplx/− mouse at the age of 20 weeks (lower two panels) in comparison to a control littermate (upper two panels). The area of one ventral horn of each section is magnified for better visualization. The scale bar is 50 μm. Below, the quantitative analysis is presented by the bar histogram. Note the reduction of both the α motor neurons (gray bars, by 38%) and the γ motor neurons (white bars, by 59%) in the mutant mice. n = 4 for each group. *, p < 0.05; **, p < 0.01.
The amounts of TDP-43 and ChAT in the spinal cords of the mutant and control mice were also analyzed by immunoblotting. As shown in supplemental Fig. S1B, the difference between the amounts of TDP-43 of the spinal cords of Tardbplx/− and HB9:Cre-Tardbplx/− mice was barely detectable (supplemental Fig. S1B) because HB9-expressing cells only accounted for 5–10% of the total cell population of the spinal cord (33, 34). On the other hand, the level of the ChAT protein was significantly reduced in the HB9:Cre-Tardbplx/− spinal cord (supplemental Fig. S1B), consistent with loss of the ChAT(+) spinal cord motor neurons in the mutant mice as described for Fig. 3A.
The numbers of motor neurons in the ventral horn of the lumbar (L3–L5) level of the spinal cords of mice were quantified with respect to the α- and γ-types. In the mammalian spinal cords, α and γ motor neurons could be distinguished both by the cell body sizes, for which the γ motor neurons were significantly smaller than the α motor neurons (43), and by the expression of specific molecular markers, e.g. NeuN for the α motor neurons (44, 45). As seen in Fig. 3B, Nissl staining showed that the numbers of both the α and γ motor neurons, as defined by their relative soma sizes, in the lumbar regions of the spinal cords of 20-week-old HB9:Cre–Tardbplx/− mice were decreased significantly when compared with the control mice.
In view of the reported changes of the cell body sizes of neurons from ALS or with TDP-43 proteinopathies (for example, Refs. 46 and 47), we also carried out immunofluorescence staining experiments in which both ChAT and NeuN were used as markers to identify the total, α-, and γ-type motor neurons, respectively, in the spinal cords of 10-week-old (Fig. 4, upper panel) as well as 20-week-old mice (Fig. 4, lower panel). As seen, a significant loss of the motor neurons, either the α- or γ-type, was also observed in the symptomatic 20-week-old HB9:Cre-Tardbplx/− mice (Fig. 4, lower panel), although the overall Nissl staining patterns (Fig. 3B) or NeuN staining patterns of the spinal cord sections were similar between the HB9:Cre-Tardbplx/− and control mice. Notably, consistent with the report that larger α motor neurons in ALS were more vulnerable to degeneration (48), the % of reduction of the α motor neurons (∼46%) in the 20-week-old HB9:Cre-Tardbplx/− mice was larger than that of the γ motor neurons (∼25%), as estimated with use of the molecular markers (Fig. 4, lower panel). Finally, a small decrease (∼10%) of spinal cord motor neurons could also be detected at the presymptomatic age of 10 weeks (Fig. 4, upper panel). This progressive loss of motor neurons during ALS pathogenesis of our mouse model was very similar to that observed before in the ALS mouse model with transgenic overexpression of hSOD1 (21). Altogether, the data of Fig. 4 suggested that the pathological symptoms of HB9:Cre-Tardbplx/− mice were associated mainly with the loss of spinal cord motor neurons at the symptomatic on-set stage (20 weeks old).
FIGURE 4.
Immunofluorescence co-staining analysis of the lumbar spinal cord motor neurons of 10-week-old and 20-week-old mice with use of anti-ChAT (white), anti-NeuN (green), DAPI (blue), and anti-TDP-43 (red). The photos are exemplified in the left 4 rows of panels. The scale bars are 50 μm. Right, the quantitative analysis is presented by the 2 bar histograms. Note the reduction of both the α (NeuN(+)/ChAT(+)) motor neurons (gray bars, by 46%) and the γ (NeuN(−)/ChAT(+)) motor neurons (white bars, by 25%) in the 20-week-old mutant mice (lower histogram, n = 4 for each groups; *, p < 0.05; ***, p < 0.005). On the other hand, the number of the total motor neurons (MN) in the lumbar spinal cord of the 10-week-old HB9:Cre–Tardbplx/− mice was reduced only marginally, by 11%, in comparison to the control (black bars of the upper histogram; n = 4 for each group; *, p < 0.05).
