Abstract
Context:
Fatigue and underperformance are common in athletes. Understanding overtraining syndrome (OTS) is helpful in the evaluation, management, and education of athletes.
Evidence Acquisition:
Relevant articles in English were searched with OVID (1948-2011) and PubMed using the following keywords: overtraining syndrome, overtraining, overreaching, unexplained underperformance, staleness, pathophysiology, management, treatment, evaluation. Bibliographies were reviewed for additional resources.
Results:
OTS appears to be a maladapted response to excessive exercise without adequate rest, resulting in perturbations of multiple body systems (neurologic, endocrinologic, immunologic) coupled with mood changes. Many hypotheses of OTS pathogenesis are reviewed, and a clinical approach to athletes with possible OTS (including history, testing, and prevention) is presented.
Conclusions:
OTS remains a clinical diagnosis with arbitrary definitions per the European College of Sports Science’s position statement. History and, in most situations, limited serologies are helpful. However, much remains to be learned given that most past research has been on athletes with overreaching rather than OTS.
Keywords: overtraining syndrome, overtraining, overreaching, unexplained underperformance, staleness, pathophysiology, management, treatment, evaluation
Generally speaking, athletes train to increase performance. Performance increases are achieved through increased training loads. Increased loads are tolerated only through interspersed periods of rest and recovery—training periodization. Overreaching is considered an accumulation of training load that leads to performance decrements requiring days to weeks for recovery.14,30 Overreaching followed by appropriate rest can ultimately lead to performance increases.14,30 However, if overreaching is extreme and combined with an additional stressor, overtraining syndrome (OTS) may result.30 OTS may be caused by systemic inflammation and subsequent effects on the central nervous system, including depressed mood, central fatigue, and resultant neurohormonal changes.2,45,46 This article summarizes previous literature and updates the European College of Sport Science’s position statement.30
Terminology
Definitions from the European College of Sport Science position statement on OTS are used30 (Table 1). Symptoms of nonfunctional overreaching (NFO) and OTS are varied, nonspecific, anecdotal, and numerous (Table 2).
Table 1.
Terminology from position statement on overtraining by European College of Sport Science.30
| Term | Synonym | Definition | Performance Decrement | Outcome |
|---|---|---|---|---|
| Functional overreaching | Short-term overreaching | Increased training leading to a temporary performance decrement and with improved performance after rest | Days to weeks | Positive (super- compensation) |
| Nonfunctional overreaching | Long-term overreaching | Intense training leading to a longer performance decrement but with full recovery after rest; accompanied by increased psychologic and/or neuroendocrinologic symptoms | Weeks to months | Negative due to symptoms and loss of training time |
| Overtraining syndrome | Consistent with extreme nonfunctional overreaching but with (1) longer performance decrement (> 2 months), (2) more severe symptomatology and maladapted physiology (psychologic, neurologic, endocrinologic, immunologic systems), (3) and an additional stressor not explained by other disease | Months | Negative due to symptoms and possible end to athletic career |
Table 2.
Symptoms of overtraining syndrome.
| Parasympathetic Alterationsa | Sympathetic Alterationsb | Other |
|---|---|---|
| Fatigue | Insomnia | Anorexia |
| Depression | Irritability | Weight loss |
| Bradycardia | Agitation | Lack of mental concentration |
| Loss of motivation | Tachycardia | Heavy, sore, stiff muscles |
| Hypertension | Anxiety | |
| Restlessness | Awakening unrefreshed |
More common in aerobic sports.
More common in anaerobic sports.
Past terminology includes burnout, staleness, failure adaptation, underrecovery, training stress syndrome, and chronic fatigue. Some researchers refer to overtraining as unexplained underperformance syndrome.5,44 Differentiation of NFO and OTS is clinically difficult and can be made often only after a period of complete rest.30,36 The difference between the 2 is based on time to recovery and not necessarily the degree or type of symptoms.30,36
Many consider overreaching and overtraining as a continuum.10 Others question anecdotal evidence suggesting that NFO precedes OTS.14 Unfortunately, much of the literature has been done on overreached rather than overtrained athletes by current terminology. Some studies used overtrained athletes but failed to show that these athletes suffered from performance impairment.14 Recent reports highlight the importance of psychological and/or social stressors in addition to physiologic stress in the development of NFO/OTS.20,28,49 An individual’s stress capacity plays a role in the development of NFO/OTS.20
Epidemiology
Epidemiologic studies use varied terminology. It appears that OTS is extremely rare, but exact prevalence and incidence data are lacking. *One study found a NFO lifetime prevalence of ~60% in elite male and female runners, compared with 33% in nonelite female runners.35 A multicenter, multicountry survey found that 35% of adolescent swimmers had been “overtrained” at least once.42 Estimates of “staleness” were reported in 5% to 30% of swimmers over a season18,33,37 and in 15% of British elite athletes.21 In the most recent survey of elite adolescent athletes, ~30% reported NFO at least once in their careers—they averaged 2 episodes lasting 4 weeks. The risk was significantly increased in individual sports, low physically demanding sports (such as golf), females, and elite athletes.27
Pathophysiology
Numerous hypotheses have been proposed for OTS, each with strengths and weaknesses (Table 3). In attempting to understand OTS mechanistically and clinically, it is crucial to seek an explanation that accounts for the many symptoms of this complicated entity.
Table 3.
