Abstract
AIM: To examine effects of chronic methadone usage on bowel visualization, preparation, and repeat colonoscopy.
METHODS: In-patient colonoscopy reports from October, 2004 to May, 2009 for methadone dependent (MD) patients were retrospectively evaluated and compared to matched opioid naive controls (C). Strict criteria were applied to exclude patients with risk factors known to cause constipation or gastric dysmotility. Colonoscopy reports of all eligible patients were analyzed for degree of bowel visualization, assessment of bowel preparation (good, fair, or poor), and whether a repeat colonoscopy was required. Bowel visualization was scored on a 4 point scale based on multiple prior studies: excellent = 1, good = 2, fair = 3, or poor = 4. Analysis of variance (ANOVA) and Pearson χ2 test were used for data analyses. Subgroup analysis included correlation between methadone dose and colonoscopy outcomes. All variables significantly differing between MD and C groups were included in both univariate and multivariate logistic regression analyses. P values were two sided, and < 0.05 were considered statistically significant.
RESULTS: After applying exclusionary criteria, a total of 178 MD patients and 115 C patients underwent a colonoscopy during the designated study period. A total of 67 colonoscopy reports for MD patients and 72 for C were included for data analysis. Age and gender matched controls were randomly selected from this population to serve as controls in a numerically comparable group. The average age for MD patients was 52.2 ± 9.2 years (range: 32-72 years) years compared to 54.6 ± 15.5 years (range: 20-81 years) for C (P = 0.27). Sixty nine percent of patients in MD and 65% in C group were males (P = 0.67). When evaluating colonoscopy reports for bowel visualization, MD patients had significantly greater percentage of solid stool (i.e., poor visualization) compared to C (40.3% vs 6.9%, P < 0.001). Poor bowel preparation (35.8% vs 9.7%, P < 0.001) and need for repeat colonoscopy (32.8% vs 12.5%, P = 0.004) were significantly higher in MD group compared to C, respectively. Under univariate analysis, factors significantly associated with MD group were presence of fecal particulate [odds ratio (OR), 3.89, 95% CI: 1.33-11.36, P = 0.01] and solid stool (OR, 13.5, 95% CI: 4.21-43.31, P < 0.001). Fair (OR, 3.82, 95% CI: 1.63-8.96, P = 0.002) and poor (OR, 8.10, 95% CI: 3.05-21.56, P < 0.001) assessment of bowel preparation were more likely to be associated with MD patients. Requirement for repeat colonoscopy was also significant higher in MD group (OR, 3.42, 95% CI: 1.44-8.13, P = 0.01). In the multivariate analyses, the only variable independently associated with MD group was presence of solid stool (OR, 7.77, 95% CI: 1.66-36.47, P = 0.01). Subgroup analysis demonstrated a general trend towards poorer bowel visualization with higher methadone dosage. ANOVA analysis demonstrated that mean methadone dose associated with presence of solid stool (poor visualization) was significantly higher compared to mean dosage for clean colon (excellent visualization, P = 0.02) or for those with liquid stool only (good visualization, P = 0.01).
CONCLUSION: Methadone dependence is a risk factor for poor bowel visualization and leads to more repeat colonoscopies. More aggressive bowel preparation may be needed in MD patients.
Keywords: Colonoscopy, Methadone, Opioid, Inadequate bowel preparation, Colonoscopy preparation, Methadone dose
INTRODUCTION
Approximately 7 million colonoscopies are performed annually in the United States[1,2]. It is the preferred modality for colorectal cancer screening[3,4]. A successful colonoscopy requires adequate pre-colonoscopy bowel preparation[5]. The adequacy of the preparation depends on its ability to reliably empty the colon of fecal material without altering the colonic mucosa[2,6]. Inability to properly visualize the lumen due to retained fecal material may result in missed pathologies, diagnostic delays and repeat procedures, with potentially adverse medical, legal, and economic implications. It has been reported that over 20% of all colonoscopies performed in the United States fail due to suboptimal bowel preparation or poor colonic visualization[7,8], leading to an estimated 12%-22% increase in long term costs related to the procedure[9].
Concurrently, opioid use in the United States has increased sharply over the last decade[10-13]. An estimated 3% of all adult population in the United States is on long term opioid treatment for chronic pain management[10,14]. Methadone, a synthetic opioid, is commonly used for analgesia in patients with malignancy[15], and intractable neuropathy[16]. It is also standard therapy for the treatment of opioid addiction[17]. However, methadone can cause constipation due to its anti-peristaltic effects on the entire gastrointestinal (GI) tract[18]. Therefore, it is reasonable to postulate that adequate colonic visualization may be difficult to accomplish in patients on chronic methadone therapy due to excessive fecal retention.
Although studies have demonstrated a variety of risk factors for suboptimal preparation and poor bowel visualization[19,20], the effects of opioids on the quality of bowel visualization has not been evaluated. Our aim was to examine the quality of colonoscopic bowel visualization in methadone dependent patients.
