Abstract
Purpose
Individuals with Parkinson’s disease (PD) exhibit differences in displacement and velocity of the articulators as compared to older adults. The purpose of the current study was to examine effects of three loudness cues on articulatory movement patterns in individuals with PD.
Methods
Nine individuals diagnosed with idiopathic PD and 9 age- and sex- matched healthy controls produced sentences in four conditions: 1) comfortable loudness, 2) targeting 10dB above comfortable, 3) twice as loud as comfortable, and 4) in background noise. Lip and jaw kinematics and acoustic measurements were obtained.
Results
Both groups significantly increased sound pressure level (SPL) in the loud conditions as compared to comfortable. For the loud conditions, both groups had the highest SPL in background noise and 10dB and the lowest in twice as loud. Control participants produced the largest opening displacement in background noise and the smallest in twice as loud. Conversely, individuals with PD produced the largest opening displacement in twice as loud and the smallest in background noise.
Conclusions
Control participants and individuals with PD responded to cues to increase loudness in different ways. Changes in SPL may explain differences in kinematics for the control participants, but do not for individuals with PD.
Keywords: Parkinson’s disease, articulatory kinematics, cueing
INTRODUCTION
Parkinson’s disease (PD) affects about 1.5 million people in the United States (Yorkston, Beukelman, Strand, & Bell, 1999) and is one of the most common degenerative diseases of neurological origin (Burn, 2000). Parkinson’s disease results in motor impairments including rigidity, tremor at rest, bradykinesia, reduced range of movement, masked facial expressions and difficulty initiating movement; cognitive impairments including deficits in set-switching and verbal working memory; speech impairments including hypokinetic dysarthria; and swallowing impairments leading to aspiration pneumonia (Darley, Aronson, & Brown, 1969; Hochstadt, Nakano, Lieberman, & Friedman, 2006; Squire Bloom, McConnell, Roberts, Spitzer, & Zigmond, 2003; Yorkston et al., 1999).
The majority of individuals with PD manifest some type of speech impairment during the course of the disease (Ho, Iansek, Marigliani, Bradshaw, & Gates, 1999; Logemann, Fisher, Boshes, & Blonsky, 1978; Sapir, Pawlas, Ramig, Countryman, O’Brien, Hoehn, et al., 2001). Hypokinetic dysarthria, the most common type of dysarthria in individuals with PD, is characterized by deficits in the articulatory, laryngeal, and respiratory system (Ackermann & Ziegler, 1991; Baker, Ramig, Luschei, & Smith, 1998; Bunton, 2005; Dromey, Ramig, & Johnson, 1995; Forrest, Weismer, & Turner, 1989; Kleinow, Smith, & Ramig, 2001; Sadagopan & Huber, 2007; Smith, Ramig, Dromey, Perez, & Samandari, 1995; Solomon & Hixon, 1993). The cardinal perceptual characteristics associated with hypokinetic dysarthria are monopitch, monoloudness, reduced stress, inappropriate silences, reduced loudness, harshness, breathiness, low pitch, imprecise consonants, inappropriate silences, variable rate, short rushes of speech, and syllable repetitions (Darley et al., 1969). Hypokinetic dysarthria in individuals with PD typically begins with some type of voice impairment (Logemann et al., 1978; Sapir et al., 2001). In a study of two hundred patients with PD, Logemann et al. (1978) found that 89% of subjects had some type of voice impairment. As the disease progresses, individuals with PD also begin to have abnormal articulation (Logemann et al., 1978; Sapir et al., 2001). However, there is new evidence for speech (vowel) articulation abnormalities in early stages of Parkinson’s disease (Skodda, Visser, & Schlegel, in press).
Loudness Control in Individuals with PD
Despite the numerous deficits associated with hypokinetic dysarthria, the deficit most commonly treated in speech therapy is hypophonia (reduced vocal loudness) (Ramig, Countryman, Thompson, & Horii, 1995; Ramig, Fox, & Sapir, 2004; Ramig, Sapir, Fox, & Countryman, 2001). Control of vocal loudness requires the articulatory, laryngeal, and respiratory systems to work as an integrated unit. To increase vocal loudness, typical speakers breathe to higher lung volumes thus taking advantage of higher passive recoil forces to generate higher subglottal pressures (Hixon, Goldman, & Mead, 1973; Huber, 2007, 2008). To generate higher subglottal pressure, resistance at the level of the vocal folds must increase as well (Stathopoulos & Sapienza, 1997). During loud speech, changes to articulation include larger excursions and faster velocities (Huber & Chandrasekaran, 2006; Schulman, 1989). Given that individuals with PD typically have difficulty breathing to higher lung volumes, have decreased glottal closure, and have reduced amplitude and velocity of articulatory movements, it is easy to understand why individuals with PD are often perceived as hypophonic (Bunton, 2005; Dromey, 2000; Sadagopan & Huber, 2007; Smith, et al., 1995; Solomon & Hixon, 1993). While the laryngeal and respiratory systems are crucial for the production of loud speech, this paper will focus on the role of the articulatory system in the production of loud speech in individuals with PD.
Articulatory Changes in Individuals with PD
Changes in the function of the lip and jaw have been observed in studies of individuals with PD (Forrest, Weismer, & Turner, 1989; Forrest & Weismer, 1995, Walsh, 2007). During sentence and word repetition, lower lip and jaw displacements and velocities were decreased in individuals with PD between Hoehn and Yahr (H&Y) stage two and three as compared to healthy, age-matched controls (Forrest et al., 1989; Forrest & Weismer, 1995, Walsh, 2007). Differences in lower lip movement may correlate with the perceived severity of speech impairment. In one study, individuals with severe dysarthria had greater reductions in lower lip displacement and velocity than those with mild dysarthria (Forrest et al., 1989).
In addition to reductions in the displacement and velocity of articulatory movements, the stability of repeated speech movements may be affected by PD (Dromey, 2000; Kleinow, Smith, & Ramig, 2001; Walsh, 2007). The Spatiotemporal Index (STI) is one measure designed to reflect the underlying pattern or stability of movements (Smith, Johnson, McGillem, & Goffman, 2000). A high STI reflects a more variable pattern of movements, whereas a low STI reflects a more stable pattern of movements. Using this index, Kleinow et al. (2001) found that typically aging adults and individuals with PD demonstrated less trial-to-trial lip and jaw movement stability as compared to young adults. However, Kleinow et al. (2001) did not find differences in the lip and jaw movement stability between individuals with PD and typically aging adults. Walsh (2007) used a potentially more sensitive measure to observe the articulatory stability of repeated movements. The lip aperture (LA) variability index reflects the trial-to-trial variability in the coordination of the upper lip, lower lip, and jaw. Walsh (2007) found that individuals with PD with mild-moderate dysarthria had significantly higher LA variability indices as compared to control participants. Higher LA variability indices indicate individuals with PD demonstrate less coordinative stability across the articulatory mechanism (Walsh, 2007). Taken together, these results indicate that while the stability of the movement of the lower lip and jaw alone may not differ in individuals with PD with respect to age-matched control subjects, the stability of coordination across the articulatory mechanism does decline as a function of disease.