Common hypotheses of overtraining syndrome etiology (arranged in order of complexity).
| Hypothesis | Theory | Strengths | Weaknesses |
|---|---|---|---|
| Glycogen hypothesis | Decreased glycogen causes fatigue and decreased performance | Low glycogen can be correlated with decreased performance and exercise-induced fatigue | No proven correlation in the literature between low glycogen and overtrained athletes Athletes with normal glycogen levels still become overtrained Does not account for all symptoms |
| Central fatigue hypothesis | Increased tryptophan uptake in the brain leads to increased 5-HT centrally and mood symptoms | Exercise correlated with increased tryptophan, 5-HT, and fatigue Rats undergoing intense training have increased 5-HT Selective serotonin reuptake inhibitors decrease performance Athletes getting branched chain amino acids supplement had less fatigue |
Few studies measure 5-HT directly Mood changes/fatigue subjective and difficult to study Mood/fatigue influenced by many other factors Does not account for all symptoms |
| Glutamine hypothesis | Decreased glutamine causes immune dysfunction and increased susceptibility to infection | Glutamine does decrease after prolonged exercise In vitro immune cell function is compromised with decreased glutamine Athletes are more susceptible to upper respiratory tract infections after “intense” exercise |
In vivo, decreased plasma glutamine not necessarily correlated with decreased bioavailable glutamine Glutamine supplementation does not improve postexercise impairment of immune cells Some studies show low glutamine in athletes with upper respiratory tract infections, and some do not Glutamine can be influenced by many other factors Increased upper respiratory tract infections are seen in most athletes after intense exercise, not just overtrained ones Does not account for all symptoms |
| Oxidative stress hypothesis | Excessive oxidative stress causes muscle damage and fatigue | Resting markers of oxidative stress are higher in overtrained athletes and increase with exercise Citrate synthase (marker of oxidative capacity) decreases in overreached rats, so they are more susceptible to oxidative stress |
Studies have been small Lack of clinically relevant research Does not account for all symptoms |
| Autonomic nervous system hypothesis | Parasympathetic predominance causes many symptoms of overtraining syndrome | A study showed variability in autonomic nervous system forces (through heart rate variability) with exercise versus rest Decreased heart rate variability with awakening in overtrained athletes suggests disruption of autonomic nervous system modulation |
Decreased nocturnal catecholamines in overtrained athletes in some studies; no change or increased in others Studies looking at catecholamine excretion with methodological differences are hard to compare No difference in heart rate variability/autonomic nervous system influence between overtrained and control athletes during sleep, when free of external influences Does not account for all symptoms |
| Hypothalamic hypothesis | Dysregulation of the hypothalamus and hormonal axes cause many symptoms of overtraining syndrome | Endurance athletes have activation of the hypothalamic-pituitary-adrenal axis compared with controls | Contradictory data in terms of activation of hypothalamic-pituitary-adrenal/hypothalamic-pituitary-gonadal axes in overtrained athletes and levels of ACTH, cortisol, testosterone Other factors can influence hypothalamic-pituitary-adrenal/hypothalamic-pituitary-gonadal axis activation Does not account for all symptoms |
| Cytokine hypothesis | Inflammation and cytokine release causes most of the above effects and symptoms of overtraining syndrome | Unified theory accounting for many symptoms of overtraining syndrome and “why” it develops Cytokines may act on hypothalamic centers to regulate “sickness” behavior, causing mood changes and fatigue Subacute muscle injury and cytokines decreases glucose transport into muscles, decreases glycogen, and causes fatigue Tryptophan is used to synthesize inflammatory proteins and decreases with systemic inflammation Decreased tryptophan is associated with depressive symptoms Increased cytokine levels are found in depressed patients Giving participants cytokines caused depressive symptoms Cytokines activate the hypothalamic-pituitary-adrenal system (increasing cortisol) and inhibit the hypothalamic-pituitary-gonadal system (decreasing testosterone) Inflammation causes activation of glucose/protein metabolism and decreased glutamine Increased cytokines that favor TH2 lymphocyte activation lead to increased humoral/decreased cell-mediated immunity and more upper respiratory tract infection |
Little evidence actually verifying increased cytokines in overtrained athletes No studies looked at long-term responses to training/overtraining One study showed no change in cytokine levels in overtrained cyclists Cytokine studies to date look predominantly at very fit athletes with questionable application to the general population |
Glycogen Hypothesis
Low muscle glycogen can impair performance because of inadequate fuel for the workload.47 Low muscle glycogen also results in increased oxidation and decreased concentrations of branched chain amino acids. This can alter synthesis of central neurotransmitters involved in fatigue.7 Because decrements in performance and fatigue are hallmarks of OTS, decreased muscle glycogen may cause OTS.
While this association seems plausible, it has not been substantiated in the literature. Swimmers who consume inadequate carbohydrates have more fatigue during training but do not necessarily suffer a performance decrement needed to diagnose OTS.7 Even athletes who consume increased amounts of carbohydrates and maintain normal glycogen levels can still become overtrained.47 While low muscle glycogen levels may be associated with exercise-induced fatigue, the link with OTS appears weak.
Central Fatigue Hypothesis
OTS nearly always includes disrupted mood, sleep, and behavior.2,18,22,33,45 The neurotransmitter serotonin (5-HT) is implicated in regulation of these functions; therefore, alterations in 5-HT could lead to OTS.2-4,22,30,45 5-HT is derived from tryptophan. With exercise, there is increased unbound tryptophan, which competes with branched chain amino acids for entry into the brain.2-4,45 Exercise decreases levels of branched chain amino acids due to increased oxidation, favoring tryptophan entry into the brain and conversion to 5-HT.5
Increases in unbound tryptophan have been positively correlated with fatigue, presumably due to increased serotonin synthesis in the brain.3,4,45 Giving serotonin reuptake inhibitors to athletes artificially increases 5-HT in the brain and reduces performance.5 Conversely, marathon runners receiving branched chain amino acids supplementation felt more energized and mentally clear, possibly due to dampened 5-HT synthesis.6
Fatigue in overtrained athletes may be due to increased sensitivity to 5-HT rather than increased 5-HT.4 Increased neurologic sensitivity to a 5-HT agonist has been reported in overtrained athletes.4 Well-trained athletes usually are less sensitive to 5-HT; this adaptation may be lost in OTS.4
There is fairly consistent evidence that concentrations of 5-HT precursors and prolactin, an indirect measure of central 5-HT activity, are altered in excessive exercise,4 but few studies have actually measured 5-HT activity in overtrained athletes. Mood changes and fatigue are subjective, difficult to measure, and influenced by many confounding factors.4 Therefore, 5-HT activity requires cautious interpretation.
