Abstract
Heterotopic ossification (HO) is a process by which bone forms in soft tissues, in response to injury, inflammation or genetic disease. This usually occurs by initial cartilage formation, followed by endochondral ossification. A rare disease called Fibrodysplasia Ossificans Progressiva (FOP) allows this mechanism to be induced by a combination of genetic mutation and acute inflammatory responses. FOP patients experience progressive HO throughout their lifetime and form an ectopic skeleton. Recent studies on FOP have suggested that heterotopic cartilage and bone is of endothelial origin. Vascular endothelial cells differentiate into skeletal cells through a mesenchymal stem cell intermediate that is generated by endothelial-mesenchymal transition (EndMT). Local inflammatory signals and/or other changes in the tissue microenvironment mediate the differentiation of endothelial-derived mesenchymal stem cells into chondrocytes and osteoblasts to induce HO. Here we discuss the current evidence for the endothelial contribution to heterotopic bone formation.
Keywords: endothelial-mesenchymal transition, EndMT, EMT, heterotopic ossification, fibrodysplasia ossificans progressiva, FOP, BMP, TGF-beta, stem cells
Introduction
Heterotopic ossification (HO) is a debilitating condition common in cases of inflammation associated with severe injury, surgery, or disease.(1–3) HO most commonly occurs in muscle, tendons and ligaments,(1,2) although it is unclear as to why these specific tissue environments foster the formation of ectopic bone.
Perhaps the most extreme cases of HO can be found in patients with fibrodyplasia ossificans progressiva (FOP), a genetic disorder that causes progressive HO throughout the patients’ lifetime, rendering them immobile.(4) These patients experience periodic episodes of acute inflammatory processes that cause tumor-like swellings in muscle tissue, which lead to the formation of cartilage and bone in the inflamed region.(4) Until recently, little was know regarding the molecular and cellular mechanisms that mediate this process.
A genetic mutation that causes FOP was first reported by Eileen Shore and colleagues in 2006.(5) Patients with FOP carry an autosomal dominant heterozygous germ-line mutation in the ALK2 (also known as ACVR1) gene.(5) Activin-like kinase 2 (ALK2) is a transforming growth factor-beta (TGF-β)/bone morphogenetic protein (BMP) type I receptor known to be activated by cytokines such as BMP2, BMP4, BMP7 and TGF-β2 in various cell types.(6) The FOP mutation causes an argenine to histidine change in amino acid 206 (R206H) in the ALK2 protein.(5) Studies have shown that this mutation, found in a glycine-serine rich region, causes the receptor to be constitutively active.(7) Constitutive phosphorylation of the mutant ALK2 receptor has been reported, as well as phosphorylation of downstream Smad proteins.(7–9)
Several studies have been conducted in order to identify the cellular origin of the ectopic bone in FOP. The logical candidate would be local tissue mesenchymal stem cells stem cells or some other progenitors recruited to the lesions from bone marrow. However, bone marrow transplant studies have been shown to be ineffective in perturbing HO in mice, effectively ruling out a role for hematopoietic cells or bone marrow progenitors.(10,11)
More recently, vascular endothelial cells have emerged as the leading candidate for the cellular origin of heterotopic cartilage and bone in FOP. Approximately 50% of the cartilage and bone cells found in heterotopic lesions appear to be of endothelial origin based on Tie2-Cre lineage tracing and expression of various endothelial markers (Tie1, Tie2, vWF, VE-cadherin) in these cells.(9,12) It is still unclear as to what other cell types might give rise to the remaining 50% of the total cellular population. Lineage tracing with MyoD-Cre reporter mice showed a minimal contribution (<5%) of skeletal muscle cells to HO.(12) Smooth Muscle Myosin Heavy Chain-Cre mice showed no significant contribution of vascular smooth muscle cells in giving rise to heterotopic bone.(12) Other potential candidates could be pericytes, which have been show to have osteogenic potential,(13) or resident stem cells within muscle tissue. A recent study has suggested that such resident stem cells within the skeletal muscle interstitium may have a role in forming heterotopic bone.(14) Here we will focus our discussion on the endothelial contribution to HO.