Effects of Loudness on Individuals with PD
One of the distinctive characteristics of the dysarthria associated with PD is the perception of decreased loudness (Darley et al., 1969). Increasing sound pressure level (SPL) has been shown to change articulatory kinematic patterns in young adults and individuals with PD (Dromey, 2000; Huber & Chandrasekaran, 2006; Tasko & McClean, 2004). In young adults, increased SPL resulted in increased velocity and displacement of the articulators, particularly the jaw (Huber & Chandrasekaran, 2006; Schulman, 1989; Tasko & McClean, 2004). Dromey (2000) demonstrated that in individuals with PD, lower lip velocity and displacement increased significantly with increased SPL. Increasing SPL has also been found to increase the stability of repeated speech movements in individuals with PD (Dromey, 2000; Kleinow et al., 2001).
Self-Monitoring of Motor Tasks in Individuals with PD
In addition to the dysarthria associated with PD, individuals with this disease often demonstrate reduced ability to monitor their speech production as compared to typically aging adults and young adults (Ho, Bradshaw, & Iansek, 2000; McNamara, Obler, Au, Durso, & Albert, 1992). McNamara et al. (1992) found that during picture description, individuals with PD made significantly more speech errors than control subjects and only corrected these errors 25% of the time compared to 72–92% corrected errors by control participants. Individuals with PD are also less likely to adequately judge how loudly they are speaking which may contribute hypophonia associated with PD. Ho, Bradshaw, and Iansek (2000) found that while individuals with PD were found to speak at significantly lower SPL than control participants, they consistently rated themselves louder than control participants. The authors suggested that the tendency to overestimate loudness is related to deficits in sensory integration.
However, not all studies of individuals with PD suggest that speech monitoring skills are impaired. Fox and Ramig (1997) found that individuals with PD rate themselves as more severely impaired when asked to rate their speech and voice characteristics as compared to the ratings of typically aging adults, suggesting intact self-monitoring abilities. However, information was not given on how accurate the participants were in rating their own speech as compared to formal assessment ratings of speech and voice characteristics. Participants could have rated themselves as more severely impaired due to external influences including knowledge that PD affects speech function and familial concerns about speech function, while at the same time misjudging the severity of the problem. When asked to perform judgment tasks regarding vocal loudness during sustained phonation, Dromey and Adams (2000) did not find significant differences between older adults and individuals with PD. However, the authors suggest that while individuals with PD may have a preserved ability to judge loudness in a highly structured, simple task such as sustained phonation they may still be unable to accurately self-monitor during an unstructured, more complex task such as conversation.
Effects of Cueing on Individuals with PD
The potential that individuals with PD do not adequately self-monitor motor output has led researchers to search for possible methods of cueing, to call attention to motor planning and output without relying on an individual’s self-monitoring abilities. Numerous studies have been conducted to observe changes to motor planning and output in response to different external and internal cues in individuals with PD (Morris, Iansek, Matyas, & Summers, 1996; Oliveira, Gurd, Nixon, Marshall, & Passingham, 1997; Sadagopan & Huber, 2007; Sidaway, Anderson, Danielson, Martin, & Smith, 2006; Willems, Nieuwboer, Chavret, Desloovere, Dom, Rochester, et al., 2006). Tasks that are performed under “the guidance of visual, auditory, or somatosensory feedback” are considered to be guided by external cues whereas movements based on internal cues are guided by unconscious information or “self-cues” (Gazzaniga, Ivry, & Mangun, 1998, 471).
Researchers have observed improvements in gait patterns as a result of the influences of external and internal cues in individuals with PD (Morris et al, 1996; Sidaway et al., 2006; Willems et al., 2006). Internal cues such as visualization of the movement were found to be effective in normalizing gait patterns in individuals with PD (Morris et al., 1996). However, internal cues did not generalize as well as external cues. External cues have resulted in stride length regulation and a decrease in stepping frequency (Morris et al, 1996; Sidaway et al., 2006; Willems et al., 2006). Based on the effectiveness of these types of cues, the use of external cues is supported in gait therapy for individuals with PD. Similarly, auditory and visual cues were shown to increase movement amplitude for handwriting, and individuals with PD were able to maintain this increased movement amplitude on the non-cued trial following the cued trials (Oliveira et al., 1997). Just as visual and auditory cues have been shown to improve limb movement in individuals with PD, external cues used to address loudness and articulation have been found to modulate speech behavior in individuals with PD (Dromey, 2000; Ho, Bradshaw, Iansek, & Alfredson, 1999; Kleinow et al., 2001 Sadagopan & Huber, 2007).
While external cues have been shown to effect change in the motor behavior of individuals with PD, different types of cues may affect motor behavior differently. For example, Willems et al. (2006) demonstrated that different cues elicit the most effective gait pattern in two subgroups of individuals with PD, those who have freezing symptoms and those who do not. Individuals who experienced freezing benefited the most from a metronome cue set 10% above the frequency of baseline stepping whereas those who did not experience freezing benefited the most from an metronome cue set 10% below the frequency of baseline stepping.
Different types of cues have also been shown to result in different loudness changes and different respiratory patterns in individuals with PD. Sadagopan and Huber (2007) found that the amount of SPL increase differed across three cues to increase loudness. When background noise was used, individuals with PD were significantly louder than when they were asked to speak twice as loud as comfortable or when they were asked to target a specific SPL using an SPL meter for feedback. Also, background noise was also found to elicit more typical respiratory patterns from individuals with PD than the other cues.
Although the effects of different types of cues to increase loudness have been observed for SPL changes and respiratory patterns in individuals with PD, most studies of the articulatory system in individuals with PD compared cues to elicit alterations in loudness and rate (Dromey, Ramig, & Johnson, 1995; Dromey, 2000; Kleinow et al., 2001), without an examination of how cues to achieve the same goal (for example, increased loudness) differ from one another relative to the motor patterns they elicit. However, the studies which have been done support the effectiveness of external cues in changing articulatory kinematics in individuals with PD. Dromey (2000) asked individuals with PD to either hyperarticulate or to speak at twice their comfortable loudness. Both hyperarticulating and increasing loudness resulted in a significant increase in displacement and velocity of lip movements. However, the hyperarticulate condition resulted in a more variable displacement and velocity of lip movements across utterances than both the twice as loud and habitual conditions (Dromey, 2000). Asking individuals with PD to decrease their rate of speech also led to more variable lip and jaw movement patterns than asking them to talk twice as loud or at habitual loudness and rate (Kleinow et al., 2001). Taken together with the results from the respiratory system, these findings provide evidence not only for the importance of using cues to elicit improved motor patterns in individuals with PD, but also the importance of the type of cue used to elicit these patterns (Dromey, 2000; Kleinow et al., 2001; Sadagopan & Huber, 2007).
