Abstract
Objectives
To evaluate the risk of virologic failure conferred by suboptimal adherence to non-nucleoside reverse transcriptase inhibitors (NNRTI) and minority NNRTI resistance mutations.
Design
Pooled analysis of the risk of virologic failure conferred by minority NNRTI resistance mutations and NNRTI adherence from three studies of treatment-naïve individuals initiating an NNRTI-based regimen.
Methods
Participants from each study were categorized into both adherence quartiles (Q1–Q4) and four strata: ≥95%, 80–94%, 60–79%, and <60%. Weighted Cox proportional hazard models were used to estimate the risk of virologic failure.
Results
The majority of participants (N=768) had high measured adherence, but those in the lowest adherence quartile had the highest proportion of participants with virologic failure and the risk of virologic failure increased step-wise with adherence below 95%. Detection of minority NNRTI drug resistance mutations increased the proportion of participants with virologic failure across adherence quartiles (Cochran-Mantel-Haenszel P<0.001) and adherence strata (Cochran-Mantel-Haenszel P<0.001; <60% adherence, HR 1.7 [1.1–2.7], P=0.02; 60–79% adherence, HR 1.2 [0.5–3.2], P=0.67; 80–94% adherence, HR 2.5 [0.98–6.3], P=0.06; ≥95% adherence, HR 3.6 [2.3–5.6], P<0.001). On multivariate analysis, the effect of minority variants was also most prominent at higher levels of medication adherence.
Conclusions
The presence of minority NNRTI resistance mutations and NNRTI adherence were found to be independent predictors of virologic failure, but also modify each other’s effects on virologic failure. In addition to the focus on medication adherence counseling, ultrasensitive HIV-1 drug resistance assays could play a role in optimizing the success rates of first-line antiretroviral therapy.
Keywords: HIV-1 drug resistance, minority variants, medication adherence, antiretroviral therapy, virologic failure
INTRODUCTION
Non-nucleoside reverse transcriptase inhibitor (NNRTI)-based regimens are recommended as first-line antiretroviral therapy (ART) and are the most commonly prescribed regimen for treatment-naïve patients [1–3]. These regimens are generally well tolerated and effective, but a substantial proportion of participants treated with an NNRTI-based regimen experience virologic failure. In the AIDS Clinical Trials Group (ACTG) study A5095, virologic failure was seen in 11% of participants receiving an efavirenz-based regimen at a median of 32 weeks [4] and in 25% of participants at a median 3-years of follow-up [5]. More recently in the STARTMRK trial, 11% of participants in the efavirenz treated arm were found not to have achieved virologic suppression by 48 weeks [6]. Despite the expanded range of available antiretroviral therapies, treatment failure of first-line regimens can have serious consequences. These include the accumulation of NNRTI or nucleoside reverse transcriptase inhibitor (NRTI) resistance mutations, which can result in cross-resistance to second-generation NNRTI agents (e.g. etravirine and rilpivirine) and diminished effectiveness of the NRTI “backbone” of subsequent regimens. Other potential consequences of delayed effective ART include suboptimal immunologic recovery and increased morbidity and mortality as most individuals in both developed and developing countries still enter HIV care late in the course of disease [7–9].
There are several potential causes of treatment failure for NNRTI-based ART regimens. NNRTI adherence is closely linked with the risk of virologic failure, but it is still unclear what level of NNRTI adherence is needed to maintain optimal rates of virologic suppression [10,11]. The presence of low-frequency, or minority, HIV-1 drug resistance mutations also contributes to the increased risk of virologic failure. In a recent pooled analysis of 985 participants from ten studies, the presence of a minority HIV-1 drug resistance mutation before starting ART was associated with 2–3 times the risk of virologic failure in treatment-naïve participants initiating an NNRTI-based regimen [12]. The risk of virologic failure also increased with suboptimal medication adherence (defined as an overall adherence rate <95%), but the risk was substantially higher in the presence of both drug-resistant minority variants and suboptimal medication adherence. Using data from this pooled analysis, we report one of the largest studies evaluating the effect of NNRTI adherence on the risk of virologic failure for first-line NNRTI-based regimens. We also performed a detailed examination of the relationship between minority NNRTI resistance mutations, medication adherence (including at various adherence levels below 95%), and the risk of virologic failure.
PATIENTS AND METHODS
Study Population
This is a substudy of a previously reported pooled analysis [12]. A systematic review of the literature and conference proceedings was performed through December, 2010 to identify studies evaluating the impact of minority HIV-1 drug resistance mutations in ART-naïve participants initiating an NNRTI-based regimen. A total of ten studies were identified, of which three had available NNRTI adherence data [13–15]. Investigators from all studies agreed to provide patient-level data for this pooled analysis. Participants were excluded with any pre-treatment evidence of reduced NRTI or NNRTI drug susceptibility by standard genotyping based on the Stanford Resistance DB mutation scoring system (score ≥10 for any antiretroviral medication). The definition of virologic failure was standardized for all patients to a plasma HIV-1 RNA level of ≥200 copies/ml at two consecutive time points at least 16 weeks after treatment initiation. Patients were also counted as virologic failures if the last available HIV-1 RNA level was ≥200 copies/ml without a confirmatory measurement. A complete description of the systematic review and pooled analysis methodologies can be found in the initial report [12].
