Abstract
Waldenström’s macroglobulinemia is an indolent, lymphoproliferative disease, characterized by a heterogeneous lymphoplasmacytic bone marrow infiltrate and high immunoglobulin M production. While technological advances over the past several decades have dramatically improved the possibilities of studying the molecular basis of Waldenström’s macroglobulinemia, the pathogenesis of the disease remains fragmented. Undoubtedly, research has been successful in uncovering underlying aberrations and deregulated mechanisms in this disease, providing useful information for identifying biomarkers for disease diagnosis, risk stratification and therapeutic intervention, but there is still a long way to go before the pathogenesis of Waldenström’s macroglobulinemia is fully revealed. In addition, the low number of in vitro or in vivo models significantly challenges extensive analysis. In this manuscript, we review the molecular basis of this disease.
Keywords: WM, NF-kB signaling pathway, Jak/STAT, PI3K/Akt, aCGH, IL-6
Introduction
Waldenström’s macroglobulinemia (WM) is an indolent and incurable B-cell neoplasm defined by the accumulation of lymphoplasmacytic cells in the bone marrow and characterized by the hypersecretion of monoclonal immunoglobulin M (IgM) protein.1 Representing 1–2% of hematologic cancers, WM is seen more frequently in white males, with a median age at diagnosis of 63 years.2,3
The bone marrow infiltrate consists of a heterogeneous population of post-germinal center (hypermutated), mature B cells, ranging from small B lymphocytes (CD19+, CD20+) to completely differentiated plasma cells (CD138+), half of which may have visible cytoplasmic inclusions, known as Dutcher’s Bodies.2,3
The clinical presentation of WM patients varies. Approximately 30% of patients with WM demonstrate indolent disease (smoldering WM). In the remaining WM patients, the clinical features can include hyperviscosity (due to hypersecretion of IgM approximately 100-fold higher than normal range values), anemia, organomegaly (specifically hepatosplenomegaly), neuropathy, parasthesia, mucosal bleeding, and retinopathies with visual disturbances.2
Accurate and definitive diagnostic classification has been hampered by a lack of a definitive criterion for this lymphoplasmacytic lymphoma. The World Health Organization classification categorizes WM alongside other non-lymphoplasmacytic lymphomas and classifies the monoclonal protein secretions of WM to consist of excess IgM, IgG, or IgA, as opposed to IgM alone.3 The Second International Workshop on Waldenström’s Macroglobulinemia came to the conclusion that WM is limited to lymphoplasmacytic lymphomas with a detectable amount of monoclonal IgM secretion.3
While multiple prognostic models have been developed, the indolent nature of the disease, median patient age, and associated confounding factors make survival analysis difficult. Across multiple analytical models, age (>65 years), increased β2 microglobulin, and organomegaly (hepatomegaly or splenomegaly) have been identified as poor prognostic factors.4 Additional adverse factors include anemia, low albumin levels, hyperviscosity and presence of constitutional symptoms such as fever, night sweats, fatigue, and weight loss.5 The free light chain (FLC) concentration shows significantly higher values in symptomatic WM patients. In addition, the FLC concentration was higher in patients with increased β2 microglobulin, hypoalbulinemia and anemia.6,7 Small patient series showed a correlation between FLC, time to treatment and overall survival,6,8 but larger studies are needed to confirm these findings.
Another biological factor found to be associated with prognosis was the von Willebrand factor which showed increased levels in cases of poor prognosis. In addition, von Willebrand factor concentration was associated with the existence of a microenvironment that stimulates the growth and survival of tumor cells (i.e. increased BM microvessel density and VEGF).9
Because of the heterogeneity of WM, the treatment approach must consider several different clinical profiles. As currently defined, WM is heterogeneous and may include more than one type of B-cell lymphoma. A consensus panel agreed that initiation of therapy was appropriate for patients with constitutional symptoms, such as fever, night sweats or weight loss. The median survival ranges from five to more than ten years, depending on the series analyzed.10–13
The existence of somatic hypermutation in WM indicates a role for antigenic stimulation in the development of WM.14–16 The IgH variable region genes are commonly mutated and the VH3 gene is often used, suggesting antigen exposure and selection.14,17 Clonality studies have shown clonal IgH and IgL gene rearrangements. The IgH variable region is commonly mutated in WM, but intra-clonal variation is usually absent and IgH switching usually does not occur.18,19
Environmental and inherited factors may contribute to familial IgM-MGUS/WM clusters. Patients with monoclonal gammopathy of undetermined significance (MGUS) are at increased risk for progression to WM.20 In a population-based study of 1,384 individuals with MGUS, researchers showed an increased risk factor of 46 for developing WM.20 The rate of progression from IgM-MGUS to WM was further noted to be 1.5–2% a year.21–23 While the development of WM is thought to be sporadic, there are a few studies demonstrating familial linkage and predisposition to the disease.2,24–26 Both familial clustering of WM and a notable increase (~10 fold) of IgM-MGUS frequency in first-degree relatives of WM patients is suggestive of familial risk.27 Using the assumption that WM and IgM-MGUS share common susceptibility genes, strong linkages involving chromosomes 1q, 3q, and 4q have been identified.21 A causal relationship between MGUS/WM and chronic antigenic stimulation has been suggested by the results of several studies.14–16,28,29 Recently, it was shown that 11% of WM/IgM-MGUS reacted with paratarg-7 (P-7), a protein of unknown function.30 Family analyses of relatives of patients with IgM MGUS/WM with an anti-P-7- paraprotein showed that the hyperphosphorylated state of this protein (pP7) is inherited as a dominant trait.30 Because only 2% of healthy controls reacted against P-7, carriers of pP7 are associated with a more than 6 times increased risk of developing MGUS/WM (P=0.001). Thus, pP-7 is the first molecularly characterized structure that provides a plausible explanation for the familial clustering of cases of IgM MGUS/WM.
While genetic analysis has helped distinguish entities across B-cell neoplasms, there are nevertheless similarities in presentation. Whereas IgM-MGUS shares with WM the presentation of an elevated IgM secretion, the evidence of bone marrow infiltration by lymphoplasmacytic lymphoma on a bone biopsy is characteristic of WM.31 Additional entities to consider in the differential diagnosis include marginal zone lymphoma (MZL), B-cell chronic lymphocytic leukemia (B-CLL) and multiple myeloma (MM). Flow cytometry analysis has been a useful tool in helping to distinguish WM from these diseases.3
Molecular analysis has been increasingly used to analyze the biological basis and progression of WM, yet we still do not completely understand the genetic origins and active pathways of WM. This paper aims to provide an overview of the molecular basis of this disease.
Genetic studies
The low rate of proliferation of malignant WM cells was historically a major obstacle in the study of the genetic basis of WM, and this has only been surpassed in recent years with the incorporation of techniques focused on the study of interphase nuclei. The conventional cytogenetic (CC) approach provided the first insights into whole genome analysis of WM.32,33 However, the low resolution of the technique combined with the need for tumor cell division lead to very low rates of successful analysis (<10% of cases). Further development of fluorescence in situ hybridization (FISH) overcame the absence of tumor cell division; however, it only provided targeted validation of already known abnormalities. More recent whole-genome approaches, such as array-based genomic hybridization (aCGH) and massively parallel DNA sequencing, mean that a comprehensive, high-resolution analysis can be performed without the need for tumor cell division, thus overcoming the low proliferation of the disease. While knowledge of the genetic basis of WM remains fragmented, understanding of the genetic landscape has significantly improved in recent years.
Practical aspects of genetic testing
Comprehensive, high-resolution tests are very powerful techniques in the analysis of WM genetics. They provide a complete analysis at DNA (aCGH/SNP arrays, sequencing), RNA (GEP, QPCR, sequencing) or microRNA (microRNA array) levels. Nevertheless, some points need to be considered before it can become part of the analysis. A critical point to be highlighted is that only tumor cells must be analyzed when the molecular tests are performed in WM, in order to avoid false negative results as a consequence of contamination with non-tumor cells. In order to obtain reliable data in any of these techniques, the tests must be performed in purified tumor cells by sorting or using immunomagnetic beads.