Endothelial-mesenchymal transition (EndMT)
EndMT is characterized by loss of cell-cell adhesion and a strong change in cell polarity, generating elongated spindle-shaped cells. Expression of endothelial markers such as CD31, VE-cadherin, Tie1, and vWF is reduced, while mesenchymal markers such as FSP-1, α-SMA, N-cadherin, and vimentin increase. These newly formed mesenchymal cells are highly invasive and motile, and give rise to various tissue types in embryonic development and disease.(15,16)
EndMT was initially discovered as an essential embryonic mechanism for cardiac development.(17) Endothelial cells in the atrioventricular canal and the outflow tract undergo EndMT and invade the underlying tissue to form the valves and septa of the heart.(17,18) More recently, EndMT has been shown to have essential roles in pathological conditions such as fibrosis of the heart,(19) kidney,(20) and lung.(21) EndMT also occurs in tumors to cause formation of cancer-associated fibroblasts (CAFs) as part of the tumor stroma, which regulates cancer progression.(22) EndMT has also been shown to have a role in formation of scar tissue during wound healing.(23)
Until recently, EndMT was thought to be a process of endothelial cells turning into fibroblasts, and that this fibroblast phenotype was the ultimate fate of these cells. However, new studies have shown that EndMT represents a dedifferentiation of endothelial cells to a stem cell phenotype, which can subsequently redifferentiate into bone, cartilage or fat cells.(9) These studies suggest that in FOP, heterotopic cartilage and bone arises from such stem cells derived from vascular endothelial cells via EndMT.(9) The results of two other recent studies indicate that the vascular endothelium in adipose tissue can give rise to white and brown fat cells.(24,25) It would not be surprising if these insights lead to the discovery of other cellular lineages derived from endothelium in developmental and pathological systems. Such studies may lead to novel therapeutic strategies to inhibit EndMT-mediated cell differentiation in conditions such as HO.
The role of EndMT in heterotopic ossification
Studies in mouse models of HO induced by recombinant BMP2, BMP4 or transgenic expression of mutant ALK2 show that ectopic cartilage and bone cells express endothelial biomarkers such as Tie1, Tie2, vWF, and VE-cadherin.(9,12) Similar results were observed in pathologic specimens from FOP patients.(9) Osteoblasts and chondrocytes from normal bone or cartilage tissue do not express these endothelial markers.(9) Furthermore lineage tracing in Tie2-Cre reporter mice have demonstrated that chondrocytes and osteoblasts in heterotopic lesions express the reporter (enhanced green fluorescent protein or lacZ),(9,12) strongly suggesting that they are of endothelial origin.
Over-expressing ALK2 carrying the FOP mutation (R206H) in mature vascular endothelial cells in culture induced endothelial-mesenchymal transition (EndMT).(9) The endothelial cells dramatically changed their morphology from round, cobblestone-like cells to elongated, spindle shaped cells characteristic of mesenchyme. Furthermore, the cells showed reduced expression of endothelial markers and gained expression of mesenchymal markers. Notably, in the early stages of HO, a mesenchymal condensation forms in the lesions prior to chondrogenesis and endochondral ossification. Studies of Tie2-Cre reporter mice showed that most of the mesenchymal cells in these lesions were of endothelial origin.(9,12)
Endothelial cells expressing the mutant ALK2 gene showed positive expression for mesenchymal stem cell markers such as STRO-1, CD10, CD44, CD71, CD90, and CD117. Moreover, the cells demonstrated multipotency in culture and were easily differentiated into ostoblasts, chondrocytes or adipocytes when exposed to the appropriate differentiation media.(9) In vivo, Tie2-Cre lineage tracing suggests that this EndMT-mediated acquisition of stem cell characteristics promotes the differentiation of endothelium into the heterotopic skeletal cells. Although Tie2 is also expressed in hematopoietic cells, heterotopic cartilage and bone cells also express other endothelial markers such as vWF and VE-cadherin(9,12), which were found to not be expressed in hematopoietic stem cells.(9) Furthermore, bone marrow transplant studies have suggested no significant role for hematopoietic cells in HO.(10,11)
