Abstract
Macrophage secretion of VEGF in response to hypoxia contributes to tumor growth and angiogenesis. In addition to VEGF, hypoxic macrophages stimulated with GM-CSF secrete high levels of a soluble form of the VEGF receptor (sVEGFR-1), which neutralizes VEGF and inhibits its biological activity. Using mice with a monocyte/macrophage-selective deletion of HIF-1α or HIF-2α, we recently demonstrated that the anti-tumor response to GM-CSF was dependent on HIF-2α-driven sVEGFR-1 production by tumor-associated macrophages, while HIF-1α specifically regulated VEGF production. We therefore hypothesized that chemical stabilization of HIF-2α using an inhibitor of prolyl hydroxylase 3 (PHD3; an upstream inhibitor of HIF-2α activation) would increase sVEGFR-1 production from GM-CSF-stimulated macrophages. Treatment of macrophages with the PHD3 inhibitor AKB-6899 stabilized HIF-2α and increased sVEGFR-1 production from GM-CSF-treated macrophages, with no effect on HIF-1α accumulation or VEGF production. Treatment of B16F10 melanoma-bearing mice with GM-CSF and AKB-6899 significantly reduced tumor growth compared to either drug alone. Increased levels of sVEGFR-1 mRNA, but not VEGF mRNA, were detected within the tumors of GM-CSF- and AKB-6899-treated mice, correlating with decreased tumor vascularity. Finally, the anti-tumor and anti-angiogenic effects of AKB-6899 were abrogated when mice were simultaneously treated with a sVEGFR-1 neutralizing antibody. These results demonstrate that AKB-6899 decreases tumor growth and angiogenesis in response to GM-CSF by increasing sVEGFR-1 production from tumor-associated macrophages. Specific activation of HIF-2α can therefore decrease tumor growth and angiogenesis.
INTRODUCTION
Elevated numbers of tumor-associated macrophages are associated with poor clinical outcome in a number of human cancers, including melanoma, breast, ovarian, and prostate cancer (1–4). Tumor-associated macrophages are well known to contribute to tumor progression by secreting the potent angiogenic molecule vascular endothelial growth factor (VEGF), which occurs largely in response to the low O2 concentration within the tumor microenvironment (5, 6). Development of the vertical growth phase in primary melanoma lesions correlates directly with vascular density and VEGF expression (7). VEGF is upregulated during melanoma progression and dissemination and is expressed at high levels in greater than 90% of metastatic lesions (8). Furthermore, in the clinical setting, serum levels of VEGF correlate with advanced disease, degree of tumor burden, and poor overall survival in melanoma patients (9). For this reason, VEGF is considered an attractive therapeutic target in melanoma, and numerous strategies have been employed to inhibit VEGF, including neutralizing Abs such as bevacizumab (Avastin), RNA interference, oral VEGF receptor inhibitors, and anti-VEGF receptor vaccines (10–13). Although these methods slow the growth and metastasis of human melanoma xenografts and B16F10 melanomas in immunocompetent mice, these strategies have been unsuccessful at limiting disease progression in melanoma patients.
Besides VEGF, low O2 upregulates the expression of the VEGF receptor (VEGFR-1), as well as genes involved in anaerobic metabolism, cell survival, and proliferation (14). In addition to the membrane-bound isoform of VEGFR-1, macrophages secrete a soluble form of the receptor (sVEGFR-1), which results from alternative splicing of the same gene transcript (15). sVEGFR-1 comprises the extracellular ligand-binding domain of the membrane-bound form of the receptor and serves as a potent antagonist of VEGF signaling by sequestering VEGF and inhibiting its interaction with the transmembrane receptors (16). We previously reported that in addition to VEGF, macrophages stimulated with GM-CSF at hypoxia secrete large quantities of sVEGFR-1, which binds the VEGF and inhibits angiogenesis (17). Using mice with a monocyte/macrophage-selective deletion of HIF-1α or HIF-2α, we demonstrated that the anti-tumor response to GM-CSF was dependent on HIF-2α-driven sVEGFR-1 production by tumor-associated macrophages, while HIF-1α in tumor macrophages specifically regulated VEGF production (18). These findings suggest that hypoxia, canonically thought to promote angiogenesis, can inhibit angiogenesis through HIF-2α stabilization, by inducing sVEGFR-1 production from tumor-associated macrophages.
The transcriptional response to hypoxia is driven primarily by a family of transcription factors known as the hypoxia inducible factors (HIFs). The HIFs are constitutively transcribed but are rapidly degraded under normoxic conditions, principally through the hydroxylation of proline residues by prolyl hydroxylase domain (PHD) proteins, of which there are three isoforms (PHD1–3). This modification allows binding of the von Hippel-Lindau (VHL) E3 ubiquitin ligase, which targets the HIF for ubiquitination and proteasomal degradation. Hypoxia promotes HIF protein accumulation by inhibiting PHD-mediated proline hydroxylation, as O2 is the rate-limiting co-factor in the hydroxylation reaction. Numerous studies in solid tumor cells have demonstrated that PHD2 preferentially hydroxylates HIF-1α, while hydroxylation of HIF-2α is mainly mediated by PHD3 (19). This implies that selective stabilization of HIF-2α could be achieved by specifically targeting PHD3. Because our previous results indicated that HIF-2α controls sVEGFR-1 production (18), we hypothesized that stabilization of HIF-2α (via inhibition of PHD3) would increase macrophage production of sVEGFR-1. In the current report, we demonstrate that combined treatment of tumor-bearing mice with GM-CSF and a small molecule inhibitor of PHD3 resulted in decreased tumor growth and angiogenesis as compared to either treatment alone, an effect which was dependent on the HIF-2α-driven production of sVEGFR-1 by tumor-associated macrophages. To our knowledge, these findings are the first to demonstrate that activation of a HIF protein can decrease tumor growth and angiogenesis, and provide support for the specific targeting of HIF-1α or HIF-2α as a therapeutic strategy. Furthermore, these data provide support for the administration of GM-CSF and AKB-6899 as a means of decreasing angiogenesis and inhibiting tumor growth in malignant melanoma.
MATERIALS AND METHODS
Purification of peripheral blood monocytes
Human peripheral blood mononuclear cells (PBMCs) were isolated from fresh peripheral blood leukocyte source packs (American Red Cross, Columbus OH) by density gradient centrifugation over Lymphocyte Separation Medium (Cellgro). Monocytes were purified from total PBMCs by layering over FBS, as previously described (20). Monocytes were cultured in endotoxin-free RPMI 1640 supplemented with 1% fetal bovine serum (FBS), 1% PSA (penicillin G sodium, streptomycin sulfate, and amphotericin B), and 10 μg/mL of the endotoxin inhibitor polymyxin B. Monocytes were treated for 24 hours with 10 ng/mL GM-CSF, 10 μM AKB-6899, or an equivalent volume of the vehicle controls (PBS or DMSO, respectively). Cell-free culture supernatants were harvested and analyzed for VEGF or sVEGFR-1 by ELISA (R&D Systems). AKB-6899 at concentrations up to 50 μM had no effect on the viability of peripheral blood monocytes, as determined by flow cytometric staining for annexin V and propidium iodide following 24 hours of incubation (data not shown).
AKB-6899 and AKB-4924
AKB-6899 is a novel substituted 3-hydroxypyridine-2-carbonyl glycine conjugate. This group of compounds was found to preferentially inhibit PHD3, resulting in HIF-2α stabilization. In contrast, AKB-4924 is a 4-substituted 3-hydroxy-2-oxo-1,2-dihydropyridine derivative. These compounds were found to strongly stabilize HIF-1α through PHD2 inhibition.