Adherence Measurements
NNRTI adherence data were available from three studies, which evaluated participants from the FIRST study [13], ACTG A5095 [14], and GS-934 [15]. In the FIRST study, NNRTI adherence measurements were based on self-reported adherence over the previous 7 days and recorded at months 1, 4, and every 4 months thereafter [16]. In ACTG A5095, NNRTI adherence was determined based on self-reported adherence over the previous 4 days at weeks 4, 12, 24, and every 24 weeks thereafter [4]. In GS-934, clinic-based pill count was performed at weeks 4, 8, every 8 weeks through week 48, and then every 12 weeks thereafter [17,18].
Minority Variants
To identify NNRTI minority variants, two of the studies used allele-specific PCR [14,15] and one used 454 deep sequencing [13]. All three studies examined the frequencies of the K103N mutation; two examined the frequencies of Y181C mutation [13,14]; and the study using 454 sequencing searched for the presence of additional NNRTI mutations [13]. The study using 454 sequencing identified three participants with additional mutations associated with NNRTI resistance (G190A, K101E, and P225H) and they were included in the analysis as harboring a minority NNRTI-resistant variant. A detailed description of the limits of detection for each assay and the NNRTI minority variants detected can be found in the original report of the pooled analysis [12]. NRTI-resistant minority variants were only evaluated in a small subset of participants and not included in this analysis.
Statistical Analysis
Fisher’s exact tests and Cochran-Mantel-Haenszel statistics (stratified by study) were used to compare the risk of virologic failure by adherence categories. Cox proportional hazard models stratified by study were used to estimate the risk of virologic failure across multiple factors: the presence of baseline minority NNRTI resistance mutations, NNRTI adherence, ethnicity, baseline CD4 cell count, and viral load. To avoid bias induced by targeted sampling, non-randomly sampled cases (virologic failures) in the analysis of A5095 contributed to the Cox proportional hazard models only at their time of failure and were not included in the calculation of virologic failure prevalence. The resulting analysis framework may be considered analogous to a Prentice weighted analysis for a case-cohort study [19,20]. Cox proportional hazard models were evaluated using a time-updated recent adherence, which was defined as the average adherence in the most recent 60 days. Missing values were imputed from the most proximal adherence measurement. Only documented adherence data to the time of virologic failure or censoring was used to categorize participants into adherence quartiles and strata. The presence of an interaction between recent adherence and baseline NNRTI resistance was also evaluated. Statistical analysis was performed using SAS 9.2 (SAS Institute, Cary, NC) and PASW Statistics 18 (IBM SPSS, Chicago, IL). Figures were created using GraphPad Prism 5 (GraphPad Software, La Jolla, CA). Findings with a P-value <0.05 were considered to be statistically significant except for the test of interaction for which a P-value <0.10 was considered to be statistically significant.
RESULTS
Patient and study characteristics
Of the ten studies included in the original pooled analysis [12], medication adherence data were available from three studies comprising 78% (768/985) of the total participants [13–15]. The baseline characteristics of the participants are described in Table 1. Low frequency NNRTI resistance mutations were detected by 454 deep sequencing in one study [13] and by allele-specific PCR in two studies [14,15]. The median follow-up time was 32 months [IQR, 12–34 months].
Table 1.
Baseline characteristics of participants included in the study.