MATERIALS AND METHODS
Patient selection
This was a retrospective cohort study of patients selected from Beth Israel Medical Center (BIMC) at Albert Einstein College of Medicine endoscopy database who had an in-patient colonoscopy performed between October, 2004 and May, 2009. The study was approved by BIMC Institutional Review Board. Patients were cross referenced using BIMC’s computerized medical records, out-patient records, and discharge summaries to obtain demographic, clinical, medications and laboratory data. Search filters for “methadone” were applied to select for patients. Patients with risk factors known to cause constipation or gastric dysmotility were excluded (Table 1). Eligible patients were divided into two groups: (1) methadone dependent (MD); and (2) those without any history of opioid usage (C). MD patients and their dosages were verified from their respective institutional methadone maintenance treatment program. Patients whose methadone dose was not verified or those who received methadone on an as needed basis were also excluded. The colonoscopy reports of all eligible patients were analyzed for degree of bowel visualization, assessment of the bowel preparation (good, fair, or poor), and whether a repeat colonoscopy was required. Age and gender matched controls who were free of any opioid exposure for at least 4 wk before colonoscopy were selected from our database as controls.
Table 1.
Exclusion criteria for patient enrollment
| Patient with history of |
| Diabetes, neurological or any thyroid dysfunction |
| Active inflammatory bowel disease |
| Scleroderma |
| Chronic constipation |
| Chronic diarrhea |
| Any acute or chronic renal insufficient or on hemodialysis |
| Radiation to colon |
| Colectomy, or hemi-colectomy |
| Positive stool studies for any pathogen |
| Iron replacement therapy |
| Fentanyl, dilaudid, or any other opioids |
| Pancreatic insufficiency or pancreatic enzyme replacement therapy |
| Tricyclic antidepressants |
| Colonoscopy performed on emergent basis or on unstable patients |
| Colonoscopy report without comment on adequacy of bowel preparation or bowel images |
Evaluation of bowel visualization
Bowel visualization was scored on a 4 point scale utilized in multiple prior studies examining quality of bowel visualization[20-22]. Points were assigned as following: excellent = 1, good = 2, fair = 3, or poor = 4[20-22]. Excellent visualization was defined as a clean bowel without presence of any stool and small to moderate amount of clear liquid. Good was defined as presence of small amount of liquid fecal material able to be easily suctioned. Fair was defined as semisolid stool (fecal particulate), able to be washed out with more than 90% of the colon visualized. Poor was defined as presence of large amounts of solid stool obscuring more than 90% of the colonic mucosa.
Statistical analysis
Descriptive statistics of mean and standard deviation were used to describe the continuous variables. Percentage and frequency were used to describe the categorical variables. Analysis of variance (ANOVA) compared for differences in the continuous variables. Pearson χ2 test compared for differences in the categorical variables. P values were two sided, and < 0.05 were considered statistically significant. All variables significantly differing between MD and C groups were included in both univariate and multivariate logistic regression analyses. ANOVA was used to compare methadone dose to bowel visualization level. Least significant difference post-hoc comparisons were performed. All analysis were performed with IBM SPSS Statistics Version 19.
RESULTS
Demographics and clinical characteristics
A total of 178 MD patients underwent colonoscopy during the designated study period, out of which 57 were excluded for concomitant use of other opioids or prescribed methadone on an as needed basis, 48 were excluded for unverifiable methadone dosage and 17 for either incomplete colonoscopy reports, colonoscopies performed urgently, or for prematurely terminated examinations for unknown reasons. A total of 115 colonoscopies were performed during the same study period on opioid naïve patients who passed the exclusion criteria for the study. Age and gender matched patients were randomly selected from this population to serve as controls in a numerically comparable group. In the final analysis, 56 patients were included in the MD group and 68 in control group. This yielded a total of 67 colonoscopy reports for MD patients and 72 for C, as some patients underwent repeat examinations. Baseline characteristics of all patients are listed in Table 2. The average age for MD patients was 52.2 ± 9.2 years (range: 32-72 years) years compared to 54.6 ± 15.5 years (range: 20-81 years) for C (P = 0.27). Sixty nine percent of patients in MD and 65% in C group were males (P = 0.67).
Table 2.
Clinical characteristics of the enrolled patients n (%)
| Methadone patients | Controls | P value | |
| Number of patients | 56 | 68 | |
| Number of colonoscopies | 67 | 72 | |
| Average age (yr) | 52.2 ± 9.2 | 54.6 ± 15.5 | 0.27 |
| Gender | 0.67 | ||
| Male | 46 (68.7) | 47 (65.3) | |
| Female | 21 (31.3) | 25 (34.7) | |
| Indication | |||
| Bleeding | 22 (32.4) | 47 (40.9) | |
| Anemia | 3 (4.4) | 25 (21.7) | |
| Screening | 31 (45.6) | 11 (9.6) | |
| Abdominal pain | 5 (7.4) | 8 (7) | |
| Other | 7 (10.3) | 24 (20.9) |
Colonoscopy evaluation
Table 3 compares bowel visualization, overall assessment of bowel preparation, and need for repeat colonoscopies between MD and C groups. Bowel visualization quality significantly differed between the two groups, with data suggesting greater percentages of solid stool (poor visualization) for MD compared to C (40.3% vs 6.9%, P < 0.001). Poor bowel preparation (35.8% vs 9.7%, P < 0.001) and need for repeat colonoscopy (32.8% vs 12.5%, P = 0.004) were significantly higher in MD group compared to C.