HIF-1αflox/flox/LysMcre and HIF-2αflox/flox/LysMcre mice and culture of bone marrow-derived macrophages
The generation of HIF-1αflox/flox/LysMcre and HIF-2αflox/flox/LysMcre mice has been previously described (18). In these mice, the HIF-2α gene is flanked by loxP sequences and cre recombinase is expressed from the macrophage lysozyme (LysM) promoter, resulting in a myeloid lineage-specific deletion of HIF-2α. LysMcre recombinase mice, which express no floxed alleles, were used as controls. Deletion of HIF-2α in HIF-2αflox/flox/LysMcre macrophages, but in not the LysMcre control macrophages, was confirmed at the transcript level by real-time PCR (data not shown). To generate bone marrow-derived macrophages (BDMs), femoral bone marrow was isolated and progenitor cells were plated in RPMI 1640 supplemented with 10% FBS, 1%PSA, 10 μg/mL polymyxin B, and 20 ng/mL of recombinant murine M-CSF. Cells were cultured for 5days with the addition of fresh M-CSF every other day. Differentiated BDM were serum-starved for 2 hours and then treated with 100 ng/mL of murine GM-CSF and/or 10 μM AKB-6899 in RPMI 1640 containing 1% FBS, 1% PSA, and 10 μg/mL polymixinB. Culture supernatants were collected after 24 hours and assayed for VEGF and sVEGFR-1 content by ELISA (R&D Systems). AKB-6899 at concentrations up to 50 μM had no effect on the viability of peripheral blood monocytes or of bone marrow-derived macrophages from LysMcre control, HIF-1αflox/flox/LysMcre, or HIF-2αflox/flox/LysMcre mice, as determined by annexin V/propidium iodide exclusion after 24 hours of culture (data not shown).
Real-time PCR
Human monocytes were left untreated or were stimulated with 100ng/mL GM-CSF, 10 μM AKB-6899, or the combination, at 21% O2. At various time-points, cells were harvested in Trizol reagent (Invitrogen) and RNA was extracted in chloroform and then purified using the RNeasy Minikit (Qiagen). In murine studies, organs harvested at the time of euthanasia were flash-frozen in liquid nitrogen, pulverized in liquid nitrogen, and then dissolved in Trizol. cDNA was generated from 1 μg of RNA using the Superscript First Strand Synthesis System (Invitrogen) and was used for real-time PCR with previously described primers (21) and SYBR Green PCR Master Mix (Applied Biosciences), according to the manufacturer’s instructions. Data were analyzed according to the comparative threshold method and normalized against the β-actin internal control transcript.
Murine melanoma tumor models
6–8-week-old C57BL/6 mice or SCID mice were injected with 1 × 105 B16F10 murine melanoma cells or 1 × 106 A375 human melanoma cells, respectively, subcutaneously on the left flank. Once tumors become palpable (approximately 5–7days), mice were randomly allocated to receive treatment with either: 20% PEG-400 in 5% sucrose (vehicle for AKB-6899) and PBS (vehicle for GM-CSF), 20% PEG-400 and GM-CSF (100 ng per mouse in a 50 μL volume), AKB-6899 (17.5 mg/kg mouse weight in a 100 μL volume) and PBS, or AKB-6899 and GM-CSF (same concentrations). AKB-6899 (or the vehicle control) was administered intraperitoneally, while GM-CSF (or the vehicle control) was administered intratumorally. Mice were treated intratumorally 3 times per week until tumors reached a size of 20 mm in any dimension (approximately 2.5 weeks), at which point mice were be euthanized, in accordance with institutional policy. Tumor diameters were measured 3 times per week with calipers, and tumor volumes will be calculated as follows: Tumor volume = 0.5 × [(large diameter) × (small diameter)2]. For experiments analyzing the effect of neutralizing sVEGFR-1 in combination with AKB-6899 treatment, mice were treated intraperitoneally 3x/week with either AKB-6899 or vehicle control, and intratumorally with 4 μg of either an anti-VEGFR-1 neutralizing antibody (R&D Systems) or a polyclonal goat IgG isotype control (Santa Cruz Biotechnology) in a 50 μL volume. All protocols were approved by the Ohio State University Animal Care and Use Committee, and mice were treated in accordance with institutional guidelines for animal care.
Evaluation of lung metastases
Lung metastases were evaluated in B16F10-bearing mice by detection of mRNA for murine melanocyte-specific proteins within the lungs of tumor-bearing mice. B16F10 tumor-bearing mice were treated with intratumoral GM-CSF and/or intraperitoneal AKB-6899 (or vehicle controls), as described above. At the time of sacrifice, lungs were excised and flash-frozen in liquid nitrogen. Frozen lungs were homogenized in liquid nitrogen and the pulverized material was dissolved in Trizol reagent (Invitrogen). RNA was extracted in chloroform and purified using the RNeasy Minikit (Qiagen). cDNA was generated from 1 μg of RNA using the Superscript First Strand Synthesis System (Invitrogen) and used for real-time PCR using SYBR Green PCR MasterMix (Applied Biosciences) according to the manufacturers’ instructions. The melanocyte-specific mRNA Pmel17 was detected by nested PCR using a modification of the protocol described by Tsukamoto et al. (22). For the initial reaction, 30 cycles of PCR were carried out (95°C for 1 minute, 58°C for 1 min, 72°C for 1 min) in a 20 μl reaction volume containing 2 μl of cDNA. For reamplification with the nested primers, 1 μL of the first reaction product was amplified in a 20 μl reaction volume for a further 30 cycles. Data were analyzed according to the comparative threshold method and normalized against the β-actin internal control transcript. Results are semi-quantitative and represent the fold difference in transcript levels in AKB-6899-and/or GM-CSF-treated mice as compared with levels in vehicle control mice.
Statistical analyses
The ANOVA test was used to compare independent measurements between multiple treatment groups. The data was log-transformed to normalize the variance across groups. P-values were adjusted using the Holm’s procedure to conserve the type I error at 0.05 due to multiple comparisons. For tumor growth data, changes in tumor volume over time were assessed via a longitudinal model. Tumor values were log-transformed, and estimated slopes (changes in tumor volume over time) were calculated with 95% confidence intervals. Estimated differences in tumor volume were calculated by a random-effects regression of the longitudinal data. For all analyses, p ≤ 0.05 was considered statistically significant.
RESULTS
Inhibition of PHD3 with AKB-6899 stabilizes HIF-2α and enhances monocyte and macrophage production of sVEGFR-1
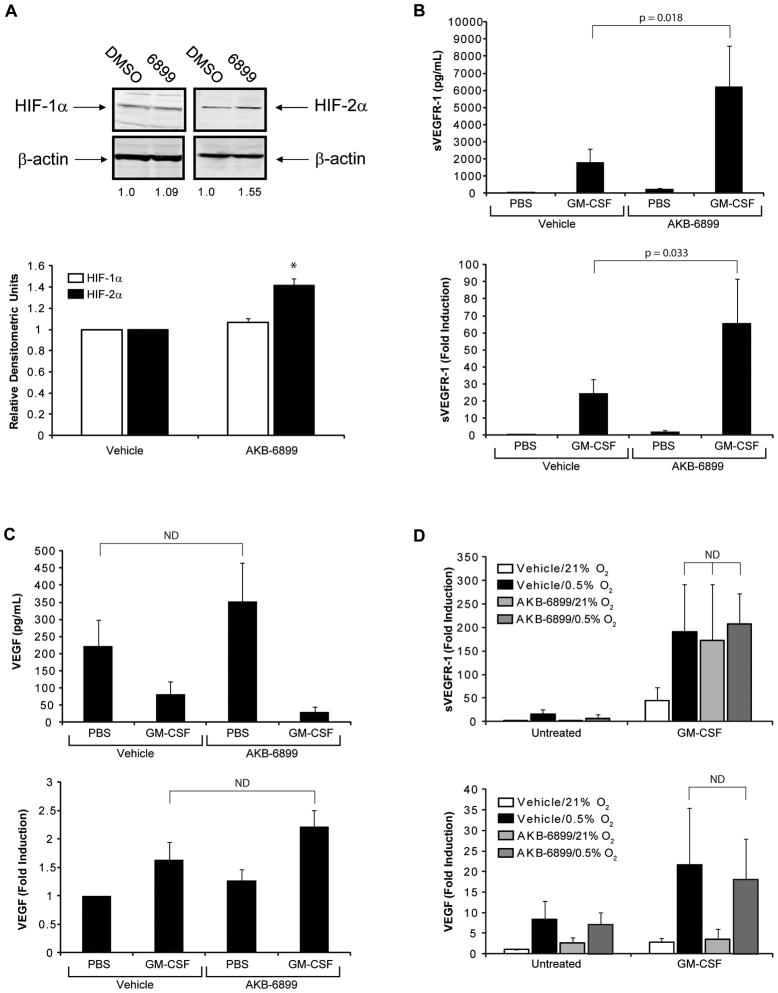