Although little is known about the effects of different cues to increase loudness on the articulatory system in individuals with PD, data from the articulatory system in young adults shows that different loudness cues elicit different articulatory movement patterns (Huber & Chandrasekaran, 2006). When asked to target a loudness level approximately 10dB above their comfortable level, young adults exhibited significantly decreased movement variability during a sentence task than when performing the task at a comfortable loudness level (Huber & Chandrasekaran, 2006). Based on these results, it is possible that different cues to increase loudness will result in different articulatory patterns in individuals with PD, as was shown in the respiratory kinematic data (Sadagopan & Huber, 2007).
Changes to articulation as a result of cues to elicit increased loudness are of clinical importance. Due to the prevalence of perceived hypophonia, therapy techniques that target increased loudness are commonly prescribed for individuals with PD, most commonly the Lee Silverman Voice Treatment (LSVT LOUDR) program. Based on acoustic data, it has been suggested that LSVT LOUDR produces increased movement amplitude and stability in the articulatory system. For example, there is evidence for improved vowel articulation with increased vocal loudness following LSVT LOUDR, as reflected in acoustic measures of vowel formants, including the F2i/F2u ratio and the Formant Centralization Ratio, as well as improved perception of vowel goodness, with significant correlations between the SPL and vowel acoustic and perception measurements (Dromey et al., 1995; Sapir, Ramig, Spielman, & Fox., 2010; Sapir et al., 2007). Investigating how different types of cues affect the movement and stability of the articulatory system may reveal that one type of cue results in more stable articulatory patterns or patterns more similar to that of typical, elderly speakers.
Hypotheses
The current study investigated the effects of three loudness cues on lip and jaw movement patterns in individuals with PD. It was hypothesized that both control participants and individuals with PD would alter articulatory patterns in response to the different cues to increase loudness. Further, due to their susceptibility to cueing, it was hypothesized that individuals with PD would not respond in the same way as control participants to the different cues to increase loudness. Lastly, it was hypothesized that overall articulatory movement stability would be lower in individuals with PD than in control participants, although less so in the loud conditions.
METHODS
Participants
Nine individuals diagnosed with idiopathic Parkinson’s disease (PD) and 9 age- and sex-matched healthy control participants served as the participants in this study. Each group consisted of 6 men and 3 women. The age range for the participants was 68–90. The mean age of the individuals with PD was 78 years with a standard deviation of 6.6 years. The mean age of the control participants was 73.6 years with a standard deviation of 4.3 years.
All participants had been non-smokers for at least the past 5 years, reported no head or neck cancer or surgery, and no formal training in singing or speaking. All participants were free of infections, colds, and allergies on the day of testing. Participants were required to be living independently and be ambulatory. Each participant was required to pass the Mini-Mental Status examination (Folstein, Folstein, & McHugh, 1975) with a score of 24 to ensure adequate cognitive function. The control participants reported no neurological disease and demonstrated normal speech, language, and voice (as determined by the second author, a certified speech-language pathologist). Control participants also had normal hearing as assessed by a hearing screening at 40dB HL for 500Hz, 1000Hz, and 2000Hz frequencies, bilaterally, in a quiet room (Ventry & Weinstein, 1983). Although hearing status was not an exclusionary criterion for individuals with PD, participants with PD were also given hearing screenings. All participants with PD passed the hearing screening at 40dB HL for the 500Hz and1000Hz frequencies, while 6 of the 9 participants with PD passed the hearing screening at 40dB HL for the 2000Hz frequency. Those who did not pass at the 2000Hz frequency had values ranging from 50dB to 60dBHL. All participants with PD were tested within 1–3 hours of taking their anti-Parkinsonian medication, even though medication cycle is not likely to affect speech (Skodda, Visser, & Schlegel, 2010; Solomon & Hixon, 1993). All participants with PD were classified as H&Y stage two or three. Demographics such as age, time since diagnosis, and medications for participants with PD are presented in Table 1.
Table 1.
Demographics and speech impairment rating of individuals with PD
| Participant | Age | Time Since Diagnosis | Medications | Reduced Loudness | Speech Rate | Articulatory Precision |
|---|---|---|---|---|---|---|
| F01PD | 72 | 9 months | Mirapex | Mild | Mild | Normal |
| F02PD | 74 | 5 years | Sinemet, Eldeypryl, Dopamine agonist (Bromocriptine), Co-enzyme Q-10 (antioxidant) | Normal | Normal-Mild | Normal |
| F03PD | 79 | 6 years | Eldepryl, Stalevo, Requip | Normal | Normal-Mild | Normal |
| M01PD | 83 | 4 years 11 months | Sinemet | Severe | Moderate | Mild |
| M02PD | 76 | 5 years | Sinemet, Requip, Comtan | Moderate | Moderate-Severe | Moderate |
| M03PD | 73 | 11 years | Benztropine, Carbidopa, Levadopa | Moderate | Moderate | Moderate |
| M04PD | 68 | 3–4 years | Permax, Stalevo | Mild | Normal | Normal |
| M05PD | 90 | 3 years | Carbidopa, Levadopa, Amantadine | Mild | Normal-Mild | Mild |
| M06PD | 82 | 3 years 8 months | Amantadine, Sinemet | Mild | Normal | Normal-Mild |
Three experienced speech-language pathologists (not affiliated with the study) evaluated the speech of the participants with PD and rated the characteristics of reduced loudness, speech rate, and articulatory precision (Table 1). Ratings of reduced loudness were obtained from sentences, presented without equalizing intensity, while ratings of speech rate and articulatory precision were obtained from sentences, equalized for intensity (70 dB). These ratings were made on a scale from 1 (normal) to 7 (severe impairment). Overall ratings for each dimension represent an average of the raters’ scores. Individuals with PD represented an overall mild to moderate speech impairment.
Procedures and Speech Stimuli
Participants were asked to say the sentences “Buy Bobby a puppy” (short sentence) and “You buy Bobby a puppy now if he wants one” (embedded sentence). Inclusion of these sentences allowed researchers to observe potential effects of sentence length and complexity on lip and jaw kinematics. Participants were instructed to say each sentence one time per breath at a level that was clear and audible to the experimenters. These sentences were produced fifteen times each in the following four conditions:
COMF: Participants read the sentences at a comfortable loudness and pitch. No feedback about loudness was provided.
COMF+10: Participants were asked to read the sentences while targeting a specific loudness level, which was approximately 10dB (+/− 2dB) above their comfortable loudness level. A SPL meter was used to provide visual feedback in real-time.
2XCOMF: Participants were instructed to read the sentences at a level that they perceive to be as “twice as loud” as their comfortable loudness level. When this condition was not preceded by the COMF condition, the participants were acclimated to their comfortable loudness level before the “twice as loud” cue was given. No feedback about loudness was provided.
NOISE: Background multi-talker noise (AUDiTEC of St. Louis) was introduced at a level of 70dBA through field-free speakers positioned in front of the participant at a distance of about 40 inches. The participants were then asked to read the sentences. No feedback about loudness was provided.