A recent study has argued against the role of endothelial cells in BMP2-dependent HO, suggesting that Tie2-positive resident mesenchymal stem cells in the skeletal muscle interstitium give rise to the cartilage and bone cells.(14) This study has shown that CD31-negative mesenchymal stem cells in the muscle tissue can generate cartilage and bone, yet CD31 usually disappears during EndMT, so it is unclear as to whether these cells could have arisen from endothelial cells based on this marker. VE-cadherin-Cre mice were used to analyze HO induced by BMP2/matrilgel injection into the muscle tissue. The data showed no expression of the GFP reporter in heterotopic cartilage and bone cells.(14) However, in the same BMP2-induced HO model, another study has shown positive expression of VE-cadherin protein in the heterotopic bone and cartilage cells.(12) Similar expression of VE-cadherin protein in ectopic skeletal cells was found in a BMP4-induced model of HO.(9) Both studies also showed expression of other endothelial markers in these cells such as Tie1 or vWF.(9,12) If VE-cadherin gene expression is sufficient to give rise to the protein detected in heterotopic skeletal cells,(9,12) it is unclear as to why VE-cadherin-Cre-dependent recombination and GFP reporter expression was not detected.(14)
It is also possible that endothelial progenitor cells (EPCs) might contribute to HO in FOP, rather than mature endothelium from existing blood vessels. EPCs express the same markers, albeit at lower levels than in mature endothelial cells.(26) Studies have shown that circulating EPCs can undergo EndMT and give rise to smooth muscle cell progeny during angiogenesis.(27) So, the possibility exists that circulating EPCs could be recruited to early FOP lesions as part of the inflammatory response and via EndMT contribute to the formation of heterotopic bone.
Therapeutic strategies for inhibtion of EndMT-mediated heterotopic ossification
The ALK2 mutations in FOP, including the most common mutation R206H, are gain of function mutations.(4,7) Therefore it is not surprising that in the absence of mutation, ALK2 activating cytokines such as BMP4 or TGF-β2 can stimulate EndMT and the endothelial acquisition of stem cell phenotype.(9) The ALK2 ligands BMP2 and BMP4 have been show to induce HO when mixed with matrigel and injected into skeletal muscle in mice.(9,12) In contrast, siRNA-mediated knockdown of ALK2 expression has been shown to block EndMT induced by BMP4 or TGF-β2.(9) Dorsomorphin, a chemical inhibitor of ALK2, also inhibits EndMT.(9) Dorsomorphin and a similar ALK2 inhibitor known as LDN-193189 can both successfully inhibit HO in mice.(28)
Like for ALK2, siRNA-mediated knockdown of ALK5 was also able to inhibit EndMT.(9) ALK2 signals through Smad1/5/8, whereas ALK5 signals through Smad2/3,(29) so it was surprising that both receptors are required for the process. Immunoprecipitation experiments demonstrated an interaction between ALK5 and ALK2 as a result of activation by the R206H ALK2 mutation or by the ligands BMP4 or TGF-β2.(9) Other studies have also shown that inhibition of ALK5 can prevent EndMT of endothelial progenitor cells.(27)
Interestingly, the ALK2 activating ligand BMP7 is a well-know inhibitor of EndMT.(9,19,20) Although it is not entirely clear why this should be so, treatment of endothelial cells with recombinant BMP7 showed no interaction of ALK2 with ALK5. Furthermore, only activation of the Smad1/5/8 pathway was observed with BMP7.(9) These findings suggest that there is ligand specificity for ALK2-dependent stimulation of EndMT.
EndMT requires both Smad-dependent and Smad-independent signals to induce the transformation.(30) Expression of a dominant negative Smad4 protein has been shown to block EndMT induced by TGF-β2. Likewise, chemical inhibitors of PI3K, MEK and p38 MAPK pathways successfully blocked TGF-β2-induced EndMT.(30) Therefore, it is likely that inhibiting ALK2, ALK5 or several of their downstream signaling pathways may prove beneficial in preventing EndMT-dependent HO.