We have previously shown that monocyte production of sVEGFR-1 in response to GM-CSF and hypoxia is dependent on HIF-2α, while HIF-1α controlled monocyte production of VEGF under the same conditions (17). We therefore hypothesized that selective stabilization of HIF-2α would enhance sVEGFR-1 production from GM-CSF-stimulated monocytes without affecting VEGF production. In order to confirm the selective upregulation of HIF-2α by AKB-6899, murine bone marrow-derived macrophages were treated with 10 μM AKB-6899 for 18 hours, and cell lysates were immunoblotted for HIF-1α and HIF-2α. We observed an increase in HIF-2α protein in cells treated with AKB-6899 (p = 0.001), with no corresponding increase in HIF-1α (p = 0.105) (Figure 1A), confirming the specific effect of AKB-6899 on HIF-2α accumulation. However, AKB-6899 treatment did not increase levels of HIF-1α or HIF-2α mRNA, confirming the effect of AKB-6899 on HIF-2α protein accumulation. To determine whether stabilization of HIF-2α increased sVEGFR-1 production, human peripheral blood monocytes were stimulated with 100 ng/mL GM-CSF in the presence or absence of 10 μM AKB-6899. sVEGFR-1 production by GM-CSF-treated monocytes increased significantly when monocytes were also treated with AKB-6899, at both the protein and the transcript level (p = 0.018and p =0.033, respectively) (Figure 1B). VEGF levels in the same supernatants were then measured using an ELISA that detects free (bioavailable) VEGF, but does not detect VEGF bound to sVEGFR-1. Treatment of cells with AKB-6899 did not significantly increase production of VEGF (p = 0.133). Detection of VEGF protein was reduced in the supernatants of GM-CSF-stimulated monocytes, due to neutralization of VEGF by sVEGFR-1 (21) (Figure 1C). Evaluation of VEGF transcript levels by real-time PCR revealed that while GM-CSF slightly increased VEGF production, there was no difference in VEGF expression between monocytes stimulated with GM-CSF alone or with GM-CSF/AKB-6899 (p = 0.558) (Figure 1C). Finally, human monocytes were stimulated with GM-CSF at 0.5% O2 or with GM-CSF/AKB-6899 at normoxia, in order to compare the effect of chemical HIF-2α stabilization with the effect of HIF-2α stabilization by hypoxia. As previously observed, 100 ng/mL GM-CSF increased sVEGFR-1 production, which increased further when cells were stimulated with GM-CSF at 0.5% O2 or when cells were stimulated with GM-CSF at ambient O2 in the presence of 10 μM AKB-6899. However, the amount of sVEGFR-1 production from monocytes stimulated with GM-CSF at 0.5% O2 was equivalent to the amount produced by monocytes stimulated with GM-CSF at ambient O2 in the presence of AKB-6899 (Figure 1D). Furthermore, stimulation of monocytes with AKB-6899 at 0.5% O2 did not further increase sVEGFR-1 production compared to monocytes stimulated with AKB-6899 at normoxia, suggesting that maximal stabilization of HIF-2α was achieved with AKB-6899. The combination of GM-CSF and 0.5% O2 (which activates both HIF-1α and HIF-2α) also increased monocyte production of VEGF, as observed previously (17), whereas stimulation with AKB-6899 (which selectively stabilizes HIF-2α) at normoxia did not (Figure 1D). Furthermore, stimulation of monocytes with AKB-6899 at 0.5% O2 did not further increase VEGF production over that which was observed with hypoxia alone, suggesting that AKB-6899 had no effect on VEGF production, regardless of O2 concentration. These results demonstrate that inhibition of PHD3 with AKB-6899 stabilizes HIF-2α and selectively induces sVEGFR-1 from GM-CSF-stimulated monocytes to the same extent as hypoxia, while HIF-1α accumulation and VEGF production are unaffected by AKB-6899 treatment.
Figure 1. AKB-6899 stabilizes HIF-2α and increases macrophage production of sVEGFR-1 in response to GM-CSF.
(A) Murine bone marrow-derived macrophages stimulated for 24 hours with AKB-6899 or an equivalent volume of DMSO (vehicle control) were immunoblotted for HIF-1α or HIF-2α. The numbers below the immunoblots represent the fold increase in HIF levels, normalized to β-actin protein and expressed in relative densitometric units. Immunoblots from one representative donor are shown; the graph represents the mean ± SEM relative densitometric units from 3 independent experiments. (B) Human peripheral blood monocytes were cultured in media containing PBS or 10 ng/mL GM-CSF, and DMSO (vehicle control) or 10 μM AKB-6899. After 24 hours, culture supernatants were harvested and analyzed for sVEGFR-1 content by ELISA (top panel), while cells were lysed in Trizol and analyzed for sVEGFR-1 transcript by real-time PCR (bottom panel). Results shown are the mean ± SEM of a total of 12 healthy donors from 3 independent experiments. (C) The same supernatants as in (B) were analyzed using an ELISA that detects only bioavailable VEGF (i.e., VEGF that is not bound to sVEGFR-1) (top panel). RNA from the cells in (B) was also analyzed for VEGF expression by real-time PCR (bottom panel). (D) Human monocytes were treated with 10 ng/mL GM-CSF and/or 10 μM AKB-6899 at normoxia (21% O2) or hypoxia (0.5% O2). sVEGFR-1 and VEGF induction were analyzed by real-time PCR. Results represent the mean ± SEM of 4 donors from 2 independent experiments. *, p = 0.001 vs. vehicle control-treated macrophages. **, p = 0.036 vs. vehicle-treated macrophages from LysMcre control mice. ND, no difference.
Because our previous results indicated that monocyte production of VEGF was dependent on HIF-1α, we further hypothesized that selective stabilization of HIF-1α via inhibition of PHD2 would increase monocyte production of VEGF but not sVEGFR-1. To address this hypothesis, human peripheral blood monocytes were stimulated with GM-CSF in the presence of AKB-4924, a selective inhibitor of PHD2, which results in HIF-1α stabilization. As previously observed, GM-CSF induced monocyte production of sVEGFR-1. However, there was no difference in sVEGFR-1 production from monocytes stimulated with GM-CSF alone or monocytes co-stimulated with AKB-4924, at either the protein (p = 0.306) or transcript level (p = 0.566) (Figure 2A). However, AKB-4924 increased monocyte production of VEGF protein (p = 0.011) and mRNA (0.007, respectively)(Figure 2B). These observations confirm the preferential effect of PHD3 on HIF-2α stabilization (and of PHD2 on HIF-1α stabilization) and confirm that sVEGFR-1 production from human monocytes and macrophages is specifically dependent on HIF-2α.
Figure 2. The HIF-1α-stabilizing compound AKB-4924 increases macrophage production of VEGF but does not affect sVEGFR-1 production.

(A) Human peripheral blood monocytes were stimulated with GM-CSF and/or AKB-4924 (or the appropriate controls). After 24 hours, culture supernatants were harvested and analyzed for sVEGFR-1 content by ELISA (top panel). Cells were simultaneously harvested and sVEGFR-1 transcript expression was evaluated by real-time PCR (bottom panel). (B) The same supernatants and cells as in (A) were analyzed for VEGF content by ELISA (top panel) or for VEGF transcript by real-time PCR (bottom panel), respectively. All results shown are the mean ± SEM of a total of 5 healthy donors from 2 independent experiments. ND, no difference.