Table 3.
Comparisons of bowel visualization, preparation assessment, and rate of repeat colonoscopy between methadone and control patients n (%)
| Variables | Methadone patients (n = 67) | Control (n = 72) | P value |
| Bowel visualization | < 0.001 | ||
| Excellent | 12 (17.9) | 30 (41.7) | |
| Good | 14 (20.9) | 28 (38.9) | |
| Fair | 14 (20.9) | 9 (12.5) | |
| Poor | 27 (40.3) | 5 (6.9) | |
| Bowel preparation assessment | < 0.001 | ||
| Good | 22 (32.8) | 52 (72.2) | |
| Fair | 21 (31.3) | 13 (18.1) | |
| Poor | 24 (35.8) | 7 (9.7) | |
| Repeat colonoscopy | 22 (32.8) | 9 (12.5) | 0.004 |
Results for univariate and multivariate analysis are listed in Table 4. In the univariate analysis, factors significantly associated with MD group were presence of fecal particulate [odds ratio (OR), 3.89, 95% CI: 1.33-11.36, P = 0.01] and solid stool (OR, 13.5, 95% CI: 4.21-43.31, P < 0.001). Bowel preparation assessment had significantly greater odds ratios for fair and poor status in the MD group (Table 4). Requirement for repeat colonoscopy was also significant in the MD group (OR, 3.42, 95% CI: 1.44-8.13, P = 0.01). However, in the multivariate analyses, the only significant variable independently associated with MD group was presence of solid stool (OR, 7.77, 95% CI: 1.66-36.47, P = 0.01).
Table 4.
Univariate and multivariate analysis in methadone dependent patients
| Variables | Univariate OR (95% CI) | P value | Multivariate OR (95% CI) | P value |
| Bowel visualization | ||||
| Excellent | 1.00 | 1.00 | ||
| Good | 1.25 (0.50, 3.16) | 0.64 | 1.07 (0.41, 2.80) | 0.89 |
| Fair | 3.89 (1.33, 11.36) | 0.01 | 2.51 (0.74, 8.50) | 0.14 |
| Poor | 13.50 (4.21, 43.31) | < 0.001 | 7.77 (1.66, 36.47) | 0.01 |
| Bowel preparation assessment | ||||
| Good | 1.00 | 1.00 | ||
| Fair | 3.82 (1.63, 8.96) | 0.002 | 2.29 (0.86, 6.10) | 0.10 |
| Poor | 8.10 (3.05, 21.56) | < 0.001 | 2.61 (0.56, 12.21) | 0.22 |
| Repeat colonoscopy | 3.42 (1.44, 8.13) | 0.01 | 0.73 (0.19, 2.77) | 0.64 |
OR: Odds ratio.
Methadone dosage and bowel visualization
A subgroup analysis limited to MD patients was performed to evaluate the relationship between methadone dosage and quality of bowel visualization. There was a general trend towards poorer visualization with higher dosage (Figure 1). Mean methadone doses for quality of bowel visualization levels were: 76.7 ± 54.3 mg for clean bowel, 75.4 ± 45.8 mg for liquid stool, 108.6 ± 46.1 mg for fecal particulate, and 116.5 ± 42.3 mg for presence of solid stool (Figure 2). ANOVA analysis demonstrated statistically significant differences in mean methadone dosage between the quality of bowel visualization (P = 0.02). Least significant difference post-hoc comparisons showed that mean methadone dose associated with the presence of solid stool (poor visualization) was significantly higher compared to mean dosage for clean colon (excellent visualization, P = 0.02) or for those with liquid stool only (good visualization, P = 0.01).
Figure 1.
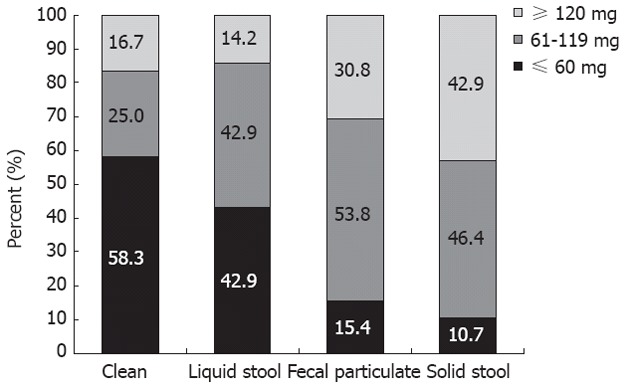
Bowel visualization according to methadone dose.