Participants were required to practice at the start of each condition by saying one sentence after being given the cue for the condition. The trials of the short sentence were always completed first, followed by the trials of the embedded sentence in each condition. In order to establish a baseline level of loudness and pitch, the COMF condition was always performed first. The order of the three loudness conditions (COMF+10, 2XCOMF, and NOISE) was counterbalanced across participants.
Equipment
An omnidirectional, condenser microphone (Quest model 1700), held at a constant 6 inches from the participant’s mouth and at a 45 degree angle, was used to capture the acoustic signal. The microphone signal was recorded on a digital audiotape (DAT tape), which was later digitized using the computer program, Praat (Boersma & Weenink, 2003). The signal was digitized at 44.1 kHz and resampled at 18 kHz. A low-pass filter at 9000 kHz was applied during resampling for anti-aliasing. An SPL meter, coupled to the microphone, was used to provide visual feedback to the participants during the COMF+10 condition. The SPL meter had a visual display screen that was projected on a television screen during the COMF+10 condition. The television screen was placed next to the computer monitor projecting the sentence to be produced.
The SPL meter was calibrated to output a 94dB signal at 1000 Hz before each participant. The calibration signal was digitized to the DAT tape, and later to the computer, to determine the calibration for measurement of SPL. The SPL meter amplified the microphone and the gain provided by the SPL meter was factored in during the calibration of the acoustic signal.
Lip and jaw kinematic data were collected using light emitting diodes (IREDs) that were tracked using a three camera system (Opotrak 3020 system, Northern Digital Inc.). IREDs were placed on the skin’s surface at midline of the vermillion border of the upper and lower lip using EMG adhesive tape. The IRED on the upper lip reflected the movement of the upper lip, and the IRED on the lower lip reflected the movement of the lower lip and jaw. To correct lip and jaw motion for head movements, five IREDs were used to determine three-dimensional axes of each participant’s head. One of these IREDs was attached at the midline of the forehead, while the other four were attached to specially-modified transparent sports goggles (Walsh & Smith, 2002). Two of the IREDs were attached on the right and left side of the goggles at the level of lateral side of the eyes, while the other two were attached on the right and left side of the goggles at the level of the angle of the mouth. Data from the IREDs was digitized by the Optotrak system at 250Hz.
The superior-inferior dimension of motion of each articulator was analyzed. The superior-inferior dimension was chosen in order to analyze the whole phrase during the two sentences since the primary dimension of lip and jaw motion for bilabial stops is the superior-inferior dimension. An audio signal, digitized at 2000 Hz, was collected in synchrony with the lip and jaw kinematic data and was used to ensure the correct identity of utterances based on the kinematic events during data measurement.
Measurements
Ten out of the 15 utterances in each trial were measured. The first 2 trials of each utterance in each condition were discarded. The following 10 consecutive utterances in each trial that were free of additions, deletions, disfluencies, or hesitations were chosen for analysis.
Sound pressure level (SPL) was measured as the average intensity across each sentence. The measurement of SPL provided information on the amount of change in vocal loudness during each loudness condition.
Articulatory kinematic measurements were completed using an algorithm designed to run within MATLAB. The kinematic measures were low-pass filtered at 40Hz. In order to measure articulatory kinematics, a portion of each utterance was segmented from the entire kinematic signal. The segmented portion began with the maximum opening velocity for the vowel [ai] in “buy” and the maximum opening velocity for the vowel [i] in “puppy” (Smith et al., 1995) (see Figure 1, lines A and B). Kinematic measurements, except the LA index, were made from the segment “Bob” in “Bobby” which could be reliably measured due to the complete closure of the lips and the readily identifiable open-close movement in the kinematic signal. The LA index was measured across the entire segmented portion of the utterance.
Figure 1.
Displacement and velocity waveforms from short sentence. Line A to line B represents the speech segment beginning at [ai] in “buy” and ending with the maximum opening velocity for the vowel [i] in “puppy”. Opening displacement for [ɑ] is represented as the difference from point C to point D and peak opening velocity is represented at point E.
Segment duration was measured as the length of the segmented portion of the utterance. For both utterances (short and embedded), this duration was taken across the same number of words (from lip opening for “Buy” to lip opening for the final vowel in “puppy”).
Opening displacement (ODIS) and opening velocity (OVEL) were measured at peak displacement and velocity for [ɑ] in “Bobby.” Signals from the lower lip IRED were measured; the lower lip IRED reflected movements of both the lower lip and the jaw. As shown in Figure 1, displacement for [ɑ] was measured as the difference from point C to point D and velocity was measured at point F.
The Lip Aperture (LA) variability index was measured across the “buy Bobby a puppy” segments from the 10 productions of each sentence in each condition. The lip aperture was defined as the difference in the signals from the upper lip and the lower lip IREDs. Since the lower lip IRED reflects the lower lip and jaw movements, this measure reflects the stability of the upper and lower lips and the jaw in controlling the size of the lip aperture during production of the utterance.
Statistics
Two-factor mixed-model analyses of variance (ANOVAs) with repeated measures were used to assess the differences between subject groups and conditions. The between factor was group (control participants and individuals with PD). The within factor was condition (COMF, COMF+10, 2XCOMF, and NOISE). Initially task (short sentence and embedded sentence) was included as a within factor. However, sentence length was not found to be a significant factor for any of the dependent measures in this study. In a recent dissertation, Walsh (2007) found that articulatory stability in individuals with PD was not differentially affected by changes to length and complexity in sentence production, as compared to age-matched control subjects. Given these findings, task was not included as a within factor and data from the two sentence types were analyzed together. Tukey’s HSD tests were used to assess pairwise comparisons for significant ANOVAs. The alpha level for the ANOVAs and Tukey’s HSD tests was set at p < .05.
Data from 2 control participants (one male and one female) and 2 individuals with PD (one male and one female) were randomly selected and reanalyzed by a second measurer. SPL, opening displacement, and peak opening velocity were remeasured. Intermeasurer reliability was determined by using an ANOVA to assess the difference between the original measures and the reliability measures. There were no significant differences between the original measures and the reliability measures indicating good inter-measurer reliability (SPL: F(1,16) = .002, p = .97, mean difference = .023; Opening Displacement: F(1,16) = .0003, p = .99, mean difference = .002; Peak Opening Velocity: F(1,16) = .0002, p = .99, mean difference = .023). Reliability measures for segment duration and LA Index were not performed. The program designed to measure these variables is an automated program that reads the entire file and calculates the final values.
RESULTS
The means, standard deviations, and ranges for each measure by group and condition are provided in Table 2.
Table 2.