sVEGFR-1 secretion in response to AKB-6899 is dependent on HIF-2α
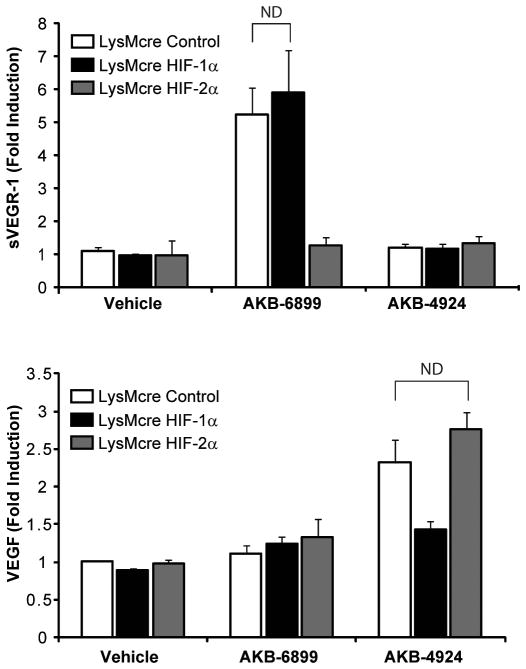
In order to confirm the specificity of AKB-4924 and AKB-699 in stabilizing HIF-1α and HIF-2α, respectively, we utilized bone marrow-derived macrophages from mice with a myeloid-specific deletion of HIF-1α or HIF-2α (HIF-1αflox/flox/LysMcre or HIF-2αflox/flox/LysMcre mice). AKB-6899 induced comparable levels of sVEGFR-1 in control macrophages and HIF-1α-deficient macrophages, but did not induce sVEGFR-1 in macrophages lacking HIF-2α. These results indicate that HIF-2α, but not HIF-1α, is required for AKB-6899-induced sVEGFR-1 production (Figure 3). In addition, AKB-4924 induced VEGF transcription in control macrophages and HIF-2α-deficient macrophages but not in macrophages deficient in HIF-1α (Figure 3), confirming that AKB-4924-induced VEGF expression is dependent on HIF-1α but not HIF-2α. These results are in agreement with our previous findings that sVEGFR-1 transcription is HIF-2α-dependent, while HIF-1α controls VEGF transcription (17). Furthermore, these findings confirm the specificity of AKB-6899 in inducing HIF-2α-dependent, but not HIF-1α-dependent, gene transcription.
Figure 3. sVEGFR-1 production from AKB-6899-stimulated macrophages is dependent on HIF-2α but not HIF-1α.
Bone marrow-derived macrophages from LysMcre control mice, HIF-1αflox/flox/LysMcre mice, or HIF-2αflox/flox/LysMcre mice were stimulated for 24 hours with 10 μM AKB-6899 or AKB-4924, and sVEGFR-1 and VEGF transcript levels were analyzed by real-time PCR. ND, no difference.
Stabilization of HIF-2α increases the anti-tumor effects of GM-CSF and enhances survival in a murine melanoma model
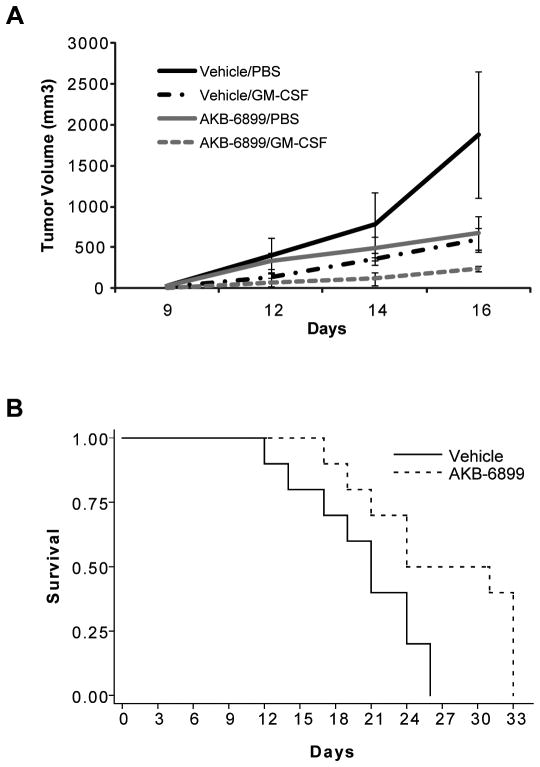
We previously demonstrated that the anti-tumor effects of GM-CSF are dependent on HIF-2α-mediated sVEGFR-1 production from tumor-associated macrophages in a murine melanoma model (18). We therefore hypothesized that chemical stabilization of HIF-2α would increase sVEGFR-1 production from tumor-associated macrophages and enhance the anti-tumor effects of GM-CSF. Mice bearing subcutaneous B16F10 melanomas were treated 3x/week with GM-CSF (100 ng/mouse, intratumoral), AKB-6899 (17.5 mg/kg, intraperitoneal), or the combination (or the appropriate vehicle controls). Based on a longitudinal model using log-transformed values, no significant differences in tumor volume were found between the four groups at baseline. However, at day 16 of treatment, the average tumor volumes for mice receiving either GM-CSF or AKB-6899 were significantly smaller than for mice treated with the vehicle controls (p < 0.001). Furthermore, combined treatment with GM-CSF plus AKB-6899 further decreased tumor growth compared to either treatment alone (p < 0.001 for GM-CSF or AKB-6899 vs. GM-CSF plus AKB-6899) (Figure 4A). Because we have previously shown that GM-CSF alone enhanced survival in a murine breast cancer model (23), we wanted to evaluate the effect of AKB-6899 on survival. As shown in Figure 4B, AKB-6899 increased the median survival (defined as the time to a tumor diameter of 20 mm3) by 3 days in B16F10 melanoma-bearing mice (p = 0.023).
Figure 4. AKB-6899 enhances the anti-tumor effect of GM-CSF in a murine melanoma model.
(A) Mice with subcutaneous B16F10 melanoma tumors were treated 3 times per week intratumorally with PBS or GM-CSF (100 ng/mouse) and intraperitoneally with AKB-6899 (17.5 mg/kg) or the vehicle control (20% PEG in 5% dextran). Tumor dimensions were measured 3 times per week, and tumor volumes were calculated as described in Materials and Methods. Each data point represents the mean tumor volume of at least 15 mice/group, total, from 2 independent experiments. (B) Kaplan-Meier survival analysis of AKB-6899 in B16F10 melanoma. Mice were inoculated with subcutaneous B16F10 tumors and treated 3 times weekly with 17.5 mg/kg AKB-6899 or the vehicle control (n = 10 mice/group, total, from 2 independent experiments). Survival was determined as the time to a tumor size of 20 mm in any dimension.
AKB-6899 enhances sVEGFR-1 production and decreases tumor angiogenesis in response to GM-CSF
We had hypothesized that chemical stabilization of HIF-2α with AKB-6899 would increase sVEGFR-1 production in response to GM-CSF, thereby reducing tumor growth and angiogenesis. Real-time PCR was therefore used to evaluate the levels of sVEGFR-1 and VEGF mRNA within tumors from mice treated with GM-CSF, AKB-6899, or the combination. Increased levels of sVEGFR-1 were detected within the tumors of mice treated with both GM-CSF and AKB-6899 (p = 0.001) (Figure 5A). Conversely, GM-CSF (alone or in combination with AKB-6899) failed to increase levels of intratumoral VEGF over the levels observed in vehicle control-treated mice (p = 0.456) (Figure 5B). To confirm that the increased sVEGFR-1 production resulted in decreased tumor angiogenesis, tumors from each of the mice were stained by immunohistochemistry for the endothelial cell marker CD31. As shown in Figure 5C, combination treatment with GM-CSF and AKB-6899 significantly reduced tumor vascularity in melanoma-bearing mice (p < 0.001), possibly through the induction of sVEGFR-1. We have previously demonstrated that GM-CSF induced macrophage infiltration into B16F10 melanoma tumors (18). In order to determine whether the observed differences in sVEGFR-1 levels were due to differences in numbers of sVEGFR-1-secreting macrophages within the tumor, tumor sections were stained for the murine macrophage marker F4/80 antigen by immunohistochemistry. Consistent with previous observations (18), an increase in tumor-infiltrating macrophages was observed in response to GM-CSF treatment (p = 0.048). However, there was no difference in macrophage infiltration into the tumors of mice treated with GM-CSF alone or with GM-CSF plus AKB-6899 (p = 0.806) (Figure 5D). These results indicate that HIF-2α stabilization does not affect macrophage trafficking into the tumor and indicate that the differences in tumor growth observed between GM-CSF and GM-CSF/AKB-6899-treated mice are due to differences in the magnitude of sVEGFR-1 production by tumor-associated macrophages, not due to differences in the total numbers of tumor-associated macrophages. Because increased angiogenesis is associated with an increased risk of metastasis, and because the B16F10 melanoma preferentially metastasizes to the lungs (24), we also evaluated lung metastasis by assessing transcript levels of the melanocyte-specific gene Pmel17 in the lungs of mice treated with GM-CSF, AKB-6899, or the combination. Significantly reduced levels of Pmel17 were detected within the lungs of mice treated with GM-CSF and AKB-6899, as compared to vehicle control-treated mice (p = 0.048) (Figure 5E). These results demonstrate that AKB-6899 enhances the anti-angiogenic effects of GM-CSF, possibly by increasing sVEGFR-1 production from tumor-associated macrophages.