Figure 2.
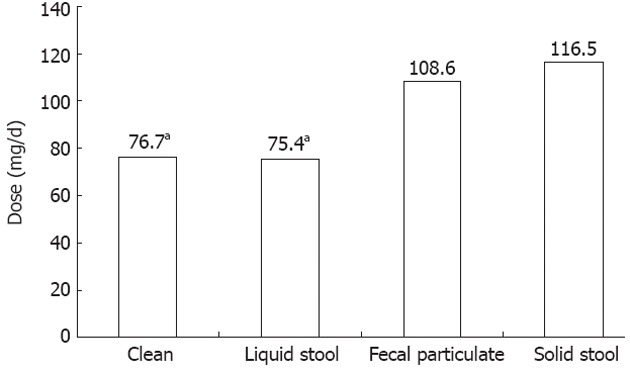
Mean methadone dose for quality of bowel visualization. aP < 0.05 vs solid stool.
DISCUSSION
To our knowledge, this is the first study to examine the effects of methadone on bowel visualization. After applying strict exclusionary criteria eliminating many known causes of GI dysmotility, our results show that chronic methadone use is an independent risk factor for poor bowel visualization due to higher incidence of retained solid stool. Bowel visualization may also be dose dependent since mean methadone dose for colonoscopies containing solid stool was significantly higher compared to the clean colonoscopies or those with only liquid stool.
Prior studies on bowel visualization have used a scoring system to evaluate for the degree of colonic stool retention[20-22]. We used the identical scoring system to examine bowel visualization in methadone dependent patients. Compared to opioid naïve patients, methadone dependent patients had a significantly higher rate of retained solid stool (poor visualization) and fecal particulate (fair visualization). Even under multivariate analysis, methadone patients had approximately an 8-fold higher occurrence for presence of solid stool compared to controls. Methadone, as with other opioids, binds to μ receptors in the central nervous system[23], and peripheral sensory nerve fibers terminals[24], including enteric neurons[10,25]. Opioids inhibit both gastric emptying and intestinal propulsive motor activity[17] throughout the entire GI tract while stimulating contraction of the pyloric and ileo-cecal sphincters[10,26]. The densest concentrations of mu receptors are found in the stomach and colon[27], therefore decreased colonic propulsion is also likely. Furthermore, it is well known that one of the major functions of a normal colon includes water absorption[28]. Opioids stimulate fluid and water absorption mainly due to delayed transit time and increased luminal contact time[10]. The combination of multiple effects of methadone throughout the GI tract in conjunction with increased colonic fluid resorption from decreased propulsion most likely explains the significantly higher presence of solid stool in methadone dependent patients.
We further examined whether there was a relationship between methadone dosage and bowel visualization. Our results show that methadone patients whose colonoscopies contained solid stool were consuming a significantly higher dose compared to methadone patients with clean bowel or liquid stool. Methadone pharmacokinetics has demonstrated a strong linear relationship between plasma concentration and methadone dose[29,30]. Therefore, it might be possible that consuming higher methadone dose led to higher plasma concentration and subsequently more side effects, including decreased transit time and increased water absorption, leading to higher presence of solid stool. Unfortunately, studies on the relationship between opioid dosage and GI motor function are limited. A study by Delgado-Aros et al[31] examined the effects of asimadoline, a κ receptor opioid agonist, in patients with irritable bowel syndrome and found no difference in GI motor function with increasing dosage. However, κ receptors are involved in visceral pain perception [32,33] and do not affect GI motility. A study by Ness et al[19] on factors affecting colonic preparation demonstrated no association between narcotics use and inadequate bowel preparation. However, it is unclear whether their study population had other comorbidities which may have confounded the results[19]. Furthermore, information on the type of narcotics (opioid vs non-opioid), dosage, and duration of usage were lacking, and the number of patients on narcotics were small (n = 14)[19].
The presence of solid stool may also have factored into the requirement for repeat colonoscopies. Methadone dependent patients were significantly more likely to require repeat colonoscopies compared to non-opioid patients under univariate analysis. However, results were non-significant when controlling for other variables under multivariate analysis. Therefore, the requirement for a repeat colonoscopy in methadone patients is dependent on other factors, most likely the presence of solid stool obscuring proper luminal visualization. Another explanation for the lack of significant results under multivariate analysis may be due to the relatively small sample size, since the range of the odds ratio was large under univariate analysis. Further studies using a larger sample size are needed to validate these results. Nonetheless, our study underscores the importance of proper bowel visualization and the substantial costs associated with repeat colonoscopies. According to a cost analysis study by Rex et al[9], the economic impact of repeat colonoscopies resulted in a 12%-22% increase in potential long term costs due to decreased time interval for subsequent colonoscopies and associated charges for polypectomy and histological examinations.