The immuno-phenotype of WM consists of expression of pan-B-cell markers (CD19, CD20, CD22), cytoplasmic immunoglobulin (cIg), FMC7, CD38, and CD79a.31,34,35 The number of plasma cells in patients with WM is generally in the normal range, but these plasma cells differ from normal and myeloma cells because although they are positive for CD38, they also commonly express CD19, CD45, and CD20, but lack CD56.34 With that in mind, a multiparametric sorting approach should be considered when a highly purified clonal population is required. The biggest limitations of this approach are the low number of cells obtained for further molecular studies (see below) and the complexity of the approach, which is not available in all laboratories. A more conservative method of collecting all tumor cells would be to perform serial enrichment using anti-CD19 and subsequently anti-CD138 beads. Importantly, CD19 and CD138 markers are expressed in normal B-cell lymphocytes and plasma cells, respectively. Thus, by using these cell surface markers, both normal and malignant cell populations will be enriched together. Interestingly, most of the molecular techniques can deal with moderate levels of contamination with non-tumoral cells (~20–30%), but when that threshold is passed the molecular results might be compromised. In FISH analysis, tumor cells must be either sorted or identified using FISH in combination with immunoflourescence detection of cytoplasmic immunoglobulin M (e.g. cIgM-FISH).36
Another limiting factor for high-throughput approaches is the need for a minimum number of tumor cells for DNA or RNA extraction. Even though the amount of material needed for molecular tests varies according to the techniques used, a minimum of 1–3 μg of DNA/RNA from tumor-enriched cells is desirable for most methods. Unfortunately, a significant number of patients are excluded at this point because of the low amount of DNA/RNA recovered. Finally, the recovered molecules must meet minimum quality requirements, especially when RNA studies are planned. Thus, RNA integrity number higher than 7.5 is required for array-based and next-generation sequencing techniques. Regarding DNA, a 260/280 ratio of 1.75–1.85 and 260/230 ratio higher than 1.9 is desirable for molecular tests.
Cytogenetics and FISH
Initial cytogenetic studies reported chromosomal abnormalities in around 10–20% of patients with WM, spanning observations from t(14;18) to trisomy 12 to deletion of 6q.32,37 Earlier CC analyses determined the deletion of 6q to be the most common recurrent chromosomal abnormality identified in approximately 50% of patients.37 Schop et al. observed 23% of patients with an abnormal karyotype to have a 6q deletion, while FISH analysis confirmed deletion of 6q in 42% of patients.32 Further studies used FISH analysis to assess minimal areas of deletion using multiple probes on the 6q arm and a minimal deleted region at 6q23–24.3 was suggested.38
In the most comprehensive cytogenetic study to date, Nguyen-Khan et al. analyzed a cohort of 171 untreated WM patients enrolled in a prospective randomized trial from the French Cooperative Group on Chronic Lymphocytic Leukemia and Waldenström’s Macroglobulinemia by CC and FISH.39 Overall, 46% of cases showed abnormalities by CC. Interestingly, 33.8% of WM cases with abnormal karyotype showed chromosomal translocations. By combining CC and FISH, deletion of 6q was found in 22% of cases, deletion 13q14 in 13%, trisomy 18 in 11%, trisomy 4, deletion of TP53 and deletion of ATM in 8% each.39 IgH rearrangements were only found in 3% of cases.
Although the deletion of 6q is present in around 50% of WM patients, its presence cannot be used for diagnosis as it is widely observed in several B-cell malignancies, such as marginal zone lymphomas, multiple myeloma and chronic lymphocytic leukemias.40–43 On the other hand, data from CC and FISH analyses has consistently indicated that translocations of immunoglobulin heavy chain (IgH) are infrequently detected in WM. While prevalent in many other B-cell neoplasms, such as MM, translocations involving IgH are very rare in WM (<3%).38,39,44,45
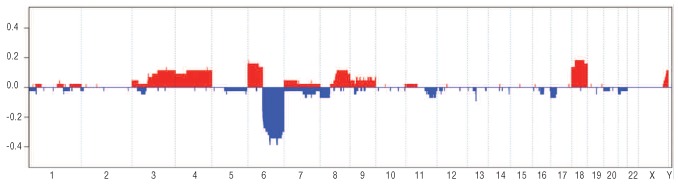
Most of the other recurrent chromosomal abnormalities observed in WM are shared with other low-grade B-cell neoplasms (trisomies 3 and 18 and deletions of 7q and 11q), such as MZL and CLL40,46 (Figure 1). Conversely, trisomy 4, which is identified in 10–20% of WM, seems to be unique in WM across low-grade B-cell neoplasias.40,47 Furthermore, trisomy 4 has occasionally been found to be the sole genetic abnormality in one patient.45,46 Further analyses have not been able to identify a minimal gained region of chromosome 4, even using high-resolution techniques.
Figure 1.
Overview of the copy-number abnormalities identified in WM by aCGH. Chromosomes 1 to Y are represented from left to right. Red blocks represent chromosome gains, whereas blue blocks represent chromosome losses. The amplitude in each abnormal region represents the frequency of each copy-number abnormality. Adapted from Braggio et al.46
The presence of 6q deletion may have prognostic significance, although some controversy remains. Nguyen-Khan et al. found that patients with 6q deletion more frequently had albumin less than 3.5 g/dL (P=0.005), B2m more than 3 mg/L (P=0.04) and an associated longer time to progression (P=0.04) in responding patients. Trisomy 4 had significantly more frequently B2M levels over 3 mg/L (P=0.02). Finally, TP53 deletion was associated with a shorter time to progression (P=0.0007) in a multivariate analysis.39 Ocio et al. analyzed the status of 6q by FISH on 98 WM patients.48 They found that patients with deletion 6q had higher B2M levels than patients without deletion 6q (>40 mg/L in 52% vs. 14% of cases, respectively, P=0.001), anemia (Hb <110 g/L in 66% vs. 40%, P=0.01) and lower albumin (< 40 g/L in 90% vs. 54%, P=0.001). In other studies, patients with a 6q deletion have been reported to have lower IgM production than those without, yet significant correlations between 6q deletions, disease progression, or prognosis for WM patients were not shown.49,50
While the clinical implications of trisomy 4 are not well understood, it has been suggested that 4q may play a role in increased susceptibility to WM.21 In a genome-wide linkage analysis consisting of 122 individuals from 11 families identified as high-risk for WM, high non-parametric linkage was found on cytoband 4q33-q34, suggesting both linkage and common susceptibility between IgM-MGUS and WM patients.21 The results of this single study lay the groundwork for further study of chromosome 4 and the potential role it plays in the etiology of WM.
Array-based comparative genomic hybridization (aCGH)
The use of aCGH has increased our knowledge of chromosomal abnormalities in WM. A study including 42 patients found that 83% of cases had chromosomal abnormalities, with a median of 3 abnormalities per case.46 Overall, 16 copy-number changes were found in over 5% of patients (Figure 1). As previously noted, partial or whole deletion of 6q proves to be the most commonly observed chromosomal abnormality. The loss of four separate minimal deleted regions (MDR) in 6q has been seen across several B-cell neoplasms. Interestingly, two of these MDR in WM include PRDM1 (BLIMP) and TNFAIP3, respectively.46 PRDM1 is well known for its repression of cell proliferation genes and also down-regulates PAX5 that in turn suppresses XBP1. While PAX5 has not been found to be mutated or over-expressed in WM, XBP1 has been shown to be over-expressed in a subset of WM patients (61%) by quantitative RT-PCR.51 TNFAIP3 is a tumor suppressor gene and its inactivation results in the constitutive activation of the NF-κB signaling pathways (Table 1).46,52,53 Biallelic inactivation of TNFAIP3 was found in approximately 5% of WM patients. Furthermore, 38% were found to have monoallelic deletions of TNFAIP3 and, as a result, significantly lower transcript expression when compared to cases with both copies of the gene. This finding may suggest TNFAIP3 haploinsufficiency in a subset of WM patients. The second most common observation by aCGH was gain of 6p (16.6%), always occurring as a secondary event following the loss of 6q.46
Table 1.