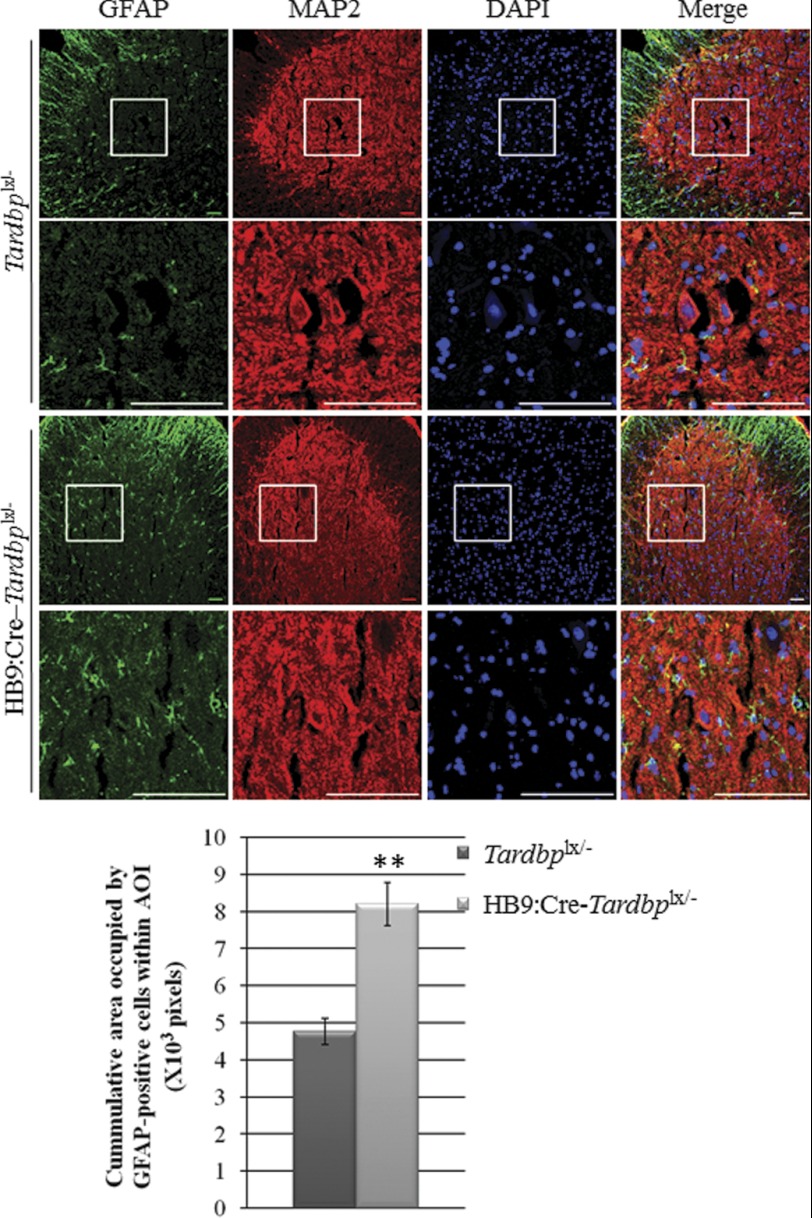
Because reactive astrocytosis and microglia activation were often taken as indications of neuronal toxicity or neuronal death (49), we first quantified the level of the glial fibrillary acidic protein (GFAP) expression by immunofluorescence staining. As seen in Fig. 5, enhanced GFAP immunoreactivity was detected in sections of the lateroventral lumbar spinal cords of HB9:Cre–Tardbplx/− mice when compared with the controls. Quantification indicated an ∼2-fold increase of GFAP-positive cells in the spinal cords of HB9:Cre–Tardbplx/− mice. Interestingly, microglia activation was also observed, as indicated by the enhanced immunoreactivity of ionized calcium binding adaptor molecule 1 (Iba1) in sections of lateroventral lumbar spinal cords of the HB9:Cre–Tardbplx/− mice as compared with controls (supplemental Fig. S4).
FIGURE 5.
Reactive astrocytosis in the spinal cords of HB9:Cre–Tardbplx/− mice. Multiple sections of the ventral spinal cords from 20-week-old HB9:Cre–Tardbplx/− mice and control littermates were immunofluorescence stained with anti-GFAP and anti-MAP2, a marker of neurons. The patterns of one representative section each from the two groups of mice are shown. DAPI stainings indicate the locations of the nuclei. Note the enhanced level of anti-GFAP fluorescence in the mutant mice (lower panels) when compared with the control mice (upper panels). The scale bars are 50 μm. Below, quantitative analysis showing an ∼2-fold increase of the area occupied by the GFAP-positive cells within a specific AOI (area of interest) in the HB9:Cre–Tardbplx/− mice. Data are presented as mean ± S.E. **, p < 0.001, n = 4 for each group.