Lastly, we evaluated the overall assessment of the adequacy of bowel preparation. The American Society of Gastrointestinal Endoscopy and the American College of Gastroenterology Taskforce on Quality in Endoscopy suggest that every colonoscopy report should include an overall assessment, although acknowledge that it lacks standardized definitions due to endoscopist subjectivity[34]. Multiple prior studies have evaluated the adequacy of bowel preparation based on the endoscopist’s overall assessment of the colonoscopy[8,19,35]. Harewood et al[8] demonstrated that approximately 25% of colonoscopies examining bowel preparation and detection of colonic neoplasia were considered “inadequate,” defined as “fair” or “poor”. Standardized definitions for preparation quality were not provided to the endoscopists[8]. Similar criteria for bowel assessment were applied in the study by Kazarian et al[35] examining adequacy of bowel preparation in urban populations. In our study, a significantly higher percentage of colonoscopies performed on methadone patients were assessed as “poor” compared to opioid naive patients. While the assessment of poor preparation was not independently associated with methadone patients, the results were significant under univariate analysis. This suggests that the significantly higher presence of solid stool in the methadone patients most likely contributed to the overall poor bowel assessment. Inter-observer variability in the assessment of the bowel preparation quality between endoscopists may also have contributed to the lack of significant results under multivariate analysis. Colonoscopy reports from multiple endoscopists were evaluated, which represents a limiting factor in our study. However, other studies on the quality of bowel preparations have also been limited by similar inter-observer bias due to multiple endoscopists[7,8]. Further studies on methadone’s effect on quality of bowel assessment based on a single endoscopist are needed to validate our results.
Due to the retrospective nature of our study, there were several other limitations. Information on the time elapsed between bowel preparation to procedure, and type of purgative ingested were not available. Therefore, bowel preparation may not have been standardized. Our study was also limited due to potential inter-observer bias since there were multiple endoscopists whose colonoscopy evaluations were utilized. This raises the concern that one endoscopist may have a more stringent assessment of the quality of bowel preparation than others. However, these limitations applied to both methadone and control groups equally, thereby reducing their confounding effects. Data on colonoscopic findings and need for therapeutic interventions were not available since it was outside the scope of our investigation into bowel visualization and preparation. We were also unable to apply the Ottowa[36] or Boston Bowel Preparation[37] Scales. These scales utilize a cumulative scoring system based on a combination of fluid quality and bowel location to determinate the quality of bowel preparation. While it is a validated system, the aims of our study were to examine the need for repeat colonoscopies and the presence of retained fecal matter instead of its location. Another limitation is the small sample size, including the number of controls, in our study. Although this is a preliminary study, the most likely explanation for the limited number of patients, specifically the controls, is due to the extensive exclusionary criteria applied. We excluded all individuals with any pathology that may affect GI motility, including many common diseases, such as diabetes and thyroid dysfunction, surgical interventions, and medications. We also excluded all partial colonoscopy reports that did not contain information on bowel assessment or those without images. Lastly, our study was performed at a single institution, thereby limiting a broad generalization of the results. Nevertheless, these limitations underscore the importance of the effects of methadone, and other opioids, on bowel visualization. Currently, studies in this area are lacking. Further prospective studies in methadone and other opioids on colonoscopic outcomes are needed to validate the results of our preliminary study.
In conclusion, methadone dependent patients may require more repeat colonoscopies due to poor bowel visualization and retained stool. Poor bowel visualization may also be dose dependent. Methadone and other opioid use has risen drastically over the last decade in the United States, and an estimated 3% of all adults are currently on chronic opioid treatment[10,14]. Considering the high numbers of colonoscopies performed annually in the United States, it is reasonable to conclude that clinicians will encounter a significant number of patients on chronic opioid therapy awaiting a colonoscopy. In fact, the average age for methadone dependent patients in our study was in the range where a screening colonoscopy should be performed on all individuals. Our results also suggest that a more aggressive approach to bowel preparation may be needed in methadone, or other opioid dependent patients. Current bowel cleansing practices may need to be revisited in these patients to not only improve the purgative effects, but also decrease the need for repeat colonoscopies. Recent investigations on peripherally acting μ-opioid receptor antagonists, such as methylnaltrexone or alvimopan[38], have shown benefit in treating opioid induced constipation, and may aid in improved bowel preparations in these patients. The utility of these antagonists in the setting of pre-colonoscopy bowel preparation for opioid dependent patients are lacking and is an area of potential future research.
ACKNOWLEDGMENTS
Dr. Verma would like to thank Rita Prasad Verma, MD for her tireless guidance into the preparation and development of this manuscript.
COMMENTS
Background
While approximately 7 million colonoscopies are performed in the United States annually, over 20% of all colonoscopies fail due to suboptimal bowel preparation or poor colonic visualization. This has led to an estimated 12%-22% increase in long term costs associated with colonoscopy. Concurrently, opioid use has increased sharply over the last decade in the United States. It is estimated that 3% of all adults are currently opioid dependent.