Most significant recurrent chromosomal abnormalities and mutations observed in Waldenström’s macroglobulinemia.

Although not unique to WM, inactivating mutations of TRAF3 (located on cytoband 14q32.32) have significant implications leading to constitutive activation of NF-κB pathways and are recurrent findings in a small percentage (~5%) of WM patients.46 Increased activation in this pathway has been observed in several B-cell tumors, such as DLBCL, MM and others.52,54,55
Recurrent deletions on 13q14 and 17p13 have mostly been seen in more advanced stages of the disease. Loss of 13q14 is found in approximately 10% of WM patients by aCGH. The MDR is similar to the region found in CLL and splenic MZL,41,46,47,56 and is different from what is observed in MM. This region contains the microRNAs MIRN15A and MIRN16-1, both of which negatively regulate BCL2 and, therefore, function as tumor suppressors of this anti-apoptotic protein.57 Deletion of 17p13, which includes TP53, has been found in 5–10% of WM patients.39,46 If p53 does not bind to damaged DNA, its role as a gatekeeper is lost; p21 is not activated and, therefore, does not interact with cdk2 to stop the cell cycle at specific checkpoints (Table 1).58 Overall, high-throughput analyses have unfolded a plethora of information regarding chromosomal abnormalities and potentially affected pathways in WM.
Massively parallel sequencing
Preliminary data obtained from whole genome sequencing of 30 WM cases have recently been reported.59 Strikingly, MYD88 mutation leading to a leucine to proline substitution in codon 265 (L265P) were found in 90% of WM cases (46 of 51). The MYD88 mutations provide a potential biomarker for differentiating WM from other related entities such as MZL and IgM-MGUS where MYD88 L265P was detected in less than 10% of cases.60 MYD88 is an adapter protein that affects the IL-1 and Toll-like receptor pathway.61,62 This mutation provides a potential target for patient treatment. Furthermore, the low prevalence of MYD88 mutations in IgM-MGUS suggests that the abnormality is either associated with disease progression or that there is more than one type of IgM-MGUS, with only some types progressing to WM.
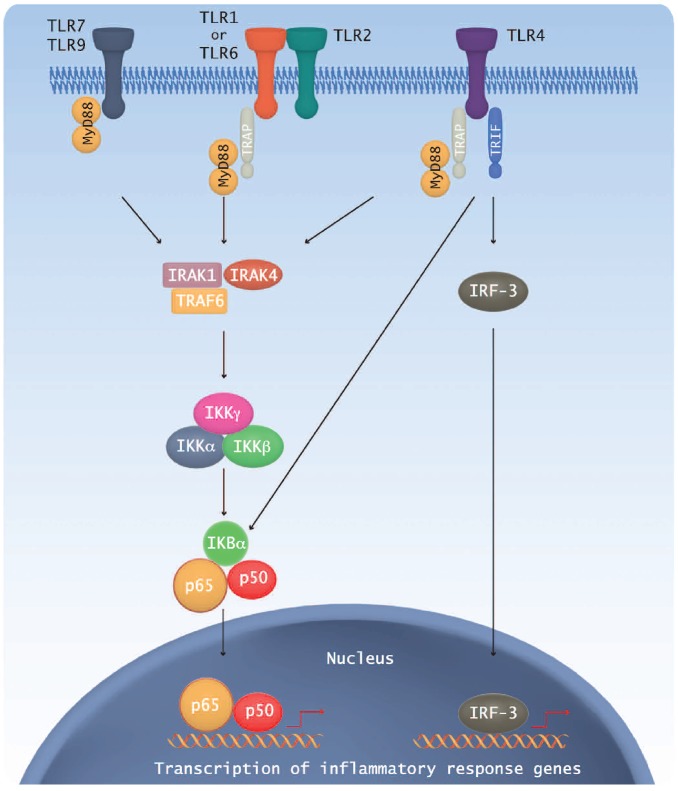
MYD88 activating mutations have been initially discovered in nodal DLBCL.63 Interestingly MYD88 mutations are found in around 40% of activated B-cell-like (ABC) subtype but are rare in the germinal center B-cell-like (GCB) subtype. This study showed that the MYD88 L265P mutant promotes cell survival by spontaneously assembling a protein complex containing IRAK1 and IRAK4, leading to IRAK4 kinase activity, IRAK1 phosphorylation, NF-κB signaling (Figure 2), JAK kinase activation of STAT3, and secretion of IL-6, IL-10 and interferon-β.63 The dysregulation of this pathway opens a novel therapeutic window, leading to the development of IRAK4 kinase inhibitors and other upstream proteins of this pathway.
Figure 2.
Schematic representation of MyD-88 dependent TLR signaling pathway.
By massively parallel sequencing, somatic mutations were described in over 10% (range 10–23%) of WM cases in an additional 7 genes (CXCR4, TAP2, LRP1B, MXLN, ARID1A, HIST1H1E and RAPGEF3) although further analysis in larger validation cohorts are needed to confirm these findings.59
MicroRNAs (miRNAs)
miRNAs regulate mRNAs via antisense mechanisms, inhibiting or modulating protein production.64,65 miRNAs play important roles in physiological processes such as cell differentiation and anti-apoptotic mechanisms.65,66 Several miRNAs (155, 206, and 9) appear to be of particular interest to WM.67 Roccaro et al. performed a comparative miRNA profile between BM CD19+ selected cells from 20 WM patients versus CD19+ cells isolated from BM and peripheral blood mononuclear cells of healthy donors.68 They identified a WM-specific microRNA signature characterized by increased expression of miRNAs-155/184/206/363/494 and 542-3p, and decreased expression of miRNA-9 (ANOVA, P<0.01). Interestingly, increased expression of miRNAs-155/184/206/363/494 and 542-3p was significantly elevated in patients with poor prognostic factors according to the International Prognostic Staging System (P<0.015).68 In addition, WM cell lines treated with antineoplastic agents rituximab, perifosine and bortezomib show a reduction in miRNAs-155/184/363/494 and 542-3p, and increased expression of miRNA-9.68 Furthermore, these expression level changes affect downstream targets of these miRNAs, such as RAS oncogene family members, transcription factors, cell-cycle and antiapoptotic regulators.68
miRNA-155 has been reported to be oncogenic in its role in various B-cell cancers.68,69 It has recently been shown that microRNA-155 regulates proliferation and growth of WM cells in vitro and in vivo, by inhibiting MAPK/ERK, PI3/AKT and NF-kB signaling pathways. Potential microRNA-155 target genes were identified using gene expression profiling and included genes involved in cell-cycle progression, adhesion and migration.68 Sacco et al. described miRNA-155 knockdown cells to have a decrease in Mdm2 resulting in an increase in p53, as well as an increase in CDK inhibitors in WM.69 Additional findings included inhibition of fibronectin adhesion and decreased response to stromal derived factor-1, suggesting miRNA-155 plays a strong role in WM cell survival.69 miRNA-206 and-9 were shown to respectively up and down-regulate histone acetylation in WM cells.68–70 It is well known that increased histone acetylation (performed by histone acetylase, HAT) increases gene transcription whereas histone deacetylase (HDAC) removes acetyl groups from chromatin, making it more condensed and reducing transcription. WM cells were found to have increased HDAC and decreased HAT expression, correlated to higher levels of miRNA-206 and lower levels of miRNA-209, respectively.69,70 It was proposed that epigenetic modification of increased deacetylase activity may impact cell cycle regulators including p21 and p53, promoting cell proliferation.69 Given the recent detailed investigation of the role miRNAs play in WM, further analysis will highlight their function in the initiation and progression of the disease.