Accumulation of Ubiquitinated Proteins in the Spinal Cord Motor Neurons with Elimination of TDP-43 Expression
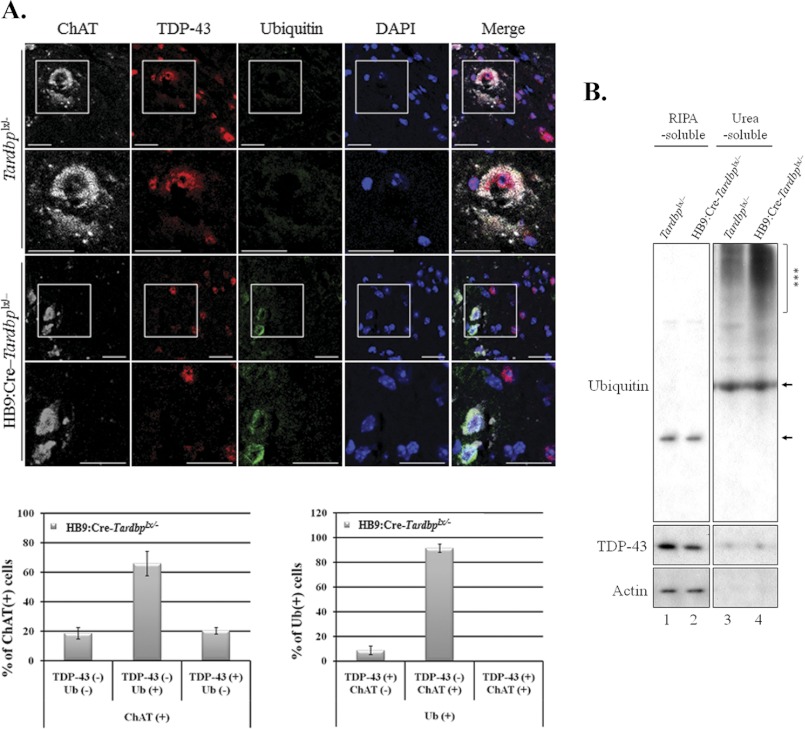
One hallmark of the diseased cells of TDP-43 proteinopathies was the accumulation of polyubiquitinated proteins, including TDP-43 itself, in the cytoplasm (29). To see if polyubiquitinated proteins would also accumulate, in the absence of TDP-43 in the spinal cord motor neurons of HB9:Cre-Tardbplx/− mice, we carried out immunofluorescence staining experiments. When anti-ChAT and anti-ubiquitin were used for co-staining, strong anti-ubiquitin signals were observed in the lumbar spinal cord cells of the HB9:Cre–Tardbplx/− mice but not in the control mice, as exemplified in Fig. 6A (Fig. 6A, compare the lower 2 rows to the upper 2 rows). Furthermore, when co-staining experiments were carried out with anti-ChAT, anti-TDP-43, and anti-ubiquitin, it was found that the cytoplasmic ubiquitin-positive deposits detected in spinal cords of 20-week-old HB9:Cre-Tardbplx/− mice were mostly present in ChAT(+)/TDP-43(−) cells (Fig. 6A), suggesting the accumulation of polyubiquitinated proteins in the spinal cord motor neurons with elimination of TDP-43. On the other hand, the accumulation of the immunofluorescent ubiquitin(+) signals was not observed in the spinal cord motor neurons of 10-week-old mice, either the control or the HB9:Cre-Tardbplx/− mice (data not shown).
FIGURE 6.
Accumulation of ubiquitinated proteins in the spinal cord motor neurons of HB9:Cre–Tardbplx/− mice. A, the lumbar sections of the spinal cords of 20-week-old control (upper 2 rows of panels) and HB9:Cre–Tardbplx/− (lower 2 rows of panels) mice were immunofluorescence co-stained with anti-ubiquitin (green), anti-TDP-43 (red), and anti-ChAT (white). DAPI (blue) stainings indicate the locations of the nuclei. The patterns of one representative section of the control mice and one representative section of HB9:Cre–Tardbplx/− are shown. The boxes mark the regions of the ventral horn magnified for better visualization of the staining signals. n = 4 for each group. Note that increased anti-ubiquitin signals were present mostly in the cytosol of ChAT(+)/TDP-43(−) cells of the HB9:Cre–Tardbplx/− mice, as exemplified by the two ChAT(+) cells in the lower left corners of the bottom row of panels. Almost all of the ChAT(+)/TDP(+) spinal cord cells of the control mice exhibited low ubiquitin signals, as exemplified by the ChAT(+) cell in the 2nd row from the top. The scale bars are 25 μm. The quantitative comparison is shown by the bar histograms below the photographs. Note that most, although not all, of the TDP-43(−)/ChAT(+) motor neurons of the HB9:Cre-Tardbplx/− mice are Ub(+), and vice versa. Furthermore, separate analysis of the immunofluorescence staining data showed that whereas ∼6% of the total cell population in the spinal cord of the HB9:Cre–Tardbplx/− mice were Ub(+), only 0.4% of the control Tardbplx/− spinal cord cells were Ub(+). B, representative immunoblots of polyubiquitinated proteins in fractionated spinal cord extracts. The total protein extracts were isolated from the spinal cords of Tardbplx/− control and HB9:Cre–Tardbplx/− mice, fractionated into the RIPA- and urea-soluble fractions, and analyzed by immunoblotting with use of anti-ubiquitin (top) and anti-actin (bottom). Note that the increase of high molecular weight ubiquitinated protein species (***) are in the urea-soluble fraction of the HB9:Cre–Tardbplx/− sample in comparison to the control sample. The arrows point to the two nonspecific bands on the anti-ubiquitin blots.