Means, standard deviations, and ranges for each measure by group and condition.
| Measure | Group | COMF | COMF+10 | 2XCOMF | NOISE | ||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| Mean (SD) | Range | Mean (SD) | Range | Mean (SD) | Range | Mean (SD) | Range | ||
|
| |||||||||
| Sound Pressure Level | OC | 86.43 (11.85) | 72.23–118.29 | 94.11 (10.71) | 80.61–122.45 | 91.79 (10.62) | 80.39–121.57 | 94.93 (10.23) | 86.61–122.35 |
| PD | 75.21 (8.79) | 58.01–87.62 | 85.43 (8.43) | 64.96–96.50 | 83.61 (9.35) | 62.71–97.29 | 85.32 (6.93) | 71.89–95.00 | |
|
| |||||||||
| Segment Duration | OC | 1.00 (0.03) | 0.84–1.25 | 1.01 (0.03) | 0.87–1.40 | 1.00 (0.02) | 0.85–1.32 | 1.02 (0.03) | 0.86–1.37 |
| PD | 0.90 (0.04) | 0.73–1.20 | 0.93 (0.03) | 0.74–1.16 | 0.94 (0.04) | 0.79–1.19 | 0.93 (0.03) | 0.81–1.18 | |
|
| |||||||||
| Opening Displacement | OC | 11.33 (3.92) | 4.67–27.48 | 14. 28 (5.04) | 5.81–29.39 | 13.62 (4.81) | 6.51–27.41 | 14.12 (4.89) | 7.18–30.08 |
| PD | 8.23 (2.44) | 4.12–13.15 | 11.37 (3.70) | 6.82–21.10 | 12.02 (4.16) | 6.18–23.63 | 11.16 (3.82) | 6.35–24.72 | |
|
| |||||||||
| Peak Opening Velocity | OC | −141.05 (46.25) | −300.33-−48.03 | −177.60 (63.18) | −325.71-−56.56 | −172.53 (59.25) | −322.92-−77.57 | −169.07 (58.83) | −355.05-−77.53 |
| PD | −113.53 (38.34) | −218.54-−57.67 | −151.17 (54.57) | −299.68-−87.05 | −161.68 (59.25) | −294.32-−85.65 | −146.83 (57.05) | −322.99-−77.02 | |
|
| |||||||||
| LA index | OC | 12.00 (4.72) | 5.90–23.36 | 9.96 (2.69) | 6.68–14.85 | 10.74 (4.33) | 6.38–22.46 | 10.90 (3.33) | 5.98–17.73 |
| PD | 14.21 (3.57) | 8.63–22.74 | 13.32 (3.89) | 8.41–19.90 | 14.32 (4.17) | 9.37–25.40 | 13.11 (2.13) | 8.30–18.33 | |
Sound pressure level was measured in decibels. Segment duration was measured in seconds. Opening displacement was measured in millimeters (mm). Peak opening velocity was measured in mm/sec.
Sound Pressure Level (SPL)
There was a significant condition effect (F(3, 47) = 995.96, p < .01) and a nearly significant group effect (F(1, 16) = 4.42, p = .05). Compared to comfortable, both groups significantly increased SPL in the loud conditions (COMF+10 (t(47) = −43.56, p < .01), 2XCOMF (t(47) = −37.78, p < .01), and NOISE (t(47) = 49.13, p < .01)). Mean SPL was nearly significantly lower for individuals with PD as compared to control participants. There was also a significant group by condition effect (F(3, 47) = 22.22, p < .01) (see Figure 2). Mean SPL in NOISE was significantly louder than in 2XCOMF for both groups (control participants: t(47) = 12.60, p < .01; PD: t(47) = 4.63, p < .01). Seven of the nine control participants and five of the nine individuals with PD followed this pattern. For the control group, SPL in COMF+10 was also significantly higher than in 2XCOMF (t(47) = −9.78, p < 0.01), but there was no difference between COMF+10 and 2XCOMF for individuals with PD (t(47) = −0.92, p = 0.98). Seven of the nine control participants followed this pattern. For individuals with PD, mean SPL in COMF+10 was significantly higher than in NOISE (t(47) = 3.52, p = 0.02), but this difference was very small (~0.1 dB) and significance seems to relate mainly to the small standard error for the NOISE condition. Only three of the nine individuals with PD followed this pattern. There was no difference between COMF+10 and NOISE for control participants (t(47) = 2.80, p = 0.12).
Figure 2.
Means and standard deviations for Sound Pressure Level (SPL) in decibels (dB) for the group by condition interaction.
Segment Duration
There were no significant differences between the groups (control participants: M= 1.00 s, SD=0.14 s; PD: M=0.93, SD=0.12 s) (F(1, 16) = 2.69, p = 0.12) and no significant differences across the loudness conditions (F(3, 47) = 2.72, p = 0.06).
Opening Displacement
There was no significant group effect (F(1, 16) = 2.70, p = 0.12). There was a significant condition effect (F(3, 47) = 254.73, p < .01). For both groups, opening displacement was significantly larger for the loud conditions (COMF+10 (t(47) = −21.78, p < .01), 2XCOMF (t(47) = −22.98, p < .01), and NOISE (t(47) = −22.39), p < .01) as compared to COMF. There was also a significant group by condition effect (F(3, 47) = 15.71, p < .01) (see Figure 3). For control participants, opening displacement was significantly greater in NOISE than in 2XCOMF (t(47) = 4.30, p < 0.01). Seven of the nine control participants followed this pattern. However, individuals with PD demonstrated change in the opposite direction. For individuals with PD, opening displacement was significantly smaller in NOISE than in 2XCOMF (t(47) = −4.27, p < 0.01). Six of the nine individuals with PD followed this pattern.
Figure 3.
Means and standard deviations for Opening Displacement in mm for the group by condition interaction.
Peak Opening Velocity
There was no significant group effect (F(1, 16) = 1.25, p = 0.28). There was a significant condition effect (F(3, 47) = 230.18, p < .01). Peak opening velocity for the loud conditions (COMF+10 (t(47) = 21.24, p < .01), 2XCOMF (t(47) = 22.95, p < .01), and NOISE (t(47) = 19.01, p < .01)) was significantly greater than for COMF. There was also a significant group by condition effect (F(3, 47) = 8.36, p < .01) (see Figure 4). Individuals with PD had a significantly smaller peak opening velocity in NOISE than in 2XCOMF (t(47) = 5.86, p < 0.01). Seven of the nine individuals with PD followed this pattern. There were no significant effects across the three loud conditions for control participants (COMF+10 vs. 2XCOMF (t(47) = 1.58, p = 0.76), NOISE vs. 2XCOMF (t(47) = −0.66, p = 0.1), and NOISE vs. COMF+10 (t(47) = 0.90, p = 0.98)).
Figure 4.
Means and standard deviations for Peak Opening Velocity in mm/sec for the group by condition interaction.