Gene expression profiling (GEP)
GEP analysis of WM provides valuable information regarding the transcriptional signature of the disease. Data gathered from two independent studies highlight the similarities and differences in GEP across WM, CLL, MM, normal B cells, and normal plasma cells.71,72 These studies specifically highlight similarities between GEP in malignant WM cells and CLL. When analyzed in unsupervised clusters, WM clustered far more with CLL expressions than to MM.71 Both WM and CLL have strong B-cell signatures, characterized by the common marker CD20, and are defined by low proliferation rates and a lack of IgH translocations (Table 2).71 The GEP of both WM and CLL shared similar profiles, particularly with regard to cell markers and IL-10.71,72
Table 2.
Summary of the most significant findings obtained from gene expression and microRNA analyses in Waldenström’s macroglobulinemia.

One of the most significant findings in both studies was the high level of IL-6 expression in WM compared to MM, CLL, and normal B cells.71,72 IL-6 is a potent inflammatory cytokine that stimulates both local and systemic activating physiological functions in a multitude of cells.73 Locally, it acts to increase lymphocyte activity, including antibody production. Additionally, IL-6 plays a key role by activating the MAPK pathway.71 While there were no specific mutations found in MAPK, its activity was notably increased, likely correlating to the upregulation of IL-6.71 The increase of IL-6 expression in WM cells, more so than normal B cells, is suggestive of its autocrine activity.71
Interestingly, IL-6 binds to the tyrosine kinase receptor Janus kinases (JAK) 1 and 2, which act via the transcription factor Stat3 to increase IgM production.74 Activation of the JAK/STAT pathway by IL-6 increases gene transcription and IgM secretion in addition to activating other signaling pathways.74 Recently, a functional, stimulating relationship between IL-6, Rantes (CCL5), and IgM secretion was observed and appears to be mediated through the JAK/STAT and PI3K pathways.75 For the moment, the specific mechanisms of hyper Ig secretion in WM are still not known. GEP data combined with studies of the JAK/STAT pathway could be useful in future investigations into the pathogenic role of IL-6 and JAK/STAT pathways in WM.
In addition to IL-6, CD1c was up-regulated in GEP studies.71 While also expressed in normal B cells, upregulation of CD1c was significantly higher in WM than in MM or CLL and it was thought this was a potential disease marker.71 The relation between these disease expressions suggests that IGM-MGUS precedes WM.71 Through GEP, differences and similarities between WM and other B-cell neoplasias can be further extrapolated and perhaps used as disease markers.
Proteomic analysis
Proteomic analyses in WM are still limited to small studies. In an analysis performed in 10 WM cases, 6 polypeptides, all of which regulate signal transduction, were found to be dysregulated (>2-fold) in over 60% of cases.76 Interestingly, Ras family proteins, including p62Dok and Rab4 were up-regulated (Table 3). This increase correlated with the activation of MAPK pathways previously recognized in WM. Rho family protein levels were also found to be elevated, suggestive of an increase in adhesive properties of malignant WM cells.76 Using a lower cut off (>1.3-fold), a much larger number of dysregulated proteins were identified. Notably, cyclin-dependent kinases, regulators of apoptosis, HDAC, and proteins involved in the PI3 kinase pathway were all up-regulated. The cumulative effect of this protein dysregulation leads to loss of cell cycle restriction, increased cell survival, and proliferation. The upregulation of the above proteins was found in both symptomatic and asymptomatic patients, indicating that this deregulation occurs early within disease pathogenesis.76 Symptomatic disease progression was marked in this study by three proteins: p43/EMAPII, CDC25c, and HSP90. Each of these proteins furthers disease advancement, replication and migration.
Table 3.
Summary of the most significant findings obtained from protein analyses in Waldenström’s macroglobulinemia.

Dysregulated molecular pathways
PI3K/Akt/mTOR
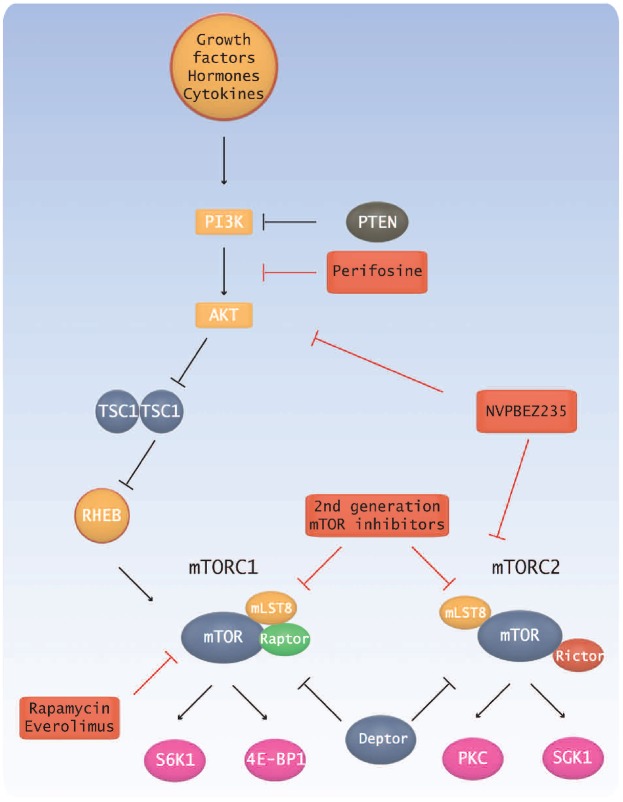
The PI3/Akt pathway regulates cell survival, increasing proliferation while inhibiting apoptosis.77 Furthermore, Akt activity increases cell migration and adhesion.78–80 Several studies have found increased Akt expression in WM.78,81 This agrees with findings of elevated expression of PI3K pathway proteins.76 While not unique to WM (Akt expression is also increased in MM), constitutive activation results in cell resistance by maintaining cell cycle pathways, inhibiting apoptosis, and supporting proliferation and cell survival.81 Both IL-6 and IGF-1 activate Akt pathways and provide strong targets for therapy.78 Recently, it was suggested that PTEN negatively regulates this pathway.81 In WM, though no mutation has been observed, PTEN gene and protein expression were found to be decreased, and it was suggested that low levels of PTEN lead to consistent activation of the PI3K/Akt pathway.81 Additionally, PTEN acts to negatively regulate mTOR which, like Akt, has increased activation due to phosphorylation in WM. Importantly, these pathways also provide targets for specific therapies (Figure 3). The first-generation mTOR inhibitors rapamycin and its analog everolimus use an allosteric mechanism to block mTORC1 output.82 A phase II trial using everolimus as a single-agent therapy on relapsed/refractory WM patients showed high activity, with an overall response rate of 70% and a moderate toxicity.83 In contrast, second mTOR inhibitors target the ATP binding site to impede kinase activity of both mTORC1 and mTORC2 (Figure 3).82
Figure 3.
Schematic representation of the PI3K/Akt/mTOR pathway. The pathway provides several targets for specific therapies. Drugs targeting the pathway are shown in red boxes.
Another promising inhibitor is NVPBEZ235 which targets both Akt and mTOR pathways, successfully reversing the activation of and dually inhibiting these overactive pathways.81 These studies provide encouraging data for the use of individualized treatment strategies to target overactive pathways in WM.
JAK/STAT
The Akt/PI3K pathway is up-regulated when STAT5 proteins are elevated.84 The JAK/Stat pathway is a critical cytokine-initiated cascade that uses several STAT proteins to achieve normal physiological and biological function, including hematopoiesis.85 Loss of these receptors and second messenger functions can be fatal and has been shown to severely disrupt erythropoiesis and lymphoid development.85 When constitutively active, cells exhibit an increase in antiapoptotic genes, cell cycle progression, and tumor evasion.74 Each of these characteristics is noted in WM. JAK1 and STAT3, both of which are activated by IL-6, show increased expression in WM, despite no known mutations in their respective genes.74 The continuous study of these dysfunctional pathways in WM will help provide targeted therapies.