| Characteristic | Simen 2009 |
Paredes 2010 |
Goodman 2011 |
Total |
|---|---|---|---|---|
| Study Design | Cohort | Case cohort |
Cohort | |
| Adherence Method | 7-day self-report |
4-day self-report |
Pill count | |
| Virologic Failure, N (%) | 44 (64) | 148 (53) | 44 (10) | 236 (31) |
| Total Participants, N | 69 | 278 | 421 | 768 |
| Age, median, yr (IQR) | 37 (31–42) |
38 (30–42) |
37 (32–43) |
37 (31–42) |
| Male Sex % (n/N) | 81 (56/69) |
81 (225/278) |
86 (363/421) |
83 (1052/1263) |
| Ethnicity % (n/N) White |
22 (15/69) |
39 (108/277) |
60 (252/420) |
49 (375/766) |
| Black | 55 (38/69) |
40 (110/277) |
22 (94/420) |
32 (242/766) |
| Hispanic | 20 (14/69) |
19 (54/277) |
14 (60/420) |
17 (128/766) |
| Others | 3 (2/69) |
2 (5/277) |
3 (14/420) |
3 (21/766) |
| CD4+ Count, median (IQR) | 252 (38–344) |
227 (127–319) |
202 (68–331) |
227 (127–319) |
| log10 HIV RNA cp/ml, median (IQR) | 5.3 (4.9–5.8) |
5.0 (4.7–5.4) |
4.8 (4.5–5.5) |
5.0 (4.7–5.4) |
Abbreviations: IQR, Inter-quartile range
Risk of virologic failure by NNRTI adherence levels
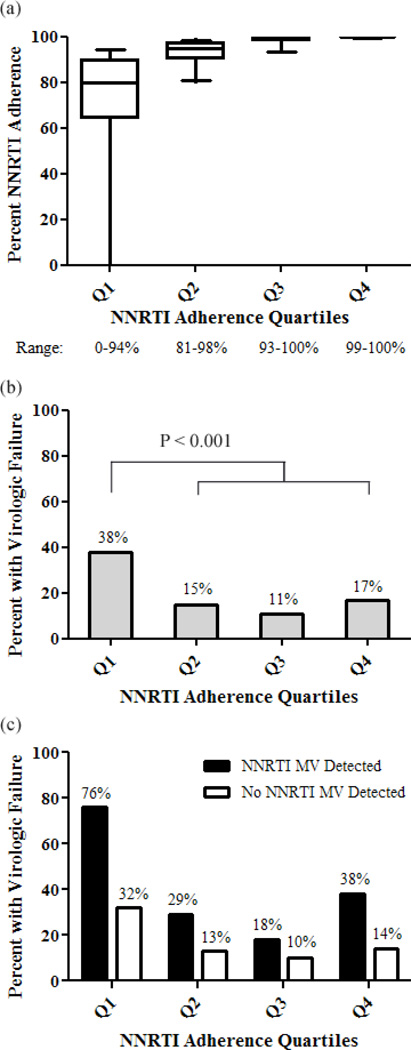
The overall level of NNRTI adherence was high within all three studies with a median adherence rate of 93% [IQR 84–100%] for participants of the FIRST study [13], 98% [IQR 94–99%] for participants of GS-934 [15], and 94% [IQR 81–100%] for participants of ACTG A5095 [14]. To account for potential differences in adherence measurement methodologies between studies (clinic-based pill count versus 4- or 7-day recall), an initial analysis was performed by stratifying participants from each study into NNRTI adherence quartiles. The combined adherence data from the three studies showed excellent adherence within the top three quartiles, all of which had median NNRTI adherence ≥95% (Fig. 1a). Individuals in the lowest adherence quartile were found to have the highest virologic failure rate: 38% (Q1), 15% (Q2), 11% (Q3), and 17% (Q4) (Fisher’s Exact P<0.001 for Q1 virologic failure rate compared to virologic failure rates of either Q2, Q3, or Q4, Fig. 1b).
Fig. 1. NNRTI adherence and virologic failure rates by adherence quartiles.
(a) Box plot of NNRTI adherence by adherence quartiles. Participants from each study were stratified into quartiles by adherence and participants from each quartile then combined. (b) Rates of virologic failure by adherence quartiles. P values represent Fisher’s exact test comparing virologic failure rate for quartile 1 versus each of the other quartiles. (c) Virologic failure rates by adherence quartile stratified by presence of NNRTI minority variants. MV, minority variants.
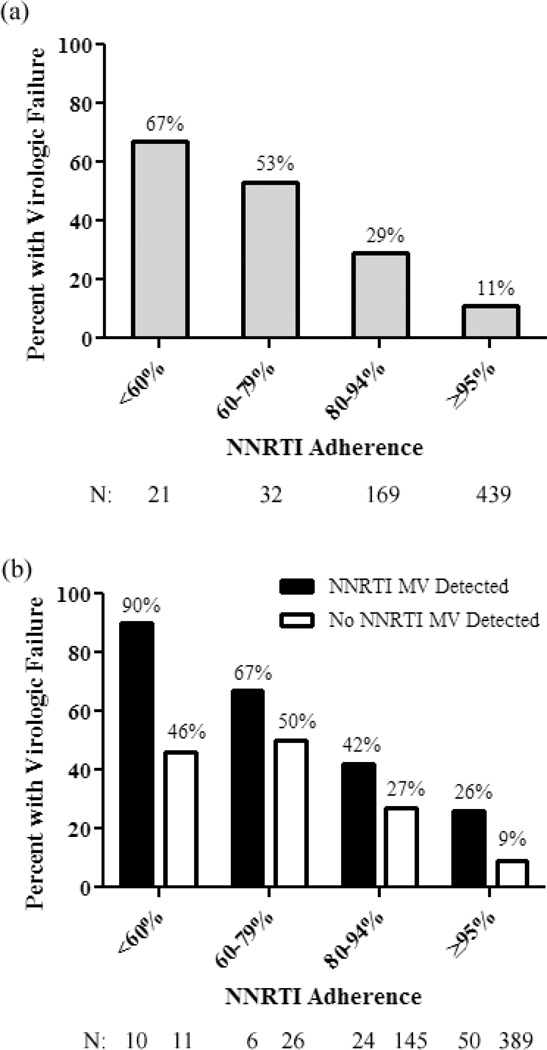
Participants were also categorized by NNRTI adherence strata: <60%, 60–79%, 80–94%, and ≥95%. An increased risk of virologic failure was seen at every NNRTI adherence stratum below 95% (Fisher’s Exact and Cochran-Mantel-Haenszel P<0.001, Fig. 2a). Using the Cox proportional hazard model and ≥95% NNRTI adherence as the reference category, the risk of virologic failure at lower strata of adherence were: 80–94% hazard ratio (HR) 2.0 [95% CI 1.3–3.2], P=0.004; 60–79% HR 3.1 [1.9–5.0], P<0.001, and <60% HR 12.6 [9.4–17.0], P<0.001. This trend was also observed within each of the three studies (data not shown).