Glutamine Hypothesis
Glutamine is integral for immune cell function.14,16,45 It also plays a role in DNA/RNA synthesis, nitrogen transport, gluconeogenesis and acid-base balance.14,16,53 Decreased glutamine after exercise may be responsible for increased incidences of upper respiratory tract infections in overtrained athletes.14,25,46
Prolonged exercise (> 2 hours) or repeated bouts of high-intensity exercise can transiently decrease plasma glutamine concentrations.16,53 Low plasma glutamine concentrations have been reported specifically in overtrained athletes.14,25 This may represent overutilization and/or a decreased production by overworked muscles.16
It is unclear whether decreased glutamine influences immune cell function. In vitro, immune cell function can be compromised when glutamine concentrations are below physiologic levels.16 Despite decreased glutamine concentrations after exercise, the amount of glutamine available to immune cells does not necessarily change.16 Glutamine supplementation can restore physiologic levels but does not improve postexercise impairment of immune cells.16 Nevertheless, glutamine supplementation may decrease early rates of infection among athletes.6
Epidemiologic data have shown that athletes may be more susceptible to upper respiratory tract infections after “excessive exercise,”53 but there is no evidence that immunosuppression is greater in athletes with NFO/OTS. In one study, 13% of overreached athletes compared with 56% of nonoverreached athletes reported upper respiratory tract infections.25 Low plasma glutamine has been seen in athletes with upper respiratory tract infections, while others demonstrate the opposite.53 Finally, glutamine levels can be influenced by nutritional state, trauma, and infection.16
Oxidative Stress Hypothesis
Some oxidative stress is desired during exercise because reactive oxygen species released from damaged muscles regulate cellular repair.50 When oxidative stress becomes pathologic, however, reactive oxygen species (ie, superoxide, hydrogen peroxide, and hydroxyl radical) can cause inflammation, muscle fatigue, and soreness with resultant inhibition of athletic performance.48
Resting markers of oxidative stress are higher in overtrained athletes compared with controls.17,26,48 Moreover, oxidative stress markers increase with exercise in the overtrained athletes.26,48 Citrate synthase reflects oxidative capacity and is expected to increase during endurance training. In an overreached rat model, citrate synthase activity actually decreases.17 OTS athletes may have diminished responses to exercise-induced stress and be more susceptible to oxidative damage.17
It is unclear if the increased oxidative stress state is a trigger or result of OTS. Clinically relevant research is limited. Moreover, confounding factors such as menstrual cycles and estrogen’s antioxidative properties are unknown.48
Autonomic Nervous System Hypothesis
An imbalance in the autonomic nervous system can explain some symptoms of OTS. Specifically, decreased sympathetic activation and parasympathetic dominance can lead to performance inhibition, fatigue, depression, and bradycardia.14,22,45
Reduced sympathetic activation in overtrained athletes is supported in some studies by decreased nocturnal urinary catecholamine excretion.14 Catecholamine excretion decreases with increasing fatigue and returns to baseline during recovery.14,20,45 However, not all studies find this trend.12,20 A decreased organ sensitivity to catecholamines may also be responsible for symptoms of decreased sympathetic activation.22
Heart rate variability (HRV) has also been used as an indicator of autonomic function.9,14,51 One study showed no difference in HRV between overtrained and control athletes during sleep.19 However, a reduced HRV was seen soon after awakening in overtrained athletes, suggesting increased sympathetic tone.19 Using HRV, studies have indicated that the effects of intense training on automatic control may be reversible. Balance between sympathetic and parasympathetic forces may be restored after a week of rest.41
Hypothalamic Hypothesis
Alterations in the hypothalamic-pituitary-adrenal (HPA) and hypothalamic-pituitary-gonadal axes may be responsible for OTS. Endurance athletes may show subtle changes in function of the HPA axis, and overtrained athletes can have alterations in cortisol, adrenocorticotropic hormone, testosterone, and other hormone levels.1,14,22,45,51 Unfortunately, the current data are contradictory as to patterns of these hormonal changes.14,22,51 Alterations in the HPA and hypothalamic-pituitary-gonadal axes are individualized and depend on other factors, including exercise capacity, inherent vulnerability to stressors, and other hormonal levels.14,51
Cytokine Hypothesis
No single hypothesis explains all aspects of OTS. The cytokine hypothesis suggests that OTS is a physiologic adaptation/maladaptation to excess stress initiated by an imbalance between training and recovery.44-46
Muscle contraction and repetitive joint action cause microtrauma to tissues.44,45 Adaptation through tissue healing and strengthening occurs via activation of a local inflammatory response and recruitment of cytokines.44,45 With continued intense training and absence of adequate rest, this inflammatory response can become amplified, chronic, and pathologic.44-46 Eventually a systemic inflammatory response can result with negative consequences throughout the body.44,45 Implicated cytokines in OTS include interleukin 1 beta (IL-1b), IL-6, and tumor necrosis factor α (TNF-α)45 (Figure 1).
Figure 1.
