Maternal obesity affects 1 in 5 pregnant women in the United States1. Maternal obesity is associated with increased circulating total, but not fetal, cf DNA2. This may be a result of increased production or decreased clearance of cf DNA in obese women. It is more likely that this is due to increased production of total cfDNA, because decreased clearance would also likely lead to an increase in cell-free fetal cfDNA. In a prior study performed on obese pregnant women, we showed that active remodeling of adipose tissue via adipocyte necrosis and/or apoptosis of the stromal vascular fraction results in an increased release of cfDNA of maternal origin into the circulation3. The focus of the prior study was on the mechanisms underlying the release of the cfDNA. In this study we more closely examined the correlation between maternal weight and total DNA levels.
This study was approved by the Institutional Review Boards at Tufts Medical Center and Metrohealth Medical Center. The samples are from the same cohort reported previously3, but the analysis is different. Briefly, sixteen obese (mean=39.2; pre-gravid BMI range 31–51) and 14 lean (mean 21.8; pre-gravid BMI range 17–24) women carrying male fetuses and 10 women carrying female fetuses (negative controls) were recruited at term (37–40 weeks) prior to an elective cesarean section. Written informed consent was obtained prior to obtaining the samples. Women with a multiple gestations, placenta previa or invasive placentation, labor, infection, fetal anomalies or aneuploidy, intrauterine growth restriction, or preeclampsia were excluded.
Maternal peripheral venous blood was collected at MetroHealth Medical Center on admission to labor and delivery, prior to placement of an intravenous line for hydration. All subjects had the same instructions prior to admission and had nothing to eat or drink for 6–8 hours prior to the blood draw. The blood was collected in an EDTA tube and plasma was separated by centrifugation and kept frozen at −20°C prior to being shipped to Tufts Medical Center for further analysis. DNA was extracted from 400 uL of plasma using the QIAamp DNA Blood Mini Kit (Qiagen, Valencia, CA) according to the blood and body fluid protocol. DNA was eluted in 50 μL of the elution buffer. Real time quantitative PCR amplification was performed as previously described4 to amplify glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and the Y chromosome sequence DYS14 as markers of total and fetal DNA, respectively. All samples were analyzed in triplicate. Analysis was blinded, and a female processed and handled all samples so that there was no risk of contaminating samples with male DNA. Conversion of raw PCR data to genome equivalents per mL of plasma was performed using the methods of Lo et al.5
The levels of total and fetal cf DNA in plasma from lean and obese women were compared using the t test. In addition, the maternal plasma volume from both obese and lean subjects was adjusted for blood volume6 and compared using the t test, as prior experiments in our laboratory suggested an increased blood volume as a function of weight7. Finally, we performed a regression analysis between total and fetal cf DNA and maternal BMI.
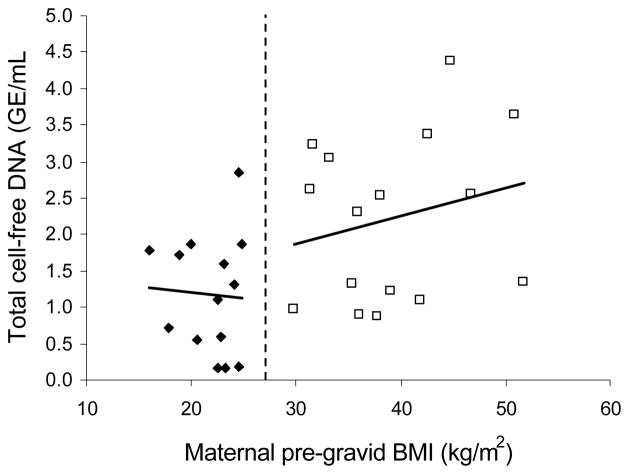
Analysis of the raw data from all subjects showed that there was a 1.7% increase in total cell-free DNA per BMI unit (kg/m2). Following adjustment for blood volume, there was a 3.2% increase per BMI unit (Table 1). When the subjects were categorized into lean and obese groups, however, there was a decrease in total cf DNA per BMI unit in lean women and an increase in obese women (Table 1 and Figure 1). Neither of these changes were statistically significant. However, this lack of significance may be a function of the small sample size. Further studies with more subjects are warranted to further characterize this relationship between cf DNA and BMI.
Table 1.
Change in maternal cell-free DNA by body mass index
| % increase per BMI unit | ||
|---|---|---|
| Subjects | Raw data1 | BV corrected2 |
| All | 1.7 | 3.2 |
| Lean (BMI<25) | −2.8 | −0.7 |
| Obese (BMI>30) | 1.5 | 2.6 |
Measured as genome equivalents per mL maternal plasma
Data corrected for blood volume using Lemmens et al. (2006) BMI: body mass index (kg/m2)
Figure 1.
Graph showing relationship between total cell-free DNA levels and body mass index [BMI]. The correlation coefficient between BMI and cfDNA for the lean population is -0.061 (p=0.41) and for the obese population is 0.23 (p=0.20).