Figure 5. AKB-6899 increases intratumoral sVEGFR-1 and decreases angiogenesis and distal metastasis in a murine melanoma model.
B16F10 tumor-bearing mice were treated with GM-CSF, AKB-6899, or the combination, as described in Figure 4A. At the time of sacrifice, tumors were harvested and flash-frozen in liquid nitrogen. Frozen tissue was pulverized in liquid nitrogen and dissolved in Trizol (Invitrogen), RNA was extracted, and real-time PCR was performed for sVEGFR-1 (A) or VEGF (B). Results represent the mean ± SEM mRNA level as compared with the levels in Vehicle/PBS-treated mice. Each group contains at least 15 tumors. A section of the tumor was harvested and fixed in 10% formalin and analyzed for CD31 (C) or the macrophage marker F4/80 antigen (D) by immunohistochemistry. For both C and D, representative images from mice in each of the treatment groups are shown at 10X magnification. Immunostaining was digitally quantified by comparing the number of positive (brown) pixels to the total number of pixels in each high-powered field (HPF) for stitched images taken across entire tumors. The graphs show the average percentage of CD31-positive or F4/80 antigen-positive pixels for at least 15 mice per treatment group. (E) At the time of sacrifice of mice from Figure 4A, lungs were flash-frozen in liquid nitrogen and dissolved in Trizol to extract RNA, and a nested real-time PCR was performed for the melanoma-specific gene Pmel17. Results represent the mean ± SEM mRNA levels in each treatment group as compared to the level in Vehicle/PBS-treated mice, for at least 15 mice/group. *, p = 001. **, p < 0.001. #, p = 0.456. ND, no difference.
The anti-tumor effects of AKB-6899 are dependent on sVEGFR-1 production
We observed increased sVEGFR-1 levels in the tumors of mice treated with GM-CSF and AKB-6899, correlating with decreased tumor growth and angiogenesis. To confirm that the modulation of tumor growth and angiogenesis was due to sVEGFR-1 production in response to AKB-6899, mice were treated with AKB-6899 in the presence or absence of asVEGFR-1 neutralizing Ab. As previously observed, AKB-6899 decreased tumor growth in mice treated with an isotype control antibody (p < 0.001), but had no effect on tumor growth in mice also treated with the anti-sVEGFR-1 neutralizing antibody (p = 0.245) (Figure 6A). To confirm the role of sVEGFR-1 production in tumor angiogenesis, tumor sections were immunostained for the endothelial cell marker CD31. As shown in Figure 6B, AKB-6899 decreased tumor vascularity in the mice treated with the control antibody (p = 0.048) but not in the mice treated with the sVEGFR-1 neutralizing Ab (p = 0.128). These results demonstrate that AKB-6899 decreases tumor angiogenesis by inducing sVEGFR-1.
Figure 6. The anti-tumor effect of AKB-6899 is dependent on sVEGFR-1 production.
(A) Mice with subcutaneous B16F10 melanomas were treated 3 times per week intraperitoneally with 17.5 mg/kg AKB-6899 (or the vehicle control) and intratumorally with an anti-sVEGFR-1 neutralizing antibody (4 μg/mouse) or an equivalent amount of a polyclonal goat isotype control antibody. Tumor dimensions were measured, and tumor volumes were calculated as in Materials and Methods. (B) At the time of sacrifice, tumors from mice in A were fixed in 10% formalin and immunostained for the endothelial cell marker CD31. Positive immunostaining was digitally quantified as described for Figure 5C. Results represent the mean ± SEM of a total of at least 10 mice/group from 2 independent experiments.
AKB-6899 and GM-CSF reduce tumor growth in a mouse model of human melanoma
We next evaluated the anti-tumor effects of AKB-6899, GM-CSF, or the combination in immunodeficient (SCID) mice bearing human melanoma xenografts of the A375 cell line, using the same treatment schema described above for the B16F10 murine tumor cell line. GM-CSF/AKB-6899 treatment significantly decreased tumor growth in this model (p = 0.05) (Figure 7). These data demonstrate that AKB-6899 can enhance the anti-tumor effects of GM-CSF in both murine and human melanoma.
Figure 7. AKB-6899 and GM-CSF decrease growth of a human melanoma xenograft.

SCID mice bearing subcutaneous A375 tumors (a human melanoma cell line) were treated with intraperitoneal AKB-6899 and/or intratumoral GM-CSF, as described for Figure 4A. Tumor dimensions were measured three times weekly, and tumor volumes were calculated as described in the Materials and Methods section. Results represent the mean ± SEM of at least 10 mice/group, total, from 2 independent experiments.
DISCUSSION
Recently we described the therapeutic potential of activating the HIF pathway in macrophages for the purpose of inhibiting tumor angiogenesis and that HIF-1α and HIF-2α had competing roles for regulating vascularization (17). Subsequently, we illustrated in a model of murine melanoma that GM-CSF regulates HIF-2α stability, even in normoxia, to up-regulate the expression of the soluble form of VEGF receptor-1 (sVEGFR-1) from mononuclear phagocytes (18). The suggestion that HIF-2α could play a role in tumor suppression was first identified by Acker et al, who described that HIF-2α overexpression in rat glioma tumors, while augmenting vascularization, actually led to increased tumor cell apoptosis, while HIF-2α-deficiency increased angiogenesis (25). In our current study, we extend our understanding of HIF pathway regulation by introducing a novel small molecule PHD3 inhibitor, AKB-6899, which selectively stabilizes HIF-2α and leads to a synergistic increase in GM-CSF-induced sVEGFR-1.
sVEGFR-1 is secreted by a limited number of cell types, including monocytes/macrophages, vascular endothelial cells, vascular smooth muscle cells, placental trophoblasts, corneal epithelial cells, and proximal tubular cells of the renal epithelia (26). Of those cell types, only vascular endothelial cells and mononuclear phagocytes are present within the tumor microenvironment and could possibly contribute to the intratumoral sVEGFR-1 expressed following AKB-6899/GM-CSF co-treatment. We have previously demonstrated that vascular endothelial cells fail to upregulate sVEGFR-1 in response to 0.5% O2, suggesting that these cells would also fail to secrete sVEGFR-1 in response to AKB-6899 (17). Furthermore, vascular endothelial cells do not express GM-CSF receptor subunits (27), and therefore are unlikely to contribute to the increased sVEGFR-1 production observed in response to GM-CSF and AKB-6899. We assessed sVEGFR-1 production from human umbilical vein endothelial cells (HUVECs) cultured with GM-CSF and/or AKB-6899. HUVECs secreted a low basal amount of sVEGFR-1, which did not increase in response to GM-CSF, AKB-6899, or the combination (data not shown). As a control, the VEGF content of the same supernatants was analyzed. While VEGF was secreted by HUVECs cultured at 0.5% O2, AKB-6899 did not induce VEGF production from HUVECs, either alone or in combination with GM-CSF. These results indicate that tumor-infiltrating macrophages are the primary source of sVEGFR-1 within the tumors of GM-CSF-and AKB-6899-treated mice.
GM-CSF increases the proliferation and activation of tumor-specific T cells and increases antigen presentation from dendritic cells and macrophages, and therefore has been considered as a potential cancer therapeutic. However, intravenous or subcutaneous recombinant GM-CSF (Leukine) was ineffective at limiting melanoma growth in phase I/II studies, and can be accompanied by severe dose-limiting toxicities (28, 29). Because systemic administration of GM-CSF is ineffective at inhibiting cancer growth, we examined the effect of local administration of GM-CSF on tumor growth and angiogenesis. Intratumoral treatment with GM-CSF induces a slight increase in VEGF production from tumor-infiltrating macrophages but a large concomitant increase in intratumoral sVEGFR-1, an effect that was associated with an overall decrease in tumor growth and angiogenesis (18, 23) VEGF inhibition in melanoma patients is relevant as studies show that serum VEGF is prognostic of outcome (30, 31). These studies suggest that local administration of GM-CSF to induce the endogenous production of sVEGFR-1 might be a novel means of targeting VEGF in melanoma. In fact, studies using chemotherapy compounds such as dacarbazine (32) or solvent-free Nab-Paclitaxel (33) in combination with bevacizumab displayed promising effects in clinical trials in patients with late-stage melanoma. However, the need for new alternatives to the anti-angiogenic component of such treatment protocols persists.