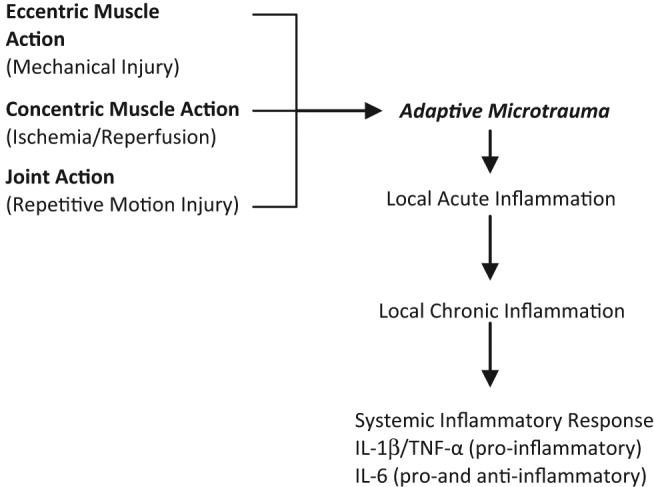
Proposed etiology of OTS through exercise and resultant inflammation. There is a time-dependant sensitization with amplification at each step to repeated intermittent stimuli over time.44,45
Reduced muscle glycogen levels are frequently observed in overtrained athletes.7,47 Decreased glycogen may be a consequence of OTS through cytokine-mediated effects.45 Cytokines acting on hunger centers in the hypothalamus induce anorexia, resulting in decreased glycogen stores.45 Cytokines themselves may interfere with glucose transport into muscle cells for glycogen synthesis. Studies have shown decreased concentrations of GLUT-4 transporters in stressed muscles due to downregulation of protein synthesis by TNF-α.45 Decreased glycogen stores may account for feelings of heavy legs and muscular fatigue in overtrained athletes.45
Increased uptake of tryptophan into the brain and enhanced sensitivity to serotonin have been postulated to induce fatigue and depression.45 The cytokine hypothesis argues that serum tryptophan levels are actually decreased with systemic inflammation like that seen in OTS.45 The reason is that tryptophan is used for the synthesis of inflammatory-related proteins. Reduced tryptophan levels have actually been associated in many studies with depressive symptoms.45
Behavioral and psychological changes seen in OTS can also be attributed to cytokines. Proinflammatory IL-1b and TNF-α act on the brain to cause decreased appetite, sleep disturbance, and depression, referred to as “sickness” behavior. Cytokines may act directly on central receptors, or they may activate the HPA axis and release of stress hormones with similar peripheral effects.45 There is evidence of elevated cytokine levels in depressed patients and a precipitation of depressed mood with cytokine administration to participants, offering credibility to the notion of cytokine-induced mood changes in OTS.45
Alterations in the HPA and hypothalamic-pituitary-gonadal axes with a resultant decrease in testosterone:cortisol ratios have been implicated in OTS. Proinflammatory cytokines are potent activators of the HPA system, which cause release of corticotropin-releasing hormone, adrenocorticotropic hormone, and cortisol. These cytokines suppress testosterone through central inhibition.45 Although patterns of hormonal changes in OTS are varied, cytokine mediators may be responsible for some of these changes.
Plasma glutamine is decreased in overtrained athletes, and this can be attributed to its increased utilization for various cytokine-controlled processes.14,16,45 Glutamine is a precursor for synthesis of inflammatory proteins, which are upregulated with overtraining. IL-6 and TNF-α actually stimulate glutamine uptake into hepatocytes for this purpose.45 In addition, systemic inflammation induces a catabolic state with a necessary increase in glucose and protein metabolism. Glutamine is a crucial element for gluconeogenesis and essential in aiding renal nitrogen excretion with increased proteolysis.14,16,45
As previously mentioned, increased susceptibility to infection in overtrained athletes has been blamed on decreased glutamine without much support.14,25,46 There are 2 subsets of T helper cells: TH1 lymphocytes associated with cell-mediated immunity and TH2 lymphocytes linked to humoral immunity.46 Which subset predominates depends on the prevailing cytokine milieu. OTS causes activation of cytokines that favor a TH2 lymphocyte profile. Indeed, multiple studies show increases in TH2-favoring cytokines (IL-6, TNF-α, IL-10) after a marathon and no detectable levels of TH1-related cytokines.46 TH2 predominance causes increased humoral immunity but with a concomitant downregulation of TH1-sponsored cell-mediated immunity and reduced protection against upper respiratory tract infection.45,46
The cytokine theory does provide a comprehensive paradigm to explain many features of OTS. It views systemic inflammation as the underlying basis for overtraining and draws parallels between overtraining and other stress conditions. Unlike many other theories, it proposes to answer “why” OTS develops. It presents a primary stimulus that causes activation of many biochemical pathways and correlates activation of these pathways with symptoms observed in overtrained athletes.45
Although mechanistically sophisticated, the theory does have some concerning limitations. Despite a plausible correlation between cytokines and symptoms of overtraining, there is scant evidence showing elevated cytokine levels in overtrained individuals.45 Some studies have demonstrated acute increases in IL-2 and IL-6 following exercise, but long-term responses to training were not explored.8,9,44 One study specifically looked at overtrained cyclists but found no changes in IL-6 or TNF-α concentrations.15 It argued that because muscle contraction during cycling is largely concentric, perhaps there is not a great-enough degree of microtrauma to induce an inflammatory cascade. This seems reasonable, but other studies have found increased IL-6 levels in cyclists.8 Athletes in many of the cytokine studies were quite fit at baseline and so it is questionable whether conclusions drawn are similarly applicable to a general population of athletes.
These are justifiable concerns, but it is uncertain whether they are enough to discredit the mounting body of evidence linking cytokines with OTS. More research is needed before a definitive correlation is demonstrated between elevated cytokine levels and overtrained athletes.