Another potential candidate for therapeutic inhibition of HO is the EndMT-inducing transcription factor Snail, which binds to the promoter of genes associated with cell-cell adhesion and represses their transcription.(31) siRNA-mediated knockdown of Snail expression has been shown to inhibit TGF-β2-induced EndMT. Over-expression of Snail was insufficient to induce EndMT, but blocking the function of the Snail inhibitor GSK-3β with lithium chloride allowed EndMT to occur by Snail over-expression.(30)
Vascular endothelial growth factor (VEGF) is another known inhibitor of EndMT, as it has been shown to counteract the effects of TGF-β2 signaling.(9,32) It is unclear what the downstream effectors of VEGF are in this context, but these studies suggest that recombinant VEGF could be a potential inhibitor of HO.
FOP patients show no symptoms of the disease until they experience inflammation,(4) yet it is unclear as to how inflammatory responses trigger EndMT. Recent studies have suggested that neuronal activity may be important.(33,34) TRPV1 null mice, which lack sensory neurons exhibit dramatically reduced BMP2-induced HO.(33) BMP2 has been shown to promote release of substance P and calcitonin gene related peptide (CGRP), proteins known to induce recruitment of mast cells as part of a neuroinflammatory response. Inhibiting mast cell degranulation with cromolyn prevents BMP2-induced HO in mice.(33) Blocking the substance P receptor NK1r has also been shown to inhibit HO.(34) It is unclear what direct effects these neuroinflammatory responses have on HO, but it is feasible to consider that mast cell degranulation might have a role in regulating EndMT or the differentiation of endothelial-derived stem-like cells into chondrocytes or osteoblasts.
Taken together, these studies suggest that endothelial cells play a critical role generating cartilage and bone in heterotopic lesions through ALK2-dependent EndMT. Activation of ALK2 by ligands or by a gain-of-function mutation stimulates this process, while chemical inhibitors of ALK2 or its downstream signaling pathways can perturb EndMT-dependent cell differentiation (Figure 1). Therapeutic targeting of molecules responsible for inducing EndMT may prove beneficial for preventing the formation of ectopic bone in patients with FOP or inflammation-induced HO.
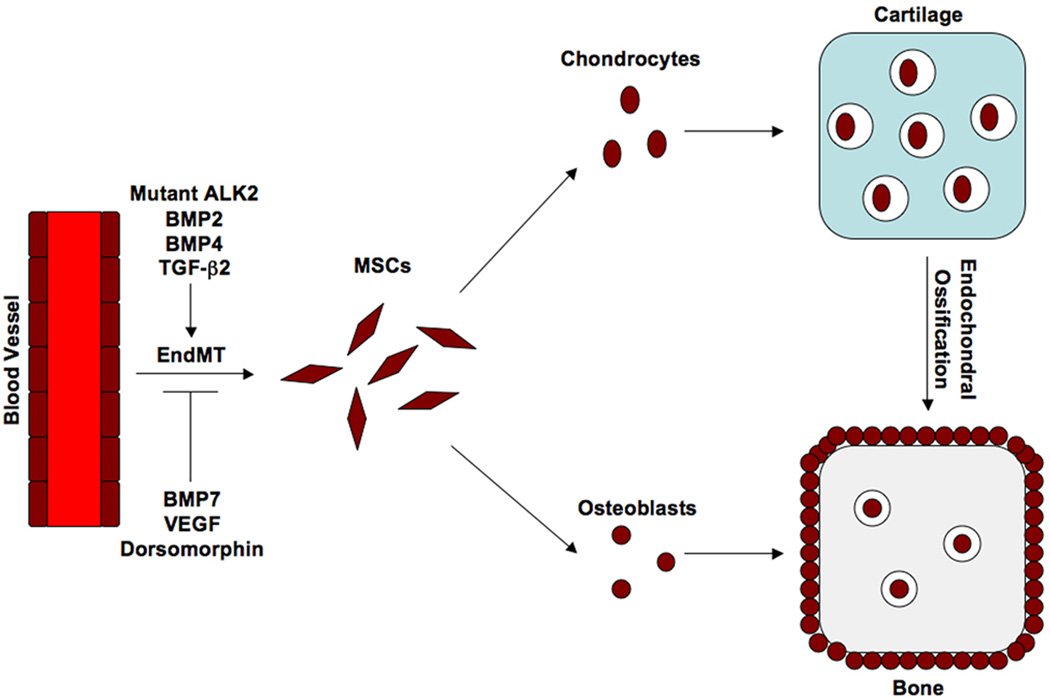
Fig. 1.
