Abstract
To date, approximately one half of acute myeloid leukaemia (AML) patients do not have a suitable specific molecular marker for monitoring minimal residual disease (MRD). The Wilm’s tumour gene (WT1) has been suggested as a possible molecular marker of MRD in AML. The expression of WT1 in peripheral blood (PB) was measured using quantitative real-time reverse transcription-polymerase chain reaction in peripheral leukocytes from 151 patients with AML at diagnosis. WT1 expression was significantly elevated, i.e. up to 3 orders of magnitude in the majority (80%) of AML patients at diagnosis compared to the PB of healthy donors. Sequence samples of the long-term followed-up AML patients treated with chemotherapy and/or allogeneic bone marrow transplantation were analysed for WT1 expression. The results revealed that the hematological relapses were preceded (median, 1.8 months) by an increase in WT1 gene expression. For the practical utility of this gene as a molecular marker of relapse, it was necessary to determine an upper remission limit, crossing which would signal hematological relapse. The upper remission limit was determined in our set of patients to be 0.02 WT1/ABL. The AML patients who consequently relapsed crossed this upper remission limit; however, those in permanent remission did not. Therefore, this upper remission limit could be taken as the border of molecular relapse of AML patients. Moreover, insufficient decline of WT1 expression under the upper remission limit following induction and/or consolidation therapy was associated with markedly high risk of relapse. The results show that our upper remission limit can be taken as the border of molecular relapse of AML patients and WT1 levels following initial therapy as a beneficial prognostic marker.
Keywords: Wilm’s tumor gene, acute myeloid leukemia, minimal residual disease, prognosis, relapse
Introduction
In the majority of adult leukemic patients it is possible to achieve a complete remission (CR). In the case of acute myeloid leukemia (AML) 60–80% of patients reach CR. However, the majority of these patients subsequently relapse. Currently, there is an effort to predict relapse by monitoring minimal residual disease (MRD) and subsequently to begin the treatment of the patients during their clinical and hematological remission prior to overt hematological relapse. Treatment of MRD has a greater probability of success, is less distressing for the patients and also has a lower cost than the treatment of a probable hematological relapse. Monitoring of the MRD thus contributes to the success of the treatment of leukemic patients. The so-called molecular techniques, based on an analysis of nucleic acids, appear to be most useful for MRD monitoring. Molecular techniques are highly specific and in connection with polymerase chain reaction (PCR) also very sensitive. These techniques enable the detection of a proliferation of the leukemic clone weeks or several months prior to the hematological relapse, when the patient is still in hematological and clinical remission. However, not all leukemic patients have a specific molecular marker for the detection of MRD. Intensive effort is therefore being applied to finding new molecular nonspecific markers for these patients.
The Wilms’ tumor gene (WT1) appears to be highly promising as a molecular MRD marker in leukemic patients, particularly those with AML (1). The WT1 gene was originally identified as a tumor suppressor gene (2). This gene functions also as a transcription factor for a number of genes. It inhibits apoptosis via p53 and Bcl-2 and also inhibits the differentiation of leukemic cells (3). It is expressed in a small number of human tissues (4–6) and in various cancer cells (7) including leukemia cells (8). WT1 is highly expressed in the cells of the majority leukemic patients at diagnosis and it appears to participate in leukemogenesis (9). WT1 expression is markedly low in cells of normal healthy individuals, with the exception of the CD34+ hematopoietic progenitors (10). The expression of the WT1 gene in leukemic cells is up to 10-fold higher than in normal bone marrow or peripheral blood (PB) cells (11). Consequently, this gene is a suitable candidate as a marker for monitoring MRD in AML, either in bone marrow or in PB, particularly in patients without specific molecular markers (1,10–22). The appropriate marker for MRD monitoring should be overexpressed minimally 2 logs higher in diagnostic samples compared to those from normal healthy individuals. Therefore, estimation of the level of the WT1 expression could be beneficial for predicting hematological relapse. As the WT1 gene is often expressed in patients in permanent remission, it was necessary to establish an upper remission limit which, when exceeded, signals a high risk of hematological relapse. In fact, the upper remission limit defines the molecular relapse level. In this study, we describe an estimation of the upper remission limit in PB which could prove beneficial when deciding on an optimal treatment for AML patients. The exceeding of this level predicts an impending relapse. Conversely, when WT1 expression does not fall below this level following induction and/or consolidation therapy, it signals future hematological relapse (20).