AKB-6899 was developed for the treatment of chronic anemia, as HIF-2α also controls the production of the red blood cell growth factor erythropoietin. In ongoing phase II clinical trials, a related compound AKB-6548 is well-tolerated and effective at inducing the transcription of HIF-2α-dependent genes such as erythropoietin with little effect on HIF-1α-dependent genes such as VEGF (personal communication, Akebia Therapeutics). Our observation that inhibition of PHD3 with AKB-6899 resulted in HIF-2α accumulation and sVEGFR-1 production, while inhibition of PHD2 with AKB-4924 resulted in HIF-1α accumulation and VEGF production, illustrates the specificity of these inhibitors for the different PHD isoforms, confirms our previously described link between HIF-2α and sVEGFR-1 (18), and validates the strategy of inhibiting PHD3 as a means of specifically inducing HIF-2α-dependent transcription. The finding that inhibition of PHD3 and accumulation of HIF-2α led to an anti-angiogenic phenotype in our model is novel and may have relevance in the treatment of human cancer.
One concern of the present research was that systemically administered AKB-6899 is not specifically targeted to macrophages and therefore would inhibit PHD in tumor cells and associated endothelial cells, not selectively in tumor-associated macrophages. While we hypothesized that HIF-2α stabilization in macrophages would enhance sVEGFR-1 production following GM-CSF treatment, the effect of HIF-2α stabilization in other cell types comprising the tumor was unknown. If HIF-2α stabilization resulted in VEGF production from other tumor-associated cell types, AKB-6899 might enhance, rather than inhibit, tumor growth. It has been demonstrated that HIF-1α, not HIF-2α, primarily controls VEGF secretion from breast tumor cells (34). Furthermore, over-expression of HIF-2α in endothelial cells did not increase VEGF production in response to hypoxia (35). In the same report, HIF-2α did not regulate VEGF production in vascular endothelial cells in a murine model of melanoma (35). Consistent with these findings, VEGF was not upregulated in AKB-6899-treated tumors. It therefore appears that HIF-2α stabilization did not increase VEGF production from other cell types within the tumor microenvironment. Furthermore, it is possible that AKB-6899 could have anti-tumor effects other than the induction of sVEGFR-1 from tumor macrophages. However, neutralization of sVEGFR-1 completely reversed the inhibitory effect of AKB-6899 on tumor growth in the present study (Figure 7). This was associated with a failure of AKB-6899 to decrease angiogenesis in the tumors of these mice. We therefore feel that, at least in this specific tumor model, it is appropriate to conclude that the anti-tumor effects of AKB-6899 are dependent on the reduction of angiogenesis resulting from sVEGFR-1 production by tumor macrophages. Studies to identify other potential anti-tumor mechanisms of AKB-6899, in macrophages as well as in other cell types of the tumor microenvironment, are ongoing in our laboratory.
Other small molecule inhibitors or interference strategies to eliminate PHD activity led to increased angiogenesis in ischemic conditions (36). In fact, in one such study, a plasmid-directed, shRNA-specific knockout of PHD1, −2, or −3 promoted revascularization in mice that had undergone right femoral artery ligation. In that report, PHD2 or PHD3 inhibition increased vessel and capillary density and foot perfusion predominantly by stabilizing HIF-1α (37). While their study employed local inhibition of PHD2 and PHD3 at the plasmid injection site, directly on the endothelium, our study focuses on macrophage activation using GM-CSF coupled with PHD3 inhibition by AKB-6899, which may explain the contradictory results. The idea that mere inhibition of the PHDs would drive uncontrolled tumor vascularization and growth was dismissed when Mazzone et al (38) showed that loss of PHD2 activity (and unregulated HIF-1α accumulation) generates a dysfunctional tumor vasculature and augments VEGFR-1 and VE-cadherin expression on endothelial cells, resulting in a reduction of metastasis. Reports such as these highlight the complexity of the HIF system and emphasize the need for further investigation into HIF regulation.
In another report, we described GM-CSF-derived mononuclear phagocyte production of sVEGFR-1 as JAK/STAT-dependent (17). In the current work, we observed a HIF2α-dependent augmentation of sVEGFR-1 when treating with AKB-6899 in combination with GM-CSF, demonstrating an alternative pathway for the production of sVEGFR-1. Work is underway in our laboratory elucidating the intersection of these signaling pathways.
The fact that GM-CSF/AKB-6899 combination therapy inhibited tumor growth in the A375 human tumor cell line, which contains the B-RAF single point mutation V600E, without the use of a B-RAF inhibitor, is encouraging. This data suggests therapeutic potential for treatment of those approximately 40% of melanoma patients who do not possess the V600E mutation and in whom PLX4032 (a small molecule inhibitor of B-RAF) actually stimulates melanoma tumor growth (39, 40). Mutations within the kinase domain of B-RAF (a serine/threonine kinase and a constituent of the MAP kinase pathway) are observed in over 60% of patients with malignant melanoma (41) and are present in approximately 20% of other malignancies (42). For this reason, B-RAF inhibitors have become an effective treatment for patients possessing these mutations, the most frequent being the V600E single substitution. For solid tumors without these mutations, especially those where targeted therapies are not possible (such as triple-negative breast cancers and certain melanomas), the discovery of novel therapies are warranted. Combination studies using GM-CSF/AKB-6899 in tumor cell lines without B-RAF mutations are currently underway in our laboratory, and the efficacy of this combination treatment will be compared to conventional cytotoxic chemotherapies like the DNA alkylating agent dacarbazine, and anti-angiogenic therapies such as bevacizumab (VEGF neutralizing antibody) or VEGFTrap (VEGFR-1:Fc gamma chimera neutralizing VEGF).
Consistent with our previous results, an increase in tumor-infiltrating macrophages was observed in GM-CSF-treated mice (18, 23). However, no difference in macrophage infiltration was observed between mice treated with GM-CSF alone or with GM-CSF/AKB-6899. These results demonstrate HIF-2α stabilization by AKB-6899 does not affect macrophage trafficking into the tumor and indicate that the differences in tumor growth observed among treatment groups are due to differences in the phenotype of tumor-infiltrating macrophages, not due to differences in their numbers. In an earlier work, we reported other potential benefits of GM-CSF besides inducing mononuclear phagocytes to produce high concentrations of sVEGFR-1. We found that GM-CSF helps maintain an M1 tumor macrophage phenotype in a mouse model of breast cancer by up-regulating iNOS and down-regulating arginase-1, IL-4, and IL-10 (23). M1 macrophages maintain a tumor-fighting program while M2 macrophages are tumor-supportive and produce factors such as VEGF and a variety of matrix metalloproteinases to enhance tumor progression (43, 44). Importantly, previous studies indicate that HIF-2α accumulation observed in tumor-associated macrophages from patients with breast cancer correlates with high tumor vascularity and tumor grade (45). The fact that HIF-2α was not observed in normal tissue macrophages suggests the important role of GM-CSF in maintaining and “re-educating” M1 macrophage polarity in the presence of AKB-6899.
Our findings that specific manipulation of macrophage HIF-1α or HIF-2α can influence blood vessel formation are applicable to not only solid tumors (where the goal would be to inhibit tumor growth by limiting blood vessel formation) but also in other diseases co-moribund with hypoxia, such as wound healing and ischemic heart disease (where the goal would be to increase tissue healing by increasing blood vessel formation through the stabilization of HIF-1α and down-regulation of HIF-2α). But, we feel the strength of our application resides in the potential to serve as therapy for those patients with malignant melanoma that do not possess the B-RAF mutations and thus have few treatment alternatives.
Acknowledgments
The authors gratefully acknowledge Dr. Robert Shalwitz of Akebia Therapeutics (Cincinnati, OH) for providing AKB-6899 and AKB-4924, as well as for guidance in experimental planning.