Although ubiquitin(+) cells in the spinal cords of the mutant mice (Fig. 6A) only accounted for a few % of the whole spinal cord cell population, we carried out immunoblotting to see if the increased ubiquitin signals came from polyubiquitinated proteins (Fig. 6B). Interestingly, most of the polyubiquitinated proteins isolated from the spinal cords of either the Tardbplx/− mice (Fig. 6B, lanes 1 and 3) or the HB9:Cre-Tardbplx/− mice (Fig. 6B, lanes 2 and 4) were in the urea-soluble fraction (Fig. 6B, lanes 3 and 4). Furthermore, the amount of polyubiquitinated proteins in the spinal cords of the HB9:Cre-Tardbplx/− mice was severalfold higher than that of the Tardbplx/− mice (compare Fig. 6B, lanes 3 and 4). The data of Fig. 6B suggested that most of the accumulated ubiquitin signals detected in spinal cord motor neurons of HB9:Cre-Tardbplx/− mice (Fig. 6A) came from relatively insoluble, polyubiquitinated proteins. The data of Fig. 6 demonstrated that HB9-directed elimination of TDP-43 expression in mouse spinal cords could lead to the accumulation of polyubiquitinated proteins at the symptom onset stage but not the pre-symptom stage.
DISCUSSION
The results described above altogether indicated that the spinal cords, in particular the ventral horn of the lumbar spinal cord, of mutant HB9:Cre-Tardbplx/− mice underwent significant motor neuron loss, reactive astrocytosis, microglia activation, and accumulation of polyubiquitinated proteins, all of which were characteristic of ALS with TDP-43(+) UBIs. Because these HB9:Cre-Tardbplx/− mice are the first mouse model with targeted depletion of TDP-43 expression in the spinal cord motor neurons and 90% of the non-SOD1, non-FUS-inclusion type ALS patients are signatures with TDP-43(+) UBIs, this mouse model we have generated should be valuable for research of ALS with TDP-43 proteinopathies.
Like other neurodegenerative diseases with inclusion formation during pathogenesis, e.g. Alzheimer disease, Huntington disease, Parkinson disease, etc. (50), neurodegeneration in patients with the TDP-43(+) UBIs has also been suggested to be the result of loss of functions of TDP-43, cytotoxicity of the TDP-43(+) UBIs per se, or a combination of both (29, 31). In vivo evidence supporting the first scenario includes observations of depletion of the nuclear TDP-43 in diseased cells of ALS and FTLD-U patients with TDP-43(+) UBIs (14, 15) and the loss of motor function (47, 51) or learning ability (47) of Drosophila with knockdown of TDP-43. On the other hand, current evidence supporting the second scenario is mainly by association, which includes the cytotoxicity observed in cell cultures upon overexpression of the carboxyl-terminal 25-kDa fragment to form cytoplasmic TDP-43(+) UBIs (52) and development of FTLD-U-like symptoms or motor-neuron degeneration in transgenic mice with overexpression of TDP-43 accompanied with formation of TDP-43(+) UBI in vivo (24–26, 28, 53–55). With respective to the above, the effects of the restricted knock-out of TDP-43 expression in spinal cord motor neurons of HB9:Cre–Tardbplx/− mice on the development of a range of ALS-like phenotypes in these mice has demonstrated an essential role of TDP-43 in the survival and functioning of the mammalian spinal cord motor neurons. Furthermore, it also provides direct evidence that loss of functions of TDP-43 in spinal cord motor neurons, and perhaps in a minor fraction of other types of cells as well, is sufficient and thus could be one major cause leading to the pathogenesis of ALS with TDP-43(+) UBIs.