Microenvironment – WM tumor cell interactions
Ngo et al. showed that WM cells express high levels of chemokine and adhesion receptors, including CXCR4 and VLA-4.86 They showed that CXCR4 was essential for the migration of WM cells. Furthermore, CXCR4 knockdown or the CXCR4 inhibitor AMD3100 showed a significant inhibition of migration. Likewise, CXCR4 or VLA-4 inhibition led to significant inhibition of adhesion to fibronectin, stromal cells, and endothelial cells.86 In addition, they showed that the CXCR4/SDF-1 axis interacts with VLA-4 in regulating migration and adhesion of WM cells in the bone marrow microenvironment.
In vitro and in vivo models
While emerging technologies have provided greater insight into genetic abnormalities observed in patients with WM, further biological studies and molecular analysis are challenged by the limited number of viable cellular models proven to be derived from WM tumors. The challenges of establishing WM cell lines that clearly demonstrate clonality to primary tumors is evident in the history of cell models for WM. Continuous establishment of authentic cell lines and further study of those that are shown to be valid will promote a greater understanding of the biological pathogenesis and treatment efficacy of WM.
To date, there are still no in vivo models of WM. Tsingotjidou et al. made an effort to establish a mouse model;87 human-derived bone particles (both disease-free and containing WM) were implanted in the back legs of mice. Approximately 50% of the mice were observed to have metastatic WM cells in the non-neoplastic bone implants as well as increased levels of serum IgM.87 Tassone et al. established an in vivo model of human WM in severe combined immuno-deficient (SCID) mice implanted with human fetal bone chips (SCID-hu mice). WM cells from patients are engrafted directly into the human bone marrow (huBM) microenvironment.88 The SCID-hu mice produce human monoclonal paraprotein IgM and/or kappa or lambda chain. Interestingly, antitumor therapies based on the use of rituximab88 or combination of Sant7 (IL6R superantagonist) and dexamethasone89 induce a reduction in serum para-protein and tumor regression. Further efforts to generate in vivo models will better illustrate their viability and validity.
Conclusions
The last decade has seen a marked evolution in our knowledge of the molecular basis of WM pathogenesis. Undoubtedly, the incorporation of high-throughput ‘omic’ analyses to the study of WM has led to the discovery of a plethora of genetic abnormalities and molecular pathways associated with the disease. These powerful tools have identified novel biomarkers that might be used in the differential diagnosis and risk stratification of the disease. Certainly, the main challenge in the post-‘omics’ era is to find the potential Achilles’ heels to be exploited for drug discovery and rapidly translate these insights into novel therapeutic strategies for WM patients. Molecular pathways such as MYD88-IRAK4, NF-κB and Akt-mTOR are promising avenues in the search for novel therapies. Multiple trials targeting these and other pathways are ongoing. Significant improvements have been seen in survival rates and in the therapeutic options available for the treatment of WM. Despite this, better in vitro and in vivo WM models and therapeutic interventions are needed to fully uncover the pathogenesis of WM and to offer better treatment options for this still incurable disease.
Footnotes
Authorship and Disclosures
The information provided by the authors about contributions from persons listed as authors and in acknowledgments is available with the full text of this paper at www.haematologica.org.
Financial and other disclosures provided by the authors using the ICMJE (www.icmje.org) Uniform Format for Disclosure of Competing Interests are also available at www.haematologica.org.
References
- 1.Gertz MA, Fonseca R, Rajkumar SV. Waldenstrom’s macroglobulinemia. Oncologist. 2000;5(1):63–7. doi: 10.1634/theoncologist.5-1-63. [DOI] [PubMed] [Google Scholar]
- 2.Ghobrial IM, Gertz MA, Fonseca R. Waldenstrom macroglobulinaemia. Lancet Oncol. 2003;4(11):679–85. doi: 10.1016/s1470-2045(03)01246-4. [DOI] [PubMed] [Google Scholar]
- 3.Ansell SM, Kyle RA, Reeder CB, Fonseca R, Mikhael JR, Morice WG, et al. Diagnosis and management of Waldenstrom macroglobulinemia: Mayo stratification of macroglobulinemia and risk-adapted therapy (mSMART) guidelines. Mayo Clin Proc. 2010;85(9):824–33. doi: 10.4065/mcp.2010.0304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ghobrial IM, Fonseca R, Gertz MA, Plevak MF, Larson DR, Therneau TM, et al. Prognostic model for disease-specific and overall mortality in newly diagnosed symptomatic patients with Waldenstrom macroglobulinaemia. Br J Haematol. 2006;133(2):158–64. doi: 10.1111/j.1365-2141.2006.06003.x. [DOI] [PubMed] [Google Scholar]
- 5.Ansell SM, Kyle RA, Reeder CB, Fonseca R, Mikhael JR, Morice WG, et al. Diagnosis and management of Waldenstrom macroglobulinemia: Mayo stratification of macroglobulinemia and risk-adapted therapy (mSMART) guidelines. Mayo Clin Proc. 2010;85(9):824–33. doi: 10.4065/mcp.2010.0304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Itzykson R, Le Garff-Tavernier M, Katsahian S, Diemert MC, Musset L, Leblond V. Serum-free light chain elevation is associated with a shorter time to treatment in Waldenstrom’s macroglobulinemia. Haematologica. 2008;93(5):793–4. doi: 10.3324/haematol.12107. [DOI] [PubMed] [Google Scholar]
- 7.Leleu X, Moreau AS, Weller E, Roccaro AM, Coiteux V, Manning R, et al. Serum immunoglobulin free light chain correlates with tumor burden markers in Waldenstrom macroglobulinemia. Leuk Lymphoma. 2008;49(6):1104–7. doi: 10.1080/10428190802074619. [DOI] [PubMed] [Google Scholar]
- 8.Leleu X, Koulieris E, Maltezas D, Itzykson R, Xie W, Manier S, et al. Novel M-component based biomarkers in Waldenstrom’s macroglobulinemia. Clin Lymphoma Myeloma Leuk. 2011;11(1):164–7. doi: 10.3816/CLML.2011.n.039. [DOI] [PubMed] [Google Scholar]
- 9.Hivert B, Caron C, Petit S, Charpy C, Fankam-Siaka C, Duhamel A, et al. Clinical and Prognostic Implications of Low or High Level of Von Willebrand Factor (VWF) In Waldenström Macroglobulinemia (WM) Patients (pts). A Clinicopathological Study on 72 Pts. Blood (ASH Annual Meeting Abstracts) 2010;116 doi: 10.1182/blood-2011-11-388256. Abstract 2438. [DOI] [PubMed] [Google Scholar]
- 10.Morel P, Monconduit M, Jacomy D, Lenain P, Grosbois B, Bateli C, et al. Prognostic factors in Waldenstrom macroglobulinemia: A report on 232 patients with the description of a new scoring system and its validation on 253 other patients. Blood. 2000;96(3):852–8. [PubMed] [Google Scholar]
- 11.Dimopoulos M, Gika D, Zervas K, Kyrtsonis M, Symeonidis A, Anagnostopoulos A, et al. The international staging system for multiple myeloma is applicable in symptomatic Waldenstrom’s macroglobulinemia. Leuk Lymphoma. 2004;45(9):1809–13. doi: 10.1080/10428190410001687512. [DOI] [PubMed] [Google Scholar]