A schematic diagram of the proposed mechanism of EndMT-induced heterotopic ossification. Endothelial cells from capillary blood vessels in muscle tissue undergo EndMT in response to inflammation and an activating mutation in ALK2 in patients with FOP. Cytokines such as BMP2, BMP4, or TGF-β2 can mimic the effects of the ALK2 mutation, while proteins such as BMP7 and VEGF, or a chemical inhibitor of ALK2 like dorsomorphin, can prevent EndMT. These endothelial-derived mesenchymal stem-like cells (MSCs) differentiate into chondrocytes and osteoblasts in response to inflammatory signals. Chondrogenesis occurs first, followed by endochondral ossification.
Acknowledgements
This work was supported by grants R01HL112860 (to D. Medici) and P01AR048564 (to B. Olsen) from the National Institutes of Health and a grant from the John Butler Mulliken Foundation (to D. Medici).
Footnotes
Disclosures
The authors state that they have no conflicts of interest.
References
- 1.Kaplan FS, Shen Q, Lounev V, Seemann P, Groppe J, Katagiri T, Pignolo RJ, Shore EM. Skeletal metamorphosis in fibrodysplasia ossificans progressiva (FOP) J Bone Miner Metab. 2008;26:521–530. doi: 10.1007/s00774-008-0879-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Potter BK, Burns TC, Lacap AP, Granville RR, Gajewski DA. Heterotopi ossification following traumatic and combat-related amputations. Prevalence, risk factors, and preliminary results of excision. J Bone Joint Surg Am. 2007;89:476–486. doi: 10.2106/JBJS.F.00412. [DOI] [PubMed] [Google Scholar]
- 3.Dudley AC, Khan ZA, Shih SC, Kang SY, Zwaans BM, Bischoff J, Klagsbrun M. Calcification of multipotent prostate tumor endothelium. Cancer Cell. 2008;14:201–211. doi: 10.1016/j.ccr.2008.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shore EM, Kaplan FS. Insights from a rare genetic disorder of extra-skeletal bone formation, fibrodysplasia ossificans progressiva (FOP) Bone. 2008;43:427–433. doi: 10.1016/j.bone.2008.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shore EM, Xu M, Feldman GJ, Fenstermacher DA, Cho TJ, Choi IH, Connor JM, Delai P, Glaser DL, LeMerrer M, Morhart R, Rogers JG, Smith R, Triffitt JT, Urtizberea JA, Zasloff M, Brown MA, Kaplan FS. A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat Genet. 2006;38:525–527. doi: 10.1038/ng1783. [DOI] [PubMed] [Google Scholar]
- 6.Chen D, Zhao M, Mundy GR. Bone morphogenetic proteins. Growth Factors. 2004;22:233–241. doi: 10.1080/08977190412331279890. [DOI] [PubMed] [Google Scholar]
- 7.Shen Q, Little SC, Xu M, Haupt J, Ast C, Katagiri T, Mundlos S, Seemann P, Kaplan FS, Mullins MC, Shore EM. Fibrodysplasia ossificans progressiva R206H ACVR1 mutation activates BMP-independent chondrogenesis and zebrafish embryo ventralization. J Clin Invest. 2009;119:3462–3472. doi: 10.1172/JCI37412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.van Dinther M, Visser N, deGorter DJ, Doorn J, Goumans MJ, de Boer J, ten Dijke P. ALK2 R206H mutation linked to fibrodysplasia ossificans progressiva confers constitutive activity to the BMP type I receptor and sensitizes mesenchymal cells to BMP-induced osteoblast differentiation and bone formation. J Bone Miner Res. 2010;25:1208–1215. doi: 10.1359/jbmr.091110. [DOI] [PubMed] [Google Scholar]
