Abstract
The present study was designed to observe the initiation of mitophagy in tumor cells after ultrasound treatment in the presence of curcumin under transmission electron microscopy. Nasopharyngeal carcinoma CNE2 cells were incubated with 10 μM curcumin and then exposed to ultrasound for 8 sec at an intensity of 0.46 W/cm2. Severely swollen mitochondria, disrupted mitochondria and mitophagy were noted in the CNE2 cells after ultrasound treatment in the presence of curcumin. Our findings demonstrated that ultrasound treatment in the presence of curcumin significantly initiated mitophagy in CNE2 cells, which suggests that mitophagy serves as an important event in the process of cell death of nasopharyngeal carcinoma cells.
Keywords: curcumin, ultrasound, mitophagy, autophagic cell death, nasopharyngeal carcinoma
Introduction
Ultrasound therapy is a safe and non-invasive therapeutic approach which shows great promise in the medical field, particularly for tumor therapy. Therapeutic ultrasound mainly includes high- and low-intensity ultrasound. At present, high-intensity focal ultrasound has been applied in the clinical management of tumors since it effectively eradicates tumor tissues through high temperatures and mechanical effects (1,2). Low-intensity ultrasound activates a sensitizer to produce sonodynamic action, which kills tumor cells directly or indirectly (3–5). Sonodynamic therapy has been considered as a feasible strategy for treating malignancies. Our previous studies also confirmed that low-intensity ultrasound activated 5-ALA, hypocrellin B and mematoporphyrin monomethyl ether to induce the cell death of tumor cells (6–9). Curcumin is a naturally ocurring spice, often prescribed in India and China to treat various diseases, such as skin, rheumatic and digestive diseases (10,11). Recently, our studies found that low-intensity ultrasound enhanced the cell death of nasopharyngeal carcinoma cells in the presence of curcumin (12,13). However, the underlying mechanisms are unclear.
Necrosis, apoptosis and autophagy are the main modes of cell death which occur when tumor cells respond to chemotherapy and physical therapy (14–16). Our previous studies showed that ultrasound exposure in the presence of curcumin significantly increased the necrotic or late apoptotic rates of nasopharyngeal carcinoma cells (12). In the present study, we observed mitophagy in nasopharyngeal carcinoma CNE2 cells under transmission electron microscopy (TEM) after ultrasound treatment in the presence of curcumin.
Materials and methods
Sensitizer
A stock solution of curcumin from Sigma (USA) was constructed in dimethyl sulfoxide (DMSO) at a concentration of 100 mM and maintained in the dark at −20°C until it was used.
Cell culture
Nasopharyngeal carcinoma CNE2 cells were purchased from the Shanghai Biology Institute and approval was obtained by the Ethics Committee of Chongqing Medical University prior to the study. The cells were cultured in RPMI-1640 medium supplemented with 10% fetal calf serum (Gibco), 50 μg/ml penicillin, 50 μg/ml streptomycin and 10 μg/ml neomycin. The cells were incubated at 37°C in a humidified CO2 (5%) incubator.
Ultrasound treatment
CNE2 cells (1.0×105/ml) grown overnight in a 24-well culture plate with a flat bottom (Corning, USA) were placed on a platform containing a 1-cm diameter plane transducer, and then exposed to ultrasound 1 h after incubation with curcumin (10 μM). The plane transducer with a central frequency of 1.7 MHz was used to generate continuous ultrasound energy. The spatial average intensity of the ultrasound was 0.46 W/cm2 and exposure time was set at 8 sec. The plane transducer was placed in a water tank filled with degassed water during ultrasonic exposure and the experiment was randomly divided into four groups as described in our previous studies (12,13).
Transmission electron microscopy (TEM)
TEM was used to observe mitophagy and ultrastructural changes in the CNE2 cells 6 h after curcumin and ultrasound treatment. Fixed cells were post-fixed in 2% OsO4, dehydrated in graded alcohol and flat-embedded in Epon 812 (Electron Microscopy Sciences, Fort Washington, PA, USA). Ultra-thin sections (100-nm) were prepared, stained with uranyl acetate and lead citrate, and examined under an electron microscope (H-600; Hitachi, Japan).