Materials and methods
Patient samples
A total of 161 AML patients were enrolled in our study, 101 of which were analysed at the time of diagnosis/presentation. In total, 760 consecutive samples of these AML patients were analysed during the follow-up. Additionally, 11 patients were not evaluated at the time of diagnosis. However, these patients relapsed with concurrent/simultaneous rise of WT1 expression. In 49 of the patients consecutive samples were not available and they were analysed only at diagnosis. The AML patients were subdivided according to the FAB classification and the particular FAB subtypes comprise: AML M0 (n=2), AML M1 (n=36), AML M2 (n=57), AML M3 (n=17), AML M4 (n=25), AML M5 (n=10), AML M6 (n=2) and biphenotypic leukemia (n=1) and 1 patient not defined. The median age of the AML patients was 51 years at presentation and the median of the follow-up was 16 months. The WT1 expression was additionally measured in the PB from 28 healthy donors. A total of 10 million white blood cells from PB were isolated using the red cell lysis method immediately following collection. The cell sediment was lysed and mRNA stabilised by guanidine isothiocyanate. Total RNA was extracted according to a previously described method (23).
cDNA synthesis
Isolated total RNA (0.5–1 μg) was incubated with 50 pmol of random hexamers at 65°C for 10 min; subsequently, 120 units of Mu-MLV reverse transcriptase (Promega, Madison, USA), 10 units of RNAse inhibitor RNAsin (Promega) and a mixture of deoxynucleotide triphosphates (final concentration 2.5 mM each) were added in a total volume of 10 μl. Synthesis of cDNA was performed at 42°C for 60 min followed by heating at 95°C for 2 min.
Real-time PCR
The quantitative real-time PCR (RQ-PCR) of WT1 was performed with the TaqMan probe labeled with fluorescein and Tamra. The expression of WT1 was related to the expression of the housekeeping gene ABL. Amplification and data analysis were carried out in the Rotor-Gene 3000A thermocycler (Corbett Research, Sydney, Australia). The primers and TaqMan probe specific for the WT1 cDNA were designed as described previously (24) and synthesized by Invitrogen (Carlsbad, CA, USA) and Metabion (Munich, Germany). Optimum reaction conditions for WT1 and ABL amplification were: 1.00 units FastStart DNA Polymerase (Roche Applied Science, Mannheim, Germany), 4.0 mM magnesium chloride, 0.5 μM primers: WT1 sense, 5′-ACA ggg TAC gAg AgC gAT AAC CA-3′; WT1 antisense, 5′-CAC ACg TCg CAC ATC CTg AAT-3′; and 0.2 μM of the specific fluorogenic probe, 5′-6-FAM-CAA CgC CCA TCC TCT gCg gAg CCC A XTph where X is Tamra and 200 μM mixture of the particular deoxyribonucleotide triphosphate in a total volume of 20 μl. Cycling was initiated by denaturation at 95°C for 5 min followed by 45 cycles of denaturation at 95°C for 20 sec and annealing/elongation at 60°C for 60 sec as recommended for the TaqMan assays. Final results were expressed as the mathematical formula: Relative WT1 expression = 2CtABL-CtWT1. Simultaneously, the dilution range standards were quantified in order to check the efficiency and sensitivity of our RQ-PCR protocol for quantification of WT1 related to the quantification of the reference gene ABL. These standards were prepared by cloning PCR products of WT1 and ABL into a plasmid and the copy number was subsequently calculated. The two RQ-PCRs, WT1 and ABL, had the same efficiency. The aforementioned RQ-PCR achieved the 10−4–10−5 sensitivity and it was confirmed and correlated with the quantification of WT1 expression using the WT1 ProfileQuant® kit (Ipsogen, Marseille, France), the protocol recommended by the European LeukemiaNet. The two methods were found to be markedly significantly correlated (P<0.001).
Statistical evaluation
Statistical evaluation of our results, i.e. determination of medians, column statistics and calculation of significance, was performed by the Prism GraphPfad software. The nonparametric Mann-Whitney test was used for comparison of expression WT1 in the particular FAB subtypes. The upper remission value of WT1 expression was estimated as 3 standard deviations (SD) above the mean expression in PB cells from patients in permanent hematological remission.