In this study, we found a correlation between BMI and total DNA levels. In lean women, the decrease in total cf DNA likely reflects a dilutional effect seen in all pregnant women due to increasing plasma volume. Conversely, in obese women, the increased levels of total cf DNA may reflect the increased adipocyte necrosis3 and stromal vascular apoptosis that is significant enough to overcome the dilutional effect that occurs in pregnant women.
Our results are similar to a previous study by Lapaire et al.2, in which a correlation was found between maternal BMI and total, but not fetal, second trimester cf DNA levels. There are several differences between our study and the Lapaire study, however. Firstly, the gestational ages of the study subjects were different. In the present study, maternal blood samples were drawn at term, in contrast to 20 to 21 weeks. Secondly, in the Lapaire et al. study there was no adjustment for maternal weight. We adjusted for the effect of maternal weight on blood volume using the method described by Lemmens et al.6
The present study and that of Wataganara et al.7 suggest that a correction factor for maternal weight is needed when cf DNA analyses are performed. It is standard practice to correct for maternal weight when performing serum screening for Down syndrome. Diagnostic laboratories typically derive their own regression curves for the relationship between serum analytes and maternal weight because of several factors, including differences in instrument sensitivity in the measurement of serum analytes, as well as differences in maternal weight distribution among centers8. Review of the literature, however, indicates that there is no consensus as to the best method to adjust serum markers, including cf nucleic acids, for maternal weight in cases of extreme obesity. We selected the Lemmens et al.6 formula because it correlated total blood volume with BMI, although that formula was used for quantification of blood volume in non-pregnant surgical patients.
In summary, we show here that maternal BMI affects total cf DNA levels in both lean and obese pregnant women. In the future, raw DNA values may need to be adjusted for maternal BMI for adequate interpretation of clinical tests that involve assessment of the fetal fraction9. In addition, the presence of increased total cf DNA levels in obese pregnant women suggests that this analyte may be a biomarker for systemic problems during gestation that may impact both maternal and fetal health.
What is already known about this topic?
Experiments in our laboratory and others have suggested that maternal weight affects levels of circulating cell-free (cf) nucleic acids in the maternal circulation.
What does this study add?
There is a decrease in total cf DNA per BMI unit in lean women, and an increase in obese women, demonstrating a multi-factorial relationship between maternal weight and cf DNA. These data suggest that a correction factor for maternal weight may be needed when analyses of maternal plasma cf DNA are performed.
Acknowledgments
We would like to acknowledge L Presley and J Minium for recruiting pregnant women into the study and obtaining plasma samples.
Funding sources: NIH R01 HD 22965 (PMC/SHM), R01 HD42053-07 (DWB); NIH T32 HD049341.
References
- 1.Kim SY, Dietz PM, England L, Morrow B, Callaghan WM. Trends in prepregnancy obesity in nine states, 1993–2003. Obesity. 2007;15:986–993. doi: 10.1038/oby.2007.621. [DOI] [PubMed] [Google Scholar]
- 2.Lapaire O, Volgmann T, Grill S, et al. Significant correlation between maternal body mass index at delivery and in the second trimester, and second trimester circulating total cell-free DNA levels. Reprod Sci. 2009;16:274–279. doi: 10.1177/1933719108327599. [DOI] [PubMed] [Google Scholar]
- 3.Haghiac M, Vora NL, Basu S, et al. Increased death of adipose cells, a path to release cell-free DNA into systemic circulation of obese women. Obesity. 2012 doi: 10.1038/oby.2012.138. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Johnson KL, Dukes KA, Vidaver J, et al. Inter-laboratory comparison of fetal male DNA detection from common maternal plasma samples by real-time PCR. Clin Chem. 2004;50:516–521. doi: 10.1373/clinchem.2003.024380. [DOI] [PubMed] [Google Scholar]
- 5.Lo YM, Tein MS, Lau TK, et al. Quantitative analysis of fetal DNA in maternal plasma and serum: implications for noninvasive prenatal diagnosis. Am J Hum Genet. 1998;62:768–775. doi: 10.1086/301800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lemmens HJ, Bernstein DP, Brodsky JB. Estimating blood volume in obese and morbidly obese patients. Obes Surg. 2006;16:773–776. doi: 10.1381/096089206777346673. [DOI] [PubMed] [Google Scholar]
- 7.Watanagara T, Peter I, Messerlian GM, et al. Inverse correlation between maternal weight and second trimester circulating cell-free fetal DNA levels. Obstet Gynecol. 2004;104:545–550. doi: 10.1097/01.AOG.0000137352.93110.15. [DOI] [PubMed] [Google Scholar]
- 8.Neveux LM, Palomaki GE, Larrivee DA, et al. Refinements in managing maternal weight adjustment for interpreting prenatal screening results. Prenat Diagn. 1996;16:1115–1119. doi: 10.1002/(SICI)1097-0223(199612)16:12<1115::AID-PD3>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 9.Palomaki GE, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study. Genet Med. 2011;13:913–920. doi: 10.1097/GIM.0b013e3182368a0e. [DOI] [PubMed] [Google Scholar]