Results
Conventional TEM is a standard method for monitoring cellular ultrastructure and autophagy (17). In the TEM observation, the untreated CNE2 cells showed integrity of the cell membrane and mitochondria, with well-developed cristae and a variety of cell nuclei (Fig. 1A). Slight swelling of mitochondria was observed in the CNE2 cells treated by ultrasound alone or curcumin alone. Severely swollen and disrupted mitochondria, and more extensive mitophagy was observed in the CNE2 cells treated with ultrasound radiation in the presence of curcumin (Fig. 1B). This demonstrated that ultrasound treatment in the presence of curcumin markedly damaged mitochondria and initiated mitophagy in the CNE2 cells.
Figure 1.

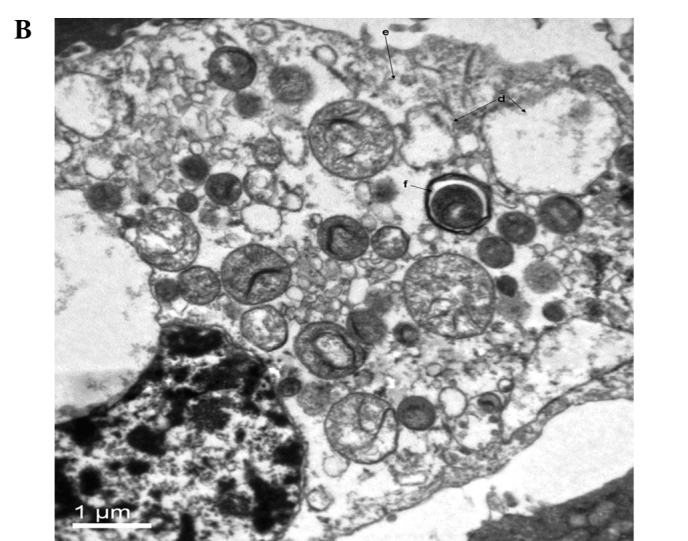
Mitophagy of CNE2 cells observed by TEM 6 h after the treatment of curcumin and ultrasound exposure under ultrasound sonication (1.7 MHz) with an intensity of 0.46 W/cm2 for 8 sec. (A) Sham control. (B) CNE2 cells treated with ultrasound in the presence of curcumin: d, a severely swollen mitochondrion; e, a disrupted mitochondrion; f, mitophagy.
Discussion
Our previous studies demonstrated that ultrasound treatment in the presence of curcumin increased the necrotic or late apoptotic rates of nasopharyngeal carcinoma cells (12). In our previous TEM study, apoptotic features, such as the disappearance of microvillin, membrane blebbing and chromatin condensation, were observed in CNE2 cells treated with both ultrasound and curcumin (13). These findings indicated that ultrasound in the presence of curcumin markedly induced the necrosis and apoptosis of CNE2 cells. Necrosis is a passive form of cell death usually accompanied by rigorous inflammation; apoptosis is an active form of cell death through programmed self-destruction of a single cell characterized by specific nuclear condensation and fragmentation (18,19). Autophagy is a mode of cell death distinct from necrosis and apoptosis. Autophagy is usually considered to be a genetically programmed cell death involving the degradation of cellular proteins and organelles often accompanied by the formation of an autophagosome (20,21). In the process of autophagy, cytoplasmic components such as mitochondria and peroxisomes are encapsulated by an isolation membrane to cause the formation of an autophagosome (20,21). Increasing evidence shows that the autophagic and apoptotic pathways may be regulated by common factors (21,22). Mitochondria are regarded as an important convergent point of apoptotic and autophagic signals, which initiate apoptotic and autophagic cell death (23). In our previous studies, a significant collapse of mitochondrial membrane potential (MMP) and ultrastructural changes in mitochondria were observed after the combined treatment of curcumin and ultrasound (12,16). In the present study, we found severely swollen and disrupted mitochondria, and more extensive mitophagy in cells after the combined treatment of ultrasound and curcumin, which demonstrated that the treatment significantly damaged mitochondria and initiated mitophagy. Mitophagy is a selective form of cell death to degrade damaged mitochondria through autophagy in mammalian cells (24). Our findings suggest that ultrasound treatment in the presence of curcumin initiates autophagy to degrade damaged mitochondria.
It is well known that autophagy plays dual roles in determining the fate of cells, such as death and survival. Certain studies have demonstrated that the induction of autophagy enhances cell death, whereas other reports reveal that autophagy sustains metabolism for promoting cell survival through the degradation of damaged proteins and organelles (25–27). Therefore, the exact mechanisms of mitophagy and autophagy regarding the bioeffects induced by ultrasound treatment in the presence of curcumin need to be determined in future investigations.