Footnotes
This study was supported by NCI K99 CA131552-01 (T.D.E) and KL2 RR025754-04 (J.M.R.) from the National Center for Research Resources. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
References
- 1.Steele RJ, Brown M, Eremin O. Characterisation of macrophages infiltrating human mammary carcinomas. Br J Cancer. 1985;51:135–138. doi: 10.1038/bjc.1985.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lissbrant IF, Stattin P, Wikstrom P, Damber JE, Egevad L, Bergh A. Tumor associated macrophages in human prostate cancer: relation to clinicopathological variables and survival. Int J Oncol. 2000;17:445–451. doi: 10.3892/ijo.17.3.445. [DOI] [PubMed] [Google Scholar]
- 3.Bingle L, Brown NJ, Lewis CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol. 2002;196:254–265. doi: 10.1002/path.1027. [DOI] [PubMed] [Google Scholar]
- 4.Torisu H, Ono M, Kiryu H, Furue M, Ohmoto Y, Nakayama J, Nishioka Y, Sone S, Kuwano M. Macrophage infiltration correlates with tumor stage and angiogenesis in human malignant melanoma: possible involvement of TNFalpha and IL-1alpha. Int J Cancer. 2000;85:182–188. [PubMed] [Google Scholar]
- 5.Leek RD, Hunt NC, Landers RJ, Lewis CE, Royds JA, Harris AL. Macrophage infiltration is associated with VEGF and EGFR expression in breast cancer. J Pathol. 2000;190:430–436. doi: 10.1002/(SICI)1096-9896(200003)190:4<430::AID-PATH538>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 6.Lewis JS, Landers RJ, Underwood JC, Harris AL, Lewis CE. Expression of vascular endothelial growth factor by macrophages is up-regulated in poorly vascularized areas of breast carcinomas. J Pathol. 2000;192:150–158. doi: 10.1002/1096-9896(2000)9999:9999<::AID-PATH687>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 7.Marcoval J, Moreno A, Graells J, Vidal A, Escriba JM, Garcia-Ramirez M, Fabra A. Angiogenesis and malignant melanoma. Angiogenesis is related to the development of vertical (tumorigenic) growth phase. J Cutan Pathol. 1997;24:212–218. doi: 10.1111/j.1600-0560.1997.tb01583.x. [DOI] [PubMed] [Google Scholar]
- 8.Salven P, Heikkila P, Joensuu H. Enhanced expression of vascular endothelial growth factor in metastatic melanoma. Br J Cancer. 1997;76:930–934. doi: 10.1038/bjc.1997.486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ugurel S, Rappl G, Tilgen W, Reinhold U. Increased serum concentration of angiogenic factors in malignant melanoma patients correlates with tumor progression and survival. J Clin Oncol. 2001;19:577–583. doi: 10.1200/JCO.2001.19.2.577. [DOI] [PubMed] [Google Scholar]
- 10.Wedge SR, Ogilvie DJ, Dukes M, Kendrew J, Chester R, Jackson JA, Boffey SJ, Valentine PJ, Curwen JO, Musgrove HL, Graham GA, Hughes GD, Thomas AP, Stokes ES, Curry B, Richmond GH, Wadsworth PF, Bigley AL, Hennequin LF. ZD6474 inhibits vascular endothelial growth factor signaling, angiogenesis, and tumor growth following oral administration. Cancer Res. 2002;62:4645–4655. [PubMed] [Google Scholar]
- 11.Tao J, Tu YT, Huang CZ, Feng AP, Wu Q, Lian YJ, Zhang LX, Zhang XP, Shen GX. Inhibiting the growth of malignant melanoma by blocking the expression of vascular endothelial growth factor using an RNA interference approach. Br J Dermatol. 2005;153:715–724. doi: 10.1111/j.1365-2133.2005.06765.x. [DOI] [PubMed] [Google Scholar]
- 12.Li Y, Wang MN, Li H, King KD, Bassi R, Sun H, Santiago A, Hooper AT, Bohlen P, Hicklin DJ. Active immunization against the vascular endothelial growth factor receptor flk1 inhibits tumor angiogenesis and metastasis. J Exp Med. 2002;195:1575–1584. doi: 10.1084/jem.20020072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Niethammer AG, Xiang R, Becker JC, Wodrich H, Pertl U, Karsten G, Eliceiri BP, Reisfeld RA. A DNA vaccine against VEGF receptor 2 prevents effective angiogenesis and inhibits tumor growth. Nat Med. 2002;8:1369–1375. doi: 10.1038/nm1202-794. [DOI] [PubMed] [Google Scholar]
- 14.Manalo DJ, Rowan A, Lavoie T, Natarajan L, Kelly BD, Ye SQ, Garcia JG, Semenza GL. Transcriptional regulation of vascular endothelial cell responses to hypoxia by HIF-1. Blood. 2005;105:659–669. doi: 10.1182/blood-2004-07-2958. [DOI] [PubMed] [Google Scholar]
- 15.Kendall RL, Thomas KA. Inhibition of vascular endothelial cell growth factor activity by an endogenously encoded soluble receptor. Proc Natl Acad Sci U S A. 1993;90:10705–10709. doi: 10.1073/pnas.90.22.10705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kendall RL, Wang G, Thomas KA. Identification of a natural soluble form of the vascular endothelial growth factor receptor, FLT-1, and its heterodimerization with KDR. Biochem Biophys Res Commun. 1996;226:324–328. doi: 10.1006/bbrc.1996.1355. [DOI] [PubMed] [Google Scholar]
- 17.Eubank TD, Roda JM, Liu H, O’Neil T, Marsh CB. Opposing roles for HIF-1alpha and HIF-2alpha in the regulation of angiogenesis by mononuclear phagocytes. Blood. 2011;117:323–332. doi: 10.1182/blood-2010-01-261792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roda JM, Sumner LA, Evans R, Phillips GS, Marsh CB, Eubank TD. Hypoxia-inducible factor-2alpha regulates GM-CSF-derived soluble vascular endothelial growth factor receptor 1 production from macrophages and inhibits tumor growth and angiogenesis. J Immunol. 2011;187:1970–1976. doi: 10.4049/jimmunol.1100841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Appelhoff RJ, Tian YM, Raval RR, Turley H, Harris AL, Pugh CW, Ratcliffe PJ, Gleadle JM. Differential function of the prolyl hydroxylases PHD1, PHD2, and PHD3 in the regulation of hypoxia-inducible factor. J Biol Chem. 2004;279:38458–38465. doi: 10.1074/jbc.M406026200. [DOI] [PubMed] [Google Scholar]
- 20.Wang Y, Zeigler MM, Lam GK, Hunter MG, Eubank TD, Khramtsov VV, Tridandapani S, Sen CK, Marsh CB. The role of the NADPH oxidase complex, p38 MAPK, and Akt in regulating human monocyte/macrophage survival. Am J Respir Cell Mol Biol. 2007;36:68–77. doi: 10.1165/rcmb.2006-0165OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Eubank TD, Roberts R, Galloway M, Wang Y, Cohn DE, Marsh CB. GM-CSF induces expression of soluble VEGF receptor-1 from human monocytes and inhibits angiogenesis in mice. Immunity. 2004;21:831–842. doi: 10.1016/j.immuni.2004.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tsukamoto K, Hirata S, Osada A, Kitamura R, Shimada S. Detection of circulating melanoma cells by RT-PCR amplification of three different melanocyte-specific mRNAs in a mouse model. Pigment Cell Res. 2000;13:185–189. doi: 10.1034/j.1600-0749.2000.130311.x. [DOI] [PubMed] [Google Scholar]
- 23.Eubank TD, Roberts RD, Khan M, Curry JM, Nuovo GJ, Kuppusamy P, Marsh CB. Granulocyte macrophage colony-stimulating factor inhibits breast cancer growth and metastasis by invoking an anti-angiogenic program in tumor-educated macrophages. Cancer Res. 2009;69:2133–2140. doi: 10.1158/0008-5472.CAN-08-1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nicolson GL, Brunson KW, Fidler IJ. Specificity of arrest, survival, and growth of selected metastatic variant cell lines. Cancer Res. 1978;38:4105–4111. [PubMed] [Google Scholar]