Diagnosis
History
Patients will primarily present with unexplained underperformance. Diagnosis of OTS is clinical and accomplished through history,30 which should demonstrate the following: (1) decreased performance persisting despite weeks to months of recovery, (2) disturbances in mood, and (3) lack of signs/symptoms or diagnosis of other possible causes of underperformance.30 The list of organic diseases that can result in underperformance is extensive and not limited to the following: undiagnosed asthma/bronchial hyperreactivity, thyroid disease, adrenal disease, diabetes mellitus or insipidus, iron deficiency with or without anemia, infection (myocarditis, hepatitis, HIV, etc), and malnutrition (due to eating disorders, celiac sprue, etc).30 An extensive nutrition history should be obtained along with an assessment of caloric expenditure.30
If an athlete presents with underperformance without a period of rest and recovery, OTS cannot be diagnosed. By definition, such patient has functional overreaching (FO) versus NFO with possible OTS.30 The diagnosis from history can be made only in retrospect given the definitions of NFO and OTS. If less than 14 to 21 days of rest are required for return to previous performance, NFO would be diagnosed. If it has been greater than 14 to 21 days of rest, OTS is diagnosed by some.30
Often, underperformance will be met with an increase in training volume and intensity to improve results. History should include assessment of possible triggers30 (Table 4). In addition, certain historical clues should raise suspicion of NFO/OTS over organic diseases. These clues include the ability to start a training session but the inability to complete and/or a loss of finishing kick.3 While mood symptoms can coexist with organic processes, the presence of pervasive mood changes in the proper setting may signal NFO/OTS versus a primary mood disorder.2
Table 4.
Potential triggers of overtraining syndrome.30
| Increased training load without adequate recovery |
| Monotony of training |
| Excessive number of competitions |
| Sleep disturbances |
| Stressors including personal life (family, relationships) and occupational |
| Previous illness |
| Altitude exposure |
| Heat injury episode |
| Severe “bonk” |
Testing
Ruling out organic diseases leading to underperformance is driven by history.30 The sports medicine provider may consider screening tests to include comprehensive metabolic panel (including kidney function, potassium, magnesium, and glucose), complete blood count, erythrocyte sedimentation rate, C-reactive protein, iron studies, creatine kinase, and thyroid stimulating hormone. Metabolic issues (ie, mitochondrial, glycogen storage, and lipid peroxidation diseases) and cardiovascular disease should be considered in athletes new to higher intensities and volumes.41 Cardiovascular disease should be investigated in master athletes or someone with a positive family history.
In one of the few studies describing evaluation of underperforming athletes, a cause for repeated infections and/or fatigue was found in 68% of regional- or higher-level athletes.43 Ninety-three percent of the athletes reported decreases in performance. This evaluation was significant for a more extensive and costly laboratory evaluation, including B12, folate, serology for viral hepatitis, toxoplasmosis, cytomegalovirus/Epstein-Barr virus titers, Epstein-Barr virus DNA in saliva, serum and salivary immunoglobulins (serum immunoglobulin G subclasses and specific serum immunoglobulin E to aeroallergens), and antinuclear antibodies.43 Such extensive testing should be considered only in the appropriate setting. In those athletes with atopic complaints (allergic rhinitis, dyspnea and/or exertional cough, eczema), pulmonary function tests should be considered.41,43
Biochemical markers have been studied in a variety of athlete populations. There have been no specific or sensitive levels defined for creatine kinase, urea, or iron.51 Recently, oxidative stress biomarkers have been found to correlate well with training load and overreached status,26 but such markers remain impractical in standard laboratories. Hematologic markers have also been disappointing. Recently plasma viscosity > 1.44 was found to be specific but not sensitive for NFO.52
Immunologic markers have also been studied with variable results. Two studies have found increased levels of T cell activation, but such testing lacks practicality.9,11 Most recent studies focus on salivary immunoglobulin A. In endurance athletes, lower levels of salivary immunoglobulin A have been correlated with increased incidence of upper respiratory tract infection symptoms.23 One study showed 18% to 32% lower levels in athletes with symptoms of overtraining, but no performance measures were reported.24 However, another study did not find a statistically significant decrease of salivary immunoglobulin A after intensified training in cyclists.15 Most would agree that excessive exercise, FO, NFO, and OTS can result in impairment in cell-mediated immunity, but there does not appear to be an immunologic marker that differentiates them.30
Hormonal markers have shown some promising results but have a multitude of confounding variables (ie, diurnal and seasonal timing, phase of menstrual cycle, nutritional status).14,30 Resting cortisol levels have not differentiated between athletes with and without NFO/OTS. Testosterone results have been contradictory. Some have suggested that a decreased testosterone:cortisol ratio can be diagnostic of NFO and/or OTS. However, the ratio represents the physiologic strain of training rather than the athlete’s maladaption to that stress. The utility of testosterone:cortisol ratio has not been supported by the literature.14
Cortisol has a peak during the day with nadir during night.12 Morning cortisol does not accurately reflect levels of free cortisol.12 Free cortisol is filtered by the kidney at a constant rate; therefore, 24-hour and overnight urinary-free cortisol have been more studied.12 However, even overnight excretion shows high interindividual variability.12 Cortisol (catabolic and anti-inflammatory) is converted to inactive cortisone by 11β-HSD2. A prospective study found a clinically significant increase in overnight urinary cortisol:cortisone ratio during a high training load period in triathletes, who subsequently underperformed and reported fatigue.12 It is proposed that cytokines may inhibit 11β-HSD2 activity and result in relative increases in cortisol and, hence, catabolism.12 However, overnight urinary cortisol:cortisone ratio remains in the research realm and awaits validation.