Fig. 2. Virologic failure rates by NNRTI adherence strata.
(a) Rates of virologic failure by adherence categories. (b) Virologic failure rates by adherence categories stratified by presence of NNRTI minority variants. MV, minority variants.
Baseline minority variants increase the proportion of participants with virologic failure across adherence categories
Participants with detectable baseline minority NNRTI resistance mutations had a higher rate of virologic failure across adherence quartiles (Cochran-Mantel-Haenszel P<0.001 and Breslow-Day test for homogeneity P=0.45, Fig. 1c). The detection of baseline minority NNRTI resistance mutations also increased the proportion of participants with virologic failure across adherence stratum (Cochran-Mantel-Haenszel P<0.001 and Breslow-Day test for homogeneity P=0.49, Fig. 2b). The increased risk of virologic failure in the presence of minority variants was reflected in the Cox proportional hazard models: <60% adherence, HR 1.7 [1.1–2.7], P=0.02; 60–79% adherence, HR 1.2 [0.5–3.2], P=0.67; 80–94% adherence, HR 2.5 [0.98–6.3], P=0.06; ≥95% adherence, HR 3.6 [2.3–5.6], P<0.001.
Multivariate analysis
In the multivariate Cox proportional hazard model reported in our initial pooled analysis, an overall average adherence value was included in the model [12]. We utilized an additional method of incorporating adherence data in the Cox model that involved a time-updated analysis of the most recent NNRTI adherence within the past 60 days as a reflection of recent adherence. The results from the additional Cox proportional hazard model closely mirror the results of the original multivariate analysis and support the original findings (Table 2). In addition, the effect estimates of NNRTI adherence on the risk of virologic failure were concordant between the Cox proportional hazard models for each of the 3 studies and ranged from a hazard ratio of 1.13 to 1.18 (per 5% lower adherence) using the time-updated most recent 60 day adherence analysis. Limiting the analysis to K103N minority variants only did not significantly alter the results.
Table 2.
Multivariate Cox proportional hazard models using two methods of analyzing NNRTI adherence.
| Overall Average Adherence |
Time-Updated Adherence (last 60 days) |
|||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value | |
| MV Present (NNRTI) | 2.3 | 1.7–3.2 | <0.0001 | 2.2 | 1.6–3.1 | <0.0001 |
| Adherence (per 5% decrease) | 1.15 | 1.13–1.18 | <0.0001 | 1.14 | 1.13–1.17 | <0.0001 |
| Ethnicity Black |
2.8 | 2.1–3.9 | <0.0001 | 2.7 | 1.9–3.6 | <0.0001 |
| Hispanic | 2.0 | 1.4–2.9 | 0.0004 | 1.9 | 1.3–2.8 | 0.001 |
| Other | 2.6 | 1.0–6.5 | 0.046 | 2.2 | 0.9–5.6 | 0.10 |
| CD4 count (per 50 cells/mm3 increase) | 1.0 | 0.98–1.04 | 0.67 | 1.0 | 0.96–1.02 | 0.46 |
| Baseline pVL (log10 copies/mL increase) | 1.0 | 0.88–1.33 | 0.44 | 1.1 | 0.90–1.36 | 0.33 |
Abbreviations: MV, minority variant