- 12.Morel P, Duhamel A, Gobbi P, Dimopoulos MA, Dhodapkar MV, McCoy J, et al. International prognostic scoring system for Waldenstrom macroglobulinemia. Blood. 2009;113(18):4163–70. doi: 10.1182/blood-2008-08-174961. [DOI] [PubMed] [Google Scholar]
- 13.Leleu X, Soumerai J, Roccaro A, Hatjiharissi E, Hunter ZR, Manning R, et al. Increased incidence of transformation and myelodysplasia/acute leukemia in patients with Waldenstrom macroglobulinemia treated with nucleoside analogs. J Clin Oncol. 2009;27(2):250–5. doi: 10.1200/JCO.2007.15.1530. [DOI] [PubMed] [Google Scholar]
- 14.Aoki H, Takishita M, Kosaka M, Saito S. Frequent somatic mutations in D and/or JH segments of Ig gene in Waldenstrom’s macroglobulinemia and chronic lymphocytic leukemia (CLL) with Richter’s syndrome but not in common CLL. Blood. 1995;85(7):1913–9. [PubMed] [Google Scholar]
- 15.Wagner SD, Martinelli V, Luzzatto L. Similar patterns of V kappa gene usage but different degrees of somatic mutation in hairy cell leukemia, prolymphocytic leukemia, Waldenstrom’s macroglobulinemia, and myeloma. Blood. 1994;83(12):3647–53. [PubMed] [Google Scholar]
- 16.Martin-Jimenez P, Garcia-Sanz R, Balanzategui A, Alcoceba M, Ocio E, Sanchez ML, et al. Molecular characterization of heavy chain immunoglobulin gene rearrangements in Waldenstrom’s macroglobulinemia and IgM monoclonal gammopathy of undetermined significance. Haematologica. 2007;92(5):635–42. doi: 10.3324/haematol.10755. [DOI] [PubMed] [Google Scholar]
- 17.Kriangkum J, Taylor BJ, Mant MJ, Treon SP, Belch AR, Pilarski LM. The malignant clone in Waldenstrom’s macroglobulinemia. Semin Oncol. 2003;30(2):132–5. doi: 10.1053/sonc.2003.50061. [DOI] [PubMed] [Google Scholar]
- 18.Kriangkum J, Taylor BJ, Treon SP, Mant MJ, Belch AR, Pilarski LM. Clonotypic IgM V/D/J sequence analysis in Waldenstrom macroglobulinemia suggests an unusual B-cell origin and an expansion of polyclonal B cells in peripheral blood. Blood. 2004;104(7):2134–42. doi: 10.1182/blood-2003-11-4024. [DOI] [PubMed] [Google Scholar]
- 19.Sahota SS, Forconi F, Ottensmeier CH, Stevenson FK. Origins of the malignant clone in typical Waldenstrom’s macroglobulinemia. Semin Oncol. 2003;30(2):136–41. doi: 10.1053/sonc.2003.50072. [DOI] [PubMed] [Google Scholar]
- 20.Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, Plevak MF, et al. A long-term study of prognosis in monoclonal gammopathy of undetermined significance. N Engl J Med. 2002;346(8):564–9. doi: 10.1056/NEJMoa01133202. [DOI] [PubMed] [Google Scholar]
- 21.McMaster ML, Goldin LR, Bai Y, Ter-Minassian M, Boehringer S, Giambarresi TR, et al. Genomewide linkage screen for Waldenstrom macroglobulinemia susceptibility loci in high-risk families. Am J Hum Genet. 2006;79(4):695–701. doi: 10.1086/507687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kyle RA, Therneau TM, Rajkumar SV, Remstein ED, Offord JR, Larson DR, et al. Long-term follow-up of IgM monoclonal gammopathy of undetermined significance. Blood. 2003;102(10):3759–64. doi: 10.1182/blood-2003-03-0801. [DOI] [PubMed] [Google Scholar]
- 23.Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, Plevak MF, et al. Long-term follow-up of IgM monoclonal gammopathy of undetermined significance. Semin Oncol. 2003;30(2):169–71. doi: 10.1053/sonc.2003.50062. [DOI] [PubMed] [Google Scholar]
- 24.Kristinsson SY, Fears TR, Gridley G, Turesson I, Mellqvist UH, Bjorkholm M, et al. Deep vein thrombosis after monoclonal gammopathy of undetermined significance and multiple myeloma. Blood. 2008;112(9):3582–6. doi: 10.1182/blood-2008-04-151076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mulligan G, Mitsiades C, Bryant B, Zhan F, Chng WJ, Roels S, et al. Gene expression profiling and correlation with outcome in clinical trials of the proteasome inhibitor bortezomib. Blood. 2007;109(8):3177–88. doi: 10.1182/blood-2006-09-044974. [DOI] [PubMed] [Google Scholar]
- 26.Treon SP, Hunter ZR, Aggarwal A, Ewen EP, Masota S, Lee C, et al. Characterization of familial Waldenstrom’s macroglobulinemia. Ann Oncol. 2006;17(3):488–94. doi: 10.1093/annonc/mdj111. [DOI] [PubMed] [Google Scholar]
- 27.McMaster ML. Familial Waldenstrom’s macroglobulinemia. Semin Oncol. 2003;30(2):146–52. doi: 10.1053/sonc.2003.50063. [DOI] [PubMed] [Google Scholar]
- 28.Royer RH, Koshiol J, Giambarresi TR, Vasquez LG, Pfeiffer RM, McMaster ML. Differential characteristics of Waldenstrom macroglobulinemia according to patterns of familial aggregation. Blood. 2010;115(22):4464–71. doi: 10.1182/blood-2009-10-247973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Goldin LR, Landgren O. Autoimmunity and lymphomagenesis. Int J Cancer. 2009;124(7):1497–502. doi: 10.1002/ijc.24141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Grass S, Preuss KD, Wikowicz A, Terpos E, Ziepert M, Nikolaus D, et al. Hyperphosphorylated paratarg-7: a new molecularly defined risk factor for monoclonal gammopathy of undetermined significance of the IgM type and Waldenstrom macroglobulinemia. Blood. 2011;117(10):2918–23. doi: 10.1182/blood-2010-09-306076. [DOI] [PubMed] [Google Scholar]
- 31.Owen RG, Treon SP, Al-Katib A, Fonseca R, Greipp PR, McMaster ML, et al. Clinicopathological definition of Waldenstrom’s macroglobulinemia: consensus panel recommendations from the Second International Workshop on Waldenstrom’s Macroglobulinemia. Semin Oncol. 2003;30(2):110–5. doi: 10.1053/sonc.2003.50082. [DOI] [PubMed] [Google Scholar]
- 32.Schop RF, Kuehl WM, Van Wier SA, Ahmann GJ, Price-Troska T, Bailey RJ, et al. Waldenstrom macroglobulinemia neoplastic cells lack immunoglobulin heavy chain locus translocations but have frequent 6q deletions. Blood. 2002;100(8):2996–3001. doi: 10.1182/blood.V100.8.2996. [DOI] [PubMed] [Google Scholar]
- 33.Mansoor A, Medeiros LJ, Weber DM, Alexanian R, Hayes K, Jones D, et al. Cytogenetic findings in lymphoplasmacytic lymphoma/Waldenstrom macroglobulinemia. Chromosomal abnormalities are associated with the polymorphous subtype and an aggressive clinical course. Am J Clin Pathol. 2001;116(4):543–9. doi: 10.1309/6U88-357U-UKJ5-YPT3. [DOI] [PubMed] [Google Scholar]
- 34.San Miguel JF, Vidriales MB, Ocio E, Mateo G, Sanchez-Guijo F, Sanchez ML, et al. Immunophenotypic analysis of Waldenstrom’s macroglobulinemia. Semin Oncol. 2003;30(2):187–95. doi: 10.1053/sonc.2003.50074. [DOI] [PubMed] [Google Scholar]
- 35.Owen RG, Barrans SL, Richards SJ, O’Connor SJ, Child JA, Parapia LA, et al. Waldenstrom macroglobulinemia. Development of diagnostic criteria and identification of prognostic factors. Am J Clin Pathol. 2001;116(3):420–8. doi: 10.1309/4LCN-JMPG-5U71-UWQB. [DOI] [PubMed] [Google Scholar]
- 36.Ahmann GJ, Jalal SM, Juneau AL, Christensen ER, Hanson CA, Dewald GW, et al. A novel three-color, clone-specific fluorescence in situ hybridization procedure for monoclonal gammopathies. Cancer Genet Cytogenet. 1998;101(1):7–11. doi: 10.1016/s0165-4608(97)00058-7. [DOI] [PubMed] [Google Scholar]