Clinically, it would be helpful to know what level of training signals the change from FO to NFO and from NFO to OTS. However, there are no specific and validated blood markers for NFO or OTS.51 Therefore, some have focused on physiologic tests. A couple recent studies show promise for discriminating NFO from OTS. A 2-bout maximal exercise protocol reportedly diagnoses FO, NFO, and OTS by investigating the HPA axis response.31,32 The bouts of maximal exercise are separated by 4 hours. It is believed that FO, NFO, and OTS represent a disturbance, an adaptation, and eventual maladaption of the HPA axis. In an early study, FO athletes had a less pronounced hormonal response to a second bout of maximal exercise in comparison with the extremely exaggerated response in NFO athletes.32 These findings were in contrast to the extreme response to the first bout of exercise and absence of response to the second bout in OTS athletes.31 This first study displayed great utility in picking up FO before NFO.32 In the most recent study of 2-bout maximal exercise protocol, reliable differentiation of NFO from OTS was achieved. However, the diagnosis of NFO and OTS was made retrospectively and arbitrarily defined OTS as greater than 1 year of symptoms. Athletes were tested after having symptoms ranging from 2 weeks to 1 year and prospectively followed for resolution of symptoms. After a second bout, NFO athletes showed very large increases in adrenocorticotropic hormone and prolactin, whereas OTS athletes showed absent or very limited increases. The study’s authors propose that the different responses are due to hypersensitivity of glucocorticoid receptors in NFO compared with insensitivity in OTS. This would seem to confirm a disturbance in the HPA and give credence to the close relationship between OTS and other stress-related syndromes, such as major depression and posttraumatic stress disorder.31 If findings can be replicated, 2-bout maximal exercise protocol may have prognostic value by identifying athletes requiring greater than 1 year to recover.
Physiologic testing with resting heart rate, maximal heart rate, and HRV do not show consistent results, nor do they allow differentiation of FO, NFO, and OTS. Researchers have argued that use of performance tests for diagnosis is “polluted” by the underperformance inherent to NFO and OTS.29 However, the most consistent finding is a diminished maximal lactate in OTS, but it was not sensitive enough to rule out NFO.30 In an analysis of the literature, greater performance decreases were found in studies that report time to fatigue versus studies that report more functional performance measures (ie, 100- or 400-m swim, 15-minute time trial).14
Most agree that psychological distress is required for diagnosis of OTS. The best-studied measure is Profile of Mood States scores. In 2 studies, 81% of “stale” swimmers were identified with the Profile of Mood States questionnaire.34,37 However, increased scores can be seen in athletes with increased training without FO/NFO34 and have not always been reported with performance measures.37 Regardless, mood questionnaires can be prospectively followed, are cheap, and are quickly available. Variables such as timing of testing in relation to training must be standardized. In a case study of an NFO athlete, it was suggested that decreased Profile of Mood States vigor score may be more specific to overreaching than the Profile of Mood States fatigue score. It is suggested that fatigue is a “normal experience” in athletes, whereas decreased vigor can be viewed as maladaptive.13
Treatment
Treatment varies based on the etiology for the underperformance,30 and any organic disease should be treated appropriately. No treatment is required for FO other than balancing overload training with appropriate recovery.30 Treatment of NFO and OTS is rest; however, some propose that relative rest is more appropriate.3 It is recommended to build up volume prior to intensity, starting from 5 to 10 minutes daily until 1 hour is tolerated.3 It is unclear which strategy is best, so the motivation for exercise, internal versus external, should be considered when recommending complete versus relative rest. Given the significant psychological overlay, one may consider involving a sports psychologist or other mental health expert in multidisciplinary management.30 If stress, depression, and/or anxiety are increased with full rest, relative rest with well-defined expectations should be provided.30
Treatment with selective serotonin reuptake inhibitor is suggested by some based on similarities between neuroendocrinologic changes between depression and OTS.2,38 However, one should be cautious of increased heat stress and possible decreased performance with antidepressant treatment in athletes. In addition, if sleep complaints are prominent, treatment with trazodone or amitriptyline could be considered.38
Prevention
Given the unethical nature of inducing OTS in athletes and uncertain pathogenesis, there are no evidence-based means of preventing OTS. However, observation of training load, performance measures, and mood questionnaires can help interrupt the progression from FO to NFO/OTS.30,33,34 A study has shown a decrease in “burnout” in collegiate swimmers from 10% to zero when altering training load in response to the Profile of Mood States questionnaire. When the mood state decreased, training load was also decreased.33,34
Major components of prevention are screening and education. One should educate athletes at risk for overtraining that one of the initial signs of overreaching is increased rating of perceived exertion for a given workload.34 In addition, sports medicine providers may consider preemptively asking if training has increased to compensate for decreases in performance. History of athletes should include inquiry about training (monotony, excessive load, sudden increase, caloric/hydration needs in relation to load) and personal stressors (interpersonal, family, sleep, travel)30 (Tables 4 and 5).
Table 5.
| Periodization of training |
| Tapering for competition |
| Adjust training volume and intensity based on performance and mood |
| Ensure adequate calories for training load |
| Ensure adequate hydration |
| Ensure adequate carbohydrate ingestion during exercise |
| Ensure adequate sleep |
| Promoting mental toughness or resilience as buffer |
| Rest period of greater than 6 hours between exercise bouts |
| Abstinence of training following infection, heat stroke/stress, periods of high stress |
| Avoid extreme environmental conditions |
| Utilize Profile of Mood States (or stress level) and alter training load |
Future Directions
Some hope that a recently developed animal model of overtraining may help guide further hypothesis testing of NFO and OTS.17 A very promising technology being studied in elite athletes is Fourier transform infrared spectroscopy. A group of researchers are monitoring athletes prospectively, and they propose that changes in metabolism can be appreciated by Fourier transform infrared spectroscopy before symptoms and objective performance decrements are clinically appreciated.39,40 Their results are very thought-provoking but require independent validation.