- 9.Medici D, Shore EM, Lounev VY, Kaplan FS, Kalluri R, Olsen BR. Conversion of vascular endothelial cells into multipotent stem-like cells. Nat Med. 2010;16:1400–1406. doi: 10.1038/nm.2252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kaplan FS, Glaser DL, Shore EM, Pignolo RJ, Xu M, Zhang Y, Senitzer D, Forman SJ, Emerson SG. Hematopoietic stem-cell contribution to ectopic skeletogenesis. J Bone Joint Surg Am. 2007;89:347–357. doi: 10.2106/JBJS.F.00472. [DOI] [PubMed] [Google Scholar]
- 11.Suda RK, Billings PC, Egan KP, Kim JH, McCarrick-Walmsley R, Glaser DL, Porter DL, Shore EM, Pignolo RJ. Circulating osteogenic precursor cells in heterotopic bone formation. Stem Cells. 2009;27:2209–2219. doi: 10.1002/stem.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lounev VY, Ramachandran R, Wosczyna MN, Yamamoto M, Maidment AD, Shore EM, Glaser DL, Goldhamer DJ, Kaplan FS. Identification of progenitor cells that contribute to heterotopic skeletogenesis. J Bone Joint Surg Am. 2009;91:652–663. doi: 10.2106/JBJS.H.01177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Levy MM, Joyner CJ, Virdi AS, Reed A, Triffitt JT, Simpson AH, Kenwright J, Stein H, Francis MJ. Osteoprogenitor cells of mature human skeletal muscle tissue: an in vitro study. Bone. 2001;29:317–322. doi: 10.1016/s8756-3282(01)00585-3. [DOI] [PubMed] [Google Scholar]
- 14.Wosczyna MN, Biswas AA, Cogswell CA, Goldhamer DJ. Multipotent progenitors resident in the skeletal muscle interstitium exhibit robust BMP-dependent osteogenic activity and mediate heterotopic ossification. J Bone Miner Res. 2012;27:1004–1017. doi: 10.1002/jbmr.1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Medici D, Kalluri R. Endothelial-mesenchymal transition and its role in contributing to stem cell phenotype. Semin Cancer Biol. 2012 doi: 10.1016/j.semcancer.2012.04.004. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Potenta S, Zeisberg E, Kalluri R. The role of endothelial-to-mesenchymal transition in cancer progression. Br J Cancer. 2008;99:1375–1379. doi: 10.1038/sj.bjc.6604662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Markwald RR, Fitzharris TP, Smith WN. Structural analysis of endocardial cytodifferentiation. Dev Biol. 1975;42:160–180. doi: 10.1016/0012-1606(75)90321-8. [DOI] [PubMed] [Google Scholar]
- 18.Mercado-Pimentel ME, Runyan RB. Multiple transforming growth factor-beta isoforms and receptors function during epithelial-mesenchymal cell transformation in the embryonic heart. Cells Tissues Organs. 2007;185:146–156. doi: 10.1159/000101315. [DOI] [PubMed] [Google Scholar]
- 19.Zeisberg EM, Tarnavski O, Zeisberg M, Dorfman AL, McMullen JR, Gustafsson E, Chandraker A, Yuan X, Pu WT, Roberts AB, Neilson EG, Sayegh MH, Izumo S, Kalluri R. Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat Med. 2007;13:952–961. doi: 10.1038/nm1613. [DOI] [PubMed] [Google Scholar]
- 20.Zeisberg EM, Potenta SE, Sugimoto H, Zeisberg M, Kalluri R. Fibroblasts in kidney fibrosis emerge via endothelial-to-mesenchymal transition. J Am Soc Nephrol. 2008;19:2282–2287. doi: 10.1681/ASN.2008050513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hashimoto N, Phan SH, Imaizumi K, Matsuo M, Nakashima H, Kawabe T, Shimokata K, Hasegawa Y. Endothelial-mesenchymal transition in bleomycin-induced pulmonary fibrosis. Am J Respir Cell Mol Biol. 2010;43:161–172. doi: 10.1165/rcmb.2009-0031OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zeisberg EM, Potenta S, Xie L, Zeisberg M, Kalluri R. Discovery of endothelial to mesenchymal transition as a source for carcinoma-associated fibroblasts. Cancer Res. 2007;67:10123–10128. doi: 10.1158/0008-5472.CAN-07-3127. [DOI] [PubMed] [Google Scholar]