The molecular and cellular basis of how loss of function of TDP-43 would lead to the development of TDP-43 proteinopathies is currently unknown and may take awhile to be fully understood in view of the multifunction nature of TDP-43. Notably, loss of the spinal cord motor neurons, albeit small, already could be detected at the presymptomatic stage of the HB9:Cre-Tardbplx/− mice (Fig. 4, upper panel). Because HB9-directed expression of Cre was as early as E9.5, this result suggested that TDP-43 is not required for biogenesis of the spinal cord motor neurons during early development. However, the maintenance/survival of the spinal cord motor neurons of the adult mice does involve TDP-43, the long term lack of which would lead to the progressive loss of these neurons. Finally, it is especially interesting to note that accumulation of ubiquitinated proteins could still occur in the cytosol of the spinal cord motor neurons with elimination of the TDP-43 expression of the HB9:Cre–Tardbplx/− mice (Fig. 6, A and B).
These ubiquitin signals, however, are not present as inclusions or aggregates as seen in the diseased samples of ALS (14, 15). Although, they do appear to be elevated from polyubiquitinated proteins in immunoblotting analysis (Fig. 6B). In any case, we suggest that accumulation of polyubiquitinated proteins in the diseased cells of TDP-43 proteinopathies might be independent of the formation of TDP-43(+) UBIs. It is likely that the initial loss of function of TDP-43, as the result of mutations in some of the ALS-associated disease genes, including TDP-43 itself, may cause impairment of the proteasome system and/or autophagy thus leading to accumulation of polyubiquitinated proteins in the diseased cells. Interestingly, this scenario is supported by a recent study showing that TDP-43 is required for the maintenance of a functional autophagy in rodent cells (56). Upon depletion of the mouse TDP-43 and the consequent destabilization of the ATG7 mRNA, the autophagy is impaired and ubiquitinated proteins accumulate in the cytosol (56), in a strikingly similar pattern as observed in Fig. 6. In the presence of TDP-43, these polyubiquitinated proteins then could form the TDP-43(+) UBIs with TDP-43 (57, 58). To what extent and how generally applicable this proposed scenario is to the ALS cases with TDP-43(+) proteinopathies awaits to be validated in the future.
Supplementary Material
Acknowledgments
We thank our colleagues and Sue-Ping Lee at the IMB Confocal Microscope Core for help and suggestions. The Tardbp gene knock-out experiments were done with help from the Transgenic Core Facility (TCF) of the Institute of Molecular Biology (IMB), Academia Sinica.
This work was supported by a Frontiers of Science Award from the National Science Council and the Academia Sinica (AS), Taipei Taiwan.

This article contains supplemental Figs. S1–S4.
- ALS
- amyotrophic lateral sclerosis
- ChAT
- choline acetyltransferase
- GFAP
- glial fibrillary acidic protein.
REFERENCES
- 1. Mulder D. W. (1982) Clinical limits of amyotrophic lateral sclerosis. Adv. Neurol. 36, 15–22 [PubMed] [Google Scholar]
- 2. Pasinelli P., Brown R. H. (2006) Molecular biology of amyotrophic lateral sclerosis. Insights from genetics. Nat. Rev. Neurosci. 7, 710–723 [DOI] [PubMed] [Google Scholar]
- 3. Wijesekera L. C., Leigh P. N. (2009) Amyotrophic lateral sclerosis. Orphanet J. Rare Dis. 4, 1–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Johnston C. A., Stanton B. R., Turner M. R., Gray R., Blunt A. H., Butt D., Ampong M. A., Shaw C. E., Leigh P. N., Al-Chalabi A. (2006) Amyotrophic lateral sclerosis in an urban setting. A population based study of inner city London. J. Neurol. 253, 1642–1643 [DOI] [PubMed] [Google Scholar]