Lip Aperture Variability Index (LA index)
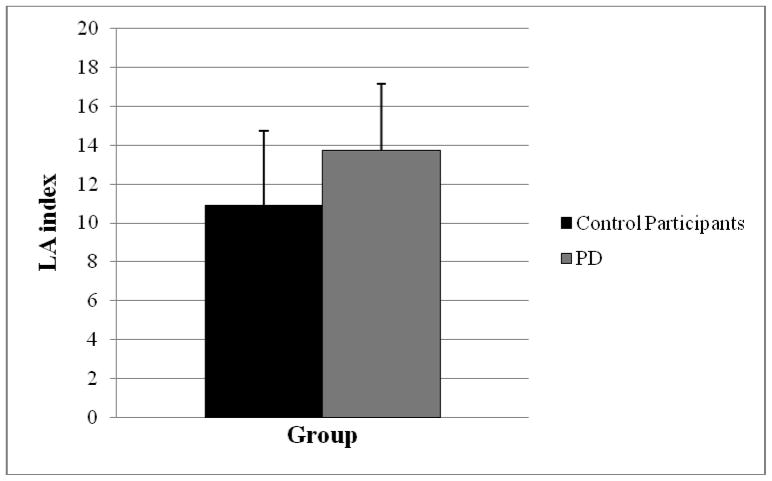
There was a significant effect of group (F(1, 16) = 6.89, p = 0.02). Individuals with PD had a significantly higher LA index than control participants (t(16) = −2.62, p = .02) (see Figure 5). Eight of the nine individuals with PD followed this pattern. There were no significant differences for LA index across the loudness conditions (F(3,47) = 1.36, p = 0.27).
Figure 5.
Means and standard deviations for LA index for the significant group effect. No interaction effects were found for LA index.
DISCUSSION
This study examined lip and jaw kinematics in response to different cues to increase loudness in individuals with PD and age- and sex-matched control participants. Results indicate that individuals with PD and control participants altered articulatory patterns and SPL in response to three types of cues to increase loudness. Both individuals with PD and control participants significantly increased SPL from COMF to the three loud conditions (COMF+10, 2XCOMF, and NOISE). Similarly, opening displacement and peak opening velocity were larger for the three loud conditions as compared to the COMF condition for both individuals with PD and control participants. Although individuals with PD are very responsive to external cueing, they did not produce greater articulatory or SPL responses to the cues to increase loudness as compared to control participants. However, individuals with PD and control participants did not respond to the three cues to increase loudness with the same articulatory patterns.
Control participants appeared to respond best to the NOISE and COMF+10 cues. They produced significantly higher mean SPL in NOISE and COMF+10 than in 2XCOMF. Control participants also demonstrated significantly greater opening displacement in NOISE and COMF+10 than in 2XCOMF. Differences in opening displacement across conditions are consistent with differences in SPL across conditions given that increased lip and jaw opening displacement is expected with increased vocal loudness (Dromey, 2000; Huber and Chandrasekeran, 2007; Schulman, 1989). It has been hypothesized that in order to reduce turbulence caused by increased airflow and pressure through the articulators during loud speech and maintain perceptually appropriate speech sounds, the jaw position becomes more open with loud speech (Schulman, 1989).
Individuals with PD also produced significantly higher mean SPL in NOISE and COMF+10 cues than in 2XCOMF. However, articulatory patterns in individuals with PD were not consistent with loudness changes. While SPL in the NOISE condition was significantly higher than in 2XCOMF, opening displacement and peak opening velocity were significantly smaller in the NOISE condition than in the 2XCOMF condition. This suggests that individuals with PD did not rely solely on mouth opening as a mechanism for increasing loudness in the NOISE condition. In theory, a decreased mouth opening would reduce SPL and overall intelligibility. Thus, individuals with PD had to utilize a different speech subsystem to increase loudness in the NOISE condition and to overcome the reduction in mouth opening.
Sadagopan and Huber (2007) observed the respiratory kinematic patterns in the same group of individuals with PD in response to the same cues to increased loudness, although a different speech task (reading) was used. The SPL results from reading were consistent with the results from the current study; SPL was highest in the NOISE condition and lowest in the 2XCOMF condition. Individuals with PD produced the most efficient respiratory patterns in the NOISE condition as compared to the other loudness conditions. Individuals with PD utilized the mid-lung volume range to the greatest extent in the NOISE condition (Sadagopan & Huber, 2007). This effective use of the respiratory system may have produced large enough gains in SPL to overcome a smaller mouth opening. These results suggest that individuals with PD may utilize the respiratory system to a greater extent than the articulatory system to effect change in loudness levels when speaking in the presence of background noise.
A potential reason behind this finding is that individuals with PD may have a relatively fixed” jaw position and could not produce a larger excursion of the articulators. Forrest et al. (1989) found that the greatest difference between individuals with PD and healthy, age-matched controls was observed in the jaw, with individuals with PD using a relatively “fixed” jaw position (Forrest et al., 1989). In young adults, Huber and Chandrasekaran (2006) found that the jaw more than any other articulator (lower lip and upper lip) demonstrated the most change across the same three cues to increase loudness. Thus, if individuals with PD are unable to change the excursion of their jaw to a large extent due to a relatively “fixed” jaw position, they may need to rely on other subsystems (i.e., respiratory and laryngeal).
In general, opening displacement and peak opening velocity did not significantly differ between the two groups. However, mean data indicate individuals with PD had smaller opening displacements and peak opening velocities across loudness conditions and tasks. Smaller opening displacements and peak opening velocities may have contributed to the perceived articulatory imprecision in some of the speakers. With an increased number of participants or a wider range of impairment severity, significant differences in opening displacement and peak opening velocity may have been found between the groups. Taken with the results relative to SPL change, these results suggest that individuals with PD utilize the articulatory system differently than older adults when increasing vocal loudness.
Control participants and individuals with PD increased loudness to the greatest extent in the external cue conditions (i.e., COMF+10 and NOISE). While loudness did increase from comfortable in the internal cue condition, 2XCOMF, this cue did not result in as large an increase in SPL in either group as the two external cues. This finding is consistent with results from previously discussed limb motor and speech motor literature. Clinicians working with individuals with PD should utilize external cueing, at least in the initial phases of therapy, to achieve increased vocal loudness. Ideally, during later stages of treatment, external cues can be replaced by internal cues.
Relative to the question of stability, individuals with PD had significantly higher LA indices, indicating their movement patterns were less stable than control participants. This finding is consistent with the findings of Walsh (2007). However, while significant, the mean differences were small, suggesting that PD may not grossly impact articulatory stability. Individuals with PD with mild-moderate speech impairment still generate relatively stable articulatory patterns.
This study has several limitations. The main limitation to this study is that the laryngeal system was not investigated. Given that increasing vocal loudness requires all three subsystems, articulatory, laryngeal, and respiratory, to work as an integrated unit, it is impossible to ignore the potential contribution of the laryngeal system to the results of the current study. For instance, while it appears from Sadagopan and Huber (2007) that individuals with PD do utilize mid-lung volume ranges to a larger extent in the NOISE condition thus compensating for decreased mouth opening, increases in laryngeal tension leading to increases in subglottal pressure could also have played a part in the increase in vocal loudness. Future work should include examinations of the laryngeal system. Another limitation of this study is the fact that perceptual ratings of reduced loudness, speech rate, and articulatory precision were not obtained during the loud speech tasks. In order to fully conclude which cue to increase loudness produced the greatest benefits to speech production, measures of perceptual characteristics need to be made using speech samples from each of the loudness conditions.