Results
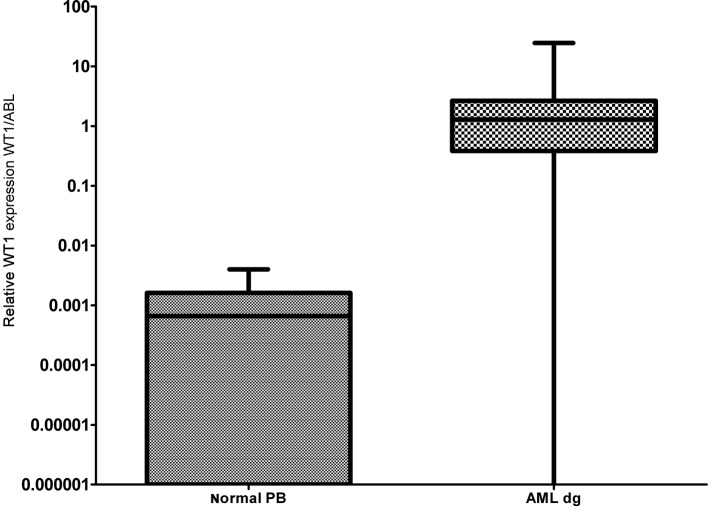
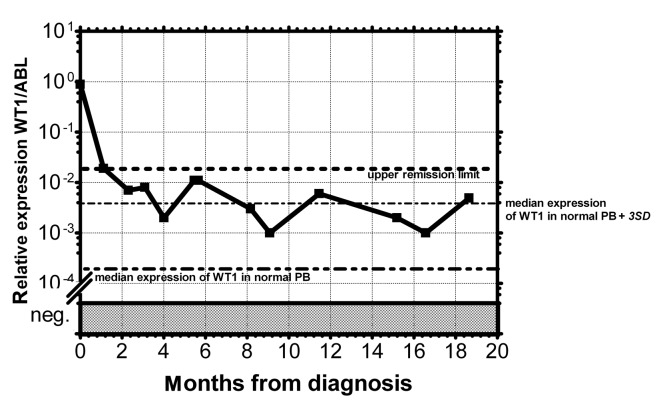
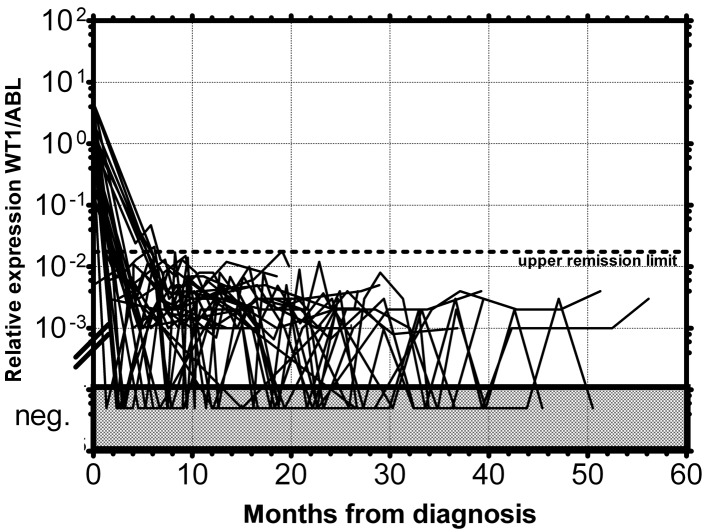
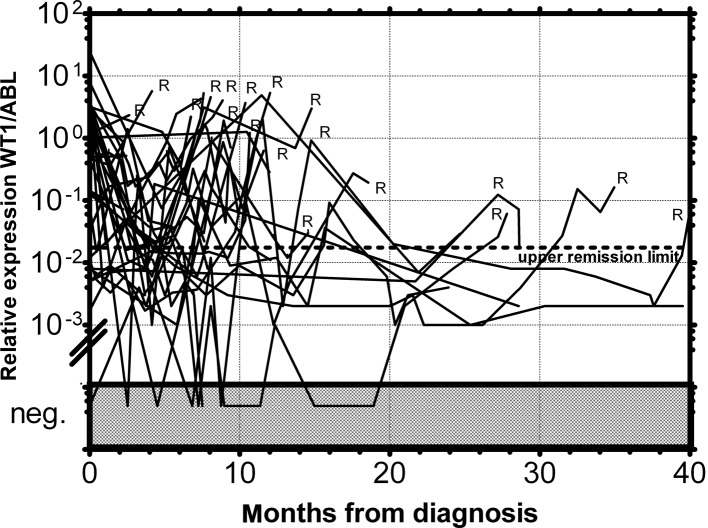
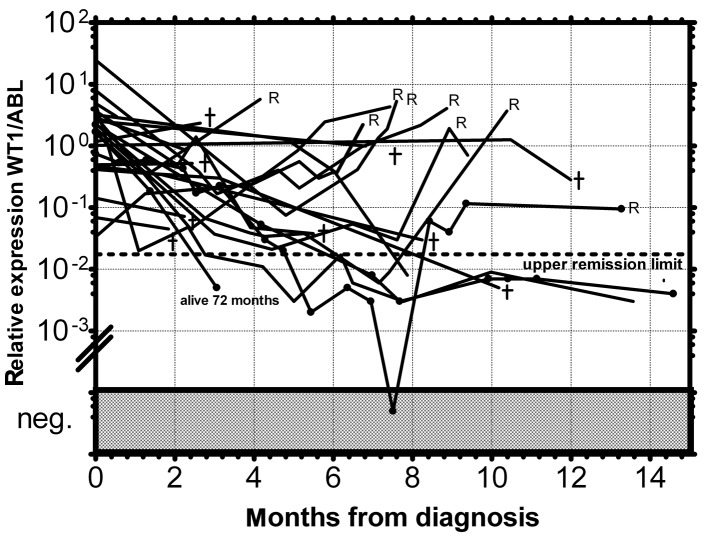
As documented in Fig. 1 more than 90% of AML patients have a two-orders-of-magnitude overexpression of the WT1 gene compared to median expression in normal PB. WT1 expression was almost uniform among the particular FAB subgroups with the exception of the AML M5 patients, whose WT1 expression varied in a broad range; this observation is in accordance with a previous study (25). We have only found a statistically significantly lower expression in AML1/ETO-positive patients at diagnosis compared to the rest of AML patients at diagnosis (P=0.0089) (Fig. 2), in accordance with the results from another study (15). Due to the significant overexpression of WT1 in more than 90% of AML patients, its decrease in patients in remission and its increase prior to a hematological relapse, WT1 expression could be used as a marker of MRD. The main objection to the utility of the WT1 expression as a marker of MRD is its expression in the PB of healthy normal individuals and AML patients in permanent hematological remission. It was therefore necessary to establish a molecular relapse border for WT1 expression. The impending hematological relapse could be signaled by exceeding this WT1 level, defined as the molecular relapse level. At the beginning we took the mean level of WT1 of healthy persons plus 3 SD. However, the WT1 level