In summary, the present study revealed that ultrasound treatment in the presence of curcumin initiated mitophagy in CNE2 cells. Our results highlight that mitophagy may be an important event involving cancer cell death induced by ultrasound treatment in the presence of curcumin.
Acknowledgments
This study was supported by grants from the National Nature Science Foundation of China (30973168) and the Affiliated Hospital of Xi’an Medical University (XYFY10-02). The authors express sincere thanks to Professor Faqi Li, Mr Eric Chuck Hey Pun, Mr Jianyong Wu, Mr Dejiang Chao and Ms. Qinglin Li for the helpful assistance.
References
- 1.Obyn C, Mambourg F. Assessment of high intensity focused ultrasound for the treatment of prostate cancer. Acta Chir Belg. 2009;109:581–586. doi: 10.1080/00015458.2009.11680492. [DOI] [PubMed] [Google Scholar]
- 2.Chaussy C, Thüroff S. High-intensity focused ultrasound in the management of prostate cancer. Expert Rev Med Devices. 2010;7:209–217. doi: 10.1586/erd.09.66. [DOI] [PubMed] [Google Scholar]
- 3.Hiraoka W, Honda H, Feril LB, Jr, Kudo N, Kondo T. Comparison between sonodynamic effect and photodynamic effect with photosensitizers on free radical formation and cell killing. Ultrason Sonochem. 2006;13:535–534. doi: 10.1016/j.ultsonch.2005.10.001. [DOI] [PubMed] [Google Scholar]
- 4.Rosenthal I, Sostaric JZ, Riesz P. Sonodynamic therapy – a review of the synergistic effects of drugs and ultrasound. Ultrason Sonochem. 2004;11:349–363. doi: 10.1016/j.ultsonch.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 5.Kuroki M, Hachimine K, Abe H, Shibaguchi H, Kuroki M, Maekawa S, Yanagisawa J, Kinugasa T, Tanaka T, Yamashita Y. Sonodynamic therapy of cancer using novel sonosensitizers. Anticancer Res. 2007;27:3673–3677. [PubMed] [Google Scholar]
- 6.Wang P, Xu CS, Xu J, Wang X, Leung AW. Hypocrellin B enhances ultrasound-induced cell death of nasopharyngeal carcinoma cells. Ultrasound Med Biol. 2010;36:336–342. doi: 10.1016/j.ultrasmedbio.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 7.Wang P, Xu C, Xia X, Xu J, Wang X, Xiang J, Leung AW. Mitochondrial damage in nasopharyngeal carcinoma cells induced by ultrasound radiation in the presence of Hypocrellin B. J Ultrasound Med. 2010;29:43–50. doi: 10.7863/jum.2010.29.1.43. [DOI] [PubMed] [Google Scholar]
- 8.He Y, Xia X, Xu C, Yu H, Bai D, Xiang J, Leung AW. 5-Aminolaevulinic acid enhances ultrasound-induced mitochondrial damage in K562 cells. Ultrasonics. 2010;50:777–781. doi: 10.1016/j.ultras.2010.03.004. [DOI] [PubMed] [Google Scholar]
- 9.Tian Z, Quan X, Xu C, Dan L, Guo H, Leung W. Hematoporphyrin monomethyl ether enhances sonokilling action of ultrasound on osteosarcoma in vivo. J Ultrasound Med. 2009;28:1695–1702. doi: 10.7863/jum.2009.28.12.1695. [DOI] [PubMed] [Google Scholar]
- 10.Anand P, Thomas SG, Kunnumakkara AB, Sundaram C, Harikumar KB, Sung B, Tharakan ST, Misra K, Priyadarsini IK, Rajasekharan KN, Aggarwal BB. Biological activities of curcumin and its analogues (Congeners) made by man and mother nature. Biochem Pharmacol. 2008;76:1590–1611. doi: 10.1016/j.bcp.2008.08.008. [DOI] [PubMed] [Google Scholar]
- 11.Alarcón de la Lastra C. Curcumin: a promising spice for therapeutics. Mol Nutr Food Res. 2008;52:985. doi: 10.1002/mnfr.200890036. [DOI] [PubMed] [Google Scholar]
- 12.Wang X, Xia X, Xu C, Xu J, Wang P, Xiang J, Bai D, Leung AW. Ultrasound-induced cell death of nasopharyngeal carcinoma cells in the presence of curcumin. Integr Cancer Ther. 2011;10:70–76. doi: 10.1177/1534735410377197. [DOI] [PubMed] [Google Scholar]