In summary, control participants and individuals with PD altered articulatory movement patterns in response to cues to increase loudness. However, the two groups responded to the cues to increase loudness in different ways. Control participants produced the smallest articulatory movements in 2XCOMF while individuals with PD produced the smallest articulatory movements in NOISE. Changes in SPL can explain the differences in kinematics for the control participants, but they do not explain the differences in kinematics for the individuals with PD. Individuals with PD may rely more on the respiratory system to drive changes in loudness than the articulatory system. These results indicate the importance of examining more than one speech subsystem to gain a complete picture of speech impairments. Further studies should include a larger number of participants, a wider range of impairment severity, and different types of speech tasks. Further, future research should examine why individuals with PD respond differently to loudness cues than typically aging adults. This difference could be the result of physiologic or cognitive impairments resulting in different interpretations of the goals of the task as a result of the cue.
Acknowledgments
This research was funded by National Institute on Deafness and Other Communication Disorders Grant 1R03DC05731. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Deafness and Other Communication Disorders or the National Institutes of Health.
References
- Ackermann H, Ziegler Z. Articulatory deficits in Parkinsonian dysarthria: an acoustic analysis. Journal of Neurology, Neurosurgery, and Psychiatry. 1991;54:1093–1098. doi: 10.1136/jnnp.54.12.1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker K, Ramig L, Luschei E, Smith M. Thyroarytenoid muscle activity associated with hypophonia in Parkinson disease and aging. Neurology. 1998;51:1592–1598. doi: 10.1212/wnl.51.6.1592. [DOI] [PubMed] [Google Scholar]
- Boersma P, Weenink D. Praat (Version 4.1). [Computer software] Amsterdam, The Netherlands: University of Amsterdam, Institute of Phonetic Sciences; 2003. [Google Scholar]
- Bunton K. Patterns of lung volume use during an extemporaneous speech task in persons with Parkinson’s disease. Journal of Communication Disorders. 2005;38:331–348. doi: 10.1016/j.jcomdis.2005.01.001. [DOI] [PubMed] [Google Scholar]
- Burn D. Parkinson’s disease: an overview. The Pharmaceutical Journal. 2000;246:333–337. [Google Scholar]
- Darley FL, Aronson AE, Brown JR. Differential diagnostic patterns of dysarthria. Journal of Speech and Hearing Research. 1969;12:246–269. doi: 10.1044/jshr.1202.246. [DOI] [PubMed] [Google Scholar]
- Dromey C, Ramig LO, Johnson AB. Phonatory and articulatory changes associated with increased vocal intensity in Parkinson disease: A case study. Journal of Speech and Hearing Research. 1995;38:751–764. doi: 10.1044/jshr.3804.751. [DOI] [PubMed] [Google Scholar]
- Dromey C. Articulatory kinematics in patients with Parkinson disease using different speech treatment approaches. Journal of Medical Speech-Language Pathology. 2000;8:155–161. [Google Scholar]
- Dromey C, Adams S. Loudness Perception and Hypophonia in Parkinson Disease. Journal of Medical Speech-Language Pathology. 2000;8:225–259. [Google Scholar]
- Gazzaniga MS, Ivry RB, Mangun GR. Cognitive neuroscience: The biology of the mind. New York: Norton; 1998. [Google Scholar]
- Hixon T, Goldman M, Mead J. Kinematics of chest wall during speech production: volume displacements of the rib cage, abdomen, and lung. Journal of Speech and Hearing Research. 1973;16:78–115. doi: 10.1044/jshr.1601.78. [DOI] [PubMed] [Google Scholar]
- Ho AK, Iansek R, Marigliani C, Bradshaw JL, Gates S. Speech impairment in a large sample of patients with Parkinson’s disease. Behavioural Neurology. 1999;11:131–137. [PubMed] [Google Scholar]
- Ho AK, Bradshaw JL, Iansek R, Alfredson R. Speech volume regulation in Parkinson’s disease: effects of implicit cues and explicit instructions. Neuropsychologia. 1999;37:1453–1460. doi: 10.1016/s0028-3932(99)00067-6. [DOI] [PubMed] [Google Scholar]
- Ho AK, Bradshaw JL, Iansek R. Volume perception in Parkinsonian Speech. Movement Disorders. 2000;15:1125–1131. doi: 10.1002/1531-8257(200011)15:6<1125::aid-mds1010>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- Hochstadt J, Nakano H, Lieberman P, Friedman J. The roles of sequencing and verbal working memory in sentence comprehension deficits in Parkinson’s disease. Brain and Language. 2006;97:243–257. doi: 10.1016/j.bandl.2005.10.011. [DOI] [PubMed] [Google Scholar]
- Huber JE. Effects of cues to increase sound pressure level on respiratory kinematic patterns during connected speech. Journal of Speech, Language, and Hearing Research. 2007;50:621–634. doi: 10.1044/1092-4388(2007/044). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber JE. Effects of utterance length and vocal loudness on speech breathing in older adults. Respiratory Physiology and Neurobiology. 2008;164:323–330. doi: 10.1016/j.resp.2008.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber JE, Chandrasekaran B. Effects of increasing sound pressure level on lip and jaw movement parameters and consistency in young adults. Journal of Speech, Language, and Hearing Research. 2006;49:1368–1379. doi: 10.1044/1092-4388(2006/098). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Folstein MF, Folstein SE, McHugh PR. Mini Mental State.” A practical method for grading the cognitive state of patients for the clinician. Journal of Psychiatry Research. 1975;12:189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- Forrest K, Weismer G, Turner GS. Kinematic, acoustic, and perceptual analyses of connected speech produced by Parkinsonian and normal geriatric adults. Journal of the Acoustical Society of America. 1989;85:2608–2622. doi: 10.1121/1.397755. [DOI] [PubMed] [Google Scholar]
- Forrest K, Weismer G. Dynamic aspects of lower lip movement in Parkinsonian and neurolgically normal geriatric speakers’ production of stress. Journal of Speech and Hearing Research. 1995;38:260–272. doi: 10.1044/jshr.3802.260. [DOI] [PubMed] [Google Scholar]
- Fox CM, Ramig LO. Vocal sound pressure level and self-perception of speech and voice in men and women with Idiopathic Parkinson disease. American Journal of Speech-Language Pathology. 1997;6:85–94. [Google Scholar]