- 23.Lee JG, Kay EP. FGF-2-mediated signal transduction during endothelial mesenchymal transformation in corneal endothelial cells. Exp Eye Res. 2006;83:1309–1316. doi: 10.1016/j.exer.2006.04.007. [DOI] [PubMed] [Google Scholar]
- 24.Tran KV, Gealekman O, Frontini A, Zingaretti MC, Morroni M, Giordano A, Smorlesi A, Perugini J, De Matteis R, Sbarbati A, Covera S, Cinti S. The vascular endothelium of the adipose tissue gives rise to both white and brown fat cells. Cell Metabolism. 2012;15:222–229. doi: 10.1016/j.cmet.2012.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gupta RK, Mepani RJ, Kleiner S, Lo JC, Khandekar MJ, Cohen P, Frontini A, Bhowmick DC, Ye L, Cinti S, Spiegelman BM. Zfp423 expression identifies committed preadipocytes and localizes to adipose endothelial and perivascular cells. Cell Metabolism. 2012;15:230–239. doi: 10.1016/j.cmet.2012.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fadini GP, Losordo D, Dimmeler S. Critical reevaluation of endothelial progenitor cell phenotypes for therapeutic and diagnosic use. Circ Res. 2012;110:624–637. doi: 10.1161/CIRCRESAHA.111.243386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Moonen JR, Krenning G, Brinker MG, Koerts JA, van Luyn MJ, Harmsen MC. Endothelial progenitor cells give rise to pro-angiogenic smooth muscle-like progeny. Cardiovasc Res. 2010;86:506–515. doi: 10.1093/cvr/cvq012. [DOI] [PubMed] [Google Scholar]
- 28.Yu PB, Deng DY, Lai CS, Hong CC, Cuny GD, Bouxsein ML, Hong DW, McManus PM, Katagiri T, Sachidanandan C, Kamiya N, Fukuda T, Mishina Y, Peterson RT, Bloch KD. BMP type I receptor inhibition reduces heterotopic ossification. Nat Med. 2008;14:1363–1369. doi: 10.1038/nm.1888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Miyazono K, Kamiya Y, Morikawa M. Bone morphogenetic protein receptors and signal transduction. J Biochem. 2010;147:35–51. doi: 10.1093/jb/mvp148. [DOI] [PubMed] [Google Scholar]
- 30.Medici D, Potenta S, Kalluri R. Transforming growth factor-β2 promotes Snail-mediated endothelial-mesenchymal transition through convergence of Smad-dependent and Smad-independent signalling. Biochem J. 2011;437:515–520. doi: 10.1042/BJ20101500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nieto MA. The snail superfamily of zinc-finger transcription factors. Nat Rev Mol Cell Biol. 2002;3:155–166. doi: 10.1038/nrm757. [DOI] [PubMed] [Google Scholar]
- 32.Paruchuri S, Yang JH, Aikawa E, Melero-Martin JM, Khan Z, Loukogeorgakis S, Schoen FJ, Bischoff J. Human pulmonary valve progenitor cells exhibit endothelial/mesenchymal plasticity in response to vascular endothelial growth factor-A and transforming growth factor-beta2. Circ Res. 2006;99:861–869. doi: 10.1161/01.RES.0000245188.41002.2c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Salisbury E, Rodenberg E, Sonnet C, Hipp J, Gannon FH, Vadakkan TJ, Dickinson ME, Olmstead-Davis EA, Davis AR. Sensory nerve induced inflammation contributes to heterotopic ossification. J. Cell. Biochem. 2011;112:2748–2758. doi: 10.1002/jcb.23225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kan L, Lounev VY, Pignolo RJ, Duan L, Liu Y, Stock SR, McGuire TL, Lu B, Gerard NP, Shore EM, Kaplan FS, Kessler JA. Substance P signaling mediates BMP-dependent heterotopic ossification. J Cell Biochem. 2011;112:2759–2772. doi: 10.1002/jcb.23259. [DOI] [PMC free article] [PubMed] [Google Scholar]