in patients in permanent remission exceeded this level in certain cases without subsequent hematological relapse. Therefore we took the mean WT1 level of patients in permanent remission plus 3 SD, as documented in 1 patient in permanent remission (Fig. 3). None of the monitored patients in permanent remission (n=73) exceeded this upper remission level (Fig. 4). Conversely, the relapsed patients (n=39) crossed this upper remission level prior to relapse (median prediction, 1.76 months prior to overt relapse; range 0.29–2.57) and 32 of them succumbed to their disease (Fig. 5). Therefore, we would suggest crossing this value as the molecular relapse in AML patients. In our experimental setting, the upper remission level was estimated to be the relative (WT1/ABL) expression WT1 = 0.02. Also in accordance with a previous study (20), the majority of our patients (21/24) whose WT1 level did not fall below our upper remission level following induction and/or consolidation therapy relapsed and the majority of them subsequently succumbed to their disease (Fig. 6). We therefore propose that our upper remission limit can be considered as the molecular relapse level and patients whose WT1 expression exceeds this level should be monitored more carefully and, if possible, the treatment of the impending relapse should be begun in advance.
Figure 1.
Relative expression of the WT1 gene in PB of normal healthy donors and AML patients at diagnosis. The bars represent medians and the boxes represent 25 and 75 percentiles of WT1 expression.