Research frontiers
Opioids, such as methadone, both inhibit intestinal propulsive motor activity throughout the entire gastrointestinal (GI) tract and stimulate fluid and water absorption in the colon. These effects lead to constipation and fecal retention. Fecal retention can obscure proper bowel visualization, thus potentially resulting in missed pathologies, diagnostic delays and repeat procedures. Studies on colonoscopy outcomes and bowel visualization in methadone dependent patients are lacking. In this study, the authors suggest that chronic methadone usage is a risk factor for poor bowel visualization and preparation, thereby leading to more repeat colonoscopies. These effects appear to be dose dependent.
Innovations and breakthroughs
Studies on the relationship between opioids and colonoscopy outcomes are limited. A prior study demonstrated no association between narcotics use and inadequate bowel preparation. However, there were several limitations, including small number of patients on unspecified narcotics without information on dosage and duration of narcotic use. This is the first and largest study to examine the effects of methadone dependence on bowel visualization and need for repeat colonoscopies. Furthermore, they examined whether there was a relationship between methadone dosage and colonoscopy outcomes.
Applications
By demonstrating that methadone is another risk factor for failure of colonoscopies, pre-colonoscopy bowel preparation methods may need to be re-evaluated in methadone dependent patients to improve bowel visualization and reduce the need for repeat colonoscopies. Mu-receptor antagonists used to treat opioid induced constipation may serve a role in pre-colonoscopy bowel preparation to improve visualization and reduced rates of repeat colonoscopies in methadone dependent patients.
Terminology
Methadone, a synthetic mu receptor agonist, is a commonly used opioid for analgesia in patients with malignancy, and intractable neuropathy. It is also standard therapy for the treatment of opioid addiction. In addition to slowing GI motility and increased fluid resorption in the colon, methadone pharmacokinetics has demonstrated a strong linear relationship between methadone dose and plasma concentrations.
Peer review
In this novel study, the authors retrospectively examined bowel visualization and preparation assessment, and need for repeat colonoscopies in a small cohort of methadone dependent patients. The study revealed that chronic methadone usage may be an additional risk factor for failure of colonoscopy due to poorer visualization and bowel preparation, and leads to a higher need for repeat colonoscopies. This association may also be dose dependent. Larger, prospective studies are needed to affirm these findings.
Footnotes
Peer reviewer: Dr. Paulino Martínez Hernández Magro, Department of Colon and Rectal Surgery, Hospital San José de Celaya, Eje Vial Norponiente No 200-509, Colonia Villas de la Hacienda, 38010 Celaya, México
S- Editor Gou SX L- Editor A E- Editor Li JY
References
- 1.Rossi F, Sosa JA, Aslanian HR. Screening colonoscopy and fecal occult blood testing practice patterns: a population-based survey of gastroenterologists. J Clin Gastroenterol. 2008;42:1089–1094. doi: 10.1097/MCG.0b013e3181599bfc. [DOI] [PubMed] [Google Scholar]
- 2.Seeff LC, Richards TB, Shapiro JA, Nadel MR, Manninen DL, Given LS, Dong FB, Winges LD, McKenna MT. How many endoscopies are performed for colorectal cancer screening? Results from CDC’s survey of endoscopic capacity. Gastroenterology. 2004;127:1670–1677. doi: 10.1053/j.gastro.2004.09.051. [DOI] [PubMed] [Google Scholar]
- 3.Wexner SD, Beck DE, Baron TH, Fanelli RD, Hyman N, Shen B, Wasco KE. A consensus document on bowel preparation before colonoscopy: prepared by a task force from the American Society of Colon and Rectal Surgeons (ASCRS), the American Society for Gastrointestinal Endoscopy (ASGE), and the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) Dis Colon Rectum. 2006;49:792–809. doi: 10.1007/s10350-006-0536-z. [DOI] [PubMed] [Google Scholar]