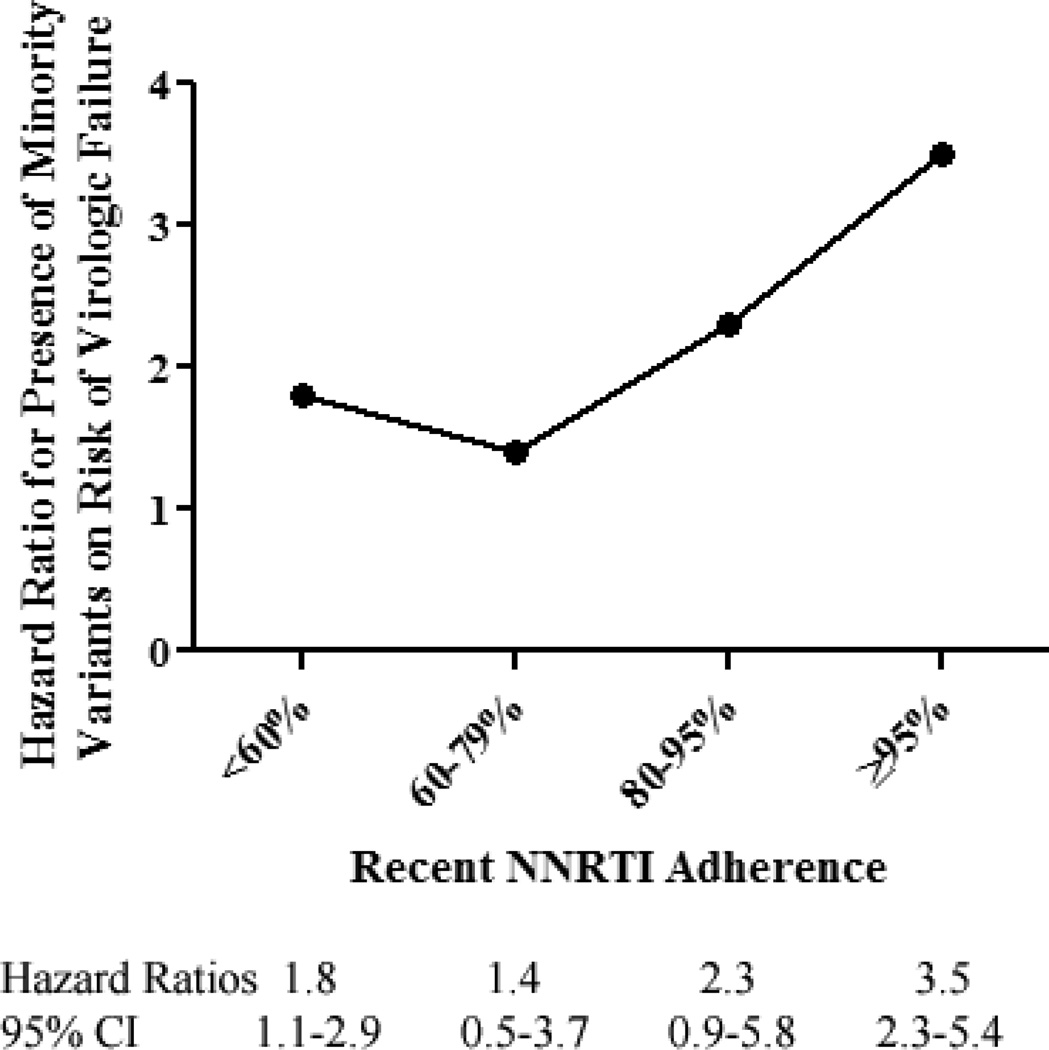
A significant interaction was found in the Cox model between the presence of baseline minority variants and recent medication adherence (P = 0.09). When NNRTI adherence was stratified into previously defined categories, the effect of minority variants on virologic failure was greatest with higher levels of medication adherence (Fig. 3). The association between minority variants and virologic failure was significantly greater in participants with ≥95% recent adherence than in those with <60% recent adherence (P=0.03).
Fig. 3. Relationship between the presence of NNRTI minority variants and risk of virologic failure differs by levels of medication adherence.
Hazard ratios are from the Cox proportional hazard model using NNRTI adherence over the most recent 60 days in a time-updated analysis.
DISCUSSION
In this study, we evaluated the relationship between NNRTI minority variants, medication adherence, and the risk of virologic failure in a subset of participants from three randomized HIV-1 treatment trials. The majority of participants had high measured NNRTI adherence and an increased proportion of participants were found to have virologic failure in the lowest quartile of measured adherence and at every adherence strata below 95%. Minority NNRTI drug resistance mutations increased the risk of virologic failure across adherence categories. We also detected a significant interaction between the presence of minority variants and medication adherence and found that the effect of minority variants on virologic failure appears to be most pronounced with higher levels of adherence.
It is clear that there are ART class-specific relationships between adherence and virologic failure. In contrast to the adherence-failure relationship for protease inhibitors, there is evidence that <95% adherence to NNRTI therapy may be sufficient for virologic suppression [10,11]. In this analysis, we found that most participants had excellent adherence, but that there was an increased risk of virologic failure at every adherence stratum below 95%. One possible explanation for the discrepancy lies in the method used for measuring adherence. While one of the prior studies analyzed self-reported adherence [11], another study used both electronic bottle cap monitoring and unannounced pill counts [10]. The use of self-report and clinic-based pill counts for adherence measurements may over-estimate the levels of medication adherence [21]. However, there is also evidence that patient self-report and electronic medication monitoring may yield similar results [22,23] and that electronic medication monitoring may underestimate actual adherence [21]. Although unannounced pill counts or electronic medication monitoring are likely optimal methods of measuring adherence, the cost associated with these methods also limits the number of participants who can be evaluated.