Figure 2.
Relative expression of the WT1 gene in PB of particular FAB sub-types of AML patients at diagnosis. The bars represent medians and the boxes represent 25 and 75 percentiles of WT1 expression.
Figure 3.
Monitoring of MRD in AML patients by relative expression of the WT1 gene in PB. – •• – •• –, Median of WT1 expression in normal PB; – • – • –, median of WT1 expression in normal PB plus 3 SD in normal healthy donors; •••••••••••, upper remission limit of AML patients in permanent remission, relative level = 0.02.
Figure 4.
Monitoring of MRD of AML patients in permanent remission by relative expression of WT1 gene in peripheral blood. The dotted line represents the upper remission limit.
Figure 5.
MRD monitoring of AML patients who relapsed by relative expression of the WT1 gene in PB. The dotted line represents the upper remission limit. R, hematological relapse.
Figure 6.
Monitoring of MRD of AML patients whose WT1 level did not fall below the upper remission limit after induction or consolidation therapy. The dotted line represents the upper remission limit. R, hematological relapse; †exitus.
Discussion
The expression of WT1 gene is considerably increased in the vast majority of AML patients. There were no significant diferences in WT1 expression observed among particular FAB subtypes with one exception: The AML1/ETO-positive patients displayed lower expression of WT1 compared to the rest of the AML patients at diagnosis. This observation is in accordance with a previous study (15). Additionally, the AML M5 patients exhibited a broad range of WT1 expression, as has also been shown in a previous study (25). The quantitation of the WT1 gene expression in PB made it possible to detect MRD in AML patients regardless of the presence or absence of tumor-specific DNA markers. The only disadvantage of the WT1 gene as a marker of MRD in AML patients is its expression in leukocytes of normal healthy persons and AML patients in permanent remission. Therefore, for practical use of measuring the WT1 expression as the MRD marker, it was necessary to determine the level of WT1 expression that, when exceeded, signals a high risk of relapse. This upper normal limit was originally established in a previous study (26). Additionally, the critical remission level was also defined previously (14). At the begining of our study, we attempted to similarly define the molecular relapse level as a mean expresion of WT1 in normal PB plus 3 SD. As certain patients crossed this level without subsequent relapse, the level estimated by this approach was not applicable for monitoring MRD in our patients. The WT1 level in our remission patients was higher in some cases than in normal healthy individuals and therefore we used the mean of WT1 expression plus 3 SD in AML patients who did not subsequently relapse and were in permanent remission. According to our results, crossing this level signalises a high risk of relapse and almost all (95%) of these patients subsequently relapsed. Therefore this level, determined on the basis of patients in permanent remission, could be taken as the border of molecular relapse. As has been shown by previously (20,26), an insufficient decrease of WT1 expression that does not bring its level below the upper normal level after induction and/or consolidation therapy, signalises the future hematological relapse. This was also found in 88% (21/24) of our patients who did not fall below our upper remission limit. As previously described, the determination of WT1 expression following cytoreductive treatment allows for the identification of patients with markedly poor prognosis (14,21), i.e., those whose WT1 expression did not reach normal level, i.e., below the upper remission limit. An alternative approach could be an absolute decrease of the WT1 level following the initial treatment. Consequently, the decrease in the WT1 level by fewer than 2 orders of magnitude also signals the high risk of relapse (15,26,27).
The WT1 level following induction and consolidation is therefore a significant prognostic factor of patient outcome. Patients whose WT1 expression was below the upper remission limit in their follow-up samples remained in complete remission. Our results document the utility of monitoring MRD in AML patients by quantitative estimation of the expression of the WT1 gene.
Acknowledgments
The authors thank Ms. Eva Kohoutová and Ms. Olga Nosková for their skilled technical assistance. The study was supported by grants IGA MZ CR NS10632-3/2009 and IHBT00023736.