- 25.Acker T, Diez-Juan A, Aragones J, Tjwa M, Brusselmans K, Moons L, Fukumura D, Moreno-Murciano MP, Herbert JM, Burger A, Riedel J, Elvert G, Flamme I, Maxwell PH, Collen D, Dewerchin M, Jain RK, Plate KH, Carmeliet P. Genetic evidence for a tumor suppressor role of HIF-2alpha. Cancer Cell. 2005;8:131–141. doi: 10.1016/j.ccr.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 26.Wu FT, Stefanini MO, Mac Gabhann F, Kontos CD, Annex BH, Popel AS. A systems biology perspective on sVEGFR1: its biological function, pathogenic role and therapeutic use. J Cell Mol Med. 2010;14:528–552. doi: 10.1111/j.1582-4934.2009.00941.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yong K, Cohen H, Khwaja A, Jones HM, Linch DC. Lack of effect of granulocyte-macrophage and granulocyte colony-stimulating factors on cultured human endothelial cells. Blood. 1991;77:1675–1680. [PubMed] [Google Scholar]
- 28.Locke F, Clark JI, Gajewski TF. A phase II study of oxaliplatin, docetaxel, and GM-CSF in patients with previously treated advanced melanoma. Cancer Chemother Pharmacol. 2010;65:509–514. doi: 10.1007/s00280-009-1057-y. [DOI] [PubMed] [Google Scholar]
- 29.Ravaud A, Delaunay M, Chevreau C, Coulon V, Debled M, Bret-Dibat C, Courbon F, Gualde N, Nguyen Bui B. Granulocyte-macrophage colony-stimulating factor alone or with dacarbazine in metastatic melanoma: a randomized phase II trial. Br J Cancer. 2001;85:1467–1471. doi: 10.1054/bjoc.2001.2120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ascierto PA, Leonardi E, Ottaiano A, Napolitano M, Scala S, Castello G. Prognostic value of serum VEGF in melanoma patients: a pilot study. Anticancer Res. 2004;24:4255–4258. [PubMed] [Google Scholar]
- 31.Grignol VP, Olencki T, Relekar K, Taylor C, Kibler A, Kefauver C, Wei L, Walker MJ, Chen HX, Kendra K, Carson WE., 3rd A phase 2 trial of bevacizumab and high-dose interferon alpha 2B in metastatic melanoma. J Immunother. 2011;34:509–515. doi: 10.1097/CJI.0b013e31821dcefd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Testori A, di Pietro A, Munzone E, Mosconi M, Gandini S, Passoni C, Verrecchia F, Minchella I, Nole F, Ferrucci PF. FC14 First-line Dacarbazine (DTIC) plus Bevacizumab (B) combination therapy in advanced melanoma (MM) patients (pts): a phase II study. Melanoma Research. 2010;20:e35. doi: 10.1097/CMR.0000000000000146. [DOI] [PubMed] [Google Scholar]
- 33.Boasberg P, Cruickshank S, Hamid O, O’Day S, Weber R, Spitler L. Nab-paclitaxel and bevacizumab as first-line therapy in patients with unresectable stage III and IV melanoma. J Clin Oncol. 2009;27 doi: 10.1097/COC.0b013e318287bbae. [DOI] [PubMed] [Google Scholar]
- 34.Sowter HM, Raval RR, Moore JW, Ratcliffe PJ, Harris AL. Predominant role of hypoxia-inducible transcription factor (Hif)-1alpha versus Hif-2alpha in regulation of the transcriptional response to hypoxia. Cancer Res. 2003;63:6130–6134. [PubMed] [Google Scholar]
- 35.Yamashita T, Ohneda K, Nagano M, Miyoshi C, Kaneko N, Miwa Y, Yamamoto M, Ohneda O, Fujii-Kuriyama Y. Hypoxia-inducible transcription factor-2alpha in endothelial cells regulates tumor neovascularization through activation of ephrin A1. J Biol Chem. 2008;283:18926–18936. doi: 10.1074/jbc.M709133200. [DOI] [PubMed] [Google Scholar]
- 36.Nangaku M, Izuhara Y, Takizawa S, Yamashita T, Fujii-Kuriyama Y, Ohneda O, Yamamoto M, van Ypersele de Strihou C, Hirayama N, Miyata T. A novel class of prolyl hydroxylase inhibitors induces angiogenesis and exerts organ protection against ischemia. Arterioscler Thromb Vasc Biol. 2007;27:2548–2554. doi: 10.1161/ATVBAHA.107.148551. [DOI] [PubMed] [Google Scholar]
- 37.Loinard C, Ginouves A, Vilar J, Cochain C, Zouggari Y, Recalde A, Duriez M, Levy BI, Pouyssegur J, Berra E, Silvestre JS. Inhibition of prolyl hydroxylase domain proteins promotes therapeutic revascularization. Circulation. 2009;120:50–59. doi: 10.1161/CIRCULATIONAHA.108.813303. [DOI] [PubMed] [Google Scholar]
- 38.Mazzone M, Dettori D, Leite de Oliveira R, Loges S, Schmidt T, Jonckx B, Tian YM, Lanahan AA, Pollard P, Ruiz de Almodovar C, De Smet F, Vinckier S, Aragones J, Debackere K, Luttun A, Wyns S, Jordan B, Pisacane A, Gallez B, Lampugnani MG, Dejana E, Simons M, Ratcliffe P, Maxwell P, Carmeliet P. Heterozygous deficiency of PHD2 restores tumor oxygenation and inhibits metastasis via endothelial normalization. Cell. 2009;136:839–851. doi: 10.1016/j.cell.2009.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Halaban R, Zhang W, Bacchiocchi A, Cheng E, Parisi F, Ariyan S, Krauthammer M, McCusker JP, Kluger Y, Sznol M. PLX4032, a selective BRAF (V600E) kinase inhibitor, activates the ERK pathway and enhances cell migration and proliferation of BRAF melanoma cells. Pigment Cell Melanoma Res. 2010;23:190–200. doi: 10.1111/j.1755-148X.2010.00685.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hatzivassiliou G, Song K, Yen I, Brandhuber BJ, Anderson DJ, Alvarado R, Ludlam MJ, Stokoe D, Gloor SL, Vigers G, Morales T, Aliagas I, Liu B, Sideris S, Hoeflich KP, Jaiswal BS, Seshagiri S, Koeppen H, Belvin M, Friedman LS, Malek S. RAF inhibitors prime wild-type RAF to activate the MAPK pathway and enhance growth. Nature. 2010;464:431–435. doi: 10.1038/nature08833. [DOI] [PubMed] [Google Scholar]
- 41.Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S, Teague J, Woffendin H, Garnett MJ, Bottomley W, Davis N, Dicks E, Ewing R, Floyd Y, Gray K, Hall S, Hawes R, Hughes J, Kosmidou V, Menzies A, Mould C, Parker A, Stevens C, Watt S, Hooper S, Wilson R, Jayatilake H, Gusterson BA, Cooper C, Shipley J, Hargrave D, Pritchard-Jones K, Maitland N, Chenevix-Trench G, Riggins GJ, Bigner DD, Palmieri G, Cossu A, Flanagan A, Nicholson A, Ho JW, Leung SY, Yuen ST, Weber BL, Seigler HF, Darrow TL, Paterson H, Marais R, Marshall CJ, Wooster R, Stratton MR, Futreal PA. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949–954. doi: 10.1038/nature00766. [DOI] [PubMed] [Google Scholar]
- 42.El-Osta H, Falchook G, Tsimberidou A, Hong D, Naing A, Kim K, Wen S, Janku F, Kurzrock R. BRAF Mutations in Advanced Cancers: Clinical Characteristics and Outcomes. PLoS One. 2011;6:e25806. doi: 10.1371/journal.pone.0025806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tjiu JW, Chen JS, Shun CT, Lin SJ, Liao YH, Chu CY, Tsai TF, Chiu HC, Dai YS, Inoue H, Yang PC, Kuo ML, Jee SH. Tumor-associated macrophage-induced invasion and angiogenesis of human basal cell carcinoma cells by cyclooxygenase-2 induction. J Invest Dermatol. 2009;129:1016–1025. doi: 10.1038/jid.2008.310. [DOI] [PubMed] [Google Scholar]
- 44.Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23:549–555. doi: 10.1016/s1471-4906(02)02302-5. [DOI] [PubMed] [Google Scholar]
- 45.Leek RD, Talks KL, Pezzella F, Turley H, Campo L, Brown NS, Bicknell R, Taylor M, Gatter KC, Harris AL. Relation of hypoxia-inducible factor-2 alpha (HIF-2 alpha) expression in tumor-infiltrative macrophages to tumor angiogenesis and the oxidative thymidine phosphorylase pathway in Human breast cancer. Cancer Res. 2002;62:1326–1329. [PubMed] [Google Scholar]