In the analysis of A5095, the increased risk of virologic failure associated with baseline NNRTI minority variants was only statistically significant for those individuals with high medication adherence [14]. In this larger pooled analysis, which includes participants from A5095, we found that the presence of baseline NNRTI minority variants increased the risk of virologic failure across adherence strata. We also detected an interaction between the presence of minority variants and adherence and showed that the minority variant effect on the risk of virologic failure was most prominent in individuals with higher rates of recent medication adherence. The most likely explanation for this observation is that the effect of drug-resistant minority variants on risk of treatment failure is partly masked in non-adherent patients, who have a substantial risk of virologic failure whether or not drug-resistant mutants are present in the virus population.
This analysis has some limitations. First, we combined three studies using different methods of measuring adherence: clinic-based pill count, 4-day, and 7-day recall. We were conscious of possible method-dependent measurement differences that could introduce heterogeneity to the results. Our primary analysis was therefore based on stratifying participants into adherence quartiles within each study prior to combining the data between studies to minimize heterogeneity. Reassuringly, the effect estimates of NNRTI adherence on the risk of virologic failure in the Cox proportional hazard models were nearly identical between the three studies. Another limitation involves the pooling of data from three studies that used different minority variant assay methodologies with different sensitivities for variant detection. These issues were discussed in detail in the primary pooled analysis and a number of sensitivity analyses were performed to confirm the robustness of the findings [12]. Since only a subset of participants in this analysis were tested for Y181C mutation (Goodman et. al. only tested for the presence of K103N mutation [15]) and only participants in the FIRST study were tested for NNRTI mutations other than K103N and Y181C [13], our results most likely underestimate the effect of minority NNRTI resistance mutations on the risk of virologic failure as a significant proportion of those with no detectable minority variants may have had unmeasured Y181C or other NNRTI resistance mutations. Finally, we used two methods of analyzing adherence in the Cox proportional hazard model, both of which have some limitations. The use of average adherence for each participant is insensitive to variations in adherence over time, but the use of a time-dependent analysis requires imputing adherence for missing values using the most proximal adherence measurement. The effect estimates for baseline minority variants, adherence, and the other variables in the multivariate model are relatively unchanged, however, when results of the two models are compared.
In this study, we report the largest and most in-depth analysis of the relationship between baseline NNRTI minority variants, NNRTI adherence, and the risk of virologic failure. We found that the presence of minority drug resistance mutations and suboptimal NNRTI adherence were independent predictors of virologic failure, but also modify each other’s effects on virologic failure. Not only was optimal NNRTI adherence not sufficient to overcome the effect of NNRTI minority variants, the risk associated with these minority variants was actually greatest in those individuals with the highest levels of adherence. In addition to the continued focus on medication adherence counseling, the use of ultrasensitive HIV-1 drug resistance assays could play a role in optimizing the success rates of first-line antiretroviral therapy.
Acknowledgements
We thank the study participants of all studies for their contributions to this analysis. We thank Karin Metzner (University of Zurich), Melanie Balduin (University of Cologne), Martin Jakobsen (Aarhus University), Anna Maria Geretti (University College London), Rodolphe Thiebaut (Bordeaux School of Public Health), Lars Ostergaard (Aarhus University Hospital), Bernard Masquelier (Bordeaux University Hospital), and Jeffrey Johnson (CDC) for their invaluable contributions to the original systematic review and pooled analysis. We thank Paul Bain, PhD (Countway Library of Medicine, Harvard Medical School) for his assistance with the systematic review and Christian Pou (irsiCaixa AIDS Research Institute) his help in the data collection. We wish to thank Christina Lalama, MS (Harvard School of Public Health) and the AIDS Clinical Trials Group for contributing data from ACTG A5095.
Dr Li has received research support from Bristol-Myers Squibb and has served as a consultant for Tibotec. Dr Paredes has received consulting fees from Pfizer, Merck, Roche Diagnostics; the IrsiCaixa AIDS Research Institute and the Lluita contra la SIDA Foundation have received grant support from Pfizer, ViiV Healthcare, Siemens, Merck, and Boehringer Ingelheim for studies that Dr. Paredes serves as principal investigator. Drs Svarovskaia and Miller are employees and stock-holders of Gilead Sciences, Inc. Yale University receives grant support from Merck, Pfizer, Gilead, Abbott, ViiV and Bristol-Myers Squibb for studies that Dr Kozal serves as the principal investigator. Dr Kozal receives royalties from patents owned by Stanford University for some HIV diagnostic tests. Dr Kuritzkes has served as a consultant to and/or has received research grant support from Abbott, Avexa, Boehringer-Ingelheim, Bristol-Myers Squibb, Gilead, GlaxoSmithKline, Merck, Oncolys, Pfizer, Roche, Tobira, Vertex, ViroStatistics, and ViiV Healthcare.