Abbreviations:
- WT1
Wilm’s tumor gene;
- AML
acute myeloid leukemia;
- MRD
minimal residual disease
References
- 1.Cilloni D, Gottardi E, Fava M, et al. Usefulness of quantitative assessment of the WT1 gene transcript as a marker for minimal residual disease detection. Blood. 2003;102:773–774. doi: 10.1182/blood-2003-03-0980. [DOI] [PubMed] [Google Scholar]
- 2.Call KM, Glaser T, Ito CY, et al. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome-11 Wilms’ tumor locus. Cell. 1990;60:509–520. doi: 10.1016/0092-8674(90)90601-a. [DOI] [PubMed] [Google Scholar]
- 3.Scharnhorst V, van der Eb AJ, Jochemsen AG. WT1 proteins: functions in growth and differentiation. Gene. 2001;273:141–161. doi: 10.1016/s0378-1119(01)00593-5. [DOI] [PubMed] [Google Scholar]
- 4.Menke AL, Schedl A. WT1 and glomerular function. Semin Cell Dev Biol. 2003;14:233–240. doi: 10.1016/s1084-9521(03)00026-0. [DOI] [PubMed] [Google Scholar]
- 5.Silberstein GB, Van HK, Strickland P, Roberts CT, Jr, Daniel CW. Altered expression of the WT1 Wilms tumor suppressor gene in human breast cancer. Proc Natl Acad Sci USA. 1997;94:8132–8137. doi: 10.1073/pnas.94.15.8132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Makrigiannakis A, Amin K, Coukos G, Tilly JL, Coutifaris C. Regulated expression and potential roles of p53 and Wilms’ tumor suppressor gene (WT1) during follicular development in the human ovary. J Clin Endocrinol Metab. 2000;85:449–459. doi: 10.1210/jcem.85.1.6246. [DOI] [PubMed] [Google Scholar]
- 7.Nakatsuka S, Oji Y, Horiuchi T, et al. Immunohistochemical detection of WT1 protein in a variety of cancer cells. Mod Pathol. 2006;19:804–814. doi: 10.1038/modpathol.3800588. [DOI] [PubMed] [Google Scholar]
- 8.Miwa H, Beran M, Saunders GF. Expression of the Wilms-Tumor Gene (Wt1) in human leukemias. Leukemia. 1992;6:405–409. [PubMed] [Google Scholar]
- 9.Menssen HD, Renkl HJ, Rodeck U, et al. Presence of Wilms’ tumor gene (wt1) transcripts and the WT1 nuclear protein in the majority of human acute leukemias. Leukemia. 1995;9:1060–1067. [PubMed] [Google Scholar]
- 10.Inoue K, Ogawa H, Yamagami T, et al. Long-term follow-up of minimal residual disease in leukemia patients by monitoring WT1 (Wilms tumor gene) expression levels. Blood. 1996;88:2267–2278. [PubMed] [Google Scholar]
- 11.Sugiyama H. Wilms tumor gene (WT1) as a new marker for the detection of minimal residual disease in leukemia. Leuk Lymphoma. 1998;30:55–61. doi: 10.3109/10428199809050929. [DOI] [PubMed] [Google Scholar]
- 12.Trka J, Kalinova M, Hrusak O, et al. Real-time quantitative PCR detection of WT1 gene expression in children with AML: prognostic significance, correlation with disease status and residual disease detection by flow cytometry. Leukemia. 2002;16:1381–1389. doi: 10.1038/sj.leu.2402512. [DOI] [PubMed] [Google Scholar]
- 13.Ogawa H, Tamaki H, Ikegame K, et al. The usefulness of monitoring WT1 gene transcripts for the prediction and management of relapse following allogeneic stem cell transplantation in acute type leukemia. Blood. 2003;101:1698–1704. doi: 10.1182/blood-2002-06-1831. [DOI] [PubMed] [Google Scholar]
- 14.Garg M, Moore H, Tobal K, Yin JAL. Prognostic significance of quantitative analysis of WT1 gene transcripts by competitive reverse transcription polymerase chain reaction in acute leukaemia. Br J Haematol. 2003;123:49–59. doi: 10.1046/j.1365-2141.2003.04552.x. [DOI] [PubMed] [Google Scholar]
- 15.Ostergaard M, Olesen LH, Hasle H, Kjeldsen E, Hokland P. WT1 gene expression: an excellent tool for monitoring minimal residual disease in 70% of acute myeloid leukaemia patients - results from a single-centre study. Br J Haematol. 2004;125:590–600. doi: 10.1111/j.1365-2141.2004.04952.x. [DOI] [PubMed] [Google Scholar]