- 4.Davila RE, Rajan E, Baron TH, Adler DG, Egan JV, Faigel DO, Gan SI, Hirota WK, Leighton JA, Lichtenstein D, et al. ASGE guideline: colorectal cancer screening and surveillance. Gastrointest Endosc. 2006;63:546–557. doi: 10.1016/j.gie.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 5.Froehlich F, Wietlisbach V, Gonvers JJ, Burnand B, Vader JP. Impact of colonic cleansing on quality and diagnostic yield of colonoscopy: the European Panel of Appropriateness of Gastrointestinal Endoscopy European multicenter study. Gastrointest Endosc. 2005;61:378–384. doi: 10.1016/s0016-5107(04)02776-2. [DOI] [PubMed] [Google Scholar]
- 6.Wu KL, Rayner CK, Chuah SK, Chiu KW, Lu CC, Chiu YC. Impact of low-residue diet on bowel preparation for colonoscopy. Dis Colon Rectum. 2011;54:107–112. doi: 10.1007/DCR.0b013e3181fb1e52. [DOI] [PubMed] [Google Scholar]
- 7.Lebwohl B, Wang TC, Neugut AI. Socioeconomic and other predictors of colonoscopy preparation quality. Dig Dis Sci. 2010;55:2014–2020. doi: 10.1007/s10620-009-1079-7. [DOI] [PubMed] [Google Scholar]
- 8.Harewood GC, Sharma VK, de Garmo P. Impact of colonoscopy preparation quality on detection of suspected colonic neoplasia. Gastrointest Endosc. 2003;58:76–79. doi: 10.1067/mge.2003.294. [DOI] [PubMed] [Google Scholar]
- 9.Rex DK, Imperiale TF, Latinovich DR, Bratcher LL. Impact of bowel preparation on efficiency and cost of colonoscopy. Am J Gastroenterol. 2002;97:1696–1700. doi: 10.1111/j.1572-0241.2002.05827.x. [DOI] [PubMed] [Google Scholar]
- 10.Camilleri M. Opioid-induced constipation: challenges and therapeutic opportunities. Am J Gastroenterol. 2011;106:835–842; quiz 843. doi: 10.1038/ajg.2011.30. [DOI] [PubMed] [Google Scholar]
- 11.Cicero TJ, Inciardi JA, Muñoz A. Trends in abuse of Oxycontin and other opioid analgesics in the United States: 2002-2004. J Pain. 2005;6:662–672. doi: 10.1016/j.jpain.2005.05.004. [DOI] [PubMed] [Google Scholar]
- 12.Cone EJ, Fant RV, Rohay JM, Caplan YH, Ballina M, Reder RF, Spyker D, Haddox JD. Oxycodone involvement in drug abuse deaths: a DAWN-based classification scheme applied to an oxycodone postmortem database containing over 1000 cases. J Anal Toxicol. 2003;27:57–67; discussion 67. doi: 10.1093/jat/27.2.57. [DOI] [PubMed] [Google Scholar]
- 13.Manchikanti L, Singh A. Therapeutic opioids: a ten-year perspective on the complexities and complications of the escalating use, abuse, and nonmedical use of opioids. Pain Physician. 2008;11:S63–S88. [PubMed] [Google Scholar]
- 14.Dunn KM, Saunders KW, Rutter CM, Banta-Green CJ, Merrill JO, Sullivan MD, Weisner CM, Silverberg MJ, Campbell CI, Psaty BM, et al. Opioid prescriptions for chronic pain and overdose: a cohort study. Ann Intern Med. 2010;152:85–92. doi: 10.1059/0003-4819-152-2-201001190-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Trescot AM. Review of the role of opioids in cancer pain. J Natl Compr Canc Netw. 2010;8:1087–1094. doi: 10.6004/jnccn.2010.0077. [DOI] [PubMed] [Google Scholar]
- 16.Moulin DE, Palma D, Watling C, Schulz V. Methadone in the management of intractable neuropathic noncancer pain. Can J Neurol Sci. 2005;32:340–343. doi: 10.1017/s0317167100004236. [DOI] [PubMed] [Google Scholar]
- 17.Stein C. The control of pain in peripheral tissue by opioids. N Engl J Med. 1995;332:1685–1690. doi: 10.1056/NEJM199506223322506. [DOI] [PubMed] [Google Scholar]
- 18.Yuan CS, Foss JF, O’Connor M, Osinski J, Karrison T, Moss J, Roizen MF. Methylnaltrexone for reversal of constipation due to chronic methadone use: a randomized controlled trial. JAMA. 2000;283:367–372. doi: 10.1001/jama.283.3.367. [DOI] [PubMed] [Google Scholar]
- 19.Ness RM, Manam R, Hoen H, Chalasani N. Predictors of inadequate bowel preparation for colonoscopy. Am J Gastroenterol. 2001;96:1797–1802. doi: 10.1111/j.1572-0241.2001.03874.x. [DOI] [PubMed] [Google Scholar]
- 20.Chung YW, Han DS, Park KH, Kim KO, Park CH, Hahn T, Yoo KS, Park SH, Kim JH, Park CK. Patient factors predictive of inadequate bowel preparation using polyethylene glycol: a prospective study in Korea. J Clin Gastroenterol. 2009;43:448–452. doi: 10.1097/MCG.0b013e3181662442. [DOI] [PubMed] [Google Scholar]
- 21.Sharma VK, Chockalingham SK, Ugheoke EA, Kapur A, Ling PH, Vasudeva R, Howden CW. Prospective, randomized, controlled comparison of the use of polyethylene glycol electrolyte lavage solution in four-liter versus two-liter volumes and pretreatment with either magnesium citrate or bisacodyl for colonoscopy preparation. Gastrointest Endosc. 1998;47:167–171. doi: 10.1016/s0016-5107(98)70351-7. [DOI] [PubMed] [Google Scholar]