Financial Support: Dr Li is the recipient of a Clinical Investigator Training Program Fellowship: Harvard/MIT Health Sciences and Technology - Beth Israel Deaconess Medical Center, in collaboration with Pfizer Inc. and Merck & Co. Dr Paredes was supported in part by the CHAIN, Collaborative HIV and Anti-HIV Drug Resistance Network, Integrated Project no. 223131, funded by the European Commission Framework 7 Program. Dr Ribaudo is supported in part by grants from the NIH (Statistical and Data Management Center of the AIDS Clinical Trials Group U01 AI068634 and Harvard University CFAR P30 AI060354). Dr Kozal is supported by a VA Merit Award. Dr. Huppler Hullsiek was supported in part by a grant from the NIH (U01 AI042170). Dr Kuritzkes is supported in part by grants from the NIH (U01 AI 068636, K24 RR016482) and an ACTG Virology Specialty Laboratory subcontract from the ACTG.
Footnotes
Presented in part at the 2011 International Workshop on HIV & Hepatitis Virus Drug Resistance and Curative Strategies, Los Cabos, Mexico, June 7–11, 2011
Financial Disclosures and Conflicts of Interest: No other potential conflicts of interest relevant to this article were reported.
Contributions: J.Z.L., R.P., H.R., and D.R.K. provided scientific input into the study design. J.Z.L. and H.R. performed the statistical analysis. All authors were involved in data collection, data analysis, and the editing of the manuscript.
References
- 1.McKinnell JA, Willig JH, Westfall AO, Nevin C, Allison JJ, Raper JL, et al. Antiretroviral prescribing patterns in treatment-naive patients in the United States. AIDS Patient Care STDS. 2010;24:79–85. doi: 10.1089/apc.2009.0220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Horberg MA, klein DB. An update on the use of Atripla in the treatment of HIV in the United States. HIV/AIDS - Research and Palliative Care. 2010;2:135–140. doi: 10.2147/hiv.s6366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bertagnolio S, Kelley K, Hassani AS, Obeng-Aduasare Y, Jordan M. Surveillance of transmitted and acquired HIV drug resistance using WHO surveys in resource-limited settings. 18th Conference on Retroviruses and Opportunistic Infections; February 27–March 2, 2011; Boston, MA. [Google Scholar]
- 4.Gulick RM, Ribaudo HJ, Shikuma CM, Lustgarten S, Squires KE, Meyer WA, 3rd, et al. Triple-nucleoside regimens versus efavirenz-containing regimens for the initial treatment of HIV-1 infection. N Engl J Med. 2004;350:1850–1861. doi: 10.1056/NEJMoa031772. [DOI] [PubMed] [Google Scholar]
- 5.Gulick RM, Ribaudo HJ, Shikuma CM, Lalama C, Schackman BR, Meyer WA, 3rd, et al. Three- vs four-drug antiretroviral regimens for the initial treatment of HIV-1 infection: a randomized controlled trial. JAMA. 2006;296:769–781. doi: 10.1001/jama.296.7.769. [DOI] [PubMed] [Google Scholar]
- 6.Lennox JL, DeJesus E, Lazzarin A, Pollard RB, Madruga JV, Berger DS, et al. Safety and efficacy of raltegravir-based versus efavirenz-based combination therapy in treatment-naive patients with HIV-1 infection: a multicentre, double-blind randomised controlled trial. Lancet. 2009;374:796–806. doi: 10.1016/S0140-6736(09)60918-1. [DOI] [PubMed] [Google Scholar]
- 7.Althoff KN, Gebo KA, Gange SJ, Klein MB, Brooks JT, Hogg RS, et al. CD4 count at presentation for HIV care in the United States and Canada: Are those over 50 years more likely to have a delayed presentation? AIDS Res Ther. 7:45. doi: 10.1186/1742-6405-7-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tuboi SH, Schechter M, McGowan CC, Cesar C, Krolewiecki A, Cahn P, et al. Mortality during the first year of potent antiretroviral therapy in HIV-1-infected patients in 7 sites throughout Latin America and the Caribbean. J Acquir Immune Defic Syndr. 2009;51:615–623. doi: 10.1097/QAI.0b013e3181a44f0a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lessells RJ, Mutevedzi PC, Cooke GS, Newell ML. Retention in HIV care for individuals not yet eligible for antiretroviral therapy: rural KwaZulu-Natal, South Africa. J Acquir Immune Defic Syndr. 2011;56:e79–e86. doi: 10.1097/QAI.0b013e3182075ae2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bangsberg DR. Less than 95% adherence to nonnucleoside reverse-transcriptase inhibitor therapy can lead to viral suppression. Clin Infect Dis. 2006;43:939–941. doi: 10.1086/507526. [DOI] [PubMed] [Google Scholar]