- 5. Manjaly Z. R., Scott K. M., Abhinav K., Wijesekera L., Ganesalingam J., Goldstein L. H., Janssen A., Dougherty A., Willey E., Stanton B. R., Turner M. R., Ampong M. A., Sakel M., Orrell R. W., Howard R., Shaw C. E., Leigh P. N., Al-Chalabi A. (2010) The sex ratio in amyotrophic lateral sclerosis. A population based study. Amyotroph. Lateral Scler. 11, 439–442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Shaw P. J. (2005) Molecular and cellular pathways of neurodegeneration in motor neurone disease. J. Neurol. Neurosurg. Psychiatry 76, 1046–1057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sreedharan J., Blair I. P., Tripathi V. B., Hu X., Vance C., Rogelj B., Ackerley S., Durnall J. C., Williams K. L., Buratti E., Baralle F., de Belleroche J., Mitchell J. D., Leigh P. N., Al-Chalabi A., Miller C. C., Nicholson G., Shaw C. E. (2008) TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis. Science 319, 1668–1672 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Lagier-Tourenne C., Cleveland D. W. (2009) Rethinking ALS. The FUS about TDP-43. Cell 136, 1001–1004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Mackenzie I. R., Rademakers R., Neumann M. (2010) TDP-43 and FUS in amyotrophic lateral sclerosis and frontotemporal dementia. Lancet Neurol. 9, 995–1007 [DOI] [PubMed] [Google Scholar]
- 10. Ou S. H., Wu F., Harrich D., García-Martínez L. F., Gaynor R. B. (1995) Cloning and characterization of a novel cellular protein, TDP-43, that binds to human immunodeficiency virus type 1 TAR DNA sequence motifs. J. Virol. 69, 3584–3596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Wang H. Y., Wang I. F., Bose J., Shen C. K. (2004) Structural diversity and functional implications of the eukaryotic TDP gene family. Genomics 83, 130–139 [DOI] [PubMed] [Google Scholar]
- 12. Buratti E., Baralle F. E. (2008) Multiple roles of TDP-43 in gene expression, splicing regulation, and human disease. Front. Biosci. 13, 867–878 [DOI] [PubMed] [Google Scholar]
- 13. Wang I. F., Wu L. S., Chang H. Y., Shen C. K. (2008) TDP-43, the signature protein of FTLD-U, is a neuronal activity-responsive factor. J. Neurochem. 105, 797–806 [DOI] [PubMed] [Google Scholar]
- 14. Arai T., Hasegawa M., Akiyama H., Ikeda K., Nonaka T., Mori H., Mann D., Tsuchiya K., Yoshida M., Hashizume Y., Oda T. (2006) TDP-43 is a component of ubiquitin-positive Tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem. Biophys. Res. Commun. 351, 602–611 [DOI] [PubMed] [Google Scholar]
- 15. Neumann M., Sampathu D. M., Kwong L. K., Truax A. C., Micsenyi M. C., Chou T. T., Bruce J., Schuck T., Grossman M., Clark C. M., McCluskey L. F., Miller B. L., Masliah E., Mackenzie I. R., Feldman H., Feiden W., Kretzschmar H. A., Trojanowski J. Q., Lee V. M. (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314, 130–133 [DOI] [PubMed] [Google Scholar]
- 16. Forman M. S., Trojanowski J. Q., Lee V. M. (2007) TDP-43, a novel neurodegenerative proteinopathy. Curr. Opin. Neurobiol. 17, 548–555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Neumann M. (2009) Molecular neuropathology of TDP-43 proteinopathies. Int. J. Mol. Sci. 10, 232–246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Neumann M., Kwong L. K., Sampathu D. M., Trojanowski J. Q., Lee V. M. (2007) TDP-43 proteinopathy in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Protein misfolding diseases without amyloidosis. Arch. Neurol. 64, 1388–1394 [DOI] [PubMed] [Google Scholar]
- 19. Kato S. (2008) Amyotrophic lateral sclerosis models and human neuropathology. Similarities and differences. Acta Neuropathol. 115, 97–114 [DOI] [PubMed] [Google Scholar]
- 20. Turner B. J., Talbot K. (2008) Transgenics, toxicity and therapeutics in rodent models of mutant SOD1-mediated familial ALS. Prog. Neurobiol. 85, 94–134 [DOI] [PubMed] [Google Scholar]
- 21. Gurney M. E., Pu H., Chiu A. Y., Dal Canto M. C., Polchow C. Y., Alexander D. D., Caliendo J., Hentati A., Kwon Y. W., Deng H. X. (1994) Motor neuron degeneration in mice that express a human Cu,Zn-superoxide dismutase mutation. Science 264, 1772–1775 [DOI] [PubMed] [Google Scholar]
- 22. Gurney M. E., Cutting F. B., Zhai P., Andrus P. K., Hall E. D. (1996) Pathogenic mechanisms in familial amyotrophic lateral sclerosis due to mutation of Cu,Zn-superoxide dismutase. Pathol. Biol. 44, 51–56 [PubMed] [Google Scholar]
- 23. Kato S., Kato M., Abe Y., Matsumura T., Nishino T., Aoki M., Itoyama Y., Asayama K., Awaya A., Hirano A., Ohama E. (2005) Redox system expression in the motor neurons in amyotrophic lateral sclerosis (ALS). Immunohistochemical studies on sporadic ALS, superoxide dismutase 1 (SOD1)-mutated familial ALS, and SOD1-mutated ALS animal models. Acta Neuropathol. 110, 101–112 [DOI] [PubMed] [Google Scholar]