- 22.Chiu HM, Lin JT, Wang HP, Lee YC, Wu MS. The impact of colon preparation timing on colonoscopic detection of colorectal neoplasms--a prospective endoscopist-blinded randomized trial. Am J Gastroenterol. 2006;101:2719–2725. doi: 10.1111/j.1572-0241.2006.00868.x. [DOI] [PubMed] [Google Scholar]
- 23.Callahan RJ, Au JD, Paul M, Liu C, Yost CS. Functional inhibition by methadone of N-methyl-D-aspartate receptors expressed in Xenopus oocytes: stereospecific and subunit effects. Anesth Analg. 2004;98:653–659, table of contents. doi: 10.1213/01.ane.0000099723.75548.df. [DOI] [PubMed] [Google Scholar]
- 24.Viscusi ER, Gan TJ, Leslie JB, Foss JF, Talon MD, Du W, Owens G. Peripherally acting mu-opioid receptor antagonists and postoperative ileus: mechanisms of action and clinical applicability. Anesth Analg. 2009;108:1811–1822. doi: 10.1213/ane.0b013e31819e0d3a. [DOI] [PubMed] [Google Scholar]
- 25.Wood JD, Galligan JJ. Function of opioids in the enteric nervous system. Neurogastroenterol Motil. 2004;16 Suppl 2:17–28. doi: 10.1111/j.1743-3150.2004.00554.x. [DOI] [PubMed] [Google Scholar]
- 26.De Schepper HU, Cremonini F, Park MI, Camilleri M. Opioids and the gut: pharmacology and current clinical experience. Neurogastroenterol Motil. 2004;16:383–394. doi: 10.1111/j.1365-2982.2004.00513.x. [DOI] [PubMed] [Google Scholar]
- 27.Fickel J, Bagnol D, Watson SJ, Akil H. Opioid receptor expression in the rat gastrointestinal tract: a quantitative study with comparison to the brain. Brain Res Mol Brain Res. 1997;46:1–8. doi: 10.1016/s0169-328x(96)00266-5. [DOI] [PubMed] [Google Scholar]
- 28.Irving MH, Catchpole B. ABC of colorectal diseases. Anatomy and physiology of the colon, rectum, and anus. BMJ. 1992;304:1106–1108. doi: 10.1136/bmj.304.6834.1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fonseca F, de la Torre R, Díaz L, Pastor A, Cuyàs E, Pizarro N, Khymenets O, Farré M, Torrens M. Contribution of cytochrome P450 and ABCB1 genetic variability on methadone pharmacokinetics, dose requirements, and response. PLoS One. 2011;6:e19527. doi: 10.1371/journal.pone.0019527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wolff K, Hay A. Methadone concentrations in plasma and their relationship to drug dosage. Clin Chem. 1992;38:438–439. [PubMed] [Google Scholar]
- 31.Delgado-Aros S, Chial HJ, Cremonini F, Ferber I, McKinzie S, Burton DD, Camilleri M. Effects of asimadoline, a kappa-opioid agonist, on satiation and postprandial symptoms in health. Aliment Pharmacol Ther. 2003;18:507–514. doi: 10.1046/j.1365-2036.2003.01670.x. [DOI] [PubMed] [Google Scholar]
- 32.Camilleri M, Andresen V. Current and novel therapeutic options for irritable bowel syndrome management. Dig Liver Dis. 2009;41:854–862. doi: 10.1016/j.dld.2009.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Delvaux M, Louvel D, Lagier E, Scherrer B, Abitbol JL, Frexinos J. The kappa agonist fedotozine relieves hypersensitivity to colonic distention in patients with irritable bowel syndrome. Gastroenterology. 1999;116:38–45. doi: 10.1016/s0016-5085(99)70226-x. [DOI] [PubMed] [Google Scholar]
- 34.Rex DK, Petrini JL, Baron TH, Chak A, Cohen J, Deal SE, Hoffman B, Jacobson BC, Mergener K, Petersen BT, et al. Quality indicators for colonoscopy. Am J Gastroenterol. 2006;101:873–885. doi: 10.1111/j.1572-0241.2006.00673.x. [DOI] [PubMed] [Google Scholar]
- 35.Kazarian ES, Carreira FS, Toribara NW, Denberg TD. Colonoscopy completion in a large safety net health care system. Clin Gastroenterol Hepatol. 2008;6:438–442. doi: 10.1016/j.cgh.2007.12.003. [DOI] [PubMed] [Google Scholar]
- 36.Rostom A, Jolicoeur E. Validation of a new scale for the assessment of bowel preparation quality. Gastrointest Endosc. 2004;59:482–486. doi: 10.1016/s0016-5107(03)02875-x. [DOI] [PubMed] [Google Scholar]
- 37.Lai EJ, Calderwood AH, Doros G, Fix OK, Jacobson BC. The Boston bowel preparation scale: a valid and reliable instrument for colonoscopy-oriented research. Gastrointest Endosc. 2009;69:620–625. doi: 10.1016/j.gie.2008.05.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tack J. Current and future therapies for chronic constipation. Best Pract Res Clin Gastroenterol. 2011;25:151–158. doi: 10.1016/j.bpg.2011.01.005. [DOI] [PubMed] [Google Scholar]