- Kleinow J, Smith A, Ramig LO. Speech motor stability in IPD: Effects of rate and loudness manipulations. Journal of Speech, Language, and Hearing Research. 2001;44:1041–1051. doi: 10.1044/1092-4388(2001/082). [DOI] [PubMed] [Google Scholar]
- Logemann JA, Fisher HB, Boshes B, Blonsky ER. Frequency and cooccurrence of vocal tract dysfunctions in the speech of a large sample of Parkinson patients. Journal of Speech and Hearing Disorders. 1978;43:47–57. doi: 10.1044/jshd.4301.47. [DOI] [PubMed] [Google Scholar]
- McNamara P, Obler LK, Au R, Durso R, Albert ML. Speech monitoring skillsin Alzheimer’s disease, Parkinson’s disease, and normal aging. Brain and Language. 1992;42:38–51. doi: 10.1016/0093-934x(92)90055-j. [DOI] [PubMed] [Google Scholar]
- Morris ME, Iansek R, Matyas TA, Summers JJ. Stride length regulation in Parkinson’s disease: Normalization strategies and underlying mechanisms. Brain. 1996;119:551–568. doi: 10.1093/brain/119.2.551. [DOI] [PubMed] [Google Scholar]
- Oliveira RM, Gurd JM, Nixon P, Marshall JC, Passingham RE. Micrographia in Parkinson’s disease: the effect of providing external cues. Journal of Neurology, Neurosurgery, and Psychiatry. 1997;63:429–433. doi: 10.1136/jnnp.63.4.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramig L, Countryman S, Thompson L, Horii Y. A comparison of two forms of intensive speech treatment for Parkinson disease. Journal of Speech and Hearing Research. 1995;38:1232–1251. doi: 10.1044/jshr.3806.1232. [DOI] [PubMed] [Google Scholar]
- Ramig L, Fox C, Sapir S. Parkinson’s disease: Speech and voice disorders and their treatment with the Lee Silverman Voice Treatment. Seminars in Speech and Language. 2004;25:169–180. doi: 10.1055/s-2004-825653. [DOI] [PubMed] [Google Scholar]
- Ramig L, Sapir S, Fox C, Countryman S. Intensive voice treatment (LSVT) for patients with Parkinson’s disease: A 2-year follow up. Journal of Neurology, Neurosurgery, & Psychiatry. 2001;71:493–498. doi: 10.1136/jnnp.71.4.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadagopan N, Huber JE. Effects of loudness cues on respiration in individuals with Parkinson’s disease. Movements Disorders. 2007;22:651–659. doi: 10.1002/mds.21375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sapir S, Ramig L, Spielman J, Fox C. Formant Centralization Ratio: A Proposal for a New Acoustic Measure of Dysarthric Speech. Journal of Speech, Language, and Hearing Research. 2010;53:114–125. doi: 10.1044/1092-4388(2009/08-0184). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sapir S, Spielman J, Ramig L, Story B, Fox C. Effects of Intensive Voice Treatment (the Lee Silverman Voice Treatment [LSVT]) on Vowel Articulation in Dysarthric Individuals with Idiopathic Parkinson’s Disease: Acoustic and Perceptual Findings. Journal of Speech, Language, and Hearing Research. 2007;50:899–912. doi: 10.1044/1092-4388(2007/064). [DOI] [PubMed] [Google Scholar]
- Sapir S, Pawlas A, Ramig L, Countryman S, O’Brien C, Hoehn M, Thompson L. Voice and Speech Abnormalities in Parkinson Disease: Relation to Severity of Motor Impairment, Duration of Disease, Medication, Depression, Gender, and Age. Journal of Medical Speech-Language Pathology. 2001;9:213–226. [Google Scholar]
- Schulman R. Articulatory dynamics of loud and normal speech. The Journal of the Acoustical Society of America. 1989;85:295–312. doi: 10.1121/1.397737. [DOI] [PubMed] [Google Scholar]
- Sidaway B, Anderson J, Danielson G, Martin L, Smith G. Effects of long-term gait training using visual cues in an individual with Parkinson disease. Physical Therapy. 2006;86:186–194. [PubMed] [Google Scholar]
- Skodda S, Visser W, Schlegel U. Vowel Articulation in Parkinson’s disease. Journal of Voice. doi: 10.1016/j.jvoice.2010.01.009. (in press) [DOI] [PubMed] [Google Scholar]
- Skodda S, Visser W, Schlegel U. Short- and long-term dopaminergic effects on dysarthria in early Parkinson’s disease. Journal of Neural Transmission. 2010;117:197–205. doi: 10.1007/s00702-009-0351-5. [DOI] [PubMed] [Google Scholar]
- Smith A, Johnson M, McGillem C, Goffman L. On the assessment of stability and patterning of speech movements. Journal of Speech, Language, and Hearing Research. 2000;43:277–286. doi: 10.1044/jslhr.4301.277. [DOI] [PubMed] [Google Scholar]
- Smith M, Ramig L, Dromey C, Perez K, Samandari R. Intensive Voice Treatment in Parkinson Disease: Laryngostroboscopic Findings. Journal of Voice. 1995;9(4):453–459. doi: 10.1016/s0892-1997(05)80210-3. [DOI] [PubMed] [Google Scholar]
- Solomon N, Hixon T. Speech breathing in Parkinson’s disease. Journal of Speech and Hearing Research. 1993;36:294–310. doi: 10.1044/jshr.3602.294. [DOI] [PubMed] [Google Scholar]
- Stathopoulos E, Sapienza C. Developmental Changes in Respiratory and Laryngeal Function with Variations in Sound Pressure Level. Journal of Speech, Language, and Hearing Research. 1997;40:595–614. doi: 10.1044/jslhr.4003.595. [DOI] [PubMed] [Google Scholar]
- Squire LR, Bloom FE, McConnell SK, Roberts JL, Spitzer NC, Zigmond MJ. Fundamental Neuroscience. 2. New York: Elsevier Science; 2003. [Google Scholar]
- Tasko SM, McClean MD. Variations in articulatory movement with changes in speech task. Journal of Speech, Language, and Hearing Research. 2004;47:85–100. doi: 10.1044/1092-4388(2004/008). [DOI] [PubMed] [Google Scholar]
- Walsh B. Speech Production in Individuals with Parkinson’s Disease: Basic Kinematic Parameters and Effects of Increased Linguistic Demands on Interarticulatory Coordination. Dissertation 2007 [Google Scholar]
- Walsh B, Smith A. Articulatory movements in adolescents: evidence of protracted development of speech motor control processes. Journal of Speech, Language, and Hearing Research. 2002;45:1119–1133. doi: 10.1044/1092-4388(2002/090). [DOI] [PubMed] [Google Scholar]
- Willems AM, Nieuwboer A, Chavret F, Desloovere K, Dom R, Rochester L, Jones D, Kwakkel G, Van Wegen E. The use of rhythmic auditory cues to influence gait in patients with Parkinson’s disease, the differential effect for freezers and non-freezers, an explorative study. Disability and Rehabilitation. 2006;28:721–728. doi: 10.1080/09638280500386569. [DOI] [PubMed] [Google Scholar]
- Yorkston KM, Beukelman DR, Strand EA, Bell KR. Management of motor speech disorders in children and adults. 2. Austin, TX: Pro-ed; 1999. [Google Scholar]