Others are suggesting further investigation of psychomotor speed testing as a means of diagnosing OTS.36 In a survey of the literature, psychomotor speed is known to be decreased in many different pathologies, most notably major depression and chronic fatigue syndrome, which share many characteristics with NFO/OTS.36 One study showed differences in psychomotor speed between NFO/OTS athletes and control athletes. However, a decrease in psychomotor speed was also found in overload training without FO and in a second group that did show FO.36
Conclusion
OTS remains a clinical diagnosis with arbitrary definitions per the European College of Sports Science’s position statement.30 History and, in most situations, limited serologies are paramount. Many pathophysiologic hypotheses have been proposed. Currently, it appears that OTS represents a systemic inflammatory process with diffuse effects on the neurohormonal axis affecting host immunology and mood.30 OTS is a maladapted response to exercise when excessive and not matched with appropriate rest.30
Footnotes
References
- 1. Angeli A, Minetto M, Dovio A, et al. The overtraining syndrome in athletes: a stress-related disorder. J Endocrinol Invest. 2004;27:603-612 [DOI] [PubMed] [Google Scholar]
- 2. Armstrong LE, VanHeest JL. The unknown mechanism of the overtraining syndrome: clues from depression and psychoneuroimmunology. Sports Med. 2002;32:185-209 [DOI] [PubMed] [Google Scholar]
- 3. Budgett R. Fatigue and underperformance in athletes: the overtraining syndrome. Br J Sports Med. 1998;32:107-110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Budgett R, Hiscock N, Arida R, et al. The effects of the 5-HT2C agonist m-chlorphenylpiperazine on elite athletes with unexplained underperformance syndrome (overtraining). Br J Sports Med. 2010;44:280-283 [DOI] [PubMed] [Google Scholar]
- 5. Budgett R, Newsholme E, Lehmann M, et al. Redefining the overtraining syndrome as the unexplained underperformance syndrome. Br J Sports Med. 2000;34:67-68 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Castell LM, Poortmans JR, Leclercq R, et al. Some aspects of the acute phase response after a marathon race, and the effects of glutamine supplementation. Eur J Appl Physiol. 1997;75:47-53 [DOI] [PubMed] [Google Scholar]
- 7. Costill DL, Flynn MG, Kirwan JP, et al. Effects of repeated days of intensified training on muscle glycogen and swimming performance. Med Sci Sports Exerc. 1988;20:249-254 [DOI] [PubMed] [Google Scholar]
- 8. Edwards KM, Burns VE, Ring C, Carroll D. Individual differences in the interleukin-6 response to maximal and submaximal exercise takes. J Sports Sci. 2006;24(8):855-862 [DOI] [PubMed] [Google Scholar]
- 9. Fry RW, Grove JR, Morton AR, et al. Psychological and immunological correlates of acute overtraining. Br J Sports Med. 1994;28(4):241-246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Fry RW, Morton AR, Keast D. Overtraining in athletes: an update. Sports Med. 1991;12(1):32-65 [DOI] [PubMed] [Google Scholar]
- 11. Gabriel H, Kindermann W. The acute immune response to exercise: what does it mean? Int J Sports Med. 1997;18(1)(suppl):S28-S45 [DOI] [PubMed] [Google Scholar]
- 12. Gouarne C, Groussard C, Gratas-Delamarche A, et al. Overnight urinary cortisol and cortisone add new insights into adaptation to training. Med Sci Sports Exerc. 2005;37:1157-1167 [DOI] [PubMed] [Google Scholar]
- 13. Gustafsson H, Holmberg H, Hassmen P. An elite endurance athlete’s recovery from underperformance aided by a multidisciplinary sport science support team. Eur J Sport Sci. 2008;8(5):267-276 [Google Scholar]
- 14. Halson SL, Jeukendrup AE. Does overtraining exist? An analysis of overreaching and overtraining research. Sports Med. 2004;34(14):967-981 [DOI] [PubMed] [Google Scholar]
- 15. Halson SL, Lancaster GI, Jeukendrup AE, et al. Immunological responses to overreaching in cyclists. Med Sci Sports Exerc. 2003;35(5):854-861 [DOI] [PubMed] [Google Scholar]
- 16. Hiscock N, Pedersen BK. Exercise-induced immunosuppresion: plasma glutamine is not the link. J Appl Physiol. 2002;93:813-822 [DOI] [PubMed] [Google Scholar]
- 17. Hohl R, Ferraresso RL, DeOliveira RB, et al. Development and characterization of an overtraining animal model. Med Sci Sports Exerc. 2009;41(5):1155-1163 [DOI] [PubMed] [Google Scholar]
- 18. Hooper S, MacKinnon LT, Hanrahan S. Mood states as an indication of staleness and recovery. Int J Sport Psychol. 1997;28:1-12 [Google Scholar]
- 19. Hynynen A, Uusitalo A, Konttinen N, et al. Heart rate variability during night sleep and after awakening in overtrained athletes. Med Sci Sports Exerc. 2006;38(2):313-317 [DOI] [PubMed] [Google Scholar]
- 20. Kentta G, Hassmen P. Overtraining and recovery: a conceptual model. Sports Med. 1998;26(1):1-16 [DOI] [PubMed] [Google Scholar]
- 21. Koutedakis Y, Sharp NC. Seasonal variations of injury and overtraining in elite athletes. Clin J Sport Med. 1998;8(1):18-21 [DOI] [PubMed] [Google Scholar]
- 22. Lehmann M, Foster C, Keul J. Overtraining in endurance athletes: a brief review. Med Sci Sports Exerc. 1993;25(7):854-862 [DOI] [PubMed] [Google Scholar]
- 23. Mackinnon LT. Chronic exercise training effects on immune function. Med Sci Sports Exerc. 2000;32(7)(suppl):S369-S376 [DOI] [PubMed] [Google Scholar]