- 24. Wegorzewska I., Bell S., Cairns N. J., Miller T. M., Baloh R. H. (2009) TDP-43 mutant transgenic mice develop features of ALS and frontotemporal lobar degeneration. Proc. Natl. Acad. Sci. U.S.A. 106, 18809–18814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Tsai K. J., Yang C. H., Fang Y. H., Cho K. H., Chien W. L., Wang W. T., Wu T. W., Lin C. P., Fu W. M., Shen C. K. (2010) Elevated expression of TDP-43 in the forebrain of mice is sufficient to cause neurological and pathological phenotypes mimicking FTLD-U. J. Exp. Med. 207, 1661–1673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Wils H., Kleinberger G., Janssens J., Pereson S., Joris G., Cuijt I., Smits V., Ceuterick-de Groote C., Van Broeckhoven C., Kumar-Singh S. (2010) TDP-43 transgenic mice develop spastic paralysis and neuronal inclusions characteristic of ALS and frontotemporal lobar degeneration. Proc. Natl. Acad. Sci. U.S.A. 107, 3858–3863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Zhou H., Huang C., Chen H., Wang D., Landel C. P., Xia P. Y., Bowser R., Liu Y. J., Xia X. G. (2010) Transgenic rat model of neurodegeneration caused by mutation in the TDP gene. PLoS Genet. 6, e1000887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Igaz L. M., Kwong L. K., Lee E. B., Chen-Plotkin A., Swanson E., Unger T., Malunda J., Xu Y., Winton M. J., Trojanowski J. Q., Lee V. M. (2011) Dysregulation of the ALS-associated gene TDP-43 leads to neuronal death and degeneration in mice. J. Clin. Invest. 121, 726–738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wang I. F., Wu L. S., Shen C. K. (2008) TDP-43. An emerging new player in neurodegenerative diseases. Trends Mol. Med. 14, 479–485 [DOI] [PubMed] [Google Scholar]
- 30. Buratti E., Baralle F. E. (2009) The molecular links between TDP-43 dysfunction and neurodegeneration. Adv. Genet. 66, 1–34 [DOI] [PubMed] [Google Scholar]
- 31. Chen-Plotkin A. S., Lee V. M., Trojanowski J. Q. (2010) Nat. Rev. 6, 211–220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Wu L. S., Cheng W. C., Hou S. C., Yan Y. T., Jiang S. T., Shen C. K. (2010) TDP-43, a neuropathosignature factor, is essential for early mouse embryogenesis. Genesis 48, 56–62 [DOI] [PubMed] [Google Scholar]
- 33. Yang X., Arber S., William C., Li L., Tanabe Y., Jessell T. M., Birchmeier C., Burden S. J. (2001) Patterning of muscle acetylcholine receptor gene expression in the absence of motor innervation. Neuron 30, 399–410 [DOI] [PubMed] [Google Scholar]
- 34. Arber S., Han B., Mendelsohn M., Smith M., Jessell T. M., Sockanathan S. (1999) Requirement for the homeobox gene Hb9 in the consolidation of motor neuron identity. Neuron 23, 659–674 [DOI] [PubMed] [Google Scholar]
- 35. Kramer E. R., Knott L., Su F., Dessaud E., Krull C. E., Helmbacher F., Klein R. (2006) Cooperation between GDNF/Ret and ephrinA/EphA4 signals for motor-axon pathway selection in the limb. Neuron 50, 35–47 [DOI] [PubMed] [Google Scholar]
- 36. Hess D. M., Scott M. O., Potluri S., Pitts E. V., Cisterni C., Balice-Gordon R. J. (2007) Localization of TrkC to Schwann cells and effects of neurotrophin-3 signaling at neuromuscular synapses. J. Comp. Neurol. 501, 465–482 [DOI] [PubMed] [Google Scholar]
- 37. Li X. M., Dong X. P., Luo S. W., Zhang B., Lee D. H., Ting A. K., Neiswender H., Kim C. H., Carpenter-Hyland E., Gao T. M., Xiong W. C., Mei L. (2008) Retrograde regulation of motoneuron differentiation by muscle β-catenin. Nat. Neurosci. 11, 262–268 [DOI] [PubMed] [Google Scholar]
- 38. Cheever T. R., Olson E. A., Ervasti J. M. (2011) Axonal regeneration and neuronal function are preserved in motor neurons lacking β-actin in vivo. PLoS One 6, e17768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Luria V., Laufer E. (2007) Lateral motor column axons execute a ternary trajectory choice between limb and body tissues. Neural. Dev. 2, 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Zhou C., Zhao C. P., Zhang C., Wu G. Y., Xiong F. (2007) A method comparison in monitoring disease progression of G93A mouse model of ALS. Amyotroph. Lateral Scler. 8, 366–372 [DOI] [PubMed] [Google Scholar]
- 41. Newman D. S., Aggarwal S. K., Silbergleit R. (1995) Thoracic radicular symptoms in amyotrophic lateral sclerosis. J. Neurol. Sci. 129, 38–41 [DOI] [PubMed] [Google Scholar]