- 13.Wang X, Xia X, Leung AW, Xiang J, Jiang Y, Wang P, Xu J, Yu H, Bai D, Xu C. Ultrasound induces cellular destruction of nasopharyngeal carcinoma cells in the presence of curcumin. Ultrasonics. 2011;51:165–170. doi: 10.1016/j.ultras.2010.07.006. [DOI] [PubMed] [Google Scholar]
- 14.Sasnauskiene A, Kadziauskas J, Vezelyte N, Jonusiene V, Kirveliene V. Apoptosis, autophagy and cell cycle arrest following photodamage to mitochondrial interior. Apoptosis. 2009;14:276–286. doi: 10.1007/s10495-008-0292-8. [DOI] [PubMed] [Google Scholar]
- 15.Xu CS, Leung AWN, Liu L, Xia XS. LED-activated pheophorbide a induces cellular destruction of colon cancer cells. Laser Physics Lett. 2010;7:544–548. [Google Scholar]
- 16.Garcia-Escudero V, Gargini R. Autophagy induction as an efficient strategy to eradicate tumors. Autophagy. 2008;4:923–925. doi: 10.4161/auto.6714. [DOI] [PubMed] [Google Scholar]
- 17.Apel A, Herr I, Schwarz H, Rodemann HP, Mayer S. Blocked autophagy sensitizes resistant carcinoma cells to radiation therapy. Cancer Res. 2008;68:1485–1494. doi: 10.1158/0008-5472.CAN-07-0562. [DOI] [PubMed] [Google Scholar]
- 18.Jäckel MC, Dorudian MA, Marx D, Brinck U, Schauer A, Steiner W. Spontaneous apoptosis in laryngeal squamous cell carcinoma is independent of bcl-2 and bax protein expression. Cancer. 1999;85:591–599. [PubMed] [Google Scholar]
- 19.LaMuraglia GM, Schiereck J, Heckenkamp J, Nigri G, Waterman P, Leszczynski D, Kossodo S. Photodynamic therapy induces apoptosis in intimal hyperplastic arteries. Am J Pathol. 2000;157:867–875. doi: 10.1016/S0002-9440(10)64600-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Apel A, Herr I, Schwarz H, Rodemann HP, Mayer S. Blocked autophagy sensitizes resistant carcinoma cells to radiation therapy. Cancer Res. 2008;68:1485–1494. doi: 10.1158/0008-5472.CAN-07-0562. [DOI] [PubMed] [Google Scholar]
- 21.Sy LK, Yan SC, Lok CN, Man RYK, Che CM. Timosaponin A-III induces atuophagy preceding mitochondria apoptosis in HeLa cancer cells. Cancer Res. 2008;68:10229–10237. doi: 10.1158/0008-5472.CAN-08-1983. [DOI] [PubMed] [Google Scholar]
- 22.Li M, Jiang X, Liu D, Na Y, Gao GF, Xi Z. Autophagy protects LNCaP cells under androgen deprivation conditions. Autophagy. 2008;4:54–60. doi: 10.4161/auto.5209. [DOI] [PubMed] [Google Scholar]
- 23.Yan CH, Liang ZQ, Gu ZL, Yang YP, Reid P, Qin ZH. Contributions of autophagic and apoptotic mechanisms to CrTX-induced death of K562 cells. Toxicon. 2006;47:521–530. doi: 10.1016/j.toxicon.2006.01.010. [DOI] [PubMed] [Google Scholar]
- 24.Kim EH, Choi KS. A critical role of superoxide anion in selenite-induced mitophagic cell death. Autophagy. 2008;4:76–78. doi: 10.4161/auto.5119. [DOI] [PubMed] [Google Scholar]
- 25.Ge P, Zhang J, Wang X, Meng F, Li W, Luan Y, Ling F, Luo Y. Inhibition of autophagy induced by proteasome inhibition increases cell death in human SHG-44 glioma cells. Acta Pharmacol Sin. 2009;30:1046–1052. doi: 10.1038/aps.2009.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kessel D, Vicente MGH, Jr, Reiners JJ. Initiation of apoptosis and autophagy by photodynamic therapy. Autophagy. 2006;2:289–290. doi: 10.4161/auto.2792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kondo Y, Kanzawa T, Sawaya R, Kondo S. The role of autophagy in cancer development and response to therapy. Nat Rev Cancer. 2005;9:726–734. doi: 10.1038/nrc1692. [DOI] [PubMed] [Google Scholar]