- 11.Maggiolo F, Ravasio L, Ripamonti D, Gregis G, Quinzan G, Arici C, et al. Similar adherence rates favor different virologic outcomes for patients treated with nonnucleoside analogues or protease inhibitors. Clin Infect Dis. 2005;40:158–163. doi: 10.1086/426595. [DOI] [PubMed] [Google Scholar]
- 12.Li JZ, Paredes R, Ribaudo HJ, Svarovskaia ES, Metzner KJ, Kozal MJ, et al. Low-frequency HIV-1 drug resistance mutations and risk of NNRTI-based antiretroviral treatment failure: a systematic review and pooled analysis. JAMA. 2011;305:1327–1335. doi: 10.1001/jama.2011.375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Simen BB, Simons JF, Hullsiek KH, Novak RM, Macarthur RD, Baxter JD, et al. Low-abundance drug-resistant viral variants in chronically HIV-infected, antiretroviral treatment-naive patients significantly impact treatment outcomes. J Infect Dis. 2009;199:693–701. doi: 10.1086/596736. [DOI] [PubMed] [Google Scholar]
- 14.Paredes R, Lalama CM, Ribaudo HJ, Schackman BR, Shikuma C, Giguel F, et al. Pre-existing minority drug-resistant HIV-1 variants, adherence, and risk of antiretroviral treatment failure. J Infect Dis. 2010;201:662–671. doi: 10.1086/650543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Goodman D, Zhou Y, Margot NA, McColl DJ, Zhong L, Borroto-Esoda K, et al. Low level of the K103N HIV-1 above a threshold is associated with virological failure in treatment-naive individuals undergoing efavirenz-containing therapy. AIDS. 2011;25:325–333. doi: 10.1097/QAD.0b013e3283427dcb. [DOI] [PubMed] [Google Scholar]
- 16.MacArthur RD, Novak RM, Peng G, Chen L, Xiang Y, Hullsiek KH, et al. A comparison of three highly active antiretroviral treatment strategies consisting of non-nucleoside reverse transcriptase inhibitors, protease inhibitors, or both in the presence of nucleoside reverse transcriptase inhibitors as initial therapy (CPCRA 058 FIRST Study): a long-term randomised trial. Lancet. 2006;368:2125–2135. doi: 10.1016/S0140-6736(06)69861-9. [DOI] [PubMed] [Google Scholar]
- 17.Gallant JE, DeJesus E, Arribas JR, Pozniak AL, Gazzard B, Campo RE, et al. Tenofovir DF, emtricitabine, and efavirenz vs. zidovudine, lamivudine, and efavirenz for HIV. N Engl J Med. 2006;354:251–260. doi: 10.1056/NEJMoa051871. [DOI] [PubMed] [Google Scholar]
- 18.Arribas JR, Pozniak AL, Gallant JE, Dejesus E, Gazzard B, Campo RE, et al. Tenofovir disoproxil fumarate, emtricitabine, and efavirenz compared with zidovudine/lamivudine and efavirenz in treatment-naive patients: 144-week analysis. J Acquir Immune Defic Syndr. 2008;47:74–78. doi: 10.1097/QAI.0b013e31815acab8. [DOI] [PubMed] [Google Scholar]
- 19.Barlow WE, Ichikawa L, Rosner D, Izumi S. Analysis of case-cohort designs. J Clin Epidemiol. 1999;52:1165–1172. doi: 10.1016/s0895-4356(99)00102-x. [DOI] [PubMed] [Google Scholar]
- 20.Wacholder S, Boivin JF. External comparisons with the case-cohort design. Am J Epidemiol. 1987;126:1198–1209. doi: 10.1093/oxfordjournals.aje.a114759. [DOI] [PubMed] [Google Scholar]
- 21.Liu H, Golin CE, Miller LG, Hays RD, Beck CK, Sanandaji S, et al. A comparison study of multiple measures of adherence to HIV protease inhibitors. Ann Intern Med. 2001;134:968–977. doi: 10.7326/0003-4819-134-10-200105150-00011. [DOI] [PubMed] [Google Scholar]
- 22.Oyugi JH, Byakika-Tusiime J, Charlebois ED, Kityo C, Mugerwa R, Mugyenyi P, Bangsberg DR. Multiple validated measures of adherence indicate high levels of adherence to generic HIV antiretroviral therapy in a resource-limited setting. J Acquir Immune Defic Syndr. 2004;36:1100–1102. doi: 10.1097/00126334-200408150-00014. [DOI] [PubMed] [Google Scholar]
- 23.Walsh JC, Mandalia S, Gazzard BG. Responses to a 1 month self-report on adherence to antiretroviral therapy are consistent with electronic data and virological treatment outcome. AIDS. 2002;16:269–277. doi: 10.1097/00002030-200201250-00017. [DOI] [PubMed] [Google Scholar]