- 24. Mackinnon LT, Hooper SL. Mucosal (secretory) immune system responses to exercise of varying intensity and during overtraining. Int J Sports Med. 1994;15(3)(suppl):S179-S183 [DOI] [PubMed] [Google Scholar]
- 25. Mackinnon LT, Hooper SL. Plasma glutamine and upper respiratory tract infection during intensified training in swimmers. Med Sci Sports Exerc. 1996;28(3):285-290 [DOI] [PubMed] [Google Scholar]
- 26. Margonis K, Fatouros IG, Jamurtas AZ, et al. Oxidative stress biomarkers responses to physical overtraining: implications for diagnosis. Free Radic Biol Med. 2007;43(6):901-910 [DOI] [PubMed] [Google Scholar]
- 27. Matos NF, Winsley RJ, Williams CA. Prevalence of non-functional overreaching/overtraining in young English athletes. Med Sci Sports Exerc. 2011;43(7):1287-1294 [DOI] [PubMed] [Google Scholar]
- 28. Meehan HL, Bull SJ, Wood DM, et al. The overtraining syndrome: a multicontextual assessment. Sports Psychol. 2004;18:154-171 [Google Scholar]
- 29. Meeusen R, Duclos M, Gleeson M, et al. The overtraining syndrome: facts and fiction. Eur J Sport Sci. 2006;6(4):263 [Google Scholar]
- 30. Meeusen R, Duclos M, Gleeson M, et al. Prevention, diagnosis and treatment of the overtraining syndrome: ECSS Position Statement Task Force. Eur J Sport Sci. 2006;6(1):1-14 [Google Scholar]
- 31. Meeusen R, Nederhof E, Buyse L, et al. Diagnosing overtraining in athletes using the two-bout exercise protocol. Br J Sports Med. 2010;44:642-648 [DOI] [PubMed] [Google Scholar]
- 32. Meeusen R, Piacentini MF, Busschaert B, et al. Hormonal response in athletes: the use of a two bout exercise protocol to detect subtle differences in (over)training status. Eur J Appl Physiol. 2004;91:140-146 [DOI] [PubMed] [Google Scholar]
- 33. Morgan WP, Brown Dr, Raglin JS, et al. Psychological monitoring of overtraining and staleness. Br J Sports Med. 1987;21:107-114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Morgan WP, Costill DL, Flynn MG, et al. Mood disturbance following increased training in swimmers. Med Sci Sports Exerc. 1988;20(4):408-414 [DOI] [PubMed] [Google Scholar]
- 35. Morgan WP, O’Connor P, Sparling P, et al. Psychological characterizations of the elite female distance runner. Int J Sports Med. 1987;8:124-131 [DOI] [PubMed] [Google Scholar]
- 36. Nederhof E, Lemmeink K, Visscher C, et al. Psychomotor speed, possibly a new marker for overtraining syndrome. Sports Med. 2006;36:817-828 [DOI] [PubMed] [Google Scholar]
- 37. O’Connor PJ, Morgan WP, Raglin JS, et al. Mood state and salivary cortisol levels following overtraining in female swimmers. Psychoneuroendocrinology. 1989;14(4):303-310 [DOI] [PubMed] [Google Scholar]
- 38. Pearce PZ. A practical approach to the overtraining syndrome. Curr Sports Med Rep. 2002;1:179-183 [DOI] [PubMed] [Google Scholar]
- 39. Petibois C, Cazoral G, Deleris G. FT-IR spectroscopy utilization to sportsmen fatigability evaluation and control. Med Sci Sports Exerc. 2000;32(10):1803-1808 [DOI] [PubMed] [Google Scholar]
- 40. Petibois C, Cazoral G, Poortmans JR, et al. Biochemical aspects of overtraining in endurance sports: the metabolism alteration process syndrome. Sports Med. 2003;33:83-94 [DOI] [PubMed] [Google Scholar]
- 41. Pichot V, Roche F, Gaspoz FE. Relation between heart rate variability and training load in middle-distance runners. Med Sci Sports Exerc. 2000;32:1729-1736 [DOI] [PubMed] [Google Scholar]
- 42. Raglin J, Sawamura S, Alexiou S, et al. Training practice and staleness in 13-18-year-old swimmers: a cross-cultural study. Pediatr Exerc Sci. 2000;12:61-70 [Google Scholar]
- 43. Reid VL, Gleeson M, Williams N, et al. Clinical investigation of athletes with persistent fatigue and/or recurrent infections. Br J Sports Med. 2004;38:42-45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Robson PJ. Elucidating the unexplained underperformance syndrome in endurance athletes: the interleukin-6 hypothesis. Sports Med. 2003;33:771-781 [DOI] [PubMed] [Google Scholar]
- 45. Smith LL. Cytokine hypothesis of overtraining: a physiological adaptation to excessive stress? Med Sci Sports Exerc. 2000;32:317-331 [DOI] [PubMed] [Google Scholar]
- 46. Smith LL. Overtraining, excessive exercise, and altered immunity: is this a T helper-1 versus T helper-2 lymphocyte response? Sports Med. 2003;33(5):347-364 [DOI] [PubMed] [Google Scholar]
- 47. Snyder AC, Kuipers H, Cheng B, et al. Overtraining following intensified training with normal muscle glycogen. Med Sci Sports Exerc. 1995;27(7):1063-1070 [DOI] [PubMed] [Google Scholar]
- 48. Tanskanen M, Atalay M, Uusitalo A. Altered oxidative stress in overtrained athletes. J Sports Sci. 2010;28(3):309-317 [DOI] [PubMed] [Google Scholar]
- 49. Tenenbaum G, Jones CM, Kitsantas A, et al. Failure adaptation: psychological conceptualization of the stress response process in sport. Int J Sport Psychol. 2003;34:1-26 [Google Scholar]
- 50. Tiidus PM. Radical species in inflammation and overtraining. Can J Physiol Pharmacol. 1998;76:533-538 [DOI] [PubMed] [Google Scholar]
- 51. Urhausen A, Kindermann W. Diagnosis of overtraining: what tools do we have? Sports Med. 2002:32;95-102 [DOI] [PubMed] [Google Scholar]
- 52. Varelet-Marie E, Mercier J, Brun J. Is plasma viscosity a predictor of overtraining in athletes? Clin Hemorheol Microcirc. 2006;35:329-332 [PubMed] [Google Scholar]
- 53. Walsh NP, Blannin AK, Robson PJ, Gleeson M. Glutamine, exercise and immune function. Sports Med. 1998;28(3):177-191 [DOI] [PubMed] [Google Scholar]