- 16.Weisser M, Kern W, Rauhut S, Schoch C, Hiddemann W, Haferlach T, Schnittger S. Prognostic impact of RT-PCR-based quantification of WT1 gene expression during MRD monitoring of acute myeloid leukemia. Leukemia. 2005;19:1416–1423. doi: 10.1038/sj.leu.2403809. [DOI] [PubMed] [Google Scholar]
- 17.Cilloni D, Gottardi E, De Micheli D, et al. Quantitative assessment of WT1 expression by real time quantitative PCR may be a useful tool for monitoring minimal residual disease in acute leukemia patients. Leukemia. 2002;16:2115–2121. doi: 10.1038/sj.leu.2402675. [DOI] [PubMed] [Google Scholar]
- 18.Osborne D, Frost L, Tobal K, Yin JAL. Elevated levels of WT1 transcripts in bone marrow harvests are associated with a high relapse risk in patients autografted for acute myeloid leukaemia. Bone Marrow Transplant. 2005;36:67–70. doi: 10.1038/sj.bmt.1704992. [DOI] [PubMed] [Google Scholar]
- 19.Candoni A, Tiribelli M, Toffoletti E, et al. Quantitative assessment of WT1 gene expression after allogeneic stem cell transplantation is a useful tool for monitoring minimal residual disease in acute myeloid leukemia. Eur J Haematol. 2009;82:61–68. doi: 10.1111/j.1600-0609.2008.01158.x. [DOI] [PubMed] [Google Scholar]
- 20.Cilloni D, Messa F, Arruga F, et al. Early prediction of treatment outcome in acute myeloid leukemia by measurement of WT1 transcript levels in peripheral blood samples collected after chemotherapy. Haematologica. 2008;93:921–924. doi: 10.3324/haematol.12165. [DOI] [PubMed] [Google Scholar]
- 21.Ommen HB, Nyvold CG, Braendstrup K, et al. Relapse prediction in acute myeloid leukaemia patients in complete remission using WT1 as a molecular marker: development of a mathematical model to predict time from molecular to clinical relapse and define optimal sampling intervals. Br J Haematol. 2008;141:782–791. doi: 10.1111/j.1365-2141.2008.07132.x. [DOI] [PubMed] [Google Scholar]
- 22.Sakamoto Y, Mariya Y, Sasaki S, et al. WT1 mRNA level in peripheral blood is a sensitive biomarker for monitoring minimal residual disease in acute myeloid leukemia. Tohoku J Exp Med. 2009;219:169–176. doi: 10.1620/tjem.219.169. [DOI] [PubMed] [Google Scholar]
- 23.Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162:156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- 24.Polák J, Markova J, Schwarz J, Maaloufova J, Volkova Z, Cermak J, Haskovec C. [The use of quantitative assessment of Wilms tumour gene 1 for monitoring of residual disease in acute myeloid leukemia patients] Cas Lek Cesk. 2006;145:36–42. [PubMed] [Google Scholar]
- 25.Kreuzer KA, Saborowski A, Lupberger J, Appelt C, Na IK, Le Coutre P, Schmidt CA. Fluorescent 5′-exonuclease assay for the absolute quantification of Wilms’ tumour gene (WT1) mRNA: implications for monitoring human leukaemias. Br J Haematol. 2001;114:313–318. doi: 10.1046/j.1365-2141.2001.02912.x. [DOI] [PubMed] [Google Scholar]
- 26.Cilloni D, Renneville A, Hermitte F, et al. Real-time quantitative polymerase chain reaction detection of minimal residual disease by standardized WT1 assay to enhance risk stratification in acute myeloid leukemia: a European LeukemiaNet study. J Clin Oncol. 2009;27:5195–5201. doi: 10.1200/JCO.2009.22.4865. [DOI] [PubMed] [Google Scholar]
- 27.Gianfaldoni G, Mannelli F, Ponziani V, Longo G, Bencini S, Bosi A, Vannucchi AM. Early reduction of WT1 transcripts during induction chemotherapy predicts for longer disease free and overall survival in acute myeloid leukemia. Haematologica. 2010;95:833–836. doi: 10.3324/haematol.2009.011908. [DOI] [PMC free article] [PubMed] [Google Scholar]