- 37.Mansoor A, Medeiros LJ, Weber DM, Alexanian R, Hayes K, Jones D, et al. Cytogenetic findings in lymphoplasmacytic lymphoma/Waldenstrom macroglobulinemia. Chromosomal abnormalities are associated with the polymorphous subtype and an aggressive clinical course. Am J Clin Pathol. 2001;116(4):543–9. doi: 10.1309/6U88-357U-UKJ5-YPT3. [DOI] [PubMed] [Google Scholar]
- 38.Schop RF, Van Wier SA, Xu R, Ghobrial I, Ahmann GJ, Greipp PR, et al. 6q deletion discriminates Waldenstrom macroglobulinemia from IgM monoclonal gammopathy of undetermined significance. Cancer Genet Cytogenet. 2006;169(2):150–3. doi: 10.1016/j.cancergencyto.2006.04.009. [DOI] [PubMed] [Google Scholar]
- 39.Nguyen-Khac F, Lejeune J, Chapiro E, Mould S, Barin C, Daudignon A, et al. Cytogenetic abnormalities in a cohort of 171 patients with Waldenström macroglobulinemia before treatment: clinical and biological correlations. Blood (ASH Annual Meeting Abstracts) 2010;116 Abstract 801. [Google Scholar]
- 40.Braggio E, Dogan A, Keats JJ, Chng WJ, Huang G, Matthews JM, et al. Genomic analysis of marginal zone and lymphoplasmacytic lymphomas identified common and disease-specific abnormalities. Mod Pathol. 2012;25(5):651–60. doi: 10.1038/modpathol.2011.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ferreira BI, Garcia JF, Suela J, Mollejo M, Camacho FI, Carro A, et al. Comparative genome profiling across subtypes of low-grade B-cell lymphoma identifies type-specific and common aberrations that target genes with a role in B-cell neoplasia. Haematologica. 2008;93(5):670–9. doi: 10.3324/haematol.12221. [DOI] [PubMed] [Google Scholar]
- 42.Rinaldi A, Mian M, Chigrinova E, Arcaini L, Bhagat G, Novak U, et al. Genome-wide DNA profiling of marginal zone lymphomas identifies subtype-specific lesions with an impact on the clinical outcome. Blood. 2011;117(5):1595–604. doi: 10.1182/blood-2010-01-264275. [DOI] [PubMed] [Google Scholar]
- 43.Dohner H, Stilgenbauer S, Benner A, Leupolt E, Krober A, Bullinger L, et al. Genomic aberrations and survival in chronic lymphocytic leukemia. N Engl J Med. 2000;343(26):1910–6. doi: 10.1056/NEJM200012283432602. [DOI] [PubMed] [Google Scholar]
- 44.Chang H, Samiee S, Li D, Patterson B, Chen CI, Stewart AK. Analysis of IgH translocations, chromosome 13q14 and 17p13.1(p53) deletions by fluorescence in situ hybridization in Waldenstrom’s macroglobulinemia: a single center study of 22 cases. Leukemia. 2004;18(6):1160–2. doi: 10.1038/sj.leu.2403369. [DOI] [PubMed] [Google Scholar]
- 45.Terre C, Nguyen-Khac F, Barin C, Mozziconacci MJ, Eclache V, Leonard C, et al. Trisomy 4, a new chromosomal abnormality in Waldenstrom’s macroglobulinemia: a study of 39 cases. Leukemia. 2006;20(9):1634–6. doi: 10.1038/sj.leu.2404314. [DOI] [PubMed] [Google Scholar]
- 46.Braggio E, Keats JJ, Leleu X, Van Wier S, Jimenez-Zepeda VH, Valdez R, et al. Identification of copy number abnormalities and inactivating mutations in two negative regulators of nuclear factor-kappaB signaling pathways in Waldenstrom’s macroglobulinemia. Cancer Res. 2009;69(8):3579–88. doi: 10.1158/0008-5472.CAN-08-3701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Braggio E, Keats JJ, Leleu X, Wier SV, Jimenez-Zepeda VH, Schop RF, et al. High-resolution genomic analysis in Waldenstrom’s macroglobulinemia identifies disease-specific and common abnormalities with marginal zone lymphomas. Clin Lymphoma Myeloma. 2009;9(1):39–42. doi: 10.3816/CLM.2009.n.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ocio EM, Schop RF, Gonzalez B, Van Wier SA, Hernandez-Rivas JM, Gutierrez NC, et al. 6q deletion in Waldenstrom macroglobulinemia is associated with features of adverse prognosis. Br J Haematol. 2007;136(1):80–6. doi: 10.1111/j.1365-2141.2006.06389.x. [DOI] [PubMed] [Google Scholar]
- 49.Madan RA, Chang VT, Yook C, Baddoura FK, Srinivas S, Kasimis B. Waldenstrom’s macroglobulinemia evolving into acute lymphoblastic leukemia: a case report and a review of the literature. Leukemia. 2004;18(8):1433–5. doi: 10.1038/sj.leu.2403408. [DOI] [PubMed] [Google Scholar]
- 50.Chang H, Qi X, Xu W, Reader JC, Ning Y. Analysis of 6q deletion in Waldenstrom macroglobulinemia. Eur J Haematol. 2007;79(3):244–7. doi: 10.1111/j.1600-0609.2007.00903.x. [DOI] [PubMed] [Google Scholar]
- 51.Leleu X, Hunter ZR, Xu L, Roccaro AM, Moreau AS, Santos DD, et al. Expression of regulatory genes for lymphoplasmacytic cell differentiation in Waldenstrom macroglobulinemia. Br J Haematol. 2009;145(1):59–63. doi: 10.1111/j.1365-2141.2009.07592.x. [DOI] [PubMed] [Google Scholar]
- 52.Compagno M, Lim WK, Grunn A, Nandula SV, Brahmachary M, Shen Q, et al. Mutations of multiple genes cause deregulation of NF-kappaB in diffuse large B-cell lymphoma. Nature. 2009;459(7247):717–21. doi: 10.1038/nature07968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Novak U, Rinaldi A, Kwee I, Nandula SV, Rancoita PM, Compagno M, et al. The NF-{kappa}B negative regulator TNFAIP3 (A20) is inactivated by somatic mutations and genomic deletions in marginal zone lymphomas. Blood. 2009;113(20):4918–21. doi: 10.1182/blood-2008-08-174110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Keats JJ, Fonseca R, Chesi M, Schop R, Baker A, Chng WJ, et al. Promiscuous mutations activate the noncanonical NF-kappaB pathway in multiple myeloma. Cancer Cell. 2007;12(2):131–44. doi: 10.1016/j.ccr.2007.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Annunziata CM, Davis RE, Demchenko Y, Bellamy W, Gabrea A, Zhan F, et al. Frequent engagement of the classical and alternative NF-kappaB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell. 2007;12(2):115–30. doi: 10.1016/j.ccr.2007.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Calin GA, Dumitru CD, Shimizu M, Bichi R, Zupo S, Noch E, et al. Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA. 2002;99(24):15524–9. doi: 10.1073/pnas.242606799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cimmino A, Calin GA, Fabbri M, Iorio MV, Ferracin M, Shimizu M, et al. miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc Natl Acad Sci USA. 2005;102(39):13944–9. doi: 10.1073/pnas.0506654102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Levine AJ. p53, the cellular gatekeeper for growth and division. Cell. 1997;88(3):323–31. doi: 10.1016/s0092-8674(00)81871-1. [DOI] [PubMed] [Google Scholar]
- 59.Hunter Z, Xu L, Zhou Y, Yang G, Liu X, Cao Y, et al. Whole-Genome sequencing results from 30 patients with Waldenstrom’s macroglobulinemia. Blood (ASH Annual Meeting Abstracts) 2011;118 Abstract 434. [Google Scholar]
- 60.Xu L, Sohani AR, Arcaini L, Hunter Z, Yang G, Zhou Y, et al. A somatic variant in MYD88 (L265P) revealed by whole genome sequencing differentiates lymphoplasmacytic lymphoma from marginal zone lymphomas. Blood (ASH Annual Meeting Abstracts) 2011;118 Abstract 261. [Google Scholar]