- 42. Azzouz M., Leclerc N., Gurney M., Warter J. M., Poindron P., Borg J. (1997) Progressive motor neuron impairment in an animal model of familial amyotrophic lateral sclerosis. Muscle Nerve 20, 45–51 [DOI] [PubMed] [Google Scholar]
- 43. Burke R. E., Strick P. L., Kanda K., Kim C. C., Walmsley B. (1977) Anatomy of medial gastrocnemius and soleus motor nuclei in cat spinal cord. J. Neurophysiol 40, 667–680 [DOI] [PubMed] [Google Scholar]
- 44. Friese A., Kaltschmidt J. A., Ladle D. R., Sigrist M., Jessell T. M., Arber S. (2009) γ and α motor neurons distinguished by expression of transcription factor Err3. Proc. Natl. Acad. Sci. U.S.A. 106, 13588–13593 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Shneider N. A., Brown M. N., Smith C. A., Pickel J., Alvarez F. J. (2009) γ Motor neurons express distinct genetic markers at birth and require muscle spindle-derived GDNF for postnatal survival. Neural. Dev. 4, 42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Hudson A. J., Kiernan J. A., Munoz D. G., Pringle C. E., Brown W. F., Ebers G. C. (1993) Clinicopathological features of primary lateral sclerosis are different from amyotrophic lateral sclerosis. Brain Res. Bull. 30, 359–364 [DOI] [PubMed] [Google Scholar]
- 47. Lin M. J., Cheng C. W., Shen C. K. (2011) Neuronal function and dysfunction of Drosophila dTDP. PLoS One 6, e20371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Nguyen M. D., Larivière R. C., Julien J. P. (2000) Reduction of axonal caliber does not alleviate motor neuron disease caused by mutant superoxide dismutase 1. Proc. Natl. Acad. Sci. U.S.A. 97, 12306–12311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Hall E. D., Oostveen J. A., Gurney M. E. (1998) Relationship of microglial and astrocytic activation to disease onset and progression in a transgenic model of familial ALS. Glia 23, 249–256 [DOI] [PubMed] [Google Scholar]
- 50. Ross C. A., Poirier M. A. (2005) Opinion. What is the role of protein aggregation in neurodegeneration? Nat. Rev. Mol. Cell Biol. 6, 891–898 [DOI] [PubMed] [Google Scholar]
- 51. Feiguin F., Godena V. K., Romano G., D'Ambrogio A., Klima R., Baralle F. E. (2009) Depletion of TDP-43 affects Drosophila motoneurons terminal synapsis and locomotive behavior. FEBS Lett. 583, 1586–1592 [DOI] [PubMed] [Google Scholar]
- 52. Yang C., Tan W., Whittle C., Qiu L., Cao L., Akbarian S., Xu Z. (2010) The C-terminal TDP-43 fragments have a high aggregation propensity and harm neurons by a dominant-negative mechanism. PLoS One 5, e15878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Shan X., Chiang P. M., Price D. L., Wong P. C. (2010) Altered distributions of Gemini of coiled bodies and mitochondria in motor neurons of TDP-43 transgenic mice. Proc. Natl. Acad. Sci. U.S.A. 107, 16325–16330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Stallings N. R., Puttaparthi K., Luther C. M., Burns D. K., Elliott J. L. (2010) Progressive motor weakness in transgenic mice expressing human TDP-43. Neurobiol. Dis. 40, 404–414 [DOI] [PubMed] [Google Scholar]
- 55. Xu Y. F., Gendron T. F., Zhang Y. J., Lin W. L., D'Alton S., Sheng H., Casey M. C., Tong J., Knight J., Yu X., Rademakers R., Boylan K., Hutton M., McGowan E., Dickson D. W., Lewis J., Petrucelli L. (2010) Wild-type human TDP-43 expression causes TDP-43 phosphorylation, mitochondrial aggregation, motor deficits, and early mortality in transgenic mice. J. Neurosci. 30, 10851–10859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Bose J. K., Huang C. C., Shen C. K. (2011) Regulation of autophagy by neuropathological protein TDP-43. J. Biol. Chem. 286, 44441–44448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Pesiridis G. S., Tripathy K., Tanik S., Trojanowski J. Q., Lee V. M. (2011) A ”two-hit“ hypothesis for inclusion formation by carboxyl-terminal fragments of TDP-43 protein linked to RNA depletion and impaired microtubule-dependent transport. J. Biol. Chem. 286, 18845–18855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Furukawa Y., Kaneko K., Watanabe S., Yamanaka K., Nukina N. (2011) A seeding reaction recapitulates intracellular formation of Sarkosyl-insoluble transactivation response element (TAR) DNA-binding protein-43 inclusions. J. Biol. Chem. 286, 18664–18672 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.