- 61.Jeong E, Lee JY. Intrinsic and extrinsic regulation of innate immune receptors. Yonsei Med J. 2011;52(3):379–92. doi: 10.3349/ymj.2011.52.3.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhu J, Mohan C. Toll-like receptor signaling pathways--therapeutic opportunities. Mediators Inflamm. 2010;2010:781235. doi: 10.1155/2010/781235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ngo VN, Young RM, Schmitz R, Jhavar S, Xiao W, Lim KH, et al. Oncogenically active MYD88 mutations in human lymphoma. Nature. 2011;470(7332):115–9. doi: 10.1038/nature09671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281–97. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 65.He L, Hannon GJ. MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet. 2004;5(7):522–31. doi: 10.1038/nrg1379. [DOI] [PubMed] [Google Scholar]
- 66.Gangaraju VK, Lin H. MicroRNAs: key regulators of stem cells. Nat Rev Mol Cell Biol. 2009;10(2):116–25. doi: 10.1038/nrm2621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Hodge LS, Elsawa SF, Grote DM, Price Troska TL, Asmann YW, Fonseca R, et al. MicroRNA expression in tumor cells from Waldenstrom’s macroglobulinemia reflects both their normal and malignant cell counterparts. Blood Cancer Journal. 2011;1 doi: 10.1038/bcj.2011.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Roccaro AM, Sacco A, Chen C, Runnels J, Leleu X, Azab F, et al. microRNA expression in the biology, prognosis, and therapy of Waldenstrom macroglobulinemia. Blood. 2009;113(18):4391–402. doi: 10.1182/blood-2008-09-178228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sacco A, Issa GC, Zhang Y, Liu Y, Maiso P, Ghobrial IM, et al. Epigenetic modifications as key regulators of Waldenstrom’s macroglobulinemia biology. J Hematol Oncol. 2010;3:38. doi: 10.1186/1756-8722-3-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Roccaro AM, Sacco A, Jia X, Azab AK, Maiso P, Ngo HT, et al. microRNA-dependent modulation of histone acetylation in Waldenstrom macroglobulinemia. Blood. 2010;116(9):1506–14. doi: 10.1182/blood-2010-01-265686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Chng WJ, Schop RF, Price-Troska T, Ghobrial I, Kay N, Jelinek DF, et al. Gene-expression profiling of Waldenstrom macroglobulinemia reveals a phenotype more similar to chronic lymphocytic leukemia than multiple myeloma. Blood. 2006;108(8):2755–63. doi: 10.1182/blood-2006-02-005488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gutierrez NC, Ocio EM, de Las Rivas J, Maiso P, Delgado M, Ferminan E, et al. Gene expression profiling of B lymphocytes and plasma cells from Waldenstrom’s macroglobulinemia: comparison with expression patterns of the same cell counterparts from chronic lymphocytic leukemia, multiple myeloma and normal individuals. Leukemia. 2007;21(3):541–9. doi: 10.1038/sj.leu.2404520. [DOI] [PubMed] [Google Scholar]
- 73.Hodge DR, Hurt EM, Farrar WL. The role of IL-6 and STAT3 in inflammation and cancer. Eur J Cancer. 2005;41(16):2502–12. doi: 10.1016/j.ejca.2005.08.016. [DOI] [PubMed] [Google Scholar]
- 74.Hodge LS, Ansell SM. Jak/Stat pathway in Waldenstrom’s macroglobulinemia. Clin Lymphoma Myeloma Leuk. 2011;11(1):112–4. doi: 10.3816/CLML.2011.n.023. [DOI] [PubMed] [Google Scholar]
- 75.Elsawa SF, Novak AJ, Ziesmer SC, Almada LL, Hodge LS, Grote DM, et al. Comprehensive analysis of tumor microenvironment cytokines in Waldenstrom macroglobulinemia identifies CCL5 as a novel modulator of IL-6 activity. Blood. 2011;118(20):5540–9. doi: 10.1182/blood-2011-04-351742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hatjiharissi E, Ngo H, Leontovich AA, Leleu X, Timm M, Melhem M, et al. Proteomic analysis of Waldenstrom macroglobulinemia. Cancer Res. 2007;67(8):3777–84. doi: 10.1158/0008-5472.CAN-06-3089. [DOI] [PubMed] [Google Scholar]
- 77.Vivanco I, Sawyers CL. The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev Cancer. 2002;2(7):489–501. doi: 10.1038/nrc839. [DOI] [PubMed] [Google Scholar]
- 78.Leleu X, Jia X, Runnels J, Ngo HT, Moreau AS, Farag M, et al. The Akt pathway regulates survival and homing in Waldenstrom macroglobulinemia. Blood. 2007;110(13):4417–26. doi: 10.1182/blood-2007-05-092098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yoeli-Lerner M, Toker A. Akt/PKB signaling in cancer: a function in cell motility and invasion. Cell Cycle. 2006;5(6):603–5. doi: 10.4161/cc.5.6.2561. [DOI] [PubMed] [Google Scholar]
- 80.Brader S, Eccles SA. Phosphoinositide 3-kinase signalling pathways in tumor progression, invasion and angiogenesis. Tumori. 2004;90(1):2–8. doi: 10.1177/030089160409000102. [DOI] [PubMed] [Google Scholar]
- 81.Sacco A, Roccaro A, Ghobrial IM. Role of dual PI3/Akt and mTOR inhibition in Waldenstrom’s macroglobulinemia. Oncotarget. 2010;1(7):578–82. doi: 10.18632/oncotarget.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Guertin DA, Sabatini DM. The pharmacology of mTOR inhibition. Sci Signal. 2009;2(67) doi: 10.1126/scisignal.267pe24. pe24. [DOI] [PubMed] [Google Scholar]
- 83.Ghobrial IM, Gertz M, Laplant B, Camoriano J, Hayman S, Lacy M, et al. Phase II trial of the oral mammalian target of rapamycin inhibitor everolimus in relapsed or refractory Waldenstrom macroglobulinemia. J Clin Oncol. 2010;28(8):1408–14. doi: 10.1200/JCO.2009.24.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Harir N, Pecquet C, Kerenyi M, Sonneck K, Kovacic B, Nyga R, et al. Constitutive activation of Stat5 promotes its cytoplasmic localization and association with PI3-kinase in myeloid leukemias. Blood. 2007;109(4):1678–86. doi: 10.1182/blood-2006-01-029918. [DOI] [PubMed] [Google Scholar]
- 85.Ward AC, Touw I, Yoshimura A. The Jak-Stat pathway in normal and perturbed hematopoiesis. Blood. 2000;95(1):19–29. [PubMed] [Google Scholar]
- 86.Ngo HT, Leleu X, Lee J, Jia X, Melhem M, Runnels J, et al. SDF-1/CXCR4 and VLA-4 interaction regulates homing in Waldenstrom macroglobulinemia. Blood. 2008;112(1):150–8. doi: 10.1182/blood-2007-12-129395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Tsingotjidou AS, Emmanouilides CE, Siotou E, Poutahidis T, Xagorari A, Loukopoulos P, et al. Establishment of an animal model for Waldenstrom’s macroglobulinemia. Exp Hematol. 2009;37(4):469–76. doi: 10.1016/j.exphem.2008.12.007. [DOI] [PubMed] [Google Scholar]
- 88.Tassone P, Neri P, Kutok JL, Tournilhac O, Santos DD, Hatjiharissi E, et al. A SCID-hu in vivo model of human Waldenstrom macroglobulinemia. Blood. 2005;106(4):1341–5. doi: 10.1182/blood-2004-11-4477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Tassone P, Neri P, Burger R, Savino R, Shammas M, Catley L, et al. Combination therapy with interleukin-6 receptor superantagonist Sant7 and dexamethasone induces antitumor effects in a novel SCID-hu In vivo model of human multiple myeloma. Clin Cancer Res. 2005;11(11):4251–8. doi: 10.1158/1078-0432.CCR-04-2611. [DOI] [PubMed] [Google Scholar]